Abstract
Posterior paresis/paralysis in farmed mink is responsible for significant morbidity and mortality, with individual farms reporting the loss of as many as 700 animals each year. Although this disease has been recognized by North American mink farmers for approximately 40 years, there are few published reports focusing on this entity. The objective of this study was to investigate the etiology and pathogenesis of the disease. Complete necropsy examinations were done on 40 clinically affected mink, ranging from 7 to 10 weeks of age, and on three normal animals in the same age range from two mink farms. Thirty-two of the 40 clinically affected animals had an isolated vertebral lesion characterized by bone lysis and proliferation that usually was centered on an intervertebral disk space in the midthoracic area. An inflammatory reaction, composed primarily of neutrophils, was present within the vertebral sections in 25 of the 40 affected animals (62.5%), and the presence of gram-positive cocci was confirmed in 8 of 10 animals (80%) in which bacterial organisms were observed histologically. Bacterial cultures from 15 affected animals yielded Streptococcus sp. from the intervertebral disk space in 13 of 15 (86.7%) animals and from heart blood in 6 of 8 (75%). A farm visit revealed no history or evidence of traumatic wounds as a source of infection in these animals, and the diet appeared to be adequate for skeletal development. We conclude that posterior paresis/paralysis in farmed mink is associated with bacterial diskospondylitis, likely occurring secondary to bacteremia/septicemia.
Keywords
A clinical problem characterized by posterior paresis/paralysis of unknown etiology has been reported to occur in growing North American farmed mink since the 1960s. 16 This condition appears to be widely recognized in the mink industry and affects as many as 700 mink per year on some farms (D. Armstrong, personal communication). On one large mink farm (average = 50,000 mink; range = 45,000–60,000 mink), in which yearly records of the disease have been documented for the past 15 years, the cumulative yearly incidence has ranged from 0.17 to 2.0% (mink farmer, personal communication). Although a general description of the disease was published nearly 40 years ago, 17 the scientific literature contained no information on the cause or pathogenesis of this entity before a recent abstract by our group. 15 The clinical signs associated with this disease include progressive posterior paresis/paralysis with a subsequent loss of general body condition. Severely affected animals develop urinary incontinence. Because these animals are unable to stand on their hind legs, they are unable to reach their food, which is routinely placed on the top of their wire-mesh cages. Affected animals either die or are euthanatized. Many of these animals have focal dorsoventral (DV) vertebral deviations, particularly in the thoracic region, that are palpable on gross examination. The condition affects both males and females and occurs in multiple color phases of mink.
Posterior paresis/paralysis in mink appear to be underrecognized and is therefore likely to be underreported. One reason for this is that several of the clinical signs associated with this disease can be caused by various etiologies, e.g., urinary incontinence secondary to urolithiasis. In addition, although there is no evidence to date of a familial predisposition, concern regarding the possibility of a genetic link appears to have adversely affected the willingness of mink farmers to report it. Perhaps in part because of these concerns, there are no reliable data regarding the prevalence of this condition in the United States.
In the Midwest region of the United States, clinical signs are first seen in July and August, during the period of rapid growth and in an age range of 7–10 weeks. Animals younger than seven weeks of age are rarely affected. Although the majority of the cases are reported to occur in mink that are 7–14 weeks of age, the disease has been reported to occasionally affect individual animals as old as 24 weeks of age. Farm visits have revealed no history or evidence of traumatic wounds as a source of infection in these animals. In addition, variations in diet composition (particularly manipulations in dietary calcium, phosphorus, and electrolyte levels) over the past 10 years have had no significant influence on the incidence of this disease on the two affected farms from which animals for this study were examined (D. Armstrong, personal communication). Previous diagnoses on this condition from other veterinary diagnostic laboratories, as well as our initial assessments, included fractures secondary to a presumed reduction in vertebral cortical bone (osteopenia) or vertebral osteomyelitis, or both. The purpose of this investigation was to carefully evaluate this condition in farmed mink to determine its etiology and pathogenesis.
Materials and Methods
Animals
All mink for this study were collected from two mink farms, both with a relatively high incidence of disease, over a period of four consecutive years (2000–2003). The mink on both these farms are fed (up to four times daily) a mixture of moistened manufactured dry cereal and meat by-products such as fish, poultry, eggs, cheese, and crackers on a wire mesh on top of their cages. On one farm, the mink are annually vaccinated subcutaneously (SQ) with a three-way vaccine including Pseudomonas, botulism (Clostridium botulinum), and mink enteritis virus (parvovirus) at 6–7 weeks of age. An aerosolized distemper virus vaccine is administered in the nest boxes at 10 weeks of age. The second farm vaccinates the mink SQ with a four-way vaccine including Pseudomonas, botulism (Clostridium botulinum), mink enteritis virus (parvovirus), and distemper at 10–11 weeks of age. The animals on both farms are housed outside in multirow sheds that are approximately 250 ft long and contain an average of 100–150 cages per row. The cages are 12 in. wide, 15 in. deep, and 24–36 in. long, and each pen contains a wooden nest box bedded with sawdust. Typical litter sizes on these two farms range from five to eight kits. The mink kits are weaned at 37–39 days of age, and the animals are housed in groups of two to three per cage from the age of 8–10 weeks until pelting (at approximately 28 weeks of age). Both farms use antibiotics as needed to treat conditions such as mastitis, diarrhea, and pneumonia, with the antibiotic usage being widely variable from year to year.
The animals that were necropsied included a total of 43 mink, all of which were between 7 and 10 weeks of age, from the two affected mink farms. The gender was not recorded for the majority of these mink; however, one group included nine male and two female mink. Forty of these animals had clinical signs of variable severity, from slight gait abnormalities to complete hind limb paralysis. The remaining three mink were age-matched, clinically normal mink that were examined for comparison. All mink were euthanatized on the mink farms using a carbon monoxide chamber and were delivered the same day to the Minnesota Veterinary Diagnostic Laboratory on ice for necropsy examination.
Radiology
DV and lateral radiographs of the spine were taken from all 43 animals (after removal of the viscera) using a cabinet radiography unit (Faxitron series, Hewlett Packard, McMinnville, OR) and high detail film (X-Omat TL, Eastman Kodak Company, Rochester, NY). The radiographic technique included the following: exposures of 30–55 kvp; mAs = 3 mA continuous for 30–35 seconds; and film to source distance = 61 cm.
Necropsy and histology
Complete diagnostic necropsies were done on all 43 animals. All internal tissues were examined grossly, and any abnormalities were noted. To investigate the possibility that bite wounds may have been the source of the bacterial infection, the skin from 30 of the mink was carefully removed and the subcutis was examined for evidence of trauma. In cases in which a radiographic lesion was present, longitudinal midsagittal sections from each area of the affected vertebrae and intervertebral disk from each animal were fixed in 10% neutral-buffered formalin, decalcified in 10% ethylene diamine tetraacetic acid, and processed for histology. For animals with no gross or radiographic vertebral lesions, sections taken from midthoracic vertebrae (T6–T7) were examined. Routine paraffin-embedded histologic sections were cut at 5 μm and stained with hematoxylin and eosin (HE), and selected histologic sections were stained with Gram's stain.
Bacteriology
At necropsy, routine bacterial cultures of lung, liver, intestine, and mesenteric lymph node were done on 10 of 40 of the affected animals and routine bacterial cultures of the vertebral lesion (identified radiographically) were done on 7 of 40 of the affected animals. For an additional 8 of 40 affected animals, a more rigorous bacterial sampling and culture procedure was used. Heart blood was collected aseptically on the farm from each animal immediately postmortem into tryptic soy broth and agar, Cary Blair anaerobic media, and Campylobacter transport media in screw-top tubes. The carcasses were transported on ice to the Minnesota Veterinary Diagnostic Laboratory that same day and, within 5 hours of death, swabs for bacterial culture were obtained from the oral cavity, urinary bladder, and affected intervertebral site at necropsy (either from the site of the radiographic lesion or the site of grossly localized vertebral inflammation, or both) from each animal into the same aerobic and anaerobic transport media. In cases in which there was no radiographic lesion and no gross evidence of localized vertebral inflammation, swabs for bacterial culture were taken from the midthoracic spine (T6–T7). Routine aerobic (including Salmonella sp.) and anaerobic cultures were done on all collected samples in addition to specific/selective cultures for Campylobacter and Brucella sp.
Results
Radiology
Radiographically, 32 of 40 affected animals had radiographic lesions that were localized to the thoracic spine (T2–T14), most commonly in the midthoracic area. These lesions were characterized by focal loss of a definable intervertebral disk space with collapse of the immediately adjacent vertebral bodies, accompanied by moderate lytic and proliferative bony changes. Similar bony changes were also present in the adjacent rib heads (Fig. 1). The majority of these mink (28 of 32) had a single lesion that appeared to center on an intervertebral disk space and involved only the two adjacent vertebral bodies. The remaining four mink with radiographic vertebral lesions had evidence of multifocal bony changes centered on multiple, usually consecutive, intervertebral disks. No radiographic lesions were observed in the remaining eight affected animals or in the control animals.
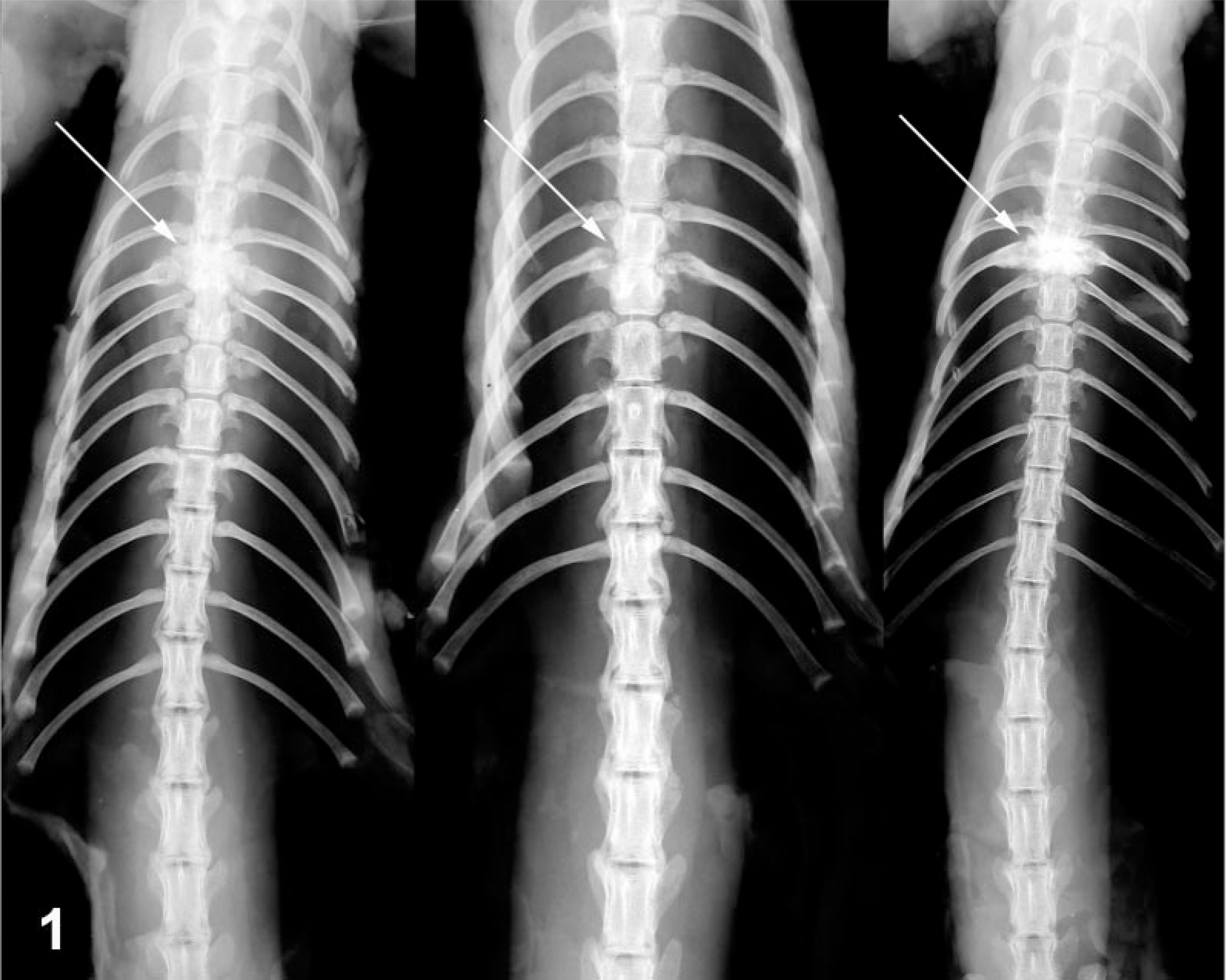
DV radiograph (with the viscera removed). Vertebral column; 8-week-old mink with posterior paralysis. A single lesion is present in the thoracic spine and is centered on the intervertebral disk space, involving the two adjacent vertebral bodies in all three animals. The affected vertebral bodies are variably decreased in height and have an increased radiodensity and evidence of bony proliferation. Intervertebral disk space has collapsed (arrows), and there are lytic and proliferative changes in the adjacent rib heads.
Necropsy and histology
Grossly, all affected mink that had radiographic lesions (32 of 40) also had grossly identifiable enlargement of one or more thoracic vertebral sites. Ten of these 32 animals also had variable degrees of focal DV deviation of the spine at the level of the midthorax and four of 32 had pale tan exudate at the site of the thoracic vertebral enlargement. One affected animal that did not have a radiographic lesion had exudate at the atlanto-occipital junction. The remaining 7 of 40 affected animals that had no radiographic lesions had no grossly evident lesions. Twelve of the 40 affected animals, which had radiographically or grossly visible vertebral lesions, or both, had wounds resulting in the absence of the extreme distal end of the tail, or loss of the distal hind limb extremities, or both. These were attributed to bite wounds or self-mutilation secondary to loss of sensation to the extremities. No other significant gross lesions were observed in the musculo-skeletal system of any of the animals, including the three control animals, and no significant macroscopic lesions were identified in any nonmusculoskeletal organs. No cutaneous or subcutaneous lesions were identified in any of the animals from which the skin was removed.
Histologically, there was focal collapse and lysis of the intervertebral disk with collapse and bony proliferation involving the adjacent vertebral bodies (Fig. 2). On higher magnification, areas of fibrosis, deposits of fibrin, remnants of intervertebral disk material, and marked bony remodeling with increased numbers of osteoclasts and osteoblasts were evident. In the majority of the vertebral sections (25 of 40 mink, 62.5%), there was evidence of subacute, suppurative inflammation characterized by degenerate neutrophils, fibrin deposits, cellular debris, fibrous connective tissue, and a variable presence and number of gram-positive bacterial cocci in eight of 10 mink (80%) in which Gram's stains were done (Fig. 3). This inflammation often involved the vertebral endplates (osteitis/spondylitis), the marrow cavity (osteomyelitis), the intervertebral disks (diskitis), and the spinal cord (myelitis). The thoracic spinal cord immediately adjacent to the affected disk and vertebrae often contained histological evidence of myelomalacia of the white and gray matter, characterized by swollen, empty axonal sheaths, swollen axons, and occasional aggregates of gitter cells. Fragments of mineralized bone surrounded by moderate numbers of neutrophils and macrophages were present within the meninges and subjacent white matter of the spinal cord. Interestingly, in one case, bacterial colonies were present within a morphologically normal intervertebral disk that was one vertebral body distant from a lesion site (Fig. 4).
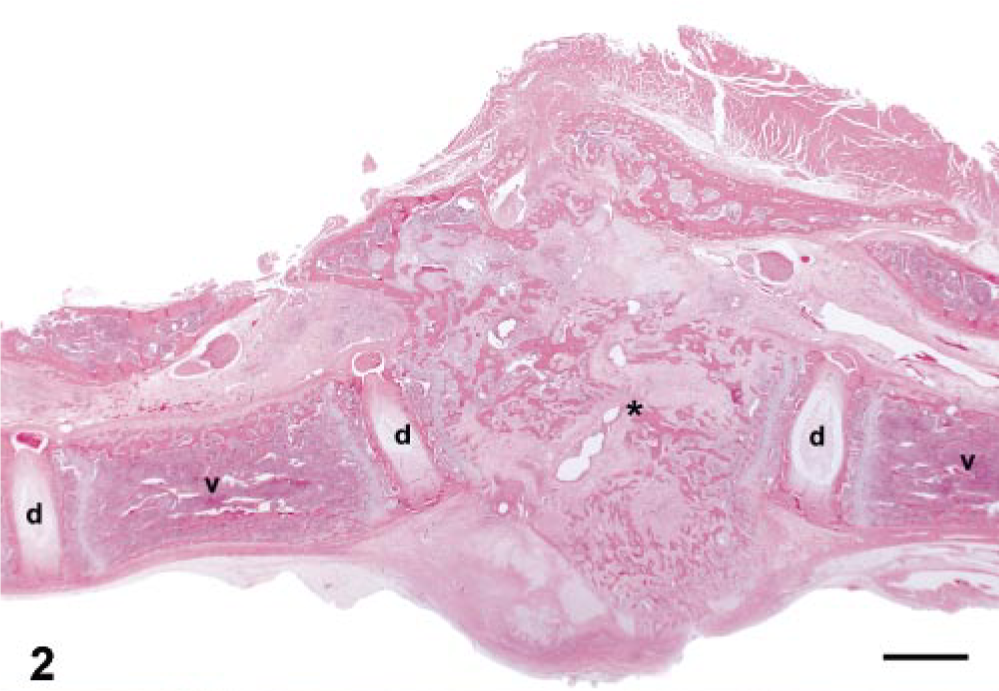
Midsagittal section of thoracic vertebra; 8-week-old mink with posterior paralysis. Vertebral bodies (v), intervertebral disk (d), and focally extensive vertebral lesion characterized by disruption of bony trabeculae of the vertebral body with loss of normal architecture and locally extensive fibrosis (∗). At higher magnification, this lesion includes fibrin, fibrous connective tissue, remnants of intervertebral disk material, and bony remodeling of the adjacent vertebral bodies on either side of the lesion. HE. Bar = 2 mm.
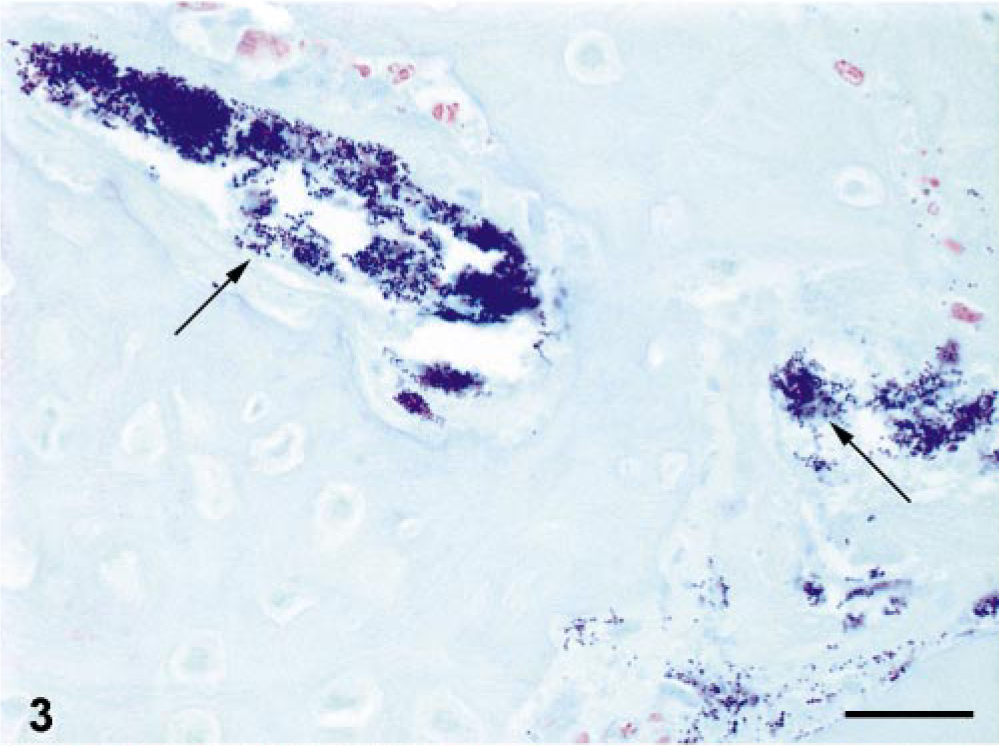
Cross-section of thoracic vertebra; 8- to 9-week-old mink with posterior paralysis. Numerous gram-positive bacterial cocci (arrows) are present in the section. Gram's stain. Bar = 25 μm.
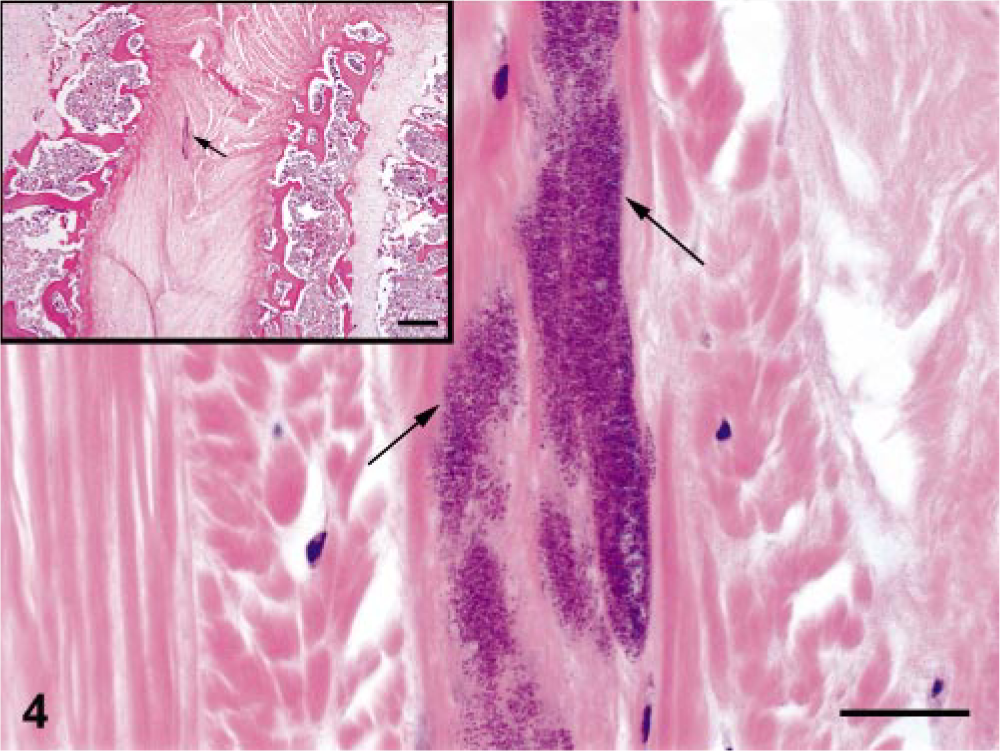
Midsagittal section of thoracic intervertebral disk; 8-week-old mink with posterior paralysis. The disk is located one disk space away from a radiographic lesion, similar to those demonstrated in Fig. 2. Clusters of bacterial cocci (arrows) are present in this otherwise normal disk. HE. Bar = 25 μm. Inset: lower power magnification demonstrates location of bacterial cocci (arrow) within intervertebral disk. HE. Bar = 250 μm.
Bacteriology
Routine bacterial cultures of soft tissues yielded a mixed, scant growth of bacterial organisms including coliforms, Staphylococcus sp., Streptococcus sp., Proteus sp., and Salmonella sp. in various combinations from the lung, liver, mesenteric lymph node, or intestine, or all, from seven of 10 of the affected animals. There was no significant bacterial growth from cultures of lung, liver, and kidney of three additional clinically affected mink. Of the seven mink with clinical signs from which routine bacterial cultures were obtained from the intervertebral disk lesion site, cultures yielded four mink with Streptococcus sp., one mink with Staphylococcus intermedius, one with scant growth of alpha-Streptococcus sp. (not group D), and one with no growth.
In the eight mink that had a more rigorous bacterial culture regimen, including intervertebral disk cultures and heart blood, eight of eight mink yielded beta-hemolytic Streptococcus sp. from the intervertebral disk cultures and six of eight yielded beta-hemolytic Streptococcus sp. from heart blood cultures (Table 1). Antibiotic sensitivity/susceptibility testing revealed that the organism was susceptible to many of the tested antibiotics, including amoxicillin/clavulanic acid, amoxicillin, penicillin, and trimethoprim/sulphamethoxazole. Bacteriology results from the oral cavity and urinary bladder of these mink were mixed and considered to be noncontributory.
Anatomic location of vertebral lesions, bacteriology, and Gram's stain results; 10-week-old mink.∗
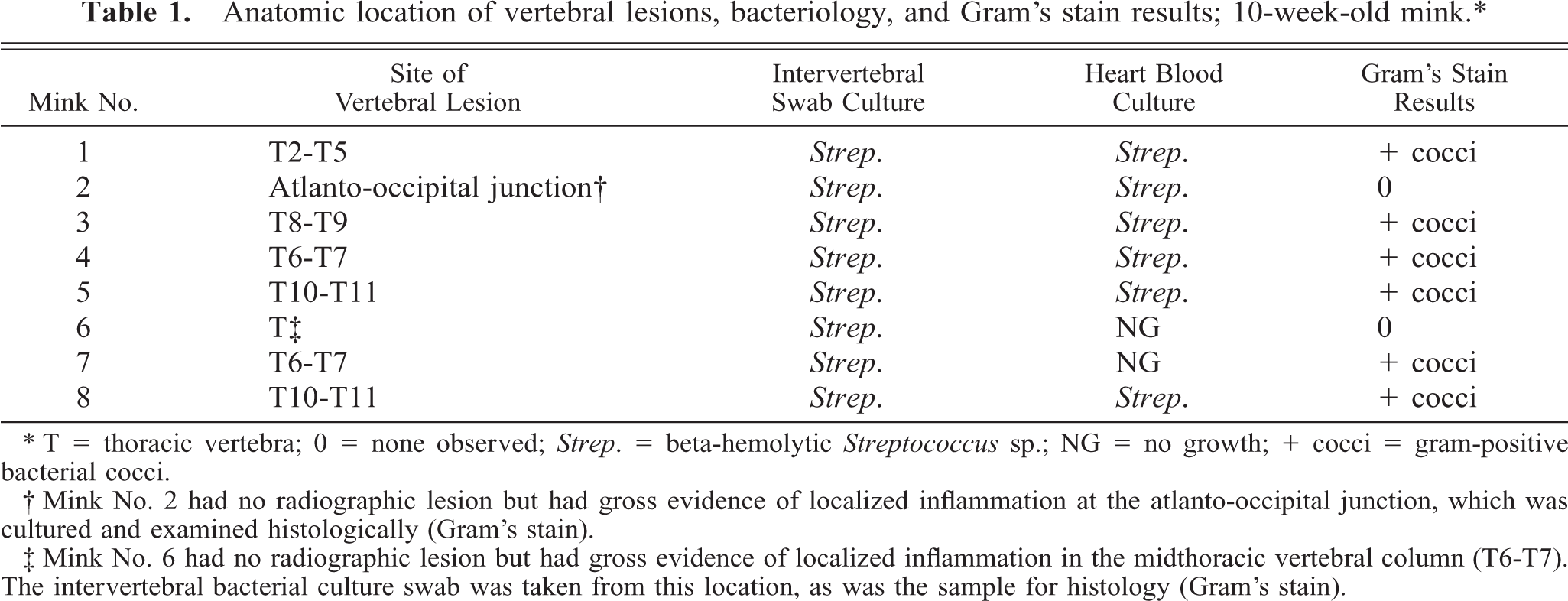
∗ T = thoracic vertebra; 0 = none observed; Strep. = beta-hemolytic Streptococcus sp.; NG = no growth; + cocci = gram-positive bacterial cocci.
† Mink No. 2 had no radiographic lesion but had gross evidence of localized inflammation at the atlanto-occipital junction, which was cultured and examined histologically (Gram's stain).
‡ Mink No. 6 had no radiographic lesion but had gross evidence of localized inflammation in the midthoracic vertebral column (T6-T7). The intervertebral bacterial culture swab was taken from this location, as was the sample for histology (Gram's stain).
Discussion
The results of this study provide strong evidence that posterior paresis/paralysis in North American farmed mink occurs secondary to bacterial diskospondylitis. Bacteriology results revealed beta-hemolytic Streptococcus sp. as the predominant bacterial organism cultured from the intervertebral sites and heart blood of clinically affected mink. To our knowledge, this is the first report in the scientific literature to suggest a pathophysiologic mechanism and potential etiologic agent for this disease.
In veterinary medicine, diskospondylitis primarily affects large, middle-aged dogs 11, 22, 23 but has also been reported in pigs, 5 horses, 1, 8 and rarely in cats. 13, 14, 22 The most common route of spinal infection is by arterial spread of bacterial organisms from other infected sites of the body. 22
On the mink farms that we have investigated to date, the disease is most commonly reported in animals in the 7- to 14-week age range. The reason for this narrow window of time during which animals are clinically affected is unknown; however, the onset of disease may be associated with a decline in maternal antibodies. In addition, the lesions occurring in this disease are nearly always located within the thoracic spine, very rarely affecting the cervical or lumbar vertebrae. We hypothesize that the highly specific thoracic vertebral predilection site and, possibly, the narrow age window during which the disease usually occurs are related to the vascular supply to the vertebral column. On the basis of data from the human literature, we suspect the intervertebral disk in very young mink has a blood supply and, thus, a portal of entry for bacteria but that this disappears as the animals mature. 2, 4, 18, 19, 24 Because of the specific thoracic vertebral predilection site, we also suspect that the blood supply to the thoracic vertebrae differs from that of the cervical and lumbar vertebrae, allowing the deposition of bacteria in thoracic sites at a vulnerable time during development. Because of the presence of numerous secondary changes observed histologically at the lesion sites, it is likely that the lesions observed in this study had first developed at least 2 weeks before necropsy.
This disease in mink appears to be very similar clinically and anatomically to juvenile diskitis (spondylodiscitis) seen in human medicine. As opposed to diskitis in adult humans, which usually occurs after spinal surgery, spontaneous diskitis typically affects children and often occurs without an identifiable initiating cause. 2, 4, 7, 25 The explanation for these differences in adult versus juvenile humans is thought to be at least in part because of age differences in the vascular supply to the intervertebral disk. 2, 4, 18, 19, 24
In the human literature, there are discrepancies regarding the blood supply of the intervertebral disk. Some authors suggest that the intervertebral disk is avascular at all ages and may be equally susceptible to hematogenous infection at any age, 18 but others claim that there is a good vascular supply to fetal and neonatal disks that is not present in older humans. 10, 20 Coventry et al. 3 found that vascular channels in the cartilaginous endplate were only normally present in the first three decades of life, and a microangiography study by Hassler 10 demonstrated that the intervertebral disks of stillborn babies and children had a much more ample vascular supply than those of adults.
There currently is a lack of published information regarding the correlation of histologic, bacteriologic, and radiopathologic results in human spondylodiscitis, and the existing literature contains many discrepancies. 12 Although some authors propose an infectious cause to spondylodiscitis, conclusive data in humans are not available. Some investigators claim that spondylodiscitis is inflammatory (noninfectious) in nature or caused by trauma. 9 Evidence against an infectious etiology includes the fact that many cultures are sterile and some patients recover without antimicrobial therapy. 6 In contrast, some authors observed that patients who were treated with intravenous antibiotics for at least 6 days had a more rapid resolution of symptoms and also had the lowest likelihood of developing recurrent symptoms. 21 In human spondylodiscitis, the diagnostic yield of detecting the etiologic agent in tissue sections by histopathology is relatively low. Lucio et al. 12 reported a percentage of 14.8%, even with special histochemical stains. The yield of culturing an etiologic agent has been reported to range from 50 to 80%. 12 Some of these problems most likely are attributed to the fact that the condition is chronic at the time of diagnosis and obtaining an appropriate culture sample requires an invasive and somewhat blinded procedure (versus collecting a postmortem sample with direct observation of the site of interest, as was done in our studies). It is also unclear from the human literature precisely where the infection is initiated (intervertebral disk versus subchondral bone or hyaline cartilage of the vertebral endplate). 9
The source and route of infection in diskospondylitis in mink are currently unknown. Bite wounds from penmates are one possibility; however, we were unable to document the presence of cutaneous lesions at necropsy. The severe wounds involving the hind limbs and tail that often were present in these animals are reported by mink farmers to occur after the animals develop posterior paresis/paralysis and appear to be the result of self-trauma or trauma from penmates secondary to anesthesia due to spinal cord damage. Another possibility that we have not yet investigated is perinatal infection, acquired from the dam during whelping. Diskospondylitis may be accompanied or preceded by other sequelae of bacteremia, such as endocarditis. A recent report from Denmark documented verrucous endocarditis associated with Streptococcus bovis in mink; however, the mink in the cases reported in this study had no gross or histologic evidence of endocarditis. 17 Because the number of affected animals on a particular farm varies rather widely from year to year, it is conceivable that certain stressors (age- or environment related) may serve as triggers of this disease. Husbandry practices also may play a role; however, despite minor differences in animal husbandry practices (e.g., vaccination schedule), the overall incidence and severity of morbidity and mortality due to posterior paresis/paralysis were similar on the two mink farms involved in this study.
Although it is our understanding that posterior paresis/paralysis occurs throughout the United States and, possibly, in other countries as well, this study was limited to data collected from two mink farms in the Midwest United States. It is unclear whether bacterial diskospondylitis is important in the pathogenesis of this condition in other geographic locations but it certainly is possible. In addition, bacteriologic surveys will be needed to determine whether or not beta-hemolytic Streptococcus sp. is an important etiologic agent on other mink farms or whether other bacterial organisms may also be involved in this disease. In this study, the majority of bacterial cultures of intervertebral swabs from affected mink (12 of 15) yielded beta-hemolytic Streptococcus sp.; however, alpha-Streptococcus sp. and Staphylococcus intermedius were isolated from two different affected mink. It also is possible that the bacterial organism may be less important in the pathogenesis of this disease than other predisposing factors such as immune status or housing density.
On the basis of these necropsy and bacteriology results, farm personnel from one affected mink farm identified and separated out approximately 50 variably paretic or paralyzed mink. One half of these mink were treated with 50 mg/mink of injectable lincomycin once a day (Durvet Inc., Blue Springs, MO) SQ and the other half of this trial group was treated with 6.25 mg/mink of injectable enrofloxacin once a day (Bayer Health Care-Animal Health Division, Shawnee Mission, KS) SQ for three consecutive days. The majority of the treated mink in both groups that were partially mobile (slightly to moderately paretic or paralyzed) showed marked clinical improvement within 24 hours after treatment and most recovered completely. The apparent dramatic response of these animals to antibiotic therapy suggests that this method of treatment may be effective in preventing or treating this disease. Further supporting this hypothesis is the fact that we have identified individual mink farms that appear to have little or no incidence of this disease, and these farms tend to use relatively high doses of prophylactic antibiotics for an extended period of time.
The results of these studies to date provide strong evidence that posterior paresis/paralysis in farmed mink is caused by bacterial diskospondylitis that probably occurs secondary to bacteremia of unknown origin. We speculate that the relatively narrow age window during which the clinical disease occurs and the highly predictable site of the lesions may be related to vascular supply to the spine, particularly the thoracic spine. A reduction in immune status secondary to a decline in maternal antibodies may play a role in the age of onset of the disease. Our findings provide strong reason to believe, for the first time in the 40+ years during which this disease has been recognized, that it will be possible to develop effective methods of treatment and prevention.
Footnotes
Acknowledgements
We would like to thank Dr. Rod Frank for sharing the initial necropsy results with us and for his scientific input into the study. We also thank Dr. Sandy McLachlan, Anne Undersander, Laura Bursch, Jessika Stadden, Joel Pettit, Laurel Deloria, and the Bacteriology and Histotechnique personnel of the Minnesota Veterinary Diagnostic Laboratory for scientific and technical assistance. Finally, we thank Dean Armstrong and Whitie Johnson of the Heger Company and the participating mink farmers (the identities of whom have been omitted because of privacy considerations) for their assistance and generous donation of mink (both affected and normal) for necropsy.
