Abstract
A 4-year-old spayed female Golden Retriever was presented for evaluation of a rostral maxillary gingival mass. An en bloc resection was performed after histologic diagnosis of ameloblastic fibroma from an incisional biopsy specimen. Histologically, the tumor was composed of (1) poorly differentiated vimentin-positive mesenchymal cells that surrounded islands and (2) thin anastomosing trabeculae of odontogenic epithelium that variably coexpressed pancytokeratin and vimentin. To the authors' knowledge, this is the first report of ameloblastic fibroma in a dog. The clinical, radiographic, and histologic findings in this case are compared to those in other domestic animals and humans.
Ameloblastic fibroma is a rare neoplasm of mixed odontogenic ectomesenchymal and odontogenic epithelial origin. 4,7 It is the most common odontogenic tumor in cattle. 7 However, it is extremely rare in companion animals, and its diagnosis in the cat has been controversial. 7,9 Although there have been several reports of the related ameloblastic fibro-odontoma, 12 ameloblastic fibroma has not, to our knowledge, been reported in the dog. The purpose of this report is to describe the clinical, radiographic, and histologic features of a maxillary ameloblastic fibroma in a dog.
Case History
A 4-year-old spayed Golden Retriever (35 kg) was presented to the Veterinary Medical Teaching Hospital for evaluation of a gingival mass, detected on routine physical examination by the primary care veterinarian, adjacent to the right maxillary first incisor tooth (No. 101). The mass apparently caused no discomfort, and except for unrelated facial pruritus, the dog was otherwise healthy.
On oral examination, the mass (9 × 9 × 4 mm) was on the labial aspect of tooth No. 101, causing palatal deviation but no loosening of the tooth (Fig. 1). The overlying gingival mucosa was ulcerated with formation of a draining tract. The tongue, buccal mucosa, soft and hard palates, tonsils, pharynx, regional lymph nodes, and extraoral facial structures were normal. Occlusal digital intraoral radiography of the right maxillary incisors did not detect alveolar bone involvement or bone density within the mass (Fig. 2).
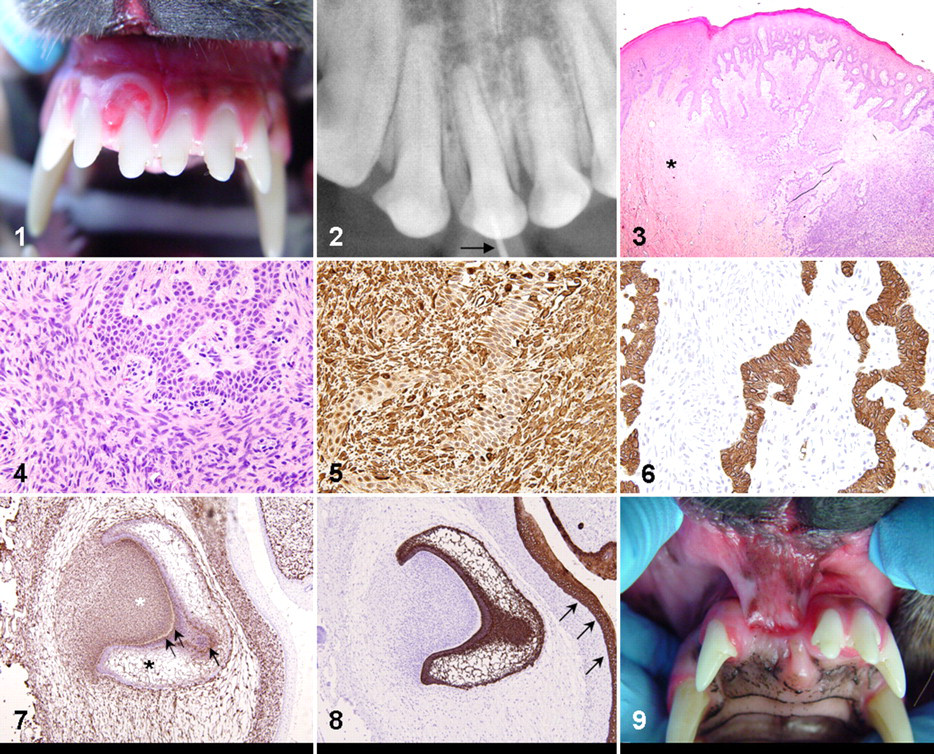
Mouth, dog. Superficial aspect of the ameloblastic fibroma just before en bloc resection. The tumor encircles the base of the maxillary right first incisor tooth; granulation tissue is apparent at its ulcerated surface.
Histologic Findings
An incisional biopsy specimen was submitted to the pathology service. Histologically, a moderately demarcated infiltrative gingival mass effaced and thickened the lamina propria and elevated the overlying gingival mucosa. The neoplastic tissue was moderately cellular and composed of a biphasic population, with poorly differentiated mesenchymal cells surrounding islands and thin anastomosing trabeculae of epithelial cells (Figs. 3, 4). The predominant tissue resembled the primitive mesenchyme of the dental papilla, with plump spindle-to-stellate cells densely and haphazardly arranged in scant immature collagenous stroma. Interconnecting or isolated cords of odontogenic epithelium were generally thin and consisted of a bilayer of short columnar cells with palisading oval nuclei, like the dental lamina of early tooth development. Odontogenic epithelium lacked apical nuclear polarity and stellate reticulum, both features of the enamel organ slightly later in tooth development. Nuclei of epithelial and mesenchymal cell populations lacked features of atypia, and mitotic figures were rare. The odontogenic epithelium was focally continuous with the deep rete pegs of the intact mucosal epithelium. No enamel matrix or dentin was detected in the mass. Inflammation and necrosis was minimal, and the overlying mucosal epithelium, where intact, was hyperplastic. The mesenchymal component blended into surrounding lamina propria, and neoplastic cells extended to the deep surgical margins of the original biopsy specimen. The diagnosis was ameloblastic fibroma.
An en bloc resection of the tumor was performed, with teeth Nos. 101 and 102 (maxillary right second incisor) and the associated alveolar bone. In a histologic section of the resected maxilla, the gingival mucosa rostral to tooth No. 101 was ulcerated, and the lamina propria was replaced by highly cellular granulation tissue with moderate neutrophilic infiltrates. Inconspicuously intermixed with reactive fibroblasts were few cords of palisading epithelium and a small amount of neoplastic primitive mesenchyme. The neoplastic tissue extended to but did not invade alveolar bone, which had mild lysis and periosteal proliferation. There was slight resorption of tooth No. 101 near the cementoenamel junction. An unremarkable periodontal ligament and several quiescent epithelial rests surrounded the tooth root. Surgical margins were free of neoplastic tissue.
Immunohistochemistry was performed on the initial incisional biopsy specimen. Antibody to vimentin (1:200 dilution, clone V9, Dako, Carpinteria, CA) strongly labeled the cytoplasm of the mesenchymal cells of the mass, with variable weak-to-moderate cytoplasmic labeling of the epithelial component (Fig. 5). With pancytokeratin antibody (1:200 dilution, clone AE1/AE3, Dako), there was strong cytoplasmic labeling of neoplastic epithelial cells, with no labeling of mesenchymal cells (Fig. 6). Immunohistochemistry for vimentin and pancytokeratins was applied to fetal canine tissue to illustrate the immunophenotype of the developing tooth. The dental papilla was vimentin positive; the entire enamel organ was cytokeratin positive; and both the inner enamel epithelium and the stellate reticulum coexpressed both markers (Figs. 7, 8).
Case Outcome and Discussion
En bloc resection was expected to be curative, so the dog received no ancillary therapy. Minor dehiscence was detected 2 weeks postoperatively, but the surgical site had granulation tissue and was healing by second intention. Healing was considered adequate 6 months after resection (Fig. 9) with no clinical or radiographic evidence of tumor recurrence.
Odontogenic tumors are histologically classified according to the presence of odontogenic epithelium, odontogenic mesenchyme, or both. In animals, tumors with both components include ameloblastic fibroma, feline inductive odontogenic tumor (a tumor unique to cats), ameloblastic fibro-odontoma, and odontoma. 8 Ameloblastic fibro-odontoma and odontoma both have some degree of tooth formation. In contrast, ameloblastic fibroma does not form dentigerous structures. 4,7 Although rare in humans and domestic animals, it is the most common odontogenic tumor in cattle. 7 Ameloblastic fibroma has been diagnosed in cats, 10,15,18,26 although most cases were reclassified as feline inductive odontogenic tumors in subsequent review articles. 7,9 Although canine ameloblastic fibro-odontomas have been reported, 1,12,23 ameloblastic fibroma has not been reported in dogs.
In humans and cattle, ameloblastic fibromas are benign, slow-growing, expansile, noninvasive neoplasms 8,13 that do not tend to infiltrate bone. 4 The typical clinical presentation in humans is jaw swelling, mainly of the caudal mandible, or intraoral swelling. 4,14,17,19,25 In about 75% of cases, the tumor is associated with an impacted tooth, 4,16,19 and it may be accompanied by ulceration, pain, tenderness, and drainage. 3,4,17 The tumor is mostly diagnosed in persons younger than 20 years of age; about 20% of the lesions are discovered with routine radiography. 4,16,25
Bovine ameloblastic fibroma usually affects the rostral mandible of young cattle (< 1.5 years). 7 Grossly, most tumors are firm, lobulated, roughly spherical masses with intratumoral cysts. 2,20 –22,24 The clinical presentation of the tumor in our case differed from reported cases in other species in that it was raised and ulcerated, rather than spherical, lobulated, or cystic, and it developed in the maxillary gingiva, in contrast to the typical mandibular location in humans and cattle.
Radiographically, human ameloblastic fibroma is a well-defined unilocular radiolucency (smaller tumors) or multilocular radiolucency (when larger), usually with a radiopaque boundary. 4,14,16,17,19 These tumors may resemble dentigerous cysts when associated with an unerupted tooth. 16 This is in stark contrast to the radiographic appearance of the tumor in our case, in which no cystic component or osseous involvement was evident.
Ameloblastic fibromas may be central or peripheral. The term peripheral implies the presence of an odontogenic tumor in the gingival soft tissue, whereas deep involvement of the tooth root and alveolar bone is a feature of central tumors. As in the present case, the mesenchymal component is generally predominant over the poorly differentiated epithelial component. 6 Owing to the lack of specific molecular markers, the diagnosis is based on histologic features and dentigerous relationships. Although not specific, immunohistochemical expression of pancytokeratin and vimentin are consistent with reports of human ameloblastic fibroma 5 and canine ameloblastic fibro-odontoma, 23 generally reflecting the degree of cellular differentiation. The primitive mesenchyme has strong expression of vimentin with little expression of cytokeratin. Both neoplastic and native mucosal epithelia express pancytokeratins, whereas the least-differentiated odontogenic epithelium coexpresses pancytokeratins and vimentin. Although only pancytokeratin was tested here, expression of specific cytokeratins by odontogenic epithelium may vary according to location within a tumor and degree of differentiation. 5 Cytokeratin 14 is most consistently expressed in primitive odontogenic epithelium in both tumors 5 and the developing tooth. 11 Cytokeratins and vimentin coexpress in parts of the enamel organ during tooth development. 11 We substantiated this in the present tumor and in the normal-developing canine tooth.
In human medicine, the surgical approach for ameloblastic fibromas is controversial. Most tumors are treated conservatively with enucleation and curettage. 4,14,16,19,25 Resection (marginal, segmental) or wide excision is considered aggressive 3,14 and mostly reserved for extensive central tumors or those that recur. 4,17,25 Because no controlled studies of the treatment of ameloblastic fibromas in domestic animals are available, treatment protocols are largely based on those for human ameloblastic fibromas, which support en bloc resection. Reappearance is unlikely after en bloc resection because this tumor is thought to regrow from residual neoplastic tissue rather than true recurrence. 4,13,16 However, periodic monitoring of the surgical site for recurrence should be performed for the remainder of the animal’s life.
The diagnosis of ameloblastic fibroma in this dog was based on the presence of a benign proliferation of primitive odontogenic mesenchyme and odontogenic epithelium without the formation of tooth-like structures. As periodic dental prophylaxis becomes increasingly routine for companion animals, more oral tumors will be detected, and their histologic classification will provide new information on the prevalence of tumors of odontogenic origin in animals.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
