Abstract
In September 2020, an outbreak of epizootic hemorrhagic disease occurred in captive reindeer (Rangifer tarandus) and was associated with neurological signs and mortality. Four reindeer died or were euthanized after acute illness over a 12-day period. Affected reindeer displayed abnormal behavior, neurologic signs, lethargy, and/or lameness. The most consistent gross finding was dark red streaks throughout the adrenal gland cortices (4/4). One animal had acute hemorrhage involving the subcutis and skeletal muscles over the ventrolateral body wall and back, and abomasal serosa. Histologically, the most common lesions were adrenal gland cortical hemorrhage (4/4) with necrosis (3/4) and lymphoplasmacytic meningoencephalitis with gliosis, glial nodules, satellitosis, and nonsuppurative perivascular cuffing (4/4). The brain lesions were most frequent in the gray matter of the cerebrum, hippocampus, and thalamus but also involved the cerebellum and brainstem. Epizootic hemorrhagic disease virus serotype 6 was detected through PCR and sequencing of the spleen in all cases.
Epizootic hemorrhagic disease virus (EHDV) is an arbovirus classified in the genus Orbivirus within the subfamily Sedoreovirinae of the family Reoviridae. It is the causative agent of epizootic hemorrhagic disease (EHD), a disease characterized in its acute form by sudden death, possibly with a fulminant hemorrhagic disease syndrome.11,12,17 The virus is closely related to bluetongue virus, which causes similar clinical signs and lesions.7,11,18,19 Epizootic hemorrhagic disease virus is spread by hematophagous midges, with Culicoides sonorensis being the only definitively identified vector although other Culicoides species are likely involved.11,17-20,22 The disease most often occurs in late summer and early fall, correlating with seasonal patterns of vector activity.7,11,20 Seven serotypes (1–2, 4–8) have been identified thus far. Epizootic hemorrhagic disease virus-1, EHDV-2, and EHDV-6 have been reported in North America.1,11,12,17-20,22 Epizootic hemorrhagic disease virus-6 was first reported in North America in 2006 from moribund or dead white-tailed deer (WTD) in Indiana and Illinois, with increasing numbers of reports over the years across a wide geographical range.1,18,21,22
Although EHD is a disease primarily affecting cervids, it has been reported in a variety of wild and domestic noncervid ruminants.18,19 Cattle may be infected, but the majority remain asymptomatic or develop mild transient clinical signs.11,12,19,20 However, outbreaks with more substantial clinical signs and death have occurred sporadically in the Mediterranean basin, Reunion Island, South Africa, and the United States in association with EHDV-2, EHDV-6, EHDV-7, and the Ibaraki strain of EHDV-2.11,12,19,20 Clinical disease has been reported in other ruminant species, including mule deer, black-tailed deer, elk, pronghorn, brocket deer, yaks, bison, alpaca, mountain goat, and Rocky Mountain bighorn sheep.11,15,16,18,20 To our knowledge, there have been no reports of EHD in reindeer (Rangifer tarandus). In this report, we describe the gross and microscopic findings of fatal EHD in captive reindeer with confirmed EHDV-6 infection.
Over a 12-day period in August and September 2020, 4 reindeer of a captive herd in Minnesota became acutely ill and were euthanized or died without any premonitory signs (Table 1). They included 3 females and 1 male, and ages ranged from 2 to 13 years. All animals had been at the zoo for 1 to 3 years. Clinical signs, when present, lasted for 1 to 2 days and ranged from neurological signs (including circling), lethargy, abnormal behavior (such as obsessively licking the ground and leg), and/or lameness. One animal was found dead without premonitory signs. All animals underwent necropsy. A wide variety of tissues were collected in 10% neutral-buffered formalin. Brain, spleen, and liver samples were saved frozen. Formalin-fixed tissues were paraffin-embedded, sectioned at 4 µm, and stained with hematoxylin and eosin.
Signalment, date of onset of clinical signs, duration of illness, clinical signs, date of death, manner of death, and molecular (PCR and partial genome sequencing) and VI results of the reindeer included in this study.
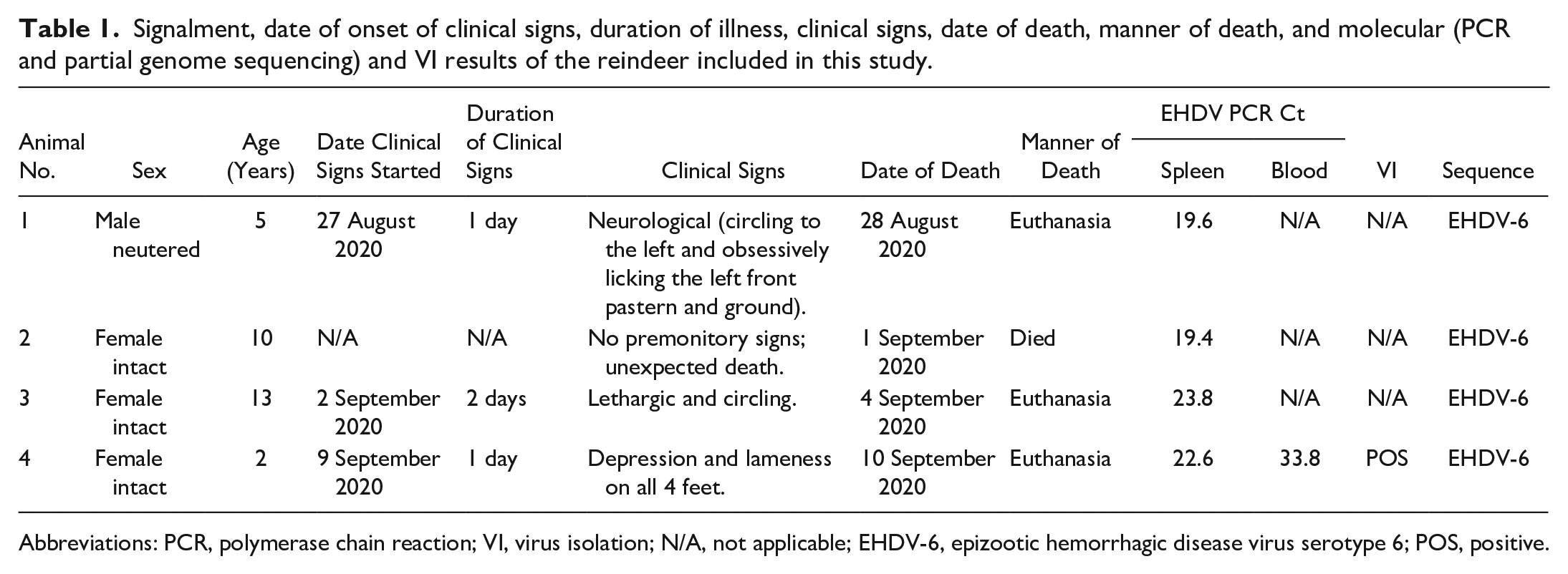
Abbreviations: PCR, polymerase chain reaction; VI, virus isolation; N/A, not applicable; EHDV-6, epizootic hemorrhagic disease virus serotype 6; POS, positive.
At necropsy, all animals were in a good nutritional state. In all cases, the adrenal glands had multiple, dark red streaks throughout the cortices (Fig. 1). Other findings included a diffusely enlarged and dark red spleen in 3 animals, pulmonary alveolar edema and congestion in 2 animals, and hemorrhages within the subcutis and skeletal muscles over the ventrolateral body wall, musculature of the back, and abomasal serosa in 1 animal.
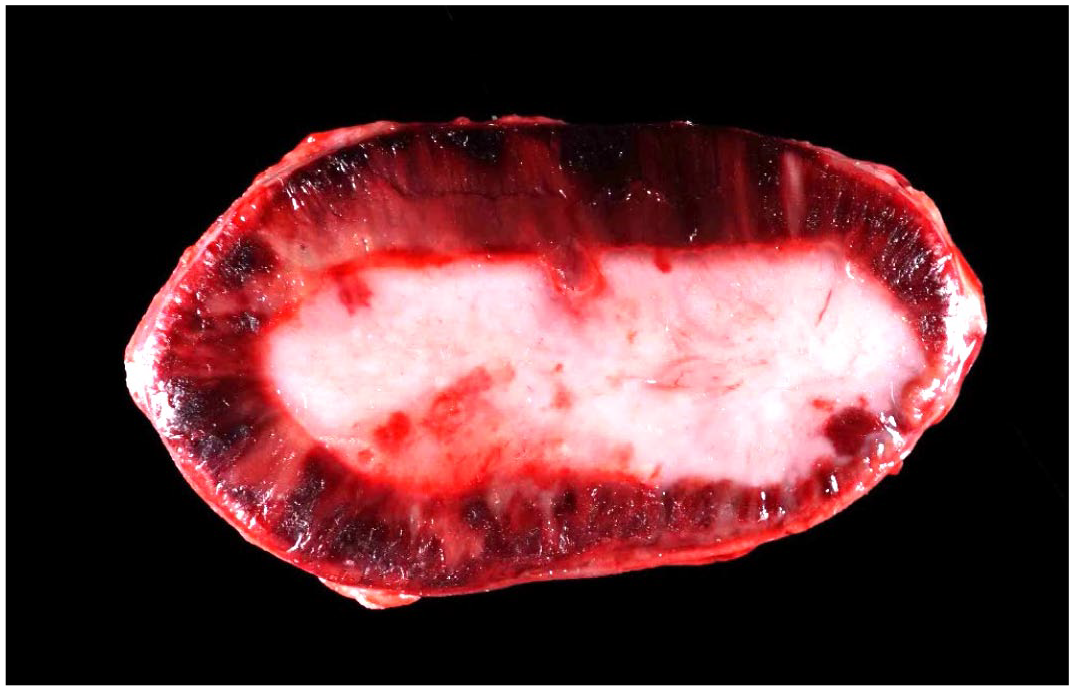
Adrenal gland, cut section, case 3. There are multiple, dark red streaks throughout the cortex.
The most significant histological lesions in all animals involved the adrenal glands and brains. In all animals, the adrenal glands had bilateral, diffuse, cortical congestion and randomly distributed, mild to marked hemorrhage affecting approximately 20% to 80% of the sections (Fig. 2a, b). The hemorrhagic foci in 3 animals (cases 1, 3, and 4) were accompanied by coagulative necrosis, characterized by eosinophilic ghostlike outlines of cortical cells with absent, karyolitic, or karyorrhectic nuclei or by lytic necrosis with eosinophilic granular debris admixed with small numbers of neutrophils. Necrotic foci were often surrounded by a thin rim of degenerate cortical cells with intracytoplasmic, feathery, indistinct vacuoles.
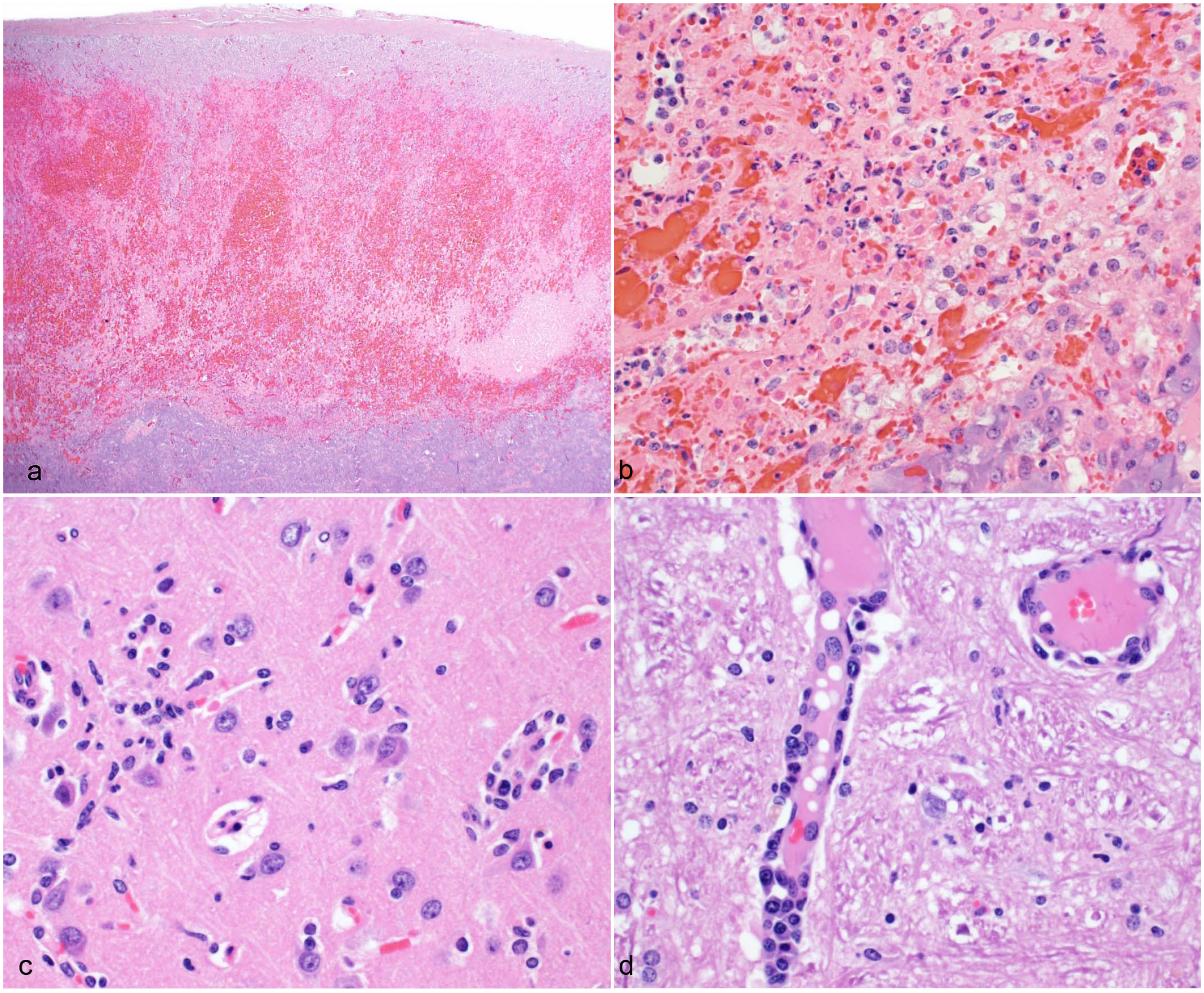
Adrenal gland and brain of reindeer with epizootic hemorrhagic disease. Hematoxylin and eosin. (a, b) Adrenal gland, case 1. There is extensive loss of cortical cells with lytic necrosis, neutrophilic infiltration, hemorrhage, and congestion. (c) Brain, case 4. There are multifocal glial nodules admixed with low numbers of neutrophils. (d) Brain, case 4. Multiple vessels are surrounded and infiltrated by lymphocytes and plasma cells, and affected vessels are lined by hypertrophied endothelial cells.
All animals had lesions in the brain that were typically more severe in the cerebral gray matter, hippocampus, and thalamus than those of the cerebellum and brainstem. The 2 reindeer that were affected later in the outbreak (cases 3 and 4) had more severe brain lesions. The lesions consisted of rare to occasional nodular aggregates of glial cells (glial nodules) that were accompanied by a small number of neutrophils (Fig. 2c). The neuroparenchyma multifocally had subtle increased cellularity due to a slightly increased number of glial cells (gliosis), especially within the hippocampus and cerebral gray matter. In the cerebral gray matter, rare neurons were surrounded by glial cells (satellitosis). Multiple small-caliber vessels within the neuroparenchyma were surrounded by up to 3 cell layers of lymphocytes, histiocytes, and fewer plasma cells (Fig. 2d). Many of the affected vessels were lined by hypertrophied endothelial cells. Two animals (cases 3 and 4) had rare foci of liquefactive necrosis in the hippocampus, cerebrum, and/or corpus callosum that were surrounded by thin rims of glial cells and neutrophils. In 1 reindeer (case 3), the neuropil surrounding areas of gliosis and/or glial nodules was edematous based on the presence of clear spaces between the intricate network of cells and cell processes. Spinal cord was examined in 2 animals (cases 1 and 4). In 1 animal (case 4), the gray matter of the spinal cord had a few, subtle glial nodules.
Two reindeer (cases 3 and 4) had mild retinitis with rare aggregates of lymphocytes and fewer plasma cells that predominantly surrounded small-caliber blood vessels. In one of these cases (case 3), the nerve fiber layer and inner plexiform layer of the retina had rare, scattered foci of liquefactive necrosis with loss of cells and replacement by amorphous, eosinophilic material, karyorrhectic nuclei, and low numbers of neutrophils. One animal (case 1) had multifocal myocarditis characterized by small aggregates of neutrophils, fewer lymphocytes, plasma cells, and a few fibroblasts accompanied with necrotic cardiomyocytes with hypereosinophilic sarcoplasm, loss of cross striation, and pyknotic nuclei.
Unfixed spleen from all animals and a whole blood sample from case 4 were submitted to the National Veterinary Service Laboratories in Ames, Iowa for EHDV-specific PCR using methods described previously.4,23 All samples tested positive for EHDV (Table 1). All positive EHDV samples were serotyped as EHDV-6 by partial genome sequencing targeting VP2.10,23 Based on the VP2 gene sequencing, all study sequences were 100% identical to one another and showed >99% nucleotide identity with an EHDV-6 strain isolated from an Ohio deer (KF570134.1; Supplemental Table S1). The phylogenetic analysis was performed on 2426 nucleotide curated sequences and compared with the published sequences of EHDV-6 available in GenBank. Case 1 was excluded for the phylogenetic analysis as only a shorter sequence over 594 bases was retrievable. Geneious Prime 2022.0.1 was used for sequence alignment and percent identity analysis. The sequences were aligned using MAFFT and a phylogenetic tree was constructed in RAxML 8.2.11 (Supplemental Figure S1). The phylogenetic tree was constructed by comparing 2426 nucleotides of the VP2 gene, using the GAMMA-based likelihood-method-based 1000 bootstraps. The sequences generated in this study (excluding case 1) were deposited in Genbank (OQ679892-679902 and OQ688969-688971).
Samples from 3 animals (cases 2, 3, and 4) underwent whole genome sequencing, using previously described methods with modifications. 3 Briefly, nucleic acid was incubated at 99°C for 5 minutes to denature dsRNA, then snap-cooled on dry ice before first-strand synthesis. Potential coinfection with bluetongue virus was excluded by whole genome sequence analysis. In addition, EHDV-6 was isolated from the whole blood sample from case 4 using a procedure adapted from previously described methods. 23 Virus isolation was not attempted on the spleen of the other animals as the VP2 gene sequencing results indicated that all animals were infected by the same virus.
Serum samples from 3 of the 4 animals collected during the outbreak and banked serum samples from cases 1 and 2 were submitted to the National Veterinary Service Laboratories for EHDV-specific serology. Serum from case 2 was not collected during the outbreak as the animal died without premonitory signs. Of the samples collected between 20 August 2020 and 10 September 2020, all 3 animals were positive for EHDV-specific antibodies by both agar gel immunodiffusion, as described previously, 5 and virus neutralization for North American serotypes 1, 2, and 6 per World Organization for Animal Health guideline (Supplemental Table S2). 23 Note that cross-reactivity is not unexpected between the EHDV serotypes when testing by virus neutralization. 23 The banked serum samples from case 1, taken on 12 May 2020, and case 2, sampled on 3 August 2020, tested negative for EHDV-specific antibodies by agar gel immunodiffusion and virus neutralization.
Unfixed brain samples from 3 animals (cases 1, 3, and 4) were submitted to the Minnesota Department of Health and tested negative for rabies virus by direct fluorescent antibody assay as per CDC guidelines, 2 negative for West Nile virus by PCR using slightly modified primer sequences from previously published methods, 9 and negative on PCR testing for Eastern equine encephalitis virus and Western equine encephalitis virus. 8
The report describes an outbreak of EHD caused by EHDV-6 in a captive reindeer herd. The clinical presentation and lesions observed in these reindeer differed slightly from previously published cases of EHD in ruminants, in that the main and most consistent lesions were within the adrenal glands and the brain. Neurological signs, such as incoordination, seizures, head tremors, hyperesthesia to sound and touch, loss of prehension, indifference to people, and apparent blindness have been infrequently reported and have been noted in WTD, black-tailed deer, pronghorn, yak, and mule deer.5,7,15,16,19 Such cases involved EHDV-2 and EHDV-6, or the EHDV serotype was not specified. Central nervous system lesions associated with EHD have rarely been reported and include thrombosis, nonsuppurative meningoencephalitis, neutrophilic to lymphoplasmacytic perivascular inflammation, hemorrhage, glial nodules, and lymphoplasmacytic vasculitis in the brain and spinal cord.7,15,19 Many of these lesions were present in the reindeer of this study. The identified lesions appeared to be more severe than in the published cases and were associated with liquefactive necrosis in 2 cases and cerebral edema in 1. In the reindeer of this study, lesions occurred throughout most regions of the brain, but were most severe in the cerebral gray matter, hippocampus, and thalamus.
Classically, in peracute and acute cases of EHD, there is hemorrhage and/or edema involving multiple tissues (most frequently involving the heart, skin, and gastrointestinal tract, in particular the abomasum), as well as gastrointestinal ulceration and erosion, necrosis in multiple tissues, and coronitis.5-7,11,15,19,20,22 The reindeer in this study were unique in that the most common and grossly striking feature involved the adrenal glands, without significant cardiac or gastrointestinal tract involvement. Adrenal gland involvement has been reported;5-7,15 however, it is usually not the main and only site of hemorrhage, unlike in 3 of the 4 reindeer. The reason for the unique lesion presentation in this report is unclear and may include host, virus, and/or vector interactions. Lesions are typically mild or not seen in cattle, and animal-to-animal variation has been reported in experimental WTD infections. 19 In WTD, the pathological findings also differ depending on the chronicity of the disease.6,20 Hence, it is possible that these reindeer did not survive long enough for some of the classic lesions to develop.
Epizootic hemorrhagic disease was first reported in Minnesota in a sick cow during 2012, when an unusually large number of EHD cases were observed in cattle and other ruminants.13,22 The majority of these cases, including the case detected in Minnesota, were caused by EHDV-2. The first major EHD outbreak (caused by EHDV-2) in Minnesota’s free-ranging WTD was documented in 2019. 14 To our knowledge, EHDV-6 had not been reported in the state until this reindeer outbreak. 13 Neither the reasons for the low incidence in certain ruminant species, nor the reasons for the increased reports of EHDV-6 cases in the United States have been established. Possible suggestions include lack of detection and routine examination of ill animals (unlikely in a zoo setting); innate resistance of the host to the virus; exposure to a new serotype or strain of EHDV; vector seasonality; numerous other environmental conditions that may affect vector distribution, size, and competence; and/or the role of maternal antibodies in regions where EHDV is endemic.5,17,19,21,22 Although Culicoides sonorensis is the only definitively identified vector for EHDV, these midges are not commonly distributed throughout the upper Midwestern and Eastern United States. 17 Despite this, EHDV-6 has continued to spread in the upper Midwestern and Eastern North America, suggesting that other Culicoides species may play a role in transmission. 17 A rise in clinical cases with EHD has been seen when EHDV spreads beyond its normal geographical distribution, resulting in exposure of immunologically naive populations. 22 In endemic areas, the seroprevalence of EHDV in WTD can be high, but disease is rare.18,21 The serology results in the 4 reindeer suggest that the outbreak began mid to late August.
This report shows that reindeer are susceptible to EHDV-6 infection and develop clinical disease with fatal outcomes. Gross and microscopic changes were dominated by adrenal gland and brain involvement.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231196797 – Supplemental material for Outbreak of epizootic hemorrhagic disease in captive reindeer (Rangifer tarandus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231196797 for Outbreak of epizootic hemorrhagic disease in captive reindeer (Rangifer tarandus) by Emma H. Torii, Arno Wünschmann, Mia Kim Torchetti, Leo Koster, Albert van Geelen, Randy Atchison and Anne Rivas in Veterinary Pathology
Footnotes
Acknowledgements
We thank Mary Lea Killian from the National Veterinary Services Laboratories, the clinicians and veterinary care staff at the Minnesota Zoo, the necropsy and histology staff at the University of Minnesota Veterinary Diagnostic Laboratory, the Diagnostic Virology staff at the National Veterinary Services Laboratories, and the staff at the Minnesota Department of Health. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official US Department of Agriculture (USDA) or US Government determination or policy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
