Abstract
Avian poxvirus infections typically manifest as 2 forms: cutaneous (“dry”) pox, characterized by proliferative nodules on the skin, and diphtheritic (“wet”) pox, characterized by plaques of caseous exudate in the oropharynx and upper respiratory and gastrointestinal tracts. Systemic spread of virus to visceral organs beyond the skin and mucous membranes is rarely reported. Out of 151 cases diagnosed with avian poxvirus over a 20-year period at a zoological institution, 22 were characterized as having systemic involvement based on histopathology and molecular findings. Gross lesions in systemic cases included soft white nodules scattered throughout the liver, spleen, and kidneys. Two histopathologic patterns emerged: (1) widespread histiocytic inflammation in visceral organs with intrahistiocytic viral inclusions and (2) severe, localized dry or wet pox lesions with poxvirus-like inclusions within dermal and subepithelial histiocytes. In situ hybridization targeting the core P4b protein gene confirmed the presence of poxvirus DNA within histiocytes in both patterns. Polymerase chain reaction was performed targeting the reticuloendothelial virus long terminal repeat (REV LTR) flanking region and the core P4b protein gene. Sequences of the REV LTR flanking region from all systemic pox cases were identical to a previously described condorpox virus isolated from an Andean condor with systemic pox. Sequences of the core P4b protein gene from all systemic pox cases grouped into cluster 2 of the B1 subclade of canarypox viruses. Systemic involvement of avian poxvirus likely occurs as a result of infection with certain strain variations in combination with various possible host and environmental factors.
Avian poxviruses are DNA viruses in the family Poxviridae, subfamily Chordopoxvirinae, genus Avipoxvirus and have been reported to infect 232 avian species from 23 orders. 3 A total of 10 species of avian poxvirus are currently recognized by the International Committee on Taxonomy of Viruses: fowlpox virus, canarypox virus, juncopox virus, mynahpox virus, pigeonpox virus, psittacinepox virus, quailpox virus, sparrowpox virus, starlingpox virus, and turkeypox virus (ictvonline.org). Numerous studies have investigated the phylogenetic relationships of avian poxvirus species and strains, predominantly using sequences of the gene encoding the core P4b protein, which divides avian poxviruses into 3 clades: clade A (fowlpox viruses), clade B (canarypox viruses), and clade C (psittacinepox viruses).8,11,15 Fowlpox virus serves as the type species for this genus and is of economic importance in commercial poultry productions, causing decreased egg production, stunted growth, and variable mortality rates in infected flocks. 21
Avian poxvirus infections are classically divided into 2 clinical manifestations: cutaneous or “dry” pox and diphtheritic or “wet” pox. Dry pox is characterized by proliferative, crusty, or scabby nodules on nonfeathered skin. In wet pox, affected birds develop erosions and plaques or pseudomembranes of thick, white to yellow, caseous exudate on the mucosa of the oral cavity, esophagus, upper respiratory tract, and sinuses. 21 Infected birds can have concurrent dry and wet pox. Regardless of the clinical form, lesions of avian poxvirus share similar characteristic histologic findings, including epithelial hyperplasia and ballooning of epithelial cells with large, intracytoplasmic, eosinophilic viral inclusions with a central clearing, referred to as Bollinger bodies. 21
An atypical, systemic form of avian poxvirus infection has also been sporadically described, most commonly in canaries causing respiratory distress and high mortality rates. 21 Lesions in this form include fibrinous pneumonia, tracheitis, and air sacculitis with edema, proliferation of respiratory epithelial cells containing Bollinger body-like inclusions and often concurrent dry or wet pox.2,4,12,17,18 These reports in canaries and other species have documented not only lower respiratory tract involvement of classic poxvirus infection but also poxvirus-like inclusions in splenic and thymic reticuloendothelial cells and mononuclear cells in the thymus, bursa of Fabricius, spleen, and bone marrow.2,4,7,18,22 While lower respiratory tract involvement could be a direct extension from oropharyngeal lesions, suspected infection of lymphoid and hematopoietic tissue would indicate systemic trafficking of virus. In these reports, the presence of poxvirus was confirmed using various diagnostic modalities including polymerase chain reaction (PCR),2,18 virus isolation,2,4,7,12,18 and electron microscopy.4,18,22
Visceral disease from systemic poxvirus infection has been reported in a captive Andean condor with atypical inflammatory nodules in the heart, lung, liver, kidney, small intestine, pancreas, and spleen. 13 Poxvirus-like inclusions were seen within histiocytes in this inflammatory population as well as in biliary epithelial cells, splenic reticuloendothelial cells, and thymic and bursal epithelial cells. Similar visceral inflammation has been reported in a chicken with dry pox and inflammatory nodules in the liver. 1
The aims of this study were to (1) confirm and characterize presumptive systemic poxvirus infections by localizing avian poxvirus genomes within systemic inflammatory lesions and (2) compare viral strains from systemic cases to those from birds with nonsystemic pox. Characterizing systemic poxvirus infections is important in zoological settings with mixed-species aviaries, where birds are in close contact with each other, indirectly exposed to native birds, and represent threatened and endangered species.
Materials and Methods
Source Population
All birds housed at San Diego Zoo Wildlife Alliance (SDZWA; comprised of the San Diego Zoo and the San Diego Zoo Safari Park) that died of natural causes or were euthanized for medical reasons between July 1999 and November 2019 were candidates for inclusion in this study. Birds were housed in a variety of exhibits with various environmental, husbandry, and species-grouping parameters. All native, wild birds that were submitted to the pathology service after being either found dead or debilitated and subsequently euthanized on SDZWA premises during this time period were also reviewed for inclusion in this study. When possible, a complete necropsy was performed for each zoo-housed and native bird. Complete sets of tissues were collected and placed in 10% neutral buffered formalin for histopathology for all zoo-housed birds. Complete sets of tissues were not consistently collected for histopathology from native birds. Variable amounts and types of fresh frozen tissues were saved at the time of necropsy from each bird. Postmortem findings were stored in the searchable database, Zoological Information Management Software (ZIMS; Species360, version 1.7, zims.Species360.org).
Case Identification
Cases of avian pox were identified by searching avian necropsy reports in ZIMS for the keyword “pox” and reviewing each case to confirm a diagnosis of poxvirus infection, including dry, wet, and systemic forms, during this 20-year period. Complete sets of slides were reviewed for cases with diagnoses in the necropsy report of widespread granulomatous or histiocytic inflammation in multiple tissues with eosinophilic, intrahistiocytic material suspicious for poxvirus (n = 26). Slides were also reviewed for a randomly selected subset of 26 birds with nonsystemic avian pox, defined by typical mucocutaneous lesions (eg, dry pox, wet pox, or both), as well as an additional 4 birds with nonsystemic pox that were not selected during randomization, but were of the same species as individuals represented in the systemically affected group. Formalin-fixed tissues were embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). The tissue distribution of inflammatory lesions associated with poxvirus-like inclusions was recorded for each case, as well as significant comorbidities diagnosed at the time of death.
DNA Isolation
DNA was extracted from frozen archived tissues from birds identified as systemic pox cases (n = 26) and birds with nonsystemic infections (n = 30) based on histopathology. A DNeasy Blood and Tissue Kit (Qiagen, Germany) was used following the manufacturer’s tissue extraction protocol to extract DNA from frozen archived tissues. Sample DNA was eluted at 100 µl and stored at −80°C prior to use.
Polymerase Chain Reaction
PCR was performed on extracted DNA from systemic pox cases (n = 26), the randomly selected nonsystemic cases (n = 26), and the additional species-matched nonsystemic cases (n = 4). A total of 2 primer sets were used to amplify 2 different areas of the avian poxvirus genome: a region flanking the reticuloendothelial virus (REV) long terminal repeat (LTR) integrated sequence and the gene encoding the core P4b protein. The REV LTR flanking region is often used to distinguish between wild-type and vaccine strains of avian poxvirus, as wild-type strains contain the full REV-integrated provirus while vaccine strains contain variable length LTRs within the genome. 21 The gene encoding the core P4b protein is conserved but varies between strains and has proven to be useful in phylogenetic analyses of avian poxvirus strains.8,11,15
The flanking REV LTR region was amplified using previously published primers to generate approximately 600 bp amplicons: F: 5’-YGGAGAYAGRSAAATATCAGA-3’ and R: 5’-ATARKAATCACMMGWATATACC-3.’ 13 Cycling conditions used for the REV LTR region assay consisted of: 95°C: 1 minute, (95°C: 45 seconds, 50°C: 1 minute, 72°C: 45 seconds) × 40 cycles, and 72°C: 6 minutes. 13 The core P4b protein gene was amplified using previously published primers to generate approximately 570 bp amplicons: F: 5’-CAGCAGGTGCTAAACAACA-3’ and R: 5’-CGGYARCTTRACRCCGAATA-3.’ 10 The reverse primer was altered with degenerate nucleotides. Cycling conditions for the core P4b protein gene assay consisted of: 95°C: 1 minute, (95°C: 15 seconds, 55°C: 15 seconds, 72°C: 10 seconds) × 40 cycles, and 72°C: 1 minute. 10 Both assays were performed using a Mastercycler Pro S thermocycling system (Eppendorf, Hauppauge, New York). Each 25 µl reaction for both assays contained the following reagents: 12.5 µl 2× MyTaq Red Mix (Bioline, Memphis, Tennessee), 400 nM forward and reverse primer each (IDT, San Diego, California), and 2 µl DNA. Each assay was run with a known positive control, a negative extraction control, and a negative reaction control (nuclease-free water). Amplicons were run on a 1.1% agarose gels stained with 2.5% ethidium bromide. Bands were isolated and subsequently extracted using the EMD Millipore Ultrafree-DA centrifugal filters (Millipore, Billerica, Massachusetts).
DNA Sequencing and Analysis
All reactions for both primer sets that yielded bands after gel electrophoresis were submitted for sequencing. Purified, gel-extracted DNA and the corresponding forward and reverse primers were submitted for Sanger sequencing (Eton Bioscience, San Diego, California) on a 3730xl DNA sequencer (Applied Biosystems, Bedford, Massachusetts). Raw sequencing files were uploaded, and both directions of the sequences were trimmed using the trim vectors function (Univec [high sensitivity]) using default parameters. Each sequence was pairwise aligned using the Geneious (Biomatters, Auckland, New Zealand) alignment program, and a consensus alignment was generated. Consensus sequences were then aligned in Geneious Prime 2020.2.2 using MUSCLE 3.8.425. 5 Phylogenetic analysis on P4B sequences was performed in MEGA-X14,19,20 using the model selection function for optimal nucleotide substitution model and maximum likelihood trees generated with 500 bootstrap replicates.
In Situ Hybridization
In situ hybridization (ISH) probes were designed based on sequences of the core P4b protein gene derived from a subset of the systemic pox cases included in this study. ISH was performed on unstained slides from all systemic pox cases. Unstained slides from dry pox lesions served as a positive control.
RNA ISH was performed using the RNAscope 2.5 HD Red Chromogenic Reagent Kit according to the manufacturer’s instructions (Advanced Cell Diagnostics, Newark, California). Target probes were designed using custom software as described previously, 23 based on a consensus alignment of amplicons generated with the above-described PCR. Five µm thick sections of formalin fixed, paraffin embedded (FFPE) tissue were mounted on AutoFrost charged adhesion slides (Cancer Diagnostics, Inc., Durham, North Carolina), baked at 60°C in a dry oven, and deparaffinized. The sections were treated with an endogenous peroxidase blocker for 10 minutes at room temperature before boiling in a target retrieval solution for 15 minutes. Protease Plus was then applied for 30 minutes at 40°C. Target probes were hybridized for 2 hours at 40°C, followed by a series of signal amplification and washing steps. Hybridization signals were detected by chromogenic reactions using Fast Red. Slides were counterstained in 50% hematoxylin for 2 minutes and decolorized with 0.2% ammonium hydroxide. After rinsing in deionized water, slides were dried in a 60°C oven, dipped in xylene, and cover-slipped using xylene-based SHUR/Mount medium (Triangle Biomedical Sciences, Durham, North Carolina). RNA staining signal was identified as red punctate dots. Negative control background staining was evaluated using a probe specific to a bacterial DapB gene.
Statistical Analysis
The proportion of zoo-housed birds with systemic pox was compared to the proportion of native birds with systemic pox using a Fisher’s exact test calculated using GraphPad QuickCalcs (http://www.graphpad.com/quickcalcs/ConfInterval1.cfm). P < .05 were considered statistically significant.
Results
Population Summaries
Between July 1999 and November 2019, 151 cases of avian poxvirus were diagnosed histologically or grossly at SDZWA in 42 different species of birds, including birds housed at SDZWA (n = 98) as well as native, wild species (n = 53). The majority of cases (98/151, 64.9%) had only the dry form of disease, followed by concurrent dry and wet pox (23/151, 15.2%) and wet pox only (8/151, 5.3%). Systemic avian poxvirus infections were diagnosed in the remaining 22 individuals (14.6%) based on a combination of histopathology, PCR, and ISH findings, spanning 14 species of zoo-housed and native birds (Supplemental Table S1).
In total, 15/53 (28%) of zoo-housed birds with poxvirus infections had the systemic form of disease, compared to only 7/98 (7%) of native birds with poxvirus infections. These proportions are statistically different (Fisher exact P = 0.001), suggesting that systemic disease is more likely to occur in zoo-housed species than native birds.
Birds diagnosed with systemic poxvirus infections exhibited a variety of comorbidities diagnosed at the time of death including aspergillosis (8/22), bacterial infections or sepsis (6/22), hemoparasitism (Plasmodium sp and Isospora sp., 4/22), intestinal coccidiosis (4/22), proventricular nematodiasis (1/22), renal nematodiasis (1/22), mycobacteriosis (1/22), fungal myositis (1/22), candidiasis (1/22), and yolk sacculitis (1/22).
Gross and Histopathologic Findings
Gross lesions in birds with systemic pox varied depending on the tissues affected and the severity of the disease (Supplemental Table S1). Internal lesions often consisted of white to pale tan, soft to friable nodules scattered throughout the parenchyma of affected organs, most commonly the liver, spleen, and kidney (Fig. 1a). Other gross lesions included mottling or enlargement of the liver and spleen and serous to fibrinous coelomic effusions. No internal lesions were seen grossly in 9/22 (40.9%) cases. The majority of systemic cases also had gross or histologic evidence of concurrent classic pox lesions: dry pox (8/22, 36.4%), wet pox (2/22, 9.1%), or both dry and wet pox (11/22, 50%). Only 1 systemic case (Northern chestnut-breasted malkoha, Phaenicophaeus curvirostris) showed no gross or histologic evidence of either dry or wet pox. The classic dry and wet pox lesions present in cases of systemic pox were similar to lesions typical of these forms: proliferative, crusty to ulcerated, cutaneous nodules (dry form), and caseonecrotic plaques or exudate in the oropharynx, sinuses, trachea, and esophagus (wet form).
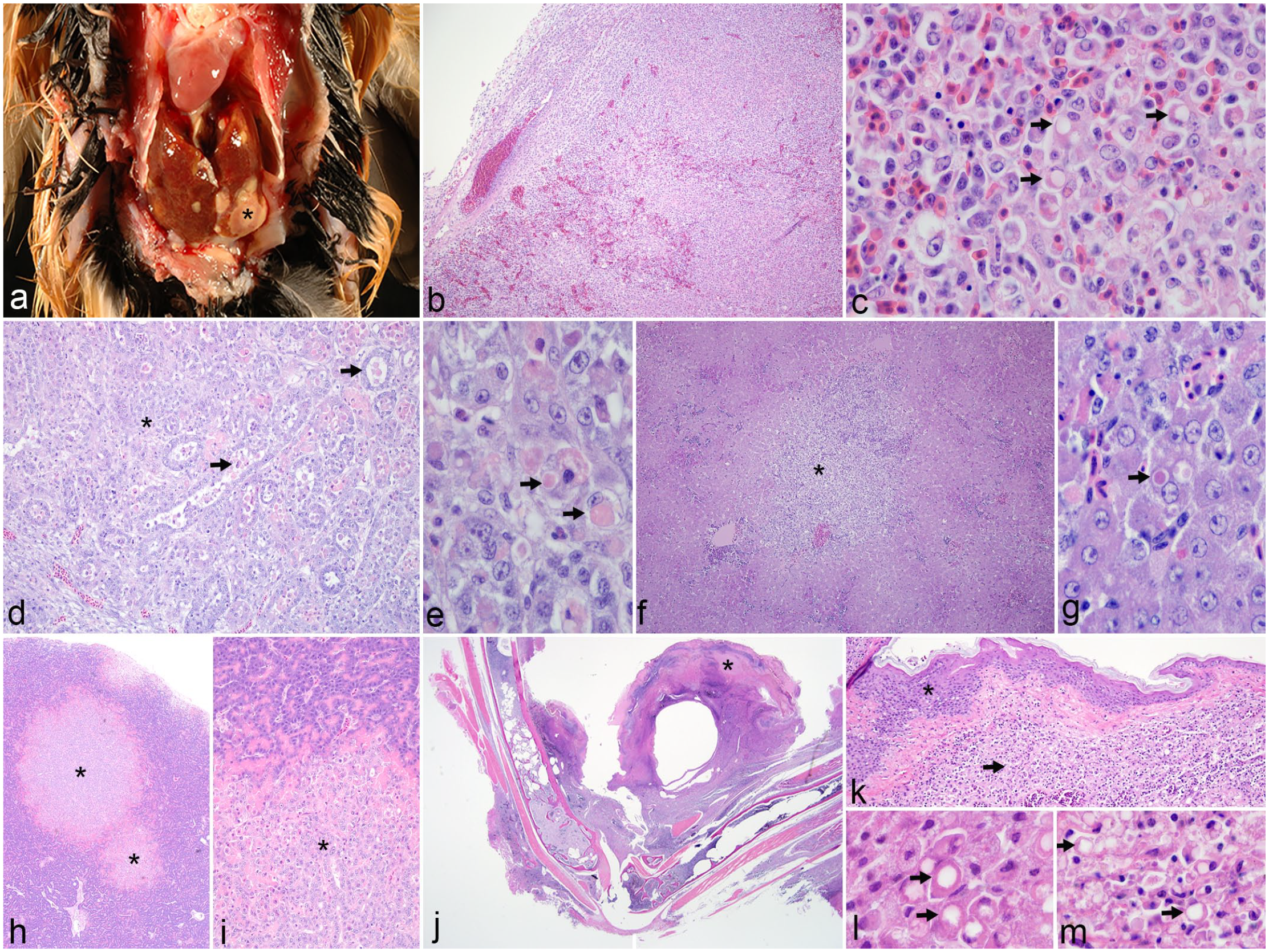
Gross and histopathologic features of systemic avian poxvirus infection. (a) Liver, Guam kingfisher. Multiple soft, pale tan to white, variably sized nodules (asterisk) are scattered throughout the liver. (b–m) Hematoxylin and eosin. (b) Spleen, northern chestnut-breasted malkoha. The normal red and white pulp architecture of the spleen is effaced by a dense population of histiocytes. (c) Spleen, northern chestnut-breasted malkoha. Many of the histiocytes within the spleen contain intracytoplasmic, brightly eosinophilic inclusions that vary from amorphous material to round inclusions with a central clear zone similar to Bollinger bodies (arrows). (d) Kidney, fawn-breasted bowerbird. The kidney is infiltrated with numerous histiocytes that separate and disrupt tubules (asterisk). Tubular epithelial cells are often degenerating to necrotic and sloughed into the lumen (arrows). (e) Kidney, fawn-breasted bowerbird. Degenerating renal tubular epithelial cells rarely contain brightly eosinophilic, intracytoplasmic inclusions (arrows). (f) Liver, lesser goldfinch. The liver has multiple nodular aggregates of histiocytes (asterisk) that efface hepatic cords and bile ducts. (g) Liver, lesser goldfinch. Hepatocytes rarely contain intracytoplasmic, brightly eosinophilic inclusions (arrow). (h) Pancreas, fawn-breasted bowerbird. The pancreas has multiple well demarcated nodules (asterisks) scattered throughout the parenchyma. (i) Pancreas, fawn-breasted bowerbird. Higher magnification of nodules in (h) shows the junction of normal exocrine pancreas (top) with proliferative epithelial cells (asterisk). (j) Leg, lesser goldfinch. A typical dry pox lesion (asterisk) is present on the skin of the leg, with inflammation extending throughout the subcutaneous soft tissues and into the medullary cavity of the bone. (k) Skin, lesser goldfinch. Proliferative epidermis (asterisk) overlays markedly inflamed dermis and subcutaneous tissue consisting of dense sheets of histiocytes (arrow). (l) Skin, lesser goldfinch. High magnification of epidermal cells from (k) containing typical cytoplasmic poxviral inclusion bodies (Bollinger bodies, arrows). (m) Skin, lesser goldfinch. High magnification of dermal and subcutaneous inflammation from (k) showing similar intracytoplasmic inclusion bodies in histiocytes (arrows).
The dry and wet pox lesions present in these 21 cases of systemic infection exhibited characteristic epithelial hyperplasia with intraepithelial, intracytoplasmic viral inclusions (Bollinger bodies). Two histopathologic patterns emerged in the 22 cases of systemic pox that were distinct from classic dry and wet pox lesions: (1) widespread histiocytic inflammation affecting multiple organs with possible intrahistiocytic inclusions, designated as the “visceral inflammatory” pattern and (2) severe dry or wet pox lesions with histiocytic inflammation and possible intrahistiocytic inclusions extending into the underlying soft tissues and bone, designated as the “deep, localized” pattern. The visceral inflammatory pattern of systemic pox occurred in 16/22 individuals (72.7%). Tissue distribution of the observed histiocytic inflammation was variable, but most commonly affected tissues were spleen, liver, and coelomic cavity (Supplemental Table S1). Histiocytes within this inflammatory population variably contained eosinophilic, intracytoplasmic material suggestive of poxvirus inclusions (Fig. 1b–g). These intrahistiocytic inclusions varied in appearance, with some forming brightly eosinophilic, round to ovoid structures with a central clear vacuole strongly resembling Bollinger bodies, while others were less discrete and consisted of granular to globular, pale to brightly eosinophilic, intracytoplasmic material. Possible viral inclusions were also rarely seen within parenchymal epithelial cells including renal tubular epithelium and hepatocytes (Fig. 1e, g). Areas of necrosis were variably present within inflamed tissues. In 2 of these cases, atypical proliferative epithelial nodules were identified in parenchymal organs: in the pancreas (acinar and ductal epithelium) of a fawn-breasted bowerbird (Chlamydera cerviniventris, Fig. 1h, i) and in the thymus (thymic epithelium) of an Andean condor (Vultur gryphus). The proliferative epithelial nodules in the thymus of the Andean condor were associated with histiocytic inflammation with intracytoplasmic poxvirus-like inclusions; however, the hyperplastic pancreatic nodules in the fawn-breasted bowerbird were not overtly associated with inflammation or suspect viral inclusions.
Six of the 22 systemic pox cases (27.3%) had the deep, localized pattern in which the inflammation and necrosis associated with a severe dry or wet pox lesion extended into the dermis or subepithelium and underlying connective tissues, skeletal muscle, periosteum, and medullary cavity of bones (Fig. 1j–l). Histiocytes within the inflammatory population in the tissues surrounding the dry or wet pox lesion frequently contained intracytoplasmic material reminiscent of poxvirus inclusions (Fig. 1m). Similar to the intrahistiocytic inclusions seen in the visceral inflammatory pattern, the material within histiocytes in the dermis, submucosa, and subcutis in these deep, localized systemic pox infections varied from Bollinger body-type inclusions to granular or globular, eosinophilic intracytoplasmic material. This deep localized pattern is in contrast to typical dry and wet pox lesions, in which milder inflammation is present in the dermis or submucosa, and viral inclusions are restricted to the epidermis or mucosal epithelium. Five of the 16 systemic pox cases with the visceral inflammatory pattern also had poxvirus-like inclusions within histiocytes in the dermis or subepithelium associated with dry or wet pox lesions.
In Situ Hybridization
ISH was performed on unstained slides of FFPE tissues that were positive for avian poxvirus by PCR. Probes targeting the core P4b protein gene showed intense labeling of viral inclusions in epidermal lesions of typical dry pox cases. The same labeling characteristics were demonstrated in histiocytes infiltrating internal organs in the visceral inflammatory pattern of systemic pox cases (Fig. 2a–e), as well as within dermal, subepithelial, and subcutaneous histiocytes in the deep, localized systemic pox infections (Fig. 2f). ISH also highlighted the presence of poxvirus DNA in epithelial populations in parenchymal organs, including renal tubular epithelium (Fig. 2c), hepatocytes, biliary epithelium, oviductal mucosa (Fig. 2g), thyroid follicular epithelium, and thymic epithelium. Visceral organs were negative for poxvirus DNA in the deep, localized systemic pox infections.
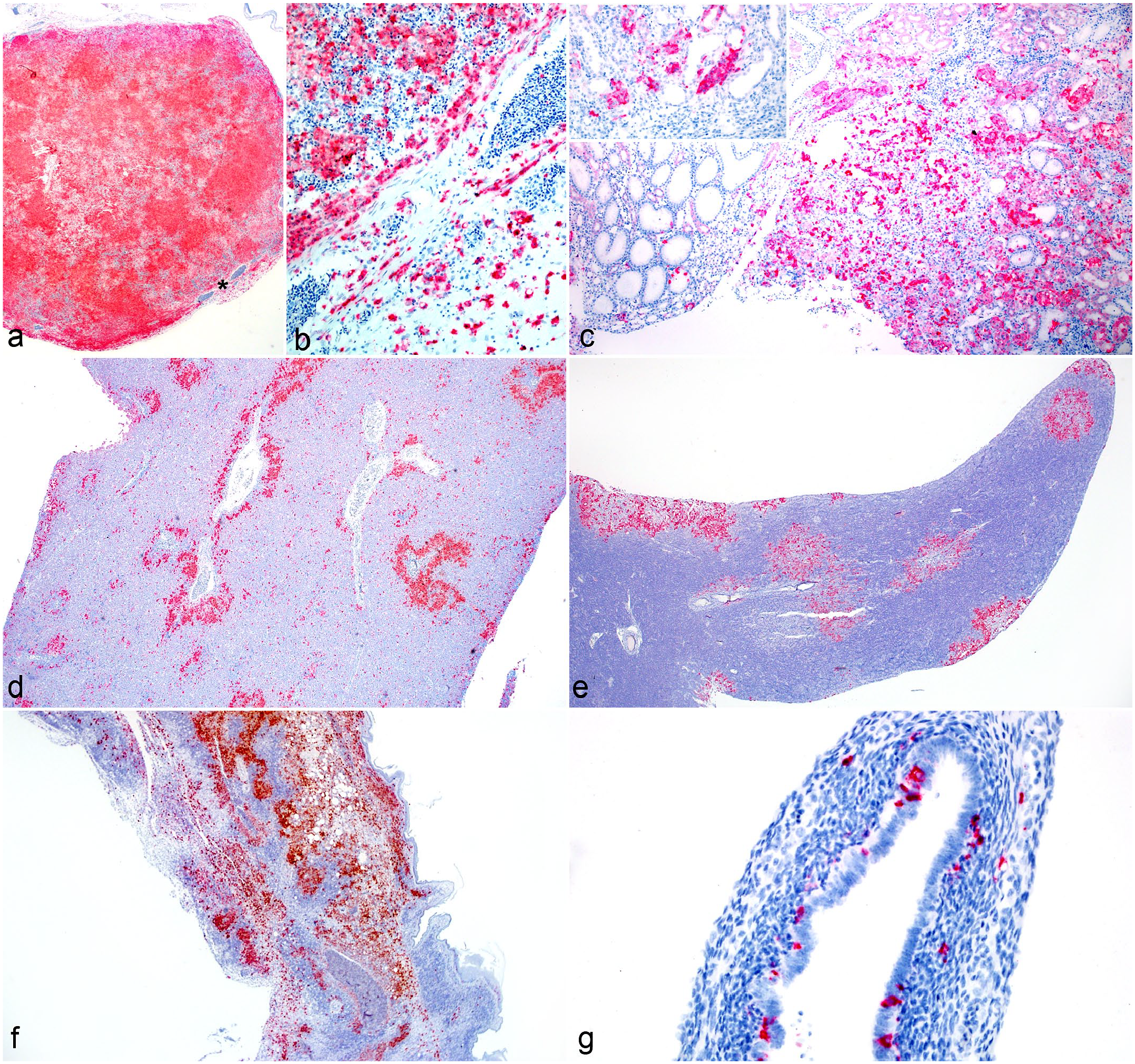
Localization of poxvirus DNA within lesions of systemic avian poxvirus infection. In situ hybridization (ISH) for core P4b gene. (a) Spleen, northern chestnut-breasted malkoha. Widespread positive ISH signal was detected throughout the spleen. Higher magnification of this region (asterisk) is seen in (b). (b) Spleen, northern chestnut-breasted malkoha. Positive ISH signal was present within infiltrating histiocytes in the spleen. (c) Kidney, fawn-breasted bowerbird. Positive ISH signal was detected within areas of inflammation. Inset: Positive signal was present in both infiltrating histiocytes as well as tubular epithelial cells. (d) Liver, Guam kingfisher. Positive ISH signal was detected in a pattern similar to infiltrating histiocytes throughout the liver. (e) Pancreas, fawn-breasted bowerbird. Positive ISH signal corresponded solely with the proliferative nodules in the pancreas. (f) Skin, lesser goldfinch. Positive ISH signal was present within infiltrating histiocytes throughout the dermis and subcutaneous tissues. (g) Oviduct, northern chestnut-breasted malkoha. Positive ISH signal was detected within the mucosa and smooth muscle layer, possibly representing mucosal epithelial cells or mucosal dendritic cells.
Polymerase Chain Reaction
Sequences of the REV LTR flanking region were amplified from 18/22 (81.8%) systemic pox cases and 17/30 (56.7%) nonsystemic cases. All 18 systemic pox sequences were >99% identical to a previously characterized condorpox virus, 13 as well as 10/17 (58.8%) nonsystemic cases (Table 1). The remaining nonsystemic pox cases yielded magpiepox virus or canarypox virus.
REV LTR sequencing results.

Abbreviation: REV LTR, reticuloendothelial virus long terminal repeat.
A total of 51 sequences of the gene encoding the core P4b protein were amplified from 19/22 (86.4%) systemic pox cases and 23/30 (76.7%) nonsystemic cases (GenBank Accession Nos. ON568587–ON568637). Sequences from all 19 systemic pox cases showed >99% identity to avian poxvirus strains within cluster 2 of the B1 subclade (canarypox virus) according to the previously proposed phylogeny (Table 2). 8 Similarly, sequences from 10/23 (43.5%) of nonsystemic cases also showed >99% identity to strains in cluster 2 of the B1 subclade. Sequences from the remaining nonsystemic cases fell into other B subclades, clade A (fowlpox virus), or the clade could not be determined (Table 1). Maximum likelihood phylogenetic analysis of the core P4B gene segment demonstrated high genetic relatedness of sequences between systemic cases and nonsystemic cases from varying host taxa. Other clades demonstrated host specificity of sequences with only corvids (American crows and ravens) in 1 group, and Anna’s hummingbirds and blackbirds occupying their own genetically distinct lineages (Supplemental Fig. S1).
Core P4b protein gene sequencing results.
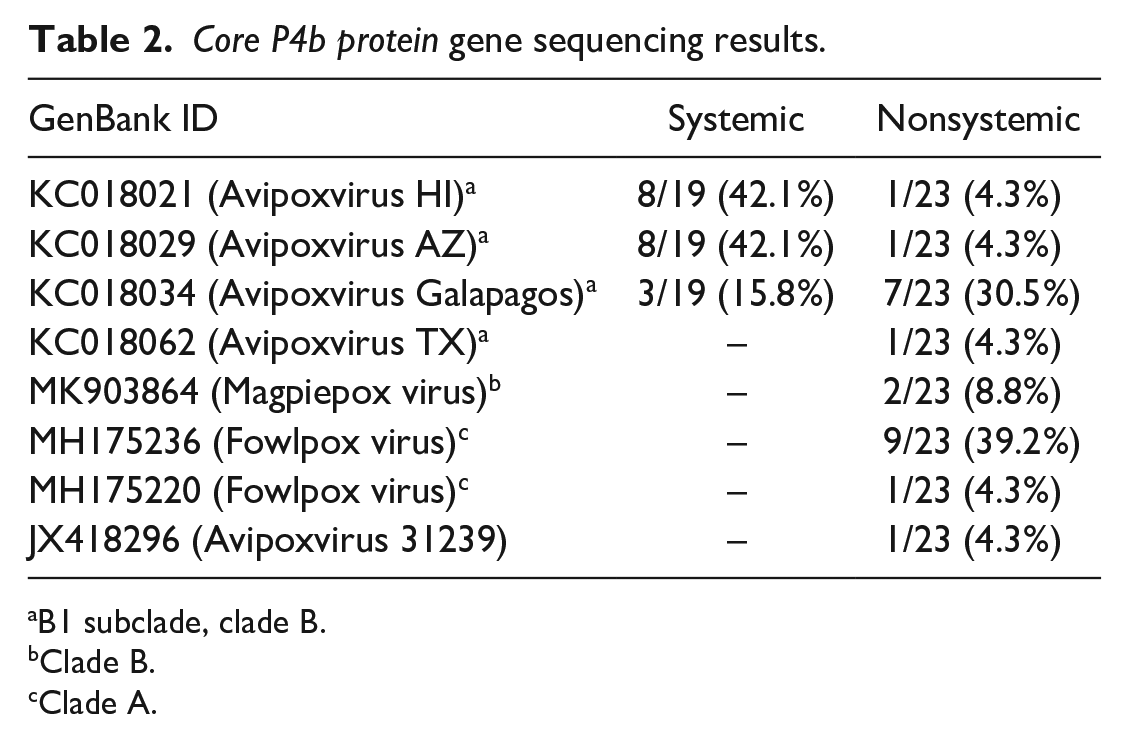
B1 subclade, clade B.
Clade B.
Clade A.
Discussion
Systemic avian poxvirus infection is a sporadically reported disease, most commonly described in canaries. This study describes the histopathologic lesions and molecular characteristics of 22 cases of systemic avian poxvirus in zoo-housed and native, wild bird species over a 20-year period from a zoological institution. During this time, the systemic form was the third most common manifestation (22/151, 14.6%) of avian poxvirus diagnosed in necropsied birds, with dry pox only as the most common manifestation (98/151, 64.9%), concurrent dry and wet pox as the second most common manifestation (23/151, 15.2%), and wet pox only as the least common manifestation (8/151, 5.3%). Two distinct patterns of systemic infection were observed in this study: a visceral inflammatory pattern and a deep, localized pattern of inflammation.
Five cases with the visceral inflammatory pattern also had poxvirus inclusions within histiocytes subjacent to severe dry or wet pox lesions in a manner similar to the deep, localized pattern. Based on these cases, in which the 2 histologic patterns overlap, it seems plausible that systemic avian pox infection begins as a severe form of localized dry or wet pox, histiocytes become infected at this localized site, and subsequently spread through the dermis or subepithelium and eventually throughout the body to parenchymal organs via leukocyte trafficking. A similar pathogenesis has been proposed for sheeppox and goatpox, 2 other systemic poxviruses of veterinary importance. 9 All but one of the 22 systemic pox cases in this study had concurrent dry or wet pox lesions. It is possible that the individual lacking dry or wet pox lesions may have had subtle disease or previously resolved lesions that could have been missed grossly or on histologic sectioning.
ISH targeting the gene encoding the core P4b protein confirmed the presence of avian poxvirus DNA within histiocytes in both histologic patterns of the disease. Interestingly, other epithelial populations within parenchymal organs were also positive for avian poxvirus DNA by ISH. This suggests that avian poxvirus may maintain some degree of epitheliotropism in the systemic form of disease, as is seen in the dry and wet forms.
Another interesting finding was the presence of nodular epithelial hyperplasia within parenchymal organs in 2 systemic pox cases: in the pancreas of a fawn-breasted bowerbird and in the thymus of an Andean condor. In the Andean condor, nodules of hyperplastic thymic epithelium were surrounded by histiocytic inflammation and presumptive poxvirus inclusions were present both within these infiltrating histiocytes as well as the hyperplastic thymic epithelial cells, suggesting that the virus was inducing the hyperplastic response similar to what is seen in the epidermis. However, ISH did not yield positive signaling in the affected thymic tissue, which may have been the result of prolonged fixation in formalin. Avian poxvirus has been implicated as the driver of tumor-like, nodular epithelial proliferation in the respiratory tract of canaries 6 and doves 16 with similar histologic findings as those in the Andean condor. Unlike the Andean condor, the epithelial nodules in the pancreas of the fawn-breasted bowerbird were not overtly associated with inflammation or poxvirus-like inclusions. However, ISH revealed the presence of abundant poxvirus DNA localized solely to the nodules of proliferative epithelium in the pancreas (Fig. 2e), again supporting the plausibility of the virus driving this epithelial proliferation.
Based on the medical record review and retrospective histopathology, 26 birds were originally considered as potential systemic poxvirus infections. Included among these were 4 individuals from a group of 9 superb parrots (Polytelis swainsonii). These 4 parrots had evidence of wet or dry pox, with extension of the inflammation, epithelial hyperplasia, and poxvirus inclusions into the lungs and air sacs, similar to what is described in canaries. These birds also had nonspecific, histiocytic inflammation of varying severity within parenchymal organs. The other 5 parrots in this group had evidence of wet or dry pox, but lacked widespread histiocytic inflammation. DNA was extracted from frozen tissues (liver, spleen, or kidney) for 8/9 parrots, but amplification for all 8 parrots was negative for poxvirus. Based on the lack of molecular evidence of poxvirus infection within visceral organs, the 4 superb parrots initially considered as potential systemic poxvirus infections were ultimately deemed nonsystemic poxvirus infections with inflammation observed within the viscera likely representing an unrelated disease process or comorbidity.
The grouping of core P4b protein gene sequences from all 22 systemic pox cases into cluster 2 of the B1 subclade may indicate that this group of closely related strains has a greater propensity to cause systemic poxvirus infection compared to other strains. The B1 subclade has previously been implicated in an outbreak of systemic avian poxvirus in canaries. 2 Many previous reports of systemic avian poxvirus did not pursue sequencing, and the molecular characteristics of strains in these cases are unknown.1,4,7,12,17,18,22 Strains in the B1 subclade are considered the “strict” canarypox viruses, and they have been documented in numerous passerine species from around the world and across a wide span of time. 8 The B1 subclade is comprised of 3 clusters, and cluster 2 is hypothesized to have originated in house finches that subsequently diverged and spread globally. 8 Further phylogenetic analyses of cases of systemic poxvirus infections are needed to elucidate the role of B1 subclade strains in the development of the systemic form of disease. While all 22 systemic pox cases clustered into the B1 subclade, 43.5% of nonsystemic cases also fell into this group, suggesting that strain alone does not determine systemic infection. Interestingly, all but one of the nonsystemic cases in this cluster (snowy-crowned robin-chat) belonged to Southern California native wildlife, while all but 3 species representing systemic cases (lesser goldfinch, Western turkey vulture, and California towhee) were non-native bird species. Along with published sequence data, these results suggest some degree of host specificity to these core P4b sequences, with a significantly larger proportion of zoo-housed birds developing systemic pox when infected with B1 subclade strains compared to native birds that more commonly develop nonsystemic disease when infected with these strains. More research is needed to adjust for confounders and determine the true host specificity of avian poxviruses and to determine whether being infected with nonhost-adapted strains is truly a risk factor for the development of systemic disease. This is particularly relevant to institutions harboring endangered birds in conservation breeding programs that may be exposed to poxvirus strains harbored by local wild birds.
Over half of the systemic avian poxvirus cases in this study belonged to the taxonomic order Passeriformes (12/22, 54.5%), followed by Coraciiformes (5/22, 22.7%), Cathartiformes (4/22, 18.2%), and Cuculiformes (1/22, 4.5%) (Supplemental Table 1). Since strains belonging to cluster 2 of the B1 subclade of avian poxviruses are believed to have originated from house finches, these strains may be host adapted to passerines resulting in their overrepresentation in systemic poxvirus infections. Infection in other orders may be due to the wide host range of avian poxviruses in general or to the exposure of exotic species in a zoological setting to strains that they are not typically exposed to in their natural habitats. Although it is difficult to determine based on the small number of individuals represented in this study, it is possible that certain species or orders of birds have a greater likelihood of developing the systemic form of disease if infected with avian poxvirus, particularly if infected with strains within cluster 2 of the B1 subclade.
Many of the individuals with systemic pox in this study had significant coinfections (15/22, 68.2%). Similar concurrent bacterial, fungal, viral, and parasitic infections have been diagnosed in other reports of systemic pox in canaries and other species.2,7,12,17,18,22 Stress due to new or unnatural environments might also be considered as a comorbid factor for exotic, captive birds in zoological settings. Although speculative, coinfections and physiologic or environmental stress could possibly predispose individuals to developing the systemic form of avian poxvirus when infected with particular strains. Additional species-matched studies are needed to more definitively show the relationship of these host factors to systemic avian poxvirus infections.
The majority (20/22, 90.9%) of systemic pox cases in this study occurred between July and November (Supplemental Table S1), as did 69.7% (90/129) of dry or wet pox cases. This seasonality may reflect the fluctuations in mosquito and other biting insect populations, which act as mechanical vectors in the transmission of avian poxvirus. 21 The time of year and abundance of vectors may also serve as predisposing factors for avian poxvirus infections, including the systemic form.
The systemic form of avian poxvirus infection may develop less commonly than other forms, but this disease could have conservation impacts on populations of critically endangered species kept in zoological institutions. The majority of reported cases of systemic avian pox have described severe respiratory disease in canaries and other species. This study adds many new species and tissues affected by systemic avian poxvirus infection, as well as highlights the utility of ISH in linking inflammatory lesions to avian poxvirus infection. Systemic avian poxvirus infection should be considered as a differential diagnosis for widespread histiocytic disease in birds in which other infectious and noninfectious causes cannot be found. It may also prompt more rigorous vector control strategies and aviary design to limit contact with native wildlife at institutions housing more critically endangered species or those in captive breeding programs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231190639 – Supplemental material for Systemic avian poxvirus infections associated with the B1 subclade of canarypox virus
Supplemental material, sj-pdf-1-vet-10.1177_03009858231190639 for Systemic avian poxvirus infections associated with the B1 subclade of canarypox virus by Devinn M. Sinnott, Jennifer Burchell, Carmel Witte, Rachel Burns and Steven Kubiski in Veterinary Pathology
Footnotes
Acknowledgements
The authors specially thank necropsy technicians at San Diego Zoo Wildlife Alliance (SDZWA), particularly April Gorow, Rachael Keeler, and Megan Takahashi, for collecting and archiving tissues; SDZWA pathologists past and present for initial review and diagnosis of cases; and Yvonne Cates for her histotechnology skills.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
