Abstract
Two 12.5-year-old castrated male Persian cats from the same household, whose dams were littermates, presented simultaneously with gastric adenocarcinoma associated with proliferative and fibrosing gastritis. Intralesional adult Ollulanus tricuspis nematodes and rare surface-associated spiral-shaped bacteria were identified in one cat. No etiologic agents were identified in tissues from the second cat; however, gastric mucosa was examined following anthelmintic treatment. Clinical signs in each cat had commenced 2 months apart and included vomiting, hematemesis, intermittent melena, and weight loss. This is the first report of gastric adenocarcinoma occurring in housemate cats or cats of common descent. Carcinogenesis may have been influenced by shared undetermined genetic and environmental factors, possibly including Ollulanus tricuspis, spiral-shaped bacteria, or other etiologies for chronic gastritis that remain unidentified.
Gastric adenocarcinoma is exceedingly uncommon in cats. No predisposing factors have been identified in cats that might be associated with gastric mucosal transformation and tumorigenesis. This manuscript reports the simultaneous occurrence of gastric adenocarcinoma in two related cats from the same household and discusses some potential contributory factors.
Two 12.5-year-old, castrated male Persian cats were presented to Colorado State University Veterinary Teaching Hospital (CSU-VTH), on the same day. Clinical signs had emerged 2 months apart and included vomiting, hematemesis, intermittent melena, hyporexia, and weight loss of 4–6 months duration. The cats were of common maternal lineage because they were born from queens that were littermates, although the sires were unrelated. The cats were raised strictly indoors within the same household. They had been the only cats in the household from the time they were young adults. There was no known exposure to cats outside of the household. Neither cat had been administered anthelmintic medication as an adult.
Cat No. 1 was referred to CSU-VTH with a diagnosis of mild, chronic, multifocal, superficial lymphoplasmacytic gastritis based on endoscopic biopsies taken 3 months prior. The referring veterinarian described the gross appearance of the fundic mucosa as irregular. The cat had been treated with sucralfate, cimetidine, cefazolin, enrofloxacin, chlorpheniramine, prednisone, metronidazole, and amoxicillin, with no response.
Upon presentation to CSU-VTH, cat No. 1 was in good body condition and no abnormalities were detected by physical examination. No significant abnormalities were identified with a complete blood count, serum biochemistry panel, or urinalysis. Endoscopy identified coarsely nodular fundic mucosa and three 2- to 4-cm-diameter polypoid masses located in the fundic and cardiac gastric mucosa. Scant spiral-shaped bacteria were identified in an impression smear made from the cut surface of one fundic mass; however, a urea slant bacterial culture grew no urease-producing bacteria. The cat was treated with metronidazole, amoxicillin liquid drops, and famotidine for 2 weeks and sucralfate suspension for 5 days.
Endoscopic biopsies revealed severe, diffuse, fibrosing, and proliferative gastritis with intralesional nematode parasites and rare surface-associated spiral-shaped bacteria (Fig. 1). The mucosa was markedly thickened with elongated, tortuous to ectatic gastric pits lined by hyperplastic mucous neck cells. The lamina propria contained abundant fibrous connective tissue that replaced and separated gastric pits. Lymphocytes, plasmacytes, and few follicular lymphoid aggregates, neutrophils, and globule leukocytes were distributed throughout the lamina propria. Profiles of nematodes were most often located within superficial dilated gastric pits. Nematodes averaged 30 µm in diameter and had thin, ridged cuticles, tricuspid tails, and poorly discernable platymyarian-meromyarian musculature. The nematodes were identified as Ollulanus tricuspis based on their morphologic characteristics and location in the stomach. Rare spiral-shaped bacteria that stained with cresyl echt violet (CEV) were associated with the surface of luminal epithelial cells. Both cats were treated with fenbendazole.
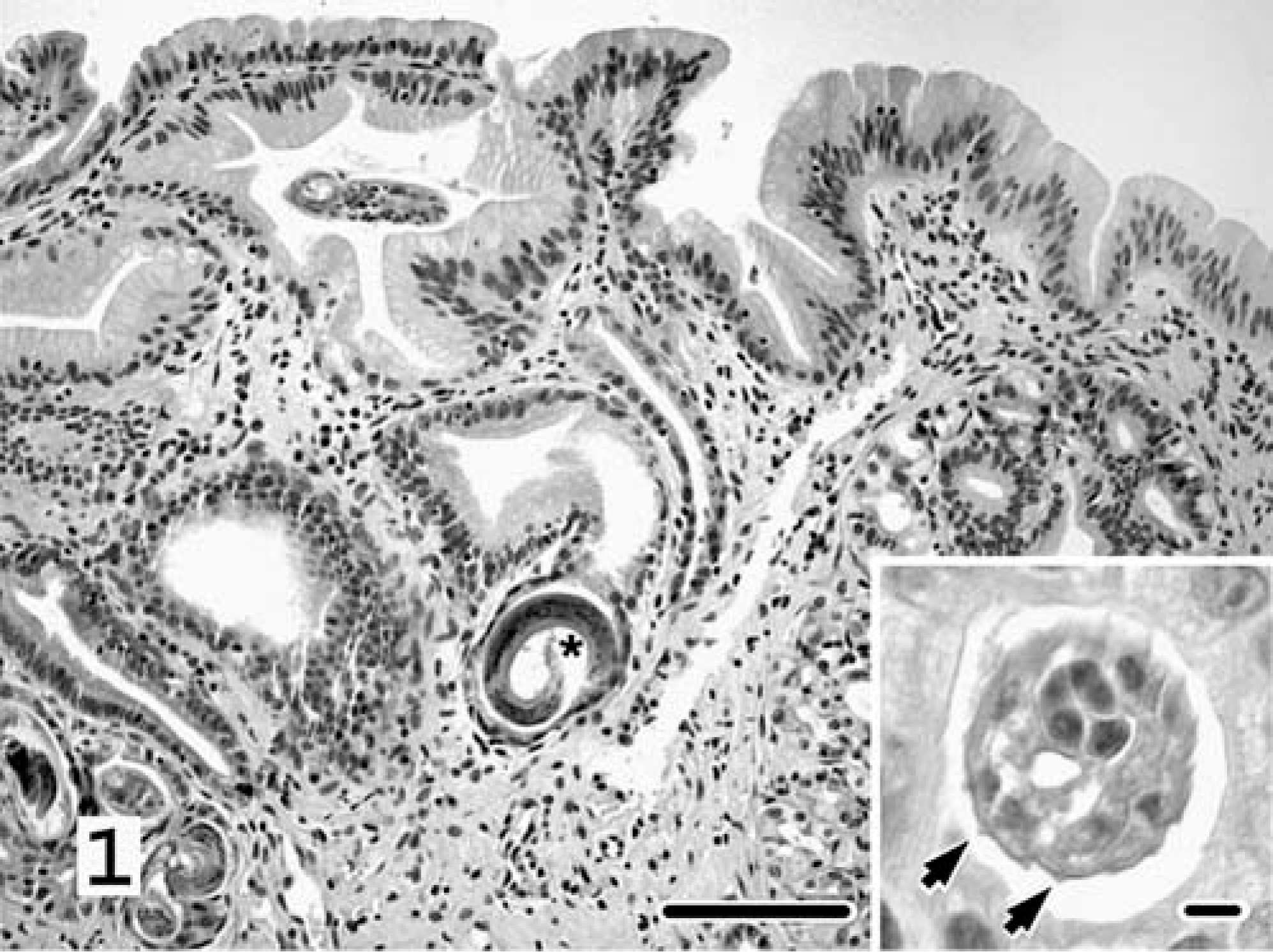
Stomach, endoscopic biopsy; cat No. 1. The mucosa contains dilated gastric pits that are separated by abundant connective tissue and are lined by hyperplastic mucous neck cells. Profiles of nematodes are within multiple glands. Asterisk denotes the characteristic tricuspid tail. Inset emphasizes distinctive longitudinal cuticular ridges (arrows). HE. Bar = 100 µm. Inset bar = 10 µm.
Despite immediate clinical improvement following fenbendazole treatment, clinical signs recommenced and worsened, and cat No. 1 returned 2 weeks later to CSU-VTH for an exploratory laparotomy. Gastric adenocarcinoma was identified in surgical biopsies taken from a dark brown, 1-cm-diameter polypoid mass that protruded from the fundic mucosa (Fig. 2). Dilated gastric pits were segmentally lined by neoplastic cuboidal to polygonal epithelial cells that piled multiple cell layers thick. There was segmental loss of the glandular basement membrane. The cells had distinct cell margins; plump, deep basophilic cytoplasm; and large, ovoid, basilar, hypochromatic nuclei with large and multiple nucleoli. Neoplastic cells had high nuclear ° cytoplasmic ratio and marked anisokaryosis, and averaged two mitoses per five high powered fields. A few of these cells had a “signet ring” appearance, characterized by large cytoplasmic vacuoles that peripherally displaced and compressed nuclei. Tubuloacinar structures and nests of similar neoplastic epithelium invaded the deep lamina propria, submucosa, and muscularis and were within vessels. Noncancerous areas had fibrosing and proliferative gastritis as previously described. Rare surface-associated spiral-shaped bacteria, which stained with CEV, were within gastric pits. Intralesional nematodes were not identified in the sections examined. In addition, parasites were not identified with examination of gastric juice (2.5 ml) that was aspirated at surgery and concentrated using the Baermann technique.
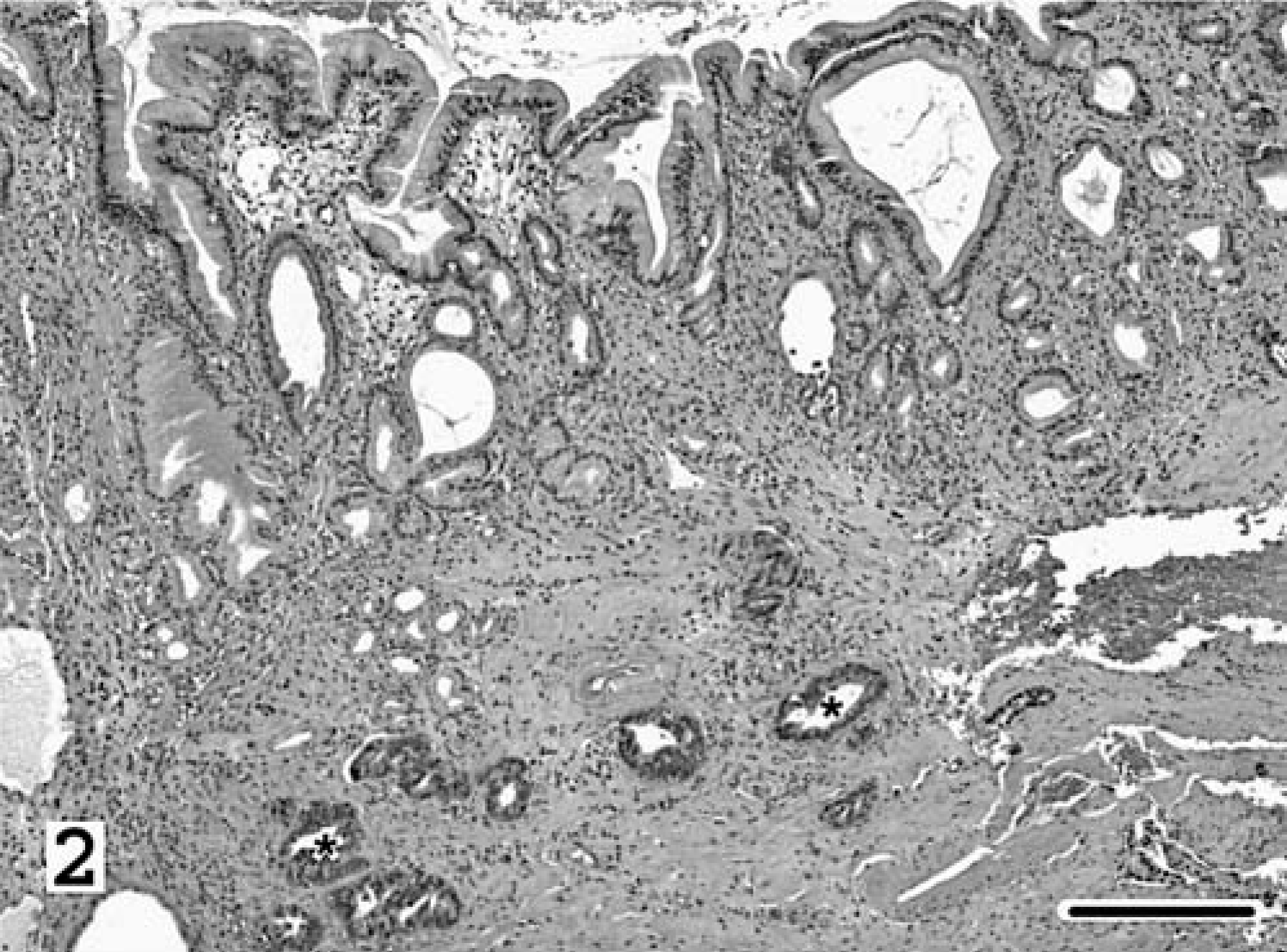
Stomach, surgical biopsy; cat No. 1. Asterisks denote tubuloacinar structures comprised of ← neoplastic epithelium that invade the submucosa. The overlying mucosa is affected with mucous neck cell hyperplasia, lymphoplasmacytic inflammation, and fibrosis. HE; Bar = 250 µm.
Cat No. 1 was euthanatized twelve days after surgery. At necropsy, omentum was adhered to the serosal surface of a 3-cm-long × 1.5-cm-thick, oblong, white, firm mural mass that regionally expanded the wall of the cranial aspect of the gastric fundus. The mucosa overlying the mass was thickened and lacked rugal folds. The remaining mucosa, except in the pyloric area, was diffusely thickened and coarsely nodular. Histologic evaluation of the stomach confirmed gastric adenocarcinoma and chronic proliferative and fibrosing gastritis. Intralesional nematodes were not observed, although spiral-shaped bacteria were. Metastasis was identified in the liver and the kidney.
Cat No. 2 had developed similar clinical signs 2 months before cat No. 1, but was referred to CSU at the same time as cat No. 1 with a diagnosis of moderate, chronic, regional, ulcerative and pleocellular gastritis with moderate epithelial dysplasia, based on endoscopic biopsies taken 5 months prior. The referring veterinarian described the gross appearance of the fundic mucosa as irregular. The cat had been treated with famotidine, sucralfate, and prednisone with limited response.
On presentation to CSU, cat No. 2 had lost approximately 2.5 kg of body weight, despite being fed six to seven meals of pureed, canned cat food per day. Physical examination revealed a palpable mass in the right cranial abdomen. Abdominal ultrasonography revealed diffuse thickening of the stomach wall (20 mm) with heterogenous echogenicity and loss of gastric layer distinction. A 3-cm × 1.5-cm mass extended from the gastric wall into the lumen of the stomach. There were also several heteroechoic, ill-defined nodules throughout the hepatic parenchyma. Cytology from ultrasound-guided percutaneous fine-needle aspirates of gastric and hepatic masses was consistent with carcinoma. Four days after discharge, the cat was administered fenbendazole, as described above, based on the endoscopic biopsy results of cat No. 1 that had demonstrated Ollulanus tricuspis.
Cat No. 2 was euthanatized 3 weeks later. At necropsy, examination of the stomach revealed a firm, white, invasive, transmural mass that spanned the caudal surface of the body of the gastric fundus and regionally extended into the greater and lesser omentum, spleen, and left limb of the pancreas. The gastric mucosa overlying the mass was thickened up to four times normal with a central area of necrosis (Fig. 3). Except in the area of the pylorus, the remaining mucosa was diffusely thickened and nodular. Hepatic and pulmonary nodules consistent with metastases were present. Histologic evaluation identified gastric adenocarcinoma, similar to that described for cat No. 1, and confirmed hepatic and pulmonary metastasis. There was severe, diffuse, chronic proliferative and fibrosing gastritis, strikingly similar to that of cat No. 1. Nematode parasites were not seen in sections examined. Spiral-shaped bacteria were not identified with hematoxylin and eosin, Warthin-Starry silver, or CEV stains.
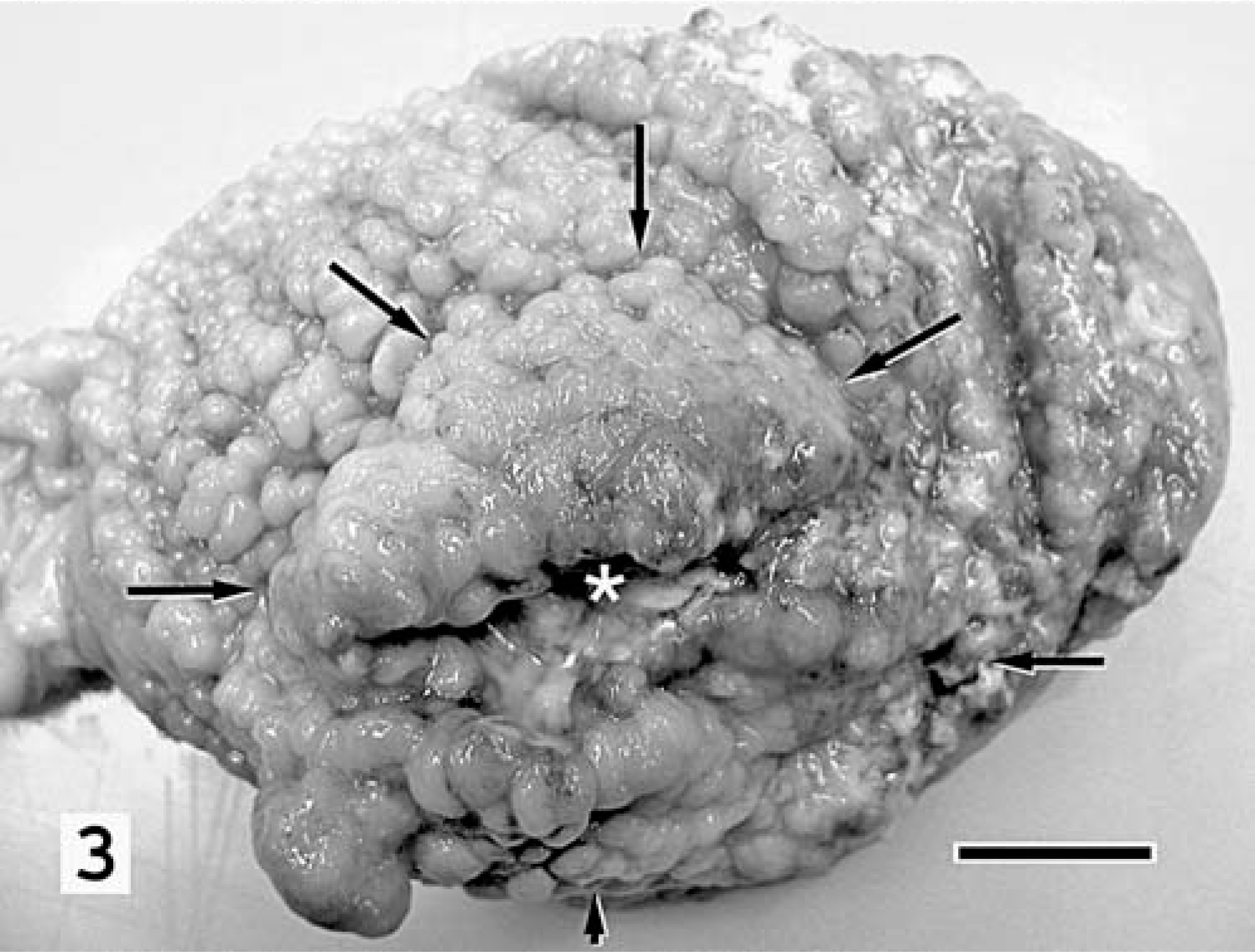
Stomach, mucosal surface; cat No. 2. The glandular mucosa is diffusely thickened and coarsely nodular. Regionally, nodules coalesce to form polypoid projections that overlay a transmural mass (arrows) with a central area of necrosis (asterisk). Bar = 1 cm.
Gastric adenocarcinoma occurring simultaneously in two cats of the same age and from the same household has not been reported. The unlikelihood of such an event suggests a common environmental, genetic, or infectious influence, or a combination of these factors. Gastrointestinal adenocarcinoma is the most common nonhematopoietic gastrointestinal tumor found in the cat. 24 However, gastric adenocarcinoma represents only 1% of all feline gastrointestinal adenocarcinomas, 24 with only eight reported cases. 1– 3, 17, 24 Therefore, it is surprising to detect two cats from the same household with this disease. Two retrospective studies have reported a male predilection for gastrointestinal adenocarcinoma. 3, 18 Although approximately two-thirds of feline gastrointestinal adenocarcinoma is reported in Siamese cats, a heritable pattern has not been recognized. 18, 24 A familial predisposition for gastric adenocarcinoma has recently been identified in Chow Chow dogs (E. McNeil, personal communication). Because these cats shared maternal lineage, the possible influence of genetic factors on carcinogenesis should be considered.
Ollulanus tricuspis is a viviparous trichostrongyle nematode of the feline stomach with a characteristic life cycle that is capable of completion within a single host. Cats usually become infected through ingestion of vomitus that contains any life stage of Ollulanus. 11 Long-haired cats may transmit these parasites more efficiently because of their propensity for hairball-associated vomiting related to frequent grooming. 8 Antemortem diagnosis of the infection requires microscopic examination of the vomitus, stomach contents, or gastric mucosa, because eggs and larvae are not typically passed in the feces. 11 In one study, evaluation of three histologic sections identified organisms in only half of infected cats; 9 therefore, histology is not a sensitive diagnostic approach.
The pathogenicity of O. tricuspis is poorly understood. O. tricuspis has been considered clinically insignificant by some authors. 9, 19 Clinical signs may be intermittent and include chronic vomiting, anorexia, or weight loss. 8, 11 The gastric mucosa may appear grossly normal, or may have erosions, excess mucus, or a nodular appearance. 7, 10, 19 There may be increased follicular lymphoid aggregates, globule leukocytes, and reactive fibrosis throughout the lamina propria. 9, 11 Dilated gastric pits or glandular atrophy may accompany these lesions. Adult O. tricuspis nematodes are usually localized to the mucus coating the luminal surface, or to the lumina of dilated gastric pits. Although epithelial hyperplasia has not been a consistent finding, trichostrongylids infecting other species, including Ostertagia ostertagia in cattle, Ostertagia circumsincta in small ruminants, Trichostrongylus axei in horses, and Nochtia nochti in Asian macaques, have been associated with proliferative gastric or abomasal lesions. 20, 22 Trichostrongylid gastritis in most species is typically characterized by similar mucous neck cell hyperplasia and by mild lymphocytic inflammation.
Helicobacter spp. have been associated with gastric adenocarcinoma in humans, ferrets, and a Syrian hamster. 5, 15 Gastric Helicobacter-like organisms (GHLOs) were associated with gastric adenocarcinoma in a cougar. 25 Helicobacter spp. have also been associated with gastric mucosal fibrosis and with eosinophilic, lymphoplasmacytic, or lymphofollicular gastritis in cats. 4, 6, 21 The failure of a urea slant bacterial culture prepared from the gastric mucosa of cat No. 1 to detect urease-producing organisms may indicate that organisms were present in numbers below detection level or were not Helicobacter spp. Although GHLOs may have contributed to gastritis and carcinogenesis in cat No. 1, GHLOs are commonly found in normal, healthy cats, and it is not unusual to incidentally observe small numbers in normal feline gastric biopsies. 16, 21 Rare spiral-shaped bacteria were microscopically associated with the gastric mucosa of cat No. 1; however, prior antibiotic therapy may have reduced bacteria numbers. In contrast, GHLOs are generally present in large numbers when associated with gastric inflammation. 12
In the two cases described herein, the characteristics of the gastritis, although nonspecific, were consistent with either gastric spirochetosis or ollulanosis. It is common for multiple cats within the same household to be simultaneously infected with Ollulanus tricuspis. 9 Nematodes were not detected in cat No. 2. However, histologic examination was performed after fenbendazole therapy, which is considered highly efficacious. 26 It is possible that cat No. 2 was never infected with Ollulanus. However, the clinical history and histologic changes, in the absence of spiral-shaped bacteria, support infection in cat No. 2 as well. The involvement of other unidentified etiologies cannot be definitively excluded.
There have been no reports of gastric adenocarcinoma associated with O. tricuspis infection in the cat. The relationship of these conditions remains uncertain, but the possible association is compelling because other helminths, such as Spirocerca lupi, Schistosoma sp., and Taenia taeniaeformis larvae, are associated with malignancy in various species. 13, 23 Noninfectious chronic inflammatory lesions affecting the cat have a propensity for malignant transformation, including vaccine-associated sarcoma and trauma-induced uveal sarcoma. 14, 27 Chronic gastritis, alone or in combination with other environmental and genetic factors, may have facilitated the development of gastric adenocarcinoma in these two related cats.
