Abstract
Cutaneous round cell neoplasms (RCNs) are among the most common skin tumors of dogs. Although a diagnosis can be made in most cases following the examination of H&E-stained routine samples, less-differentiated neoplasms can exhibit overlapping morphologic features that may be challenging to the pathologist, and require immunohistochemistry or molecular testing to reach a final diagnosis. Many patients with cutaneous RCNs are initially seen by a general practitioner and are not referred to a veterinary teaching hospital until a diagnosis has been made. For this reason, anatomic pathology residents at some academic or other training institutions may not be exposed frequently to RCNs during the surgical biopsy service as part of their residency training. In an attempt to fill that gap, here we review the key routine histologic features of canine cutaneous RCNs.
Cutaneous round cell neoplasms (RCNs), also referred to as cutaneous round cell tumors, are among the most common neoplasms of dogs in the veterinary surgical biopsy routine.4,8,9,17 RCNs encompass mainly mast cell tumor (MCT), plasma cell tumor (PCT), lymphoma, and histiocytic neoplasms, especially histiocytoma and, less often, histiocytic sarcoma.4,8,9,17 Although transmissible venereal tumor (TVT) is endemic to, and more commonly diagnosed, in some parts of the world, sporadic cases are submitted to diagnostic laboratories in the United States, and should also be considered in the differential diagnosis of a cutaneous RCN. 20 Amelanotic melanoma and neuroendocrine carcinoma can exhibit a spectrum of histologic features that could occasionally resemble a RCN. 20
Although a final diagnosis can be achieved, or at least suspected, in most cases after systematic examination of H&E-stained tissue sections, less-differentiated RCNs can exhibit overlapping morphologic features that may be challenging to the pathologist and preclude a routine diagnosis.4,17,20 Given the differences in prognosis among these neoplasms, cases in which a diagnosis could not be achieved typically require immunohistochemistry (IHC) or other ancillary testing for diagnostic confirmation.4,9,20 For these reasons, a good understanding of the morphologic features of each type of cutaneous RCN is fundamental for a routine diagnosis or for a critical consideration of the most likely possibilities and the specific IHCs to be requested in a particular case.4,20 Most diagnostic laboratories are equipped with a variety of antibodies and IHC panels that can be utilized for diagnostic confirmation, helping clinicians and oncologists to implement the appropriate clinical and therapeutic protocols based on tumor type and grade.4,17,20
As has been the case with other groups of cutaneous neoplasms of dogs, 23 many patients with cutaneous RCNs are initially seen by a general practitioner and are not necessarily referred to a veterinary teaching hospital until a final diagnosis has been made. For this reason, anatomic pathology residents at some academic or other training institutions may not be exposed to a sufficient number of RCNs during the surgical biopsy service as part of their residency training, which leads to deficiencies in recognizing the histologic hallmarks of these neoplasms in routine diagnostic samples (Rissi DR, pers. comm., 2022 Feb 10). To seek to fill that gap, here we review the key morphologic and diagnostic features of canine cutaneous RCNs. We take a “big-to-small” approach when examining each tumor type, evaluating their subgross features, followed by the overall cell arrangement and distribution within affected tissues, and finally by the characteristic cellular features.
When grading systems are available, we briefly mention the most important features that should be assessed to classify specific neoplasms. However, the scope of this article is not to review the grading systems applied to each group of neoplasms, and we encourage readers to consult the original publications for that purpose. We will discuss the classic RCNs of dogs (MCT, PCT, lymphoma, histiocytoma, and histiocytic sarcoma) and also a subset of neoplasms that can occasionally share morphologic features with RCNs (amelanotic melanoma and neuroendocrine carcinoma). We also discuss briefly the most useful diagnostic immunomarkers and their limitations for each tumor category.
The big picture
As with most observational decisions in diagnostic pathology, pattern recognition is key, and the degree of difficulty in diagnosing a cutaneous RCN may be related to the intrinsic morphologic variations that exist among individual neoplasms and on the experience of the trainee or pathologist examining the biopsy sample. Systematic training in descriptive histopathology, with continuous exposure to RCNs, will eventually amend the latter. 2 However, RCNs often have overlapping morphologic characteristics that hinder a routine diagnosis, thus requiring the use of confirmatory ancillary testing (typically IHC). For these reasons, determining the general patterns of a RCN should always precede detailed histologic evaluation of the tissue section.
Preliminary assessment of the overall subgross pattern of a possible RCN on a glass slide or digital image is highly indicated as the first step in evaluating the surgical biopsy specimen. 2 Unless there is significant ulceration and intratumoral hemorrhage or necrosis, the first feature that is typically noticed in a RCN is its deeply basophilic staining on an H&E-stained section, which is the result of the nuclear staining throughout the extensive sheets of monomorphic neoplastic round cells expanding the dermis and/or subcutaneous tissue. Although not diagnostic (other neoplasms such as basal cell tumors usually have the same characteristic), basophilic staining is an important finding that can be used as a heuristic tool in association with the subsequent and more detailed morphologic evaluation of a RCN (Fig. 1). The hallmark features discussed in our review will not always be present in every RCN, but we hope that the information in this article provides the general guidelines necessary to direct pathology trainees and pathologists to a more systematic evaluation of RCNs during their diagnostic routine.
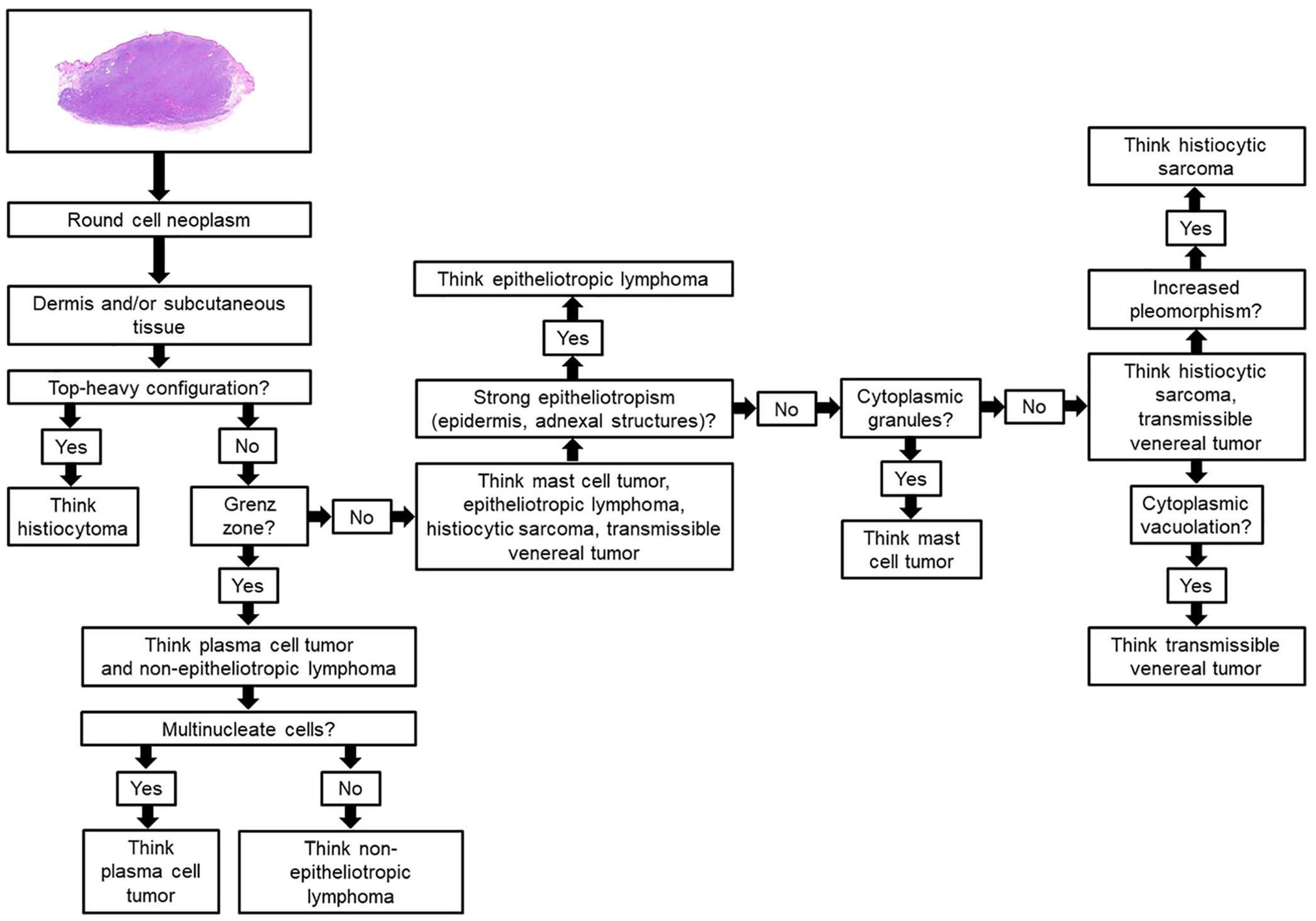
Flowchart highlighting the main decision-making thought processes to be assessed when evaluating a cutaneous round cell neoplasm.
Mast cell tumor
Subgross features
The anatomic localization of MCTs (cutaneous vs. subcutaneous) bears important implications for the diagnosis and prognosis of these neoplasms.10,16,22 Cutaneous MCTs occur as either well-demarcated or poorly demarcated neoplasms that affect the dermis primarily, with occasional involvement of the subcutaneous tissue and deeper tissues such as skeletal muscle.8–10,16 Two grading systems have been validated for cutaneous MCTs.10,16 According to the widely utilized 3-tier MCT grading system developed in the 1980s, 16 grade 1 MCTs are restricted to the superficial dermis and interadnexal spaces (Fig. 2), grade 2 and 3 MCTs infiltrate the lower dermis and subcutaneous tissue (Fig. 3), or the dermis, subcutaneous tissue, and skeletal muscle (Fig. 4). A more recent 2-tier grading system for cutaneous MCTs is based on cellular features and mitotic count, but does not consider the location of neoplastic mast cells (see below). 10
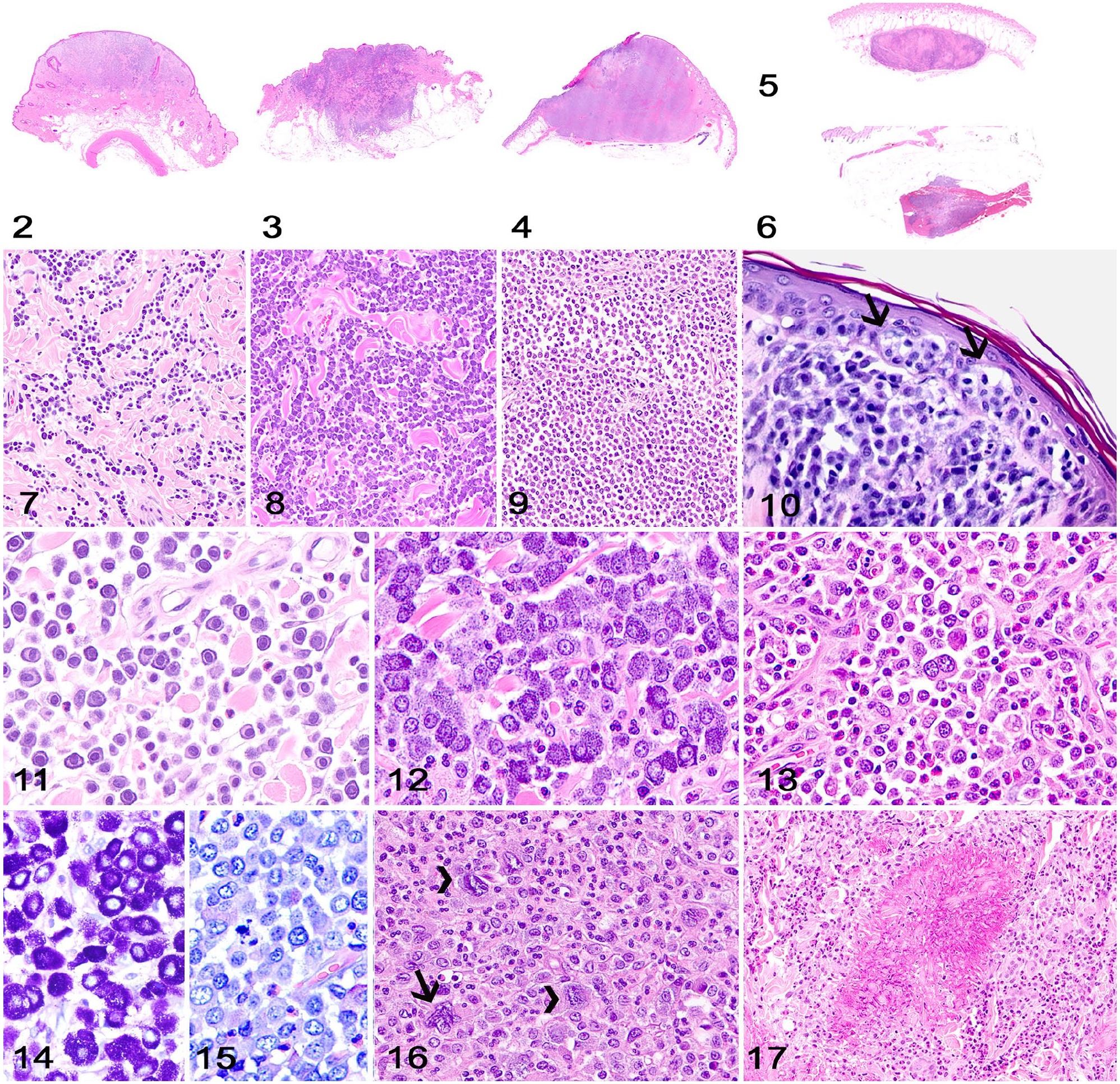
Cutaneous and subcutaneous mast cell tumor (MCT).
Subcutaneous MCTs are restricted to and surrounded by subcutaneous adipose tissue, but a few neoplastic cells can occasionally infiltrate the overlying dermis. These tumors can be well-demarcated (Fig. 5) or infiltrative (Fig. 6). The 2 existing grading systems that are routinely applied for cutaneous MCTs have not been evaluated for, or applied to, subcutaneous MCTs. 22 Subcutaneous MCTs have a more favorable prognosis than cutaneous MCTs, with prolonged survival time and low rates of recurrence and metastasis. 22
Overall cell arrangement
For cutaneous MCTs, neoplastic cell density tends to increase with tumor grade (Figs. 7–9). Neoplastic mast cells are arranged in small groups, sheets, or cords that dissect dermal collagen bundles or deeper subcutaneous tissue and skeletal muscle. Neoplastic mast cells usually spare the dermal-epidermal junction, but epitheliotropism, characterized by clusters or individual neoplastic mast cells infiltrating the epidermis or follicular epithelium, can be observed (Fig. 10). 15 Neoplastic mast cells in subcutaneous MCTs are arranged in sheets supported by a scant fibrovascular stroma or preexisting tissues and are surrounded by subcutaneous adipose tissue.
Cellular features
Grade 1 cutaneous MCTs consist of round neoplastic mast cells with abundant, faintly eosinophilic cytoplasm with medium-sized granules and distinct cell borders. Nuclei are round and have dense chromatin (Fig. 11). Mitotic activity is absent. Neoplastic cells in grade 2 MCTs are similar to those in grade 1 MCTs, but scattered cells can have indistinct cytoplasm with large granules. Nuclei are round-to-indented and have finely stippled chromatin with 1 or 2 nucleoli (Fig. 12). Binucleate neoplastic cells and rare mitoses can be present. Grade 3 MCTs are composed of round-to-oval neoplastic mast cells with moderate-to-high pleomorphism and distinct cytoplasm with fine-to-indistinct granules. Nuclei are typically round-to-indented with clear chromatin and 1–3 nucleoli. Binucleate cells and mitoses are common (Fig. 13).8,9,16
Cytoplasmic granules are evident in lower-grade MCTs (Fig. 14) but frequently need to be highlighted with special stains (Giemsa, toluidine blue) in higher-grade tumors (Fig. 15).8,9,16 According to the 2-tier grading system, 10 a cutaneous MCT should be classified as low-grade unless there are >7 mitoses in ten 400× high-power fields (HPF), >3 multinucleate cells in ten 400× HPFs, >3 bizarre nuclei in ten 400× HPFs, or karyomegaly is present (Fig. 16).
Other features
A variable number of eosinophils is usually present among neoplastic mast cells in cutaneous and subcutaneous MCTs. In many cases, eosinophilic degranulation entraps brightly eosinophilic collagen bundles, a feature that is referred to as collagenolysis (a misnomer, given that collagen is not a cell and therefore cannot suffer lysis) or flame figure (Fig. 17). Lymphocytic inflammation, hemorrhage, edema, and necrosis, with or without epidermal ulceration, can be frequent.10,16,22
Plasma cell tumor
Subgross features
Most PCTs are well-demarcated neoplasms that efface and expand the dermis (Fig. 18). However, some tumors can be poorly demarcated and infiltrate the subcutaneous tissue, leading to a characteristic entrapment of adipocytes among neoplastic cells (Fig. 19).
8
Neoplastic plasma cells often spare the superficial dermis and dermal-epidermal junction (Fig. 20),8,9 a feature often referred to as the grenz zone (
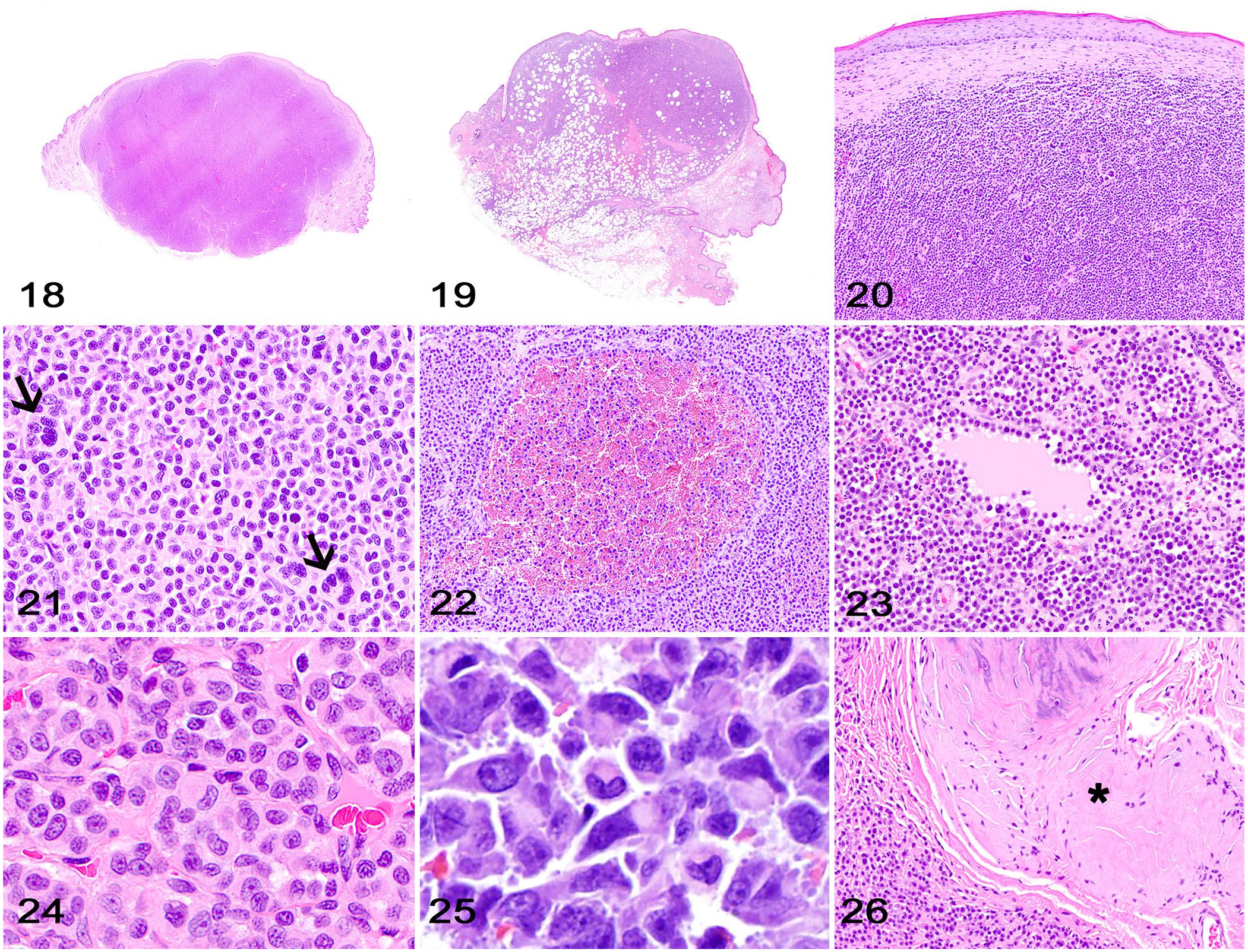
Cutaneous plasma cell tumor (PCT).
Overall cell arrangement
Neoplastic plasma cells form sheets and packets supported by a fine fibrovascular tissue stroma, and efface the dermis or subcutaneous tissue (Fig. 21).3,8,11,18 Subsets of PCTs have a distinct cell arrangement characterized by the typical sheets and packets intercalated with pseudoglandular structures containing central accumulations of blood (Fig. 22) or eosinophilic proteinaceous material (Fig. 23). 11
Cellular features
Cellular features (Fig. 24) can be better visualized at the periphery of the neoplasm. 8 Neoplastic plasma cells have a moderate-to-abundant amount of eosinophilic, and often glassy or granular, cytoplasm that can have a clear perinuclear area that corresponds to the Golgi apparatus (Fig. 25). Nuclei are round-to-oval to crescent-shape, central or eccentric, and have a dense chromatin arrangement with single or several nucleoli that has been historically referred to as clock face chromatin pattern. Binucleate and multinucleate neoplastic cells are observed commonly throughout the neoplasm. The mitotic activity varies from absent-to-low in most cases, but some PCTs can have a high number of mitoses. 8 Anisocytosis and anisokaryosis are usually evident.3,11,18 Although a classification system for canine cutaneous and mucocutaneous PCTs has been reported based on distinct cellular morphology, 18 no association between tumor type and behavior, prognosis, or treatment has been found to date.3,18
Other features
Subsets of PCTs can exhibit extensive amyloid production with or without chondroid and/or bone metaplasia (Fig. 26) and mixed inflammation, features that can dramatically efface the neoplastic cell proliferation and make the diagnosis difficult.8,9
Lymphoma
Subgross features
The main categories of cutaneous lymphoma commonly observed in the routine diagnostic caseload are epitheliotropic and non-epitheliotropic lymphoma.5,6,13 In epitheliotropic lymphoma, neoplastic lymphocytes efface the superficial dermis and the dermal-epidermal junction (Fig. 27), with a clear tropism (invasion) for the epidermis and adnexal structures, particularly hair follicle epithelium and apocrine gland epithelium.5,6
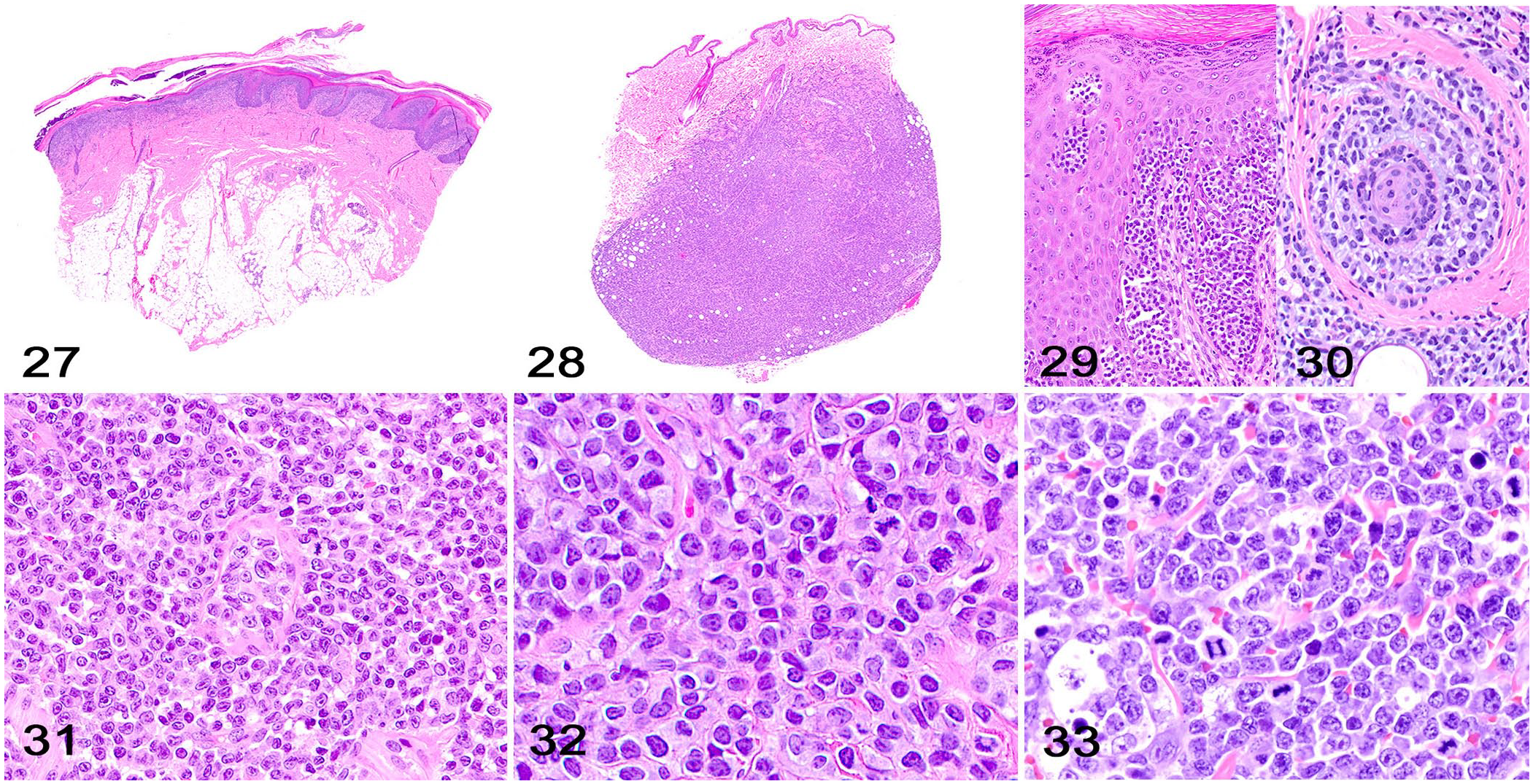
Cutaneous lymphoma.
Non-epitheliotropic lymphoma consists of sheets of neoplastic lymphocytes that more extensively efface the dermis and the subcutaneous tissue (Fig. 28).8,13 Similar to PCTs, non-epitheliotropic lymphomas typically have an area of uninvolved superficial dermis (grenz zone). 8 The classification of different subtypes of epitheliotropic and non-epitheliotropic lymphoma is heavily based on human medicine, is considered controversial in veterinary medicine, 8 and is beyond the scope of this article.
Overall cell arrangement
In epitheliotropic lymphomas, neoplastic cell invasion is characterized by distinct clusters or diffuse infiltration of neoplastic lymphocytes within the epidermis (Fig. 29) and/or follicular and apocrine gland epithelium (Fig. 30).5,6 The distinct clusters of neoplastic lymphocytes in the epithelium have been referred to historically as Pautrier microabscesses.5,8,9 However, there have been discussions within academic circles to abandon the use of eponyms in science, 24 and we would encourage pathologists and residents to use a more descriptive term for these epidermal clusters of neoplastic lymphocytes. Dr. Lucien M. Pautrier (1876–1959), a renowned French dermatologist, did not originally describe Pautrier microabscesses, but was given credit for a discovery that now bears his name. 7 Further, the small clusters of neoplastic lymphocytes within the epidermis are clearly not microabscesses.
Neoplastic cells in non-epitheliotropic lymphoma are arranged in cohesive sheets and packets supported by a fine fibrovascular stroma or preexisting tissues (Fig. 31).8,13
Cellular features
Cellular features of cutaneous lymphomas may vary greatly. 8 Neoplastic lymphocytes usually have scant, round, eosinophilic, homogeneous or finely vacuolated cytoplasm with variably distinct cell margins. Neoplastic cells tend to be small or medium in epitheliotropic lymphomas (Fig. 32) and larger in non-epitheliotropic lymphomas (Fig. 33). 8 Nuclei are round, irregular, and frequently indented. Small neoplastic lymphocytes have dense chromatin; medium-to-large lymphocytes have finely stippled chromatin and 1 or 2 nucleoli. Anisocytosis and anisokaryosis are low when small neoplastic lymphocytes dominate but tend to increase in lymphomas with a predominantly large cell population. The mitotic activity varies and will be a determinant in the tumor grade.5,6,8,9,13
Other features
Similar to MCTs, scattered eosinophils are usually observed among neoplastic lymphocytes in cutaneous lymphomas.5,8
Histiocytoma
Subgross features
Cutaneous histiocytomas are well-demarcated neoplasms that efface the dermis and often part of the subcutaneous tissue in a characteristic top-heavy configuration (Fig. 34) that resembles an inverted trapezoid. 8 In most histiocytomas, neoplastic cells form loose cords near the dermal-epidermal junction (Fig. 35) and can invade the epidermis (Fig. 36). Tumors typically push against the overlying epidermis, and longer-standing tumors are often ulcerated and hemorrhagic. Clusters of lymphocytes are present in the majority of histiocytomas and are more evident at the base of the tumor (Fig. 37). Scattered areas of tumor necrosis can be observed frequently. Although histiocytomas occur in dogs of all ages, a great proportion of histiocytomas occur in younger dogs. 12
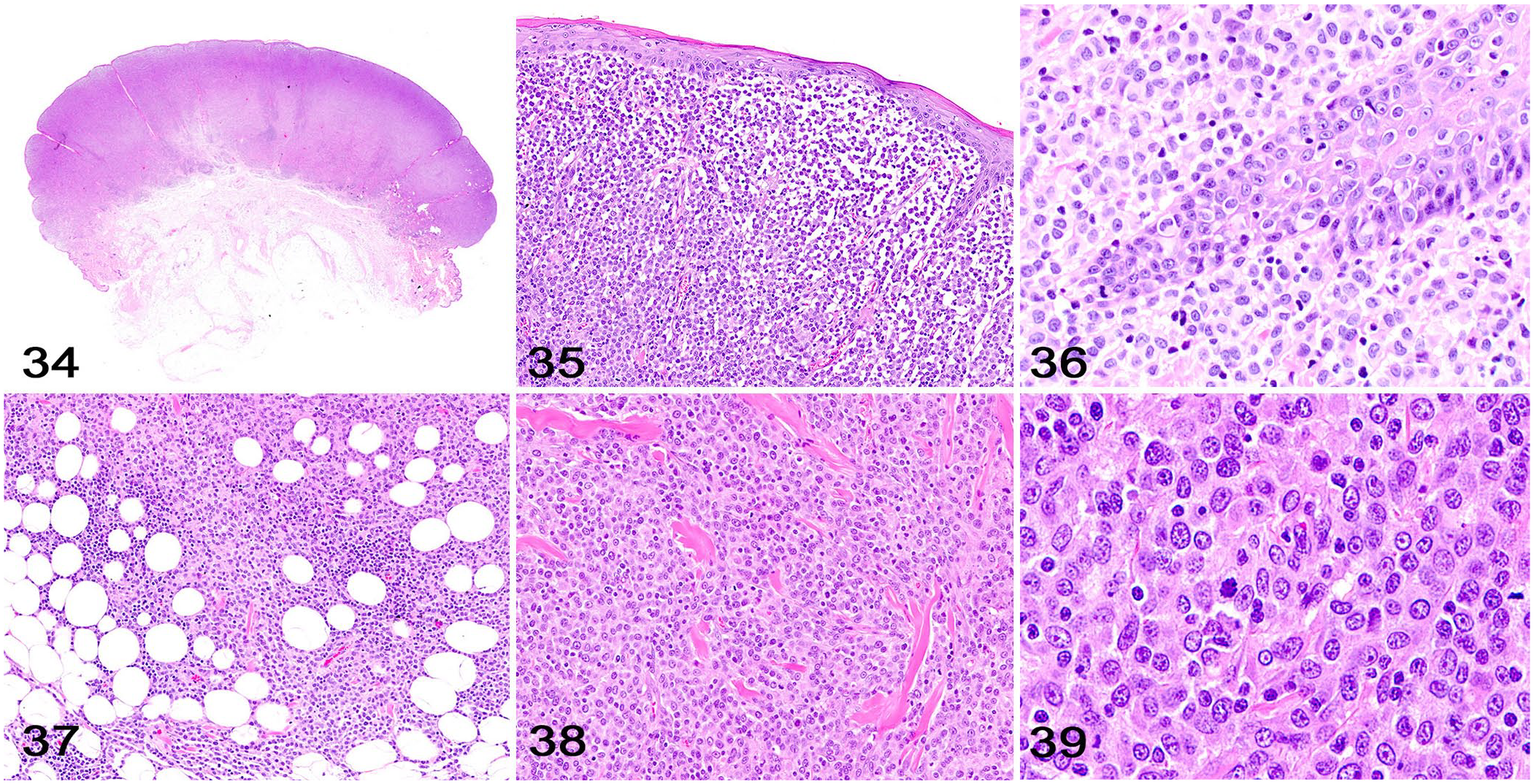
Cutaneous histiocytoma.
Overall cell arrangement
Neoplastic cells form solid sheets that almost completely efface the dermal architecture and the affected subcutaneous tissue, and are arranged in closely packed sheets supported by preexisting collagen or a scant fibrovascular stroma (Fig. 38).
Cellular features
Neoplastic cells have moderate pleomorphism and a moderate-to-abundant amount of round, eosinophilic, homogeneous or finely vacuolated cytoplasm with indistinct cell borders (Fig. 39). Nuclei are round, indented, or folded, and have finely stippled chromatin with 1 or 2 nucleoli. Anisocytosis and anisokaryosis are moderate-to-marked. Mitotic activity is typically high.
Other features
Most canine cutaneous histiocytomas regress spontaneously. Regressing histiocytomas typically have epidermal ulceration, areas of tumors necrosis, and dense lymphocytic inflammation that can obscure or outnumber the neoplastic cell population, making the diagnosis more difficult. 8 In such cases, a search for residual superficial cords of neoplastic cells near the dermal-epidermal junction may be helpful in making the diagnosis.
Histiocytic sarcoma
Subgross features
Cutaneous histiocytic sarcomas can be localized or multiple, and can be part of disseminated disease affecting internal organs. Tumors are poorly demarcated and locally invasive, and expand the dermis and subcutaneous tissue. 12
Overall cell arrangement
Neoplastic dendritic cells are arranged in cohesive sheets or coalescing nodules supported by scant fibrovascular stroma. 8
Cellular features
Cellular morphology varies among cases or within the same case, but neoplastic cells have abundant, round-to-polygonal, eosinophilic, homogeneous or vacuolated cytoplasm with distinct cell borders. Nuclei are round-to-oval to indented and have finely stippled chromatin with one-to-several nucleoli (Fig. 40). Phagocytosis by neoplastic cells can be observed throughout the neoplasm. A polygonal to spindle-cell component with prominent cell processes can also be present in some tumors, which may help in the differentiation from a RCN. There is marked anisocytosis and anisokaryosis, with prominent giant neoplastic cells containing several irregular nuclei. The mitotic count is variable but typically high.8,12
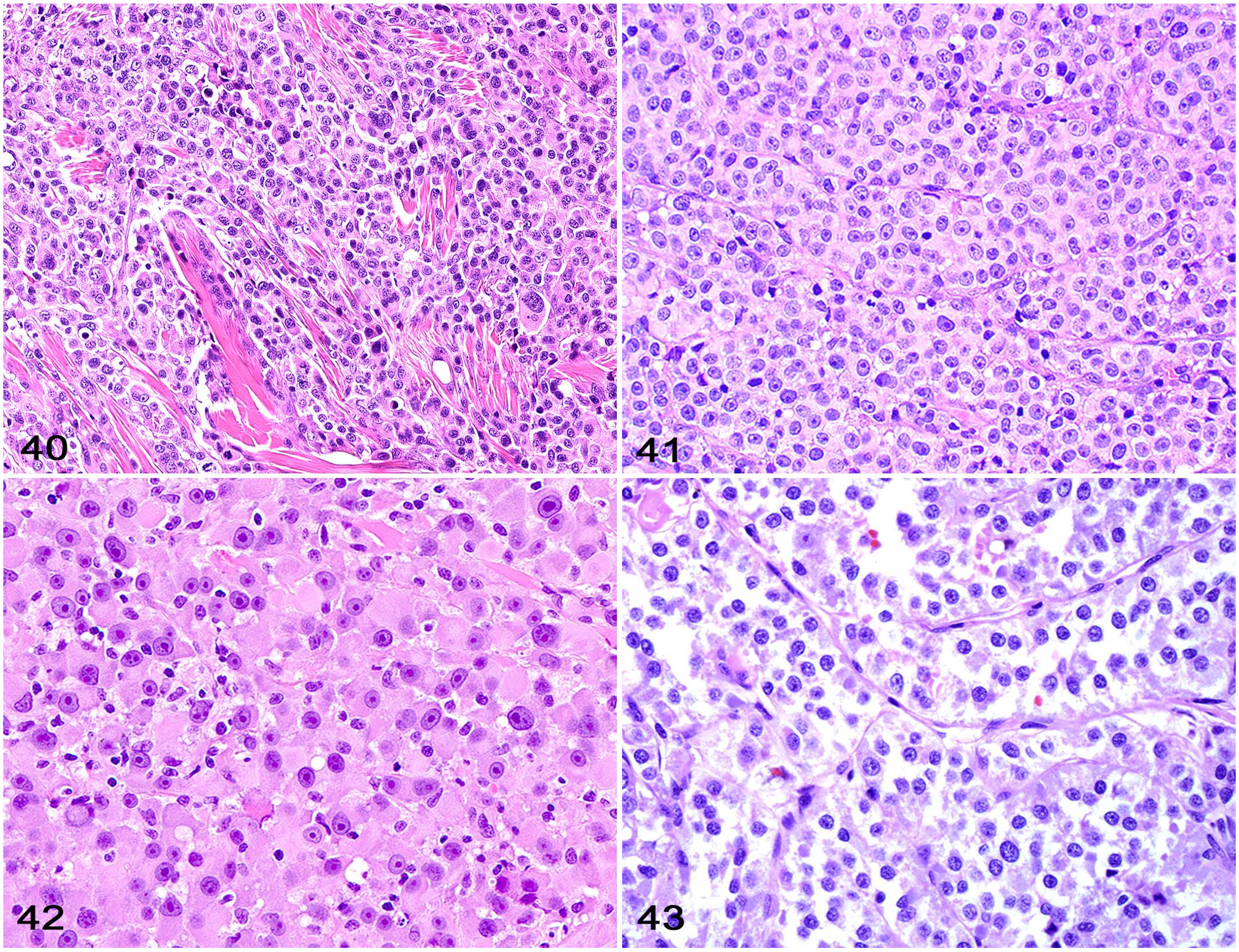
Histiocytic sarcoma. Neoplastic cells form solid sheets and have abundant, round-to-polygonal cytoplasm with distinct cell borders and round-to-indented nuclei with finely stippled chromatin. There is marked anisocytosis and anisokaryosis, with prominent giant neoplastic cells containing several irregular nuclei. H&E.
Transmissible venereal tumor
Subgross features
TVTs are poorly demarcated and typically efface the dermis and subcutaneous tissue. Invasion of the epidermis can be present rarely. Clusters of lymphocytes with fewer plasma cells and neutrophils are often distributed throughout the tumor.9,14
Overall cell arrangement
Neoplastic TVT cells form sheets that efface the dermis and subcutaneous tissue. A delicate fibrovascular stroma is usually present and often separates neoplastic cells into small packets.9,14
Cellular features
Neoplastic TVT cells are round-to-polyhedral and have a moderate amount of round-to-polyhedral, eosinophilic, and finely vacuolated cytoplasm with indistinct cell margins. Nuclei are uniform, round, and central, and have finely stippled chromatin with one prominent nucleolus (Fig. 41). The mitotic activity is typically high.9,14
Amelanotic malignant melanoma and neuroendocrine carcinoma
Although not true RCNs, amelanotic malignant melanomas and neuroendocrine carcinomas can occasionally mimic RCNs and thus should be considered when other possibilities have been ruled out.17,20
Subgross features
These neoplasms can be either well demarcated, poorly demarcated, or locally invasive, and typically efface the dermis and occasionally the subcutaneous tissue. Neoplastic melanocytes can occasionally invade the epidermis or follicular epithelium. 17
Overall cell arrangement
Neoplastic melanocytes are arranged in sheets or packets supported by preexisting tissues or a fine fibrovascular stroma (Fig. 42). In neuroendocrine carcinomas, neoplastic cells form distinct groups or packets supported by a fine fibrovascular stroma (Fig. 43).
Cellular features
Most melanomas consist of spindle cells with variable degrees of cytoplasmic melanin, and offer a straightforward path to a routine diagnosis. However, other melanomas are composed of sheets of round cells with abundant glassy cytoplasm and distinct cell margins with no cytoplasmic pigment. Nuclei are round-to-oval and have finely stippled chromatin with a large nucleolus. Anisocytosis and anisokaryosis, as well as mitotic activity, are variable among tumors. 21
Similarly, a routine diagnosis of neuroendocrine carcinoma can be achieved in most cases, but tumors with less-evident packeting of neoplastic cells may closely resemble a RCN.17,20 Neoplastic cells are polygonal-to-round and have a moderate amount of eosinophilic, often vacuolated, cytoplasm. Nuclei are round and have coarse chromatin with indistinct nucleoli. Anisocytosis and anisokaryosis are low-to-moderate. Mitoses are typically frequent.
Poorly differentiated round cell tumors
The accurate classification and grading (if applicable) of a RCN is of paramount importance for the determination of the most appropriate treatment and prognosis. 4 A final diagnosis can often be reached by histology through systematic evaluation of the key morphologic hallmarks of a particular neoplasm.4,17,20 However, histologic evaluation alone may be challenging even for the experienced pathologist, especially in cases in which neoplasms lack the key morphologic features necessary for a routine diagnosis.4,17,20 Cutaneous RCNs that exhibit no morphologic features utilized for a specific diagnosis are frequently referred to as poorly differentiated RCNs. In contrast, other RCNs may exhibit some morphologic features that, while suggestive of a specific diagnosis, are not confirmatory. In either case, IHC will be necessary for a final diagnosis.4,19
A great number of IHC protocols have been validated and successfully utilized to diagnose RCNs in dogs (and other animal species) in many diagnostic laboratories. 19 The use of particular IHCs to diagnose a RCN may vary according to the pathologist, and will typically depend on the availability of specific antibodies and on the standardized IHC protocols offered by different institutions. The immunomarkers listed in Table 1 are those routinely used by the authors on diagnostic samples submitted to the Athens Veterinary Diagnostic Laboratory (Athens, GA, USA) and Antech Diagnostics (various locations); most are commonly suggested by scientific publications describing cutaneous RCNs.4,12,14,17,19,20
Expected immunohistochemistry features of canine cutaneous round cell tumors.
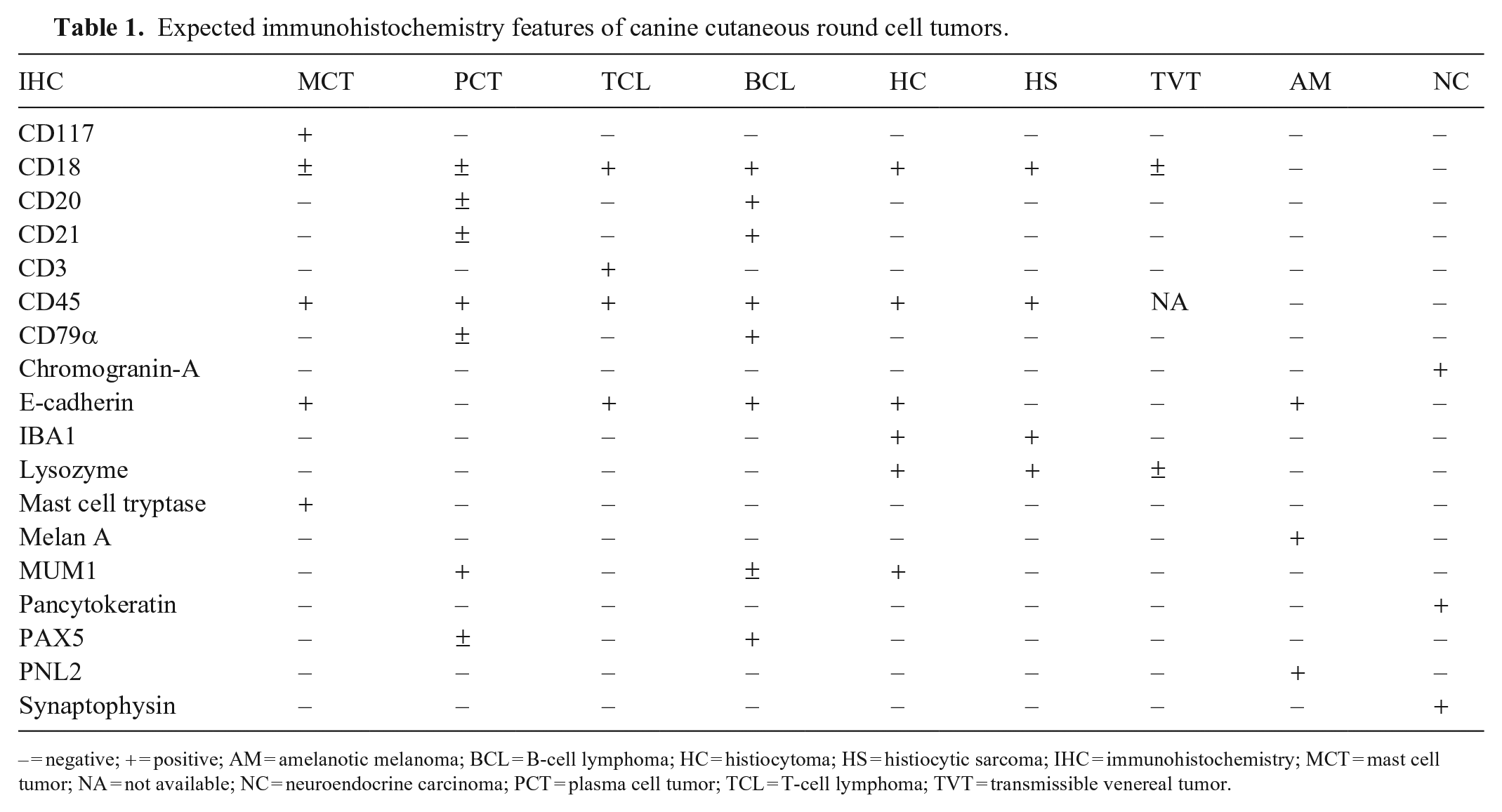
– = negative; + = positive; AM = amelanotic melanoma; BCL = B-cell lymphoma; HC = histiocytoma; HS = histiocytic sarcoma; IHC = immunohistochemistry; MCT = mast cell tumor; NA = not available; NC = neuroendocrine carcinoma; PCT = plasma cell tumor; TCL = T-cell lymphoma; TVT = transmissible venereal tumor.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
