Abstract
A fundamental goal of research into the basic mechanisms of aging is to develop translational strategies that improve human health by delaying the onset and progression of age-related pathology. Several interventions have been discovered that increase life span in invertebrate organisms, some of which have similar effects in mice. These include dietary restriction and inhibition of the mechanistic target of rapamycin by treatment with rapamycin. Key challenges moving forward will be to assess the extent to which these and other interventions improve healthy longevity and increase life span in mice and to develop practical strategies for extending this work to the clinic. Companion animals may provide an optimal intermediate between laboratory models and humans. By improving healthy longevity in companion animals, important insights will be gained regarding human aging while improving the quality of life for people and their pets.
Keywords
Research on the basic biology of aging aims to understand mechanisms that cause organisms to decline in function over time and lead to increasing risk of morbidity and mortality. 61 This is, of course, intimately connected to pathology because aging promotes disease. Most leading causes of mortality in developed nations share a single greatest risk factor, and it isn’t how much you eat, drink, smoke, or exercise; it’s how old you are. 39 Diabetes, heart disease, kidney disease, stroke, Alzheimer disease, Parkinson disease, and most forms of cancer, along with several other diseases, all show an exponential increase in risk with age over much of the human life span (Figs. 1, 2).
Aging drives disease. Age is the greatest risk factor for most causes of death and disability in developed countries.
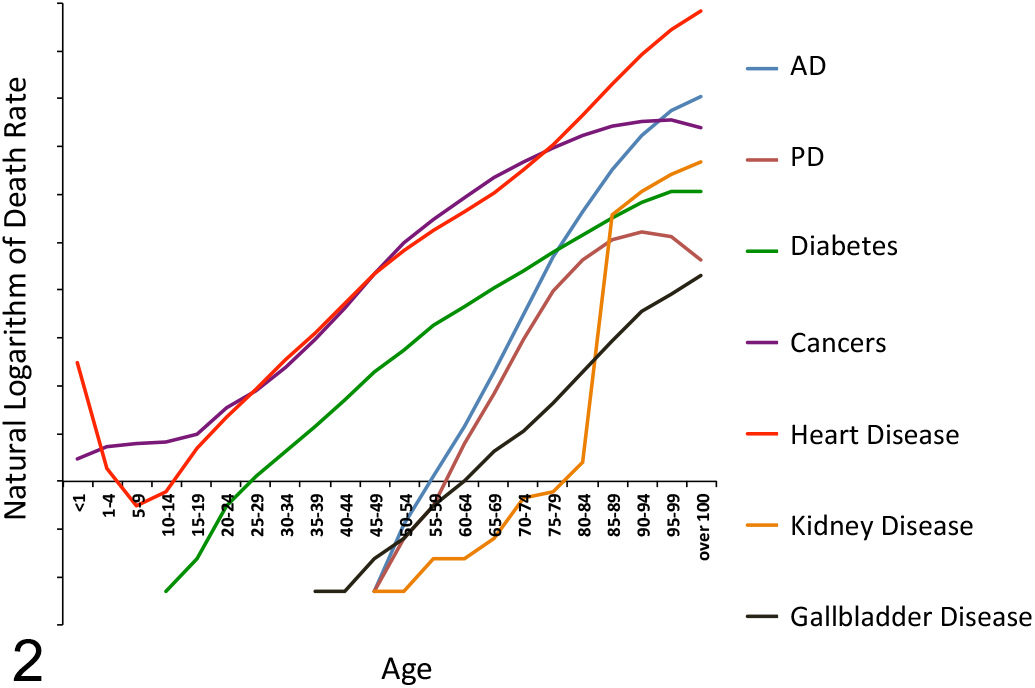
Age-specific death rates for some leading causes of death increase exponentially with age. Natural logarithm of the death rate refers to an individual’s risk of dying from a given disease at the specified age. Death rate data from the US Centers for Disease Control and Prevention. AD, Alzheimer disease; PD, Parkinson disease.
Understanding why this relationship between age and disease exists and, ultimately, intervening in the aging process at a molecular level to promote healthy longevity is a primary goal of aging research. In this way, aging research is the ultimate form of preventative medicine: if the rate of aging can be slowed, then many of the diseases and declines in function associated with aging should be simultaneously delayed. Thus, interventions that target the molecular mechanisms of aging have the potential to increase both life span and health span, which can be defined as the period of life free from chronic disease and disability.
The field of pathology plays a critical role in aging research—from the perspective of understanding the spectrum of diseases that occur during aging in different model organisms and different individuals within a population and in evaluating the impact of genetic and environmental factors on health span. This is particularly true in laboratory animal models where it is often challenging to quantify health span. Diagnosing and quantifying disease rates in aged animals in particular is essential for understanding the impact of factors that may increase the risk of age-associated disease, as well as for assessing whether interventions designed to delay aging are actually having the desired effect.
Conserved Longevity Pathways
Studies of aging in yeast and invertebrate model organisms such as Caenorhabditis elegans and Drosophila melanogaster have had a large impact on the field by allowing detailed mechanistic studies of molecular mechanisms of aging and high-throughput genetic analysis of modifiers of life span. 52,86 One of the most important findings to emerge from these model systems is that many aspects of aging are highly conserved across broad evolutionary distances. This includes genetic modifiers of longevity, as well as specific cellular processes that degrade with age and likely contribute to age-associated pathology. Indeed, a recent review categorized these conserved features of aging into a set of 9 “hallmarks of aging,” 57 many of which span the evolutionary distance from yeast to people (Fig. 3). The 9 hallmarks of aging identified by Lopez-Otin and colleagues are as follows: genomic instability, mitochondrial dysfunction, deregulated nutrient sensing, loss of proteostasis, epigenetic alterations, cellular senescence, stem cell exhaustion, altered intracellular communication, and telomere attrition. Among these, only telomere attrition has not been definitively associated with aging in invertebrate model organisms.
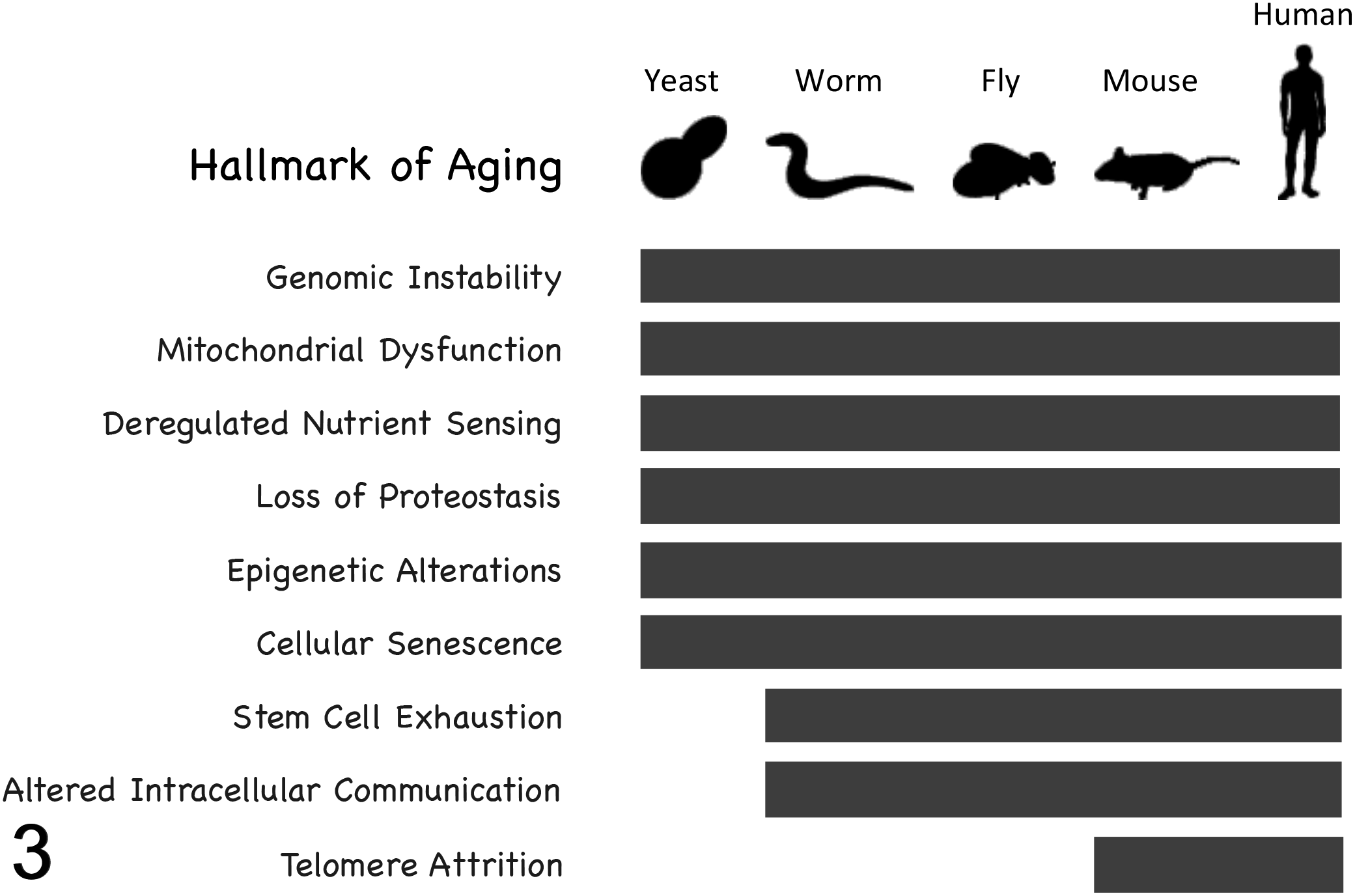
Conserved mechanisms of aging. Eight of the 9 identified “hallmarks of aging” have been definitively implicated in aging of nonmammalian species.
In addition to providing insight into fundamental mechanisms of aging, there are several examples where studies of aging in nonmammalian model systems have led to the identification of interventions that extend life span in mammals. Among these are genetic or pharmacologic inhibition of the mechanistic target of rapamycin (mTOR), deletion of the mTOR substrate S6 kinase, mutations that reduce insulin/IGF-1-like signaling (including loss-of-function mutations to the insulin/IGF-1 receptors), overexpression of sirtuin family enzymes, and treatment with the antidiabetic drug metformin 72 (Fig. 4; see additional references*). The discovery of pathways and pharmacologic interventions that delay aging in nonmammalian species and the subsequent demonstration of their efficacy in rodent models (Text Box 1) further support the idea that fundamental aspects of aging are conserved at a genetic and molecular level, suggesting that screening for longevity-promoting compounds in yeast or invertebrate species may prove fruitful for initial identification of candidates for further analysis in mice.
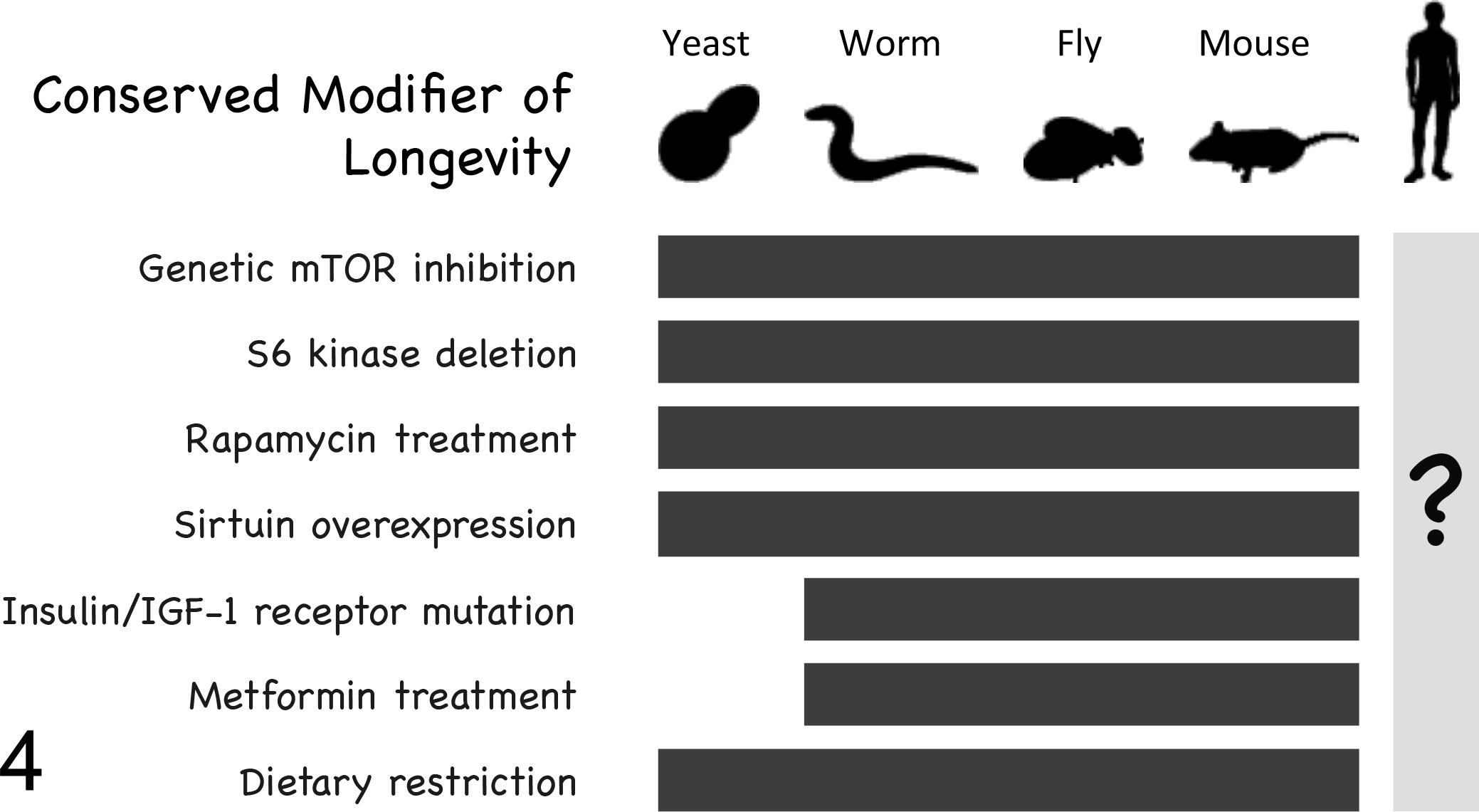
Conserved modifiers of aging. Seven genetic and environmental modifiers of longevity modulate life span similarly in yeast, worms, flies, and mice. The question mark (?) under the human figure indicates that we do not yet know whether these factors similarly modulate human aging. mTOR, mechanistic target of rapamycin.
Many mouse models of extended longevity were first shown to modulate aging in nonmammalian organisms. These include components of the insulin/IGF-1 signaling pathway, the sirtuins, and the mechanistic target of rapamycin (mTOR).
Among genetic models of enhanced longevity in rodents, mutations that perturb growth hormone and insulin/IGF-1 signaling are the best documented and most robust. 5,38 This pathway was first implicated in aging of Caenorhabditis elegans, with studies showing that mutation of the insulin-like receptor gene, daf-2, or the PI-3-kinase gene, age-1, is sufficient to increase life span. 26,51 These long-lived mouse models include the Ames and Snell dwarf mice, growth hormone receptor knockout mice, growth hormone–releasing hormone knockout mice, IGF-1 receptor heterozygous mice, fat-specific insulin-receptor knockout mice, and IGF-1-deficient mice. 9,17,23,33
The sirtuins are a family of NAD+-dependent enzymes that remove posttranslational modifications from proteins. 28 The major catalytic activity of most sirtuins is protein deacetylation 34 ; however, sirtuins also have ribosyltransferase activity and, in the case of mammalian SIRT5, the ability to remove additional posttranslational modifications from lysine residues, including glutaryl, malonyl, and succinyl groups. 28 Sirtuins first became implicated in aging based on studies in budding yeast demonstrating that overexpression of the yeast Sir2 gene was sufficient to extend lifespan. 43 Mammals have seven sirtuin genes, SIRT1–SIRT7. SIRT1 is the ortholog of yeast Sir2; however, whole body overexpression of SIRT1 does not extend life span in mice. A recent report describes increased life span from overexpression of SIRT1 only in specific regions of the brain, 79 and whole body overexpression of another sirtuin, SIRT6, can extend life span in male mice. 46
In recent years, the mTOR pathway has emerged as perhaps the most important modulator of healthy aging in mice, with many clinically relevant parallels in a variety of human diseases. 36,40 The mTOR protein is a nutrient- and growth factor–responsive kinase that exists in 2 complexes: mTOR complex 1 (mTORC1) and mTOR complex 2. Genetic or pharmacologic (ie, rapamycin) inhibition of mTORC1 extends life span in yeast, worms, flies, and mice. 90 In mice, life span extension from genetic inhibition of mTORC1 has been documented through heterozygous knockout of mTOR along with the mTORC1 component mLST8, 53 as well as in a second study of mice carrying hypomorphic alleles of mTOR. 99 Deletion of the gene encoding the mTORC1 substrate protein S6k1 also increases life span in female mice. 83 The mechanisms by which mTORC1 modulates aging remains an area of active research and probably involves cell-intrinsic processes, including regulation of mRNA translation, autophagy, and mitochondrial function, as well as systemic effects on metabolism, inflammation, and immunity.
Dietary Restriction and Rapamycin: Two Interventions That Enhance Longevity
Dietary Restriction
The best-characterized intervention for increasing life span in mammals is dietary restriction (DR; also referred to as caloric or calorie restriction), which can be defined as a reduction in nutrient availability in the absence of malnutrition. First described to increase life span in rats in the 1930s, 65 DR has since been shown to also increase life span in a plethora of other species, including yeast, worms, fruit flies, mice, fish, spiders, bats, and rhesus monkeys. 1,50,63,70 Several paradigms for restricting nutrients in rodents have been described, including chronic restriction of chow from 20% to 50%, intermittent or every-other-day feeding, and diets low in methionine or tryptophan. 74,89 Each of these has been proposed to improve health during aging and, to a varying extent, also increase life span; however, the extent to which these different forms of nutrient restriction have similar causal mechanisms is still unclear. Chronic restriction is both the most studied and the most robust DR paradigm for extending life span, with 30% to 50% restriction typically increasing median life span by approximately 30% to 50%.
In addition to having their life spans extended, rodents subjected to DR have dramatically reduced rates of most age-associated cancers, do not become obese, maintain glucose homeostasis and insulin sensitivity, and are protected against age-associated cognitive and cardiac declines. 27,75,87 DR has also been reported to confer protection against age-related disease models in mice, including models of Alzheimer and other neurodegenerative diseases. 82 Similar reports of improved health span have also been seen in 2 independent studies of DR in rhesus monkeys, with the DR groups showing lower rates of diabetes, obesity, cardiovascular disease, and cancer. 16 Interestingly, DR increased life span in only 1 of these 2 studies, although the reasons for this difference remain unclear. 15,64
Although DR has been shown to increase life span and delay age-associated decline in a variety of species and numerous independent studies, there is growing recognition of the importance that genetic background may play in individual response to DR. A group of studies performed in 41 recombinant inbred lines of mice, for example, found that roughly as many of the lines had their life spans shortened by a 40% reduced-calorie DR regimen as had their life spans extended. 54 This is a similar distribution as seen in a study of DR response in >100 single-gene deletion mutants in budding yeast. 80 This study is noteworthy, as it identified functional categories that showed differential responses to DR: mitochondrial mutants tended to show the largest life span extensions, while superoxide dismutase–deficient cells or mutants defective for acidification of the vacuole (yeast lysosome) tended to have their life spans shortened by DR. 81 Whether these functional relationships observed in yeast hold true in mammals has yet to be established, although a follow-up study based on the yeast work found that inhibition of mTOR (a downstream effect of DR; see below) rescued a severe mitochondrial disease in mice caused by deficiency of complex I of the electron transport chain. 37 It has been speculated that genetic background differences may account for the failure of DR to increase life span in some inbred mouse lines, as well as in a wild-derived mouse strain. 21,31,91 It is worth noting, however, that in nearly every case where DR failed to extend life span in mice, only a single DR level and regimen were assessed. Thus, it may be that many genotypes alter the response profile for DR such that a different level of restriction is necessary to obtain an optimal (or perhaps any) beneficial effect on aging. This concern illustrates the seminal importance of dose-response considerations in biology, something that is often lacking from studies of aging-related biology.
Rapamycin
Among pharmacologic interventions that extend life span in mice, rapamycin is the most documented and effective to date. Rapamycin is a specific inhibitor of the mTOR complex 1 (see Text Box 1) that is approved by the US Food and Drug Administration for clinical use to prevent organ transplant rejection, in cardiac stents, and for certain forms of cancer. 40 Rapamycin was first shown to increase mouse life span by the National Institute on Aging Intervention Testing Program by delivering it to UMHET3 mice in an encapsulated form in the diet beginning at 20 months of age. 32,42 This regimen increased median life span of male mice by about 9% and that of female mice by about 14%. Subsequent studies showed that a similar dietary regimen increases life span comparably when initiated at 9 months of age and that dietary delivery at a 3-fold-higher concentration increases median life span in male and female mice by about 25%. 66,67 Several other groups have reported life span extension from rapamycin delivered in the diet and by injection in C57BL/6 and 129/Sv mice. 3,25,68,100
In addition to extending life span, rapamycin has been shown to positively affect multiple age-related declines in function in mice as well as mouse models of specific age-related diseases. 36 This includes improved cognitive function during normative aging and in mouse models of Alzheimer disease. 11,30,55,60,88 Of particular note, treatment with rapamycin during midlife can at least partially reverse age-related declines in immune function and cardiac function within 6 weeks of treatment in mice. 13,18,24 Despite these improvements in multiple organ systems during aging, it has remained somewhat controversial whether rapamycin generally slows aging and improves health span or is primarily increasing life span through reduction in cancer incidence (discussed in the next section). 8,19,76
The Importance of Quantifying Health Span in Addition to Life Span
There is growing recognition within the field that it is important to obtain quantitative measures of health span in addition to assessing the effects of different interventions on life span. 10,49 As mentioned above, the interventions that robustly increase life span in mice also tend to delay multiple age-related declines in function along with major age-related diseases, especially cancer; however, there have been relatively few studies that have attempted to assess overall quality of life in these animals. There are at least 2 major reasons for this deficiency: first, mouse longevity studies to assess life span are already expensive, and the addition of animals and assays to measure health span is often prohibitive; second, there is no consensus as to which assessments best capture overall health during aging in rodents.
Recent studies on rapamycin provide a particularly good example of challenges associated with definitively assessing the effect of an intervention on health span in mice. As described above, rapamycin treatment has been shown to extend life span in several mouse strains and to improve outcome in age-related disease models. In addition, at least 2 studies attempted to directly address the question of whether rapamycin “slows aging” by assessing a breadth of age-related parameters encompassing several organ systems. Although both studies detected significant increases in life span and improvements in at least a subset of the parameters measured, they reached opposite conclusions: Wilkinson et al 98 reported that “rapamycin slows aging in mice,” while Neff et al 68 reported that rapamycin “has limited effects on aging.”
Of the 2 studies, Neff et al 68 performed the more comprehensive assessment of health, looking at around 40 distinct measures of health span, including histopathologic assessments of liver, brain, muscle, heart, kidney, and endocrine organs, as well as precancerous and cancerous lesions. They detected improvements in about 40% of these health span measures following rapamycin treatment. On the basis of their inability to detect positive changes in the remaining measures of health span, in combination with the fact that rapamycin induced changes in some health span measures even in young animals, they concluded that rapamycin does not generally slow aging. 68 One criticism of such an interpretation is that it relies on what is essentially a series of negative results (failure to detect a change) in the absence of a positive control (something that elicits the change that the researchers were trying to detect). The strength of this interpretation is further limited by the suboptimal dosing employed in the study. Rapamycin is known to more robustly increase life span at a 3-fold-higher dose than that used by either Neff et al 68 or Wilkinson et al, 98 and it seems likely that some health span measures may be similarly dose responsive. 41
These examples raise some important issues that should be considered in the design and interpretation of experiments aimed at assessing healthy aging in mice. The field will need to work toward consensus regarding which measures of health span are most informative and how many measures of health span are needed to obtain a meaningful picture of overall health. It will also be important to consider what it means if an intervention improves a measure of health span in both young and old animals. Although Neff et al 68 argued that such a result indicates that the intervention is not affecting aging, others have argued that there is no strong justification for such an interpretation. 35 Researchers should be cautious not to overinterpret their data in either direction, particularly when most interventional studies of aging in mice have not been optimized for dose or delivery and are lacking in positive control. In addition, it may be the case that some interventions increase life span and delay most age-associated phenotypes while simultaneously exacerbating other age-related traits or even causing new pathologic conditions. Again, rapamycin presents an interesting example, as there is evidence that chronic rapamycin treatment in mice impairs the ability of animals to respond to a glucose challenge, causes testicular atrophy, and increases risk of cataracts, 7 and there are several side effects in patients taking high doses of rapamycin to prevent organ transplant rejection. 48
A noteworthy advance in the area of experimental health span assessment is the recent description of a noninvasive frailty index for quantifying health span in mice. 97 Indices of frailty have been commonly used in geriatric medicine but are rarely applied in studies of aged rodents. The 31-point mouse frailty index developed by Whitehead et al 97 has the major advantages of being relatively inexpensive and noninvasive. This index has been applied to assess the effects of DR and resveratrol on health span of mice 45 and may prove useful for quantifying healthy aging across a variety of parameters if it becomes widely utilized.
Companion Animals and Citizen Science in Aging Research
As discussed in prior sections, the field has done an admirable job of characterizing key pathways and molecular mechanisms of aging in laboratory models and, in some cases, identifying interventions that appear to slow the rate of aging in these same systems. In addition to the need for improved and more consistent assessments of health span in rodents, a major barrier exists in the translation of these discoveries to improve quality of life for people. There has been much discussion of the importance of targeting human aging to increase health span and reduce the burden of age-related disease, including the anticipated economic and social consequences. 29,49,69 Unfortunately, the path toward accomplishing this goal remains unclear, with significant hurdles associated with testing putative longevity- or health span–promoting interventions directly in people. 72
My colleagues and I believe that companion animals, particularly pet dogs, represent an outstanding intermediate between laboratory animals and humans for understanding the basic mechanisms of aging and, in limited cases, for validating interventions expected to promote healthy longevity. Dogs have several traits that make them particularly compelling in this regard. From an evolutionary standpoint, dogs are roughly as distant from humans as mice are; however, companion dogs experience an environment strikingly similar to that of their owners, something that cannot be mimicked in rodents. 94 Companion dogs also offer a wealth of genetic and phenotypic diversity, with known breed-specific disposition to certain diseases and causes of death. 22,95 Dogs suffer from many of the same age-associated declines as humans, albeit at a much accelerated rate, which makes the time frame for studies of aging in dogs much more reasonable than similar studies in humans. Importantly, the understanding of geriatric ailments and the breadth of treatment options within the canine veterinary community are second only to that of human medicine. Perhaps most important, there is significant intrinsic value to improving healthy aging in companion dogs, as it would not only facilitate future efforts toward similarly affecting human aging but would also significantly enhance the quality of life for the dogs and their owners.
On the basis of this concept, we recently launched the Dog Aging Project (http://www.dogagingproject.com), an initiative with the primary goal of enhancing healthy longevity in companion dogs. 12 There are 2 major goals of the Dog Aging Project: a longitudinal study of aging in companion dogs and an intervention trial to assess the ability of rapamycin to promote health span and increase life span in middle-aged dogs.
The longitudinal study of aging in dogs will follow companion dogs throughout life to define the major genetic and environmental features that determine why some animals age well and have relatively long health span and life span while other animals age poorly. The final design of the longitudinal study of aging is currently being determined, with the support of a National Institute on Aging–funded R13 Networking Grant, which has allowed the formation of the Canine Longevity Consortium, directed by Dr Daniel Promislow (University of Washington, Seattle). The Canine Longevity Consortium is hosting a series of meetings around this topic, and key experimental details were discussed at the most recent conference in Bethesda, Maryland, in March 2015. Current plans for this study involve including up to 100 000 dogs, with assessments of genotype and environmental parameters along with lifetime health measures for all animals. Subsets of animals would participate in more extensive testing of age-associated health parameters, including activity monitoring, cognitive function, metabolic health, cardiac and kidney function, and cancer incidence, as well as additional phenotypic assessments, such as serum metabolomics. In cases where owners are willing to allow autopsy, assessment of tissue pathology will play a critical role in determining disease burden at time of death and will be important for correlating disease with genotypic and phenotypic information.
The rapamycin intervention is a smaller, more focused study designed to assess the effects of low-dose rapamycin on healthy longevity in middle-aged, mid- to large-size companion dogs. To be eligible for the initial study, dogs must weigh at least 40 lb and be at least 6 years old with no major preexisting health conditions. There are no breed exclusions as long as the animals meet these 2 criteria. Owners can enroll their animals for consideration in the study through the Dog Aging Project website. Between November 2014 and February 2015, approximately 1000 animals have been enrolled.
The current study design call for 2 phases. The first phase is primarily a dosing and safety study to ensure that the low-dose rapamycin regimen does not result in significant side effects. A relatively small cohort of at least 32 animals from the Seattle area will be enrolled into the placebo or treatment group for 10 weeks, with an initial veterinary examination prior to enrollment, which will include echocardiography, blood sampling, and sampling of hair, nails, and feces. A second exam after 2 weeks of treatment will verify no significant adverse effects and provide for a second sampling of blood, hair, nails, and feces. A third exam after 10 weeks of treatment will include similar sampling along with a second echocardiogram. In addition to the veterinary care, owners will be asked to carefully monitor their dogs and to immediately report any abnormal behaviors or side effects of the treatment. Echocardiograms are included in the short-term study on the basis of data indicating that 10 weeks of rapamycin treatment in 20-month-old mice is sufficient to reverse some measures of age-related cardiac decline. 18,24 The short-term trial is anticipated to be in progress before publication of this article.
The second phase of the rapamycin intervention study is intended to be a national study that will include several hundred dogs. The age and weight entry criteria will be similar to those of the short-term trial, and the rapamycin treatment duration is anticipated to be from 1 to 3 years. As in the short-term study, adverse events will be closely monitored through a combination of owner reporting and veterinary care, and blood, hair, nails, and feces will be obtained at periodic veterinary examinations. In addition to cardiac function, a more detailed assessment of healthy aging will be obtained, including activity monitoring, cognitive function, metabolic health, cardiac and kidney function, and cancer incidence and mortality rates.
A notable feature of both the longitudinal study of aging and the rapamycin intervention trial is that they incorporate pet owners into the study as citizen scientists. Not only are owners invested in these studies through the participation of their pets, but they also participate directly in the collection of data regarding overall health, behavior, and activity. My colleagues and I believe that this feature of the Dog Aging Project, combined with the close connection that many dog owners feel to their pets, creates a unique dynamic that resonates with the general public in a way that few other scientific endeavors have.
Conclusion
Substantial progress has been made in understanding the cellular mechanisms of aging, which has translated to the identification of several genetic and environmental interventions capable of extending life span in mice. Two of the most robust longevity-enhancing interventions are chronic DR and treatment with the mTOR inhibitor rapamycin. One challenge that the field will need to address in the near future is how best to assess the impact of these and other such interventions on health span and overall quality of life. Quantification of disease burden and end-of-life pathology will undoubtedly be an important part of any such metric. A second major challenge will be to develop tractable approaches to translating these discoveries to improve human health. Companion animals may be a particularly important bridge in this regard, providing informative avenues for future human studies while improving the quality of life for both the animals and their owners.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
