Abstract
Sporotrichosis is a mycotic infection of the cutaneous and subcutaneous tissues caused by Sporothrix spp. that can also cause extracutaneous manifestations. This study aimed to characterize cutaneous and extracutaneous sporotrichosis lesions in cats. Over 1 year, 102 cats rescued by the Zoonoses Control Center of Belo Horizonte, Brazil, euthanized with clinical suspicion of feline sporotrichosis were evaluated. After euthanasia, the animals were evaluated by macroscopic, cytological, histopathological, and immunohistochemistry (IHC) examinations; fungal culture; and polymerase chain reaction (PCR). Sporothrix infection was identified by at least one diagnostic technique in all cats (n = 102) evaluated by postmortem examination, including 26/28 cases (93%) evaluated by IHC, 66/90 cases (73%) evaluated by cytology, 70/102 cases (68.6%) evaluated by histopathology, and 62/74 cases (84%) evaluated by fungal culture. Two cats had positive results only by fungal culture. Cytology and histopathology examinations were effective in diagnosing sporotrichosis, although IHC was needed to confirm the diagnosis in cats with low fungal loads. Sporothrix brasiliensis was confirmed by the sequencing of 3 samples. Skin lesions were characterized mainly by pyogranulomatous to granulomatous dermatitis (frequently with subcutaneous inflammation) with different intensities of Sporothrix spp. yeast. Extracutaneous findings associated with sporotrichosis included rhinitis or rhinosinusitis, lymphadenitis, pneumonia, meningitis, periorchitis, conjunctivitis, and glossitis. Extracutaneous infections were observed in 74/102 cases, and a possible association between the chronicity of the disease and the higher pathogenicity of this fungal species in cats requires further investigation.
Keywords
Sporotrichosis is a mycosis of the cutaneous and subcutaneous tissues caused by Sporothrix spp. that can also cause extracutaneous manifestations and mainly affects cats and humans. 21 Sporothrix infections can be acquired by traumatic inoculation of fungal propagules from soil or by bites and scratches of infected cats.21,40 Considering the advances in identifying Sporothrix spp., these organisms are now divided into clinical and environmental clades. The clinical clade includes S brasiliensis, S schenckii, S globosa, and S luriei, which are frequently isolated from humans and animals. The environmental clade includes S chilensis, S mexicana, S humicola, and S pallida, which rarely cause infections in mammals.31,41
In recent years, the vast majority of human cases of sporotrichosis in Brazil have been caused by zoonotic transmission by cats and are due to S. brasiliensis. 38 Sporotrichosis has different clinical manifestations: cutaneous (localized or disseminated), lymphocutaneous, extracutaneous, or systemic.17,25,53 These manifestations can co-occur in domestic cats. 45 The extracutaneous form of feline sporotrichosis has been reported by histopathological examination of the lungs,23,49 lymph nodes, spleen, 49 and nasal region with mucosal and bone involvement. 20 Using only culture and isolation, the agent has been identified in the liver, spleen, kidneys, heart, 49 and bone. 14 However, its frequency and the most affected organs or tissues remain unclear. Furthermore, some studies have diagnosed visceral involvement based on clinical respiratory signs without macroscopic and microscopic evaluation of the internal organs. 45
The techniques used for diagnosing sporotrichosis include cytologic examination (because the fungal load in skin lesions in cats is often high), 52 histopathology with special stains, when necessary,5,52 and isolation of the etiologic agent in culture. 20 Polymerase chain reactions (PCRs),18,39 immunohistochemistry (IHC),19,52 and serology 3 have also been used. Diagnosing sporotrichosis can be difficult in cats with low fungal burdens and the cases may require the use of multiple techniques to identify the yeast.5,52
Considering the scarce knowledge of extracutaneous lesions of feline sporotrichosis, this study aimed to characterize the cutaneous and extracutaneous lesions in 102 cats suspected of sporotrichosis using macroscopic and microscopic examinations, and ancillary tests.
Materials and Methods
Ethical Approval
This study was part of a project approved by the Ethics Committee on Animal Use of the Universidade Federal of Minas Gerais (CEUA - 111/2017).
Geographical Area, Animals, and Samples
From March 2018 to March 2019, samples were obtained from 102 indoor/outdoor and stray cats with clinical suspicion of feline sporotrichosis. The cats were received or rescued by the Centro de Controle de Zoonoses of the municipality of Belo Horizonte (CCZ-BH). The cats were euthanized when treatment was impossible.
Clinical History, Postmortem Examination, and Sample Collection
The clinical history of the cats submitted for postmortem examination was obtained from the owners or veterinarians of CCZ-BH. All 102 euthanized cats were examined macroscopically with a detailed description of the lesions, and samples were collected for laboratory testing. The inclusion criterium for cats enrolled in this study was the presence of cutaneous ulcerations suspected of sporotrichosis in rescued cats.
Cytology
Samples for cytologic examination came from impression smears of ulcers. Cutaneous ulcers were dried with paper to remove excess exudate, while crusts were removed before pressing the glass slide lightly on the lesion’s surface. Slides were stained with Romanowsky’s rapid stain 32 and read with a standard light microscope. Confirmation of Sporothrix spp. was performed by visualization of 3 to 5 µm by 5 to 9 µm oval, rounded, or cigar-shaped yeast cells, with or without narrow-based budding, that were either extracellular or in the cytoplasm of macrophages, multinucleated giant cells, or neutrophils. 9 Yeast intensity was determined as absent, mild (up to 5 yeasts per 1000× field), moderate (6-50 yeasts per 1000× field), or marked (>50 yeasts 1000× field) 5 by examining at least 5 random non-overlapping fields (field area of 31.4 µm2).
Topography of the Skin Lesions
The ulcers and papules were counted by anatomical region to identify the most affected lesions (Supplemental Figure S1).
Macroscopic Examination of Lymph Nodes
All lymph nodes from each cat were evaluated for size and cut surface. For the intensity of the lymph node enlargement the following analysis was performed: mild for lymph nodes with enlargement less than twice the size of the normal lymph node, moderate when the lymph node enlargement was twice the normal size, and marked when the enlargement was greater than twice the normal size.
Histopathology
Samples from the skin, lung, heart, liver, spleen, kidneys, intestine, brain, bone marrow, and, superficial cervical, mandibular, popliteal, and mesenteric lymph nodes were collected for histological examination. In cats with macroscopic lesions in the nasal planum, rostral and medial nasal turbinates, conjunctiva, tongue, and testes, these organs were also collected. All tissues were fixed in 10% neutral buffered formalin for 48 hours, routinely processed, and stained with hematoxylin and eosin (HE) 28 for analysis under a standard light microscope. Morphological criteria for yeast identification were similar to those previously described. 5 For tissues where it was difficult to visualize the yeasts in HE sections, periodic acid-Schiff (PAS) reactions 28 were performed to better identify the yeasts. 5
Inflammation in the skin was classified as pyogranulomatous, granulomatous (with a predominance of epithelioid macrophages), histiocytic, or plasmacytic, according to the predominant cell types, based on subjective evaluation. To quantify the presence of the yeasts in the lesions, an average count of 10 consecutive high-power fields (HPF, ×400, field area of 196 µm2), was performed. The criteria used to classify the intensity of yeasts are provided in Supplemental Table S1. All organs collected for histologic examination were evaluated for Sporothrix associated lesions. Cytologic and histologic preparations were compared to verify the frequency of yeasts in the lesions.
Immunohistochemistry
Tissues with histologic lesions, but without yeasts in HE sections were subjected to IHC to confirm the agent in the lesion. IHC was performed similar as previously described. 33 A polyclonal rabbit anti-Sporothrix spp. antibody (Instituto Oswaldo Cruz, Fiocruz, Rio de Janeiro, RJ, Brazil) was used for IHC. Slides were incubated in phosphate-buffered saline with 0.05 g/mL of skimmed milk powder for 40 minute to block endogenous proteins. A 3% hydrogen peroxide solution in phosphate-buffered saline was used to block endogenous peroxidases. The tissue sections were incubated with anti-Sporothrix serum diluted in 1.5% bovine serum albumin in a humid chamber overnight (16 h) at 4°C. The yeasts were labeled with anti-Sporothrix serum at an optimal concentration of 1:8000. The secondary antibody was obtained from Envision Flex HRP (DAKO, Carpinteria, California, USA). 3, 3’-diaminobenzidine (DAB substrate System, Spring Bioscience) was used as the chromogen substrate, and counterstaining was performed with Mayer’s hematoxylin. For each run, sections from a sample confirmed positive by isolation of Sporothrix spp. in culture and containing yeast-like cells were included as a positive control. The primary antibody was replaced with phosphate-buffered saline as a negative control. The presence of yeasts consistent with Sporothrix spp. was evaluated using standard light microscopy at 400× magnification.
Polymerase Chain Reaction (PCR) and Sequencing
Frozen samples of cutaneous lesions from 15 cats and enlarged superficial cervical lymph nodes from 11 of these 15 cats were collected and subjected to DNA extraction and PCR. Tissues stored individually in microtubes and frozen at −20°C were subjected to DNA extraction according to previously described protocols 36 that were modified by Klassen et al. 24 Total DNA from each sample was amplified using primers specific for S brasiliensis according to previously described methods. 42 Subsequently, amplification of the partial calmodulin (CAL) gene was performed using previously described primer pairs. 39 DNA from S brasiliensis, previously identified by PCR and sequencing was used as the positive control. As a negative control, reactions were performed with all reagents except DNA template. The final product of each reaction was subjected to electrophoresis on 1.5% agarose gel containing ethidium bromide, along with a molecular weight marker of 100 bp (LowRanger100bp DNA Ladder Norgen®).
Three amplification products positive for CAL were purified using a PureLink PCR Purification Kit (Invitrogen), and the DNA was sequenced using an automated sequencer (ABIPrism 3500 Genetic Analyzer). The obtained chromatograms were visualized and assembled using the Gap4 program in the Staden software package (Staden, 1996). PCR products from 3 cats with differences in the lesion distribution and yeast quantity were selected for sequencing. Of these, the first cat had multiple skin lesions with numerous yeasts, which were also present in the extracutaneous tissues (conjunctiva, eye, nasal turbinates, lung, peripheral lymph nodes, and meninges). The second cat had skin lesions with a mild quantity of yeast without extracutaneous involvement, and the third cat had skin lesions in the head with numerous yeasts and involvement of the nasal turbinates.
Fungal Culture
Swabs were collected from skin lesions of all cats before euthanasia. These samples were seeded on Sabouraud agar containing chloramphenicol and cycloheximide, incubated at 28°C, and observed daily for up to 10 days. Hyphae from plates with growth suggestive of Sporothrix spp. were analyzed on a slide stained with lactophenol cotton blue for microscopic confirmation of the genus. 2 The visualization of septate hyphae and clustered thin conidia with the typical appearance of the inflorescence, named daisy plants, confirmed the isolation of the genus Sporothrix. 4
Statistical Analysis
To assess a relationship between cutaneous and extracutaneous lesions, the variables of “skin lesions,” “distribution of organ lesions,” “type of inflammatory infiltrate,” “intensity of Sporothrix spp. lesions,” “test positivity,” and “lymph node involvement related to skin lesions” were organized in a spreadsheet (Microsoft Office Excel 2016). The quantitative variables, means, standard deviations, and relative frequencies were calculated, and the distribution of frequencies for categorical variables was analyzed. Dunn’s test was used to assess differences between quantitative variables, number of organs, and lymph nodes affected, according to the intensity of yeast in the skin lesions. The kappa test was performed to analyze the diagnostic test agreement. 26 A proportion test was performed to verify the differences between the presented relative frequencies. Fisher’s exact test was used to evaluate for significant associations between the categorical variables. All analyses were performed according to Fávero and Belfiori 12 using Stata software version 15 (StataCorp, 2017, Stata Statistical Software: Release 15). Results were considered statistically significant at a P-value of < .05.
Results
Clinical History and Gross Findings
According to the history obtained from the owners or veterinarians of the cats submitted for postmortem examination, the clinical signs were characterized by progressive lethargy, decreased appetite, weight loss, and respiratory signs in cats with nasal mucosal lesions. Regarding treatment, 5 indoor cats (5.0%) were treated (no information on the type of medication used) and 59 outdoor cats (55.0%) were not treated. There was no information about treatment for the remaining 42 stray cats (40%).
Among the 102 cats examined, 75 (74%) were male (P < .01), and 27 (26.4%) were female. Intact males corresponded to 70% of the affected cats. More than 90% of the cats were underweight.
Gross skin lesions in all cats were papulonodular or ulcerative and hemorrhagic, draining purulent exudate, and some were crusted nodules. These lesions were single, multiple, or multifocal to coalescing. For all cats, these lesions were classified by location, distribution, size, and aspect. The site varied among the cats, and Fig. 1 demonstrates the frequency of skin lesions based on anatomic location. The most commonly affected anatomic regions were the thoracic limbs (63/102), nasal planum (53/102), ears (52/102), and face (60/102). Forty-eight cats had ulcers in the pelvic limb, 43 cats had periocular ulcers, and 27/102 cats had dorsal thoracolumbar ulcers. Lesions in the dorsal thoracolumbar region were statistically more frequent (P < .05) than those in the tail (18/102) and dorsal cervical (14/102) regions. Multiple skin lesions were more frequent (88/102; 86.3%; P < .001) than single lesions (14/102;13.7%). The presence of multiple ulcers was associated with the presence of extracutaneous lesions (P < .001).
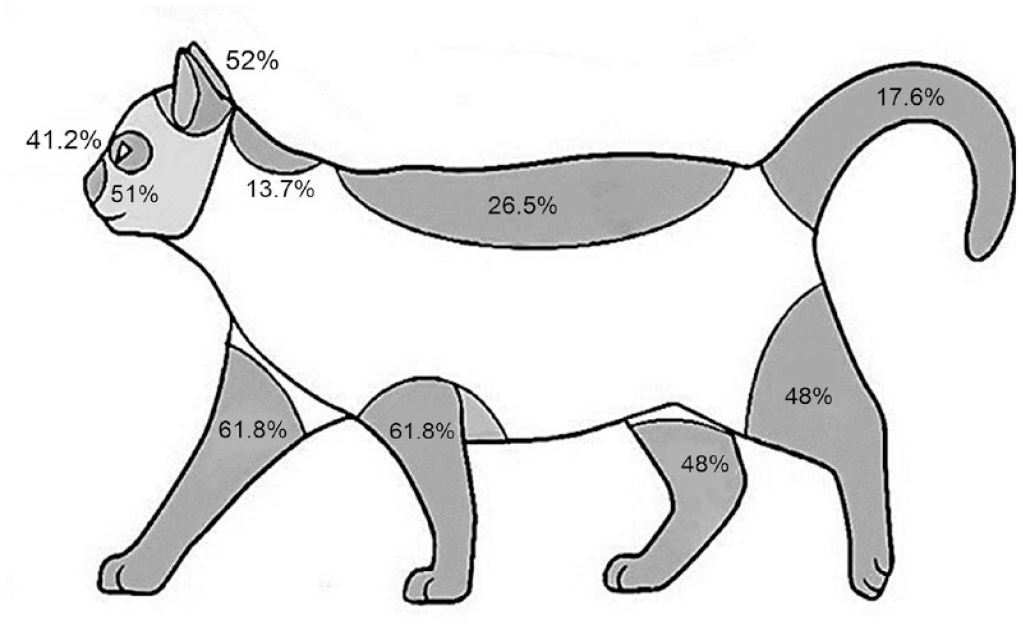
Schematic drawing of a cat showing the distribution of the skin lesions of the 102 cats with confirmed sporotrichosis. The relative frequency of lesions by anatomical region is indicated in percentages.
There was marked variation in the macroscopic characteristics of the cutaneous lesions, as aforementioned. Papules and focal non-ulcerated nodular lesions were found less frequently. Deep hemorrhagic ulcerations varied from 0.3 cm to 10 cm in diameter, and were focal, focally extensive (Fig. 2a) or multifocal to coalescing (Fig. 2b, c). In cats with focally extensive or multifocal distribution in the nose, pinnae, legs, and digits or tail, there was exposition of cartilage, muscles, and bone, respectively. Exudate associated with ulcers varied from hemorrhagic to purulent, or both, and some ulcers were elevated, dry and crusted. The underlying dermis and frequently the subcutaneous were thick, firm, and white. The edges of ulcers were raised and white in several cases.
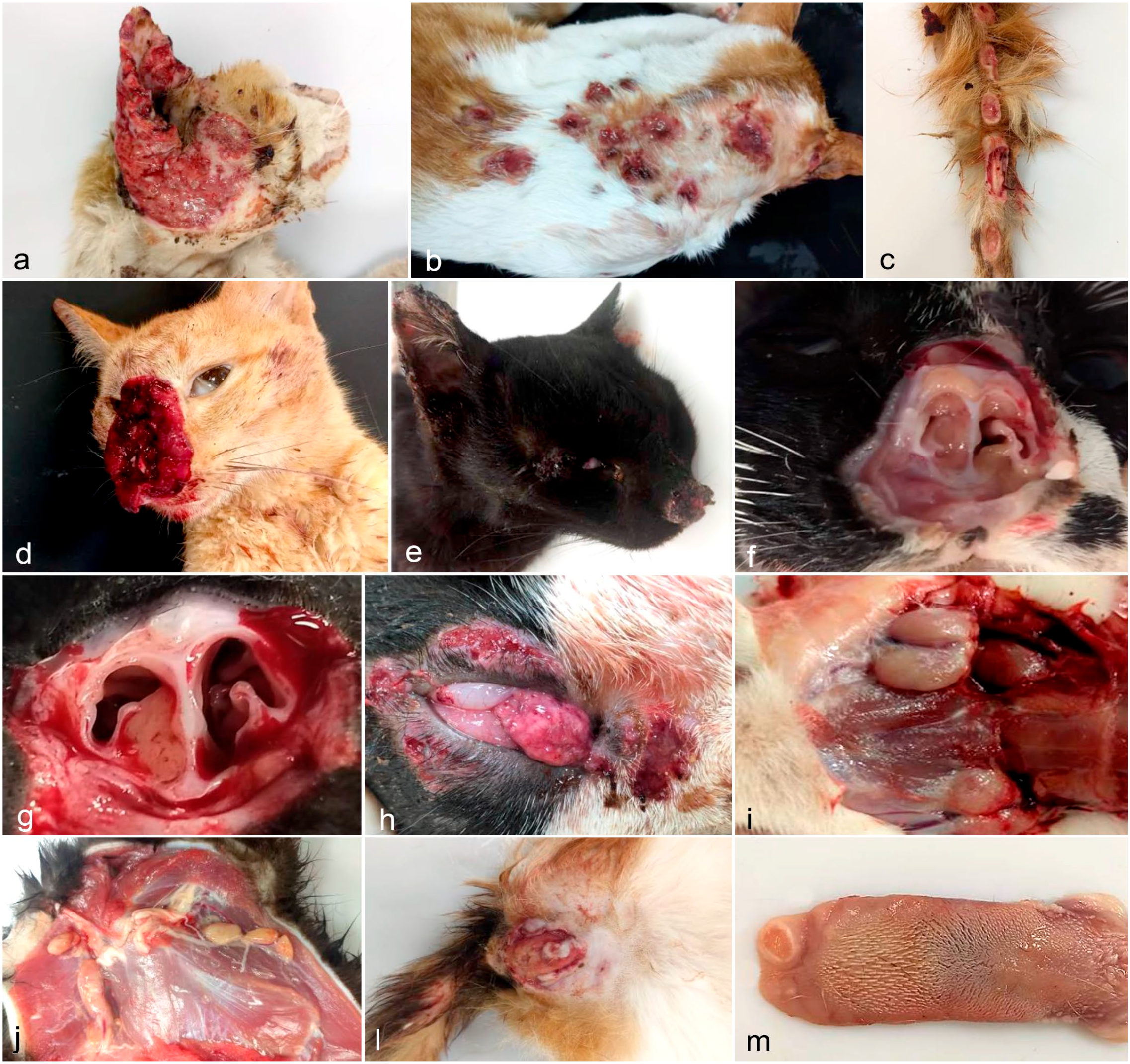
Macroscopic lesions in cats with sporotrichosis: (a) Extensive ulceration of the face and over the entire auricular surface draining hemorrhagic and purulent exudate, with raised and irregular borders, and thickened dermis and subcutis. (b) Multifocal serohemorrhagic ulcerated nodules on the dorsum of cervical and thoracic regions. (c) Multifocal ulcerative and granulomatous dermatitis with raised white edges and exposition of the tail bone. (d) Severe hemorrhagic ulcerated nasal planum with nasal cavity and septal exposure. (e) Enlarged and crusted nasal planum and nose. (f) The nasal turbinates of a cat with sporothrichosis. The right turbinate is thickened with a bright homogeneous yellow exudate. (g) The nasal cavity was filled with dense yellowish exudate and hemorrhage. (h) Thickened, hyperemic, and edematous conjunctiva and third eyelids, and hemorrhagic ulcers on the periorbital and facial skin. (i) The mandibular lymph nodes and (j) the cervical and axillary lymph nodes are markedly enlarged. (l) The skin of the scrotal region with ulceration and hyperemia. (m) In the dorsum of the tongue (apex), there is a circular ulcerative lesion with loss of the epithelium and white and raised edges.
In 29/102 cats (28%), there was an enlargement of the nasal bridge, that was variably ulcerated and hemorrhagic (Fig. 2d). In some cats, the lesion was mainly located in the nasal planum (Fig. 2e). The skin and turbinates were thickened (Fig. 2f), and purulent exudate was also frequent in the nasal cavity (Fig. 2g). In 9 cats (9%), the conjunctivae were thickened, edematous, and hyperemic (Fig. 2h). On cut section, the conjunctivae were edematous (7/9) or were white and firm (2/9). Generalized lymphadenomegaly was identified in 97/102 (95%) cats (Fig. 2i, j) and was the most frequent extracutaneous finding and ranged from mild (22/102; 21.6%), moderate (42/102; 41.2%), and marked (33/102; 32.4%), with no statistically significant difference between these categories. In 11 of the 75 male cats (15%) had focally extensive ulcers on the scrotum with marked purulent exudation that extended into the tunica vaginalis (Fig. 2l). In the oral cavity of 3 cats (3%), ulcers (Fig. 2m) were found at the apex of the tongue (especially on the dorsal surface). The lesions were white, firm, well-demarcated, and deeply depressed, reaching the muscular layer.
Fifty-nine of 102 (58%) cats had pulmonary lesions, and hyperemia and edema were grossly identified in 5 of these cats. Splenomegaly due to white pulp hyperplasia was present in 51/102 (50%) cats.
Cytology, Histopathology and IHC of the Skin
In 102 examined cats, Sporothrix infection was identified by at least one diagnostic technique. The yeast-like form of Sporothrix was identified in HE sections based on their oval to cigar-shaped yeast cell morphology with occasional buds in 70 (69%) cats. In the remaining 28 (27.4%) cats, Sporothrix yeasts were not detected using HE or other stains, and the confirmation was made using IHC in 26 (92.9%) of these cats. Two cats in which Sporothrix yeasts were not detected by cytology and histopathology, had positive results by IHC, and other 2 cats had positive results only for fungal culture.
Ninety cats were examined by cytology, of which 66 (64.7%) had identifiable yeasts. The yeasts were numerous and located extracellularly and in the cytoplasm of macrophages, accompanied by numerous plasma cells and neutrophils. The intensity of yeasts in the skin was greater (P < .01) in cats with multiple ulcers (46.5%) than in cats with single lesions (37.5%) (Fig. 3). In general, pyogranulomatous or granulomatous inflammation was present in the superficial and deep dermis, and in some cases, in the subcutis. Yeasts were intra-histiocytic or extracellular, and the intensity was variable in different classifications of the inflammatory infiltrates. The distribution of skin lesions according to the type of predominant inflammatory infiltrate and qualitative classification of Sporothrix (yeasts) was evaluated in 100 cats with yeasts visualized by histopathology and/or IHC (Supplemental Table S2). The relationship between the intensity of yeasts of Sporothrix and the predominant type of inflammatory infiltrate in skin lesions was compared and statistically evaluated (Supplemental Table S3).
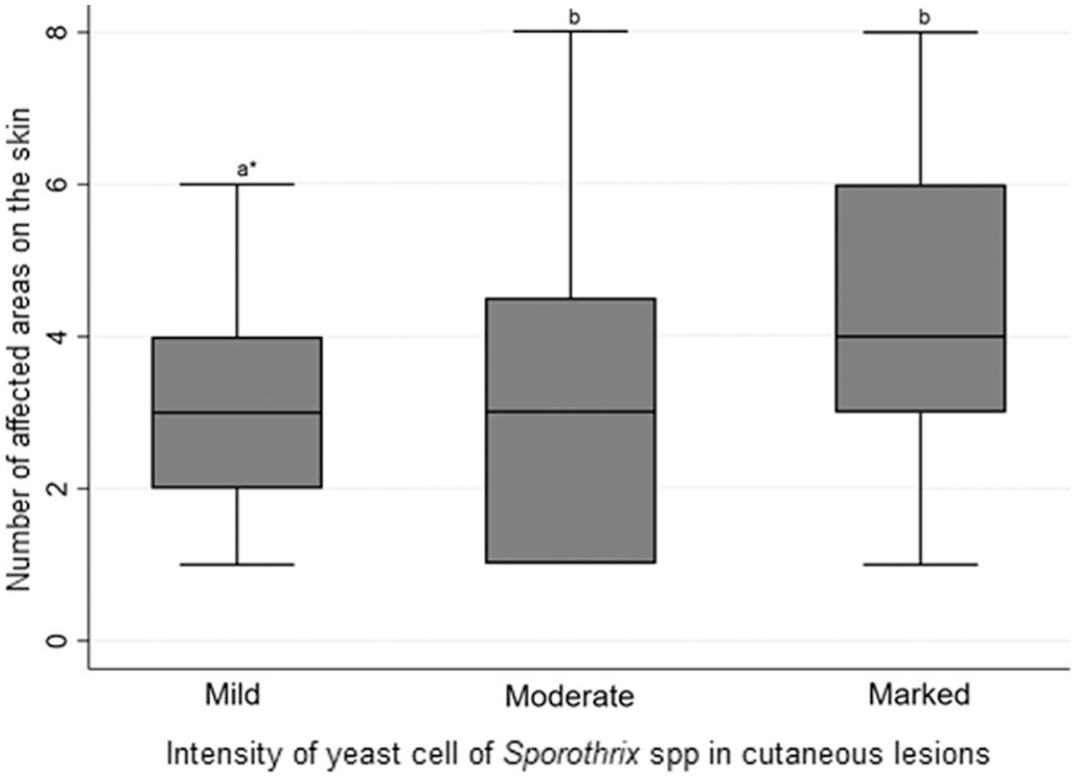
Graph showing the distribution of skin lesions and the intensity of Sporothrix spp. yeast in cutaneous lesions in cats. Dunn’s test, n=75. * Different lower-case letters indicate a statistically significant difference (P >.05) by Dunn’s test at a 5% significance level.
Of the 61 cats with pyogranulomatous (n = 50) or granulomatous (n = 11) inflammation, only 14 (22.9%) of the cats had well-formed granulomas. The intensity of the pyogranulomatous infiltrate was proportional to the intensity of the yeast in the skin lesions (P < .05). No significant association was observed between the intensity of Sporothrix yeasts and well-formed granuloma (Supplemental Table S4). However, there was a significant (P = .05) association between yeast and epithelioid macrophage intensity. When there was a mild number of yeasts, the frequency of epithelioid macrophages was 9 times higher (37.4%) than the frequency of other inflammatory cells (4.0%) (Supplemental Table S5).
Frequency, Histopathology and IHC of Extracutaneous Lesions
The presence of extracutaneous lesions was correlated with the intensity of yeast in the skin (P < .001). The location of extracutaneous lesions were as follows, in descending order of frequency: nasal turbinates, lymph nodes, lung, brain, tunica albuginea, and tongue. A summary of the histopathology and IHC results in the different organs is shown in Table 1.
Results of organ positivity by hematoxylin and eosin (HE) staining and immunohistochemistry (performed only on organs with lesions that were negative in HE) in 102 cats diagnosed with sporotrichosis from Belo Horizonte, Brazil (March 2018 to March 2019).
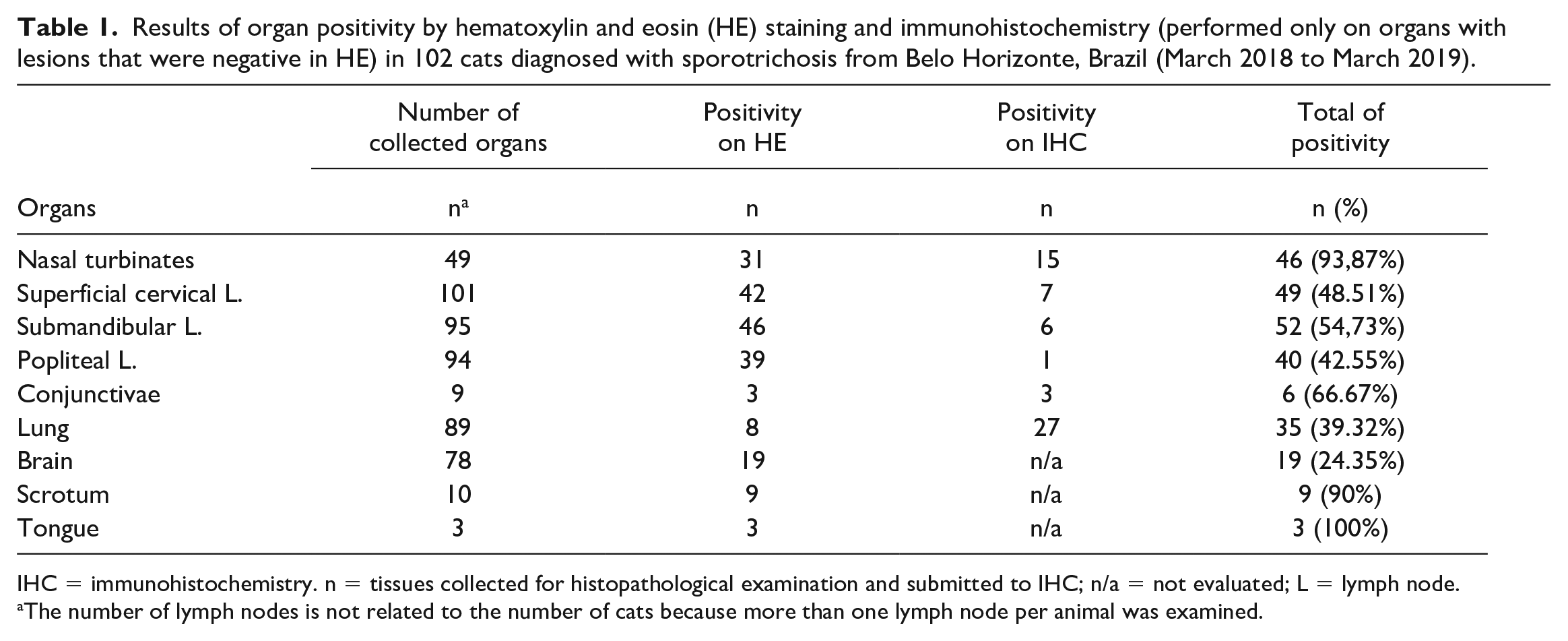
IHC = immunohistochemistry. n = tissues collected for histopathological examination and submitted to IHC; n/a = not evaluated; L = lymph node.
The number of lymph nodes is not related to the number of cats because more than one lymph node per animal was examined.
Of the 49 cats with nasal turbinates examined, there were lesions in 46 (80.3%) cats, and 31 (67.4%) cats contained yeasts that were visible in HE. The most frequent lesions were bilateral or unilateral thickening of the nasal turbinates and nasal septum by inflammatory cells in the mucosa and submucosa. In these cats, rhinitis, and/or rhinosinusitis was plasmacytic and histiocytic (17/46, 37.0%), pyogranulomatous (14/46, 30.4%), granulomatous (6/46, 13.0%), predominantly histiocytic (5/46, 10.9%), and histiocytic and lymphoplasmacytic (4/46, 8.7%). Of the 15 nasal turbinates that lacked visible yeast-like forms of Sporothrix in HE sections, positive immunolabeling (Fig. 4a, b) was obtained.
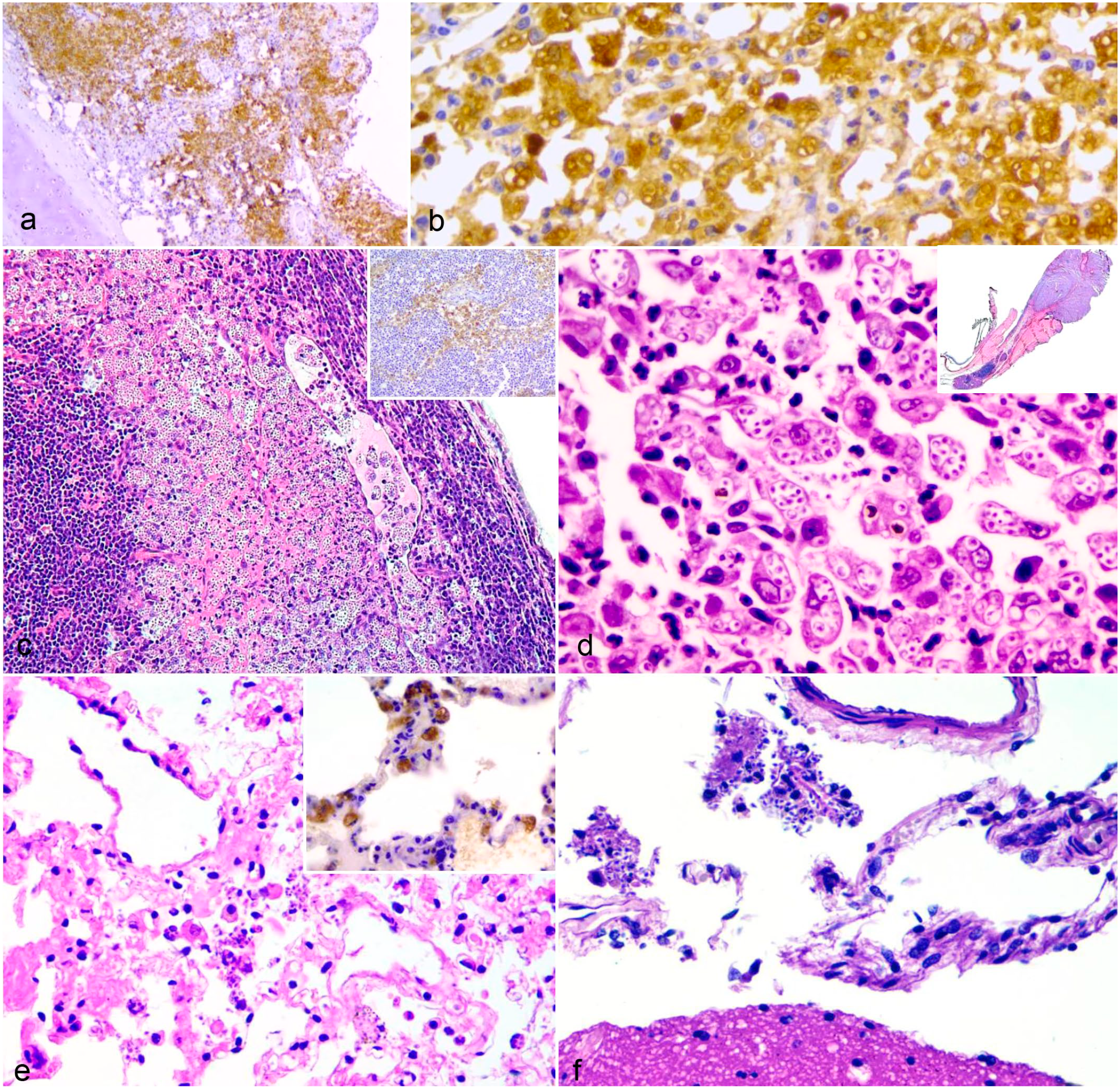
Histologic lesions of sporotrichosis in cats. (a and b) Nasal turbinate with lamina propria thickened by intense infiltration of epithelioid macrophages containing strongly positive (brown) yeast of Sporothrix spp. by immunohistochemistry. DAB with hematoxylin counterstain. (c) Lymph node with a subcapsular lymphatic sinus dilated by numerous macrophages loaded with yeasts, extended into the follicular region. Hematoxylin and eosin (HE). Inset: Medullary region of lymph node immunolabeled for Sporothrix spp. DAB with hematoxylin counterstain. (d) Granulomatous blepharitis and conjunctivitis associated with numerous intrahistiocytic Sporothrix spp. yeast. Inset: marked thickening of the third eyelid due to granulomatous inflammation. HE. (e) Lung with necrosis, fibrin exudate, and macrophages with intracytoplasmic and extracellular yeasts. HE. Inset: intrahistiocytic yeasts immunolabeled for Sporothrix spp. DAB with hematoxylin counterstain. (f) Brain. Macrophages with cytoplasmic yeasts in the cytoplasm in the subarachnoid space. HE.
Fifty-six of 100 cats (56%) had yeast in at least one of the peripheral lymph nodes. The positivity frequencies for yeast in the lymph nodes examined by HE were 50.5% (46/91) in the mandibular lymph nodes, 48.51% (49/101) in the superficial cervical, and 43.3% (39/90) in the popliteal lymph nodes. Yeasts were not visualized in the mesenteric lymph nodes. In 120 of 276 (43.5%) total affected lymph nodes in all examined cats, there were macrophages with intracytoplasmic yeasts in the subcapsular and medullary sinuses (Fig. 4c), and in some cases (6/276), also in the center of the lymphoid follicles. In 30 peripheral lymph nodes (7 cervical, 12 mandibular, and 11 popliteal), there was pyogranulomatous lymphadenitis with or without lytic necrosis, and moderate to marked plasmacytosis.
Out of 30 lymph nodes with granulomatous lesions, no yeast-like forms of Sporothrix were observed in 8 (2 mandibular and 6 popliteal). There was a significant association (P = .05) between lymph nodes with yeasts (mandibular, superficial cervical, and popliteal) and cutaneous sporotrichosis lesions. The number of lymph nodes with sporotrichosis lesions was higher in cats with multiple skin lesions (P < .001).
Lymph nodes with granulomatous lymphadenitis and/or predominantly sinusoidal histiocytic lymphadenitis without visible yeast in HE sections were also assessed by IHC (43/87). Of these, 23/43 (53.49%) were positive for yeast-like forms of Sporothrix spp. in mild quantities.
Lymphocytic and histioplasmacytic conjunctivitis was diagnosed in 9 cats (9/100). Multifocal epithelial necrosis and variable numbers of macrophages with intracytoplasmic yeast-like forms of Sporothrix were found. In 1 cat, there was also adenitis of the lacrimal gland in the left eye and marked multifocal histiocytic blepharitis of the third eyelid with numerous yeasts (Fig. 4d). Macrophages with intracytoplasmic yeasts were present in the trabecular meshwork of the eye. In 6 (6/9) conjunctivae with no visible yeasts in HE sections, it was possible to find a mild number of yeasts in 3 cats by IHC.
In the lungs of 8/85 cats, yeast-like forms of Sporothrix were identified. Infected macrophages were visualized within alveoli or interstitium in HE sections (Fig. 4e). In 3 cats, there was increased cellularity in the pulmonary interstitium due to infiltration by plasma cells, neutrophils, and macrophages. Bronchial or bronchiolar involvement was not identified in any case. Pneumonia was more severe in 1 cat that had been treated with corticosteroids for several days before death. The lungs of 33 cats that had yeast-like forms of Sporothrix in the skin, turbinates, and lymph nodes were subjected to IHC. The yeasts were identified in 27 (82%) lungs (Fig. 4e inset).
Sporothrix was found in the meninges of 19/74 (25.67%) cats using HE. Macrophages with intracytoplasmic yeasts, consistent with histiocytic meningitis, were multifocally distributed in the subarachnoid space (Fig. 4f). Hyperemia and leukocyte margination in meningeal vessels were also present. No lesions were identified in the neuroparenchyma.
In the scrotum of 9 cats (9/100), there were multifocal to coalescing ulcers, and pyogranulomatous and necrotizing periorchitis associated with yeast-like forms of Sporothrix. No yeast or inflammatory infiltrates was identified in the seminiferous tubules or epididymis.
On the tongue of 3 cats (3/100), there were multifocal ulcers, especially at the apex. Beneath the ulcers, there were cellular aggregates predominantly composed of yeast-filled macrophages, plasma cells, and epithelioid macrophages associated with tissue necrosis that extended into the muscular fibers (ulcerative and granulomatous glossitis).
No yeast-like forms of Sporothrix or lesions were identified in the liver (n = 95), spleen (n = 95), kidney (n = 95), intestine (n = 95), heart (n = 89), or bone marrow (n = 35).
Comparison of Diagnostic Assays and Molecular Analysis
The IHC results of the skin lesions were compared to those of other tests performed on the cutaneous lesions (Supplemental Table S6). The comparative statistical evaluation of the tests showed concordance between culture and IHC and between histopathology and cytology (Supplemental Table S7). However, some cases were negative on the fungal culture and positive on histopathology and/or IHC (n = 4), or positive on the fungal culture and negative on cytology, histopathology and IHC (n = 2). Skin swabs from 74 cats were tested by fungal culture and 62 samples resulted positive for Sporothrix spp.
Skin from 15 cats were evaluated by PCR, and 7 samples were amplified for CAL gene in the specific reaction for S brasiliensis. Viable DNA for sequencing was obtained from 3 lymph nodes, and the sequenced CAL amplicons showed 100% nucleotide identity with S brasiliensis when aligned with other reference sequences from GenBank. The sequences are available in GenBank under accession numbers ON010665 (Cat 1), ON010666 (Cat 2), and ON010667 (Cat 3).
Discussion
Concurrent cutaneous and extracutaneous lesions associated with detecting Sporothrix yeasts were conspicuous findings in most cats in this study. The confirmatory results demonstrated the importance of using a combination of techniques for the definitive diagnosis of Sporothrix infection. Furthermore, the study showed the capacity of the yeasts to reach the internal organs.
The disseminated cutaneous form was the most frequent, and the regions of the skin most frequently affected were the head, nasal planum, and periocular regions, which is consistent with a previous report, 47 in addition to the thoracic and pelvic limbs and tail.2,21,47 These sites are the most commonly affected because they are more exposed during territorial disputes and fights, making traumatic inoculation possible. 43 Traumatic inoculation can occur through scratching because the retractability of the nails can retain the fungus. 11 Also, inoculation can occur by biting and self-cleaning (licking) because cats can contaminate the oral cavity with the fungus present in the exudate of ulcerative lesions.4,44 In some cats, it was possible to identify sites with bite wounds, especially on the back and with multiple distributions in the body, including the scrotum, raising the possibility of self-spread, as previously described.21,34,47
Regarding the inflammatory characteristics of cutaneous lesions, the minimal number of yeasts compared to the intensity of the epithelioid macrophage infiltration found in the cats of this study may be due to the immune response, as therapy has not been reported for these cats. Other studies have suggested that the granulomatous response is related to more efficient disease control.5,10,34
Similar to this study, multiple cutaneous ulcers in cats were associated with high number of Sporothrix yeasts in a previous study. 34 Additionally in this study, a significant association was observed between the presence of multiple cutaneous ulcers and visceral lesions. The predominance of cats with multiple lesions has also been reported in previous studies; 6 however, no studies on the correlation between the frequency of cutaneous lesions and visceral involvement have been found.
Among the extracutaneous lesions of sporotrichosis, rhinitis or rhinosinusitis were frequent findings. Sporotrichosis lesions in the turbinates may occur due to drainage from cutaneous lesions on the face and nasal planum. 37 Nevertheless, sporotrichosis of turbinates can occur without ulcerative lesions on the nasal surface and adjacent areas, as observed occasionally in the present investigation. Inhalation of conidia, as described in humans, 7 may be an important form of infection for developing nasal turbinate lesions in cats.
Most lymphoid organs were markedly hyperplastic (generalized lymphadenomegaly and splenomegaly), possibly due to intense antigenic stimulation by Sporothrix infection. However, only peripheral lymph nodes were affected by yeast-associated inflammation. Lymph nodes were the most frequent site of extracutaneous infections (52.8%) in this study. These cats had at least one of the peripheral lymph nodes with yeasts in the lymphatic sinuses, indicating the importance of lymphatic drainage in the pathogenesis of sporotrichosis, either in the establishment of the disease, causing a higher frequency of the lymphocutaneous form, or in its resolution, since lymphatic circulation is responsible for the removal of cellular debris and pathogens. 16 Granulomatous lymphadenitis, with a predominance of epithelioid macrophages, often with lower yeast burden, was also a frequent finding. The predominance of epithelioid macrophages and the reduction in yeasts may be related to cellular immunity.22,54 In sporotrichosis, CD4+ T cells produce cytokines that attract macrophages to the lesion and reduce the fungal burden.22,35
Interstitial pneumonia was seen in cats with high cutaneous fungal burdens and in several that had rhinitis/rhinosinusitis. Other studies in cats with similar histopathological findings in the lungs suggested that the route of infection in these cases was inhalation, similar to that observed for other fungi that reach the lung. 52 There is also the possibility of yeast migration through the hematogenous or lymphatic route, as observed in an experimental study in rats with intranasal inoculation of Sporotrhix schenckii yeasts. 51 Another experimental study involving subcutaneous inoculation of S schenckii in cats also identified yeast in their lungs. 23 Regarding the inhalation of spores (conidia), as reported in humans, this possibility should be considered because of the spreading cycle of the fungus and the permanence of spores in soil that is rich in organic matter. 8 Several cats in this study lived in regions with access to soil and plants.
Although there are reports on the presence of Sporothrix in the brain of cats, 23 information on this lesion and its frequency are scarce. Considering that no neurological signs were reported, and all cats were euthanized before the progression of the disease to death, there is a possibility that more marked lesions were not found due to the early progression of the visceral disease. In humans, acute meningitis and cerebrospinal fluid infection have been observed in patients co-infected with the human immunodeficiency virus and S brasiliensis. 15
In this study, pyogranulomatous and necrotizing periorchitis was diagnosed in 11 cats but without extension to the testicular parenchyma. There have been studies on the isolation of Sporothrix yeasts from the testicles of cats. However, no lesions have been reported in the testicular parenchyma.29,46
Glossitis associated with sporotrichosis was also a serious lesion and possibly occurred due to traumatic self-inoculation (fights and licks of the lesions). Positivity above 40% for isolation of Sporothrix spp. in the oral cavity has been reported in cats with sporotrichosis;30,48 however, a previous report of glossitis by Sporothrix spp. was not found.
The extracutaneous lesions of Sporothrix described above also suggest the possibility of other routes of infection and dissemination, such as the hematogenous route. Yeast isolation in peripheral blood has been documented in cats with disseminated skin lesions46,50 and cats with only localized cutaneous lesions. 46 Some cats in this study had skin sporotrichosis lesions on the head, and only popliteal lymph nodes were affected, suggesting a hematogenous route. Moreover, this study indicates that extracutaneous lesions in cats are more common than the existing literature reports. In vitro experimental studies with Sporothrix schenckii have revealed that the yeast form is more virulent than the mycelial form (conidea). The transmission of yeast may increase the incidence of more severe forms of the disease. 13 Considering the differences among species, S brasiliensis of feline origin showed the highest temperature tolerance and may be more adapted and virulent to cats. 40
The Sporothrix species identification performed in 3 cats with disseminated cutaneous form and internal organ involvement in this study suggests that S brasiliensis is the species present in the cutaneous and extracutaneous mycotic infections of these cats. S brasiliensis is associated with high virulence and disseminated cutaneous lesions in humans. 1
The different diagnostic tests (cytology, histopathology, IHC, and fungal culture) used for the cutaneous lesions of the cats in this study showed high efficacy. A previous study 52 also compared different diagnostic methods in which Grocott methenamine silver (GMS) staining showed the highest sensitivity. Nevertheless, when the fungal burden was lower in this study, better results were obtained with IHC than with the other tests. Importantly, 44.3% of the cats in this study had low fungal burden despite marked cutaneous lesions, requiring IHC or fungal culture for diagnosis. Almost all of the included cats had not received antifungal therapy. Thus, other factors (such as the host immune response) may be related to the control of yeast multiplication in cats.
In the densely populated urban areas in Brazil, the frequency of zoonotic sporotrichosis increased in the last decades and became a serious public health problem. 27 The major reason is the more frequent outbreaks in cats by S brasiliensis and the high risk of cat-to-human transmission. The expensive and long treatment period may discourage the owners’ enforcement of with the recommended protocols and the care practices. This situation is aggravated when sanitary conditions and public health infrastructure are unsuitable.40,41
Conclusions
This study used macroscopic and complementary examinations in 102 cats with suspected feline sporotrichosis and presented detailed results on the distribution of extracutaneous lesions, some of which have been rarely studied and documented in cats. The most common extracutaneous findings were granulomatous rhinitis/rhinosinusitis, granulomatous and histiocytic sinusoidal lymphadenitis, meningitis, pneumonia, periorchitis, glossitis, and conjunctivitis caused by S brasiliensis. Of these, meningitis and glossitis have not been previously reported in cats. Moreover, the extracutaneous form is frequent, which may be related to the high pathogenicity of this fungus.
Cytology and histopathology have been shown to be efficacious in diagnosing the disease. However, when the fungal load of the lesion is low, additional tests such as IHC, culture, and/or PCR are needed to avoid false-negative results.
Rapid and accurate diagnosis is important for the early establishing of therapeutic protocols, because human and feline sporotrichoses are treatable diseases. Public awareness and education about responsible animal keeping and the form of transmission in endemic areas are fundamental measures to prevent and control sporotrichosis and to limit the spread of this emerging disease.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231189448 – Supplemental material for Feline sporotrichosis: Characterization of cutaneous and extracutaneous lesions using different diagnostic methods
Supplemental material, sj-pdf-1-vet-10.1177_03009858231189448 for Feline sporotrichosis: Characterization of cutaneous and extracutaneous lesions using different diagnostic methods by Agna F. Santos, Maria I. Azevedo, Camila I. Amaral, Nikollye A. Grom, Fabricio Marinho, Camila S. F. de Oliveira, Danielle F. de M. Soares, Maria H. F. Morais, Silvana T. Brandão, Rodrigo C. Menezes and Roselene Ecco in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank all the veterinarians and technicians at the Zoonoses Control Centers for their collaboration and providing cats for this study and Prof. Dr. Claudio S. L. de Barros for critically reviewing this manuscript.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil (CAPES- Coordination of Improvement of Higher Education Personnel), Finance Code 001, and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-National Council for Scientific and Technological Development), Brazil.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
