Abstract
This report presents a rare, spontaneous, heterologous, malignant mixed Müllerian tumor observed in a 98-week-old untreated Wistar rat. At necropsy, the right uterine horn was dilated and contained a mass of approximately 10 × 15 mm. Histopathologically the mass consisted of 2 parts with different morphologic characteristics, a polypoid mass protruding into the uterine lumen and a more solid part that replaced the adjacent uterine wall. The solid part exhibited characteristics of a highly infiltrative adenocarcinoma, whereas the composition of the polypoid mass was heterogeneous, consisting of a variety of benign and malignant epithelial and mesenchymal elements. The epithelial components included well-differentiated endometrial and squamous epithelium juxtaposed to carcinosarcomatous areas. The mesenchymal components consisted of well-differentiated cartilage and bone along with chondrosarcomatous, rhabdomyomatous, and other mesenchymal elements. Immunohistochemical analysis further supported the diagnosis of malignant mixed Müllerian tumor.
Keywords
Malignant mixed Müllerian tumors (MMMTs) or carcinosarcomas of the uterus are well described in women and account for 2%–5% of all malignancies of the uterine corpus. 1 MMMTs also have been diagnosed in the vagina, cervix, Fallopian tubes, and ovaries. In domestic and laboratory animals, MMMTs are very rare and have only been described in 1 cat, 4 1 rabbit, 5 and 5 Lewis rats. 6 Ernst et al 3 observed MMMT in 2 Wistar rats, with no other evidence of MMMTs reported in the literature in this strain.
Other types of uterine neoplasms occur at a relatively high frequency in the Wistar rat. The incidence of tumors recorded in the uterus of a total of 4,060 female control Wistar rats in 70 two-year studies was 18.4%. 2 Neoplasms included in this survey consisted of stromal polyps (11.9%), adenocarcinomas (5.7%), and adenomas (0.8%).
In the case presented here, an MMMT arose in a female Wistar rat (Hsd/Cpb:WU Spf) supplied by Harlan, Horst, The Netherlands. The animal was used as an untreated control in a 2-year carcinogenicity study. Animals in this study were housed in pairs of the same sex in Macrolon (type III) cages with sterilized bedding (wood shavings; Woodyclean, type 8/15, BMI, Helmond, The Netherlands) and a stainless steel shelter kept behind a barrier in a room under standard conditions (temperature 21°C ± 3°C; relative humidity ≥40%; artificial light cycle on a 12-hour light/dark base; air renewals 13–17 times per hour). The animals had free access to pelletted food (diet RHM-TM, supplied by Hope Farms B.V., Woerden, The Netherlands) and free access to regularly refreshed tap water from drinking bottles. During the in-life period the animals were observed daily for morbidity and mortality and observed for any behavioral and physical abnormalities. At the start of the study the animals were 7 weeks old.
On day 685 of the study, this female rat was found dead. Externally, the animal showed a large round mass in the low abdominal/genital region and alopecia of the right axillary region.
At necropsy a mass with a diameter of 10 × 15 mm was seen in the right uterine horn. This uterine horn was dilated and filled with a thick brownish fluid. Additional findings were seen in the abdominal cavity (some necrotic debris), skin (grossly noted subcutaneous nodule in the low abdominal/genital region of 50 × 30 mm), spleen (enlarged), left ovary (transparent cyst), and pituitary gland (slightly enlarged and pale). All organs/tissues were fixed in 4% buffered formaldehyde (formalin 10%, Klinipath, The Netherlands), eyes and optic nerves were initially fixed in Davidson’s fluid, and pituitary gland was preserved in sublimate formalin.
Following fixation, organs and tissues were dehydrated, embedded in paraffin wax, processed as approximately 5-μm-thick sections, and stained with hematoxylin and eosin (HE). Additional immunohistochemical stainings were performed to further identify the characteristics of the different cell types within the observed uterine tumor, including anti-desmin (mouse anti-human clone, D33; dilution 1:50), anti-pancytokeratin, keratin wide-spectrum screening (rabbit, polyclonal; dilution 1:1000), anti-S100 (rabbit, polyclonal; dilution 1:200), anti–smooth muscle actin (SMA) (mouse anti-human, clone 1A4; dilution 1:50), and anti-vimentin (mouse, clone V9; dilution 1:200), all supplied by DakoCytomation (Heverlee, Belgium). All slides for immunohistochemistry were pretreated using an antigen retrieval method. All antibodies were visualized using the Envision+ system (horseradish peroxidase, anti-mouse, or anti-rabbit) from DakoCytomation.
Microscopic examination of the HE-stained section of the uterus showed a tumor consisting of 2 parts, a polypoid part protruding into the lumen of the uterus and a solid part infiltrating into the uterine wall and protruding into the abdominal cavity (Fig. 1). There was a clear transition of the base of the polypoid part to the solid part of the tumor.
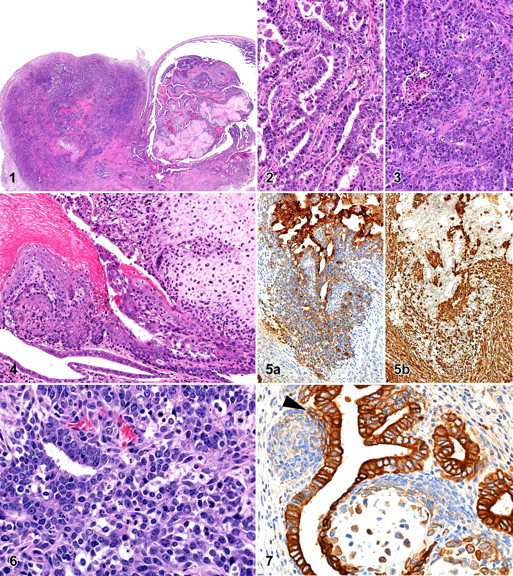
The solid part was densely cellular, poorly demarcated, and not encapsulated, and it had infiltrated into the endometrium and myometrium. It consisted of tubules of closely packed malignant epithelial cells, in some areas with a scirrhous reaction. In other areas tubular structures were less distinct, and a more solid growth pattern was observed (Figs. 2 and 3). The epithelial cells in these areas had a moderate amount of strongly basophilic cytoplasm, indistinct cell borders, and a large, round to oval centrally located nucleus with coarse chromatin and often 1 to several prominent nucleoli. Mitotic figures were prominent. In the solid areas, necrotic cellular debris was sometimes found within ill-defined lumina formed by the tumor cells (Fig. 3). A slight lymphocytic inflammation was present in the surrounding tissues. This part of the tumor had the features of a moderately to poorly differentiated adenocarcinoma of the endometrium.
The polypoid part of this biphasic tumor had a completely different composition compared with the solid part within the wall of the uterus. The polypoid part was characterized by an admixture of varying structures that were epithelial, mesenchymal, or combinations of both.
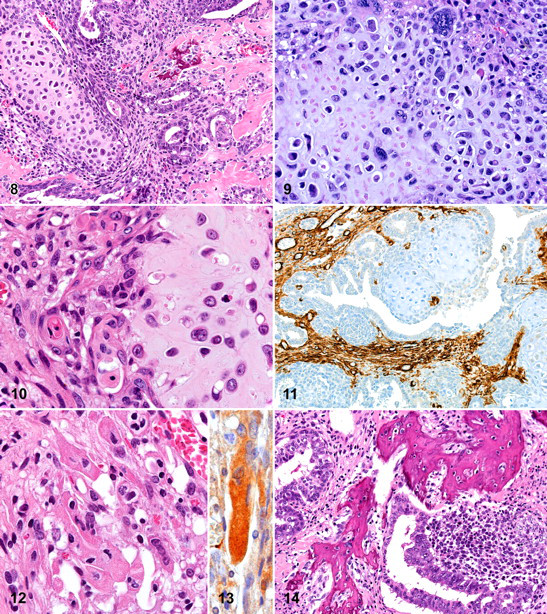
The epithelial component ranged from well-differentiated, benign-looking endometrial-type tubules, lined by a single layer of epithelial cells, to more irregular tubules lined by a multilayer epithelium. Solid nests consisting of undifferentiated cells were noted in close contact to the tubules. More or less in parallel, the epithelial cells in those different areas ranged from normal to atypical, respectively. The epithelial cells generally had round nuclei, often with prominent nucleoli. In several areas, the epithelium of the tubular structures showed squamous transformation (Fig. 4). This squamous epithelium showed normal keratinization and was considered metaplastic in nature. The nests of undifferentiated cells did not have well-defined boundaries and often were seen dispersing into the mesenchymal component of the mass (Figs. 5 and 6). The antipancytokeratin strongly stained the epithelial cells lining the tubular structures. The nests of undifferentiated cells only stained weakly positive using the antipancytokeratin, except for those cells that were most closely related to the tubular structures. Those cells stained positive using this antibody (Fig. 7).
About half of the polypoid mass consisted of areas showing mesenchymal characteristics. Rounded or lobular areas with cartilaginous differentiation were most notable, particularly because of their prominent extracellular hyaline chondroid matrix (Fig. 8). Within these areas, the cellular characteristics ranged from benign chondromatous with mononucleated to sometimes multinucleated chondrocytes (per lacunae) to chondrosarcomatous areas with atypical, slightly pleiomorphic chondrocytic cells with hyperchromatic nuclei. In the latter areas, the size of the nuclei varied greatly from very small to giant nuclei (Fig. 9). Interestingly, a number of nests composed of chondrocytic cells were surrounded by epithelial cells that did not rest on a basement membrane but appeared to have direct contact with the chondroid matrix deposited by the chondrocytic cells. In such regions, some epithelial cells clearly showed keratinization (Fig. 10). Next to these epithelial cells, which clearly stand out using the antipancytokeratin, were many chondrocytic-like cells that also expressed this epithelial marker (Fig. 7). Some of the nests of chondrocytic cells were found at similar locations as the nests of undifferentiated cells, thus closely related to the tubular structures. On some occasions undifferentiated cells were found surrounding the cartilaginous areas (Fig. 7).
Other parts of the mesenchymal component of the mass consisted of areas of undifferentiated spindle cells and anti-SMA–positive cells (probably mostly myofibroblasts, Fig. 11); in one area, obvious rhabdomyoblastic differentiation was present. The rhabdomyoblasts could readily be recognized based upon their elongated strap-like shape, abundant eosinophilic cytoplasm, and multinucleation (Fig. 12). Cross-striation was not clearly visible, possibly because the area in which those cells were found was degenerating. These cells were positive for anti-desmin (Fig. 13). Within the mesenchymal component, there were also areas with strongly hyalinized collagen and irregular shaped bony trabeculae (Fig. 14). These trabeculae with lacunae containing normal-looking osteocytes are believed to represent areas of stromal osseous metaplasia.
The anti-S100 immunohistochemical staining method was positive in the cartilaginous areas, but no morphologic signs of neuroectodermal differentiation were identified in the tumor.
Other microscopic findings of note reported in this Wistar rat were slight acute peritonitis, a thrombus in a liver vein, myeloid hyperplasia in the bone marrow, moderate lymphoid atrophy of the thymus, a cyst in 1 ovary, focal hyperplasia in the pars distalis of the pituitary gland, focal nodular hyperplasia in 1 adrenal gland, and a large fibroadenoma in the mammary gland. No metastases of the MMMT were observed.
In the embryo, 2 sets of paired tubular organs develop: the Wolffian (mesonephric or male) ducts and the Müllerian (paramesonephric or female) ducts. In the absence of testicular hormones, the Müllerian ducts undergo further development and form the female genital tract, whereas the Wolffian ducts degenerate (day 18 of gestation). 7 MMMTs are believed to arise from the pluripotent cells of the Müllerian ducts, which can give rise to cells of both epithelial and stromal types synchronously. 9 MMMTs are therefore characterized by an admixture of malignant epithelial and mesenchymal elements. The mesenchymal component can display a variety of patterns that have been referred to as homologous or heterologous. An MMMT is considered homologous when it displays differentiation toward cell types normally present in the uterus such as fibrous tissue, smooth muscle, and/or endometrial stroma-like tissue. In the heterologous type of MMMT, the mesenchymal part of the tumor contains areas with cell types not normally present in the uterus (eg, striated muscle, cartilage, bone, and/or adipose tissue).
The histopathologic characteristics of the polypoid part of the tumor described in this report, an admixture of malignant epithelial and malignant mesenchymal elements, combined with the immunohistochemical staining properties resemble those described for heterologous MMMT in human. 1,8 The mesenchymal components within this tumor included a.o. rhabdomyosarcomatous and chondrosarcomatous elements and therefore was diagnosed as a heterologous type of MMMT. The characteristics of the MMMT described here are quite similar to those described by Kaspareit-Rittinghausen and Deerberg. 6 However, chondrosarcomatous areas and areas with clear epithelial–mesenchymal transition as well as the immunohistochemical characteristics of this tumor-type, were not reported by those authors.
Various histogenetic mechanisms have been postulated to explain the biphasic carcinomatous–sarcomatous appearance of MMMTs. Speculation has centered on whether these tumors represent collision tumors, which are a mixture of 2 histogenetically distinct malignant cell populations; combination tumors, both elements of which arise from a common stem cell (referred to as “combination theory”); conversion tumors, in which the sarcomatous element derives from the carcinoma during the evolution of the tumor; or composition tumors, which are pure carcinomas with reactive, atypical, but benign stromal elements. 1,8 The combination theory is currently most widely accepted. In the case presented, the finding of undifferentiated cell nests, resembling the Müllerian blastema, supports the latter theory. The blastema-like structures suggest the presence of a pluripotential cell population as a possible source for the different elements within this tumor. The cells within these blastema-like structures seem to be able to differentiate into different epithelial and mesenchymal cell types. This is morphologically manifested by the presence of undifferentiated cells surrounding the cartilaginous areas and by the presence of squamous epithelial cells in close relationship to the cartilaginous areas, suggesting epithelial–mesenchymal transition. The immunohistochemical characteristics, the expression of epithelial markers in some cells within the undifferentiated cell nests and in the chondrosarcomatous elements of the MMMT, support this phenomenon.
The part of the tumor with features of a malignant adenocarcinoma that replaced a large part of the uterine wall and protruded in the abdominal cavity could be interpreted as being a local metastasis of the MMMT. Local or distant metastases are known to occur in MMMTs and are generally predominantly composed of the epithelial component only. 1 In some areas, however, the adenocarcinomatous part was continuous with the MMMT and no distant metastases were observed in this animal. We therefore concluded that this part represents one of the malignant epithelial components of this MMMT.
The most important differential diagnoses for MMMTs are primary or metastatic teratomas and adenosarcomas of the Müllerian duct. Teratomas are usually composed of mature and immature derivates of all 3 primitive germ layers. The absence of neuroectodermal elements and of well-differentiated organoid structures in the described tumor largely excludes this possibility. Adenosarcomas of the Müllerian duct are characterized by a combination of malignant stromal components and benign epithelial components. Heterologous parts in adenosarcomas of the Müllerian duct are uncommon, and the cellular atypia of the glandular part of the reported tumor suggests malignant instead of benign elements.
In conclusion, this report represents a histopathologic and immunohistochemical characterization of a MMMT in a Wistar rat that will further aid in the diagnosis of this very rare neoplasm in this species and strain.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
