Abstract
A 10-year-old, neutered, male Domestic Shorthair cat was presented to the teaching hospital for labored breathing, anorexia, and weight loss of several months duration. External examination revealed distortion of the bridge of the nose and pink fleshy polyps protruding from each nostril. The cat was euthanized and submitted for postmortem examination. In addition to the external findings, the nasal cavity had extensive bone and cartilage loss and contained a tan firm mass in the caudal region of the nasal cavity near the cribriform plate. On histologic examination, the mass was a nasal adenocarcinoma, and the polyps were composed of hyperplastic nasal epithelium and submucosal stroma that contained sporangia consistent with Rhinosporidium seeberi.
A 10-year-old, neutered, male Domestic Shorthair cat was presented to the veterinary teaching hospital for labored breathing, anorexia, and weight loss of several months duration. This was an exclusively outdoor “barn cat” used to hunt and kill vermin. Core vaccinations were current. Horses and other cats were kept on the property. On physical examination, the bridge of the nose was swollen and deformed; pink fleshy polyps protruded from each nostril. Limited airflow through the nares was noted, and the breathing was loud and stertorous with some wheezing. The polyps were approximately 7 × 5 × 5 mm and extended minimally into the nasal cavity. The cat was euthanized and submitted for postmortem examination. In addition to the external findings, the nasal cavity had extensive bone and cartilage loss and contained a tan, firm mass (15 mm x 7 mm x 5 mm) in the caudal region of the nasal cavity near the cribriform plate (Fig. 1).
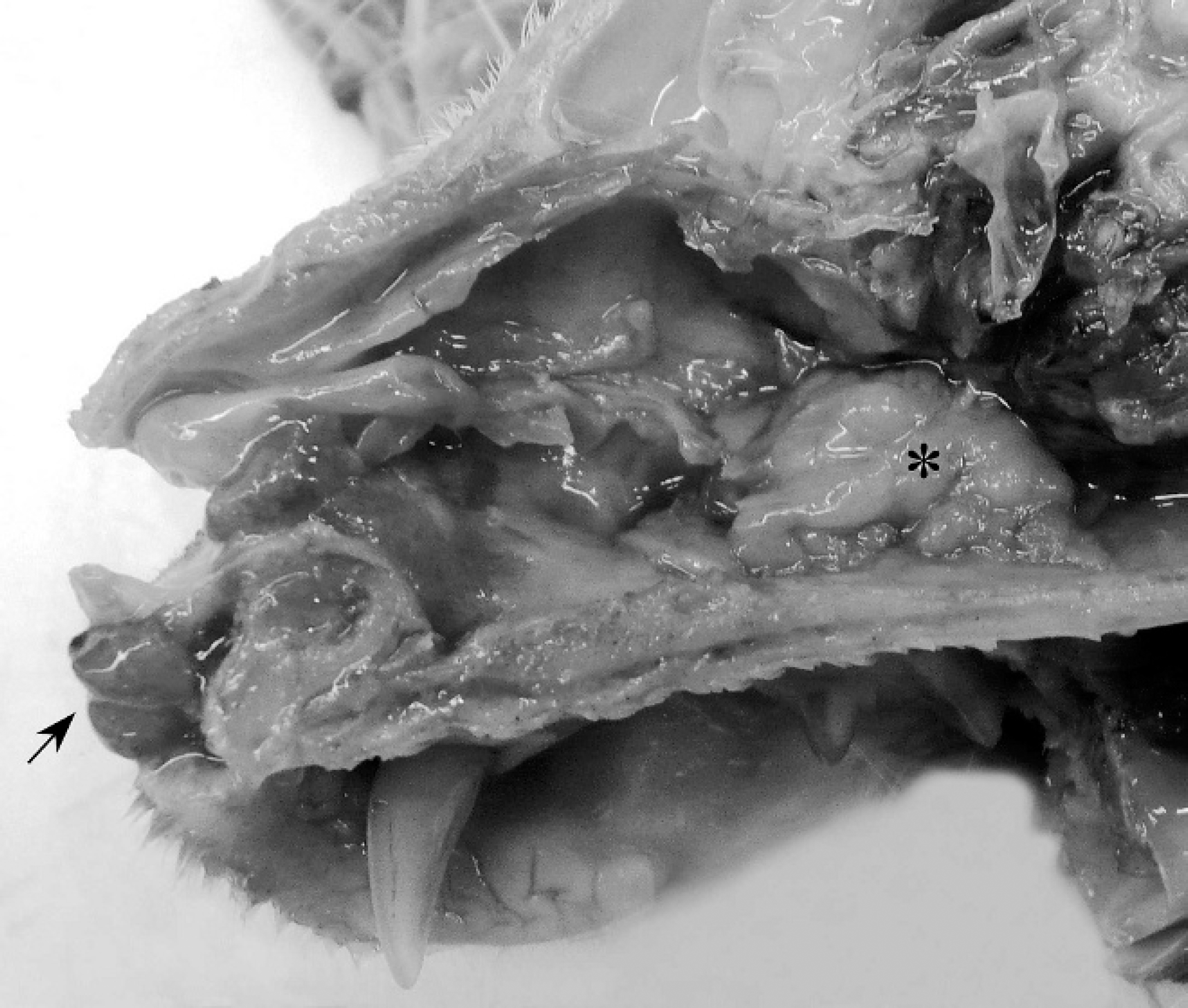
Sagittal section of the head. Polyps caused by Rhinosporidium seeberi are present in the nares (arrow), and an adenocarcinoma is located in the caudal region of the nasal cavity (asterisk).
Tissues were fixed in 10% neutral buffered formalin and routinely processed for histopathologic examination. The intranasal mass was composed of neoplastic cuboidal glandular epithelial cells that formed tubular to tubulopapillary structures within a thick collagenous stroma (Fig. 2). Individual neoplastic cells had a moderate amount of pale eosinophilic cytoplasm and indistinct cytoplasmic borders and were frequently piled on top of one another. Nuclei were generally round and open, with a single nucleolus. Mild anisocytosis and moderate aniso-karyosis were observed. Mitotic figures averaged 2 per 400x microscopic field of view. The histologic diagnosis was nasal adenocarcinoma. Numerous plasma cells and lymphocytes were present between the neoplasm and the overlying respiratory epithelium. The polypoid masses were composed of hyperplastic respiratory epithelium and thickened submucosa containing sporangia at different stages of maturation, ruptured sporangial casings, endo-spores, viable and degenerate neutrophils, macrophages, lymphocytes, and plasma cells (Fig. 3). Juvenile sporangia ranged from 10 to 50 μm in diameter, had a 2–3-μm thick eosinophilic wall, and contained a central nucleus surrounded by flocculent basophilic cytoplasm. Mature sporangia were up to 200 mm in diameter and contained numerous 2–5-μm basophilic endospores. Several mature sporangia were within the hyperplastic mucosa and opened to the surface releasing endospores into the nasal cavity and external environment (Fig. 3). Characteristics of the organism were consistent with Rhinosporidium seeberi.
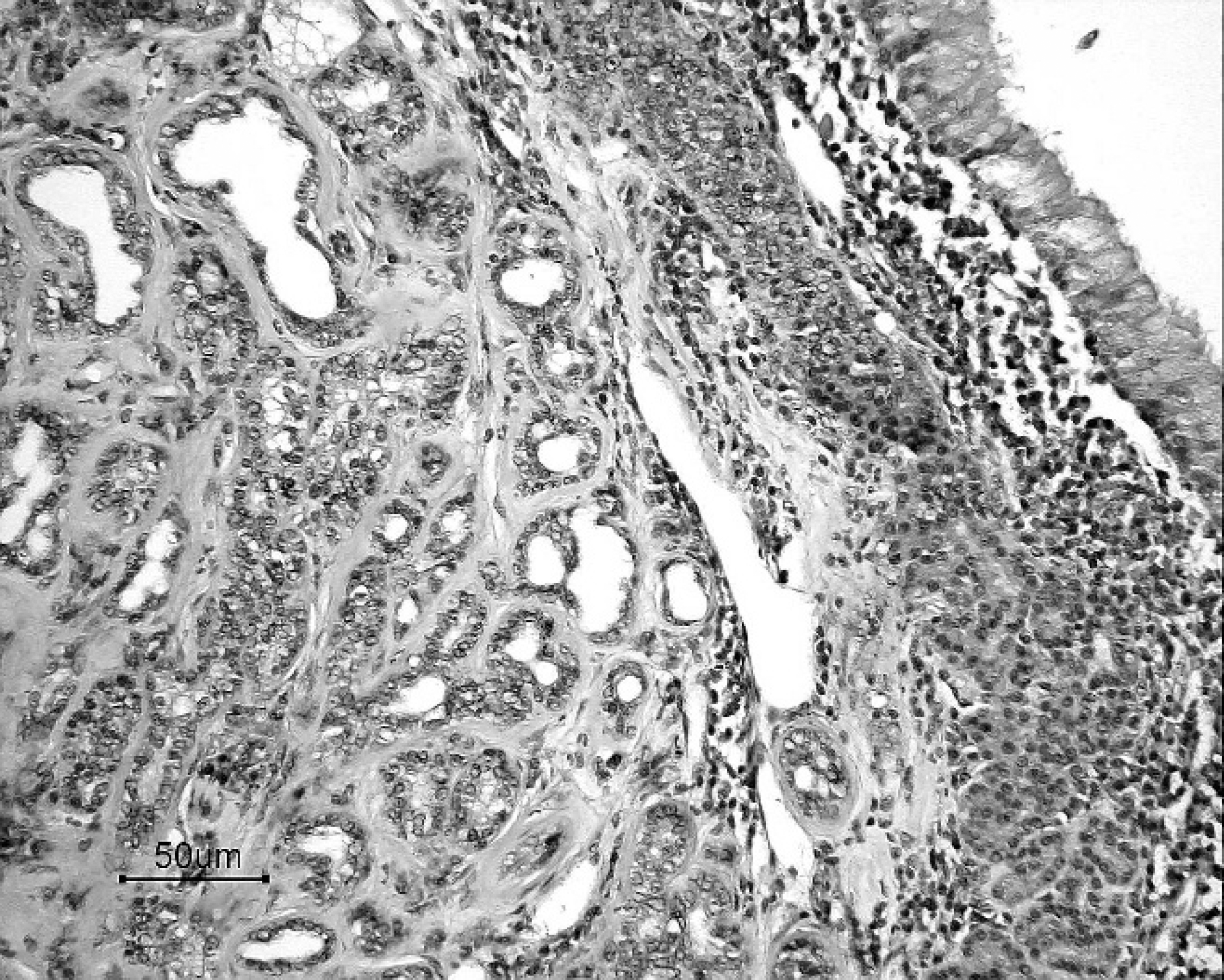
Adenocarcinoma from the caudal region of nasal cavity. Neoplastic epithelial cells form tubular and tubulopapillary structures, which are supported by a thick collagenous stroma. Many lymphocytes and plasma cells are present beneath the mucosa. Hematoxylin and eosin. Bar = 50 μm.
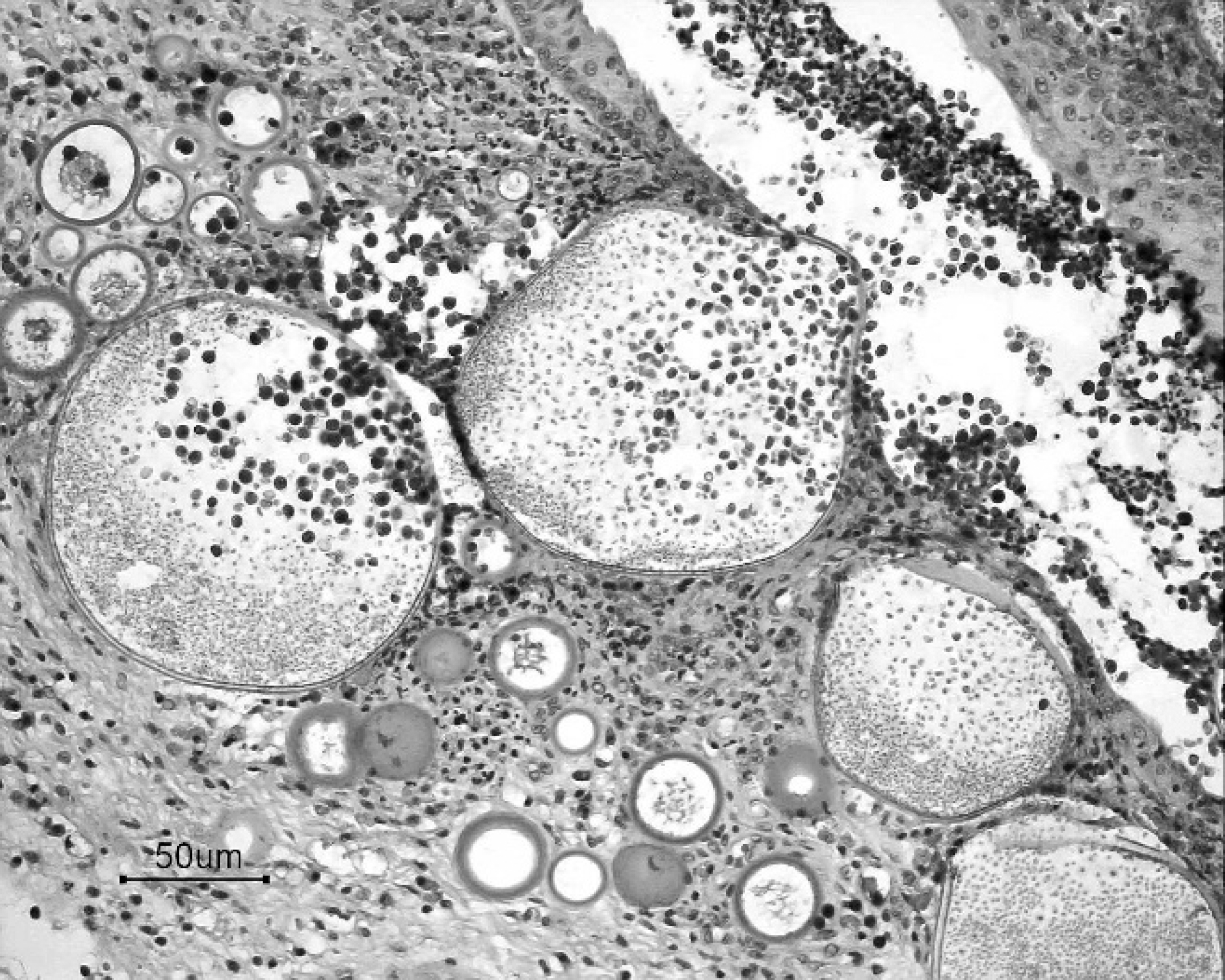
Nasal polyp; feline rhinosporidiosis. Pyogranulo-matous and hyperplastic rhinitis with mature and immature sporangia. Note the release of endospores from mature sporangium into the nasal cavity. Hematoxylin and eosin. Bar = 50 μm.
The majority of nasal neoplasms in dogs and cats are malignant, with a locally invasive and destructive behavior. The most common intranasal tumors in cats are lymphoma and adenocarcinoma. 3,6 Clinical signs include nasal discharge, dyspnea, deformation of the facial bones, anorexia, and weight loss. Malignant nasal neoplasms can be treated with radiation therapy (with or without surgery to remove or debulk the tumor, or both), chemotherapy, or both. The prognosis for untreated malignant nasal neoplasms is poor, with a survival time of only a few months in most cases. 3 Euthanasia is often requested because of declining health and diminished quality of life.
Rhinosporidiosis, caused by R. seeberi, is a cause of nasal polyps in humans, horses, and dogs, and rarely in other domestic animals and fowl. 2 The phylogenetic classification of R. seeberi has long been controversial. In the past, it was classified as a fungus; however, recent analysis places it as an aquatic protist in the class Mesomycetozoea (between fungi and animals). 1,2,4,5 Although successful attempts to grow R. seeberi in culture have been described, most microbiologists consider R. seeberi to be intractable to culture. 1
Rhinosporidiosis is endemic in the wet environments of India, Sri Lanka, and South America and is reported sporadically from other parts of the world. 1,2,4 Cases in the United States are generally localized to the southern states. Rhinosporidium seeberi is stimulated to mature and release endospores after exposure to water; thus, the disease is associated with wet environments and the host's moist mucous membranes. 1 The association between infected animals and swimming or bathing in stagnant bodies of water is strong. In arid countries, most human infections are ocular, and dust is thought to be the fomite; endospore maturation and release is stimulated by mucous secretions. Mucous membrane damage might be a predisposing factor. The infective unit is the spore that penetrates the mucosa, localizes in the subepithelial connective tissue, and undergoes maturation from an immature to intermediate to mature sporangium, which then releases endospores into tissue or out through an apical pore into the environment, and the life cycle is reinitiated. 1
Clinical findings include wheezing, sneezing, nasal discharge, and epistaxis. Polypoid masses might be visible in the nares or could be identified via rhinoscopy of the rostral nasal cavity. Diagnosis is typically accomplished by cytologic or histologic examination, or both. Surgical excision is the treatment of choice and can be curative. Pharmaceutical treatments have included dapsone, used for treating mycobacterial and some protozoal infections, and ketoconazole, an antifungal drug. 1
Feline rhinosporidiosis has been reported in 2 outdoor cats, both in the United States (North Carolina and Washington, DC). 1,4,5 Like the previous reports, the cat in the present study had spent the majority of its life outdoors and was from the East Coast. The present case is unique in that the cat also had a concurrent nasal adenocarcinoma. Mucosal damage caused by the neoplasm might have predisposed this animal to infection with R. seeberi.
