Abstract
Colorectal adenocarcinoma is an aggressive malignant tumor in cats that frequently metastasizes to the lymph nodes and/or distant organs. However, research on feline colorectal adenocarcinoma is limited, and experimental models have not been established. A novel cell line, FeLeco-G7, was established from the lymph node of a 12-year-old spayed female Maine Coon cat with metastatic colorectal adenocarcinoma. FeLeco-G7 cells were polygonal with abundant cytoplasm and adherent growth. The population-doubling time was approximately 28.3 hours, and the mean number of chromosomes was 37.6±0.1 per cell (ranging between 32 and 41). Consistent with the original tumor, FeLeco-G7 cells were immunopositive for cytokeratin (CK) 20 and CDX2, and immunonegative for CD10 and CK7. Nuclear accumulation of β-catenin was rarely observed. Mutation analysis suggested TP53 gene alterations. A subcutaneous injection of FeLeco-G7 cells into immunodeficient mice resulted in the formation of a mass at the injection site without the development of metastatic lesions. An orthotopic (intrarectal) transplantation of FeLeco-G7 cells caused cachexia and diffuse involvement of the rectal mucosa in one of the 3 mice and the formation of masses around the rectum in the other 2 mice. Metastases to the regional lymph nodes and lungs were detected in three of the 3 and one of the 3 mice, respectively. The histological findings and immunohistochemical features of these masses were similar to those of the original tumor. These results suggest that FeLeco-G7 cells and the orthotopically transplanted mouse model are valuable tools for further molecular and therapeutic research on feline colorectal adenocarcinoma.
Keywords
Adenocarcinoma is the second most common tumor in the feline colorectum. 23 Serosal infiltration and lymph node and/or distant metastatic lesions are frequently detected at the time of diagnosis.19,23 Although surgical removal is the mainstay of treatment for colorectal adenocarcinoma in cats, clinical outcomes are unfavorable, particularly in cats with distant metastases.2,19 A few clinical studies previously suggested the efficacy of chemotherapy drugs, such as doxorubicin and carboplatin, for extending survival times in cats with colorectal adenocarcinoma.2,19 However, there are currently no standard treatments for advanced colorectal adenocarcinoma in cats. Therefore, the development of novel therapeutic strategies for feline colorectal adenocarcinoma is critical.
Tumor cell lines and xenograft models are valuable tools for elucidating the molecular mechanisms underlying disease progression and developing novel anticancer therapies. Orthotopic xenograft models are widely used in human colorectal cancer research because they closely mimic malignant behaviors, such as lymph node and/or distant metastasis. 12 However, to the best of our knowledge, tumor cell lines of feline colorectal adenocarcinoma have not yet been established. The lack of an experimental model has been a barrier to feline colorectal cancer research.
In this study, we established a novel cell line obtained from the lymph node of a cat with metastatic colorectal adenocarcinoma and characterized its morphological and immunocytochemical features. Furthermore, a xenotransplant mouse model was established.
Materials and Methods
Animal Background
A tumor sample was obtained from a 12-year-old, spayed female Maine Coon with a history of hematochezia and dyspepsia. Colonoscopy revealed partially circumferential irregular elevation, ulceration, and bleeding of the colorectal mucosa (Supplemental Figure S1). The cat was diagnosed with adenocarcinoma through a rectal biopsy and histopathological examination. Subsequently, contrast-enhanced computed tomography revealed thickening of the intestinal wall at the end of the descending colon, enlarged regional lymph nodes, and small nodules ranging from 5 to 12 mm in the liver. Due to the difficulties associated with colon resection, colostomy and biopsy of the left colic lymph node and liver were performed. The cat died 26 days after surgery. Necropsy was not performed.
Histopathology and Immunohistochemistry
All tissues were fixed in 10% neutral-buffered formalin and were routinely embedded in paraffin. Consecutive sections (thickness of 4 µm) were stained with hematoxylin and eosin and were used for immunohistochemistry. For immunohistochemistry, endogenous peroxidase in sections was blocked by 3% hydrogen peroxide in methanol for 5 minutes. To unmask antigens, sections were heated in an autoclave (121°C) for 10 minutes in citrate buffer (pH 6.0, self-prepared) or incubated with 125 µg/mL proteinase K (Wako Pure Chemical, Osaka, Japan) solution at room temperature for 30 minutes. After washing with Tris-buffered saline, slides were treated with 8% skimmed milk in Tris-buffered saline. The primary antibodies used are listed in Table 1. After an incubation with each primary antibody at 4°C overnight, immunolabeled antigens were visualized using the Dako Envision+ System (Dako, Glostrup, Denmark) and reacted with 0.05% 3′3-diaminobenzidine plus 0.03% hydrogen peroxide in Tris-hydrochloric acid buffer. Sections were then counterstained with hematoxylin. All primary antibodies were examined with feline tissues to ascertain cross-reactivity, and the positive and negative controls consisted of sections from a feline tissue that were immunopositive or immunonegative for each primary antibody (Table 1).
Antibodies used for immunohistochemistry and immunofluorescence.
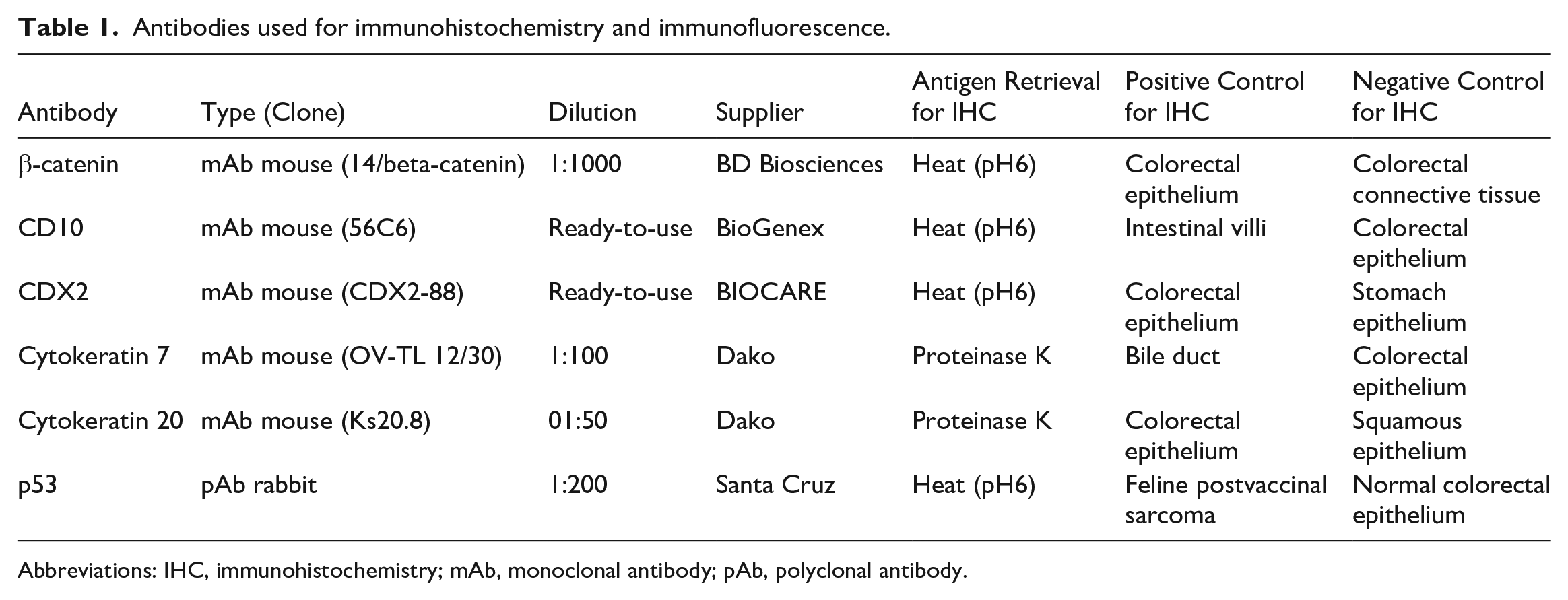
Abbreviations: IHC, immunohistochemistry; mAb, monoclonal antibody; pAb, polyclonal antibody.
Cell Cultivation
Fresh lymph node tissue with metastatic lesions was finely minced and washed 3 times with sterile phosphate-buffered saline (pH 7.4) containing 1% penicillin-streptomycin (Wako Pure Chemical, Osaka, Japan). Samples were then suspended in Dulbecco’s modified Eagle’s medium with high glucose and pyruvate (Gibco; Thermo Fisher Scientific, Tokyo, Japan) containing 0.25% crude trypsin and incubated at 4°C for 16 hours. After centrifugation, pellets were resuspended in new Dulbecco’s modified Eagle’s medium supplemented with 10% heat-inactivated fetal bovine serum (HyClone; Cytiva, Tokyo, Japan) and 1% penicillin-streptomycin, and then filtered through a cell strainer (70-µm nylon; Falcon, Tokyo, Japan). Suspended cells were seeded on 60-mm petri dishes (TrueLine; NIPPON Genetics Co., Ltd., Tokyo, Japan) and incubated at 37°C in an atmosphere of 5% CO2 and 95% air. When a confluent monolayer had grown in a petri dish, cells were trypsinized and seeded on another petri dish.
Two months after the initial cultivation (10th passage), tumor cells were cloned, using a serial dilution method. After cells had grown to cover a cell culture plate, cloned cells were removed and seeded on 60-mm petri dishes with Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum. Media exchange and cell passages were performed every 2 to 3 days. The newly established clone of feline colorectal adenocarcinoma cells was designated as FeLeco-G7. The detailed procedure to maintain FeLeco-G7 cells is described in Supplemental Table S1.
The polymerase chain reaction (PCR)-based bacterial, fungal, and mycoplasma contamination tests were performed on the 1st, 40th, and 200th passages. The PCR was conducted using the universal primer pairs for the bacterial 16S rRNA gene, 27F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-TACGGYTACCTTGTTACGACTT-3′); 21 for the fungal 18S rRNA gene, ITS3F (5′-GCATCGATGAAGAACGCAGC-3′) and ITS4R (5′-TCCTCCGCTTATTGATATGC-3′); 14 and for the intergenic spacer region between the 16S and 23S rRNA genes of mycoplasma, F1 (5′-ACACCATGGGAGCTGGTAAT-3′) and R1 (5′-CTTCWTCGACTTYCAGACCCAAGGCAT-3′), 22 as previously described. The PCR products were analyzed by agarose gel electrophoresis, using the ChemiDoc XRS+ System (Bio-Rad Laboratories, Hercules, CA).
Morphologic Characterization
FeLeco-G7 (86th passage) cells (5 × 104 cells/mL) were seeded into 2-well chamber slides (WATSON, Tokyo, Japan) and incubated at 37°C in an atmosphere of 5% CO2 and 95% air. Forty-eight hours after cultivation, the cells were stained with Giemsa. Phase contrast images of the cells from the 1st, 40th, and 200th passages were also obtained with an inverted phase contrast microscope (Carl Zeiss Meditec, Tokyo, Japan).
Growth Curve
FeLeco-G7 (69th passage) cells were seeded at 4 × 103 cells on 12-well multiplates and maintained in growth medium at 37°C for 14 days. The number of cells in each well was counted 1, 2, 4, 6, 8, 10, 12, and 14 days after the initiation of the cell culture. After the growth curve was obtained, the cell population-doubling time was calculated, using the following equation: doubling time (hours) = T × log (2)/log (Ne/Nb), where T is the duration (hours) of the exponential phase, Nb is the cell number at the beginning of the exponential phase, and Ne is the cell number at the end of the exponential phase. Three independent experiments were conducted.
Chromosome Analysis
FeLeco-G7 (66th passage) cells cultured to confluency were incubated with 0.02 µg/mL of colcemid (KaryoMAX; Gibco; Thermo Fisher Scientific, Tokyo, Japan) at 37°C for 2 hours in a humidified atmosphere of 5% CO2 and 95% air, and then treated with prewarmed hypotonic 0.075 mol/L potassium chloride. After centrifugation, cell pellets were resuspended in fixative solution (methanol/acetic acid, 1:1), dropped on a slide, and subsequently stained with Giemsa solution. Two hundred metaphases were counted under a microscope with an oil immersion objective lens (100×) to estimate the modal chromosome number.
Cell Viability Assay
The antiproliferative effects of carboplatin and SN-38, the active metabolite of irinotecan, was determined by using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) colorimetric assay. The solutions of carboplatin and SN-38 were prepared using distilled water or dimethyl sulfoxide (DMSO; Nacalai tesque, INC., Kyoto, Japan), respectively. FeLeco-G7 (50th passage) cells were seeded at 3 × 103 cells on 96-well multiplates and maintained in a growth medium at 37°C for 24 hours. After preincubation, the cells were cocultured with 12 concentrations of carboplatin (0–2700 µmol/L, Tokyo Chemical Industry Co., Ltd., Tokyo, Japan) or SN-38 (0–250 µmol/L, MedChemExpress, New Jersey) for 24 hours. For SN-38 treatment, matched DMSO concentration growth medium was used for vehicle controls. After treatment, cell proliferation was assessed using the Cell Counting Kit-8 method (Dojin-do, Kumamoto, Japan). Absorbance was measured using the iMark microplate reader (Bio-Rad Laboratories, Tokyo, Japan), and 50% inhibitory concentration (IC50) values were calculated with software R Version 3.5.0 (R Foundation for Statistical Computing, Vienna, Austria). The experiment was performed on more than 3 wells under the same conditions with different concentrations.
Immunofluorescence
FeLeco-G7 (13th, 86th, and 200th passage) cells were each seeded and grown on 8-well chamber slides (WATSON, Tokyo, Japan) 48 hours prior to immunolabeling. After fixation with 4% paraformaldehyde, the cell membrane was permeabilized by incubating cells with 0.25% Triton X-100 for the detection of intracellular antigens. After rinsing with phosphate-buffered saline, cells were treated with 1% bovine serum albumin in phosphate-buffered saline-Tween 20 to block nonspecific reactions. Primary antibodies (Table 1) were applied at 4°C overnight. Cells were further incubated with the Alexa 488-conjugated donkey antimouse or antirabbit IgG antibody (1:400; Invitrogen; Thermo Fisher Scientific, Tokyo, Japan) at room temperature for 1 hour. Cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; VECTASHIELD; Vector Laboratories, California). Negative controls were performed by omitting the primary antibodies. Slides were examined under a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec, Tokyo, Japan).
Western Blotting
For Western blotting, FeLeco-G7 (1st, 40th, and 200th passages) cell pellets were suspended in lysis buffer containing 20 mmol/L Tris (pH 8.0), 100 mmol/L NaCl, 1 mmol/L EDTA, 0.5% NP-40 (Wako Pure Chemical, Osaka, Japan), and protease inhibitor (cOmplete ULTRA; Roche Diagnostics GmbH, Mannheim, Germany). The cell suspension was frozen in liquid nitrogen, thawed at 37°C, frozen again, and placed on ice until completely thawed. After centrifugation at 23,000 rpm at 4°C for 10 minutes, supernatants were collected.
Thirty micrograms of protein samples were diluted (4:1) in Laemmli Sample Buffer (Bio-Rad Laboratories, Hercules, CA), which included 2-mercaptoethanol, and boiled for 10 minutes. The solution was electrophoresed on a 5% to 20% gradient polyacrylamide gel (ATTO, Tokyo, Japan) at 200 V and 300 mA for 50 minutes. Protein bands were transferred to a polyvinylidene fluoride membrane (Immobilon-P transfer membrane; Merck Millipore, Burlington, MA) at 25 V and 500 mA for 25 minutes. Nonspecific binding was blocked by a treatment with 1% skim milk at room temperature for 60 minutes. Membranes were probed with the following antibodies at 4°C overnight: mouse anti-β-catenin (clone 14/beta-catenin, 1:1000; BD Biosciences, Franklin Lakes, NJ), mouse anti-CDX2 (clone CDX2-88; Ready-to-use, Biocare Medical, Pacheco, CA), mouse anti-CK20 (clone Ks20.8, 1:300; Dako, Glostrup, Denmark), and horseradish peroxidase (HRP)-conjugated mouse anti-β-actin (clone 8H10D10, 1:5000; Cell Signaling Technology, Beverly, MA). After washing the membranes with Tris-buffered saline containing Tween 20, an HRP-conjugated sheep antimouse IgG antibody (1:5000; Cytiva, Tokyo, Japan) was applied. Blots were developed using ECL Select Western Blotting Detection Reagent (Cytiva, Tokyo, Japan). Immunoreactive bands were detected using the ChemiDoc XRS+ System.
Wound Healing Assay
FeLeco-G7 (1st, 40th, and 200th passage) cells were seeded at 5 × 104 cells on 24-well multiplates and grown until they reached >90% confluence. Cell cultures were scratched with a 1000 µL sterile pipette tip and the detached cells were washed away with phosphate buffered saline (PBS) twice. After scratching, cells were maintained in growth medium at 37°C. Phase contrast images were acquired with an inverted phase contrast microscope at 0, 24, and 48 hours after scratch, and the wound area was quantified, using the plugin Wound_healing_size_tool 20 of the software ImageJ Version 1.54d (National Institutes of Health, Bethesda, MD). 18 The percentage of wound closure was calculated using the following equation: wound closure % = ((A0 − At)/A0) × 100, where A0 is the initial wound area and At is the wound area after n hours of the initial scratch. Comparisons between passage numbers were performed with Student t test, using software R Version 3.5.0. The P values <.05 were considered to be significant. Three independent experiments were conducted.
Mutation Analysis of the Kirsten Rat Sarcoma Virus (KRAS) and TP53 Gene
Genomic DNA was extracted from FeLeco-G7 (1st, 40th, 81st, and 200th passage) cells, using the DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA). Purified DNA was used in subsequent PCR: from the 81st passage for Kirsten Rat Sarcoma Virus (KRAS) amplification and from the 1st, 40th, and 200th passage for TP53 amplification. Exon 1 of the feline KRAS gene was amplified as previously described. 23 Amplification of TP53 gene was conducted with specific primer pairs for feline TP53 that amplify a 533-bp fragment, which incorporates exons 5 and 6, 5′-GTAAGTGGTGCTGGTGCATGTG-3′ (forward) and 5′-TGGACGGCCGCTGACAAA-3′ (reverse), and a 945-bp fragment, which incorporates exons 7 and 8, 5′-CTTATGCAACCTCCCCACGG-3′ (forward) and 5′-CCTAATTGCAGGCAAAGCAGT-3′ (reverse). The PCR for the TP53 gene was performed as follows: for primers for exons 5 and 6, 40 cycles at 95°C for 30 seconds and 62°C for 30 seconds; and for primers for exons 7 and 8, 35 cycles at 95°C for 30 seconds and 59°C for 30 seconds. Unaffected feline tissue was also used to confirm amplification of the feline TP53 gene. The PCR products were analyzed by agarose gel electrophoresis, using the ChemiDoc XRS+ System, and purified, using the QIAquick PCR and Gel Cleanup Kit (Qiagen, Valencia, CA). Purified amplicons were submitted for a sequence analysis in the forward and reverse directions (Eurofins Genomics, Tokyo, Japan). The sequences were identified from RefSeq database, using National Center for Biotechnology Information–Basic Local Alignment Search Tool (NCBI-BLAST), 17 and a chromatogram was visually inspected for any abnormalities.
Subcutaneous and Orthotopic Transplantation
Six, 6-week-old female CB17/IcrJcl-Prkdcscid (severe combined immunodeficiency disease [SCID]) mice (CLEA Japan, Tokyo, Japan) were randomly divided into 2 groups for subcutaneous and orthotopic (intrarectal [IR]) transplantation. Experiments involving animal utilization were approved by the Animal Care and Use Committee of the Graduate School of Agricultural and Life Sciences at The University of Tokyo (approval numbers P17-017 and P20-083).
For subcutaneous transplantation, a 200 µl cell suspension (2 × 106 cells/mL, 50th passage) in PBS was subcutaneously injected into the right lower back of the mice. Tumor formation at the injection site was assessed. All mice were euthanized with isoflurane when the tumor diameter exceeded 20 mm or if the covering skin ulcerated.
For IR transplantation, a 10 µl cell suspension (3 × 105 cells/mL, 50th passage) in PBS was injected into the distal rectal submucosa of the mice as previously described. 8 All mice were euthanized with isoflurane 2 months after the injection or if mice exhibited signs of obstruction, cachexia, or any other signs of clinical decompensation.
Tumor masses and organs were collected at necropsy and fixed in 10% neutral-buffered formalin. Immunohistochemistry was performed as described above.
Results
Histopathology and Immunohistochemistry of Original Tumor
Columnar to cuboidal neoplastic cells arranged in irregular tubular structures were observed by histological examination of rectal biopsy samples (Fig. 1a). Tumor cells had abundant amphophilic cytoplasm and oval to round nuclei with prominent nucleoli (Fig. 1a). Anisokaryosis and nuclear pseudostratification were moderate, and mitotic figures were frequently detected, >50/10 high power fields (1.96 mm2), 400×. Stromal desmoplasia and neutrophilic infiltration were also observed. Tumor metastasis was found in the left colic lymph node and liver (Fig. 1b, c).
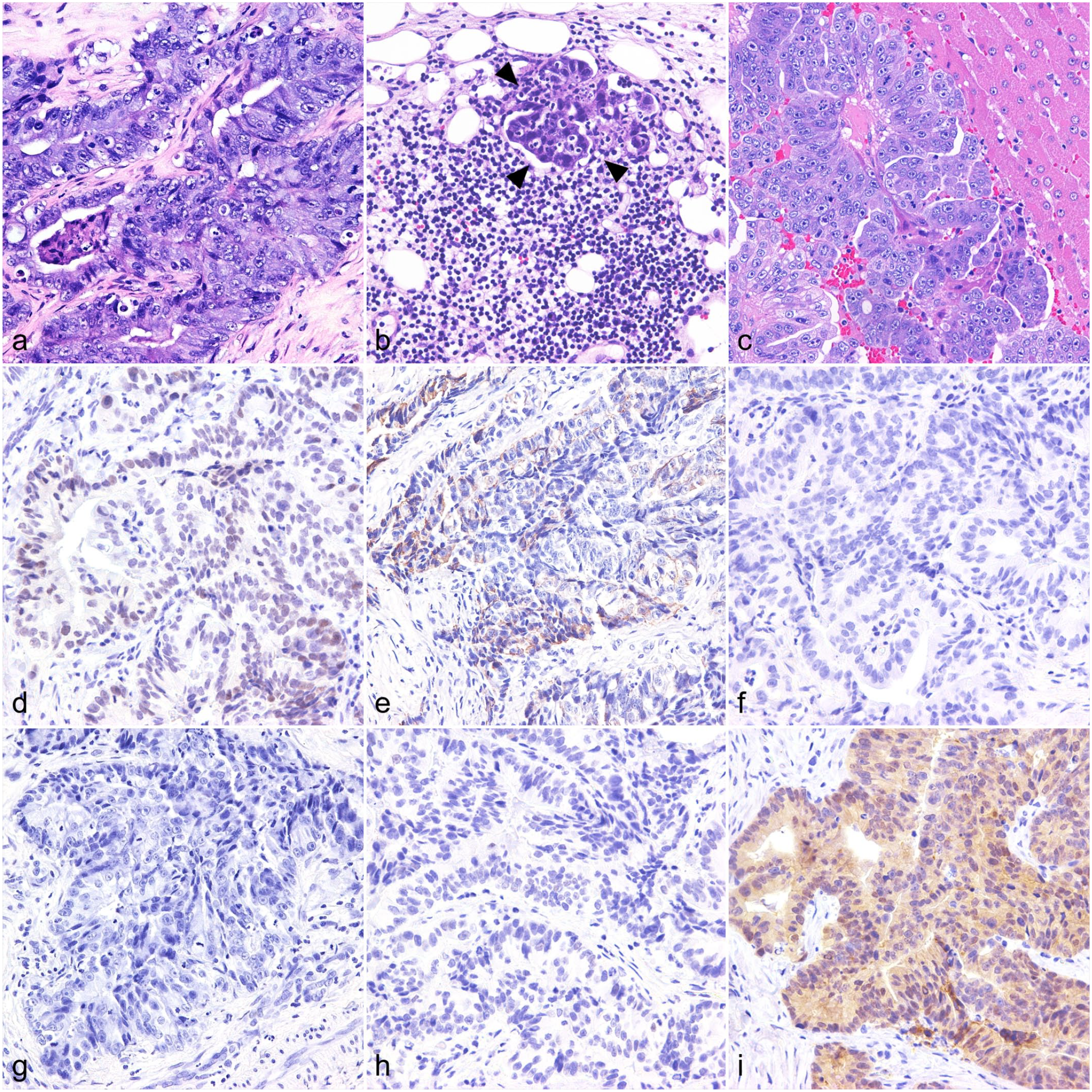
Colorectal adenocarcinoma, cat. Histological and immunohistochemical findings of primary tumor. (a) Rectum. Neoplastic cells are arranged in tubular structures with stromal desmoplasia. Hematoxylin and eosin (HE). (b) Left colic lymph node. Small metastatic focus in lymph node tissue (arrowheads). HE. (c) Liver. A metastatic lesion comprising columnar to cuboidal cells. HE. (d–i) Rectum. Immunohistochemistry. Neoplastic cells are immunolabeled for (d) CDX2 and (e) cytokeratin (CK) 20, and negative for (f) CD10, (g) CK7, and (h) p53. (i) Nuclear immunoreactivity for β-catenin is rarely observed.
Immunohistochemically, approximately 50% of tumor cells were immunopositive for CDX2 and CK20, and immunonegative for CD10, CK7, and p53 (Fig. 1d, h). Diffuse membranous and cytoplasmic immunolabeling for beta-catenin was also detected, whereas a few cells displayed nuclear accumulation (Fig. 1i).
Characterization of the Established Feline Colorectal Adenocarcinoma Cell Line (FeLeco-G7)
FeLeco-G7 cells formed an adherent monolayer of polygonal cells with abundant cytoplasm (Fig. 2a; Supplemental Figure S2). Their nuclei were round with 1 or 2 prominent nucleoli. Atypical mitoses were frequent, and anisokaryosis was mild to moderate. Bizarre, binucleated, giant neoplastic cells were occasionally present (Fig. 2a). These morphological features did not change from the 1st to the 200th passages (Supplemental Figure S2). The PCR-based bacterial, fungal, and mycoplasma contamination testing revealed no amplification product at each passage. The population-doubling time of FeLeco-G7 was approximately 17.2 hours (Supplemental Figure S3). Karyotyping revealed that the number of chromosomes of FeLeco-G7 ranged between 32 and 41 (mean 37.6, standard deviation 0.1, and mode 38). On FeLeco-G7 cells, the IC50 values of carboplatin and SN-38 were 237.1 µmol/L and 1.37 µmol/L, respectively (Fig. 2b, c). In the wound healing assay, the percentage of wound closure by the cells at 200th passage was significantly higher, compared with that at the first passage at 24 hours postscratch but not at 48 hours (Supplemental Figure S4). There were no significant differences between the percentage of wound closure at the 1st and 40th passages or the 40th and 200th passages at each time point.
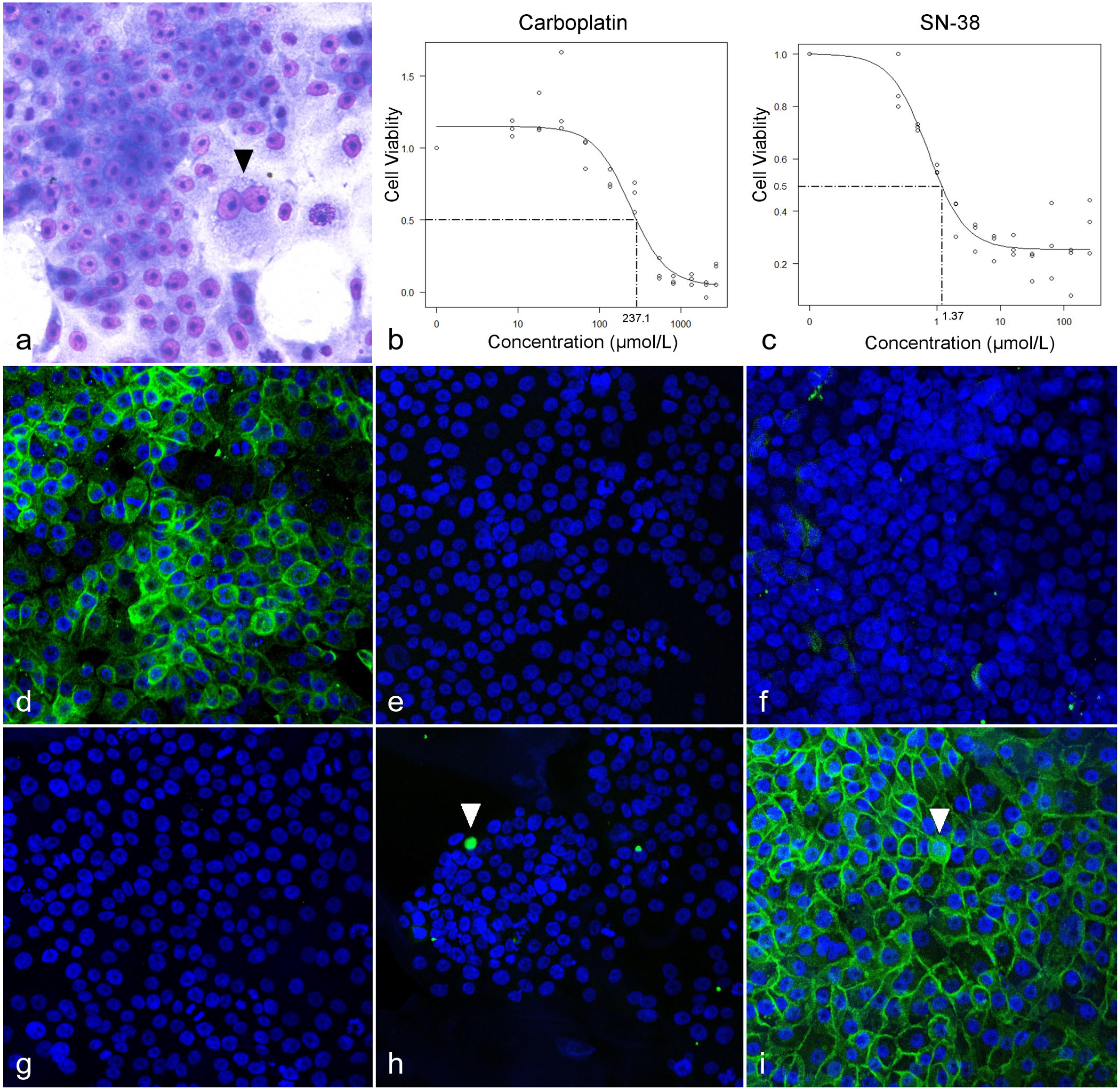
A cell line derived from feline colorectal adenocarcinoma (FeLeco-G7). (a) Cultured cells (86th passage) are polygonal with abundant cytoplasm and form an adherent monolayer. Mitotic figures and binucleated giant cells (arrowhead) are present. Giemsa stain. (b, c) Dose-response curves of FeLeco-G7 cells (69th passage) treated with (b) carboplatin or (c) SN-38. The IC50 values of carboplatin and SN-38 were 237.1 µmol/L and 1.37 µmol/L, respectively. (d–i) Immunofluorescence at 86th passage. Cells are immunolabeled (green) for (d) cytokeratin (CK) 20 and negative for (e) CD10, (f) CK7, and (g) p53. (h) Immunoreactivity for CDX2 is rarely observed (arrowhead). (i) Diffuse membranous and slight cytoplasmic (arrowhead) immunoreactivity of β-catenin is observed, whereas nuclear immunoreactivity is not found. DAPI nuclear counterstain. DAPI, 4′,6-diamidino-2-phenylindole.
Immunocytochemical examination revealed that >95% of FeLeco-G7 cells were immunopositive for CK20 and all cells were immunonegative for CD10, CK7, and p53 (Fig. 2d–g). Nuclear CDX2 was observed in <1% of cells (Fig. 2h). The β-catenin was detected mainly in the cell membrane, but not in the nuclei (Fig. 2i). These immunocytochemical features were retained at the 13th, 86th, and 200th passages (Supplemental Figures S5 and S6).
By Western blot analysis of the protein samples of FeLeco-G7 cells from the 1st, 40th, and 200th passages, anti-β-catenin, CDX2, and CK20 antibodies labeled bands consistent with the molecular weights of feline β-catenin, CDX2, and CK20, respectively (Supplemental Figure S7).
Mutation Analysis of the KRAS and TP53 Gene
Extracted genomic DNA was successfully amplified with the primers for the KRAS gene. The NCBI-BLAST result showed >99% identity between the amplicon sequence and feline chromosome B4 58593762 to 58593989. The DNA sequencing analysis identified no KRAS mutations, including KRAS codons 12 and 13.
The PCR amplification of TP53 exons 5 and 6 was successful in unaffected feline tissue, but no amplicons were detected in DNA from FeLeco-G7 cells at each passage (Supplemental Figure S8). Agarose gel electrophoresis of PCR products obtained from a primer pair for TP53 exons 7 and 8 displayed 2 extra bands in addition to the expected band in FeLeco-G7 cells at each passage (Supplemental Figure S9). The DNA sequencing analysis of each amplicon was performed; however, a chromatogram of sufficient quality was not obtained for any of the samples examined.
Xenotransplantation to SCID Mice
In subcutaneous transplantation assays, a dome-shaped subcutaneous mass was observed in all 3 SCID mice 7 to 10 days after the cell inoculation (Fig. 3a). Hemorrhage and necrosis were observed in the center and on the surface of the masses. Histologically, the masses consisted of cuboidal to columnar cells with glandular formations (Fig. 3b). Neoplastic cells had a moderate amphophilic cytoplasm and an oval to round nucleus with 1 or 2 prominent nucleoli (Fig. 3b). Anisokaryosis was moderate, and mitotic figures were frequent, >100/10 high-power fields (1.96 mm2), 400×. Distant metastases were not detected in any mice. Transplanted FeLeco-G7 cells were immunopositive for CDX2 and CK20 and immunonegative for CD10, CK7, and p53 (Fig. 3c–g). Immunoreactivity for β-catenin was found in the cytoplasm and a small number of cells also displayed nuclear immunoreactivity (Fig. 3h).
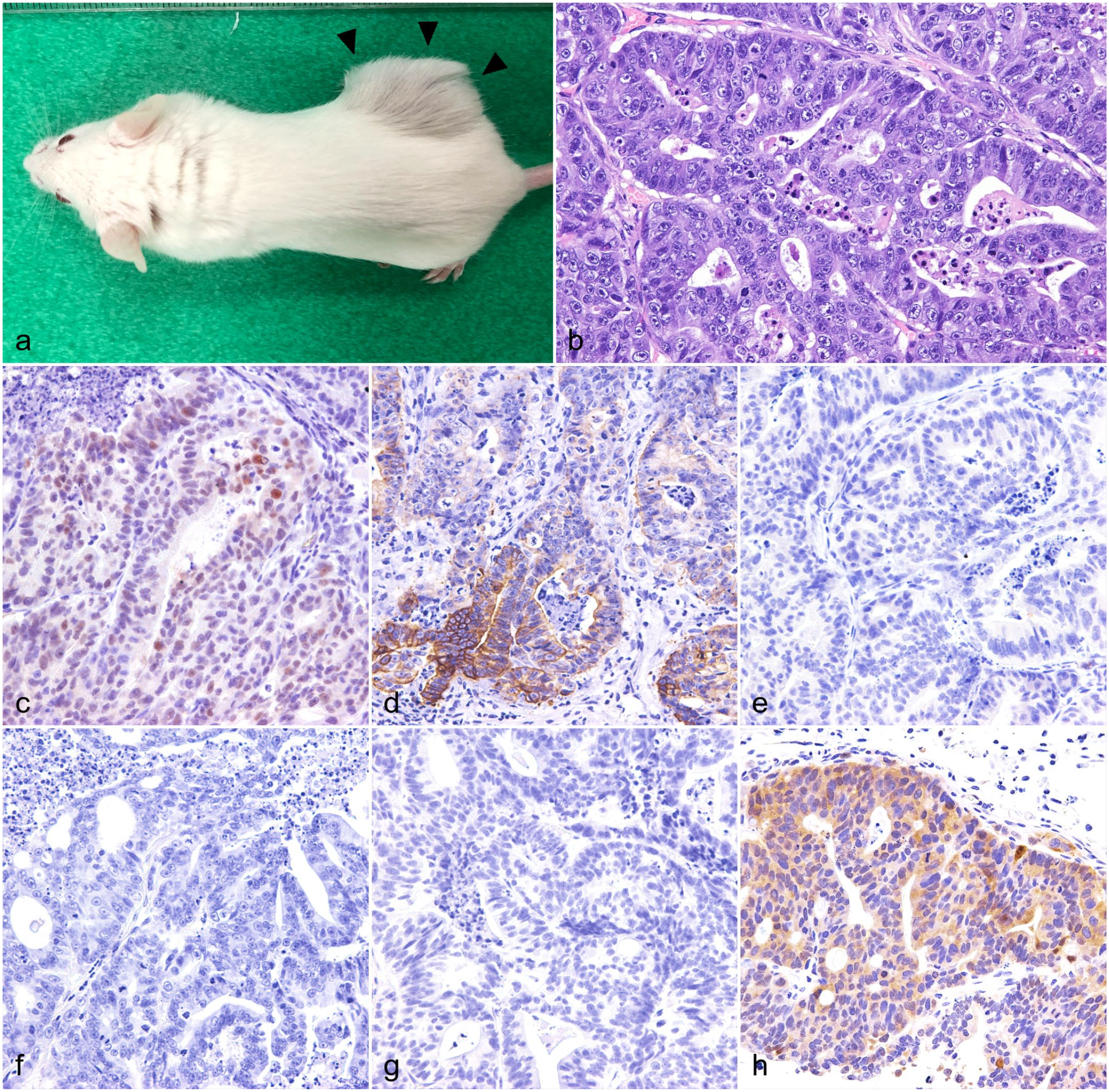
A feline colorectal adenocarcinoma cell line (FeLeco-G7, 50th passage), subcutaneous xenograft, CB17/IcrJcl-Prkdcscid mouse. (a) Subcutaneously transplanted FeLeco-G7 cells form a dome-shaped subcutaneous mass (arrowheads). (b) The mass comprises cuboidal to columnar cells with glandular formation. Hematoxylin and eosin. (c–g) Neoplastic cells are immunolabeled for (c) CDX2 and (d) cytokeratin (CK) 20 and negative for (e) CD10, (f) CK7, and (g) p53. (h) Nuclear immunoreactivity for β-catenin was rarely observed.
In IR transplantation assays, severe weight loss was detected in one of the 3 SCID mice (mouse No. 2), 1 month after cell inoculation (Supplemental Figure S10). This mouse had a slightly swollen anus without masses (Fig. 4a). Necropsy revealed a thickened rectal wall, dilated lymphatic vessels of the rectum, an enlarged medial iliac lymph node, and a mesenteric nodule (Fig. 4b). Plaque-shaped mucosal elevations were also found in the rectal mucosa (Fig. 4c). Histologically, diffuse lymphatic vessel invasion by tumor cells was observed in the rectal mucosa (Fig. 4d). Metastatic lesions were observed in the medial iliac and pancreaticoduodenal lymph nodes (Fig. 4e). Perianal swelling was observed in the two other SCID mice, 1 month after the cell inoculation (Fig. 4f). Necropsy revealed a mass surrounding the rectum and enlarged para-aortic lymph nodes in each of these 2 mice. Tumor cells invading the muscularis externa of the rectum were histologically observed (Fig. 4g). A metastatic lesion was detected in the para-aortic lymph node in 2 mice, and 1 mouse (mouse No. 3) had a metastasis to the lung (Fig. 4h). The immunohistochemical features of neoplastic cells in these lesions were consistent with those in the subcutaneous transplantation assay.
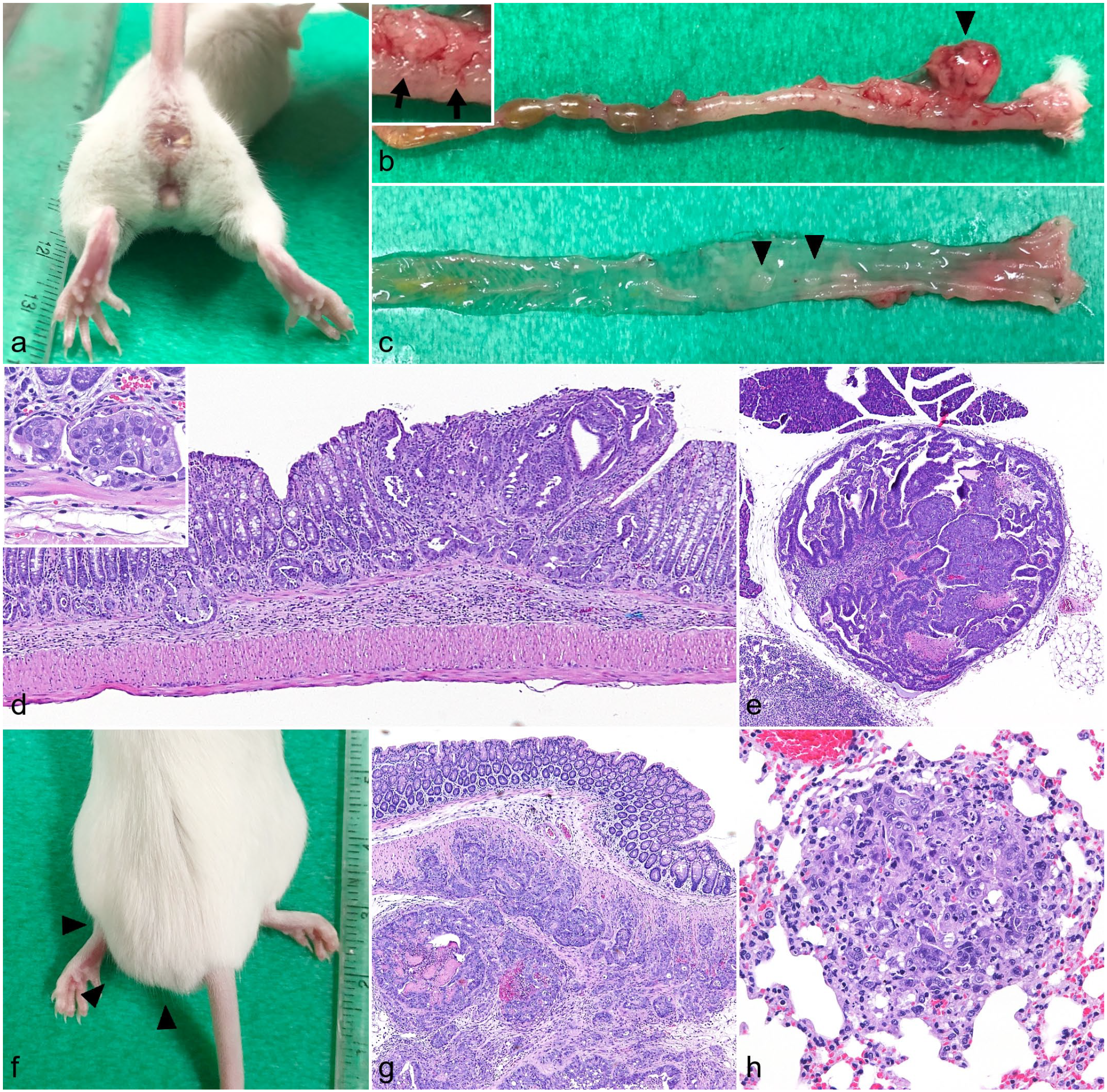
A feline colorectal adenocarcinoma cell line (FeLeco-G7, 50th passage), Orthotopic xenograft, CB17/IcrJcl-Prkdcscid mouse. (a) Slight swelling of the anus without observable masses. (b–d) Colorectum. (b) An enlarged medial iliac lymph node (arrowhead) and dilated lymph vessels (inset, arrows) are found. (c) Mucosal plaques are observed (arrowheads). (d) Orthotopic-transplanted FeLeco-G7 cells expanding the mucosa and invading lymph vessels (inset). Hematoxylin and eosin (HE). (e) Pancreaticoduodenal lymph nodes. Metastatic lesion. HE. (f) Perianal swelling (arrowheads). (g) Neoplastic cells infiltrate the muscularis externa. HE. (h) Lung. A metastatic nodule. HE.
Discussion
To the best of our knowledge, this is the first report of the successful establishment of a feline colorectal adenocarcinoma cell line, FeLeco-G7. The morphological characteristics of this cell line are similar to those of human colorectal cancer cell lines. 5 The population-doubling time of FeLeco-G7 (17.2 hours) was shorter than those of most human colorectal cancer cell lines, 4 indicating that FeLeco-G7 is a rapidly proliferating cell line. In humans, chromosomal instability is detected in approximately 70% of colorectal cancer cases and is common in colorectal cancer cell lines.5,15 Chromosomal instability is typically associated with karyotypic heterogeneity and is recognized as a common hallmark of tumorigenesis in many organs.3,24 However, the number of chromosomes of FeLeco-G7 cells was approximately 38, with few variations. This result suggests little or no chromosomal instability in FeLeco-G7 cells.
Carboplatin is a platinum agent widely used as chemotherapy for various tumors in cats. A retrospective study indicates that carboplatin may prolong the overall survival in cats with colonic adenocarcinoma. 2 However, the calculated IC50 value of carboplatin on FeLeco-G7 cells (237.1 µmol/L) was higher than what has been reported on human colorectal cancer cell lines (median 69.5 µmol/L, ranging between 25.6 and 168.5 µmol/L),9,16 suggesting that FeLeco-G7 is a carboplatin-resistant cell line.
In humans, irinotecan, an anticancer prodrug, is a key therapeutic drug for advanced colorectal cancer. 7 The SN-38 is the active metabolite of irinotecan, and inhibits DNA topoisomerase I. On FeLeco-G7 cells, the IC50 value of SN-38 (1.37 µmol/L) was higher than for most human colorectal cancer cell lines (median 0.06 µmol/L, ranging between 578.2 and 0.0003 µmol/L). 25 Moreover, the dose-response curve of FeLeco-G7 cells with SN-38 treatment did not reach 0 at highest concentration in this study. These results suggest that FeLeco-G7 is also an SN-38-resistant cell line.
The results of immunocytochemical and immunohistochemical examinations of FeLeco-G7 cells for CD10, CK7, and CK20 were consistent with those of the original tumor. The results of Western blot analysis also indicated the expression of β-catenin, CDX2, and CK20 in the cultured cells. However, the immunoreactivity of FeLeco-G7 cells for CDX2 in vitro was weaker than that of original tumor cells. Xenotransplanted FeLeco-G7 cells showed similar immunopositivity for CDX2 to that of the original tumor. These contradictory results suggest that differences in the biological environment in vivo and in vitro affected the protein expression of CDX2 in FeLeco-G7 cells.
Immunohistochemistry for β-catenin and p53 is widely used as a surrogate for the analysis of wingless-related integration site (WNT)/β-catenin signaling activity and TP53 mutations. The abnormal nuclear accumulation of β-catenin and p53 suggests the aberrant activation of WNT/β-catenin signaling and TP53 mutations, respectively.11,13 The activation of WNT/β-catenin signaling and mutations in the TP53 and KRAS genes are considered to contribute to the development of colorectal adenocarcinoma in humans.1,6,10 On the contrary, in cats, a previous study suggested that KRAS mutations were occasionally detected, and nuclear immunopositivity for β-catenin and p53 was uncommon in colorectal adenocarcinoma. 23 Consistent with the feline study, FeLeco-G7 cells and the original tumor rarely showed the nuclear accumulation of β-catenin and were immunonegative for p53. However, mutation analysis of the TP53 gene in FeLeco-G7 resulted in failure of amplification with primer pairs for exons 5 and 6, and anomalous amplicons with primer pairs for exons 7 and 8. These results suggest that there are complex mutations including structural abnormalities: deletions, insertions, inversions, or translocations of the TP53 gene in FeLeco-G7 cells. Unfortunately, sequence analysis in this study failed to clarify the details of TP53 mutations. However, sequence analysis revealed no mutations in exon 1 of the KRAS gene. Taken together, TP53 mutations may play a more important role than the activation of WNT/β-catenin signaling and KRAS mutations in the tumorigenesis of FeLeco-G7 cells. Further genetic research on FeLeco-G7 cells may contribute to a more detailed understanding of the mechanisms underlying the development of feline colorectal adenocarcinoma.
It is widely recognized that the passage number affects cell properties. In this study, the results of the wound healing assay showed a significant difference between the percentage of wound closure in the 1st and 200th passages at 24 hours. In contrast, there were no significant differences among the passage numbers at 48 hours. Based on these results, increasing the number of passages may shorten the induction period of FeLeco-G7 cells. Meanwhile, the results of morphological characterization, immunocytochemistry, Western blot analysis, and electrophoretic analysis of theTP53 gene did not vary according to the passage number. Therefore, it is suggested that FeLeco-G7 is a cell line with a stable phenotype during multiple passages.
The subcutaneous and IR xenotransplantation of FeLeco-G7 cells into SCID mice resulted in the successful development of tumor masses. These tumors exhibited similar histological and immunohistochemical features to those of the original tumor. In subcutaneously transplanted mice, tumor masses were all confined to the dermis and/or subcutis, and metastasis to the lymph nodes or distant organs was not detected. In contrast, in IR transplanted mice, metastatic lesions were found in the regional lymph nodes of all 3 mice, and lung metastasis was also detected in one of 3 mice (mouse No. 3). Furthermore, 1 mouse (mouse No. 2) showed severe cachexia and diffuse involvement of the rectal mucosa by transplanted FeLeco-G7 cells. These results on IR-transplanted mice support the invasive nature of FeLeco-G7 cells, which resembles the findings obtained in cats with colorectal adenocarcinoma.2,19 Therefore, the IR transplantation of FeLeco-G7 is a good experimental model for feline colorectal adenocarcinoma, replicating primary tumor growth and spontaneous distant metastasis.
In conclusion, we established a novel cell line of feline colorectal adenocarcinoma, FeLeco-G7. This cell line showed rapid proliferation, mild or no chromosomal aneuploidy, and high IC50 values of carboplatin and SN-38. The immunocytochemical features of FeLeco-G7 cells were consistent with the original tumor. Mutation analysis suggested TP53 gene alteration in the cells, but no mutations in KRAS gene exon 1 were found. Xenotransplantation assays showed the tumorigenicity of FeLeco-G7 cells in immunodeficient mice, and metastasis to the regional lymph nodes and/or lungs was detected in all mice, following orthotopic injection. FeLeco-G7 cells and orthotopic-transplanted mice may serve as a useful model of feline colorectal adenocarcinoma that will enable further studies on the disease.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231189858 – Supplemental material for Establishment and characterization of a novel cell line and xenotransplant mouse model derived from feline colorectal adenocarcinoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858231189858 for Establishment and characterization of a novel cell line and xenotransplant mouse model derived from feline colorectal adenocarcinoma by Mizuho Uneyama, James K. Chambers, Tadashi Fujii, Ko Nakashima and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI, Grant Number JP21J13110.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
