Abstract
A constraint on understanding the pathogenesis of malignant catarrhal fever (MCF) is the limited number of tools to localize infected cells. The amount of detectable virus, visualized in the past either by immunohistochemistry or in situ hybridization (ISH), has been modest in fixed or frozen tissues. This complicates our understanding of the widespread lymphoid proliferation, epithelial necrosis/apoptosis, and arteritis-phlebitis that characterize MCF. In this work, we developed a probe-based in situ hybridization assay targeting 2 ovine herpesvirus 2 (OvHV-2) genes, as well as their respective transcripts, in formalin-fixed tissues. Using this approach, OvHV-2 nucleic acids were detected in lymphocytes in MCF-affected animals following both natural infection (American bison and domestic cattle) and experimental infection (American bison, rabbits, and pigs). The probe did not cross-react with 4 closely related gammaherpesviruses that also cause MCF: alcelaphine herpesvirus 1, alcelaphine herpesvirus 2, caprine herpesvirus 2, and ibex–MCF virus (MCFV). No signal was detected in control tissues negative for OvHV-2. ISH will be of value in analyzing the natural progression of OvHV-2 infection in time-course studies following experimental infection and in addressing the pathogenesis of MCF.
Keywords
Ovine herpesvirus 2 (OvHV-2) is 1 of 6 known gammaherpesviruses of ungulates that can cause malignant catarrhal fever (MCF). The other 5 MCF-inducing viruses are alcelaphine herpesviruses 1 and 2 (AlHV-1, AlHV-2), caprine herpesviruses 2 and 3 (CpHV-2, CpHV-3, or MCF virus [MCFV]–white-tailed deer), and ibex-MCFV. 20 Domestic sheep are the adapted host species of OvHV-2 and shed the virus periodically and asymptomatically. Nonadapted ungulate species, such as domestic cattle and American bison, can die from severe, systemic OvHV-2–associated disease when the virus is transmitted from infected sheep. Sheep-associated MCF is an important form of MCF worldwide and has a significant economic impact on highly susceptible species. 14,20
Several obstacles block understanding of the pathogenesis of MCF. Past animal studies were mainly based on AlHV-1 because it can be propagated in cell culture as first shown in the pioneering studies of Walter Plowright. 26 However, the biology of AlHV-1 differs in important ways from several other MCFVs, particularly OvHV-2, including the time of infection in adapted host species: AIHV-1 occurs perinatally in wildebeest, 14 whereas OvHV-2 occurs largely after weaning in lambs. 16 In contrast to AlHV-1, OvHV-2 has never been isolated in cell culture. However, OvHV-2–rich secretions can be harvested from the nasal passages of sheep during episodes of natural shedding, 15 and pooled secretions from multiple lambs have been used for experimental infection of susceptible species, including domestic cattle, bison, rabbits, and pigs. 12,15,22,29
Another constraint is the difficulty in correlating the presence of OvHV-2 in tissues with pathogenesis. Plowright’s early work and numerous subsequent studies established that lymphoid hyperplasia in multiple tissues is a hallmark of MCF. 20,25 The predominant cell in inflammatory lesions of MCF is a large (10–15 μm) lymphocyte with a vesicular nucleus, variously referred to as a “lymphoblast,” “immunoblast,” or “large granular lymphocyte,” 25,28 with activation and expansion of CD8+ T cells being a consistent feature. 2,8,9,10,18,23 Studies that used in situ hybridization (ISH) or immunofluorescence to detect OvHV-2 in tissues found either a small proportion of infected cells or a limited virus distribution. 17,19,27,31 The inconsistent detection of viral particles in infected tissues led to the hypothesis that few infected cells trigger hyperplasia and/or dysregulation of many uninfected lymphocytes. 2,4,24 In contrast, severity of MCF lesions has been shown to correspond well with both increased levels of OvHV-2 DNA in tissues and expression of viral transcripts that encode a major viral capsid protein, 5,6 suggesting that OvHV-2 replication and/or its viral products are directly related to the development of lesions in MCF. For a clearer picture of OvHV-2 cell tropism and pathogenesis of MCF at various time points during infection, one needs to be able to track the virus within tissues and correlate its distribution with disease progression.
To improve our understanding of MCF pathogenesis, an assay able to reliably identify OvHV-2 within lesions is necessary. The goal in this study was to develop an ISH assay to detect OvHV-2 nucleic acids in formalin-fixed tissues of infected animals.
Materials and Methods
Cases
Tissues from 15 cases (cases 1–15) were used for this study (Table 1). Five animals with experimentally induced MCF by intranasal nebulization of OvHV-2, as previously described, included 2 pigs (cases 1 and 2), 15 2 bison (cases 3 and 4), 5 and a rabbit (case 5). 6 Natural cases of MCF were confirmed by polymerase chain reaction (PCR) 3 for OvHV-2 infection included a bull from Wyoming (case 6) and dairy cows from Davis, CA (cases 7–10). To test the specificity of the OvHV-2 probe, 4 animals with clinical MCF caused by MCFVs other than OvHV-2 (cases 11–14) were included: a rabbit with MCF experimentally induced by AlHV-1 infection (case 11), 7 a bison with MCF experimentally induced by AlHV-2 infection (case 12), 30 a duiker with naturally occurring ibex-associated MCF (case 13), and a moose with naturally occurring CpHV-2–associated MCF (case 14). Select tissues from an MCFV-uninfected cow (case 15) were also tested. All animals have been confirmed to be positive or negative for MCFV by qPCR specific for OvHV-2 3,33 (cases 1–10), or AlHV-1 32 (case 11), AlHV-2 PCR 30 (case 12), or a consensus herpesvirus PCR, 34 followed by sequencing when positive (cases 13–15).
OvHV-2 In Situ Hybridization Results in Tissues of Animals With MCF, Including Comparison to Viral Load as Measured by Quantitative PCR.
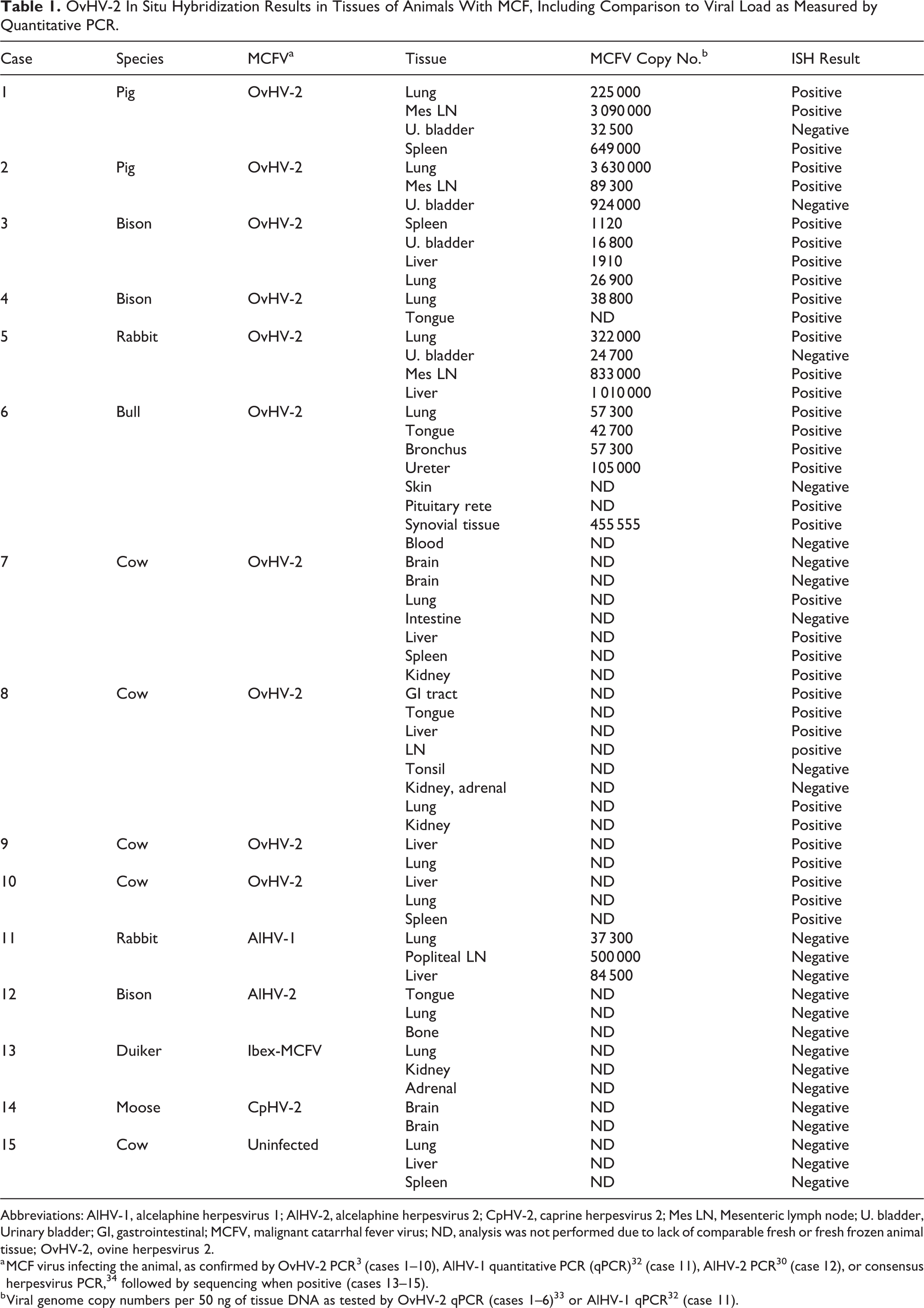
Abbreviations: AlHV-1, alcelaphine herpesvirus 1; AlHV-2, alcelaphine herpesvirus 2; CpHV-2, caprine herpesvirus 2; Mes LN, Mesenteric lymph node; U. bladder, Urinary bladder; GI, gastrointestinal; MCFV, malignant catarrhal fever virus; ND, analysis was not performed due to lack of comparable fresh or fresh frozen animal tissue; OvHV-2, ovine herpesvirus 2.
qPCR
Viral genome copies in tissues of bison, rabbits, and pigs experimentally infected with OvHV-2 (cases 1–5) or AlHV-1 (case 11) were measured by qPCR specific for OvHV-2 or AlHV-1, 32,33 respectively, and were reported previously. 5,6,7,15 For quantification of OvHV-2 genome copies in tissues of a naturally infected bull (case 6), total DNA was extracted and purified from fresh tissues using the FastDNA Kit (MP Biomedicals, Burlingame, CA, USA) and DNeasy Power Clean Pro Cleanup (Qiagen, Valencia, CA, USA), respectively, as per the manufacturers’ recommendations. Total DNA concentration was quantified using a fluorometer, Qubit (ThermoFisher Scientific, Sacramento, CA, USA). The number of OvHV-2 genome copies was quantified in tissue by qPCR as previously described 33 but with the following modifications: TaqMan Fast Advanced Master Mix (Applied Biosystems, Foster City, CA, USA) was used instead of the previous master mix reagent, the qPCR was performed in a CFX Detection System (Bio-Rad), and data were analyzed using CFX Manager Software (Bio-Rad, Hercules, CA, USA). Results are expressed as viral genome copies per 50 ng total DNA.
ISH
Tissues were selected on the basis of good tissue preservation and short fixation times: tissues were fixed for no longer than 6 days. Colorimetric ISH was performed manually on 5-μm sections of formalin-fixed, paraffin-embedded tissue on Superfrost Plus slides (Fisher Scientific, Pittsburgh, PA) using the RNAscope 2.5 Red assay kit (cat. 322360, Advanced Cell Diagnostics, Hayward, CA) as per the manufacturers’ recommendations. Deparaffinized and dehydrated tissue sections were incubated in target retrieval buffer (ACD) maintained at 98°C to 102°C using a hot plate for 15 minutes. Slides were then treated with protease plus reagent (ACD) for 30 minutes at 40°C. We designed V-OvHv2-orf25-orf50 ACD (cat. 501091) as 30ZZ paired probe sets targeting regions 49813–50552 of open reading frame (ORF) 25 and 76874–77614 of ORF50 (excluding the intronic region) in OvHV-2 (GenBank #NC_007646). Three negative controls were used to assess the specificity of the signal for OvHV-2. First, replicate sections of all slides in the study were tested with either an unrelated, GC content-matched probe or a probe to the bacterial gene diaminopimelate B (dapB). Second, the specificity of the OvHV-2 probe compared to related herpesviruses was tested against tissues infected with 4 other MCFVs (AlHV-1, AlHV-2, ibex associated, and CpHV-2; Table 1). Third, select tissues from an MCFV-uninfected animal (case 15) were tested with the OvHV-2 probe (Table 1). Slides were counterstained with hematoxylin and coverslipped using EcoMount (BioCare Medical, Concord, CA). Given that herpesviruses are double stranded, the probe could hybridize with both transcript and viral genomic DNA. Slides were digitized using an Olympus (Center Valley, PA, USA) VS120 scanner and a 40× objective with brightfield illumination.
Immunohistochemistry
Immunohistochemistry was performed using a peroxidase-conjugated immune-polymer method with primary antibody to CD3 (clone CD3-12 IgG1 [rat]) and a secondary antibody (rat-HRP-polymer, 1 step [mouse absorbed], 1:100 dilution; BioCare Medical). Antigen retrieval was performed by microwave for 20 minutes at 98°C in citrate buffer, pH 6. Chromogen development was performed with a NovaRed Peroxidase substrate kit (cat. SK-4800; Vector Laboratories, Burlingame, CA, USA). Endogenous peroxidase activity was quenched by immersing the sections in 3% hydrogen peroxide in methanol for 15 minutes. Serial sections of individual blocks were stained separately by hematoxylin and eosin (HE), CD3 immunohistochemistry (IHC), and ISH for OvHV-2 viral nucleic acid to help establish which cells were detected by ISH.
Results
Analysis was performed on a total of 44 tissues from 10 animals with experimentally induced (cases 1–5) and naturally occurring (cases 6–10) OvHV-2–associated MCF (Table 1). The diagnosis of MCF was established in each animal on the basis of typical clinical signs, characteristic gross and histological lesions, and confirmation by conventional PCR (data not shown). Tissue types were chosen based on several criteria, including quality of fixation (in formalin <6 days) and comparison of affected tissues among and between experimentally or naturally infected species. The tissues selected represented a spectrum of lesions typical of MCF, including lymphoproliferation, inflammation, arteritis-phlebitis, and ulceration.
qPCR
The experimentally infected cases were previously described (OvHV-2 infections in cases 1–5, 5,6,15 AlHV-1 infection in case No. 11, 7 and AlHV-2 infection in case 12 30 ). Viral loads (i.e., genome copy number per 50 ng total DNA) were estimated by qPCR (Table 1). Viral loads were calculated in this study for 1 naturally occurring case of OvHV-2–associated MCF (case 6); the viral loads in the tissues tested from this bull ranged from 42 700 to 455 500 OvHV-2 genome copies per 50 ng of tissue DNA, which is comparable to previously published data in various host species. 5,6,15
ISH
All animals infected with OvHV-2 were positive by ISH in most (35/45) tissues tested (Table 1). By extrapolation from previous studies, including our own, CD8+ T cells predominate in lesions in MCF and correspond to large lymphocytes seen in HE-stained preparations. Double IHC-ISH labeling was unsuccessful, so it was not possible to establish unequivocally whether ISH labeled T cells or other lymphocyte subsets. 25 Regardless, in all cases, there were a predominance of small lymphocytes, some larger (presumably immature) lymphocytes, and fewer plasma cells and histiocytes. OvHV-2 ISH-labeled cells were leukocytes and, based on nuclear morphology, were lymphocytes. Between 30% and 50% of lymphocytes were OvHV-2 ISH positive in regions of arteritis (e.g. Figs. 1b,d and 2c–d). A high proportion of lymphocytes in these same regions was CD3 positive (Fig. 1c).
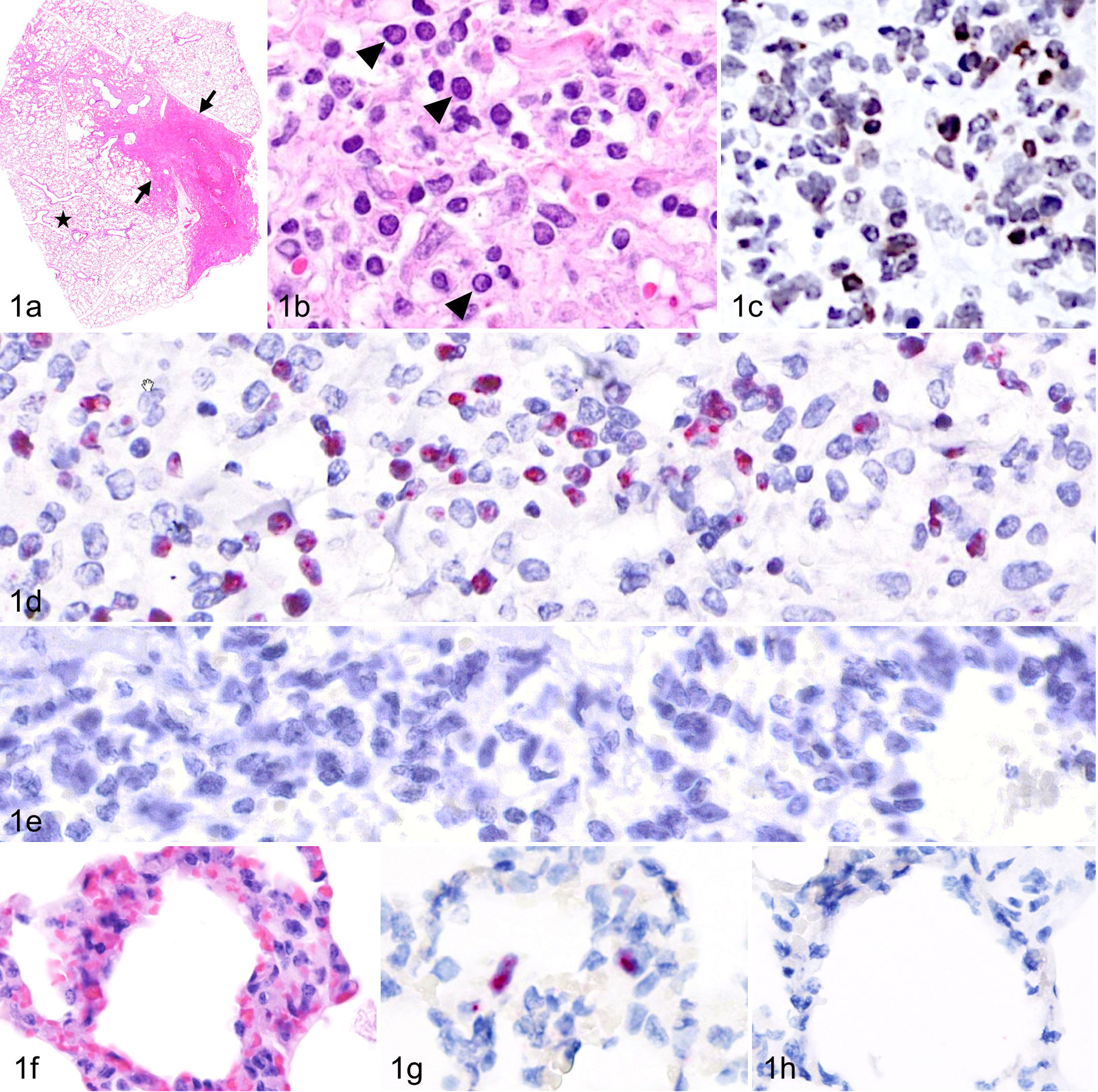
Malignant catarrhal fever, lung, American bison, case 3. (a) There is patchy consolidation, atelectasis, and inflammation. Hematoxylin and eosin (HE). (b) In heavily consolidated regions (between arrows in Fig. 1a), there is a predominance of leukocytes, including leukocytes (arrowheads). HE. (c) CD3-positive cells (brown) are abundant and exhibit anisokaryosis. Immunohistochemistry. (d) OvHV-2 nucleic acid (red) is present in nuclei of cells in regions of inflammation. In situ hybridization (ISH). (e) There is no detectable hybridization using replicate sections with an irrelevant ISH control probe (bacterial gene DapB). (f) In nonconsolidated regions (starred region in Fig. 1a), there is an increase in cellularity of the alveolar walls. HE. (g) Scattered cells contain OvHV-2 nucleic acid. ISH. (h) There is no detectable hybridization using the control, irrelevant ISH probe (DapB).
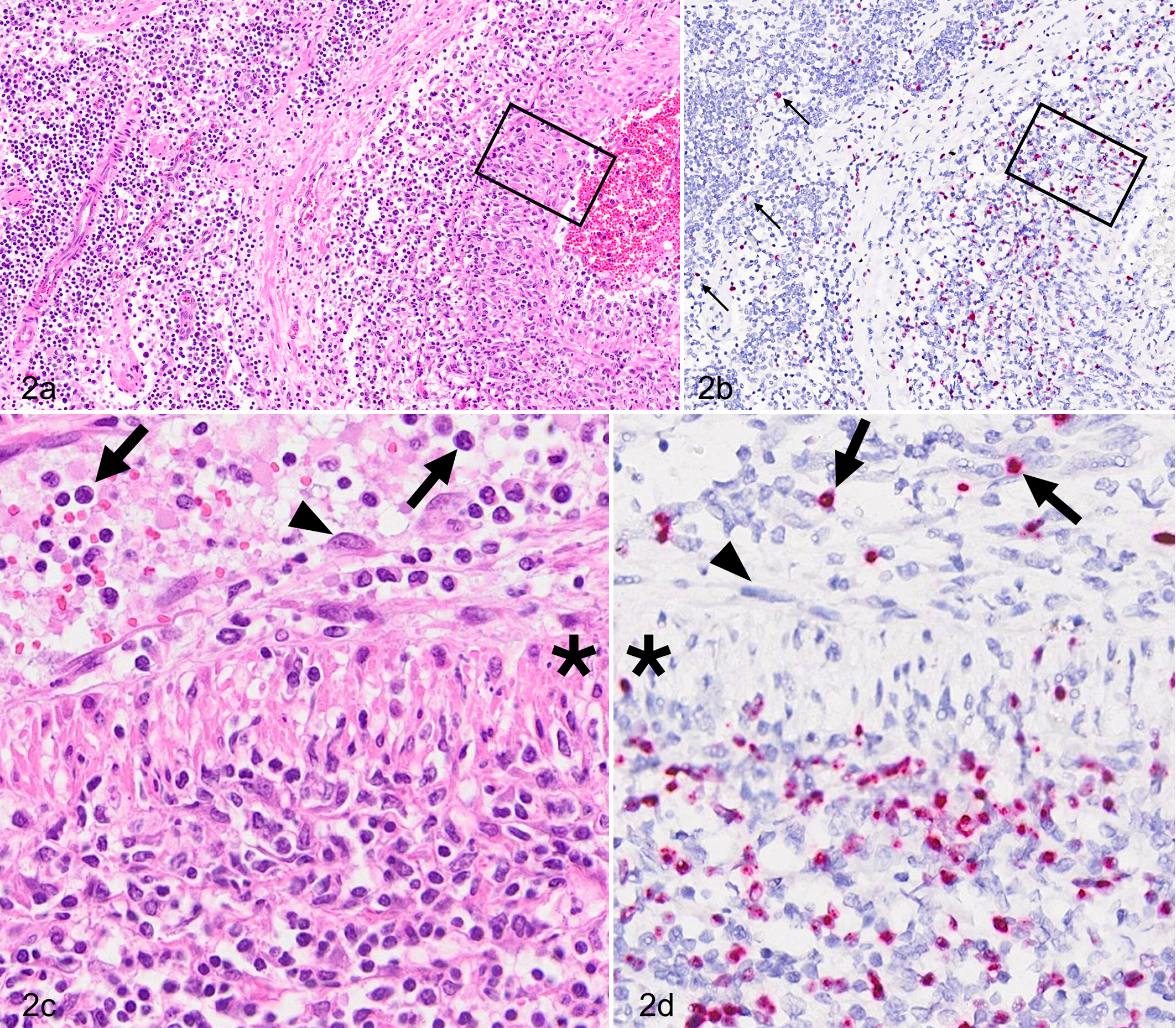
Malignant catarrhal fever, lymph node with intranodal artery, cow, case 8. (a) There is transmural arteritis with segmental disruption of the arterial wall. Hematoxylin and eosin (HE). (b) OvHV-2–positive cells are present within the tunica media and scattered throughout perivascular parenchyma of the lymph node (arrows). In situ hybridization (ISH). (c, d) Higher magnifications of the boxed insets in Fig. 2a and b. Asterisks show arterial smooth muscle. (c) Lymphocytes are present in circulation (arrows). The endothelium (arrowhead) is lifted above the internal elastic lamina (arrowhead). There is dense transmural infiltration by mononuclear cells with predominance of lymphocytes. HE. (d) OvHV-2 ISH-positive nuclei are present within the arterial lumen (arrows) and throughout the arterial wall. Endothelial cells (arrowhead) are devoid of signal. OvHV-2 probe.
The lung was inflamed in all species tested, where OvHV-2 nucleic acid–specific signal was present in the nuclei of lymphocytes in areas of consolidation, pneumonia, vasculitis, and peribronchiolitis (Fig. 1a–e). No signal, in lung or other tissue, was identifiable in epithelium, endothelium, or smooth muscle. CD3-positive cells included both large and small lymphocytes (Fig. 1b–c). In less affected regions of lung, scattered, individual lymphocytes were ISH positive (Fig. 1f–h). Inflamed arteries, regardless of tissue type, shared similar distribution and character of inflammation and OvHV-2 nucleic acid (Figs. 2, 3). Vasculitis typically involved medium-caliber arteries, with a combination of acute inflammation and segmental necrosis of the tunica media, hypertrophy of endothelium, and lymphocytic periarteritis. 21 ISH-positive cells were abundant in the adventitia and in perivascular spaces, intramurally, and in the lumen of medium-caliber arteries (Fig. 2a–d). Some ISH-positive intravascular (circulating) lymphocytes were marginated near or abutting endothelium (Fig. 2c,d). In the liver, lymphocytes (20–50%) in portal regions were ISH positive (Fig. 3). Mucosal ulcers of the tongue, rumen, bladder, and esophagus had scant positive cells adjacent to ulcerated surfaces, or the cells were restricted to inflamed, deep arteries. One bull (case 6) had polyarthritis and acute fibrinonecrotic arthritis (Figs. 4 and 5) with a high viral load in synovial tissue (Table 1). OvHV-2 nucleic acid was detected by ISH in inflamed and necrotic subsynovial tissue (Fig. 5b,c), but no signal was present within synoviocytes (Fig. 5b, arrows).
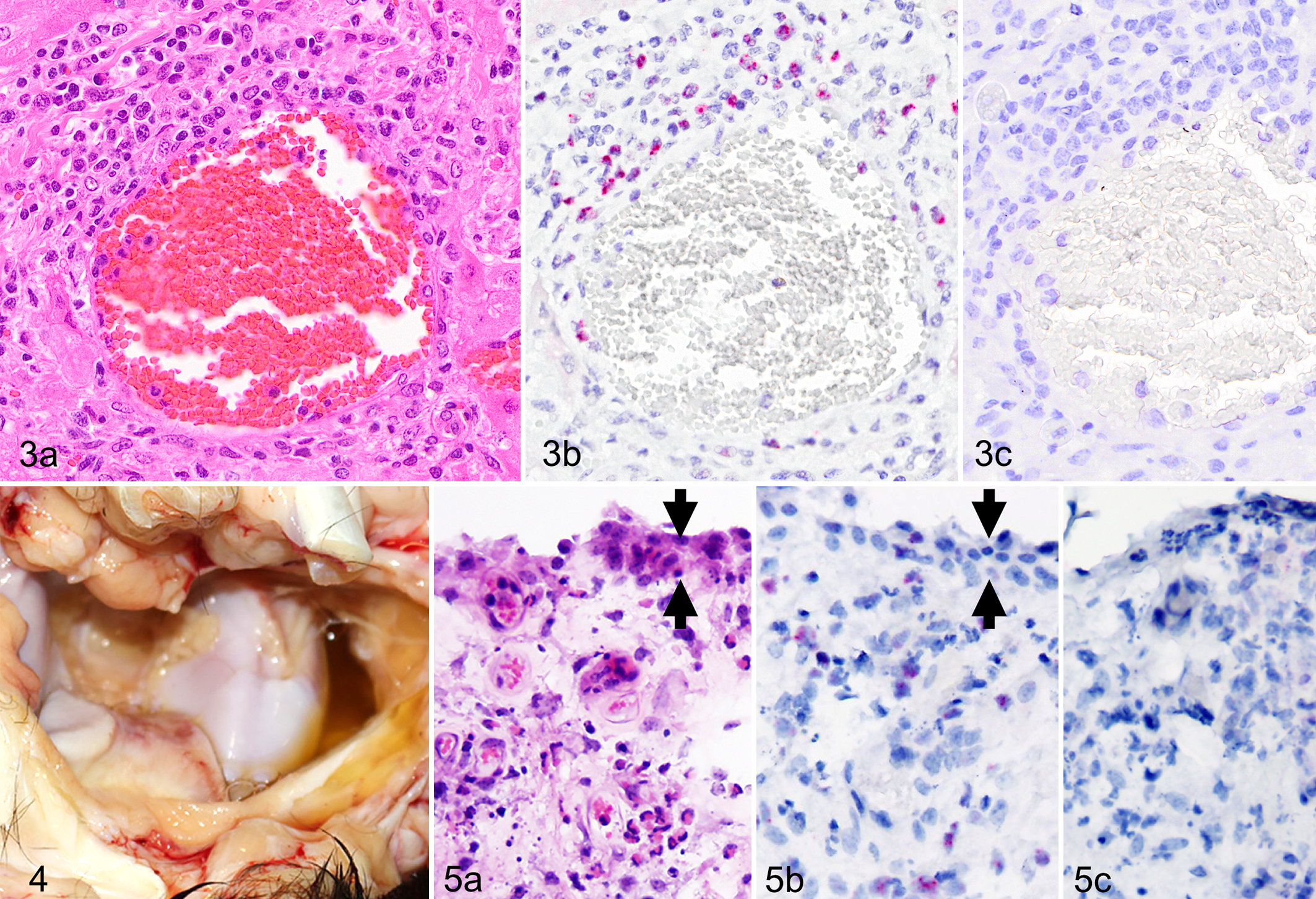
Sequential sections of all tissues probed with an unrelated, GC-matched probe were consistently negative (examples are Figs. 1e,h, 3c, 5c). Probe specificity was further tested by analysis of tissues from animals infected with 4 related MCFVs (AlHV-1, AlHV-2, CpHV-2, and ibex-MCFV). The character of histologic lesions and quantity of inflammation in these cases were similar to those in OvHV-2–positive animals (Table 1). No hybridization was detected using the OvHV-2 probe in any tissues from these cases (n = 14, Table 1), regardless of viral load (eg, case No. 11 with an AlHV-1 genome copy number of 37 300 to 500 000 per 50 ng DNA; Table 1). Sections of the liver, lung, and spleen from a cow with severe chronic, lymphocytic, plasmacytic, and neutrophilic bronchopneumonia (case 15) were uniformly negative by ISH for OvHV-2.
Discussion
The OvHV-2 ISH probe assessed in this article was highly specific. Tissues infected with other MCFVs (AIHV-1, AIHV-2, ibex-MCFV, and CpHV-2) were negative by this method. This is consistent with the use of target regions for probe hybridization (ORFs 25 and 50), which share only ∼73% (ORF25) and ∼43% (ORF50) identity with OvHV-2. Positive ISH was obtained on most of the tissues examined from OvHV-2–infected animals. In few cases, ISH failed to detect an OvHV-2 signal, even in tissues with high OvHV-2 genome copies detected by qPCR. This was likely related to the multifocal characteristic of viral distribution in tissues and the fact that different sections of the tissue were necessarily examined by each assay. In addition, ISH may be less useful for mucosal tissue because the ISH signal near ulcerated mucosa is generally limited in distribution and amount. To overcome potential false negatives when using ISH for diagnostic purposes, we suggest testing 2 or more formalin-fixed, paraffin-embedded lymph node, liver, or spleen samples. This is in contrast to PCR, where it is recommended to submit 2 fresh or cryopreserved samples: one of lymphoid tissue and one of lesional (ulcerated) mucosal tissue. Both retrospective and prospective cases can be evaluated by ISH for the presence of OvHV-2, provided fixation times in formalin are short (<7 days).
Recent advances in ISH technology have substantially improved detection of viruses in tissue, with the result that even low viral copy numbers can now be localized within tissues, whereas in the past, this was not predictable. 1,13 OvHV-2 was labeled by ISH in tissues of all animals with both experimentally induced and naturally acquired MCF due to OvHV-2. OvHV-2 nucleic acid was detectable predominantly in the nuclei of lymphocytes in affected tissues, as well as in circulating lymphocytes. This is consistent with earlier studies demonstrating susceptibility of T cells to infection. 8,27 It is possible that additional types of lymphocytes are infected, but determining this requires additional evaluation using a wider range of lymphocyte markers.
It has been previously shown that in experimentally infected animals, lesion severity is correlated with viral DNA copy number. 5,6 Here we demonstrate that ISH signals are also positively associated with lesion severity. However, because lesions in MCF are multifocal and vary from tissue to tissue, assumptions that qPCR findings predict the intensity of ISH signal should be treated with caution until additional comparative studies are performed. Interestingly, although bison are approximately 1000-fold more susceptible to MCF than are cattle, 11,29 they have a consistently lower viral genome copy number. 5 Regardless, all tissue samples examined from OvHV-2 experimentally infected bison were positive by ISH. Irregular distribution of lesions in this disease weakens comparisons between methods of detection and generally raises the possibility of false-negative results.
OvHV-2 has been challenging to detect by traditional methods, leading to a previous assumption that the mechanism of disease is indirect. Recent experimental work indicates that the proportion of infected lymphocytes is high, 8,27 which these data corroborate. The lymphocytes containing viral nucleic acid, as detected by ISH, suggest the possibility of direct viral-induced inflammation and damage.
Studies on the pathogenesis of MCF center on a fascinating juxtaposition between the clinically innocuous, persistent virus in a host species and fatal systemic disease following cross-species transmission. Even though considerable progress has been made over the years in elucidating the pathogenesis of MCF, the lack of in situ assays to identify MCFVs within lesions has restrained its complete understanding. The OvHV-2 ISH described here is not only a useful tool for investigations of the pathogenesis of MCF but also potentially valuable for diagnosis.
Footnotes
Acknowledgements
We thank Shirley Elias (Washington State University) for technical assistance and Dr Ryan Jennings (Ohio State University) for providing a tissue block from a moose with goat-associated MCF. Support funding to the Li laboratory for this project is from USDA-ARS CWU 2029-3200-037-00D. In the Pesavento laboratory, the study was supported by Boehringer Ingelheim Vetmedica and the Bernice Barbour Foundation for research in naturally occurring infectious disease. Research support for these studies was also provided by Animal Health and Hatch formula funds (University of Wyoming; WYO-568-16 and WYO-564-16).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
