Abstract
Progressive lung fibrosis in humans, typified by idiopathic pulmonary fibrosis (IPF), is a serious cause of morbidity and mortality in people. Similar diseases have been described in dogs, cats, and horses. The cause and pathogenesis of such diseases in all species is poorly understood. There is growing evidence in human medicine that IPF is a manifestation of abnormal wound repair in response to epithelial injury. Because viruses can contribute to epithelial injury, there is increasing interest in a possible role of viruses, particularly gammaherpesviruses, in the pathogenesis of pulmonary fibrosis. This review provides background information on progressive fibrosing lung disease in human and veterinary medicine and summarizes the evidence for an association between gammaherpesvirus infection and pulmonary fibrosis, especially Epstein-Barr virus in human pulmonary fibrosis, and equine herpesvirus 5 in equine multinodular pulmonary fibrosis. Data derived from experimental lung infection in mice with the gammaherpesvirus murine herpesvirus are presented, emphasizing the host and viral factors that may contribute to lung fibrosis. The experimental data are considered in the context of the pathogenesis of naturally occurring pulmonary fibrosis in humans and horses.
Keywords
In
In humans, chronic progressive fibrosing diseases are an important cause of morbidity and mortality, as well as a considerable economic cost to society. Most such conditions affect the lung, liver, kidneys, and heart. The spectrum of fibrosing diseases in humans is large compared with those recognized in nonhuman animals and doubtless reflective of people’s longevity and exposure to many potential causes of fibrosis as well as the spectrum of exposures to potential causes of fibrosis through their occupation (eg, various dusts causing pneumonconioses) or recreation (eg, alcohol causing cirrhosis).
Pulmonary Fibrosis in Humans
The spectrum and classification of progressive fibrosing pulmonary disease in humans is varied. The proper identification of such diseases is important, as the identification of the subtype often indicates the underlying cause and the expected outcome. The most recent comprehensive classification of these diseases is based on a combination of clinical and imaging data along with histopathology. 2 This classification scheme separates interstitial lung diseases into those with known causes (ie, smoking, environmentally induced lung disease, and a variety of genetic diseases) from a large group of idiopathic interstitial lung diseases. 2 Idiopathic pulmonary fibrosis (IPF) is recognized as the most clinically severe form of idiopathic diffuse parenchymal lung diseases of humans, having an average survival time of 5 years following diagnosis. 1
The pathogenesis of IPF is very poorly understood. Given the relative paucity of inflammation within the IPF lung and the failure to control the progression of disease with aggressive anti-inflammatory therapy, there has been considerable debate as to whether IPF is primarily an inflammatory disease. 28,76 The current prevailing view is that IPF is not primarily an inflammatory disease but instead is the result of abnormal wound healing in the lung in response to alveolar epithelial injury from a variety of potential causes. Viruses, in particular gammaherpesviruses (γ-HV), have received considerable attention of late as being one such cause/factor that is involved in the pathogenesis of IPF. 21,47,70,85
Pulmonary Fibrosis in Domestic Animals
Within the past 15 years, progressive fibrosing lung diseases have been recognized in the domestic dog, cat, and horse. 18,19,50,95,96,99 In these species, fibrosing lung diseases share important clinical similarities to the human disease, especially in their adult onset, progressive clinical course, and generally poor response to therapy. Unlike in human medicine, there has been no effort in veterinary medicine to classify progressive fibrosing lung diseases into subtypes and little investigation into potential causes of these diseases. The cause of the canine and feline diseases remains unknown, while a fibrosing lung disease in horses—equine multinodular pulmonary fibrosis (EMPF)—has been associated with pulmonary infection with the γ-HV equine herpesvirus 5 (EHV-5). 7,50,69,72,91,94
In the initial description of EMPF, EHV-5 was detected in 100% of affected animals. 91 This association between a γ-HV and interstitial lung disease followed an earlier description of interstitial pneumonia in donkeys associated with lung infections with the γ-HV asinine herpesviruses (AHV) 4 and 5. 44 The correlation between lung infection with EHV-5 and the development of a progressive fibrosing lung disease provides important evidence that γ-HV may be capable of playing a part in the development of such lung diseases in humans.
Myofibroblasts and Th2 Inflammation as Effectors of Pulmonary Fibrosis
It is beyond the scope of this review to cover in detail the complex biology of tissue healing and fibrosis; therefore, the reader is referred to several of many excellent review articles available on this topic. 90,95,97 While many factors determine whether tissues heal normally or move into a state of progressive fibrosis, for the purposes of this review, the myofibroblast and inflammation driven by CD4+ T-helper 2 lymphocytes will be considered in very brief detail as there are data available to suggest that both are important in the pathogenesis of lung fibrosis in the context of γ-HV infection. 52,55
Myofibroblasts are specialized mesenchymal cells that express cytoplasmic α–smooth muscle actin in well-organized stress fibers. These cells play an important role in wound healing, where they contribute to the production of extracellular matrix and cytokines—especially transforming growth factor (TGF)–β—which are important in the resolution of inflammation. 23 In organs that undergo progressive fibrosis, myofibroblasts play a crucial role in the pathogenesis of disease. Myofibroblasts can originate in tissues from a variety of sources, including resident mesenchymal cells, epithelial-mesenchymal transition, 41,42,93 endothelial-mesenchymal transition, 101 and circulating fibrocytes, which are specialized cells expressing CD45 and type I collagen that originate from a pool of bone marrow–derived mesenchymal stem cells. 13,16,18,36,62 Myofibroblasts stimulate progressive tissue fibrosis primarily through their profligate production of extracellular matrix components and cytokines that induce additional matrix production by neighboring cells. 33 –35
Fibrosis, whether in the course of wound healing or as part of progressive tissue fibrosis, is facilitated by Th2-polarized inflammation. CD4+ lymphocyte-driven Th2 inflammation is primarily directed against extracellular pathogens and characterized by the production of interleukin (IL)–4, IL-5, IL-9, IL-13, and IL-25. 60 This is in contrast to Th1 CD4+ lymphocyte-driven inflammation, which is directed against intracellular pathogens and is characterized by the production of interferon (IFN)–γ. In experimental models of lung and liver fibrosis, less fibrosis develops during Th1-driven inflammation than Th2, and this is due in part to the antifibrotic actions of IFN-γ and decreased production of Th2-associated cytokines such as TGF-β. 96
Gammaherpesviruses
Gammaherpesvirinae is 1 of 3 subfamilies in the Herpesviridae family—a large family of DNA viruses within the order Herpesvirales whose members infect mammals, birds, and reptiles. 51 There are 4 genera within the Gammaherpesvirinae subfamily: Lymphocryptovirus, Rhadinovirus, Macavirus, and Percavirus. 17 Unlike members of the other 2 subfamilies of Herpesviridae–(α and β), the γ-HV usually forgo lytic infection upon cell entry and instead establish latency within the host cell nucleus as circular episomal DNA. 3,71 Because of their propensity to rapidly establish latent infections, γ-HV usually do not result in significant clinical signs in the natural host species during the primary infection. In contrast, γ-HV infections in related nonhost species may result in more severe disease, as for example the development of malignant catarrhal fever in nonhost ungulate species infected with alcelaphine herpesvirus 1 or ovine herpesvirus 2. 3 This concept of differing responses to γ-HV infections in host vs nonhost species is important as we consider data in humans infected with Epstein-Barr virus (EBV), horses infected with EHV-5, and mice infected with murine gammaherpesvirus 68 (MHV-68). While humans and horses are the natural host of EBV and EHV-5 respectively, 5,100 the natural hosts of MHV-68 are the wood mouse (Apodemus sylvaticus) and bank vole (Myodes glareolus), not the laboratory mouse. 9,22 This is important when we consider the experimental data in mice infected with MHV-68 in the context of the larger role that γ-HV might play in the development of lung fibrosis in the various species.
Based on data largely derived from studies of MHV-68 in laboratory mice, infection with γ-HV initially results in lytic infection of respiratory epithelial cells of the nasal cavity and lung, followed by establishment of latency within lymphocytes, lung epithelial cells, macrophages, and dendritic cells. 8,24,58,74,77 In the natural host, MHV-68 also infects and replicates within the lung, but to a lower level than in BALB/c mice, and establishes latency within induced bronchus-associated lymphoid tissue (BALT) and spleen. 39 The importance of lung infection in both the lytic and latent stages of γ-HV infection, and how that might relate to development of pulmonary fibrosis, is discussed in greater detail below.
Gammaherpesviruses and Pulmonary Fibrosis in Humans
Viral infections have been suggested as one of the factors that may play a role in the underlying epithelial injury that perpetuates processes leading to progressive lung fibrosis in humans. 47,85 A variety of viruses, including hepatitis C virus, 6,83 adenoviruses, 45 β-herpesviruses (cytomegalovirus), 19 and γ-HV (EBV), 43,73,80 have been investigated in association with the development of IPF in humans. Those investigations mostly associate serologic evidence of prior viral infection or detection of viral nucleic acid within lung tissues of patients with IPF.
Lung tissue samples from 33 patients with IPF and 25 non-IPF control lungs were analyzed using a polymerase chain reaction (PCR)–based approach. 80 Using general herpesvirus primer sets, as well as virus-specific primers for 4 human herpesviruses—cytomegalovirus, EBV, human herpesvirus (HHV)–7, and HHV-8—they detected herpesvirus nucleic acid in the lungs of nearly 100% of patients with IPF compared with less than 40% of control lungs, with EBV being the most frequently detected virus. 80 More recently, analysis of lung samples from 55 patients with IPF and 41 control lungs detected herpesvirus nucleic acid more commonly within the lungs of patients with IPF; the presence of the virus was associated with poorer performance on a 6-minute walk test. 14 While these studies provide evidence for an association between herpesvirus infection and the development of progressive lung fibrosis in humans, other studies provide evidence that such infections are not common in IPF. 89,99
EBV infection is ubiquitous in people, with more than 95% of adults seropositive and harboring latent virus within B cells, thus making it difficult to ascertain the relationship between lung infection with the virus and the development of pulmonary fibrosis. Testing the pathogenicity of EBV in rodent models of lung fibrosis is difficult because γ-HV infections are often not easily established outside the natural host species; therefore, identification of γ-HV infection in association with the development of lung fibrosis in other species could be important in elucidating the relationship between such infections and disease.
Gammaherpesviruses and Pulmonary Fibrosis in Domestic Animals
Several γ-HV are known to infect domestic species. Members of the Macavirus genus include alcelaphine herpesvirus 1 and ovine herpesvirus 2, which cause malignant catarrhal fever and sheep-associated malignant catarrhal fever, respectively, 17 as well as 3 nonpathogenic porcine lymphotropic γ-HV: suid herpesviruses 3, 4, and 5. 17 Members of the Percavirus genus include EHV-2 and EHV-5, which infect horses, and EHV-7 (also known as AHV-2), which infects donkeys. 17 Two other γ-HV of donkeys, AHV-4 and AHV-5, remain unclassified and are awaiting assignment to a genus. 17
The first evidence of a γ-HV being associated with lung disease in veterinary medicine was reported in 2002. 44 Using consensus primer sets that target the herpesvirus DNA polymerase gene, herpesviral amplicons were generated from the lungs of 11 of 17 donkeys with interstitial pneumonia. Subsequent sequence analysis of the amplified DNA fragments identified 2 new γ-HV, termed AHV-4 and AHV-5. 44 Affected donkeys had moderate to severe respiratory disease typified by severe interstitial pneumonia with syncytial cells, with occasional animals having significant lung fibrosis. 44
In 2007, EMPF was identified as a severe and progressive fibrosing interstitial lung disease in adult horses associated with lung infection with the γ-HV EHV-5. 91 Affected horses develop characteristic clinical signs that include weight loss, low-grade fever, and progressive exercise intolerance. 94 The gross lesions in affected animals (Fig. 1) consist of severe nodular fibrosis that varies from discrete masses within the alveolar parenchyma to nearly diffuse consolidation of the lung by coalescent foci of interstitial fibrosis. 91 Histologically, there is marked interstitial fibrosis with retention of an “alveolar-like” architecture that is lined by cuboidal epithelial cells. Air spaces contain inflammatory cells consisting primarily of neutrophils and macrophages, with occasional macrophages having large intranuclear inclusion bodies (Fig. 2). 91 Myofibroblasts (Fig. 3) are abundant within the interstitium of EMPF-affected horses, suggesting that their presence is important in the progressive fibrosis characteristic of the disease.
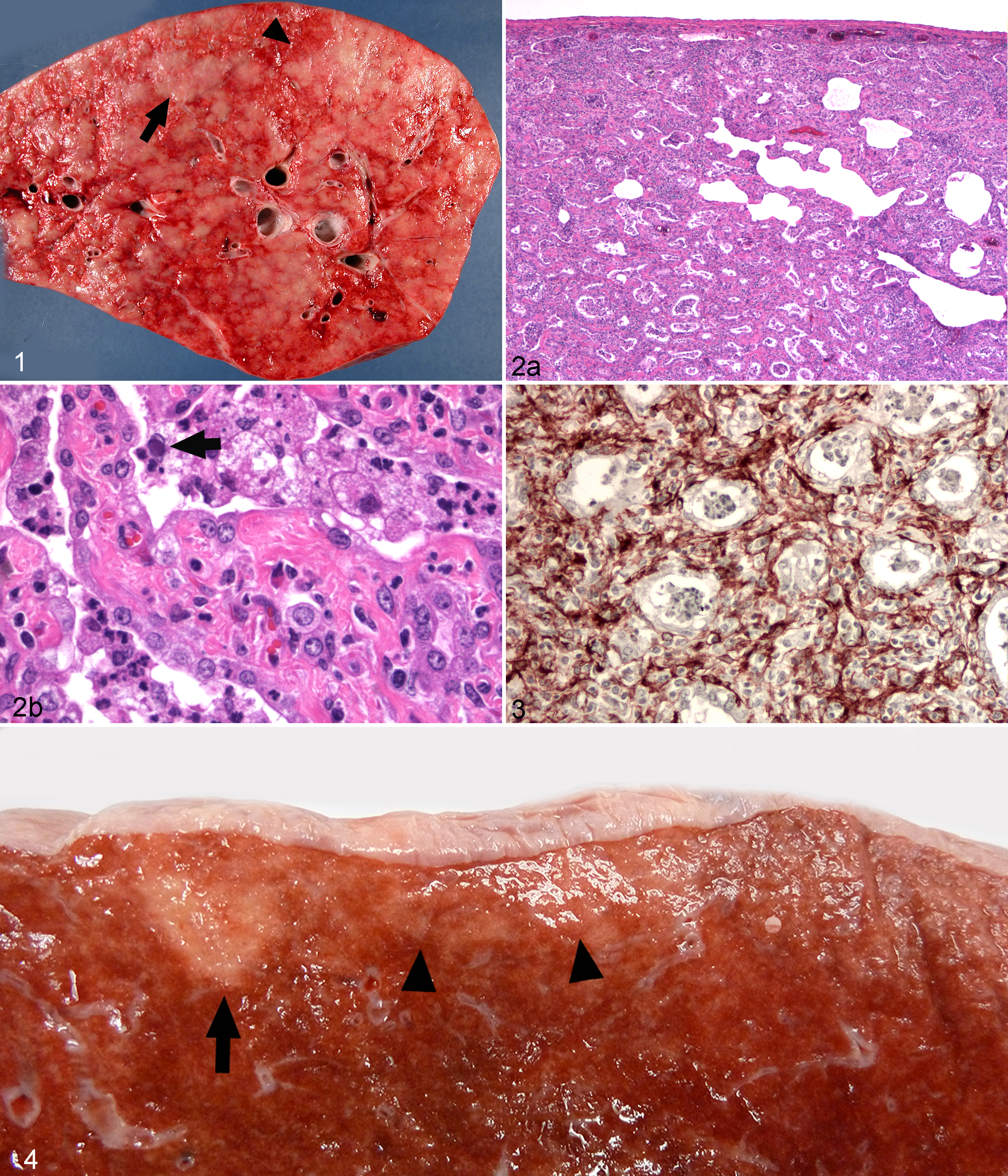
Prior to the description of EMPF, there had been no association of EHV-5 with any disease in horses. Indeed, little is known regarding the natural history of EHV-5 infections in horses, although as with EBV in humans, subclinical infections are common. 10,88 A number of PCR-based surveys of nasal swabs and peripheral blood mononuclear cells (PBMCs) have shown that infection of healthy horses with EHV-5 occurs early in life and persists in a significant percentage of adult horses. 10,49,59,81,88 In general, EHV-5 DNA is detected at higher levels in foals than in adult horses for both nasal swabs and PBMCs. In 205 weanling and 92 adult Thoroughbreds on 6 different studs in Australia, EHV-5 was detected in nasal swabs and/or PBMCs in 78% of clinically normal foals and 47% of adults. 88 When PBMCs were co-cultured with primary equine kidney cells supplemented with IL-2 to reactivate EHV-5, the prevalence of virus in PBMCs increased to 89% in foals and 100% in adult horses. 88 Similarly, a higher percentage of infected foals was detected by co-culture of PBMCs with RK-13 cells and analysis of the RK-13 cells after 3 serial passages, compared with direct detection of the virus in PBMCs at the time of sampling. 10
Apart from testing nasal swabs and PBMCs, there are few PCR surveys of other tissue sites for EHV-5. In the initial description of EMPF, EHV-5 was not detected in the lung of any of the 23 adult control horses, suggesting that the lung is not a common site of infection in horses not affected by EMPF, although the PCR methods used in the study may not detect low levels of latent virus. 91 In 2008, Torfason et al 81 detected EHV-5 and EHV 2 coinfection in the lungs of 4 healthy adult Icelandic horses and 2 healthy foals, as well as EHV-5 alone in the lungs of 2 of 5 Icelandic horses afflicted with “infectious pyrexia,” a disease of unknown cause. Regardless, as with EBV infection in humans, nearly all horses may be considered to have been infected with EHV-5, to have seroconverted to the virus, and to likely harbor latent virus within PBMCs. 10,88
The discovery of an association between lung infection with EHV-5 and the development of EMPF in adult horses provides evidence that γ-HV may contribute to the development of lung fibrosis in the natural host species. EHV-5 infection has been demonstrated in horses with EMPF by PCR assay, in situ hybridization, and ultrastructural evaluation. 91 All 24 horses in the initial description of EMPF had detectable EHV-5 within the lung compared with none of 23 age-matched controls. 91 Recently, using quantitative real-time PCR in a case of EMPF, EHV-5 DNA was more abundant in the lung compared with other organs, and within the lung, the virus was more prevalent in foci of fibrosis compared with other lung regions. 50 This further suggests a relationship between the development of lung fibrosis and the presence of EHV-5 in the lung. A recent report detected coinfection of EHV-5 and AHV-5 in horses with EMPF; the authors suggest that EHV-5 alone is not the only virus that may be associated with the development of EMPF. 7
Experimental Evidence for EHV-5 as the Cause of EMPF
While there is significant evidence to suggest a role for γ-HV in the development of progressive lung fibrosis in humans and equids, until recently there has been no direct evidence of a cause-and-effect relationship between lung infection with a γ-HV in the natural host (in any species) and the development of lung fibrosis. The close association of EHV-5 with EMPF affords the opportunity to experimentally investigate the ability of γ-HV to induce lung fibrosis in the natural host.
Recently, our laboratory undertook an experimental EHV-5 lung inoculation study in horses to attempt to induce lung fibrosis in clinically normal horses that is similar to EMPF. 92 We tested the hypothesis that isolates of EHV-5 obtained from cases of EMPF would induce lung fibrosis after direct inoculation into the lungs of clinically normal horses. Furthermore, we followed the systemic neutralizing antibody response to the virus in the inoculated horses, attempted to identify inoculum virus by comparing the genotype of the inoculated virus with that in the lungs of the horses at the end of the experiment, documented the presence of fibrosis and induction of myofibroblasts in the lung, and identified cell types harboring EHV-5 using immunohistochemistry.
To undertake this study, 22 horses were screened for neutralizing antibodies against EHV-2 and EHV-5, and the 8 adult horses (age range, 10–26 years) with the lowest titers (8–1024) to EHV-5 were selected for the study. Prior to inoculation, PBMCs and bronchoalveolar lavage fluid (BALF) from each horse were screened for EHV-5 and EHV-2 using PCR. Neither of these γ-HV was detected in BALF before inoculation, although 2 horses in the experimental infection group had detectable EHV-5 within PBMCs prior to inoculation. 92 Two separate isolates of EHV-5 from 2 cases of EMPF were grown in RK-13 cells and used for inoculation. The viruses were inoculated into the accessory lung lobes of 2 groups of 3 horses each. Two sham-inoculated horses served as controls.
None of the horses exposed to EHV-5 in this way developed clinical signs of illness, and only a modest rise in neutralizing antibody titers was documented. 92 Grossly evident nodular foci of fibrosis were present within the lungs of 3 of 6 inoculated horses (Fig. 4). Histologically, 5 of 6 horses had evident foci of fibrosis within the lungs that were accompanied by modest inflammation and accompanied by numerous myofibroblasts (Fig. 5). Unlike spontaneous EMPF, no viral inclusion bodies were visualized in the infected horses. Immunohistochemistry was performed using a rabbit polyclonal antibody generated against the inoculum viruses. This method revealed viral antigen within epithelial cells, interstitial fibroblasts/myofibroblasts, and macrophages within the foci of fibrosis in the inoculated horses. PCR analysis and attempts at virus isolation were unable to detect virus within the lungs of the inoculated horses. This inability to reisolate or detect inoculum virus by PCR within the lungs of the horses, despite the immunohistochemical evidence for viral antigen within the lung, suggests that the virus is present at a very low copy number and may be latent at onset of the development of lung fibrosis.
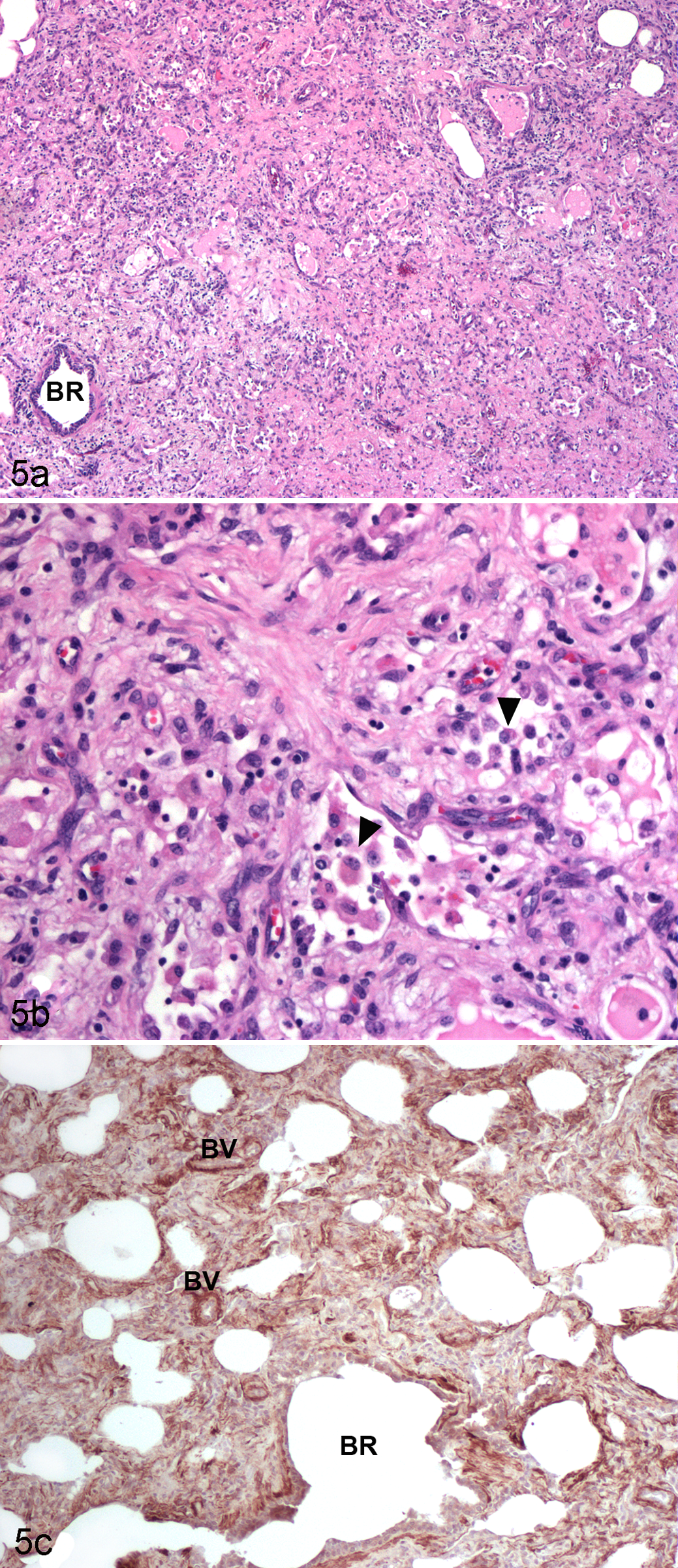
Lung, horse, experimental equine herpesvirus 5 (EHV-5) infection. Histopathology and α–smooth muscle actin (SMA) immunohistochemistry. (a) Nodule of interstitial fibrosis in the lung of a horse inoculated with EHV-5. (b) Higher magnification showing the alveolar interstitium expanded by abundant collagen and a small number of inflammatory cells. The lumens of the remodeled airspaces are lined
The results of this study provide the strongest evidence to date that EHV-5 is the cause of EMPF. These findings may have important implications vis-à-vis the development of pulmonary fibrosis in humans and other species.
Experimental Induction of Lung Fibrosis in Mice
Response to Pulmonary Infection With MHV-68 in the Natural vs Aberrant Host
As noted above, in horses and humans lung fibrosis has been associated with γ-HV (EHV-5 and EBV, respectively) indigenous to each species. Most experimental data regarding the pathogenesis of γ-HV and lung fibrosis have come from experimental exposure of mice to the γ-HV MHV-68. The natural hosts for MHV-68 are the bank vole (Myodes glareolus) and the wood mouse (Apodemus sylvaticus). 11,39 Because MHV-68 is not considered a natural pathogen of the laboratory mouse (Mus musculus), 11,39 it cannot be considered a true replica for γ-HV–associated lung fibrosis in the natural host (ie, EBV or EHV-5). Despite this, much has been learned about the biology of γ-HV infection in the lung and the development of lung fibrosis through the study of MHV-68 infection in laboratory mice. A diagram interpreting the major murine data is found in Fig. 6.
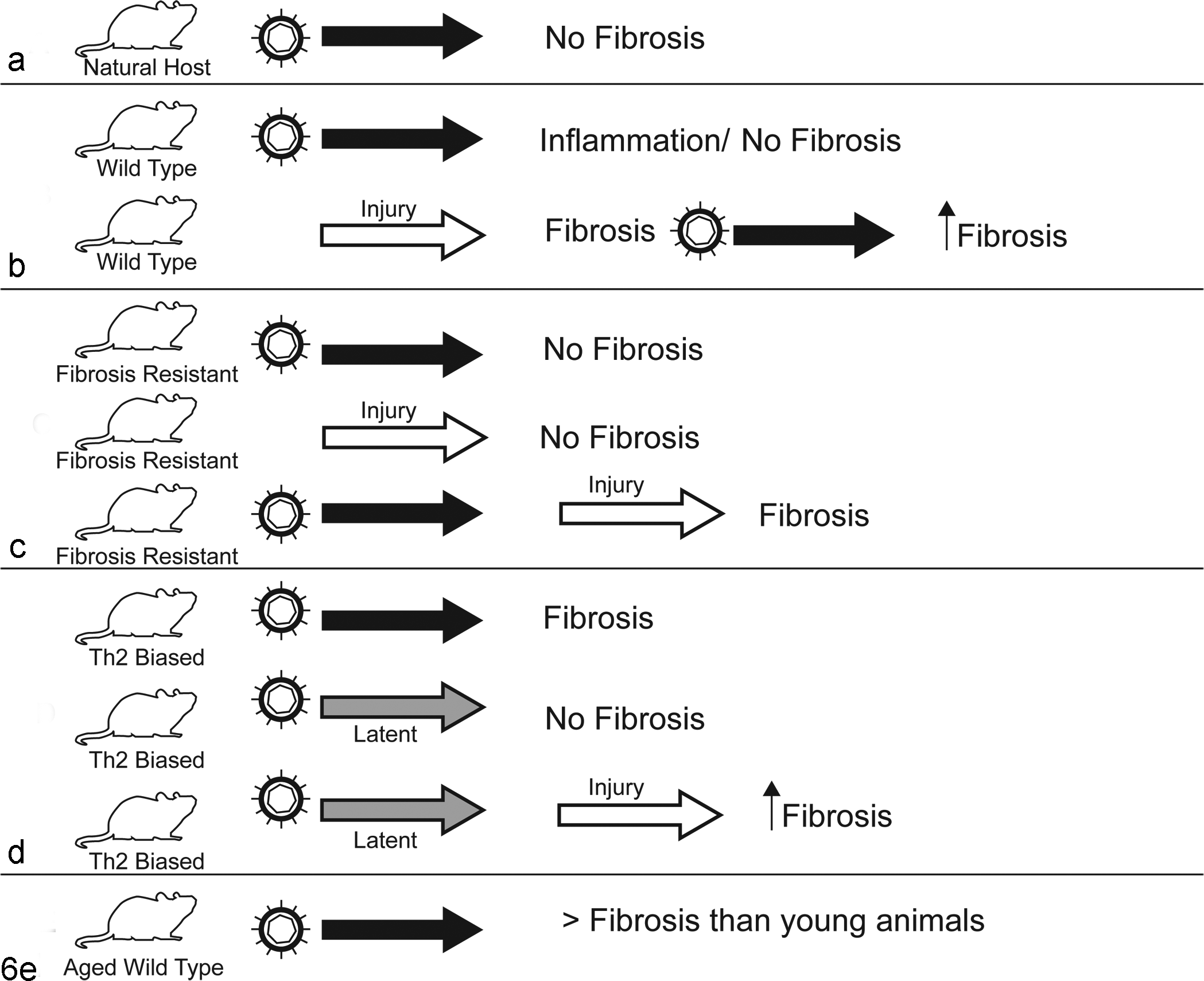
Summary of results of experimental lung infection with murine gammaherpesvirus 68 (MHV-68). (a) In the natural host, lung infection leads to little pathology and no fibrosis. 39,63 (b) Lung infection with MHV-68 alone in wild-type laboratory mice causes lung inflammation but no fibrosis, but the virus can augment lung fibrosis following exposure to lung fibrosis–inducing chemical injury from bleomycin. 39 (c) BALB/c mice are resistant to developing lung fibrosis following lung infection with MHV-68 infection or exposure to bleomycin. 48 Such mice become susceptible to lung fibrosis if infected with MHV-68 prior to exposure to bleomycin. 48 (d) Th2-biased (IFN-γR– / –) mice develop progressive lung fibrosis following lung infection with MHV-68. 20,55 Latent virus in the lungs of Th2-biased mice does not lead to fibrosis, but such virus augments fibrosis when mice are exposed to bleomycin or FITC. 75,84 (e) Aged mice develop greater lung fibrosis than younger mice after exposure to MHV-68 due to inherent profibrotic qualities present in the aged lung. 56,57
When comparing experimental lung infection with MHV-68 in a natural host—the wood mouse—with the nonhost laboratory mouse (BALB/c), considerable differences in response between the 2 species are noted. 39,63,77 In the wood mouse, lung infection with MHV-68 results in the induction of BALT, with scattered pulmonary foci of granulomatous inflammation and no fibrosis. 39 In contrast, MHV-68 induced prominent pulmonary interstitial inflammation in BALB/c mice and no fibrosis. In the wood mouse, MHV-68 replicates less efficiently, viral latency is more readily established, and production of neutralizing antibodies is enhanced compared with laboratory mice. 39 The chemokine binding protein M3 of MHV-68 has been shown to contribute to the difference in response between the wood mouse and the laboratory mouse. When viral expression of M3 is ablated, the lesions and viral latency in the wood mouse resemble the response in the laboratory mouse. 38 In totality, these data likely reflect the détente that has evolved in the relationship between MHV-68 and its natural host, the wood mouse, and is typical of the subclinical nature of γ-HV infections in general; however, the response differs considerably in a nonhost species such as the laboratory mouse. Despite these deficiencies, the laboratory mouse is the standard model species to investigate the biology of γ-HV and lung fibrosis. Interstitial pneumonia without lung fibrosis occurs in laboratory mice following lung infection with MHV-68. Active infection is self-limiting within the lung and persists for approximately 10 days, after which the virus becomes latent. 77,78 Lung infection causes alveolar epithelial cell necrosis and inflammation without fibrosis, with latency established in lung epithelial cells, macrophages, dendritic cells, and B cells. 74,77,78 The inflammation that occurs during lung infection appears approximately 7 days postinfection and is temporally associated with the upregulation of a variety of cytokines, chemokines, and their associated receptors and will resolve over time without induction of lung fibrosis. 66 –68
MHV-68 as a Cofactor in the Development of Pulmonary Fibrosis
As noted above, initial studies did not suggest that gammaherpesviral infections alone are sufficient to induce pulmonary fibrosis. Because IPF is assumed to occur as a result of multiple different injuries to the lung epithelium, Lok et al 48 combined MHV-68 lung infection with intraperitoneal bleomycin administration in BALB/c mice. Bleomycin is a glycopeptide produced by Streptococcus verticillus that is used in human medicine as an antineoplastic chemotherapeutic and is known to induce pulmonary fibrosis in mice, rats, hamsters, and some human patients with cancer. 12,102 Bleomycin-resistant BALB/c mice exposed to bleomycin alone or MHV-68 alone did not develop lung inflammation or fibrosis, while infection with MHV-68 1 week prior to bleomycin exposure resulted in significant lung inflammation and fibrosis, suggesting that γ-HV infections can act as cofactors in the development of pulmonary fibrosis. 48
Experimentally, viral infection does not have to precede a fibrogenic event to influence the degree of lung fibrosis. McMillan et al 52 infected C57Bl/6 and BALB/c mice with MHV-68 fourteen days after intratracheal administration of fluorescein isothiocyanate (FITC), at the time of peak FITC-induced lung fibrosis, and documented that viral infection increased the degree of lung fibrosis and reduced lung function to a greater extent than did FITC alone. Furthermore, this exacerbation of fibrosis was dependent on lytic viral infection of the lung.
MHV-68 as an Initiator of Pulmonary Fibrosis
In 2001, Ebrahimi et al 20 published the first study showing that infection with a γ-HV alone can result in tissue fibrosis. The authors investigated the pathogenesis of MHV-68 infection in IFN-γ receptor knockout (IFN-γR–/–) mice. IFN-γ is a Th1 cytokine that plays a role in CD4+ T-cell–mediated control of primary infection with MHV-68 and has been shown to prevent the development of bleomycin-induced lung fibrosis in mice. 29,40,66,68 IFN-γR–/– mice infected with MHV-68 developed fibrosis in the spleen, mediastinal lymph nodes, liver, and pulmonary interstitium, along with atrophy of the splenic lymphoid tissue. 20 The mice had significantly elevated levels of IL-1β, TNF-α, TNF-β, TGF-β1, and IFN-γ, and the authors suggested that this dysregulation of cytokines and chemokines drove the resultant tissue fibrosis. 20
Th2 Immune Response and MHV-68 Infection
The immunologic environment within the IPF lung is skewed toward a Th2 phenotype, with the induction of IL-4, IL-5, IL-10, and IL-13, in addition to TGF-β. These cytokines favor activation of fibroblasts and myofibroblasts and the production of extracellular matrix over its degradation. 30,86,87 The findings by Ebrahimi et al 20 therefore suggest that γ-HV infections may be capable of participating in lung fibrosis when there is a preexisting Th2-like environment established within the lung (achieved by ablating IFN-γ signaling) prior to infection. In 2005, Mora et al 55 addressed this question by establishing chronic lung infection with MHV-68 in Th2-biased mice. IFN-γR–/– mice were intranasally infected with MHV-68 and euthanized between 15 and 180 days postinfection. The infected mice developed progressive increases in interstitial lung collagen and myofibroblasts, along with decreased tidal volume and upregulation of Th2 cytokines. This study provided the first experimental evidence to suggest that chronic γ-HV infection in such mice results in progressive fibrosis—an important feature of IPF in humans. 55
The establishment of a Th2-like environment, as well as the associated production of Th2 cytokines within the lungs of IFN-γR–/– mice, appears to be necessary to promote γ-HV–induced lung fibrosis in this species. Th2 cytokines, in addition to promoting resolution of inflammation and fibrosis, can direct the differentiation of alternatively activated macrophages, which can themselves contribute to tissue fibrosis. 46 Mora et al 53 established chronic MHV-68 infection in IFN-γR–/– mice by intranasal administration of virus at 15-day intervals and sacrificed the animals 180 to 200 days postinfection. Using immunohistochemistry, Western blot analysis, and reverse transcription PCR, they documented that alveolar macrophages within the lungs of such chronically infected mice produce proteins associated with alternative activation (ie, FIZZ1, Ym1/Ym2, IGF, and arginase I) and transcription of fibronectin. 53
The induced alternative activation state in the lung macrophage may play an important role in driving fibrosis in the lungs of infected mice. Alternative activation of macrophages can also occur within the fibrotic spleens of IFN-γR–/– mice infected with MHV-68, suggesting that there may be systemic activation of macrophage subpopulations that is important in the development of multiorgan fibrosis in infected mice. 27
The effect of expression of the EBV transcription factor Zta on lung inflammation in mice was investigated. 31 Zta is a transcription factor involved in reactivating EBV from a latent state to a lytic infection. 25 Zta acts by binding to host target genes, including those that regulate inflammation such as nuclear factor (NF)–κB and p53. 31 The investigators used an adenovirus vector carrying Zta to drive expression within the lungs of mice. 31 Expression of EBV Zta did not result in lung fibrosis but did drive lung inflammation toward a Th2 phenotype and resulted in the generation of alternatively activated macrophages. The findings suggest that the presence of latent γ-HV may promote lung fibrosis.
Viral Latency and Pulmonary Fibrosis
Data in mice suggest that both lytic and latent infections with MHV-68 contribute to lung fibrosis. As noted above, primary infection by γ-HV in the natural host results in a brief lytic phase followed by latency. 3,39,58 In wild-type mice, MHV-68 establishes latency in the lung by 14 days postinfection. 84 Mora et al 54 were able to reduce lung fibrosis in IFN-γR–/– mice by blocking viral replication using the antiviral drug cidofovir or by infecting the mice with mutant virus that lacks the ability to reactivate from the latent state. The decrease in fibrosis was associated with lower levels of TGF-β, vascular endothelial growth factor, and markers of alternatively activated macrophages compared with animals experiencing persistent lytic infection by virus. 54
While the Mora et al 54 study suggests that maintaining viral latency prevents lung fibrosis, another study determined that the presence of latent MHV-68 in the lung potentiated the fibrotic effect of subsequent intratracheal FITC and bleomycin when administered 14 to 70 days postinfection. 84 The profibrotic cytokine TGF-β and cysteinyl leukotrienes were upregulated in alveolar epithelial cells of lungs latently infected with MHV-68, and increased numbers of fibrocytes (myofibroblast precursors) were recruited to the lungs. 84 Importantly, these changes occurred without reactivation of the latent virus.
A follow-up study by the same group documented that latent γ-HV in the lung stimulates the production of profibrotic mediators in the lung without a secondary stimulus. 75 In this study, wild-type mice were infected with MHV-68 and increased levels of TGF-β, CCL chemokines, tumor necrosis factor (TNF)–α, and IFN-γ were found in the lungs of latently infected mice. 75 Furthermore, they documented that these cytokines were produced by latently infected alveolar macrophages, mesenchymal cells, and CD19-expressing B cells. 75 Importantly, a profibrotic environment was established in the mice in the face of a strong IFN-γ response, suggesting that the virus modulates the lung environment to promote fibrosis upon subsequent lung injury. These studies indicate that latently infected lung is primed for fibrosis by the presence of the latent virus. This may be important given that most γ-HV are latent in the natural host species and suggests that individuals harboring latent virus may be primed to develop lung fibrosis.
Aging Promotes MHV-68–Induced Pulmonary Fibrosis
Because spontaneous pulmonary fibrosis in humans—and in equids, dogs, and cats—is a disease of adults, there is interest in comparing the response of the aged lung to the young lung in response to γ-HV infection. 56 The response of the aged murine lung to bleomycin has been investigated using the senescent-accelerated mouse (SAM), a mouse strain that ages more rapidly than wild-type mice and develops a number of aging-related diseases, including those affecting the neurologic, ophthalmic, and skeletal systems. 79 Xu et al 98 compared the responses of SAM and senescence-resistant mice to intratracheal bleomycin administration. SAM mice developed significantly greater fibrosis and inflammation and increased numbers of circulating fibrocytes compared with the senescence-resistant mice, suggesting that the aging lung is inherently primed to develop fibrosis. 98
In 2012, Naik et al 57 tested the hypothesis that the aged lung would be more susceptible to γ-HV–induced fibrosis. They infected young (4-month-old) and aged (15- to 18-month-old) wild-type C57Bl/6 mice with MHV-68. Increased lung collagen, fibrosis, and TGF-β following infection were detected in the aged lung, although there were no differences in virus clearance between the aged and young lungs, suggesting inherent differences in the aged lung that promote fibrosis. 57 Furthermore, this group demonstrated in vitro that fibroblasts from the aged lung inherently expressed greater baseline TGF-β receptors and, when infected with virus, more readily transformed into myofibroblast-like cells and produced greater amounts of collagen and fibronectin transcripts. 57
More recently, Torres-Gonzales et al 82 investigated differences in the endoplasmic reticulum (ER) stress response and alveolar epithelial apoptosis in aged (>18-month-old) and young (2- to 3-month-old) mice following infection with MHV-68. Aged mice experienced greater ER stress, type II pneumocyte apoptosis, and induction of profibrotic pathways during MHV-68 infection than young mice, along with greater lung fibrosis and decreased lung tidal volume compared with young mice. 82 The authors suggest that pulmonary γ-HV infections in aged animals may lead to increased accumulation of misfolded proteins within the ER, which induces a cascade of events leading to epithelial injury, programmed cell death, and epithelial cell loss, contributing to the progression of fibrosis in the adult lung. 82
Similar comparisons of the aged lung and the development of fibrosis have not been performed in other species afflicted with progressive fibrosing lung disease. There are, though, data on the aging equine and human immune system that could influence the interaction between γ-HV and the host that may influence the development of pulmonary fibrosis. PBMCs from aged horses do not proliferate in response to mitogens, even when supplemented with IL-2, as effectively as in younger horses 37 and have been shown to produce increased levels of the proinflammatory cytokines IFN-γ, TNF-α, IL-1β, IL-15, and IL-18. 4 This response is similar to “inflamm-aging” that has been noted in humans, which is attributed to a variety of inflammatory diseases in older people. 26 Furthermore, it has been documented that B cells from aged humans infected with EBV produce significantly greater IL-6 protein than infected B cells from younger people. 61 These data suggest that the immune system of aging individuals (horses and humans) could be primed for the development of inappropriate inflammation and that some older individuals may respond to the presence of γ-HV infection in the lung through the development of progressive fibrosis.
Summary and Conclusions
Progressive fibrosing lung diseases have been recognized as a cause of significant morbidity and mortality in human medicine for many decades, with the first description of what was likely IPF reported by Hamman and Rich in 1935. 32 Over the ensuing decades, physicians and human medical pathologists have refined the classification of such diseases into those with known causes and forms of interstitial lung diseases whose cause(s) has eluded discovery thus far. Viruses, particularly the γ-HV EBV, have been implicated in the pathogenesis of IPF in people. Studies of lung infection in mice with MHV-68, as well as the recent discovery of the association of EHV-5 with the development of EMPF in horses, have provided additional evidence for γ-HV involvement in the development of lung fibrosis.
Primary γ-HV infections in the host species occur early in life and are usually clinically silent, such that nearly all adults of the species in question carry latent virus in tissues and have serum neutralizing antibodies to the virus. Despite the prevalence of γ-HV infection, though, progressive fibrosing lung diseases remain relatively rare. When considering the potential role that γ-HV play in the development of pulmonary fibrosis, there are a number of host and viral factors to consider (Fig. 7). Host factors, such as whether the host species is the natural or aberrant host for the virus, age, the prevailing state of the immune environment within the lung, and the presence of other sources of lung injury, may determine the outcome of infection. Viral factors, such as whether the virus is latent or in a lytic phase of the infection, and the expression of virus genes such as cytokines and chemokines encoded by the virus genome may also play a role in the development of lung fibrosis.
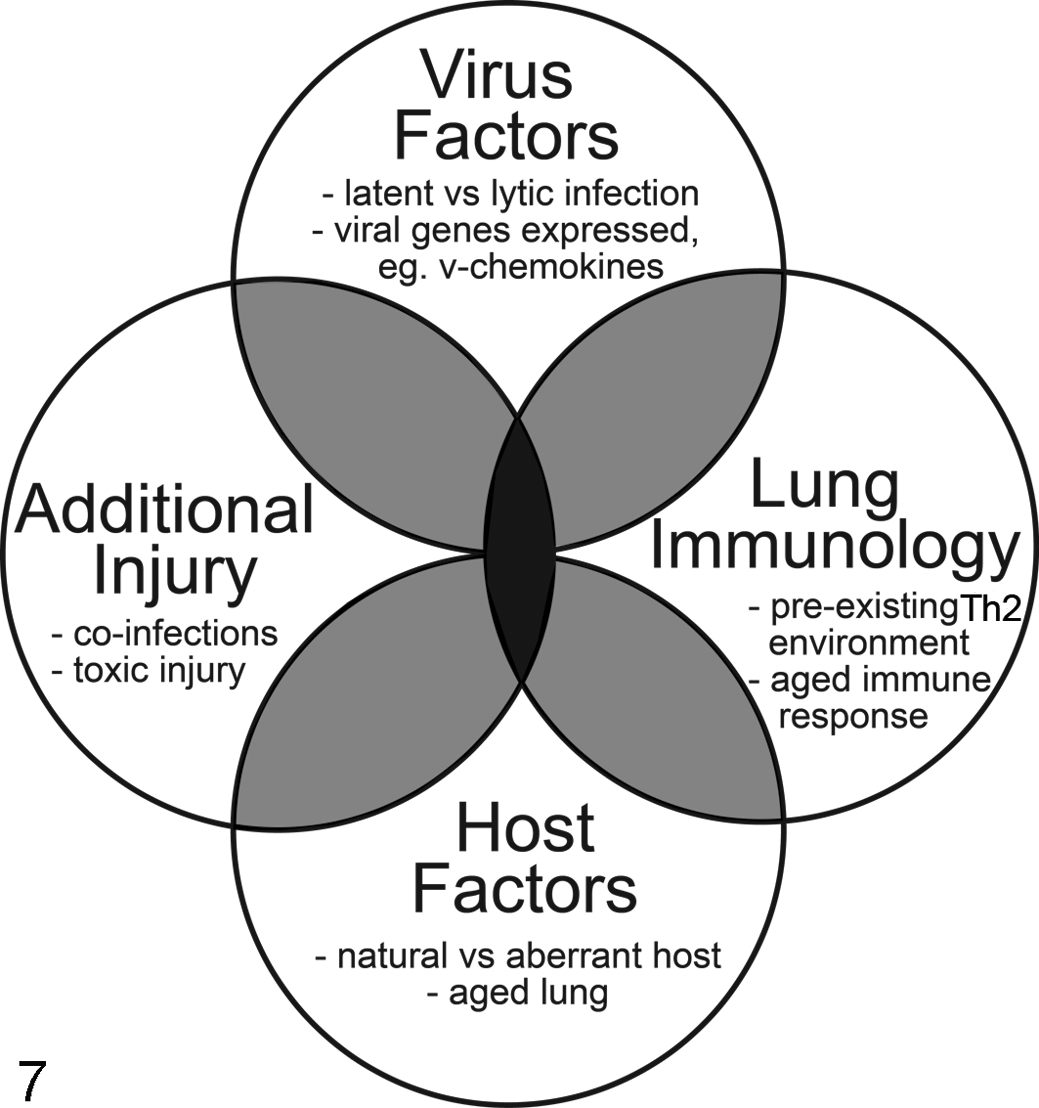
Potential factors that may determine the development of pulmonary fibrosis associated with gammaherpesvirus (γ-HV) infection in the lung based on data generated in rodents. Fibrosis (gray) may develop through a combination of host factors, viral factors, prevailing immune responses in the lung, or as a response to additional lung injury. Fibrosis may be expected to be most prominent, and in some cases progressive, when these factors occur in combination (black).
The most recent data with EHV-5 suggest that some γ-HV infections may be capable of inducing lung fibrosis on their own in their natural host species. This finding, as well as future studies investigating the biology of host–EHV-5 interactions, may yield important insights into the mechanisms of γ-HV–induced lung fibrosis.
Footnotes
Acknowledgements
The author would like to acknowledge Maggie Hoffmann for her assistance in preparing the diagrams for Figures 6 and ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: Pfizer Animal Health, Kalamazoo, MI USA, provided funding to perform the experimental EHV-5 studies summarized in this article.
