Abstract
Hepatic lesions in nondomestic felids are poorly characterized. The purpose of this study was to evaluate hepatic lesions in 90 captive, nondomestic felids including tigers, cougars, and lions. Hepatic lesions were histologically characterized as vacuolar change (lipidosis or glycogenosis), biliary cysts, biliary hyperplasia, hepatitis, necrosis, neoplasia, fibrosis, veno-occlusive disease, cholestasis, hematoma, congestion, or hemorrhage. Stepwise logistic regression analyses were performed for vacuolar change, benign biliary lesions, hepatitis, lipogranulomas, extramedullary hematopoiesis, and hepatic stellate cell hypertrophy and hyperplasia, with species as the outcome variable. Ninety cats met the inclusion criteria. Seventy livers (78%) contained 1 or more lesions. Hepatocellular vacuolar change (41/90 [46%]) was the most common lesion overall. Extramedullary hematopoiesis, lipogranulomas, and hepatic stellate cell hyperplasia were also common. One snow leopard had veno-occlusive disease. Tigers were more likely than other felids to have no significant hepatic histologic lesions (odds ratio [OR], 12.687; P = .002), and lions were more likely to have biliary cysts (OR, 5.97; P = .021). Six animals (7%) died of hepatic disease: cholangiocellular carcinoma (n = 2) and 1 each of hepatic lipidosis, hepatocellular necrosis, pyogranulomatous hepatitis, and suppurative cholecystitis. Hepatocellular iron and copper accumulations were present in 72 of 90 (80%) and 10 of 90 (11%) sections, respectively. Sinusoidal fibrosis was common (74/90 [82%]) and primarily centrilobular (65/74 [88%]). Hepatocellular iron, copper, and fibrosis were not significantly associated with hepatic lesions. Primary hepatic disease was not a common cause of death in nondomestic felids in this study.
Previous reports of liver disease in nondomestic felids include veno-occlusive disease in captive cheetahs and snow leopards 3,4,16,17,26 and telangiectasia, amyloidosis, lipomas, and biliary cysts in cheetahs and lions. 2,11,15,29 Gallbladder adenocarcinomas and hepatocellular carcinoma have been reported in lions. 19,20
In domestic animals, iron and copper accumulation have been linked to hepatic disease. Iron and copper are both potent oxidants and can be toxic in high concentrations. As a result of oxidative damage, excess hepatic iron and copper contribute to ongoing hepatocyte loss, inflammation, and fibrosis. 21 Copper accumulation in the liver is recognized as a cause of hepatitis in dogs, and breed predispositions have been described. 8,9,25 More recently, copper accumulation has been described as a cause of hepatopathy in domestic cats. 7,14 A recent study of a large group of domestic cats found no association between histologic abnormalities and the presence or quantity of hepatic iron. 27 Studies of iron and copper accumulation in the liver of snow leopards with veno-occlusive disease have been performed; 17 however, studies looking at hepatic iron and copper accumulation in a large group of nondomestic felids have not been performed.
This study describes postmortem histologic findings in liver tissues of nondomestic felids. In addition, the presence and distribution of hepatic iron, copper, and fibrosis were evaluated and correlated with other histologic abnormalities.
Materials and Methods
Autopsy records from the University of Tennessee Veterinary Medical Center’s pathology service were searched for submissions of nondomestic felids, older than 1 month, from January 1989 through November 2011. All cats in this study had been housed at a municipal zoo; a local large, nondomestic cat sanctuary; or at the home of a private owner prior to their death. Most of the animals were similarly housed in earth-floored enclosures, and all were fed all-meat diets. Cases were included in the study if paraffin-embedded liver tissues were available for histologic examination and if severe hepatic autolysis was absent. The age, body condition score (BCS), and cause of death were recorded. The reason for death or euthanasia was obtained from the medical record, autopsy request form, or autopsy results.
Histologic Evaluation
Sections were obtained from tissue blocks and stained with hematoxylin and eosin (HE), rubeanic acid, Perl’s Prussian blue, and Masson’s trichrome using routine techniques. 12,24 Sections were prospectively evaluated by the primary investigator (J.M.B.) and a board-certified pathologist (K.M.N.), who were blinded to the animals’ medical histories and previous histologic diagnoses. Slides receiving discordant diagnoses were reviewed jointly to achieve consensus. Histologic lesions were described using established criteria based on the HE-stained slides. 23,28 The major hepatic findings were characterized as vacuolar change (lipidosis or glycogenosis), biliary cysts, biliary hyperplasia, hepatitis, necrosis, neoplasia, fibrosis, veno-occlusive disease, cholestasis, hematoma, congestion, or hemorrhage. Glycogen and lipid were differentiated based on histologic appearance only. For lipid accumulation, glycogen accumulation, and inflammation, the degree of change was characterized as mild, moderate, or severe, based on the extent of hepatic involvement. The primary zone of lipid and/or glycogen accumulation and inflammation was also recorded. Inflammatory lesions were further characterized by the predominant cell type. Biliary hyperplasia was defined as portal areas containing more than 2 biliary duct profiles.
Grading of Iron and Copper
Slides stained with Perl’s Prussian blue and rubeanic acid were examined (J.M.B.) to determine the extent of iron and copper accumulation, respectively. Barton’s semiquantitative histologic grading system for iron was modified to reflect the amount of staining in each zone, not just the frequency of staining in zones, as previously described. 27 The amount and distribution of Prussian blue staining was graded separately in the hepatocytes and sinusoids. Staining in the portal areas (connective tissue) was not assessed. A modified copper grading system 27 was used for consistency with the previously described iron grading system.
The amount of staining for each element was graded on a scale of 0 to 3 (0 = no staining; 3 = frequent, dense staining). The distribution of staining was categorized as centrilobular, midzonal, periportal, or random based on the predominant pattern. A designation of random included those specimens where staining was diffuse.
Grading of Fibrosis
Slides stained with Masson’s trichrome were examined (J.M.B.) to determine the extent of sinusoidal fibrosis. Fibrosis was graded on scale of 0 to 3 (0 = none, 1 = focal, 2 = bridging, 3 = bridging with architectural distortion or cirrhosis). 18 The distribution of fibrosis was categorized as described above for hepatic copper and iron. 27
Data Analysis
Descriptive statistics were generated for age, BCS, and iron score by species (cougar, lion, tiger, and other). Samples were analyzed for normality by the Kolmogorov-Smirnov test. Outliers noted on box-and-whisker plots were double-checked to rule out data entry errors. Associations between species and age or BCS were assessed by analysis of variance (age) or the Kruskal-Wallis test (BCS). Associations between species and vacuolar change, benign biliary lesions, hepatitis, lipogranulomas, extramedullary hematopoiesis, and hepatic stellate cell hypertrophy and hyperplasia were assessed by stepwise multinomial logistic regression analysis using species as the outcome variable. The association between species and presence of a liver with no significant findings was evaluated in a separate model because “no significant findings” is exclusive of all other variables. For species represented by fewer than 12 animals, statistical analyses were not performed. Fisher exact tests were performed to assess for association between hepatocellular iron score and the presence of no significant findings, vacuolar change, benign biliary lesions, hepatitis, extramedullary hematopoiesis, and hepatic stellate cell hypertrophy and hyperplasia. A Fisher exact test was performed to assess for an association between sinusoidal iron score and extramedullary hematopoiesis. Stepwise logistic regression analysis was performed to assess for an association between hepatocellular iron score and species using iron score as the outcome variable. No statistical evaluations were performed regarding copper accumulation due to a low number of positive samples.
P values less than .05 were considered significant. For significant associations, odds ratios (ORs) were generated. Statistical analysis was performed using commercially available statistical software packages, including SPSS version 19 (SPSS, Inc, an IBM Company, Chicago, IL; SAS, Cary, NC).
Results
Liver sections were available from 90 nondomestic felids. Species included 30 tigers (Panthera tigris), 17 cougars (Felis concolor), 12 lions (Panthera leo), 8 leopards (Panthera pardus), 6 servals (Felis serval), 5 snow leopards (Panthera uncia), 3 cheetahs (Acinonyx jubatus), 3 lynx (Felis lynx), 2 caracals (Felis caracal), 2 ocelots (Felis pardalis), 1 bobcat (Felis rufus), and 1 clouded leopard (Felis nebulosa) (Suppl. Tables S1–S4). There were 8 spayed females, 41 intact females, 9 altered males, and 32 intact males. Known ages ranged from 9 months to 22 years, with a mean age of 13.5 years. Their BCS ranged from 1 to 5, with a median BCS of 3/5. Age and BCS were normally distributed for all species. There were no significant differences in age or BCS by species. Thirty-one cats (34%) died naturally, and 59 (66%) cats were euthanized. Primary cause of death or euthanasia was categorized as renal (25/90 [28%]); neoplastic (24 [27%]); infectious, including bacterial, fungal, or viral (19 [21%]); neurologic (9 [10%]); degenerative (6 [7%]); inflammatory (5 [6%]); and nonneoplastic hepatic (2 [2%]) disease.
Hepatic lesions, iron scores, copper scores, fibrosis scores, and cause of death or euthanasia are summarized by species in Supplemental Tables S1 to S4. Twenty livers (22%) had no significant histologic findings; the remaining 70 livers (78%) contained 1 or more histologic abnormality. No significant associations were found between species and hepatitis or extramedullary hematopoiesis.
Hepatocellular iron accumulation was predominantly centrilobular (n = 37), although midzonal (n = 14), periportal (n = 11), and random (n = 10) iron accumulation was also present. The median hepatocellular iron score was 1. No significant associations were found between hepatocellular iron score and species or hepatic findings. Sinusoidal iron accumulation within macrophages was common and predominantly random (n = 37), although midzonal (n = 27), centrilobular (n = 22), and periportal (n = 1) sinusoidal iron accumulation was also present. The median sinusoidal iron score was 2. All tigers had iron accumulation within sinusoids. No significant association was found between sinusoidal iron score and extramedullary hematopoiesis.
Hepatocellular copper accumulation was absent in most animals. Copper distribution was random in 8 cases and periportal in 2 cases.
Sinusoidal fibrosis was common. The median score was 2 with a range from 0 to 3. The predominant distribution was centrilobular (n = 65), although midzonal (n = 5), periportal (n = 2), and random (n = 2) distributions occurred. Fibrosis was usually identified in Masson’s trichrome–stained sections and was identified with HE staining in only 1 case (case No. 50, graded as 3 with Masson’s trichrome staining). In the absence of other major findings, a fibrosis score of 1 was considered insignificant.
Tigers
The mean age and median BCS for tigers were 13.5 years and 3. Sixteen of 30 tigers (53%) had hepatic lesions, and diagnoses were lipidosis (8/30 [27%]), portal hepatitis (3 [10%]), metastatic adenocarcinoma (3 [10%]), glycogen accumulation (2 [7%]), and 1 each of lymphoma (lymphosarcoma), hematoma, hepatocellular adenoma, congestion, and biliary hyperplasia. Two tigers had both lipidosis and glycogen accumulation (case Nos. 20 and 30). Of the 8 cases with lipid accumulation, the distribution was diffuse in 3 cases, random in 2, periportal in 2, and centrilobular in 1. No tiger with hepatic lipidosis died as a result of hepatic disease. Portal hepatitis was observed in 3 tigers: 2 had mild lymphocytic or lymphoplasmacytic inflammation (case Nos. 22 and 23), and 1 (case No. 24) with pulmonary histoplasmosis had moderate histiocytic inflammation, although no yeasts were identified in the liver.
Cause of death was attributed to hepatic disease in 1 tiger (case No. 13) with suppurative cholecystitis (Fig. 1) and cellulitis. Biliary culture isolated more than 1000 colonies of Escherichia coli and Trueperella pyogenes (formerly Actinomyces and Arcanobacterium pyogenes), while more than 1000 colonies of Clostridium sp and a variety of other Gram-positive rods and cocci were isolated from the cervical abscess. Both infections contributed to the decision to euthanize that animal.
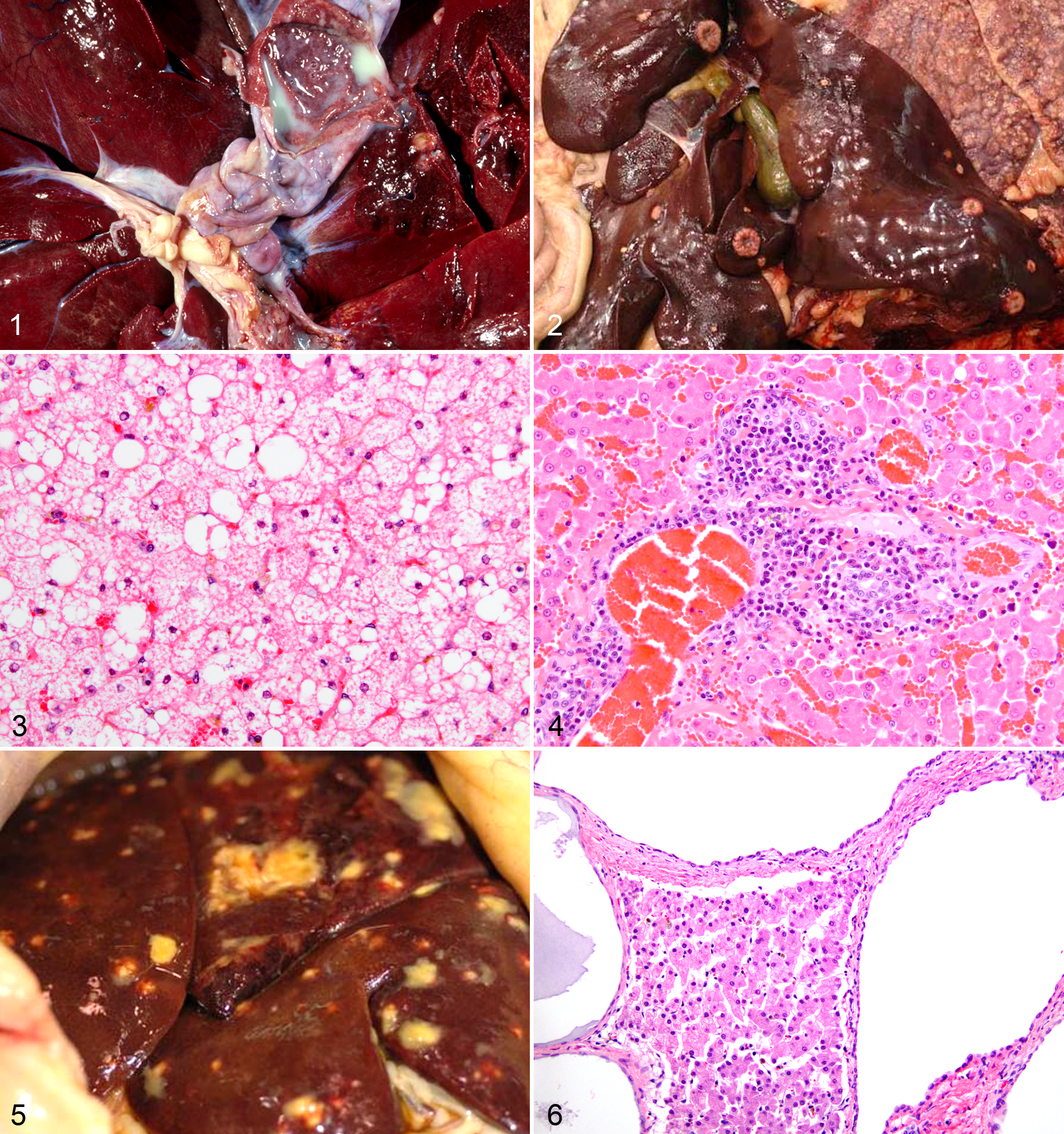
Mammary adenocarcinomas metastasized to the liver in 3 tigers (case Nos. 25, 26, and 27; Fig. 2) and to other organs but not the liver in 2 cases (case Nos. 5 and 7). Hepatocellular adenoma was identified in a tiger that died of blastomycosis (case No. 30). Lymphoma effaced the liver in 1 tiger (case No. 28).
Tigers were significantly more likely than other species to have no significant findings (OR, 12.8; P = .002). Tigers were significantly less likely than other species to have vacuolar change (OR, 0.19; P = .005) or hepatic stellate cell hypertrophy and hyperplasia (OR, 0.21; P = .016).
Cougars
The mean age and median BCS for cougars were 13.3 years and 3. Thirteen of 17 cougars (76%) had a hepatic lesion, and diagnoses were hepatic lipidosis (6/17 [35%]) (Fig. 3), portal hepatitis (4 [24%]), biliary hyperplasia (3 [18%]), biliary cysts (2 [12%]), and 1 each of multifocal random hepatitis, telangiectasia, hepatic necrosis, and cholestasis. Of the 6 cases with lipid accumulation, lipid distribution was diffuse in 3 cases, random in 2, and centrilobular in 1. There was no glycogen accumulation in any cougar. Portal hepatitis was mildly (case Nos. 39 and 41) to moderately (case Nos. 42 and 43) lymphoplasmacytic in all cases (Fig. 4).
Cause of death was attributed to hepatic disease in 2 cougars (case Nos. 39 and 45). In 1 obese cougar, severe hepatic lipidosis was the cause of death (case No. 39; Fig. 3). Pyogranulomatous hepatitis, in addition to peritonitis and sepsis, was the cause of death in another cougar (case No. 45; Fig. 5); Yersinia pseudotuberculosis was isolated on antemortem cultures.
Case No. 40 also had severe hepatic lipidosis; however, death was attributed to bacterial bronchopneumonia. Hepatocellular necrosis was identified in 1 cougar that died due to uterine adenocarcinoma and had an incidental pheochromocytoma (case No. 47). Mild biliary hyperplasia was the only hepatic lesion in 1 case (case No. 44). One cougar (case No. 47) had histologic evidence of cholestasis.
Cougars were more likely than other species to have lipogranulomas (OR, 14.73; P = .002) and less likely to have hepatic stellate cell hypertrophy and hyperplasia (OR, 0.071; P = .002).
Lions
Mean age and median BCS for lions were 15.3 years and 2. All 12 lions had at least 1 hepatic lesion. Diagnoses included lipidosis (8 [67%]), biliary cysts (7 [58%]) (Fig. 6), metastatic carcinoma (2 [17%]), and 1 each of fibrosis and portal hepatitis. Of the 8 cases with lipid accumulation, the distribution was diffuse in 3 cases, random in 2, centrilobular in 2, and midzonal in 1. The 1 case (case No. 56) with portal hepatitis was mild and lymphocytic.
No lions died of hepatic disease. One lion with mild hepatic lipidosis died of lymphoma (case No. 53); however, there was no histologic evidence of hepatic involvement of the neoplasm.
Lions were significantly more likely than other species to have benign cystic biliary lesions (OR, 5.97; P = .021).
Other Nondomestic Felids
Hepatic lipidosis was common in other nondomestic felids, occurring in leopards (2/8 [25%]), servals (5/6 [83%]), snow leopards (3/5 [60%]), cheetah (2/3 [68%]), lynx (2/3 [68%]), and a bobcat (1/1 [100%]). Other hepatic lesions were biliary cysts (n = 5) (Fig. 7), glycogen accumulation (n = 6), portal hepatitis (n = 4), cholangiocellular carcinoma (n = 2) (Fig. 8), necrosis (n = 2) (Fig. 9), biliary hyperplasia (n = 2), telangiectasia (n = 2), hemangiosarcoma (n = 1), veno-occlusive disease (n = 1) (Fig. 10), hemorrhage (n = 1), and osteosarcoma (n = 1). Extramedullary hematopoiesis, lipogranulomas, and hepatic stellate cell hypertrophy and hyperplasia were observed in 20, 19, and 24 other animals, respectively.
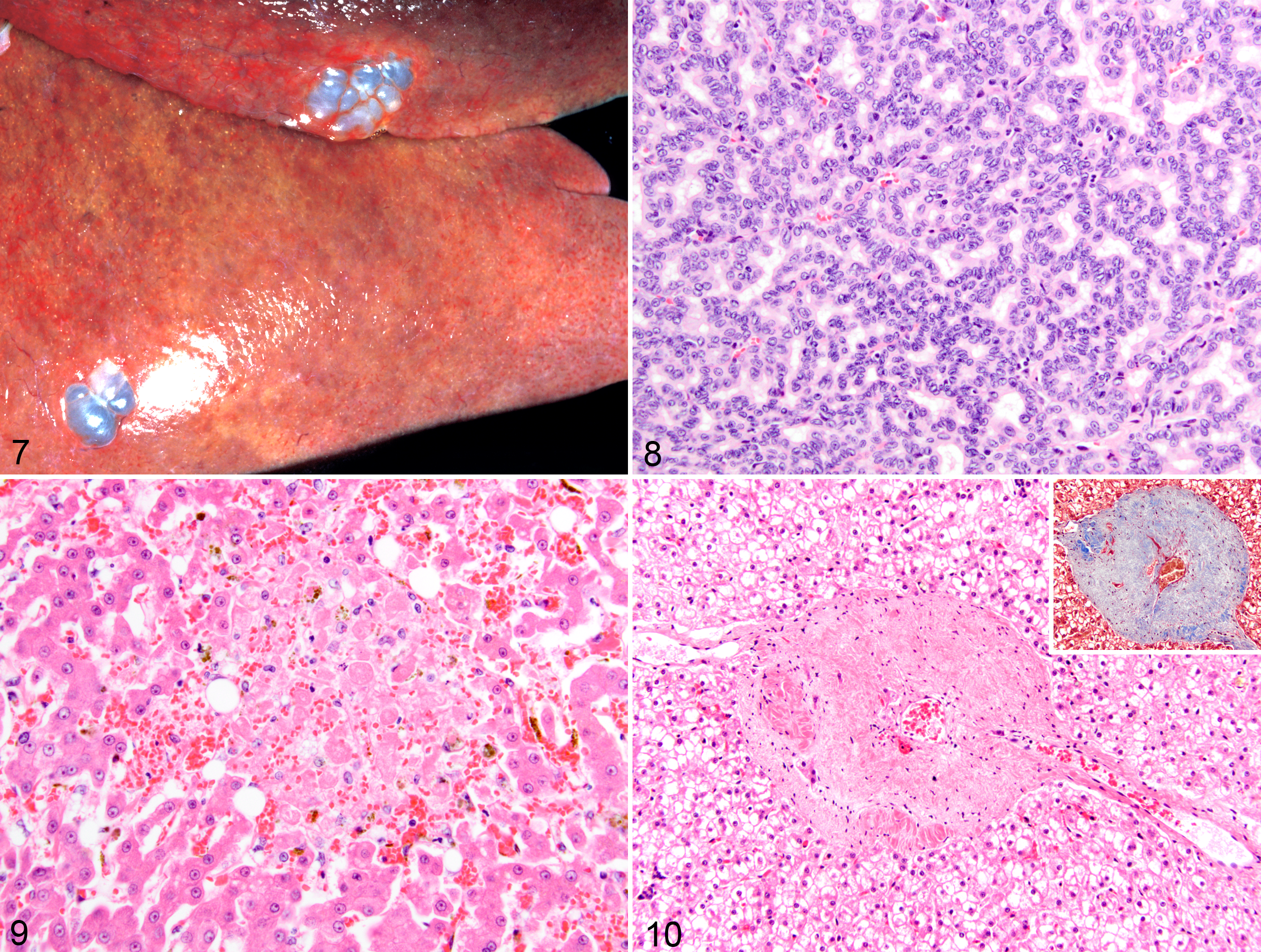
Cause of death was attributed to hepatic disease in 1 serval that had severe coagulative hepatocellular necrosis with sepsis (case No. 73; Fig. 9). Cause of euthanasia was attributed to cholangiocellular carcinoma in 2 leopards (case Nos. 66 and 67). Both these animals also had biliary cystic lesions.
One serval (case No. 72) had severe hepatic lipidosis; however, the cause of euthanasia was renal disease. Mild portal hepatitis was present in 1 leopard, serval, ocelot, and clouded leopard each. Portal hepatitis was lymphoplasmacytic in the leopard, serval, and clouded leopard and lymphocytic in the ocelot. There was a single case of a metastatic hemangiosarcoma in a snow leopard (case No. 77). The snow leopard (case No. 78) was the only case of veno-occlusive disease in this study. This animal also had severe glycogen accumulation (Fig. 10) and telangiectasia.
Discussion
This study indicates that primary hepatic disease in nondomestic cats is uncommon. In only 6 of 90 animals was hepatic disease related to cause of death.
In this study, vacuolar change was common, occurring in 46% of felids. Vacuolar change was characterized as microvesicular as is typical of lipid in 33 cases, feathery as is typical of glycogen in 4 cases, and a mixture of both in 4 cases. Hepatocellular swelling or vacuolar change from lipid or glycogen accumulation is a common and nonspecific finding. 28 Most cases of lipid accumulation were mild and consistent with increased peripheral mobilization of lipid rather than hepatocellular degeneration. Severe hepatic lipidosis was present in 3 cases and was considered the cause of death in 1 obese cougar. That cat was hypothesized to have experienced idiopathic hepatic lipidosis due to anorexia, excessive peripheral mobilization of free fatty acids, and hepatic dysfunction with icterus, as has been described in domestic cats. To our knowledge, hepatic lipidosis has not been reported as a cause of death in nondomestic cats. Idiopathic hepatic lipidosis is likely a phenomenon of captivity and secondary obesity.
Glycogen accumulation was present in 8 cases. Changes were moderate to severe in 3 leopards and 1 snow leopard. All other cases had mild accumulation, which occurred in a variety of species. A leopard and snow leopard with glycogen accumulation received steroids within 1 month of autopsy. The other 6 cases with glycogen accumulation did not have histories of steroid administration, endocrine disease, or glycogen storage disorders. Other possible causes of glycogen accumulation in this series may include a meal immediately prior to death, causing glycogen to be stored in hepatocytes.
There are 3 recent case reports of biliary cysts in lions. 2,11,29 In 1 case report, renal cysts were also present, and a mutation of the PKD1 gene, similar to domestic cats, was suspected but could not be identified. 2 In our study, lions were 6 times more likely to have cystic biliary lesions than were other nondomestic felids. Six of 7 cystic lesions in lions were multifocal, and adult-type polycystic disease (von Meyenburg complexes) was suspected. However, in the lions in this study, renal cysts were not identified. In the current study, only 1 of the 14 felids with biliary cysts had concurrent renal cysts (a cougar; data not shown). We are unable to determine if biliary cysts were present since birth in any case and therefore might represent a juvenile onset of disease.
Despite several studies describing the spontaneous occurrence of veno-occlusive disease in cheetahs and snow leopards, 3,15,17,26 only 1 snow leopard in our study had this lesion. The cause for the lower incidence in our study is unknown and may be related to genetic factors or the fewer numbers of cheetahs and snow leopards in our population. Veno-occlusive disease was reported to be the primary cause of death in 5 of 52 captive cheetahs in South Africa 16 and 9 of 31 captive cheetahs in North America; however, in the North America study, 82% of deceased cheetahs had mild evidence of veno-occlusive disease whether or not it was the primary cause of death. 15 In a study of 54 snow leopards, 42 had evidence of veno-occlusive disease. 17
In our study, there was no association between age and lipogranulomas. However, there was a significant species association. Cougars were 14.7 times more likely to have lipogranulomas than other nondomestic felids. The reason for the apparent predisposition is unclear, because most cougars lack concurrent lesions that might result in increased hepatocyte turnover. One affected cougar had pyogranulomatous hepatitis and another had multifocal necrosis, but lipogranulomas were also present in many other animals with no evidence of hepatocellular loss.
Hepatic stellate cell hypertrophy and hyperplasia commonly occur in older cats, and one known cause in domestic cats is chronic vitamin A intoxication. 28 In our study, there was no age association with hepatic stellate cell hypertrophy and hyperplasia, but there was a significant negative association between species and hepatic stellate cell hypertrophy and hyperplasia. Tigers were 5 times less likely and cougars were more than 10 times less likely than other nondomestic felids to have hepatic stellate cell hypertrophy and hyperplasia. In a study of snow leopards with veno-occlusive disease, there was a trend of increased hepatic stellate cell hypertrophy and hyperplasia with age, but an association between hepatic stellate cell proliferation and fibrosis could not be identified. 17
The only primary hepatic malignancies in our study were 2 intrahepatic cholangiocellular carcinomas, both in leopards. A cholangiocellular carcinoma has been reported in a margay, 13 and 2 gallbladder adenocarcinomas have been reported in lions. 20 Lombard and Witte reported 3 carcinomas of the biliary tree in captive felidae. 10 There were no hepatocellular carcinomas in our study; however, there was 1 hepatocellular adenoma in a tiger. Hepatocellular carcinomas have been reported in a lion 19 and a tiger. 22
Similar to domestic species, metastatic neoplasms were more common in the liver in this study than were primary hepatic neoplasms. 28 Mammary adenocarcinoma was the most common neoplasm to metastasize to the liver (4 cases). Melengestrol acetate exposure has been linked to mammary tumors in captive nondomestic felids. 5 Lymphoma was the cause of death in 4 animals in this study; however, it metastasized to the liver in only 1 tiger. Lymphoma with hepatic involvement has been reported in a lion that had concurrent obstructive biliary disease due to gallstones. 1 Harrison et al reported the spleen as the most common primary site of lymphoma in 11 lions. 6 Six cases in that study had hepatic metastases, and the predominant immunophenotype was CD3 positive. Other metastatic neoplasms in this study included hemangiosarcoma, pancreatic exocrine carcinoma, and osteosarcoma, and all had widespread metastasis.
Seventy-two of 90 Prussian blue–stained livers (80%) had hepatocellular iron accumulation, and 87 of 90 had iron accumulation within Kupffer cells (97%). This is higher than 1 study in snow leopards with veno-occlusive disease in which 65% of livers had iron accumulation in Kupffer cells or hepatocytes. 17 The high prevalence of iron in this study may indicate that accumulation of iron in nondomestic felid hepatocytes is normal, as has been previously suggested for domestic felids. 27 Iron accumulation was not associated with lesions or with particular species.
Copper accumulation was uncommon in our study (11%) but had a similar prevalence as was reported in domestic cats 27 and snow leopards with veno-occlusive disease. 17 The 1 case with veno-occlusive disease in our study did not have copper accumulation. In dogs, copper accumulation usually begins in centrilobular regions and is associated with hepatocellular loss and fibrosis. 28 In our study, copper accumulation tended to be random and not associated with any lesion. In 2 sections, a periportal distribution occurred, which is the predominant distribution observed in secondary copper accumulation in dogs.
Limitations of this study included the absence of clinicopathological data, hepatic and biliary cultures, and quantitative hepatic iron and copper analyses. The retrospective nature of the study limited the amount of tissue for additional tests. Routine veterinary care in our population could also lead to underestimation of hepatic disease due to a positive response to treatments and nonfatal outcomes of many hepatic diseases in cats.
Liver disease was not a common cause of death in our population of nondomestic felids. This study confirms a significantly increased occurrence of biliary cystic lesions in lions compared with other nondomestic felids. In addition, for unknown reasons, significant liver lesions in tigers were less frequent than for other species. Similar to previous reports in domestic cats, iron accumulation was common in nondomestic felids, and there was no relationship between lesion or species and hepatocellular iron or copper accumulation, or sinusoidal fibrosis.
Footnotes
Acknowledgements
We thank Ann Reed for her assistance with statistical analyses, Misty Bailey for a critical review of the manuscript, Anik Vasington for assistance with the digital images, and the staff and curators of the Knoxville Zoo and Tiger Haven for their help with the animals as well as with organizing the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by the University of Tennessee Department of Small Animal Clinical Sciences Companion Animal Fund and the University of Tennessee Center of Excellence in Livestock Diseases and Human Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
