Abstract
This retrospective study aimed to characterize and determine the prevalence of spinal disease in nondomestic felids within a sanctuary population. A review of 304 postmortem examination reports in Panthera species from 2003 to 2021 revealed that 86/304 (28%) were diagnosed with spinal disease. Spinal lesions were categorized according to pathologic process: degenerative (78/86, 91%), developmental (8/86, 9%), inflammatory (6/86, 7%), or neoplastic (8/86, 9%). Degenerative lesions included intervertebral disk disease (IVDD; 66/78, 85%), spondylosis without concurrent IVDD (4/78, 5%), and idiopathic (noncompressive) degenerative myelopathies (8/78, 10%). Fourteen individuals had lesions in more than 1 category. Developmental cases were vertebral (4/8) or spinal cord (3/8) malformations or both (1/8). Inflammatory lesions included meningitis (4/6) and meningomyelitis (2/6). Neoplasia included vertebral multiple myeloma (4/8) and others (4/8). IVDD often involved multiple disks but primarily affected the cervical (41/66, 62%) and thoracic spine (32/66, 48%). A multivariate binary logistic model predicted the diagnosis of IVDD at postmortem examination, where odds of being affected were highest for males, lions (Panthera leo), and geriatric age group (
Spinal disease is a frequent cause for clinical problems in captive large nondomestic felids. 22 Degenerative spinal disease includes intervertebral disk disease (IVDD) and spondylosis, with or without secondary Wallerian degeneration or myelomalacia due to spinal cord compression, and has been described in aged captive Panthera spp. with no apparent sex predilection.8,15,22,25,27 Vertebral malformations have included congenital occipitoatlantoaxial malformation in male juvenile lions (Panthera leo), 9 and spinal cord malformations reported consist of syringomyelia,20,35 hydromyelia, 47 or both in juvenile lions. 42 Spinal neoplasms have also been reported.7,19,21,24,26,46
The relative frequencies of spinal lesions and associated host factors have not been compared among Panthera species. This information would help guide diagnostic and treatment plans for large felids presenting with neurologic clinical signs. The objectives of this study were to determine the prevalence of spinal lesions within Panthera spp. as well as characterize their pathological features.
Materials and Methods
The population for this retrospective study consisted of large nondomestic felids, from a sanctuary facility in East Tennessee that died or were euthanized from 2003 to 2021. Enclosures consisted of earth floors with a wooden deck, which was elevated approximately 15–30 cm off of the ground, and access to a wooden floored den. All animals were in the genus Panthera, greater than or equal to 1-day-old at death, and were examined postmortem by anatomic pathologists at the University of Tennessee College of Veterinary Medicine. The age at death, sex, and species of each animal were recorded. Because the precise age was not known for some individuals, ages were subdivided into the following groups: neonate (0–4 weeks), juvenile (5 weeks–1 year), adult (2–13 years), and aged (≥14 years). Medical records and postmortem examination reports were examined for each individual to identify spinal disease. Individuals were designated as having spinal disease based on the postmortem documentation of gross or microscopic lesions involving the spinal cord or vertebral column.
The relevant clinical history and postmortem findings for each spinal disease case were recorded. Spinal lesions were categorized as degenerative, developmental, inflammatory, or neoplastic. Gross photographs, when available, and histological slides were reviewed for each case. The individual animal data analyzed in this study are not available as Supplemental Materials, but can be provided upon request.
A binomial “exact” calculation was used to determine the 95% confidence interval for spinal disease prevalence. Binary logistic regression was used to examine whether age, sex, or species were associated with the likelihood of having a diagnosis of IVDD at postmortem examination. Statistics were not performed for other lesions as there were insufficient data to reliably analyze. For this analysis, species groups were divided into lions, tigers (Panthera tigris), and other to provide sufficient sample sizes for comparisons among groups. Other included leopards (Panthera pardus), snow leopards (Panthera uncia), jaguars (Panthera onca), ligers (P. leo × P. tigris), and liligers (P. leo × liger). Preliminary analysis using chi-squared tests suggested each explanatory variable was associated with IVDD. Statistical analyses were performed using IBM SPSS Statistics, Version 28.0.0.0 (SPSS Inc., Chicago IL), and values of P < .05 were considered significant. The fit of the regression model was assessed using the Nagelkerke R2 and Hosmer-Lemeshow test.
Results
The study population included 304 Panthera spp. (Table 1), representing 7 species, 160 males and 143 females, and for those animals of known age, a median age of 16 years (range = 5 days–25 years). The sex of 1 individual was unknown. Spinal disease was diagnosed in 86 individuals, including 55 males and 31 females, and for those animals of known age, the median age was 17 years (range = 4 weeks–22 years). Spinal lesions were categorized into 4 pathologic processes: degenerative (78/86, 91%), developmental (8/86, 9%), inflammatory (6/86; 7%), and neoplastic (8/86, 9%). Fourteen animals had more than 1 spinal lesion caused by 2 distinct pathologic processes.
Frequency and prevalence of spinal disease diagnosed at postmortem examination, stratified by pathological process, in Panthera spp. from a captive population.
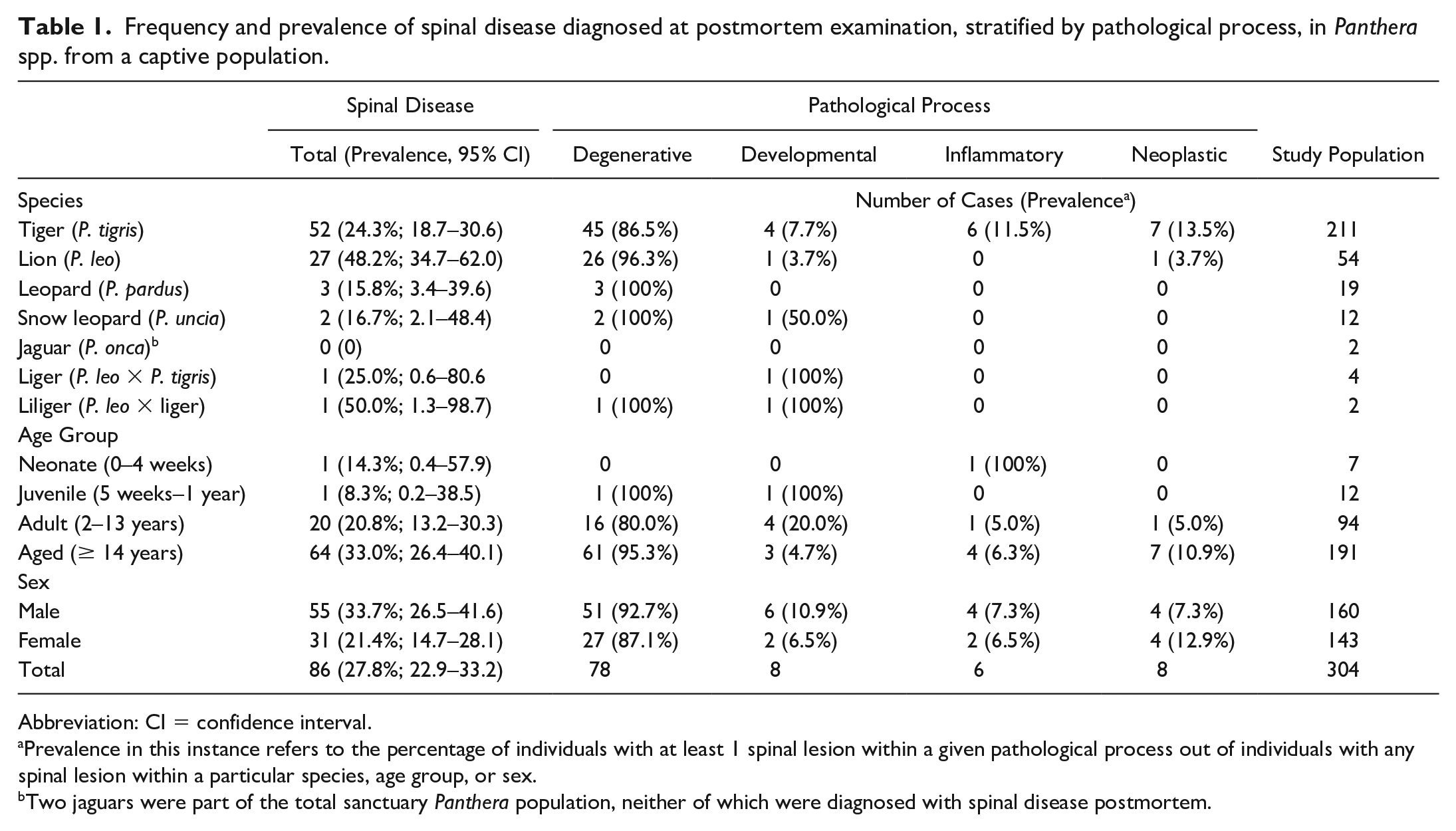
Abbreviation: CI = confidence interval.
Prevalence in this instance refers to the percentage of individuals with at least 1 spinal lesion within a given pathological process out of individuals with any spinal lesion within a particular species, age group, or sex.
Two jaguars were part of the total sanctuary Panthera population, neither of which were diagnosed with spinal disease postmortem.
The majority of animals with spinal lesions had associated clinical signs (73/86, 85%), including ataxia (45/86, 52%) and paresis (57/86, 66%), with 37 of 57 paretic cases (65%) progressing to nonambulatory. More specifically, 24/45 (53%) ataxic and 33/57 (58%) paretic patients’ signs were reported to be confined to the pelvic limbs. Other less commonly observed clinical signs included self-mutilation (10/86, 12%) and fecal and/or urinary incontinence (4/86, 4.7%). Specific clinical signs did not predict a specific lesion or pathologic process (Table 2). Neurologic clinical signs were a contributing factor in the decision to euthanize at least 58/73 clinical cases (79%).
Frequency of clinical signs for spinal disease categories in 86 Panthera spp.
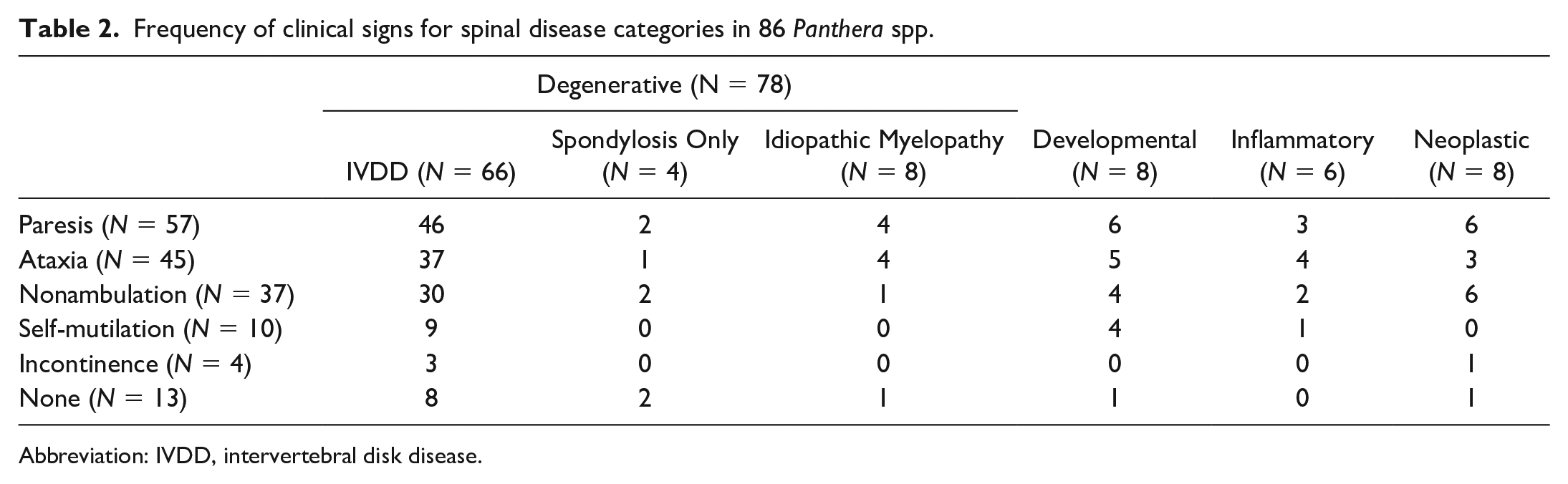
Abbreviation: IVDD, intervertebral disk disease.
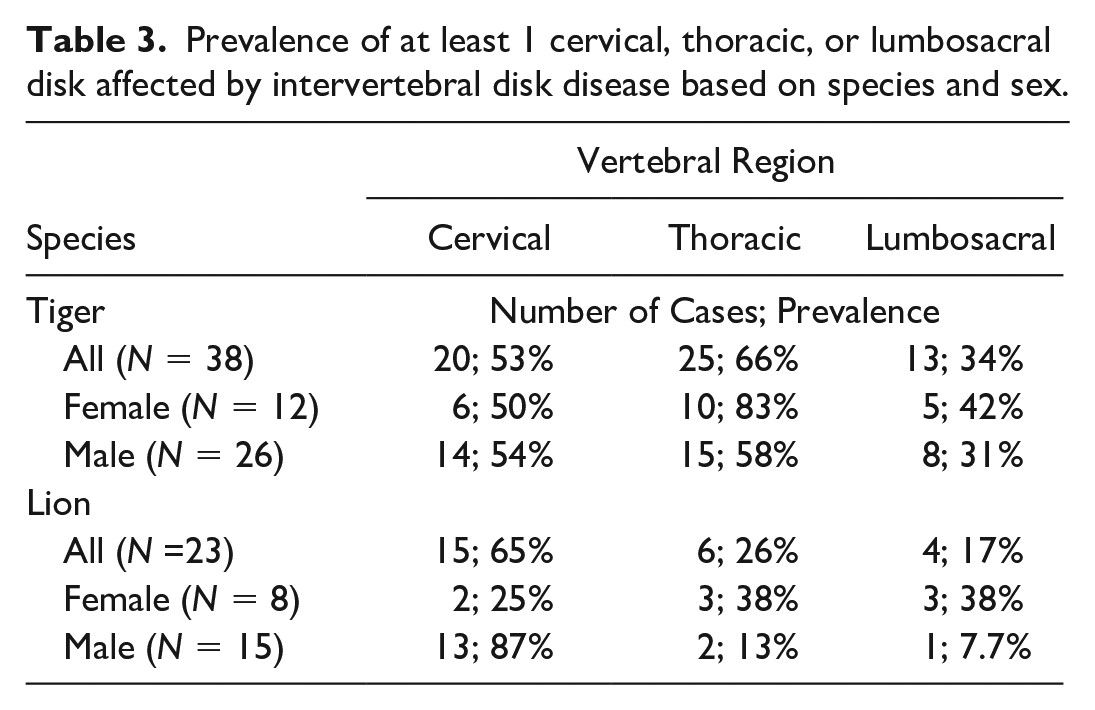
Degenerative lesions included IVDD (Fig. 1a and b; 66/78, 85%), spondylosis without concurrent IVDD (4/78, 5%), and idiopathic (noncompressive) degenerative myelopathy (8/78, 10%). Gross features of IVDD included partial to complete disk loss and/or collapse (60/66, 91%), spondylosis (43/66, 65%), end plate sclerosis (41/66, 62%), and disk herniation (32/66, 48%). Ventral bridging spondylosis (27/66, 41%) was most frequently seen, followed by dorsal bridging (12/66, 18%), ventral nonbridging (10/66, 15%), and dorsal nonbridging (9/66, 14%) spondylosis. There were 10 cases for which at least 1 disk had both dorsal and ventral spondyloses. In cases with grossly evident disk herniation (56 disks), herniation was dorsal (31/56, 55%), ventral (8/56, 14%), or both (18/56, 32%). Grossly, herniated disks were characterized by bulging of the exterior annulus fibrosis or dorsal longitudinal ligament, except in 3 instances where small masses of soft to friable disk material was extruded into the spinal canal. Schmorl’s nodes, indicative of herniation into a vertebral end plate, were described in a few cases. Spinal cord compression by dorsally herniated disk or dorsal spondylosis was seen affecting at least 1 disk in around half of the IVDD cases (30/66, 44%). The aforementioned lesions associated with IVDD were often multifocal (44/66, 67%); the median number of affected disks was 2 (range: 1–12). Overall, IVDD was most commonly found in the cervical (C2–T1) region (41/66, 62%), followed by the thoracic (T1–L1) region (32/66, 48%) and the lumbosacral (L1–S1) region (15/66, 23%) of the spine. Lions (15/23, 65%), especially male lions (13/15, 87%) had the highest prevalence of cervical IVDD, while tigers (25/38, 66%), particularly female tigers (10/12, 83%), were more frequently affected by thoracic IVDD (Table 3). The most frequently affected disk was C6–C7 (28/66, 42%), closely followed by C5–C6 (25/66, 38%) and C4–C5 (19/66, 29%) (Fig. 2). The majority of individuals with IVDD (58/66, 88%), and 2 of the 4 with spondylosis without concurrent IVDD, had related clinical signs.
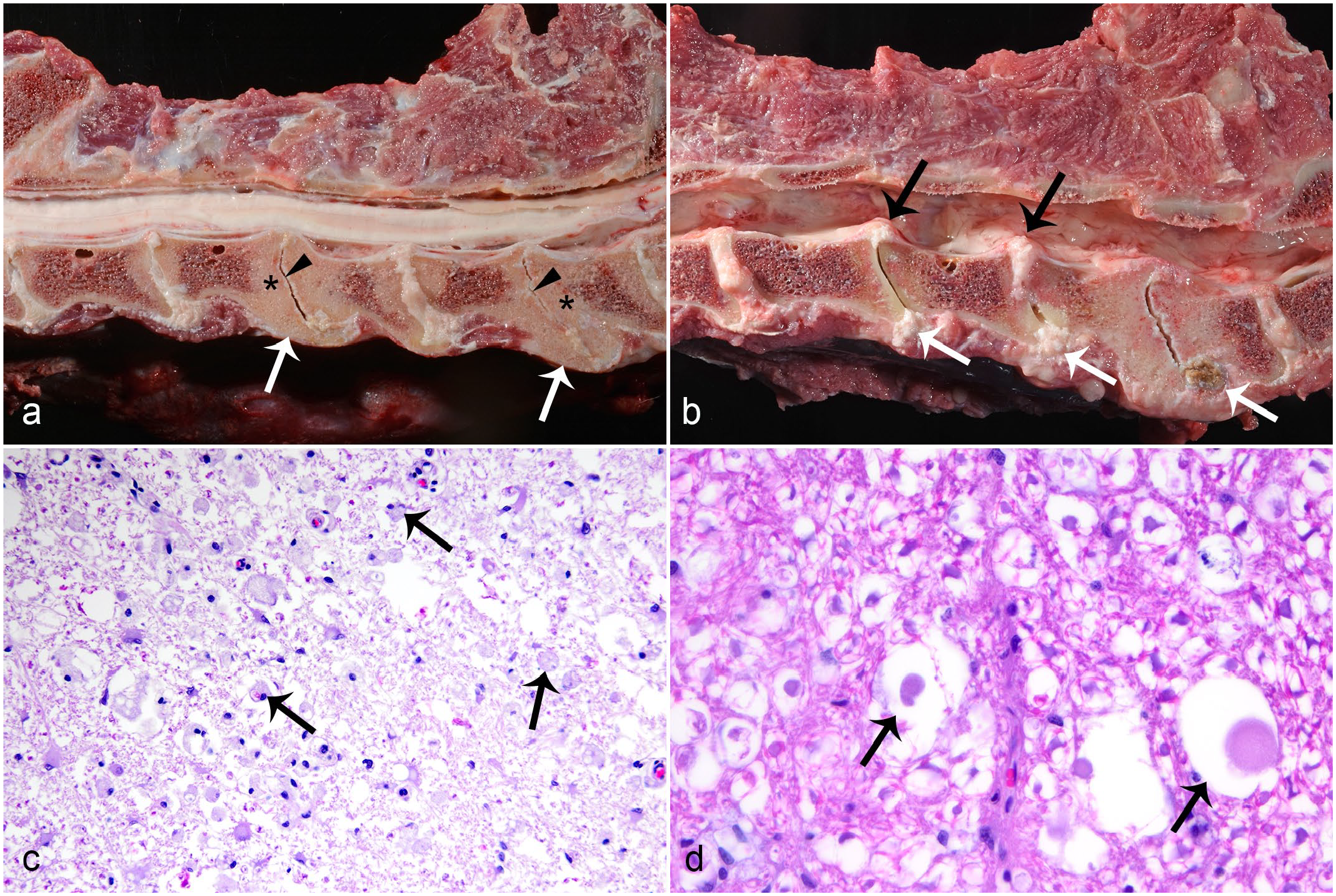
Degenerative spinal disease in Panthera spp. (a) Intervertebral disk disease (IVDD) in an aged male tiger. Multiple foci of ventral bridging spondylosis (arrows), loss of intervertebral disk with collapse of disk space (arrowheads), and vertebral end plate sclerosis (asterisks). (b) IVDD in an adult male tiger. Multiple foci of ventral (white arrows) and dorsal (black arrows) protrusion of intervertebral disk, collapse of disk space, and vertebral end plate sclerosis. (c) IVDD-associated myelomalacia in an aged male tiger. White matter is vacuolated, rarified, and contains gitter cells (arrows). Hematoxylin and eosin (HE). (d) Moderate idiopathic axonal degeneration in an adult male snow leopard. Dilated myelin sheaths contain swollen axons (arrows). HE.
Prevalence of at least 1 cervical, thoracic, or lumbosacral disk affected by intervertebral disk disease based on species and sex.
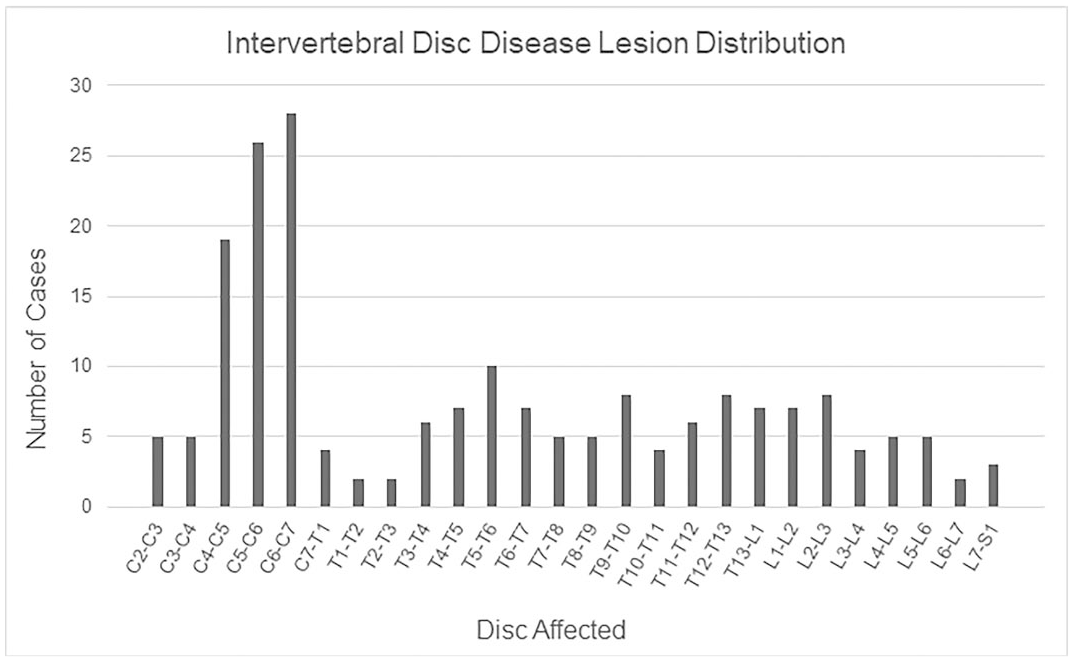
Affected disks in 64 Panthera spp. with intervertebral disk disease (disks affected were not recorded for 2 individuals).
Fifty-two cases of IVDD underwent microscopic examination of the spinal cord: 47/52 (90%) had microscopic lesions, including axonal degeneration (Wallerian) 33/52 (87%) and focal myelomalacia 2/52 (3.8%) (Fig. 1c), and 12/52 (23%) cases had both. Myelomalacia was located at the corresponding spinal segment affected by IVDD in 11/14 (79%) instances, but with grossly evident spinal cord compression described in only 7/14 (50%) cases. Three of these cases with gross compression associated with myelomalacia were those with friable disk material extruded into the spinal canal. Myelomalacia affected all segments of the cord and was either restricted to the white matter (12/14, 86%) or involved the gray and white matter (2/14, 14%). Myelomalacia was within the dorsal or dorsolateral funiculi in most instances (11/14, 79%).
Results of a logistic model examining the likelihood of a diagnosis of IVDD at postmortem examination were statistically significant, and geriatric status, sex, and species group significantly contributed to the model (Table 4). The model explained between 11.6% (Cox & Snell R2) and 17.8% (Nagelkerke R2) of the variance in the dependent variable and correctly classified 79.2% of cases.
Logistic regression predicting the likelihood of intervertebral disk disease (IVDD) diagnosis at postmortem examination in 303 Panthera spp. from a captive population, including 66 with IVDD and 237 without.
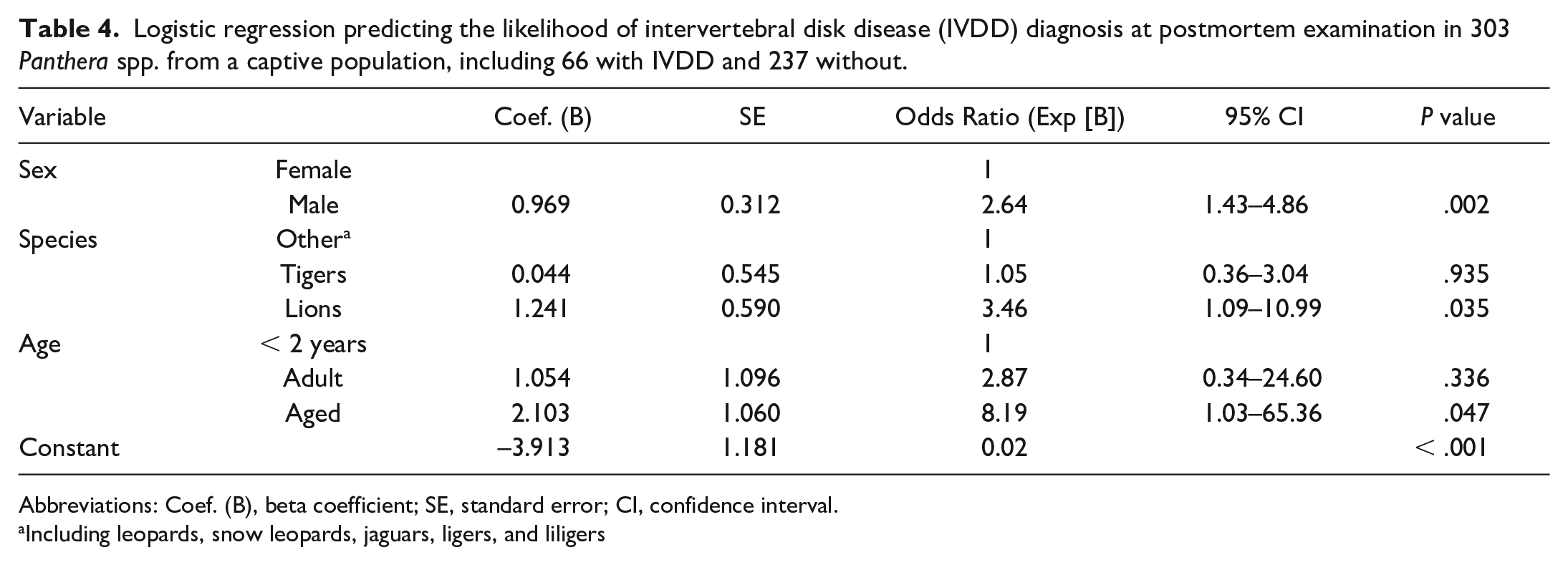
Abbreviations: Coef. (B), beta coefficient; SE, standard error; CI, confidence interval.
Including leopards, snow leopards, jaguars, ligers, and liligers
The study population included 8 cases of idiopathic (noncompressive) degenerative myelopathies that were not associated with IVDD or spondylosis, including axonal degeneration (6/8) and focal myelomalacia (2/8). Myelomalacia was diagnosed in 1 adult (10-year old) male and 1 aged (estimated 22-year old) female tiger. In both cases, the malacic area affected the lumbar spinal cord; 1 confined to the dorsal funiculi and the other involving gray and white matter. Both tigers had neurological signs that were attributed to the myelomalacia, but both had other concurrent disease, including metastatic biliary carcinoma in one and clostridial enteritis in the other. Axonal degeneration (Fig. 1d) was diagnosed in 3 tigers, 2 lions, and 1 snow leopard, 14 5 of which were aged (14- to 18-year old), and 1 that was an adult (2-year old). Five of the affected animals had associated neurological signs that were otherwise unexplained. Degenerate axons were randomly distributed throughout the white matter tracts lacking apparent spinal segment predilection or associated brain lesions (brain was histologically examined in 5/6). The severity was mild (affecting occasional axons; 3/6) or moderate (affecting several axons; 3/6), and ataxia or paresis that were otherwise unexplained by postmortem findings were present in most cases (5/6).
Developmental spinal lesions affected 8/86 felids (9%), 4 of which had more than 1 spinal malformation present, comprising either vertebral (4/8) or spinal cord (3/8) malformations, or both (1/8). There were 2 cases of vertebral dysplasia, 14 both of which had severe IVDD in associated disks, ankylosis, and stenosis of the associated spinal canal (Fig. 3a). One also had consequent kyphosis and syringohydromyelia proximal to the stenosis. The other vertebral malformations included 1 case of an anomalous protuberance from the craniodorsal body of C2 with spinal cord compression, 1 case of mild lumbar scoliosis, and 1 case of focal sacral vertebral stenosis. Spinal cord malformations (n = 5) included syringohydromyelia (2/5; Fig. 3b), 14 syringomyelia (1/5), 14 hydromyelia (1/5), and heterotopic gray matter in the dorsal funiculi (1/5). All instances of syringomyelia were associated with potentially compressive vertebral lesions in the caudo-adjacent spine, including 1 instance of vertebral dysplasia and 3 instances of IVDD.
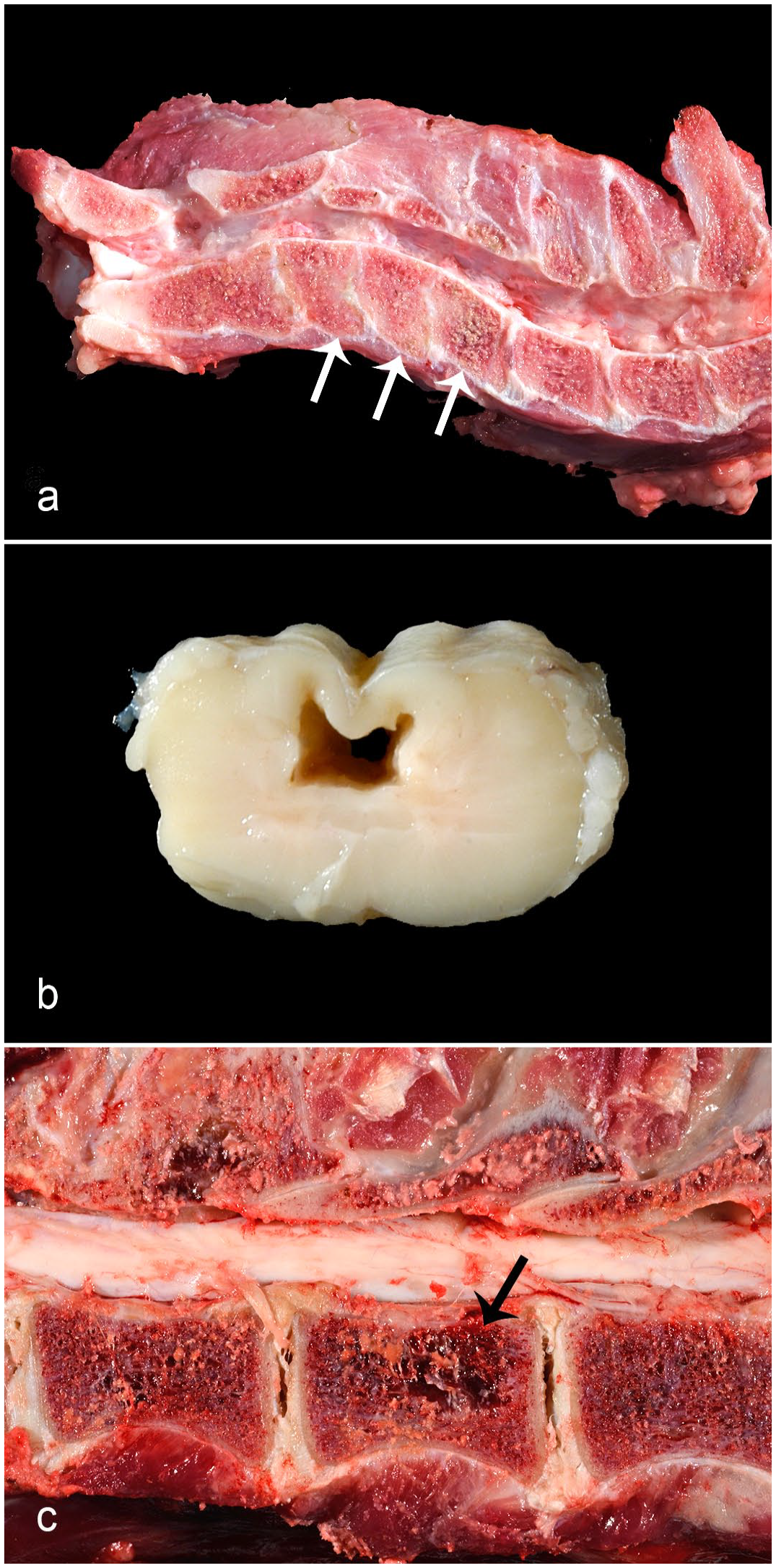
Spinal disease in Panthera spp. (a) C3–C5 vertebral dysplasia (abnormally shaped vertebrae, arrows) in a juvenile female snow leopard. Note associated narrowing and irregularity of intervertebral disks with absence of normal nucleus pulposus. (b) Syringohydromyelia in an aged male lion. (c) Vertebral multiple myeloma in an aged female tiger. An osteolytic mass is within the vertebral body (arrow).
Inflammatory spinal lesions affected 6 tigers. Two cases had known causes. The first case was a neonate with suppurative meningoencephalomyelitis with gram-negative rods attributed to Escherichia coli septicemia, and the second case was an adult with pyogranulomatous meningitis and encephalitis with intralesional Blastomyces gilchristii. 38 The remaining 4 tigers had idiopathic lymphoplasmacytic meningitis or pachymeningitis, 1 of which had associated encephalitis.
There were 8 cases of neoplasia comprised vertebral multiple myeloma (4/8; Fig. 3c),7,14 vertebral facet synovial myxoma (1/8), 21 spinal lymphoma (reportedly immunoreactive for both CD79a and CD3, 1/8), spinal nerve root neurofibroma (1/8), and spinal astrocytoma (presumptive, 1/8), all of which occurred in tigers except for the neurofibroma, which was in a lion. All neoplastic lesions except for 1 multiple myeloma case resulted in clinical compression of the spinal cord. Three of the 4 cases of multiple myeloma occurred in female tigers, and regions affected by the neoplasm included the lumbar (4/4), thoracic (3/4), and cervical (2/4) spine. 7
Amphophilic globules, with morphological features similar to those described in the cerebral cortex of captive nondomestic felids, 48 were common within the spinal cord gray matter.
Discussion
The goals of this study were to determine the relative frequencies and characteristics of spinal lesions in a population of sanctuary Panthera felids, and to identify signalment risk factors associated with these diseases. Overall, spinal disease is common among captive Panthera spp., and degenerative lesions, particularly IVDD, predominate.
Degenerative lesions comprised the majority of the spinal lesions identified, and IVDD (Hansen type II) was the most common finding. IVDD in nonchondrodystrophic animals (Hansen type II) reflects degeneration of disks with age. 6 Consistent with this and from previous reports in Panthera spp., 22 IVDD occurred most frequently in geriatric individuals and was seen in nearly three fourths of the felids with spinal lesions living beyond 13 years of age. Therefore, the high prevalence of IVDD is likely a reflection of prolonged lifespan in captivity. Panthera spp. living under human care have a much longer life expectancy relative to their free-ranging counterparts.28,45 For example, longevity in zoo-housed tigers is approximately 19 years while a study on wild Amur tigers demonstrated a maximum lifespan of 7 years in females and even less in males. 10 Environment and husbandry have also been implicated in the development of degenerative lesions, particularly the use of concrete or other hard flooring and raised platforms.22,28 The population in the current study were housed in earthen-floored enclosures with access to a low wooden deck.
In agreement with previous reports, IVDD lesions were often accompanied by a history of long-term and insidious neurological signs and were multifocal.8,22,25,27 A smaller study reported the lumbar region of the spine as most commonly affected by IVDD in large felids, 22 whereas the current study identified the cervical region, particularly C4–C7, as being most frequently involved in the disease. A sex or species predilection for IVDD in Panthera spp. has not been previously shown, but this study demonstrated an increased likelihood in males and lions. Males not only tend to weigh more than females but also possess larger physical features, such as skull size.12,44 Similarly, lions’ heads are generally greater in size and heavier compared with that of tigers. For that reason, these animals’ cervical spines likely experience greater mechanical force or mobility over time, which in other species leads to a greater risk of degenerative disk disease. 6
It is possible that this study under-estimated the frequency of sub-clinical IVDD lesions, particularly considering the predilection for the cervical spine which, owing to greater space within the spinal canal, is less prone to compressive spinal cord damage and subsequent neurological signs. 6 Postmortem observation and collection of the spine and spinal cord are not routinely performed on all animals without neurological signs. Moreover, gross identification of IVDD may not be as sensitive as advanced imaging, such as magnetic resonance imaging, radiographs, or computed tomography, especially for mild lesions. 22 The present study did not retrospectively review diagnostic imaging findings, thus the frequency of instances where IVDD was diagnosed on imaging and not detected postmortem is not known.
Spondylosis was commonly associated with IVDD in our study, as previously observed. 22 This is an infrequent finding in domestic species with IVDD and is presumptively a consequence of vertebral instability 22 or disk herniation. 6 Spondylosis also occurred without IVDD (n = 4), yet the pathogenesis in those instances is thought to similarly reflect age-associated degeneration of the annulus fibrosus. 6 In dogs, severe cases of spondylosis rarely represent diffuse idiopathic skeletal hyperostosis, which can occur independently or in combination with spondylosis. 23 It is possible that a similar entity exists and has not been recognized in nondomestic felids.
The majority of IVDD cases had significant associated spinal cord lesions, including axonal (Wallerian) degeneration (87%) or myelomalacia (27%), as well as associated clinical signs (88%). Axonal (Wallerian) degeneration reflects degeneration of an axon distal to a site of injury, 43 and is a well-known consequence of compressive spinal cord lesions, such as IVDD, including in Panthera spp. 22 In contrast, myelomalacia is indicative of more severe spinal cord injury2,34 and has not been previously described as a consequence of IVDD in Panthera spp. Myelomalacia in IVDD is typically associated with disk extrusion (where degenerated nuclear material ruptures through the annulus fibrosus into the spinal canal), and is more frequently seen with Hansen type I IVDD of chondrodystrophic dogs. 4 The disk herniations in nonchondrodystrophic animals typically represent protrusion of the degenerate annulus fibrosus (Hansen type II herniation), such as those that predominated in this study, causing slow compressions less likely to damage the spinal cord or induce severe clinical signs. 6 However, these herniations can be associated with myelomalacia when lesions are severe. 29 The pathogenesis of IVDD-associated myelomalacia is uncertain, but may involve ischemia resulting from spinal cord compression. 16 In this study, myelomalacia in individuals with IVDD was not always present at the site of IVDD, and it was only associated with grossly evident spinal cord compression in half of the cases, indicating alternative pathogeneses in some cases. The high prevalence of IVDD-associated myelomalacia highlights potential for severe sequela of the disease in these species. Because necrosis of the spinal cord is most likely caused by vascular injury, myelomalacia can occur in areas of the cord away from the primary injury site, 34 which may account for the lesions within the dorsal and dorsolateral funiculi associated with ventral compression. Furthermore, if blood flow within the cord was only partially reduced, it is expected that the parenchyma supplied by the most terminal aspect of the vessels would be affected first. 11 The vascular anatomy within the spinal cord of nondomestic felids has not been studied, but there could be an anatomical reason for the sparing of the ventral funiculi in these cases.
Some instances of myelomalacia and axonal degeneration occurred entirely without IVDD and were considered idiopathic. Myelomalacia could potentially be explained by subtle compressive lesions not grossly identified (eg, dynamic lesions, such as vertebral instability) or infarction. Similar lesions in the cerebral cortex of nondomestic felids are considered infarcts and hypothesized to be a consequence of hypertension. 48 Fibrocartilaginous embolic myelopathy has been described in a lion and a tiger, both of which had myelomalacia in the caudal cervical spinal cord associated with acute neurologic deficits that were confined to or worse on one side of the body.1,39 These findings are in contrast to the cases of idiopathic myelomalacia of the current study, which lacked unilaterally worse clinical signs, histologically identified emboli, and a cervical distribution (lesions involved the lumbar region). One case had biliary carcinoma with renal emboli that may also have been the basis for myelomalacia, although it was not microscopically identified.
Idiopathic axonal degeneration in the present study was associated with neurological signs that were otherwise unexplained, suggesting the lesion was clinically significant. It likely reflects an age-associated pathogenesis since those affected were mostly geriatric (5/6). While the distribution of axonal degeneration throughout spinal white matter tracts did not map to a spinal compressive lesion in these instances, subtle multifocal lesions cannot be ruled out. Large feline leukoencephalomyelopathy also features axonal degeneration, but is most commonly seen in cheetahs (Acinonyx jubatus), and is most pronounced in the cerebral white matter, a feature not observed in cases of idiopathic axonal degeneration in the current study.3,13,18 Other potential explanations include hypovitaminosis A, 31 hereditary myelopathy, 5 cobalamin deficiency, 41 or toxins, such as delayed organophosphate toxicity.17,43
Developmental lesions were primarily in adult and aged animals, perhaps signifying that these lesions did not clinically impact early life or that some were acquired lesions (syringohydromyelia). While developmental lesions of the spine are relatively uncommon in Panthera spp., there have been previous reports of syringomyelia in 2 unrelated pairs of Asiatic lion cub siblings,20,35 hydromyelia in 2 lions with partial cerebellar herniation, 47 and syringohydromyelia in a lion and a bobcat (Lynx rufus), both secondary to a Chiari-like malformation. 42 One lion in the current study had cerebellar herniation associated with cervical syringohydromyelia, 14 similar to a previous report. 47 Syringohydromyelia can develop as a consequence of central canal cerebrospinal fluid obstruction, 40 and the other cases in this study were associated with spinal lesions that could have potentially compressed the spinal cord, including instances of IVDD and stenosis associated with vertebral dysplasia.
Inflammatory lesions were uncommon in this study, in contrast to the brain where 20% of lesions of captive nondomestic felids consisted of meningitis or meningoencephalitis. 48 This discrepancy is probably biased by pathologist preference for not undertaking examination of the spinal cord.
While neoplasia is common in nondomestic felids,21,33,36,37 there have been few reports of neoplasms affecting the spine in Panthera spp., including a chordoma of the thoracic vertebrae in a tiger 24 and several reports of multiple myeloma in lions, tigers, and snow leopards.7,19,26,46 In the current study, multiple myeloma was the most prevalent neoplasm observed. Multiple myeloma was found in multiple vertebrae throughout the spine, and all cases involved the lumbar vertebrae. 7 Lymphoma is the most common spinal tumor in domestic cats (Felis catus) and commonly occurs as part of a multicentric process,30,32 but it appears to be uncommon in Panthera spines despite lymphoma being the third and fourth most common neoplasm overall in lions and tigers, respectively, according to one study. 21 Similar to many reports in domestic cats, the case of spinal lymphoma in the current study was extradural and involved the thoracic and lumbar spinal segments. 30 Meningioma is the most common neoplasm involving the brain of nondomestic felids, 48 but it apparently less commonly involves the spinal cord.
In summary, spinal lesions are common among Panthera spp., especially IVDD, which is often associated with clinical signs and typically involves the cervical or thoracic spine. IVDD risk is significantly increased in males, lions, and older nondomestic felids, but it can occur in females, at any age, and in other Panthera species. While less common, other disease processes, such as multiple myeloma, syringohydromyelia, vertebral dysplasia, and noncompressive idiopathic degenerative lesions should be considered in a large nondomestic felid with a neurologic presentation. Clinical signs do not appear to be specific to pathologic processes, but signalment factors, relative disease frequencies, and pathologic features identified in this study may be used to inform probable diagnoses in Panthera patients.
Footnotes
Acknowledgements
The authors thank Ms. Mary Lynn Haven and Ms. Debbie Chaffins at Tiger Haven, as well as the faculty, residents, interns, technicians, and laboratory technicians of the anatomic pathology and zoological medicine services at the University of Tennessee. They also thank Linden Craig for constructive review of an earlier version of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the University of Tennessee, College of Veterinary Medicine Center of Excellence Summer Research Program.
