Abstract
Primary pulmonary neoplasms in cattle are rare. There are few studies on the pathological findings of these neoplasms in this species. This study aimed to describe the histological and immunohistochemical findings of primary and metastatic pulmonary carcinomas in cattle. We conducted a retrospective study of 19 cases of epithelial neoplasms with pulmonary involvement. Histologically, most of the neoplasms were classified as primary pulmonary neoplasms, including different adenocarcinoma subtypes (4/19, 21%) and adenosquamous carcinomas (3/19, 16%), followed by squamous cell carcinoma (6/19, 32%), metastatic uterine adenocarcinoma (4/19, 21%), metastatic hepatocellular carcinoma (1/19, 5%), and metastatic cholangiocarcinoma (1/19, 5%). By immunohistochemistry, all neoplasms were positive for pancytokeratin, and 4/19 (21%) were positive for vimentin. Primary pulmonary neoplasms had immunoreactivity for thyroid transcription factor-1 (6/7), while only 2 of these cases were positive for napsin A. All cases with squamous differentiation (9/9) had immunoreactivity for cytokeratin (CK) 5/6, while only 7 of these cases were positive for p40. CK20, CK7, and CK8/18 showed varied immunoreactivity in the primary and metastatic pulmonary carcinomas but were important markers to confirm the diagnosis of primary mucinous adenocarcinoma and metastatic cholangiocarcinoma. HepPar-1 was only positive in the metastatic hepatocellular carcinoma. The limited number of cases of metastatic uterine adenocarcinomas in this study precluded identification of a specific immunophenotype for this tumor. Immunohistochemistry proved to be an important tool to confirm the proper classification of these neoplasms.
Primary pulmonary neoplasms are considered rare in livestock. 30 In cattle, studies on the frequency of these neoplasms are scarce and are usually comprised of incidental findings from slaughterhouses.4,46 The reported frequency of primary pulmonary tumors in cattle varies 8.3% in England, 4 0.86% in the Netherlands, 35 and 0.32% to 0.68% in southern Brazil.31,49
Primary epithelial pulmonary neoplasms may arise from alveolar cells, bronchial glands, or bronchiolar epithelium. 12 Although adenocarcinomas (ADs) are considered rare, these are the most frequent histological type reported in cattle.5,46,60 Metastatic neoplasms may be more common, as observed in a study by Anderson and Sandinson 4 in which metastatic tumors were more than twice as frequent as primary tumors in cattle. In addition, reports of pulmonary metastases in cattle are frequent.17,31,59 The lung is considered one of the main metastatic sites, both in humans and in nonhuman animals, because pulmonary capillaries are susceptible to tumor emboli and subsequent colonization by neoplastic cells.23,30
Histologic differentiation between primary and metastatic epithelial neoplasms involving the lung may be difficult. In this regard, immunohistochemistry (IHC) can be helpful, and there are several immunohistochemical markers that may help in reaching a diagnosis of primary pulmonary neoplasia. In human medicine, there is a well-established immunohistochemical panel, which can be adapted for the suspected types and subtypes of tumors, including markers for pneumocytes (thyroid transcription factor 1 [TTF-1] and napsin A) and squamous epithelial cells (CK5/6 and p40).50,65 To the best of our knowledge, there are no studies evaluating the histological and immunohistochemical features of primary and metastatic pulmonary carcinomas in cattle. Therefore, the aim of this work was to characterize, through histology and IHC, carcinomas with pulmonary involvement in cattle and to provide support to veterinary pathologists in the identification of these tumors.
Material and Methods
Case Selection
A retrospective study was carried out through the anatomopathological records of the Department of Veterinary Pathology (SPV) of the Universidade Federal do Rio Grande do Sul (UFRGS) from January 2006 to December 2021. The keywords “bovine, cattle, lung, pulmonary, carcinoma, neoplasm, and epithelial” were used to search the digital database. The inclusion criteria of the cases were as follows: (1) cattle with malignant primary pulmonary neoplasms of epithelial origin (primary tumors) or (2) cattle with malignant epithelial neoplasms from other organs with pulmonary metastases (secondary/metastatic tumors). The samples were obtained either at necropsy or during routine inspection in slaughterhouses in the state of Rio Grande do Sul, Brazil. Data, such as breed, breed aptitude (ie, dairy or beef), sex, and age, were obtained from the records. Formalin-fixed paraffin-embedded tissue blocks of the selected cases were retrieved for histological analysis. Exclusion criteria were defined as cases that had poor histological tissue quality (severe autolysis) and/or unavailable paraffin blocks.
Histologic Evaluation
Histologic sections from each case were stained with hematoxylin and eosin (HE), periodic acid-Schiff (PAS), and alcian blue (AB) at SPV-UFRGS. Four anatomic pathologists (AV, IRS, MMP, and SPP) reexamined all of the cases to establish a consensus on the final diagnoses. All metastatic tumors were evaluated together with the primary tumor, with the exception of the squamous cell carcinomas (SCCs). Not all organs were submitted for histologic evaluation in each case, and the origin of the SCCs could not be determined.
Primary pulmonary carcinomas were classified as AD (subclassified as acinar, solid, mucinous, lepidic, papillary, and micropapillary based on the predominant pattern) and adenosquamous carcinoma, according to a modified version of the current World Health Organization (WHO) classification system for human lung tumors. 50 The modification to the classification system for this study involves the omission of the terms “in situ, minimally invasive, and invasive”8,50 because of the lack of well-established definitions (ie, relationship between gross tumor size and histological characteristics/size of foci of invasive lesions) with associated clinical implications for cattle. The remaining cases were classified as SCC or according to the site of the primary tumor.3,14,64
For the primary pulmonary carcinomas and SCCs, lymphovascular invasion (LVI) was evaluated in all available tissues according to the number of vessels involved (few, less than 5 vessels; moderate, 5-10 vessels; many, greater than 10 vessels), site of invasion (intratumoral or peritumoral), and the type of vessel invaded (blood vessels, lymphatic vessels, or both). 36 For the metastatic neoplasms, LVI was classified as evident in the examined sections or not. In all cases, intratumoral necrosis was classified as absent, small foci (1%-10% of the neoplasm), medium-sized foci (11%-50% of the neoplasm), and large foci (greater than 50% of the neoplasm). 37 The amount of fibrous or fibrovascular stroma and desmoplastic response (proliferation of collagenized connective tissue) was categorized as small (1%-15% of the neoplasm), moderate (16%-50% of the neoplasm), and large (greater than 50% of the neoplasm). Intratumoral inflammatory infiltrates were manually counted in all available sections and classified according to quantity (absent; mild, 1-15 cells in the entire tumor area; moderate, 16-50 cells; or marked, greater than 50 cells) and cellular composition. The percentage of neoplastic cells with anisocytosis and anisokaryosis (form and size variation of the cytoplasm and nucleus, respectively) were considered small (1%-15% of neoplastic cells), medium (16%-50% of neoplastic cells), and large (greater than 50% of neoplastic cells). The mitotic count was determined as the total number of mitotic figures in a total area of 2.37 mm2, evaluating consecutive fields in a region with the highest mitotic activity and avoiding necrotic areas. 34 The location of PAS and/or AB-positive mucin was evaluated as intracellular or extracellular.
Immunohistochemical Evaluation
IHC for pancytokeratin (panCK), vimentin, napsin A, TTF-1, p40, HepPar-1, CK20, CK7, CK8/18, and CK5/6 was performed on all neoplastic tissue (primary and metastatic sites). IHC was performed at SPV-UFRGS and at a commercial pathology laboratory (Patologistas Reunidos, Porto Alegre, Rio Grande do Sul, Brazil). Data on the antibody, endogenous peroxidase blockage, dilution, antigen retrieval, detection system, chromogen, external positive control, and expected immunoreactivity and positive cells are detailed in Supplemental Table S1. All the slides were counterstained with Harris’ hematoxylin. The normal adjacent parenchyma was used as internal positive controls for napsin A (type 2 pneumocytes, bronchiolar epithelium, and alveolar macrophages), 40 TTF-1 (bronchiolar and alveolar epithelial cells), 41 p40 (bronchial basal cells), 22 panCK (bronchiolar and alveolar epithelial cells), and vimentin (fibroblasts). IHC for all these markers was validated using external controls of cattle tissue. For negative controls, the primary antibodies were replaced by Universal Negative Control Serum (Biocare) in tumoral sections of all cases.
IHC slides were analyzed by 3 pathologists (AV, IRS, and SPP). Expected immunoreactivities were evaluated semi-quantitatively in the entire section based on reaction intensity and percentage of labeled cells. Reaction intensity was estimated as 0 (no reaction), 1 (weak reaction), 2 (moderate reaction), and 3 (intense reaction). 43 The percentage of positive neoplastic cells for each tumor was categorized as 0 (no cells), 1 (1%-15% of cells), 2 (16%-50% of cells), and 3 (greater than 50% of cells). 43 The final score for each tumor was determined by consensus.
Results
Case Data
In this retrospective study of 15 years, 0.76% (19/2503) of the anatomopathological records of bovine tissues were diagnosed with pulmonary epithelial neoplasms. None of the cases were excluded based on the exclusion criteria. Seven cases (7/19, 37%) were identified as primary to the lung and classified as solid AD, acinar AD, mucinous AD, and adenosquamous carcinoma. The remaining neoplasms (12/19, 63.16%) were classified as SCC, metastases of uterine AD, metastasis of cholangiocarcinoma, and metastasis of hepatocellular carcinoma. Most of the affected animals were female (13/14, 93%), with ages ranging from 24 to 132 months (median of 36 months) and with dairy aptitude (11/15, 73%). The case data are detailed at Supplemental Table S2. Five cases were referred to the laboratory from field necropsies, and 14 cases were obtained from slaughterhouses.
Histologic Findings
Solid adenocarcinoma
Two cases (2/19, 11%) were diagnosed as solid AD, which were characterized by neoplastic cells predominantly arranged in solid nests (Fig. 1a). The solid nests occasionally had central necrosis (comedo-like pattern) and were admixed with small numbers of round to ovoid glandular structures (acini). The neoplastic cells were supported by fibrous stroma, with areas of moderate amounts of desmoplasia. The cells were polygonal with moderate to abundant, occasionally vacuolated cytoplasm and poorly defined borders. The nuclei varied from round to oval with coarse or finely stippled chromatin and 1 to 4 prominent nucleoli. In addition, there were small foci of necrosis associated with neutrophils. Alveolar edema, atelectasis, and hyperplasia of the bronchial-associated lymphoid tissue (BALT) were observed in the adjacent parenchyma. In 1 of the cases, the presence of intracellular and extracellular mucin was observed with both PAS and AB stains.
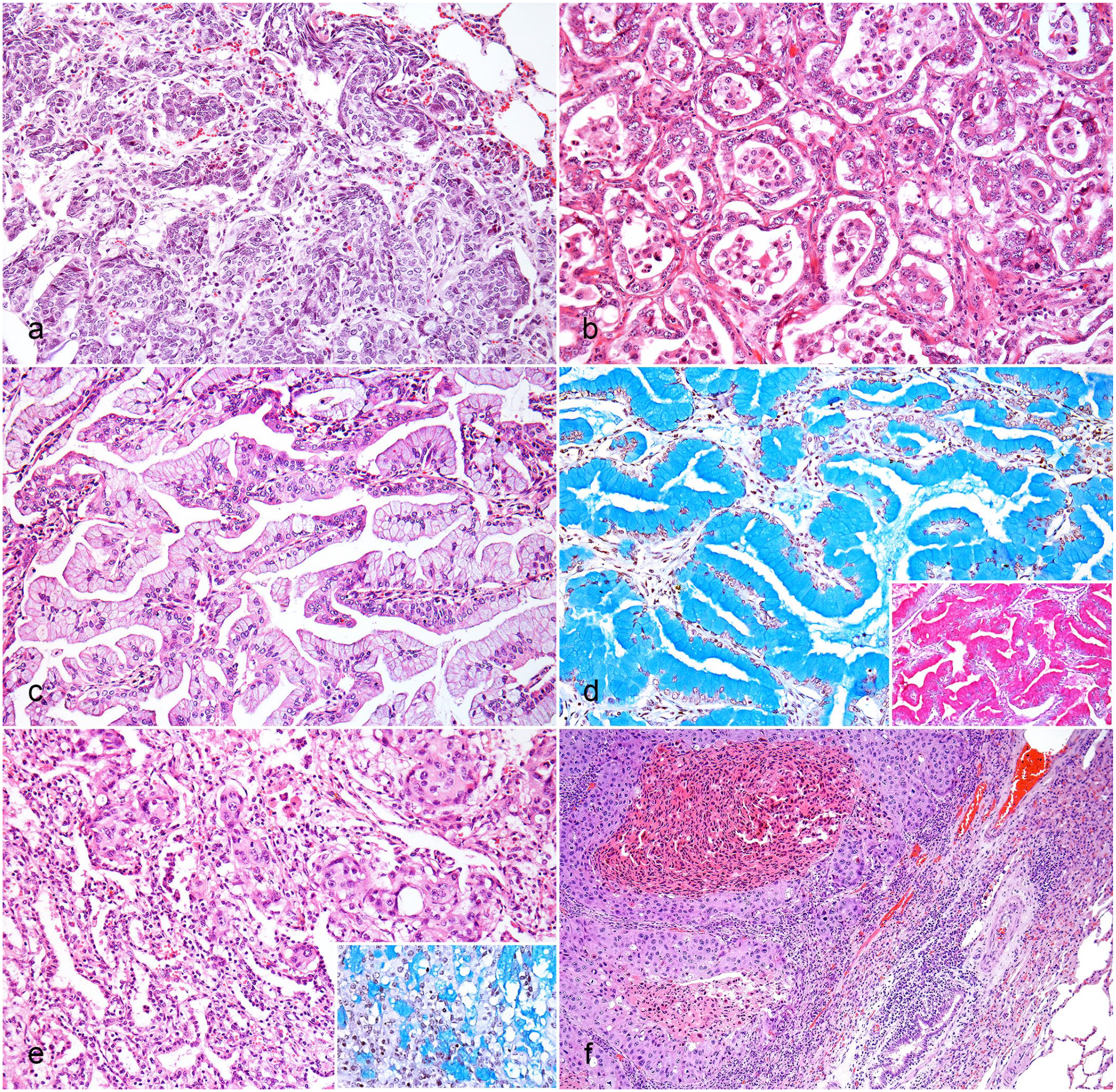
Pulmonary carcinomas, cattle: (a) Solid AD. Neoplastic cells arranged in solid nests and supported by fibrous stroma. HE. (b) Acinar AD. Neoplastic cells arranged in acini and supported by fibrovascular stroma. The acini are filled with cellular debris and foamy macrophages. HE. (c) Mucinous AD. Neoplastic cells arranged in papillae, filled with intracellular mucin, and supported fibrovascular stroma. HE. (d) Mucinous AD. Intracellular and extracellular mucin within the neoplasm. AB. Inset: abundant intracellular and extracellular mucin. Periodic acid-Schiff. (e) Adenosquamous carcinoma. Neoplastic cells arranged in large solid nests with squamous and glandular differentiation. HE. Inset: abundant intracellular and extracellular mucin. AB. (f) SCC. Neoplastic cells are arranged in solid nests, frequently with a comedo-like pattern, and supported by fibrovascular stroma. HE. AB, alcian blue; AD, adenocarcinoma; HE, hematoxylin and eosin; SCC, squamous cell carcinoma.
Acinar adenocarcinoma
The single case (1/19, 5%) diagnosed as acinar AD was characterized by circular structures composed of single to multiple layers of cuboidal epithelial cells (Fig. 1b) with central lumens often filled with cellular debris and foamy macrophages. These structures were supported by small amounts of fibrovascular stroma with areas of moderate amounts of desmoplasia. Cells ranged from cuboidal to polygonal with moderate amounts of eosinophilic cytoplasm, which was rarely vacuolated, and distinct cell margins. The nuclei varied from round to oval with coarsely stippled chromatin and 1 to 5 prominent nucleoli. In addition, foci of necrosis associated with degenerated neutrophils and mineral were observed. In the remaining parenchyma, alveolar edema was observed. No intracellular or extracellular mucin was observed with PAS or AB stains.
Mucinous adenocarcinoma
Mucinous AD was diagnosed in a single case (1/19, 5%). The neoplastic cells were arranged in acini (nonmucinous areas) and papillae (mucinous areas) (Fig. 1c) and supported by small amounts of fibrovascular stroma. The papillae were lined by a monolayer of pseudostratified columnar cells. Cells were columnar with poorly defined cell borders and abundant cytoplasm filled with intracellular mucin. In the acinar areas, the cytoplasm was vacuolated. The nuclei were basally oriented with finely stippled to condensed chromatin and inconspicuous nucleoli. In the papillary areas, the nuclei varied from oval to round with finely stippled chromatin and 1 to 2 prominent nucleoli. Within the tumor, small foci of necrosis associated with foamy macrophages and mineral were observed. Abundant intracellular and extracellular mucin was observed with the PAS and AB stains (Fig. 1d).
Adenosquamous carcinoma
Adenosquamous carcinoma was diagnosed in 3 cases (3/19, 156%) and was characterized by both glandular and squamous components. The glandular component was arranged in acini and solid nests, occasionally with a comedo-like pattern (Fig. 1e). The cells were supported by small to moderate amounts of fibrovascular stroma with small to moderate amounts of desmoplastic tissue. The cells of the squamous component were polygonal with distinct cell borders and abundant, homogeneous, eosinophilic cytoplasm. Nuclei ranged from round to oval to pleomorphic with coarse or finely stippled chromatin and 1 to 4 prominent nucleoli. Medium-sized foci of necrosis associated with inflammatory infiltrates of degenerate neutrophils were also seen. In the adjacent parenchyma, alveolar edema and BALT hyperplasia were observed. Mucin deposition was identified in only 1 case by both PAS and AB stains.
Squamous cell carcinoma
SCC was diagnosed in 6 cases (6/19, 32%). These were characterized by neoplastic cells predominantly arranged in solid nests and islands (Fig. 1f), occasionally with comedo-like patterns. The cells were supported by small to large amounts of fibrovascular stroma and desmoplastic tissue. In most cases, intercellular junctions, individual cell keratinization, and occasional keratin pearls were evident. The cells were polygonal with moderate to abundant, eosinophilic cytoplasm and distinct cell borders. Nuclei ranged from round to oval to pleomorphic with fine to coarsely stippled chromatin and 1 to 4 prominent nucleoli. It was also possible to observe small to medium-sized foci of necrosis associated with neutrophils and, occasionally, mineral. In 1 case, hypertrophy of the pulmonary arteries associated with collagen deposition was also observed. In the adjacent parenchyma, there was alveolar edema, proliferation of type 2 pneumocytes and alveolar macrophages, and BALT hyperplasia.
Metastases of uterine adenocarcinoma
Metastases of uterine AD were diagnosed in 4 cases (4/19, 21%). The neoplastic cells in the lung were arranged in acini (Supplemental Fig. S1a) supported by moderate to large amounts of fibrovascular stroma with moderate to large amounts of desmoplastic tissue. Occasionally, acini were filled with cellular debris and neutrophilic and histiocytic exudate. In 1 case, solid nests were seen. The cells ranged from cuboidal to polygonal with indistinct cell borders. The nuclei were round to oval with coarsely stippled to dense chromatin and multiple prominent nucleoli. Small foci of intratumoral necrosis and hemorrhage were seen. In 1 case, hypertrophy of the tunica media of pulmonary arteries and vasculitis were also observed. In all of these cases, the uterus had a primary epithelial neoplasm similar to that in the lung (Supplemental Fig. S1b).
Metastasis of hepatocellular carcinoma
Metastasis of hepatocellular carcinoma was diagnosed in 1 case (1/19, 5%). The neoplastic cells had an arrangement that varied from solid to trabecular (Supplemental Fig. S1c), supported by a thin fibrovascular stroma. The cells were polygonal with abundant, eosinophilic, vacuolated cytoplasm and distinct cell borders. The nuclei ranged from round to oval with coarsely stippled chromatin and 1 to 3 prominent nucleoli. Small foci of necrosis associated with mineral and degenerate neutrophils were also observed. In the remaining parenchyma, there was BALT hyperplasia. In this case, the liver had a neoplastic proliferation similar to that in the lung (Supplemental Fig. S1d).
Metastasis of cholangiocarcinoma
Metastasis of cholangiocarcinoma was diagnosed in 1 case (1/19, 5.26%). The cells were predominantly arranged in ducts (Supplemental Fig. S1d) supported by large amounts of fibrovascular stroma with moderate amounts of desmoplastic tissue. The cells were cuboidal with eosinophilic cytoplasm and poorly defined cell borders. The nuclei were round with finely stippled chromatin and 1 to 3 prominent nucleoli. In this case, the liver had a neoplastic proliferation similar to that in the lung (Supplemental Fig. S1e).
Data about the histologic distribution, anisocytosis, anisokaryosis, other criteria of malignancy, mitotic count, intratumoral inflammatory infiltrates, and LVI in each neoplastic type are available in Table 1.
Histological features of 19 bovine pulmonary carcinomas according to the histological subtype.
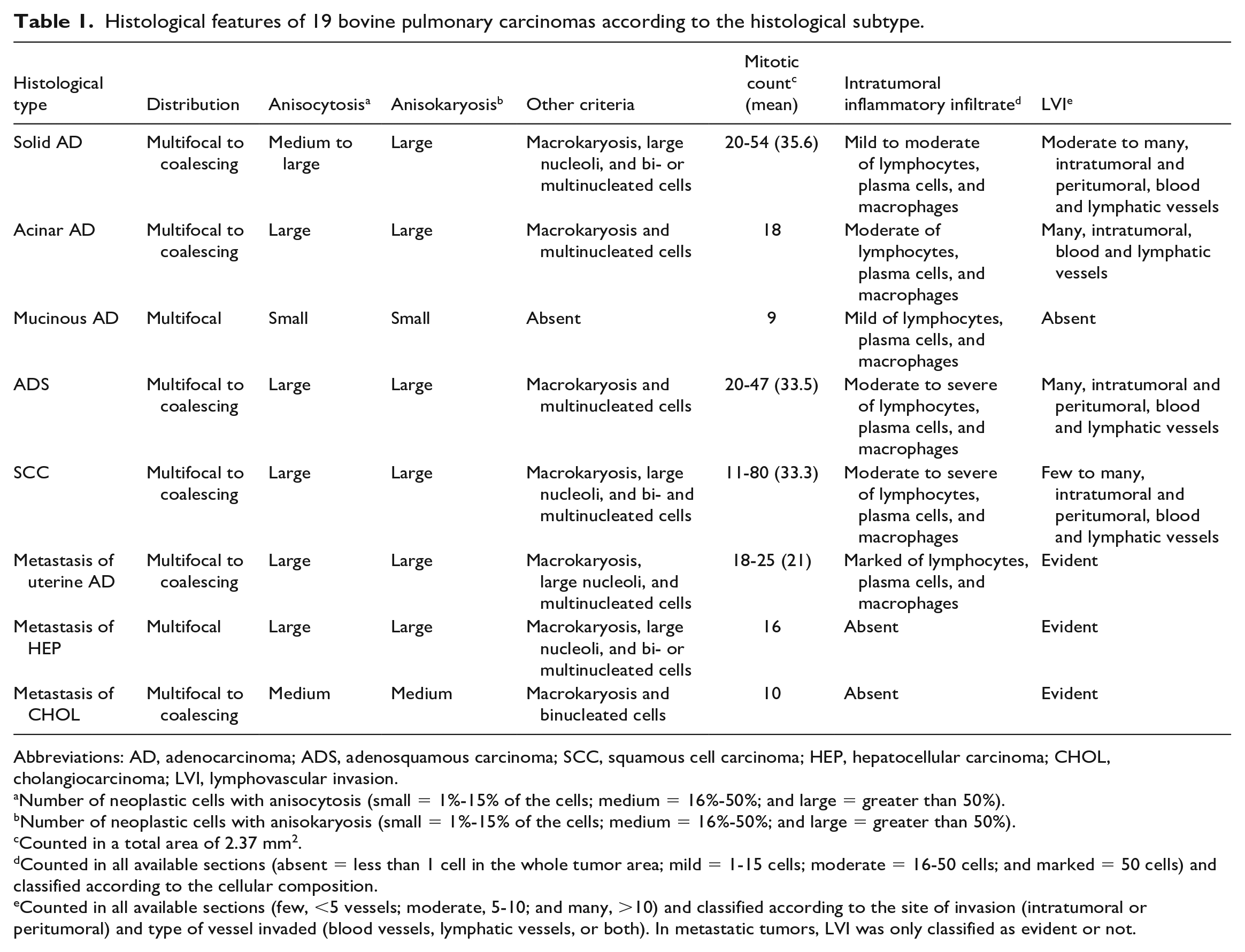
Abbreviations: AD, adenocarcinoma; ADS, adenosquamous carcinoma; SCC, squamous cell carcinoma; HEP, hepatocellular carcinoma; CHOL, cholangiocarcinoma; LVI, lymphovascular invasion.
Number of neoplastic cells with anisocytosis (small = 1%-15% of the cells; medium = 16%-50%; and large = greater than 50%).
Number of neoplastic cells with anisokaryosis (small = 1%-15% of the cells; medium = 16%-50%; and large = greater than 50%).
Counted in a total area of 2.37 mm2.
Counted in all available sections (absent = less than 1 cell in the whole tumor area; mild = 1-15 cells; moderate = 16-50 cells; and marked = 50 cells) and classified according to the cellular composition.
Counted in all available sections (few, <5 vessels; moderate, 5-10; and many, >10) and classified according to the site of invasion (intratumoral or peritumoral) and type of vessel invaded (blood vessels, lymphatic vessels, or both). In metastatic tumors, LVI was only classified as evident or not.
Immunohistochemical Findings
All external positive controls had expected immunoreactivity patterns (Supplemental Fig. S2a–f and S3a–d), except the normal kidney tissue immunolabeled for napsin A (Supplemental Fig. S3e). Data on the reaction intensity and percentage of labeled cells of each case are available in Supplemental Table S3.
Pancytokeratin
All primary and metastatic carcinomas diagnosed in the lung (19/19, 100%) had positive immunoreactivity in more than 50% positive neoplastic cells that ranged from weak (1/19, 5%) to moderate (3/19, 16%) to intense (15/19, 79%) (Fig. 2a; Supplemental Fig. S3f, S4a).
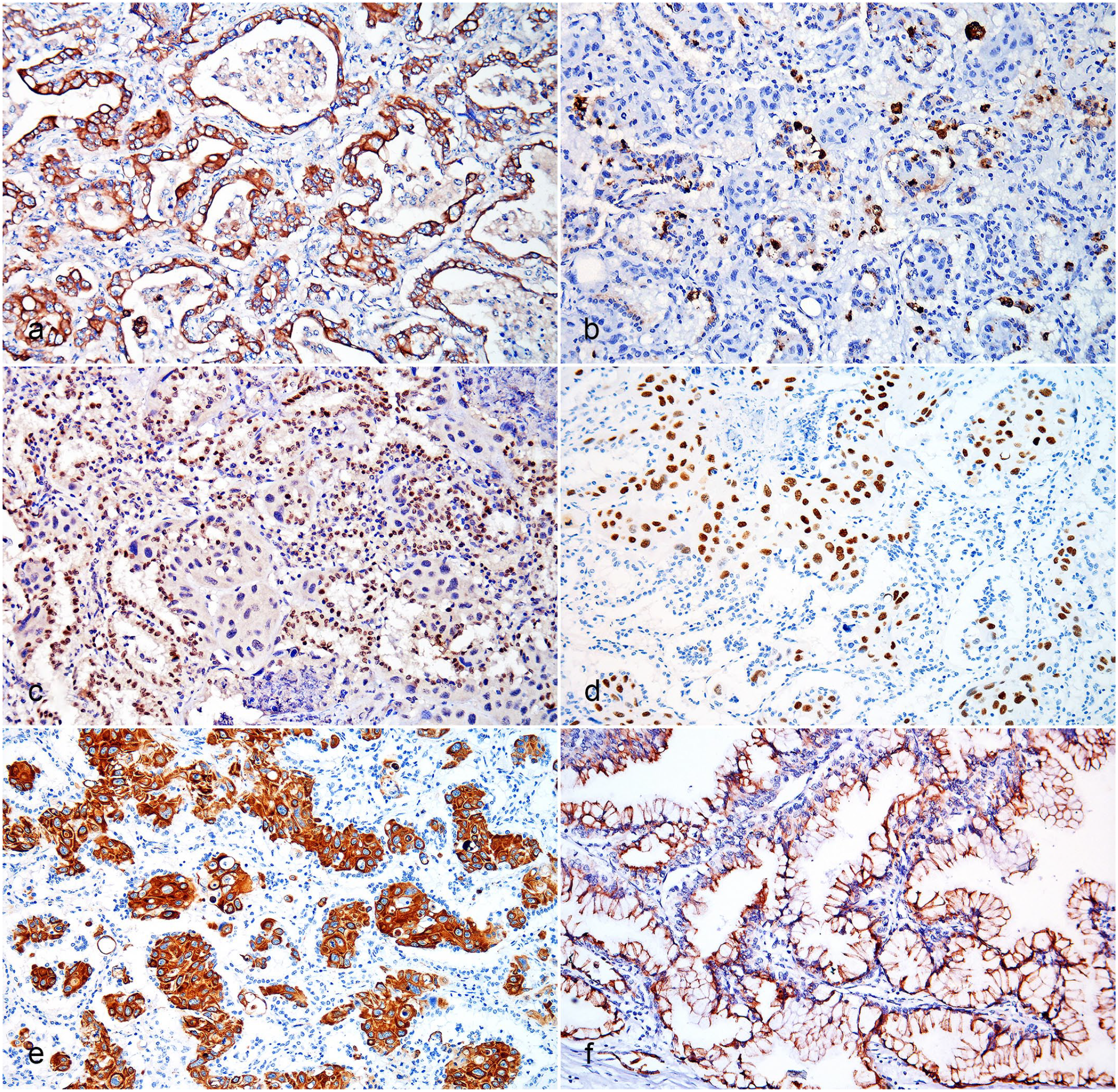
Pulmonary carcinomas, cattle: (a) Acinar AD. Diffuse cytoplasmic panCK immunoreactivity in neoplastic cells. IHC for panCK; (b) ADS. Granular cytoplasmic napsin A immunoreactivity in neoplastic cells of the glandular component and alveolar macrophages. IHC for napsin A; (c) ADS. Diffuse nuclear immunoreactivity to TTF-1 in the glandular component. IHC for TTF-1; (d) ADS. Diffuse nuclear p40 immunoreactivity in squamous neoplastic cells. IHC for p40; (e) ADS. Diffuse cytoplasmic CK 5/6 immunoreactivity in squamous cells. IHC for CK5/6; (f) mucinous AD. Diffuse cytoplasmic CK7 immunoreactivity in the neoplastic cells. IHC for CK7. AD, adenocarcinoma; ADS, adenosquamous carcinoma; CK, cytokeratin; IHC, immunohistochemistry; panCK, pancytokeratin; TTF-1, thyroid transcription factor-1.
Vimentin
There was positive immunoreactivity for vimentin in 4 of the tumors (4/19, 21%; 1 metastatic hepatocellular carcinoma, 2 SCCs, and 1 metastatic uterine AD). In the metastatic hepatocellular carcinoma, intense immunoreactivity was observed in 16% to 50% of neoplastic cells (Supplemental Fig. S4b). In the 2 cases of SCCs, there was weak immunoreactivity (2/2) in less than 15% of the neoplastic cells (2/2). In the metastatic uterine AD, moderate immunoreactivity was observed in 16% to 50% of the neoplastic cells. In all cases, vimentin expression was observed in the same cells in which panCK was expressed. The remaining cases were negative.
Napsin A
Only 2 tumors (2/19, 11%; all primary adenosquamous carcinomas) had positive immunoreactivity for napsin A. In both cases, there was intense immunoreactivity in less than 16% of neoplastic cells (2/2) (Fig. 2b). Immunoreactivity was only observed in the glandular component. The remaining cases were negative.
Thyroid transcription factor 1
Six carcinomas (6/19, 32%) had positive immunoreactivity for TTF-1. In solid ADs, immunoreactivity was weak (1/2) or moderate (1/2) and noted in less than 16% of neoplastic cells (2/2). In the acinar AD, moderate immunoreactivity was observed in less than 16% of neoplastic cells. In the 3 adenosquamous carcinomas (Fig. 2c), the immunoreactivity ranged from moderate in 16% to 50% of neoplastic cells (2/3) to intense in more than 50% of neoplastic cells (1/3). Immunoreactivity was only observed in the glandular component. The mucinous AD was the only primary pulmonary neoplasm with no immunoreactivity for TTF-1. The remaining cases were negative.
p40
Immunoreactivity for p40 was seen in 7 carcinomas (7/19, 37%; 3 primary adenosquamous carcinomas and 4 SCCs). In adenosquamous carcinomas, there was intense immunoreactivity for p40 in more than 50% of neoplastic cells (3/3), but only in the squamous component (Fig. 2d). In SCCs, immunoreactivity ranged from weak in 16% to 50% of neoplastic cells (1/4) to moderate in 16% to 50% of neoplastic cells (2/4) to moderate in more than 50% of neoplastic cells (1/4). The remaining cases were negative.
Cytokeratin 5/6
Immunoreactivity for CK5/6 was seen in 9 carcinomas (9/19, 47%; 3 primary adenosquamous carcinomas and 6 SCCs). In adenosquamous carcinomas, there was intense immunoreactivity for CK5/6 in more than 50% of neoplastic cells (3/3) and only in the squamous component (Fig. 2e), as seen for the p40 marker. In SCCs, immunoreactivity ranged from moderate in more than 50% of neoplastic cells (2/6) to intense in 16% to 50% of neoplastic cells (1/6) to intense in more than 50% of neoplastic cells (3/6) (Supplemental Fig. S4c).
Cytokeratin 8/18
Only 2 carcinomas (2/19, 11%; 1 primary adenosquamous carcinoma and 1 SCC) were positive for CK8/18. The adenosquamous carcinoma had weak immunoreactivity in 16% to 50% of neoplastic cells, specifically in the squamous component. The SCC had intense immunoreactivity for CK8/18 in less than 16% of neoplastic cells. The remaining cases were negative.
Cytokeratin 7
Immunoreactivity for CK7 was seen in 10 carcinomas (10/19, 53%; 2 primary adenosquamous carcinomas, 1 primary mucinous AD, 1 metastatic cholangiocarcinoma, 1 metastatic hepatocellular carcinoma, 3 SCCs, and 2 metastatic uterine AD). In adenosquamous carcinomas, the immunoreactivity ranged from weak (1/2) to intense (1/2) in more than 50% of neoplastic cells (2/2), specifically in the glandular component. Pulmonary mucinous AD had intense immunoreactivity in more than 50% of the neoplastic cells (Fig. 2f). There was moderate immunoreactivity in more than 50% of metastatic cholangiocarcinoma cells, while the metastatic hepatocellular carcinoma had intense immunoreactivity in less than 16% of cells. In SCCs, the immunoreactivity ranged from weak (2/3) to moderate (1/3) in more than 50% of neoplastic cells (3/3). Both uterine AD had weak immunoreactivity in 16% to 50% of neoplastic cells (2/2). The remaining cases were negative.
Cytokeratin 20
Immunoreactivity for CK20 was seen in 11 carcinomas (11/19, 58%; 2 primary adenosquamous carcinomas, 1 primary acinar AD, 1 primary mucinous AD, 1 metastatic cholangiocarcinoma, 1 metastatic hepatocellular carcinoma, and 5 SCCs). The adenosquamous carcinomas had moderate immunoreactivity in more than 50% of neoplastic cells (2/2), specifically in the glandular component. The acinar AD had weak immunoreactivity in more than 50% of neoplastic cells. In the pulmonary mucinous AD, there was moderate immunoreactivity for CK20 in more than 50% of neoplastic cells. There was moderate immunoreactivity in more than 50% of metastatic cholangiocarcinoma cells. An intense immunoreactivity in less than 16% of the metastatic hepatocellular carcinoma cells was seen. In SCCs, immunoreactivity was weak and was distributed either in 16% to 50% of neoplastic cells (1/5) or in more than 50% of neoplastic cells (4/5). The remaining cases were negative.
HepPar-1
Immunoreactivity for HepPar-1 was seen in only 1 carcinoma (1/19, 5%; metastatic hepatocellular carcinoma). This case had intense immunoreactivity for HepPar-1 in 16% to 50% of neoplastic cells (Supplemental Fig. S4d). The remaining cases were negative.
Discussion
Although the initial diagnoses of all pulmonary neoplasms in this study were based on histological findings (Fig. 3), IHC was used in the diagnostic confirmation. An immunohistochemical panel containing markers for pneumocytes and squamous cells was applied to confirm pulmonary origin and/or differentiate between AD, adenosquamous carcinoma, and SCCs, mainly when the tumor did not allow reliable morphologic classification. In the cases of metastatic uterine AD, the absence of TTF-1 and napsin A labeling helped to exclude primary pulmonary carcinoma. The use of CK20 and CK7 helped rule out metastatic colorectal AD in the case of pulmonary mucinous AD. The use of HepPar-1 and CK7 helped in the confirmation of the case of metastatic hepatocellular carcinoma and metastatic cholangiocarcinoma, respectively. Therefore, IHC was a valuable complementary tool in determining the origin of pulmonary tumors and the final diagnosis in most of our bovine cases. IHC has been widely used for this purpose in human medicine21,25,27,32,42,47,50–53 and in companion animal veterinary medicine.8,11,15,41 To the best of our knowledge, this is the first time it has been applied to pulmonary tumors in cattle.
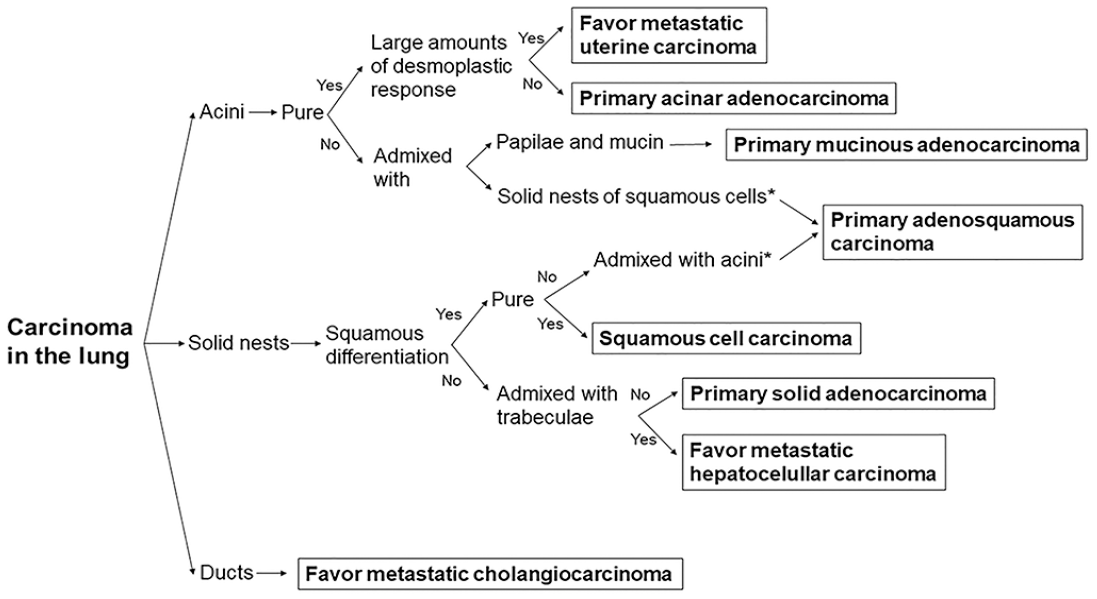
Diagnostic algorithm for diagnosing carcinomas with pulmonary involvement in cattle based on the histological features. The asterisk represents that a minimum of 10% of each pattern (glandular and squamous) is required for the diagnosis of adenosquamous carcinoma. The histological types of neoplasms included only reflect the results of this study.
Adult cows were the most affected group in this study, similar to that reported by other authors.4,31,35 The authors believe that females were overrepresented in this study because most samples were obtained from slaughterhouses in which cows are usually slaughtered at an older age than bulls, as previously observed.4,59 The large number of cattle with dairy aptitude probably also contributed to this frequency.
Pulmonary AD was the main primary carcinoma diagnosed in this study, followed by adenosquamous carcinoma. These data corroborate previous literature, which reported that AD is the most frequent primary pulmonary neoplasm diagnosed in cattle,4,31 as well as in companion animals8,11,15,43 and in humans.51,52 Unlike what has been reported in dogs and cats, in which papillary AD is the main subtype,1,8,11,43 solid AD was the most frequent subtype in our work. The less common subtypes were acinar and mucinous AD. Given the small number of cases in this study, the actual frequency of the various subtypes of primary pulmonary carcinoma remains uncertain.
Histologically, pulmonary solid AD is characterized by polygonal cells forming nests, as described in this study, and may have intracellular or extracellular mucin. 50 In the cases of our study, only 1 solid AD had extracellular mucin with both PAS and AB stains. On the contrary, acinar AD, another subtype observed in our study, is histologically characterized by oval to rounded glands with a central lumen surrounded by neoplastic cells, which may or may not contain mucin.5,46,50 These features were observed in our case, except for mucin production, which was not detectable by either PAS or AB stains.
Although mucinous AD is considered the rarest pulmonary neoplasm in humans, 32 and there are no reports in domestic animals, we diagnosed 1 case in this study. This subtype is histologically characterized by globular and/or columnar cells with abundant intracytoplasmic mucin and small basally oriented nuclei. 50 Nuclear atypia is usually mild or absent, 50 and the surrounding alveolar spaces are often filled with mucin.32,50,51 This subtype may also have a variety of histological patterns and may coexist with nonmucinous areas, 38 as was observed in our study. Primary pulmonary ADs with abundant mucin production may be histologically indistinguishable from mucin-producing metastatic ADs, 66 so an immunohistochemical panel is important in differentiating these neoplasms, especially in regard to intestinal carcinoma, which lacks CK7 immunoreactivity. 50
Pulmonary adenosquamous carcinomas are neoplasms that have both squamous and glandular components and require at least 10% of each component for the diagnosis. This criterion is based on the current WHO classification for pulmonary tumors in humans 50 and adapted by the authors for pulmonary neoplasms in cattle. Confirmation can be performed with an immunohistochemical panel that contains both squamous cell and pneumocyte markers. 50 In humans, immunoreactivity for CK5 and p40 has been used as an indicator of squamous differentiation. 58 In this study, the squamous component of all adenosquamous carcinomas showed immunoreactivity for both CK5/6 and p40. In addition, IHC for TTF-1 and/or napsin A confirmed the glandular component and pulmonary origin. Moreover, 1 of the neoplasms had weak immunoreactivity for CK8/18 in the glandular epithelium. These results are consistent with what has been described in the human literature. 50
In this study, TTF-1 was an excellent immunomarker for primary pulmonary neoplasms in cattle. There are no previous studies evaluating the frequency of immunoreactivity for this marker in primary pulmonary neoplasms in cattle. In humans and dogs, IHC for TTF-1 has a relatively high specificity and moderate sensitivity for diagnosing primary pulmonary neoplasms.41,42,47 Thyroid tumors may also be immunoreactive for TTF-1, but these are rare in cattle and were not detected in any cases of the current investigation. In our study, only 1 primary pulmonary neoplasm showed no immunoreactivity for TTF-1, specifically the mucinous AD. The absence of immunoreactivity for TTF-1 and napsin A in this case was expected, since these neoplasms have a different immunohistochemical profile from other pulmonary ADs19,66 and are typically positive for CK7 and CK20.19,50
Napsin A, a relatively new immunomarker, is an aspartic proteinase expressed in the lung and kidney. 13 There are few investigations evaluating this immunomarker in domestic animals and no studies in cattle; however, it is used for the diagnosis of nonsmall cell pulmonary AD in humans with 94.3% specificity. 53 A recent study on napsin A in dogs also demonstrated a high sensitivity of 92%. 8 This marker is also used in the diagnosis of canine renal carcinomas and human renal and thyroid carcinomas.9,39,40 Herein, positive immunoreactivity in bovine primary pulmonary carcinomas was infrequently observed; only 1 solid AD and 1 adenosquamous carcinoma were positive. Thus, napsin A does not appear to be a good marker for primary pulmonary neoplasms in cattle, although normal bovine respiratory tract cells (internal control) have positive immunoreactivity. Unfortunately, the only technique for validation of the protocol in our study was the use of normal canine kidney tissue, which is known to be positive. 39 Other attempts using more refined techniques (eg, correlation with western blot, corroborating IHC results with supportive data from other assays or bioinformatics approaches) 63 should be used in future works.
The origin of the SCCs diagnosed in our study is unknown because not all organs were sent for histological analysis and, for this reason, the SCCs cannot be classified as primary or metastatic. Cattle with depigmented skin and hair are highly predisposed to develop cutaneous SCCs on the face, eyelids, ears, dorsum, and vulva; 33 however, these parts may not be observed during slaughter inspection due to leather removal. In addition, although SCCs of the digestive tract are rare in most regions of the world, 54 there is a high incidence of these neoplasms associated with bracken fern consumption in cattle in southern Brazil.17,31 In these cases, metastases frequently occur in regional lymph nodes, liver, and lung. 17 SCCs usually have intense and diffuse immunoreactivity for squamous cell markers, such as p40, CK5, and CK5/6,43,50 as observed in this study. These immunomarkers have been used in humans 50 and companion animals. 43 Currently, there are no specific immunomarkers to differentiate primary from metastatic pulmonary SCC in veterinary medicine. In human medicine, p16 has been suggested as a useful marker for distinguishing these neoplasms; 61 however, p16 has not been validated in cattle tissues.
Uterine ADs occur frequently in adult cows and are frequent incidental findings in slaughterhouses. 3 Metastases of these neoplasms to the lung and lymph nodes are relatively common in cattle. 44 In a study carried out by Lucena et al., 31 13 of 17 uterine neoplasms had pulmonary metastases. Although IHC was not performed in those cases, some histological features may help to determine the origin of the neoplasms, such as marked desmoplasia and interspersed acinar structures.6,48 Another characteristic histological feature of these neoplasms is marked cellular pleomorphism, 48 which was also observed in our study. In all cases herein described, it was possible to evaluate the neoplasm in the uterus, which was histologically similar to the pulmonary metastases. Macroscopic and microscopic evaluation of the uterus is extremely important for the identification of this neoplasm, since the primary lesions are usually smaller than their metastases. 35
Hepatic carcinomas infrequently metastasize to the lung, but when present, lesions are similar to those observed in the primary site (liver). 59 A study of immunomarkers in hepatic lesions of cattle showed that HepPar-1 is a good immunomarker for hepatocellular carcinomas; however, CK7 demonstrated a low sensitivity for cholangiocarcinomas. 59 In our investigation, the CK7 and HepPar-1 were good immunomarkers for pulmonary metastases from these neoplasms in cattle. The authors believe these conflicting results regarding CK7 may be correlated to the high susceptibility of this antigen to long fixation times.7,29,62 While some neoplastic hepatocytes in our study had positive immunoreactivity for CK7, previous studies reported that this immunoreactivity can be expected in hepatocellular carcinomas, and it has no influence on the final diagnosis, due to the heterogeneity of neoplastic cells.56,57
Co-expression of panCK and vimentin was observed in 4 neoplasms in this study: 1 metastatic hepatocellular carcinoma, 2 SCCs, and 1 metastatic uterine AD. Vimentin is produced in epithelial-mesenchymal transition, a process of cellular reprogramming, in which epithelial cells acquire a mesenchymal phenotype; change their shape; and have more motility,18,28,55 increased tumor invasiveness, 2 and an additional role in tumorigenesis. 27 This could explain the co-expression of vimentin and panCK in these tumors. Increased expression of vimentin has already been reported in several tumor cell lines and tissues in humans, including endometrial tumors and liver tumors.24,26 Co-expression of vimentin has been reported in numerous neoplasms of domestic animals, including anaplastic carcinomas, amelanotic melanomas, renal carcinomas, prostatic carcinoma, hepatocellular carcinoma, and pulmonary carcinomas.10,16,20,43,45 Occasionally, the expression of vimentin is more marked in the metastasis than in the primary tumor. 26
In summary, SCCs, metastases of uterine AD, and primary solid AD were the most common pulmonary carcinomas in our study. IHC was a complementary tool in reaching the histological diagnosis, mainly to confirm primary pulmonary neoplasms, which is not always possible using histology alone. TTF-1 was an excellent marker for primary pulmonary carcinomas in cattle. The relatively small sample size and the lack of gross characterization are important limitations in our study. Further studies with different techniques for validation of the napsin A protocol are needed. Despite that, this study highlighted the possibility of using new immunohistochemical markers in cattle.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231192373 – Supplemental material for Histological and immunohistochemical features of carcinomas with pulmonary involvement in cattle
Supplemental material, sj-pdf-1-vet-10.1177_03009858231192373 for Histological and immunohistochemical features of carcinomas with pulmonary involvement in cattle by Andréia Vielmo, Igor Ribeiro Santos, Manoela Marchezan Piva, Marcele Bettim Bandinelli, Saulo Petinatti Pavarini, Welden Panziera and David Driemeier in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was supplied by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)—Finance Code 001.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
