Abstract
Introduction
Several studies of WNV-infected corvid carcasses demonstrated high detection rates of WNV in vascular feather pulp from the base of growing immature feathers.
5,35
Growing feathers are present during periods of molt, which typically occur at least once annually following the breeding season,
8
corresponding with arbovirus transmission activity in northern latitudes. However, in some cases, avian carcasses lack vascular feathers, especially where WNV transmission may occur outside of the avian breeding season. Nonvascular breast feathers from WNV-infected eastern screech-owls (
Nonvascular feathers have been used for detection of a variety of viruses, such as Avian influenza virus,
Sensitivity of
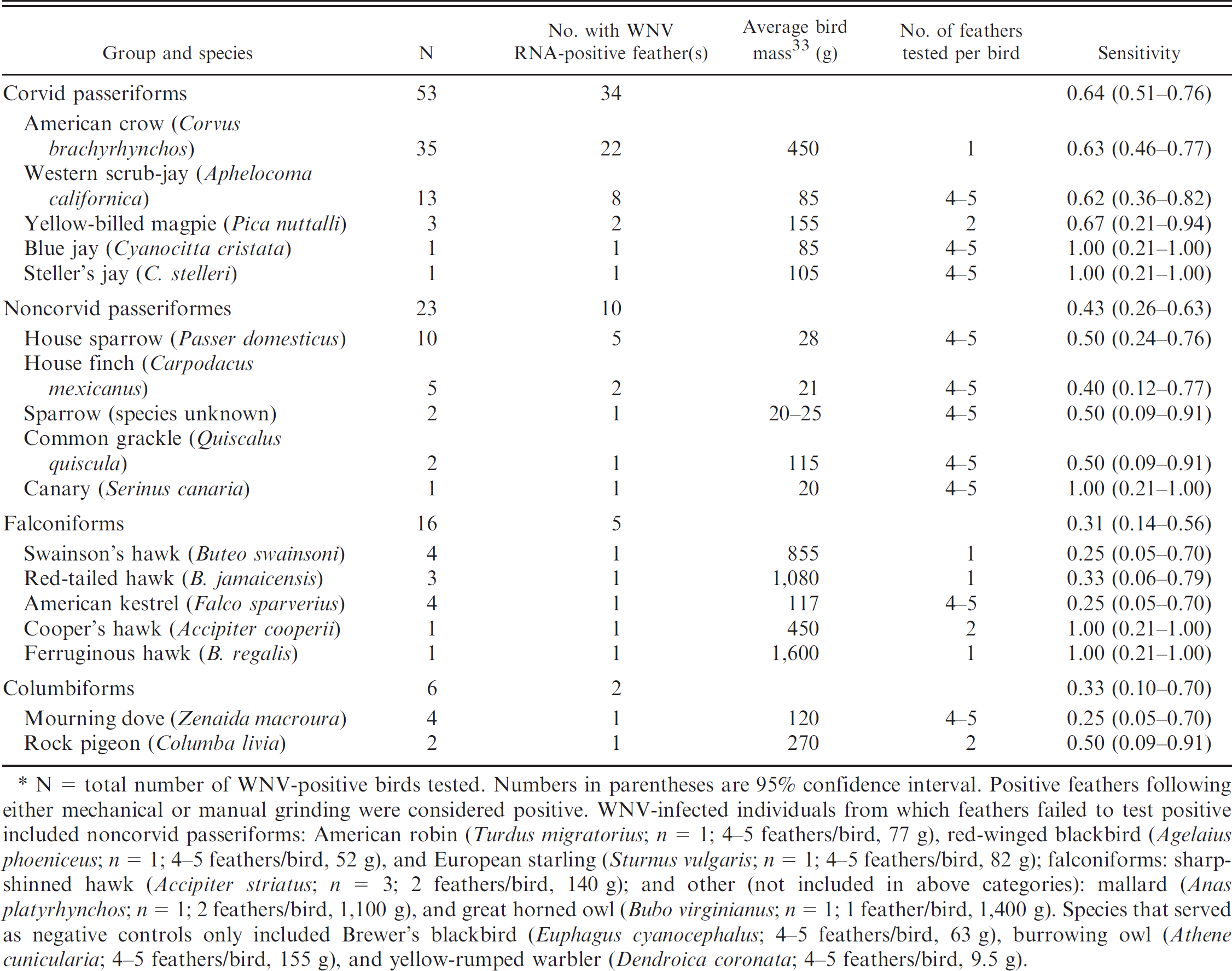
N = total number of WNV-positive birds tested. Numbers in parentheses are 95% confidence interval. Positive feathers following either mechanical or manual grinding were considered positive. WNV-infected individuals from which feathers failed to test positive included noncorvid passeriforms: American robin (
Materials and methods
Avian carcass collection and sampling
Avian carcasses from Colorado and California were sampled by oropharyngeal swab, removal of nonvascular (mature) breast feathers, and harvest of either heart (Colorado) or kidney (California) tissue. Carcasses collected for study were deemed dead <48 hr and were in good condition (e.g., intact, prior to visible decay and appearance of insect larvae), then frozen at −20°C for 1–3 days (Colorado) or shipped overnight on cold packs (California) prior to testing.
Feathers were removed from the carcass and stored in sterile cryovials (Colorado) or Whirl-Pak
a
bags (California). After feather collection, oropharyngeal swab samples were collected from Colorado birds and California crows by swabbing the oropharyngeal cavity with a cotton-tipped applicator and placing the swab into 1 ml of 15–20% fetal bovine serum prepared in BA-1 diluent (Hank's M-199 salts, 1% bovine serum albumin, 350 mg/l sodium bicarbonate, 100 units/ml penicillin, 100 mg/l streptomycin, 1 mg/l amphotericin B in 0.05 M Tris, pH 7.6). Necropsies were performed on all carcasses from Colorado and noncrow carcasses from California, at which time tissue was removed aseptically with a sterile surgical blade and either placed into cryovials with 1 ml BA-1 diluent and a ball-bearing (BB) pellet (heart) or with 1 ml lysis buffer (kidney). Heart samples (0.5 cm
3
) were ground in a mixer mill
b
at 25 cycles/sec for 5 min, clarified by centrifugation (12,000–
Feather collection and storage
Clusters of nonvascular feathers were carefully removed from the breast (i.e., ventral feather tract). The number of feathers collected per bird varied depending on the size of the bird as well as relative feather size/weight (Table 1). Mass of feathers tested per bird ranged from 0.001 g to 0.009 g.
For Colorado birds, half of the feathers collected from each carcass were held at room temperature (approximately 25°C), while the other half were held at freezer temperature (–20°C) for 1 year prior to testing. For California birds, immediately following collection, feathers were frozen at −80°C for several months until thawed for homogenization and testing.
Feather grinding techniques
Matched sets of feathers from bird carcasses collected in California (
Inhibition of WNV detection in feathers
To evaluate the possibility of test inhibition as a result of components of the feather vane that may be liberated during grinding, we compared WNV RNA detection rates in matched pairs of feathers or feather components from each of 28 naturally infected American crow carcasses. First, matched pairs of breast feather calami (i.e., tips, cut from the rest of the feather at the proximal edge of the feather vane) were ground by either mechanical or manual grinding. Mechanically ground calami was then compared with mechanically ground whole feather, again in matched pairs, as well as manually ground calami with manually ground whole feather. In all cases, homogenates were clarified by centrifugation and tested by TaqMan RT-PCR.
Reproducibility of WNV detection in feathers
To assess the reproducibility of WNV RNA detection in feathers and determine the sensitivity of single versus multiple feathers tested from a single carcass, sets of 10 whole breast feathers were mechanically ground from each of 33 carcasses of naturally infected birds. Each feather was individually ground and tested by TaqMan RT-PCR.
Distribution of WNV in feathers
To determine the distribution of WNV RNA and infectious virions within the feather, the calamus and vane (including the rachis) were tested separately for each of 6 individual feathers removed immediately following euthanasia of experimentally inoculated crows (3 fish crows [
Plaque assay
For Colorado samples, virus isolation was performed on heart tissue and oropharyngeal swabs by Vero cell plaque assay. 29 In addition, plaque assays were performed on homogenates of whole feathers and feather components. All viral plaques were harvested and identified by a commercial WNV antigen detection assay. d
Reverse transcription polymerase chain reaction
TaqMan RT-PCR methods for detection of WNV RNA were performed on oropharyngeal swabs, hearts, kidneys, and feathers. RT-PCR methods followed those of Lanciotti et al. 21 except for the use of viral RNA mini kits e for RNA extraction and a real-time detection system f for complementary DNA (cDNA) amplification. A critical threshold value of <38.0 cycles obtained for 2 distinct sets of WNV-specific primers was considered positive for target sequence amplification. Positive RNA extraction and RT-PCR controls contained viral titers of approximately 106.5 and 104.5 plaque forming units (PFU) of WNV strain NY99–4132; tap water was used as a negative control for RNA extraction and a negative control for RT-PCR contained all reaction components except for RNA template.
Statistical analyses and determination of sensitivity and test agreement
Sensitivity and k values of WNV RNA detection testing of whole nonvascular feathers were derived by comparison of feather TaqMan RT-PCR with oropharyngeal swab, heart, or kidney TaqMan RT-PCR (referred to hereafter as “standard diagnostic methods”). Sensitivity was defined as the proportion of carcasses that were positive by standard diagnostic methods and also yielded WNV-positive feathers. Test agreement was measured using the kappa statistic, k, defined as the proportion of positive and negative carcasses determined by standard diagnostic methods that matched the feather test result. The feather test sensitivity and k for each species group were compared with the corresponding values of all other species combined using a two-tailed Fisher's exact test and Bonferroni-adjusted α = 0.0125 (for 4 comparisons representing each species group). Two-tailed Fisher's exact test was also used to compare WNV detection rates between grinding methods (i.e., manual vs. mechanical) and feather components (i.e., whole feather vs. calami). Mean viral load measures were compared with a T-test of unequal sample size and unequal variance, following log transformation of viral titers.
Results
Sensitivity of WNV detection in nonvascular feathers
Feathers were collected from 109 carcasses, with 100 WNV-positive and 9 negative. Feathers from 50 individuals of 17 species tested positive. Among feathers stored frozen (–l20°C or −80°C), sensitivity of detection by RT-PCR was greatest for corvid passeriforms (64%), followed by noncorvid passeriforms (43%), columbiforms (33%), and falconiforms (31%; Table 1). Among these 4 groups, only corvid feathers were significantly more sensitive for WNV detection than other species groups (Fisher's exact test,
Storage temperature
The effect of storage temperature on WNV detectability was tested by examining sets of matched feathers held at ambient room temperature and −20°C from a subset of the WNV-positive carcasses (
Feather grinding techniques
Manual grinding was compared with mechanical grinding for matched sets of whole feathers derived from individual carcasses using TaqMan RT-PCR and virus isolation (
Inhibition of WNV detection in feathers
Because manual grinding provided better homogenization of the feathers, it was suspected that this grinding method liberated inhibitors of WNV detection and evaluated this hypothesis with 3 comparisons assessed from feathers harvested from 28 WNV-positive American crow carcasses: 1) WNV RNA detection probability in matched pairs of breast feather calami (1 pair per carcass) with one feather ground mechanically and the other manually; 2) WNV RNA detection probability in calamus versus whole feather, ground mechanically, and 3) WNV RNA detection probability in calamus versus whole feather, ground
Number of

N = number of birds. Detection probability represents the probability that WNV will be detected in a single breast feather from a WNV-positive carcass. Numbers in parentheses are 95% confidence interval.
manually. For the first comparison, manual grinding produced a higher detection rate (32%) among calami than mechanical grinding (18%), although this difference was not significant (Fisher's Exact Test,
Reproducibility of WNV detection in feathers
Evaluating the reproducibility of WNV RNA detection among sets of 10 nonvascular breast feathers indicated substantial variation among carcasses, ranging from 0 to 10 feathers testing positive per WNV-positive carcass. At the species level, however, no significant differences were discerned, with the mean number of feathers testing positive (in species with minimum
Distribution of WNV in feathers
WNV RNA was detected more frequently in the vane than the calamus in experimentally inoculated crows, whereas infectious virus was detected with greater frequency and in higher concentrations in the calamus (Table 3).
Results of
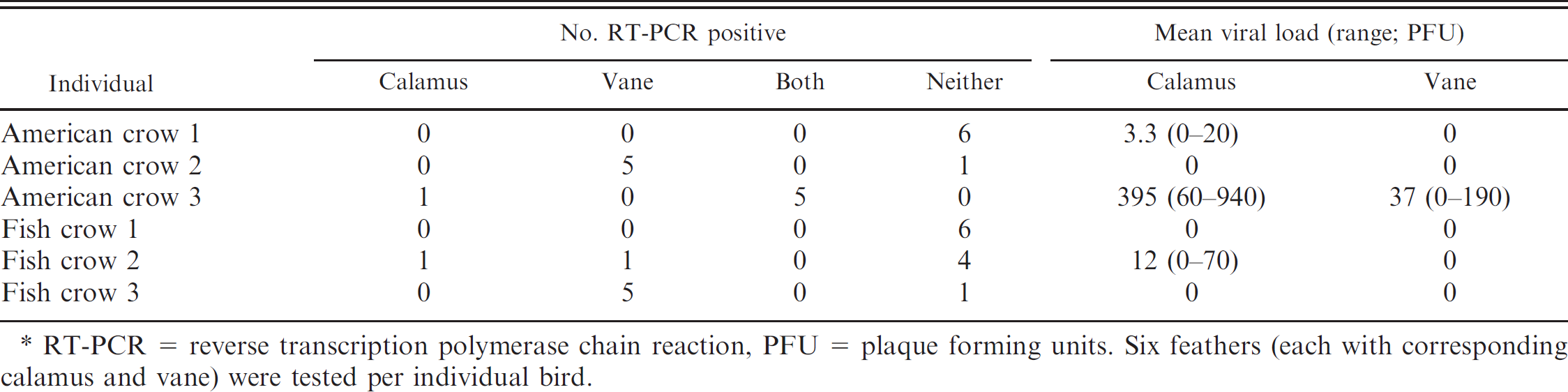
RT-PCR = reverse transcription polymerase chain reaction, PFU = plaque forming units. Six feathers (each with corresponding calamus and vane) were tested per individual bird.
Discussion
Feather testing has proven useful for diagnosis of numerous avian pathogens, and feathers may be a source of transmission. Feather follicle epithelium is a source of infectious
Feathers have been examined previously for WNV diagnosis, but within relatively limited parameters. One study compared WNV testing of vascular pulp from growing feather calami with cloacal swabs, kidney, and spleen tissues from carcasses of American crows and blue jays (
In the present study, sensitivity of WNV detection in nonvascular feathers was lower than those reported in previous studies of corvid carcasses. 5,9 Various factors may explain this reduced sensitivity, including avian species differences, feather type, feather development stage (e.g., whether pulp or blood is present in the shaft), quantity of feathers tested, feather storage conditions, and components included for testing (e.g., feather calami vs. whole feathers). The developmental stage of the feather is presumed to be the key factor. However, species selection had a clear effect in the current study. Feathers from corvids were significantly more sensitive for WNV detection than feathers from noncorvid species. The effect of feather type or anatomical location of the feather was not tested in the current study; rather, feathers were exclusively from the ventral tract. However, viral tropism for certain feathers or feather clusters could exist. In addition, reproducibility of WNV detection in ventral tract feathers was approximately 30%. Therefore, sensitivity can be increased by pooling multiple feathers from a single carcass. No effect of storage condition on RNA detection was observed, while detection varied between calamus and vane depending on grinding method. This variation may be, in part, caused by detection inhibitors derived from certain feather components.
Potential inhibition of RT-PCR by certain feather components merits further study, as mitigation of such could improve the utility of feathers in diagnosis of WNV infections. For example, melanin from the shafts of brown chicken feathers was shown to inhibit PCR detection of
The current study elucidates the potential use of feathers for WNV RNA detection in avian mortality surveillance programs. Although previous studies determined that vascular, growing feathers are the most sensitive tissue for WNV detection in corvids, it was demonstrated in the present study that nonvascular feathers are also useful, although with reduced sensitivity in corvids and relatively lower sensitivity in other species groups such as noncorvid passeriform birds, columbiforms, and falconiforms. Testing of nonvascular feathers would be most useful for surveillance in remote areas where maintaining the cold chain is unavailable (e.g., lack of dry ice or liquid nitrogen). To maximize the sensitivity of feather testing for WNV in American crows, the authors recommend testing multiple whole feathers per bird, as 6 feathers should yield approximately 90% sensitivity given a 32% detection probability per feather by RT-PCR. Because feather mass varies with body size and feather type or anatomical location, the optimal number of feathers for WNV detection may vary by species and feather tract. Potential inhibition from feather vane components should be mitigated as discussed above.
In summary, WNV particles, and more importantly RNA, persist in nonvascular feathers from a broad spectrum of bird species. Although nonvascular feathers may not be the most efficacious biological specimen for detecting WNV in avian carcasses, they offer numerous advantages to surveillance programs, including ease of collection, and simpler storage and shipping conditions. Additional potential advantages include longer period of detectability compared with tissues, application to live birds (morbidity surveillance and diagnosis in individual patients), and lower biosafety risk. Although infectious viral load in nonvascular feathers may be up to 105.7 PFU per feather, the virions are presumably sequestered within a keratinized sheath such that handlers are protected from the infectious fluids associated with most tissues, such as feather pulp and body cavities used for swabbing or removal of tissue for storage. However, to the authors’ knowledge, contamination of external feather surfaces has not been examined. Taken together, these advantages may warrant utilization of feathers for avian morbidity and mortality surveillance of WNV, HPAIV, and other important zoonotic pathogens.
Acknowledgements
The authors are grateful to the following for providing samples: Gail Kratz, Judy Scherpelz, Lisa Winta, Carin Avila, and Rebecca Bates of the Rocky Mountain Raptor Program, Bob Nightwalker and Jessica Plunkett of Wild-Kind (Larimer County Humane Society), and Jackie Parker of California Animal Health and Food Safety Laboratory. Richard Bowen (Colorado State University) and Susan Beckett (CDC) provided logistical support, and Maureen Dannen and Ying Fang (University of California, Davis) provided technical support. Funding for RNA detection platforms used for WNV RNA detection was partially provided by the Pacific Southwest Regional Center for Excellence (PSWRCE) U54 AI-65359.
Footnotes
a.
Nasco International Inc., Fort Atkinson, WI.
b.
Retsch GmbH, Haan, Germany.
c.
Tenbroek tissue grinder, Wheaton Science Products, Millville, NJ.
d.
VecTestr WNV/SLEV Antigen Assay, Medical Analysis Systems Inc., Camarillo, CA.
e.
Qiagen Inc., Valencia, CA.
f.
iCycler iQTM Real-time Detection System, Bio-Rad Laboratories, Hercules, CA.
