Abstract
Equine herpesvirus-5 (EHV-5) is commonly found in healthy asymptomatic horses worldwide. Although a cause-and-effect relationship has not been thoroughly determined, this virus has been associated with several disease conditions including equine multinodular pulmonary fibrosis (EMPF) and 1 case of interface dermatitis. The authors searched the New York State Animal Health Diagnostic Center database for cases of equine interface dermatitis between 2007 and 2022. Ten cases were identified and scrutinized for viral inclusion bodies which were present in 5 of 10 cases. Two similar cases with interface dermatitis and viral inclusion bodies, which were not part of a retrospective search, were from the Oregon Veterinary Diagnostic Laboratory. The authors describe a total of 7 horses with dermatitis characterized by crusted, alopecic, non-pruritic, non-painful, irregular to annular areas over the face, most commonly the muzzle, for up to several years duration. Histologically, there was a CD3+ T lymphocyte-dominated lymphohistiocytic interface dermatitis with hydropic degeneration, apoptotic keratinocytes, and pigmentary incontinence. Keratinocytes within the upper stratum spinosum and stratum granulosum had glassy pale basophilic intranuclear inclusion bodies consistent with herpesvirus. The presence of EHV-5 was confirmed by quantitative polymerase chain reaction (qPCR) and in situ hybridization in 7 horses and by electron microscopy in 1 horse. One horse later developed EMPF and was euthanized. EHV-5 was not detected with qPCR from 5 control horses and 5 horses with interface dermatitis without histologic evidence of viral inclusion bodies. These are the first cases of facial interface dermatitis associated with EHV-5 reported in the United States.
Keywords
Equine herpesvirus-5 (EHV-5) is a gammaherpesvirus, in the genus Percavirus that is common in horses worldwide. This virus causes acute infections in young foals and, like other herpesviruses, can establish lifelong latency. 12 Because EHV-5 is commonly found in healthy horses, it has long been thought to be non-pathogenic. In fact, EHV-5 has been detected in multiple normal equine tissues and cells, including B and T lymphocytes, the nasal cavity, lymphoid tissues, the gastric mucosal epithelium, cranial mesenteric ganglia, and the reproductive tract of mares.2,8,10,11,15,23 The distribution of EHV-5 in multiple tissues, including the skin, is yet to be established.
EHV-5 has been detected in horses with multiple different disease conditions; however, because this virus can be detected in normal tissues, the exact role this virus plays in the pathogenesis of these conditions is still largely unknown.5,6,9,11,15,16,18,26,27 In general, disease associated with EHV-5 is rare, with the most significant disease association with EHV-5 being equine multinodular pulmonary fibrosis (EMPF).26,27 EHV-5 has also been detected in cases of lymphoma although the significance of the virus in this condition is still unknown. 23
EHV-5-associated interface dermatitis was described on the face of a horse from Germany in 2012. 6 Interface dermatitis is a common reaction pattern in dermatopathology. The dermal-epidermal junction is the site of the pathology, and the classical features include varying degrees of hydropic degeneration, keratinocyte apoptosis, lichenoid cellular infiltrate, and pigmentary incontinence. 19 This reaction pattern has been linked to immune-mediated disease such as cutaneous lupus erythematosus (CLE), cutaneous adverse drug reactions, and erythema multiforme (EM). 19
Here, we describe 7 horses with interface dermatitis limited to the facial skin. In addition, there is both histologic evidence and DNA detection of EHV-5 suggesting a potential association.
Materials and Methods
A retrospective search for cases was performed through the New York State Animal Health Diagnostic Center, Cornell University, College of Veterinary Medicine between 2007 and 2022. Search parameters included all skin biopsy specimens from horses with the search terms “interface dermatitis,” “lupus erythematosus,” “lichenoid,” and “lupoid.” Once cases were identified, each hematoxylin and eosin glass slide was reviewed by a board-certified veterinary pathologist and dermatologist (JP-K) to verify the presence of interface dermatitis. To be included, each case needed to have at least 2 of the following criteria: hydropic degeneration, apoptotic keratinocytes, a lichenoid band of inflammation in the superficial dermis, and pigmentary incontinence. All cases meeting these criteria were then scrutinized under the light microscope for viral inclusion bodies. For those cases with viral inclusion bodies, we used the submission forms or contacted referring veterinarians for lesion descriptions, lesion locations, treatment, photographs, and clinical follow-up.
Skin from the muzzle of 5 horses served as controls. Controls included 3 horses with normal muzzle skin, a sarcoid from the muzzle skin of a 19-year-old quarter horse gelding, and an eosinophilic granuloma from the muzzle of a 15-year-old American saddlebred gelding. The 3 horses with normal skin were a 7-year-old quarter horse gelding, a 2-year-old thoroughbred stallion, and a 3-year-old thoroughbred gelding. These 3 samples were taken from carcasses with no skin lesions that presented for necropsy for colonic impaction, traumatic shoulder injury, and proximal sesamoid fracture, respectively.
Histology
All skin samples were collected with either a scalpel blade or a 6-mm punch biopsy instrument from representative skin lesions. Skin samples were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin wax, sectioned at a 3–5 μm thickness, and stained with hematoxylin and eosin. Histopathology was performed on all the case material, including the 5 controls.
Polymerase Chain Reaction Assays
The presence of EHV-2 and EHV-5 DNA was assessed in formalin-fixed, paraffin-embedded (FFPE) skin tissues and fresh frozen tissue (3 controls) using primer/probe sets specific for the respective viral glycoprotein B (gB) genes, as previously described. 8 In addition to the 12 horses with interface dermatitis, we also tested skin from the muzzle from 5 horses without interface dermatitis (negative method controls). For the paraffin-embedded tissue, 2 serial, 25-µm-thick scrolls were cut from one tissue block from each case. Total nucleic acid was extracted using a commercial kit (RecoverAll, Life Technologies, Carlsbad, California). Samples with nominal cycle numbers (Ct) below 35.91 were considered positive, while samples with Ct values above this value were considered negative. At this cut-off value, the analytical sensitivity of the assay is 10 copies of target sequence per microliter.
Immunohistochemistry (IHC, CD3, and CD20)
The presence and degree of immune cell infiltration were assessed by standard immunohistochemical analysis of FFPE tissues from horses 1 to 12 at the New York State Animal Health Diagnostic Center, Cornell University, College of Veterinary Medicine. IHC was not performed on the 5 control tissues. Immunolabeling was performed on 4 μm serial sections of FFPE samples using standard protocols on the Leica Bond Max Automated IHC Staining System (Leica Microsystems, Buffalo Grove, Illinois). 14 Briefly, mouse anti-human monoclonal antibody Bond Ready-To-Use Primary Antibody CD3 (LN10; Leica Microsystems, Buffalo Grove, Illinois, catalog #PA0553) and rabbit anti-human polyclonal CD20 antibody (ThermoFisher/Invitrogen catalog#PA5-16701; dilution 1:400) were used. Paraffin-embedded slides were dewaxed with Bond Dewax Solution (Leica-cat#AR9222). Heat-induced epitope retrieval with Bond Epitope Retrieval Solution 2 (Leica-cat#AR9640) was applied for 20 minutes (CD3) or 30 minutes (CD20), followed by peroxide block, which was applied for 5 minutes (Leica-cat#DS9800). After application of primary antibodies for 15 minutes, PV-HRP-Anti-Mouse IgG reagent (Leica-cat#PV6114) or Leica Bond Polymer Refine Detection Kit Polymer (Leica-cat#DS9800) was applied to the slides for 10 minutes. Application of Leica Bond Polymer Refine Detection for 10 minutes (Leica-cat#DS9800) with 3,3′-diaminobenzidine as the chromogen was followed by counterstaining with hematoxylin for 5 minutes (Leica-cat#DS9390). Normal equine lymph node and spleen served as positive antibody and methods controls, whereas omission of the primary antibody on serial sections of case slides served as negative methods controls. Positive immunolabeling was identified as brown labeling along the cell membrane.
In Situ Hybridization Assays
A highly sensitive and previously validated in situ hybridization (ISH) method (RNAscope; Advanced Cell Diagnostics, Hayward, California) was used for cellular localization of EHV-5 in FFPE tissue sections in all 7 horses with inclusion bodies and in which EHV-5 was detected via quantitative polymerase chain reaction (qPCR). 15 An ISH probe V-EHV-5-gB (Cat#463701, a 20ZZ probe targeting 259-1737 of Genbank reference sequence AF050671.1) was designed to hybridize within a 1478-nucleotide region of the EHV-5 gB gene as previously described. 15 The gB gene encodes one of the most highly expressed viral envelope glycoproteins of EHV-5.7,25 Briefly, sections of FFPE skin tissue were placed on charged slides and processed using the RNAscope 2.5 HD Detection Kit—Red according to the manufacturer’s protocol.15,24 After deparaffinization, 5-μm-thick tissue sections were permeabilized and hybridized with the EHV-5 probe followed by signal amplification, binding of a chromogenic agent, and counterstaining with hematoxylin, allowing direct visualization of EHV-5-infected cells by light microscopy. Positive signal was identified as red punctate areas within the nucleus and cytoplasm of infected cells.
Transmission Electron Microscopy
Selected FFPE tissues from horse 2 were deparaffinized, immersed in 3% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.2), stained with 2% uranyl acetate and Reynolds lead citrate, and processed routinely for transmission electron microscopy. Specimens were examined under a Tecnai G2 Spirit TWIN transmission electron microscope (Field Electron and Ion Co., Hillsboro, Oregon).
Results
Horses 1–3 and 6–12 (Table 1) all presented to the New York State Animal Health Diagnostic Center, Cornell University, College of Veterinary Medicine as diagnostic mail-in skin biopsy specimens between 2007 and 2022. Horses 4 and 5 were not part of a retrospective search. These horses were identified by pathologists at the Oregon Veterinary Diagnostic Laboratory, Carlson College of Veterinary Medicine, Oregon State University in 2016 and 2017, respectively.
Signalment, lesion location, lesion description, presence of viral inclusion bodies, and equine herpesvirus-2 and herpesvirus-5 (EHV-2/5) quantitative polymerase chain reaction results for 12 horses with interface dermatitis.
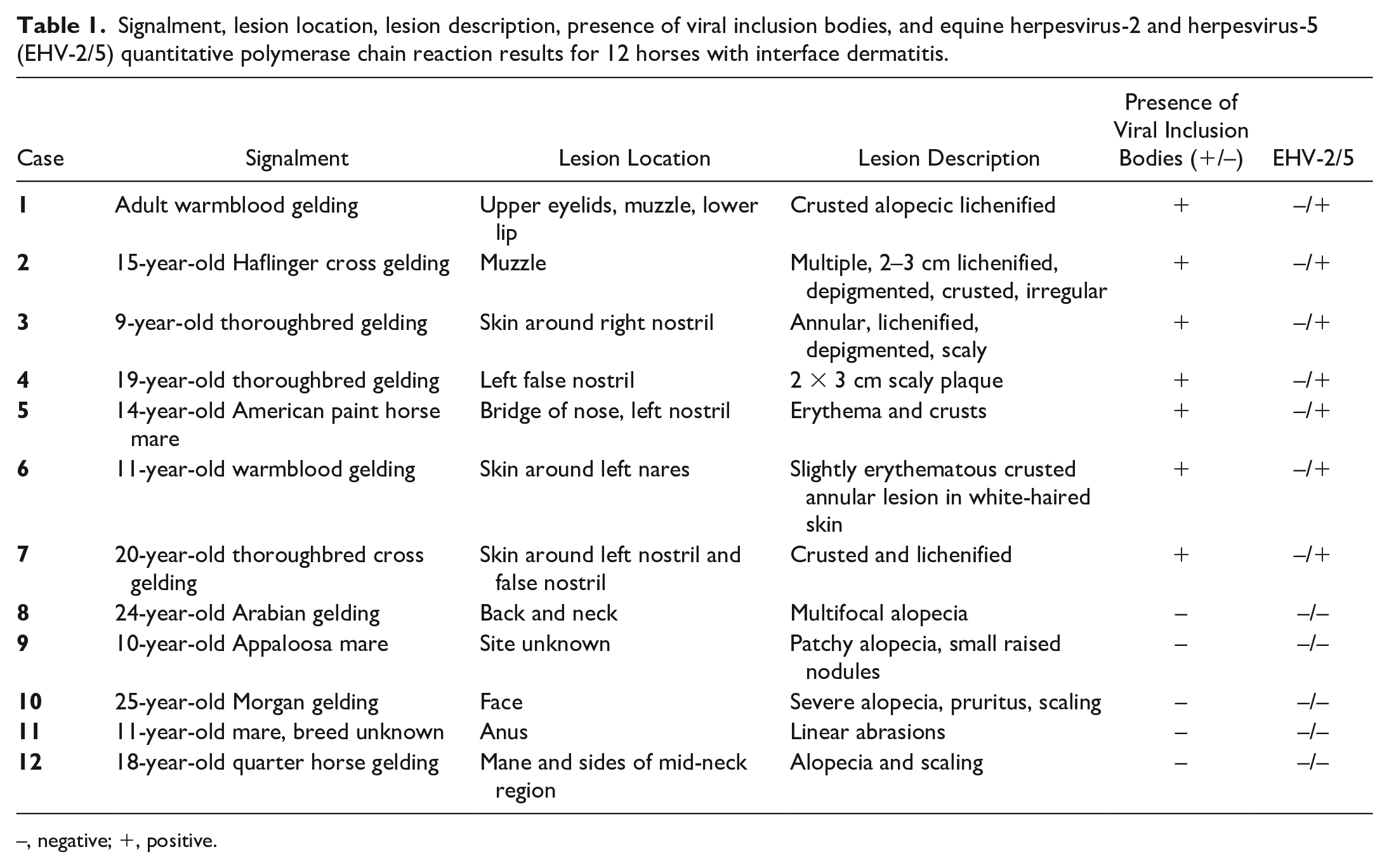
–, negative; +, positive.
Twelve horses met the inclusion criteria and 7 (horses 1–7) of those had histologically identifiable viral inclusion bodies (Table 1). Lesions were confined to the face in 8 of the horses. Crusting or scaling and alopecia were described in 9 and 7 horses, respectively. All 7 horses with viral inclusion bodies had crusted or scaly irregular to annular areas confined to the face, predominantly on the muzzle and around the nostrils (Fig. 1, Table 1). Horse 1 also had lesions over the right eye and left lip commissure (Fig. 1a–c). Lesions were described as alopecic in 3 of 7 and non-pruritic, non-painful, and lichenified in 4 of 7 horses. Depigmentation within the lesions was described in 2 of the horses and erythema was noted in 2 other horses. Six of these 7 horses were privately owned. One horse was owned by a county parks system in the state of New Jersey (horse 2). None of the horses were related nor did any of the horses live on the same farm. The age was unknown for horse 1 and a history was not provided for horse 2. The other 6 horses ranged in age from 9 to 20 years; breeds were thoroughbreds (n = 3), warmbloods (n = 2), and single cases of Haflinger-cross and American paint horse. Six of the horses were geldings, and one was a mare.
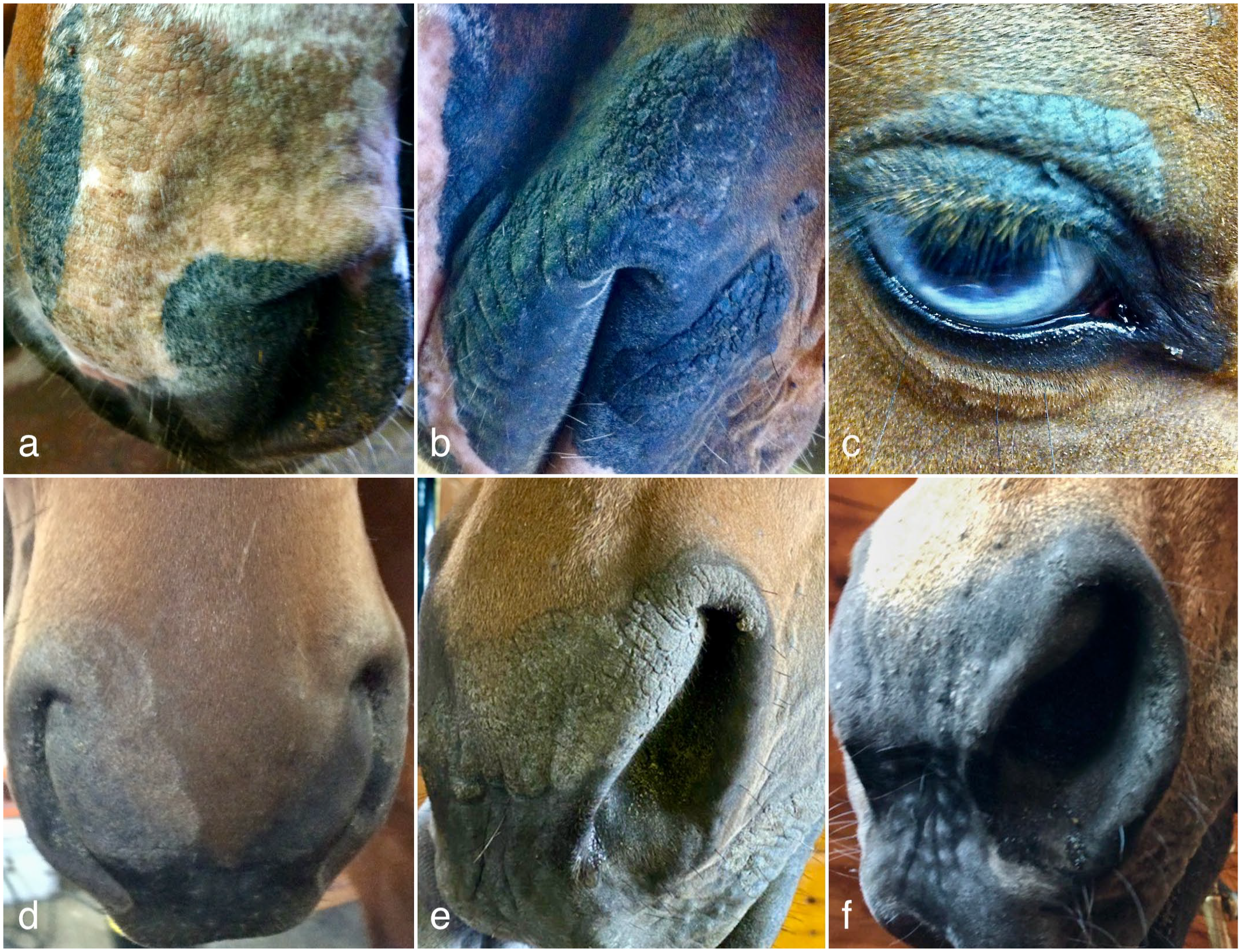
Clinical presentation of equine herpesvirus-5 (EHV-5)-associated dermatitis, horse. (a–c) Adult warmblood gelding (horse 1) with crusted lichenified well-demarcated lesions. (a) Muzzle. (b) Lips. (c) Upper eyelids. (d) 9-year-old thoroughbred gelding (horse 3) with a well-demarcated annular area of alopecia, lichenification, and scaling around the right nostril. (e) 20-year-old thoroughbred cross gelding (horse 7) with well-demarcated annular area of alopecia and lichenification surrounding the left nostril and extending into the false nostril and across midline. (f) Horse 7. Three months after the biopsy was taken, the dermatitis is greatly improved with no treatment (same horse as in e).
History, state of residence, treatment, and follow-up were recorded for the 7 horses with interface dermatitis and histologically identifiable inclusion bodies (Table 2). The duration of lesions was described as unknown to “a few years” in all cases. Post-biopsy follow-up was obtained and various treatments were described in 6 of the 7 horses (Table 2). Three years and 4 months after the initial skin biopsies (Fig. 1d), horse 3 presented to the Cornell University Equine and Farm Animal Hospital with a 2-month history of fever, cough, and weight loss. Large areas of lung consolidation were detected on ultrasound. Thoracic radiographs showed multifocal nodular areas and severe interstitial pneumonia. Aerobic bacterial culture of the lung yielded no growth. Lung histopathology revealed a severe, diffuse, chronic, pyogranulomatous pneumonia with interstitial pulmonary fibrosis and type-II pneumocyte hyperplasia. Obvious intranuclear inclusion bodies were not seen. ISH for detection of EHV-5 yielded strong nuclear and cytoplasmic signal within alveolar macrophages. The horse was treated with intramuscular injections of 50 mg dexamethasone once daily, but ultimately was humanely euthanized 1 month later due to persistent fevers and poor prognosis. Horse 4 had a 15-year history of cutaneous melanomas on the hock, tail, and perianal skin. Horse 5 had a more than 3-year history of intermittent right ocular discharge which was better in the winter months. Horse 6 had a history of chronic watery eyes and a seasonal cough in the fall. Horse 7 had alopecia, crusting, and lichenification prior to biopsy (Fig. 1e). No other horses or caretakers were affected on the farm. This horse also had a history of cetirizine-responsive seasonal pruritus with ulcers and infections in the right eye reportedly secondary to rubbing. A 3-month follow-up resulted in greater than 80% improvement with no treatment. Mild crusting waxed and waned every few days (Fig. 1f).
State of residence, history, treatment, and follow-up information for 7 horses diagnosed with EHV-5-associated dermatitis.
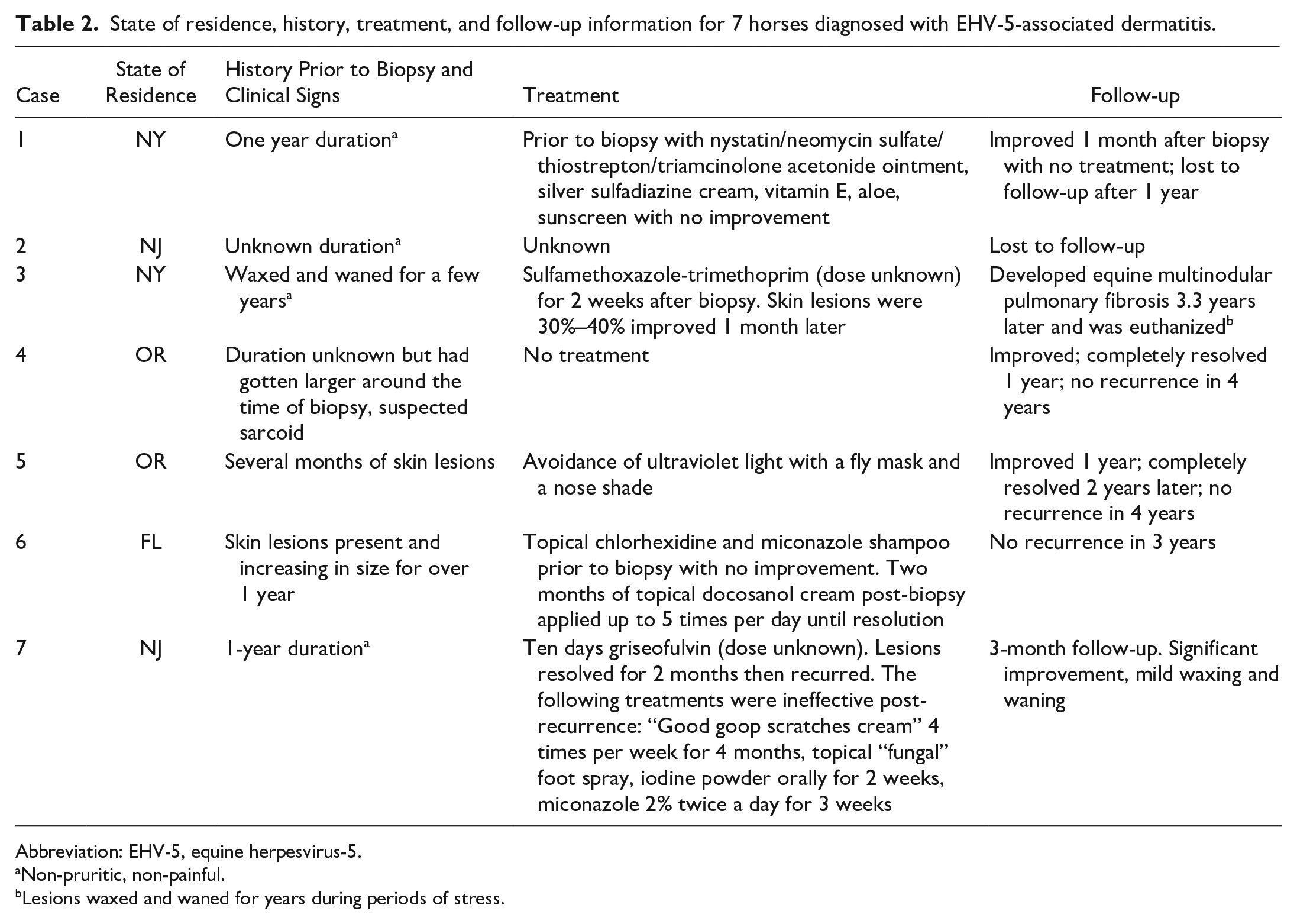
Abbreviation: EHV-5, equine herpesvirus-5.
Non-pruritic, non-painful.
Lesions waxed and waned for years during periods of stress.
Viral inclusion bodies were not noted in horses 8–12. Alopecia was described in 4 and scaling in 2 of these 5 horses. Lesions were on the neck in 2 of the 5 cases, and one each on the anus, face, and an unknown site. These horses ranged in age from 10 to 25 years; breeds were Arabian, Appaloosa, Morgan, quarter horse, and one unknown breed. There were 3 geldings and 2 mares.
Histopathology
The histopathologic changes were similar among horses 1–7. There was epidermal hyperplasia characterized by 10–15 cell layer expansion of the stratum spinosum and hyperkeratosis, which was predominantly parakeratotic measuring 15–50 layers. Variable numbers of lymphocytes and histiocytes (50–200 cells per 0.055 mm field) infiltrated the superficial dermis, epidermis, and outer root sheath of hair follicles (Fig. 2a). Melanomacrophages and free melanin (pigmentary incontinence), plasma cells, eosinophils, and neutrophils (5–20 cells per 0.055 mm field) were also present in the superficial dermis (Fig. 2b). Multifocally, the epidermal-dermal junction was obscured by the inflammatory infiltrate, and there was variable cytoplasmic vacuolation of basal keratinocytes (hydropic degeneration) (Fig. 2c). Overlying the most inflamed areas, keratinocytes within the upper stratum spinosum, stratum granulosum, and stratum corneum had glassy pale amphophilic intranuclear inclusion bodies that filled the nucleus and marginated the chromatin, suggestive of herpesvirus infection (Fig. 2d, e).
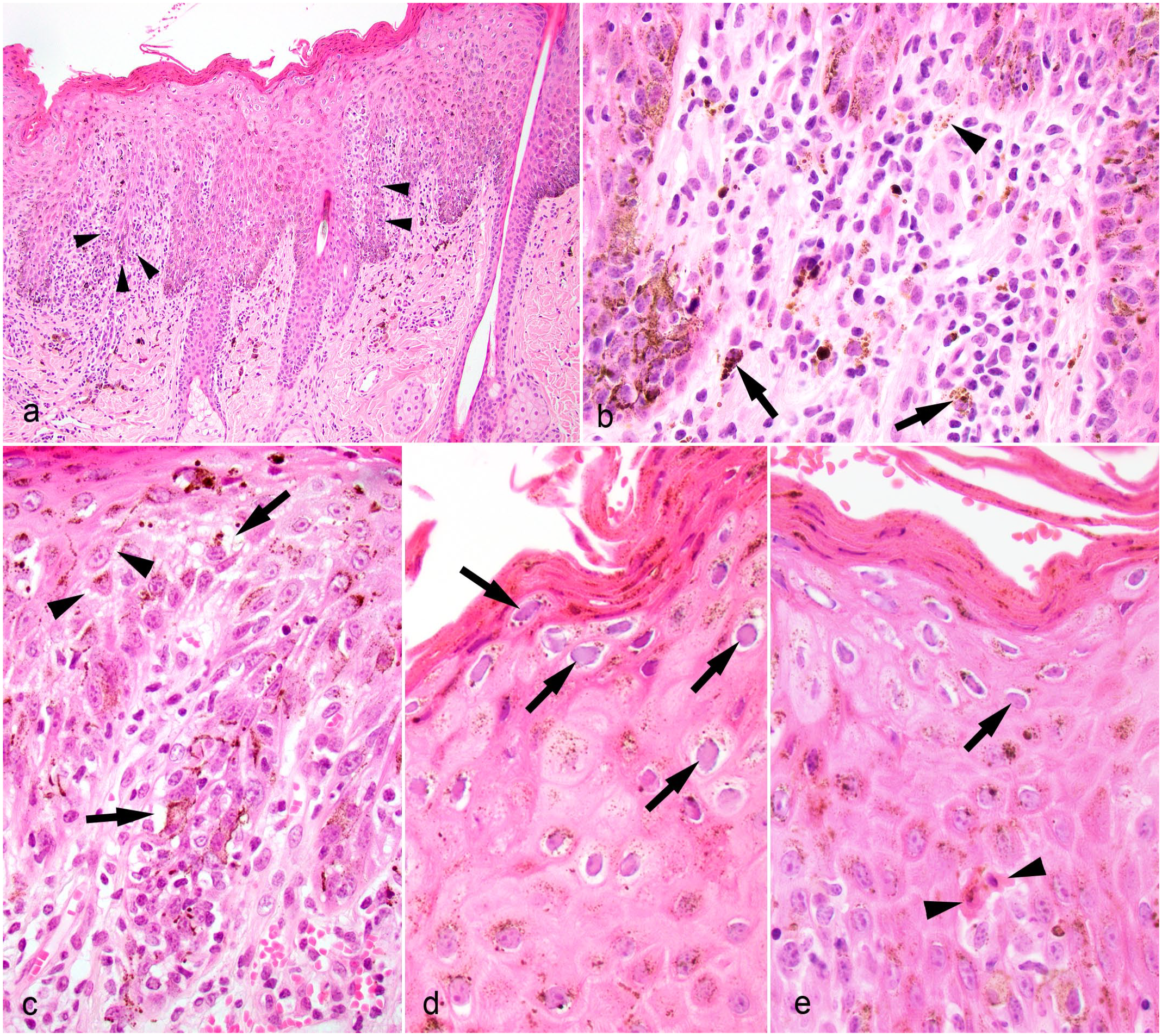
Equine herpesvirus-5 (EHV-5)-associated dermatitis, haired skin, horse. Hematoxylin and eosin. (a) Interface dermatitis with epidermal hyperplasia and parakeratotic hyperkeratosis. A moderate number of lymphocytes and histiocytes infiltrate the superficial dermis and stratum basale blurring the dermal-epidermal junction (arrowheads). Horse 7. (b) Higher magnification of (a). Macrophages containing intracytoplasmic melanin (arrows) and free melanin (arrowhead) are noted in the superficial dermis. (c) Higher magnification image of (a). Multifocal intracytoplasmic vacuoles are present within some basal keratinocytes (arrows) and spongiosis separates many keratinocytes (arrowheads). (d) Higher magnification image of (a). Keratinocytes in the strata spinosum and corneum contain amphophilic glassy intranuclear inclusion bodies that marginate chromatin (arrows). (e) Keratinocytes in the strata spinosum and corneum contain amphophilic glassy intranuclear inclusion bodies that marginate chromatin (arrows). Two apoptotic keratinocytes are present in the stratum basale (arrowheads). Horse 7.
Small numbers of apoptotic keratinocytes (1 per 0.11 mm field), some with lymphocytic satellitosis, were present throughout the epidermis, but predominantly in the stratum basale and stratum spinosum (Figs. 2e, 3a). Multifocal suppurative luminal folliculitis in horses 1 and 3 was suggestive of secondary bacterial folliculitis. Serocellular crusts were seen in horses 1, 3, and 5. Horse 5 also had thickening and hyalinization of capillary walls and occasional hemorrhage in the superficial dermis, as well as mild multifocal intra- and intercellular edema in the epidermis. Horses 5 and 6 also had moderate solar elastosis.
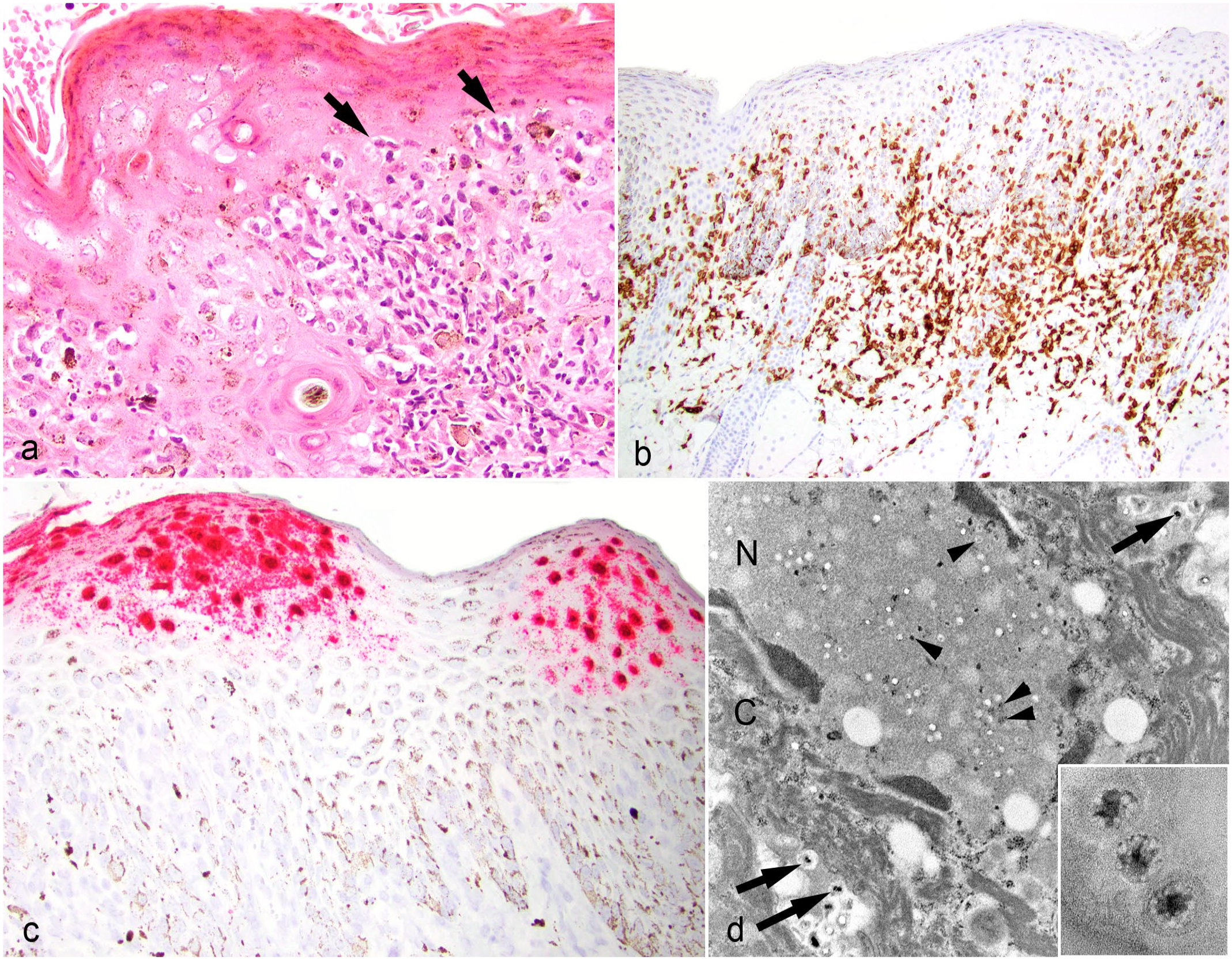
Equine herpesvirus-5 (EHV-5)-associated dermatitis, haired skin, horse. (a) Apoptotic keratinocytes are surrounded by lymphocytes (satellitosis) (arrow). Horse 3. Hematoxylin and eosin. (b) Immunohistochemistry for CD3, a marker of T lymphocytes. Strong immunoreactivity of small lymphocytes within the epidermis, outer root sheath of hair follicles, and superficial dermis. Horse 7. (c) In situ hybridization with EHV-5 probe. Viral nucleic acid within the nucleus and cytoplasm of keratinocytes within the strata spinosum, granulosum, and corneum. Horse 7. (d) Electron microscopy. Enveloped viral particles within the nucleus (N; arrowheads) and cytoplasm (C; arrow) of an infected keratinocyte. Scale bar = 500 nm; 35,900× magnification. Inset: Progeny capsids at intermediate stage of viral DNA encapsidation. Each capsid is composed of a nucleoid surrounded by an electron-lucent space and an outer capsid. 380,000× magnification. Horse 2.
Similar features of interface dermatitis were present in horses 8–12, including hydropic degeneration; pigmentary incontinence; small numbers of multilevel apoptotic keratinocytes; and variable numbers of lymphocytes, histiocytes, and plasma cells with fewer neutrophils and eosinophils in the superficial dermis and at the dermal-epidermal junction. Erosions and ulcers were noted in horse 11. Inclusion bodies were not noted in these 5 horses. Control tissues included 1 sarcoid and 1 eosinophilic granuloma, and histologically normal skin from 3 horses.
All skin biopsy specimens were initially reviewed by a dermatopathologist. Of the 7 horses with viral inclusion bodies, these inclusions were over-looked in 2 cases (horses 1 and 2) and based on the histopathologic and clinical findings the lesions were interpreted as CLE-like disease. Horses 8–12 were also diagnosed as CLE-like disease with a differential diagnosis of cutaneous adverse drug reaction.
PCR Assays
EHV-5 DNA was confirmed in FFPE skin tissue from horses 1–7 using a virus-specific qPCR assay (Table 1). All FFPE skin samples from horses 1–7 were negative for the presence of EHV-2 DNA by quantitative PCR assays. Horses 8–12 and all control skin samples were negative for the presence of both EHV-2 and EHV-5.
Immunohistochemistry
The results were similar across the 12 cases with interface dermatitis. Eighty to ninety percent of the infiltrating lymphocytes in the superficial dermis and epithelium had strong CD3 immunolabeling (Fig. 3b). Ten to twenty percent of the infiltrating lymphocytes in the superficial dermis with rare numbers in the epidermis and hair follicle epithelium had moderate to strong CD20 immunolabeling.
ISH Assays
EHV-5 nucleic acid was found in all 7 cases tested (horses 1–7) and consisted of strong diffuse hybridization signal within the nuclei of keratinocytes along the upper stratum spinosum, stratum granulosum, and stratum corneum as well as numerous pinpoint punctate foci in the cytoplasm of the same cells (Fig. 3c).
Transmission Electron Microscopy
The stratum corneum contained keratinocytes with evidence of herpesviral replication accompanied by margination of nuclear chromatin. Enveloped viral particles were found in the nucleus and cytoplasm with progeny capsids at intermediate stages of viral DNA encapsidation in the cytoplasm. Individual capsid consisted of a nucleoid surrounded by an electron-lucent space and an outer capsid (Fig. 3d).
Discussion
In this study, we describe a unique dermatitis in horses with non-pruritic, non-painful facial skin lesions and histologic features of CLE-like interface dermatitis with intranuclear inclusion bodies in keratinocytes that correlate with the detection of EHV-5 DNA by PCR and intranuclear and cytoplasmic localization of viral nucleic acids by ISH within skin lesions. The clinical presentation was characterized by variable degrees of alopecia, crusting, and lichenification involving the muzzle and nostrils, and rarely the eyelids and lip (Fig. 1).
The most widely known herpesvirus-associated skin disease in horses is coital exanthema, caused by EHV-3, a highly contagious sexually transmitted disease. Lesions include papules, vesicles, pustules, and ulcers on the genital skin of stallions and mares. Histologically, lesions are characterized by epidermal hyperplasia, perivascular dermatitis, and ballooning degeneration of keratinocytes with eosinophilic intranuclear inclusion bodies. Intraepidermal vesicles may also be present. Other forms of herpesvirus-associated dermatitis in horses are limited to rare, isolated case reports.4,8,20 Papular, crusted, and ulcerative dermatitis of the mucocutaneous junctions including the tongue, perilabial region, nostrils, penis, and prepuce have been associated with EHV-1 in 1 horse. 3 EHV-2 was identified in 1 horse with multifocal granulomatous and necrotizing nodular lesions on the neck and chest. 20 Other than the detection of intranuclear viral inclusion bodies, none of these conditions clinically nor histologically resemble what we describe in our series of cases.
The exact role that the virus is playing in the pathogenesis of this dermatitis is unclear. EHV-5 can be detected in multiple normal tissues.2,8,11,10,15,23 The distribution of EHV-5 in normal skin, however, is unknown. In this report, we show that neither EHV-2 nor EHV-5 was detected in normal muzzle skin from 3 horses nor in the muzzle skin of 1 horse with a sarcoid and 1 horse with an eosinophilic granuloma. Neither of these viruses were detected from the additional 5 cases of interface dermatitis in which viral inclusion bodies were not histologically present. Although the numbers are small, these results further support that EHV-5 is not commonly present in muzzle skin without interface dermatitis and was not detected in any of the skin samples without viral inclusion bodies (n = 10; 6 muzzle, 2 neck, 1 anus, and 1 unspecified site). EHV-5 was only present in the cases with viral inclusion bodies (n = 7). These 7 cases have some unique features in common. Clinically, the lesions were very similar among the 7 especially in terms of location and distribution. In 5 of the 7 cases, lesions were described as asymmetric around 1 nostril. In the other 2 cases, lesions were described as being on the muzzle and in 1 of those 2 cases, over the eyelids and lips. Lesions were located on the face of 8 of the 12 horses with interface dermatitis. EHV-5 and viral inclusion bodies were detected in 7 of those 8 horses. The only clinical difference between the one case in which EHV-5 was not detected on the face and the cases of EHV-5 facial interface dermatitis was the presence of pruritus. Overall, the presence of interface dermatitis with intralesional viral inclusion bodies around the nostrils and muzzle of horses are the key differentiating feature of this unique dermatitis.
To our knowledge, dermatitis associated with EHV-5 has only been described in one case report from Germany with a similar presentation to our 7 cases reported herein. 6 That horse, a 9-year-old Holsteiner stallion, had non-pruritic, pustular dermatitis around both eyes, nostrils, and the muzzle for 1 year, as well as lethargy and poor performance. Histologically, there was lymphoplasmacytic and histiocytic hydropic interface dermatitis with apoptotic keratinocytes and multiple amphophilic intranuclear inclusion bodies in the stratum granulosum and stratum spinosum. The report compared that horse’s presentation with herpes-associated EM in people, which is commonly associated with herpes simplex virus-1. Patients with herpes-associated EM present clinically with herpetic outbreaks and subsequent development of symmetric target lesions on distal extremities, distant from the herpetic lesions themselves. 4 Therefore, because the only skin lesions in these horses were on the face, the lack of symmetrical acral lesions, the lack of typical clinical lesions associated with EM-like disease in horses (urticarial papules and plaques), the lack of significant multilevel keratinocytes apoptosis and satellitosis, and the presence of lichenoid interface dermatitis, we propose that all 8 cases are more similar to CLE-like disease than EM.
There are several interesting aspects of this condition. EHV-5-associated dermatitis in horses may pose a diagnostic challenge. Two cases were incorrectly diagnosed as CLE-like disease originally based on the histologic interface pattern in addition to the clinical presentation of alopecia and crusted lesions on the face. The lesions persisted for several years in some horses. One of the horses (horse 3) developed EMPF 3 years and 4 months after the diagnosis of facial interface dermatitis and was ultimately humanely euthanized.
In horses, CLE is characterized by symmetrical annular to oval areas of erythema, scaling, and alopecia beginning on the face, especially on the lips, nostril, and periocular region. 19 Histologically, there is hydropic or lichenoid interface dermatitis or both. All cases reported here had facial lesions with lichenoid interface dermatitis with varying degrees of hydropic degeneration of basal keratinocytes. The inclusion bodies were initially missed in horses 1 and 2, leading to an erroneous diagnosis of an autoimmune skin disease. In contrast to CLE, lesions lacked a clearly documented symmetrical distribution and were actually unilateral in 5 of 7 horses. Symmetry can be an important clue in making the diagnosis of an autoimmune skin disease versus an infectious or other inflammatory condition. The cases demonstrate the importance of careful examination of all the histologic features and resisting premature closure bias based on the clinical presentation.
The mechanism by which EHV-5 infection may lead to interface dermatitis is uncertain. It is not known whether the inflammatory infiltrate is simply a reaction to the virus or an autoimmune mechanism may be contributing to the pathogenesis of the dermatitis. Although cutaneous herpesviral infection is often associated with epithelial necrosis and vesicular to ulcerative lesions, an interface dermatitis reaction pattern has been demonstrated in both herpes simplex virus-1 and acute varicella-zoster virus infections in humans. 13 Furthermore, herpesviruses can induce apoptosis in host cells by inducing DNA fragmentation. 22
Photosensitivity may play a role in the pathogenesis of EHV-5-associated dermatitis. Two of 7 horses (horses 5 and 6) had evidence of solar damage and lesions limited to white-haired skin. In humans, ultraviolet light exposure is a potent stimulus for inducing reactivation of latent herpes simplex virus. 17 An abnormal response to ultraviolet radiation contributes to the pathogenesis of CLE, suggesting that autoimmune mechanisms secondary to ultraviolet radiation may play a role in our cases of EHV-5-associated facial interface dermatitis.
The duration of EHV-5-associated facial dermatitis in our cases was long (up to multiple years) compared to other herpesviral-associated dermatitides such as herpes simplex virus-1 in people and feline herpesvirus 1 in cats with lesions generally resolving in otherwise healthy individuals in 1–3 weeks. The reason for the long duration of lesions and presumed active infection in horses is unknown. Gammaherpesviruses cause acute infections in permissive cells, such as epithelium, and lifelong latency in nonproductive cells like lymphocytes. 12 Some horses are continuously shedding EHV-5 for extended periods. In one study, EHV-5 was frequently detected in nasal swabs of individual healthy horses over 1 year. 2 However, it is unknown whether this is due to chronic active infection, reactivation of latent infections, frequent reinfections, or shedding of multiple strains of EHV-5.
The authors propose that EHV-5 interface dermatitis is a virally associated lesion that is distinct from, but has some features in common with, CLE, which could hinder accurate diagnosis if the lesion distribution is not fully described to include symmetry and if the inclusion bodies are not noted in the histopathology. A variety of factors, such as stress, underlying disease, and ultraviolet radiation, may lead to activation of the virus and clinically relevant lesions. Because the lesions were often around the nostrils and sometimes around the eye, we speculate the infection may be associated with nasal and ocular secretions in horses shedding virus from those sites.2,16,18 Although 3 of the horses had a history of ocular discharge, none of the horses had known signs of respiratory disease prior to or during the onset of the skin lesions. EHV-5 is known to infect and replicate in B and T lymphocytes and can spread to neighboring lymphocytes via cell-to cell transfer. It is thought that EHV-5-infected lymphocytes may transfer infection to epithelial cells. 22 These observations correlate with the co-localization of the lymphocytic inflammatory infiltrate together with virally infected keratinocytes in the outer layers of the epidermis of the nostrils. Given the specific facial distribution of the skin lesions, EHV-5 might be latent in the trigeminal ganglia, as reported in mice infected with murine gammaherpesvirus-68. 21 The dermatitis appears to be self-limiting but can have a long or chronically relapsing course in some cases.
Interestingly, horse 3 in our series developed EMPF more than 3 years after the diagnosis of EHV-5-associated facial interface dermatitis. This might have been a progression of the EHV-5 infection in a susceptible host or a coincidence. Genomic variability among EHV-5 isolates has been documented. 1 Such testing would have aided in ruling in or out a relationship between the EHV-5-associated facial interface dermatitis and the subsequent onset of EMPF in this horse; however, such testing was not performed.
Clinical follow-up information was not obtained from horses 8–12. The authors conclude that given the clinical presentation and lesion distribution (Table 1), as well as the histopathologic changes, these horses had CLE, systemic lupus erythematosus, or an adverse cutaneous drug reaction. Although a chronic viral infection with EHV-5 is possible, the lack of inclusion bodies and lack of PCR and ISH detection of EHV-5 makes that unlikely. Four of 7 EHV-5-positive horses had lesions for 1 year or more and they had viral inclusions as well as PCR and ISH detection of virus. Furthermore, only one of these cases had a similar lesion distribution to the EHV-5-positive horses, and this horse had clinical pruritus which was not a feature in the EHV-5-positive cases.
In conclusion, we report a specific clinical and histopathologic presentation of facial interface dermatitis associated with EHV-5. EHV-5 infection should be considered in cases of facial dermatitis in horses particularly with an asymmetric pattern of non-pruritic, non-painful, crusting, and alopecic lesions around the muzzle and nostrils. The main limitation of our study is the small sample size. Testing a larger number of normal control skin samples and a wider body distribution would give us a better indication of the normal distribution of EHV-5 in the skin. No proven effective treatment was noted in the follow-up information; in fact, lesions spontaneously resolved in 3 cases, and improved but waxed and waned in 2 others without treatment. Only 1 of 4 horses for which 3 or more years of clinical follow-up was available developed EMPF. It remains to be seen whether other horses will develop EMPF or more severe complications associated with EHV-5.
Footnotes
Acknowledgements
We thank Tom Bargar and Nicholas Conoan of the Electron Microscopy Core Facility (EMCF) at the University of Nebraska Medical Center for their technical assistance. The EMCF is supported by state funds from the Nebraska Research Initiative (NRI) and the University of Nebraska Foundation, and institutionally by the Office of the Vice Chancellor for Research. We also thank Dr Courtney Martin for information, photos, and follow-up on case 3; Dr Kathy Connell for information and follow-up on case 4; Dr Wendy Krebs for information, photos, and follow-up on case 5; Dr Laura Javsicas for photos and follow-up on case 7, Dr Dinah Skorich for clinical follow-up photos of case 7; and the staff at New York Equine for the photos of case 1. We would also like to extend a special thanks to Dr Robert Bildfell, who was the primary pathologist on one of the cases from Oregon, and Dr Danny W. Scott for his careful and insightful review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
