Abstract
Infection with feline calicivirus (FCV) is a common cause of upper respiratory and oral disease in cats. FCV infection is rarely fatal, however, virulent, systemic strains of FCV (VS-FCV) that cause alopecia, cutaneous ulcers, subcutaneous edema, and high mortality in affected cats have recently been described. Seven cats with natural VS-FCV infection all had subcutaneous edema and ulceration of the oral cavity, with variable ulceration of the pinnae, pawpads, nares, and skin. Other lesions that were present in some affected cats included bronchointerstitial pneumonia, and pancreatic, hepatic, and splenic necrosis. Viral antigen was present within endothelial and epithelial cells in affected tissues as determined by immunohistochemical staining with a monoclonal antibody to FCV. Mature intranuclear and intracytoplasmic virions in necrotic epithelial cells were identified by transmission electron microscopy. VS-FCV infection causes epithelial cell cytolysis and systemic vascular compromise in susceptible cats, leading to cutaneous ulceration, severe edema, and high mortality.
Keywords
Caliciviruses are nonenveloped, positive-sense, and single-stranded RNA viruses and include RVHDV, European brown hare syndrome virus (EBHV), and vesicular exanthema of swine viruses. Feline calicivirus (FCV) is a common pathogen of cats. Multiple strains of FCV exist and infection can cause a variety of manifestations, including fever, rhinitis, pneumonia, and oral ulcers. 1,4,10 Although morbidity is high, mortality is usually low except in kittens as a consequence of pneumonia. In 1998 an outbreak of severe disease in cats caused by an unusually virulent strain of FCV was described and reproduced in SPF cats. 7 At least six different outbreaks of similar disease characterized by high mortality have since been recognized in the United States, but the lesions have not previously been described in detail. 5 .
In contrast to the relatively mild symptoms that characterize infection of cats with the usual field strains of FCV, infection with virulent systemic FCV (VS-FCV) is devastating, with mortality rates ranging between 33% and 60%. 5 Adult cats that were previously vaccinated against calicivirus infection were prominently affected in these outbreaks, and adult cats were at higher risk for severe disease and death as compared with kittens. In addition to ulceration and nasal or ocular discharge, signs characteristic of VS-FCV infection include profound fever, anorexia, marked subcutaneous edema (especially of the limbs and face), icterus, alopecia, crusting, and ulceration of the nose, lips, pinnae, and feet. The basis of enhanced pathogenicity of VS-FCV is not known. The gross and histologic lesions, antigen distribution, and ultrastructural characteristics of fatal VS-FCV infection in seven cats are described.
Materials and Methods
Animals and case inclusion criteria
Seven cats from two VS-FCV outbreaks are included in this study. The outbreaks during which these cats were affected involved between 5 to 54 animals in a single area and occurred for periods of 1 to 3 months. In each outbreak, identical strains of calicivirus were isolated from more than one cat; and all cats had consistent clinical signs and exposure history. All seven cases were confirmed by virus isolation and sequencing of a 235-nucleotide region of the gene encoding the FCV viral capsid protein. Necropsies in four cases (cat Nos. 1–4) were performed at the University of California at Davis. In cat Nos. 5–7, fixed tissues were provided by Antech Diagnostics and IDDEXX Veterinary Services. Random, individual clinical chemistry and hematology data were available in only three cases. Because the clinical pathologic findings were incomplete and haphazard with regard to exposure, they are not reported here.
Histology
All tissue samples were fixed in 10% neutral buffered formalin. Selected tissues were embedded in paraffin, sectioned at 5 µm, and mounted on positive-charged glass slides (Superfrost/plus, Fischer Scientific). Tissue sections were stained with hematoxylin and eosin (HE).
Immunohistochemistry
All formalin-fixed, paraffin-embedded feline samples were immunohistochemically stained using the monoclonal antibody anti-feline calici virus CV8-1A (c) provided by Custom Monoclonals Inc. Sections (5 µm) were mounted on positively charged slides, deparaffinized in xylene, and placed in two changes of 100% ethanol. Endogenous peroxidase activity was quenched with 0.3% hydrogen peroxide in methanol for 10 minutes followed by decreasing concentrations of ethanol and hydration in water. Antigen retrieval included a 7-minute room temperature incubation with proteinase K (DAKO S3020). Tissues were rinsed in two changes of phosphate-buffered saline (PBS) for 5 minutes each. The primary antibody was diluted in 10% horse serum to a final protein concentration of 0.064 mg/ml. The negative control used was mouse serum of the same isotype as the primary antibody diluted to the same protein concentration. The slides were incubated with 100 µl each of either the diluted monoclonal antibody or mouse immunoglobulin G for 1 hour at room temperature in a humidified chamber. After primary antibody incubation, the slides were rinsed with two changes for 5 minutes each of tris-buffered saline with Tween 20 (TBST) (DAKO S3306). The slides were labeled with DAKO anti-mouse Envision+ System labeled with HRP (DAKO K4001) for 30 minutes at room temperature followed by two rinses (5 minutes each) with TBST. The chromagen (AEC substrate DAKO K3464) was applied for 10 minutes before a final rinse and counterstaining with Mayer's hematoxylin solution (Sigma Mhs-16), blued in PBS for 20 seconds, and coverslipped with an aqueous mounting media crystal mount (Biomea Corp. M03) and allowed to dry on a hot plate for 20 minutes before the slides were permanently mounted with Shur/Mount (American Master Technologies).
Electron microscopy
For transmission electron microscopy (TEM), sections of skin and nares that were initially preserved in 10% neutral buffered formalin were transferred to half-strength–modified Karnovsky's fixative before two washes with 0.2 M sodium cacodylate and postfixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 6 After fixation, tissues were washed in 0.2 M sodium cacodylate and dehydrated through a graded ethanol series before infiltration and embedment in Spurr's epoxy resin. 12 Thick sections were cut, mounted on 150 mesh copper grids, stained with 6% methanolic uranyl acetate and Reynold's lead citrate, and examined in a LEO 905E (Zeiss) transmission electron microscope at 60 V accelerating voltage. 11 For direct electron microscopy (DEM), polyethylene glycol–precipitated virus grown in Crandel feline kidney cells was concentrated by centrifugation in a Beckman 50Ti fixed angle rotor at 220,000 × g for 45 minutes. The pellet was resuspended in distilled water. A 5 µl aliqout of resuspended pellet material was mixed with approximately 10 µl 2% phosphotungstate (pH 7.4) and placed onto a carbon stabilized formvar grid for visualization in a LEO 905E (Zeiss) transmission electron microscope at 60 kV.
Results
Gross
Ulcers were present on all VS-FCV–infected cats, although their extent and site varied markedly (Table 1). Within the oral cavity, the dorsum of the tongue was most severely and frequently affected, and numerous smaller ulcers were scattered on the hard palate and gingiva. One or more feet also were affected with lesions ranging from circumferential hyperemia at the haired/nonhaired junction to sloughing of entire footpads (Fig. 1). Ulcers were variably present in the nasal cavity, pinnae, and haired skin. Two cats had alopecia of their limbs and ventrum. All cats had marked subcutaneous edema that affected the face and limbs. The conjunctivae in all cats were red and swollen, with crusting discharge in the region of the medial canthi. All cats had pulmonary edema of variable severity, and four cats had blood-tinged pleural effusion. Multiple, small, discrete foci of peripancreatic and omental fat necrosis were present in three cats.
Signalment, case history, and pathology of seven cats diagnosed with virulent systemic feline caliciviral infection.
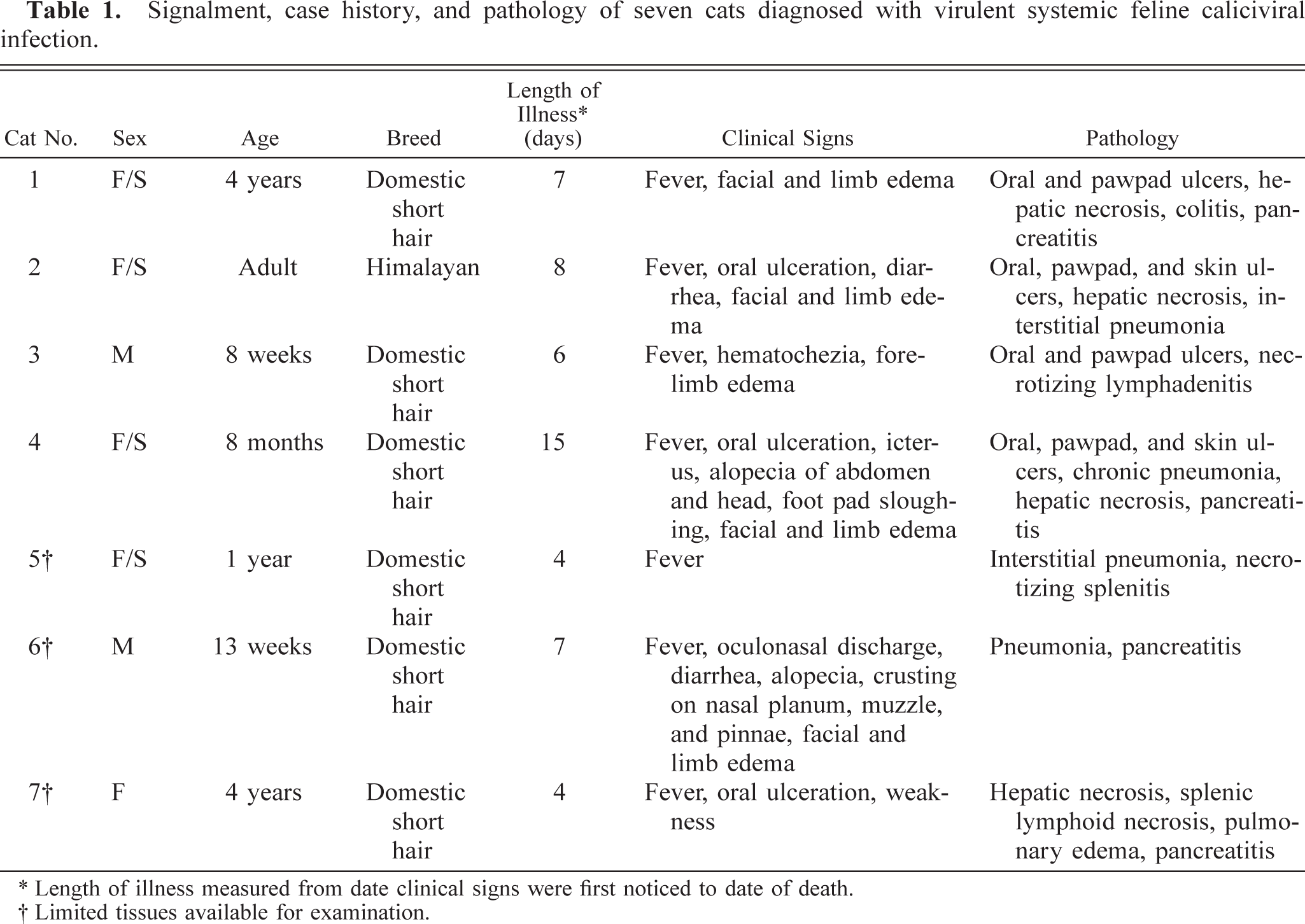
∗ Length of illness measured from date clinical signs were first noticed to date of death.
† Limited tissues available for examination.
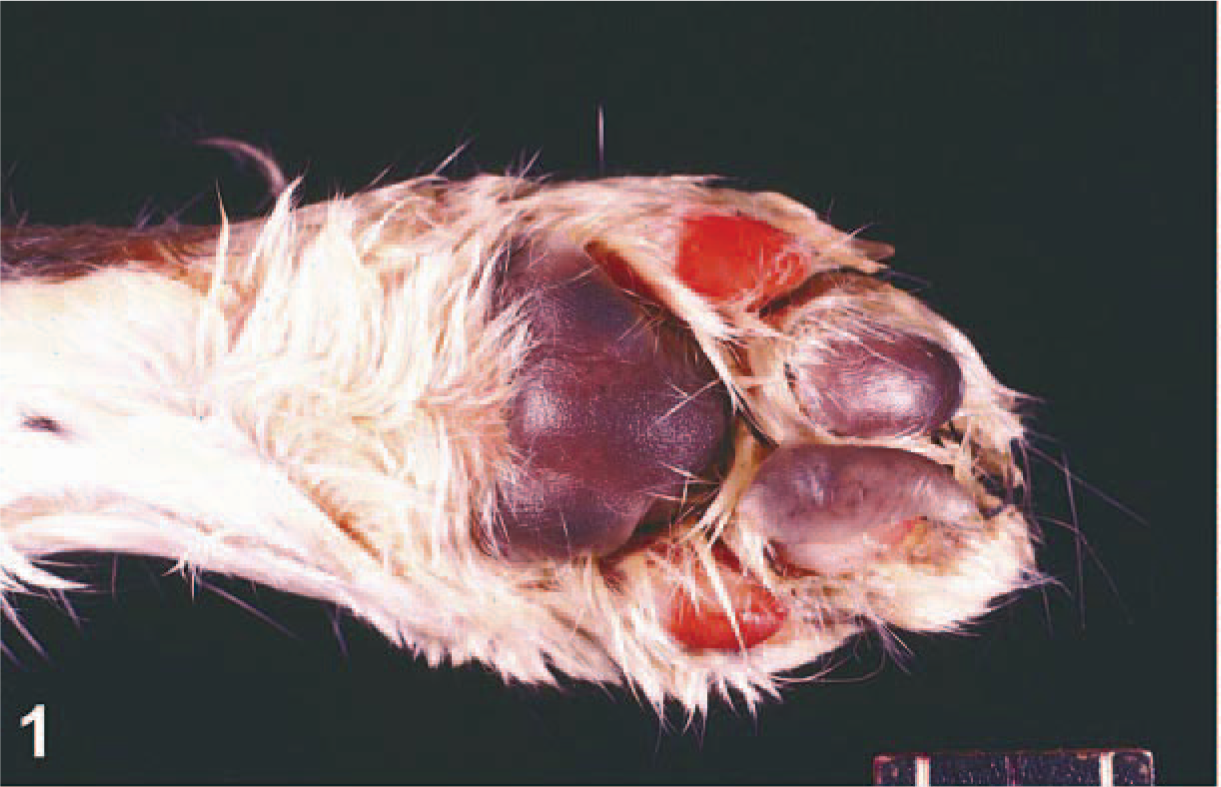
Paw; cat No. 4. The epithelial lining of the footpads of P2 and P5 have sloughed. There is peripheral ulceration at the pad-haired skin junction of P4. HE.
Histology
The most consistent histopathologic change was epithelial necrosis and ulceration. Ulcers in the nasal cavity, hard palate, tongue, pinnae, skin, and pawpads were similar. The initial lesion in haired skin was segmental epithelial necrosis of the stratum basale and stratum spinosum (Figs. 3, 6, 7). Follicular epithelium was also necrotic (Fig. 2). In early lesions of the pawpads, necrosis and neutrophils were most marked at the junction between haired and nonhaired skin (Fig. 3). In more chronic lesions, there was full thickness necrosis of epithelium, with ballooning degeneration in superficial layers and loss of distinct epithelial-subepithelial margin. Multiple microscopic ulcers were also present in regions where no gross evidence of ulceration was observed. Four of the seven VS-FCV–infected cats had bronchointerstitial pneumonia. In severely affected cats, the alveolar interstitium was expanded by type II pneumocyte hyperplasia, accumulation of leukocytes in alveolar capillaries, and microthrombi (Fig. 4). Alveoli were variably filled with a mixture of foamy histiocytes, cell debris, fibrin, and red blood cells. In acute cases, lung lesions were limited to increased accumulations of leukocytes within alveolar capillaries, regional alveolar edema, and scattered, individual necrotic epithelial cells within alveolar spaces. Hepatic changes ranged from mild disruption of the parenchyma with diffuse individualization of hepatocytes to extensive disruption of hepatocellular plates (Fig. 5) with individual cells and, in one case, centrilobular hepatic necrosis. Scattered accumulations of intrasinusoidal fibrin were present in most cases (inset, Fig. 5). Inflammation was limited to small clusters of intrasinusoidal neutrophils adjacent to necrotic foci. Four of the seven cats had multifocal, peracute, pancreatic necrosis with saponification of adjacent fat. Each cat had extensive splenic and lymphoid necrosis.
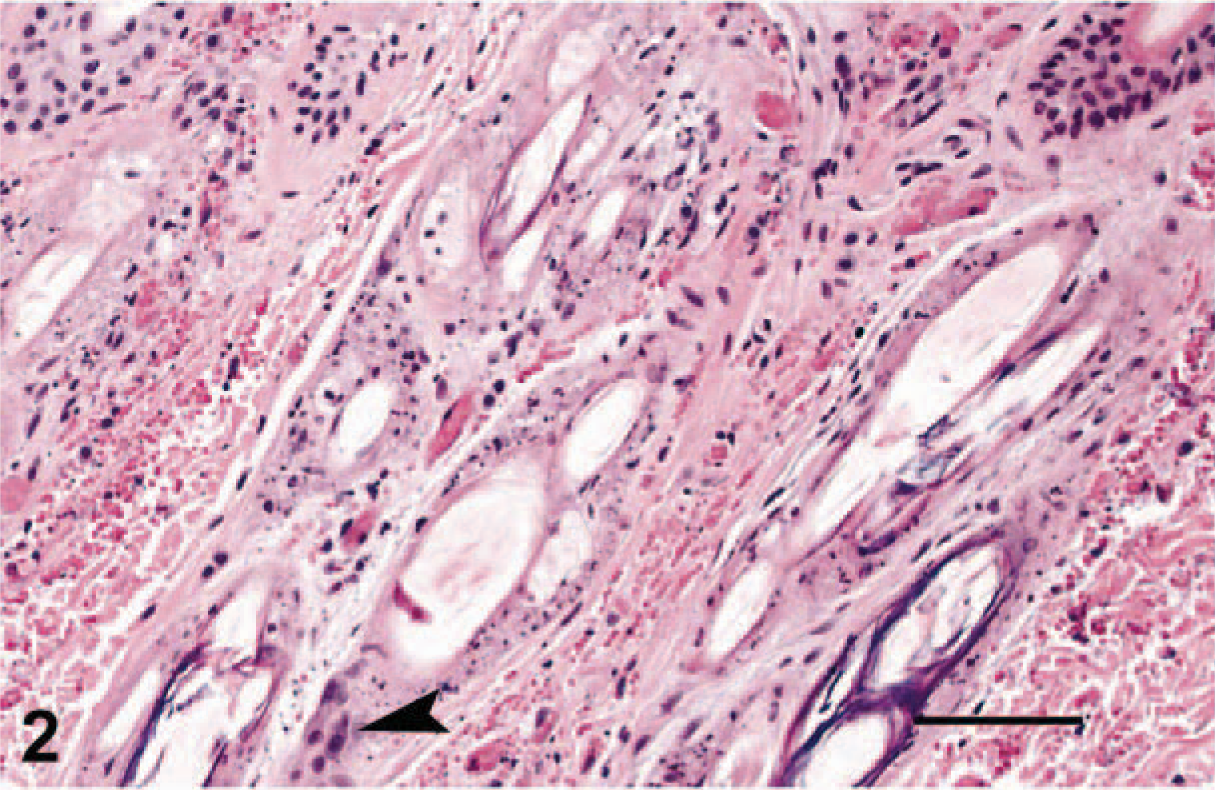
Skin; cat No. 2. Extensive necrosis of the follicular epithelium. Arrowhead indicates remnant viable epithelium. HE. Bar = 70 µm.
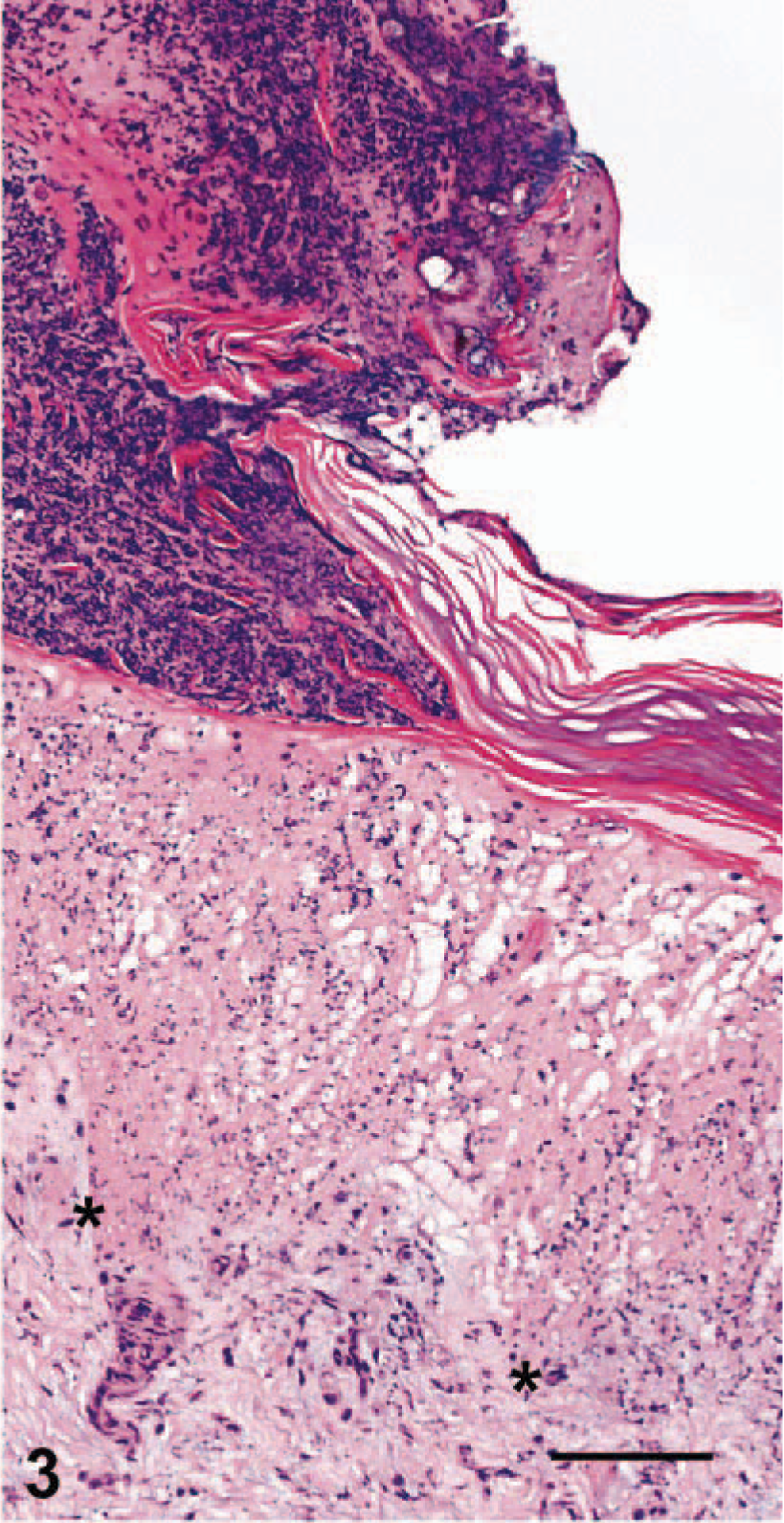
Pawpad, haired skin junction; cat No. 1. Extensive infiltration of neutrophils with accompanying karyorrhexis and karyolysis of epithelial cells at the junction between haired skin (left) and footpad (right). Asterisks indicate dermal-epidermal junction, which is obscured by necrosis of the epithelium. HE. Bar = 140 µm.
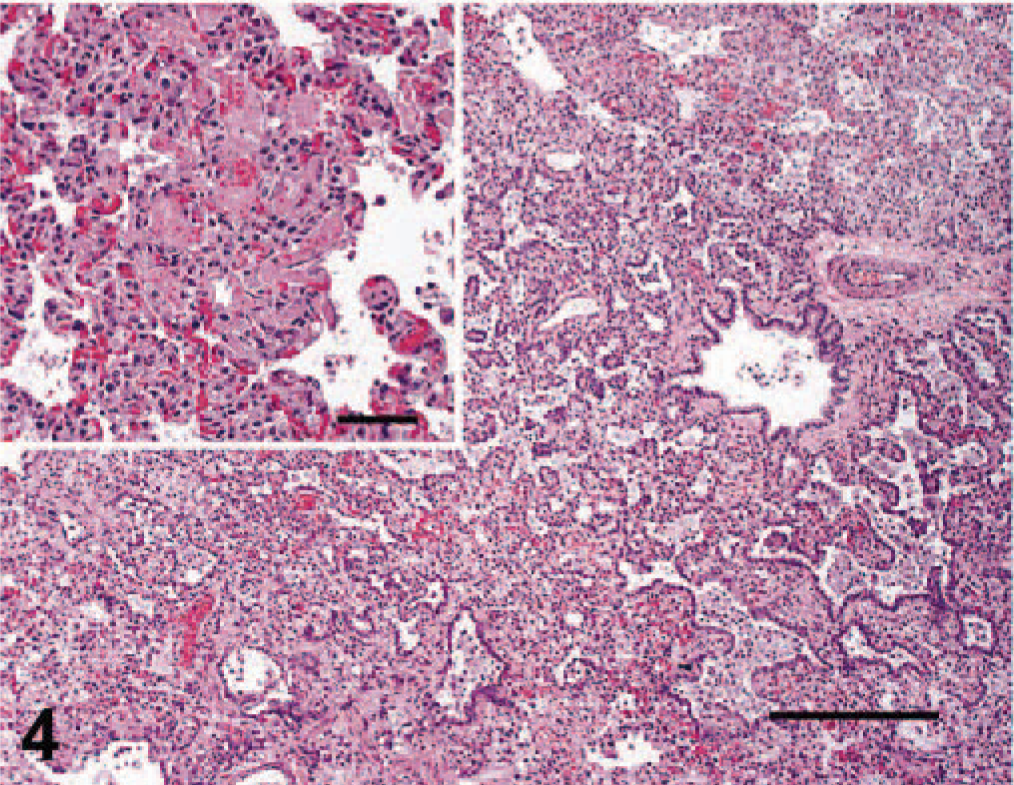
Lung; cat No. 4. Interstitial pneumonia with marked type II pneumocyte hyperplasia. HE. Bar = 92 µm. Inset: Fibrin deposits within alveolar capillaries. Bar = 45µm.
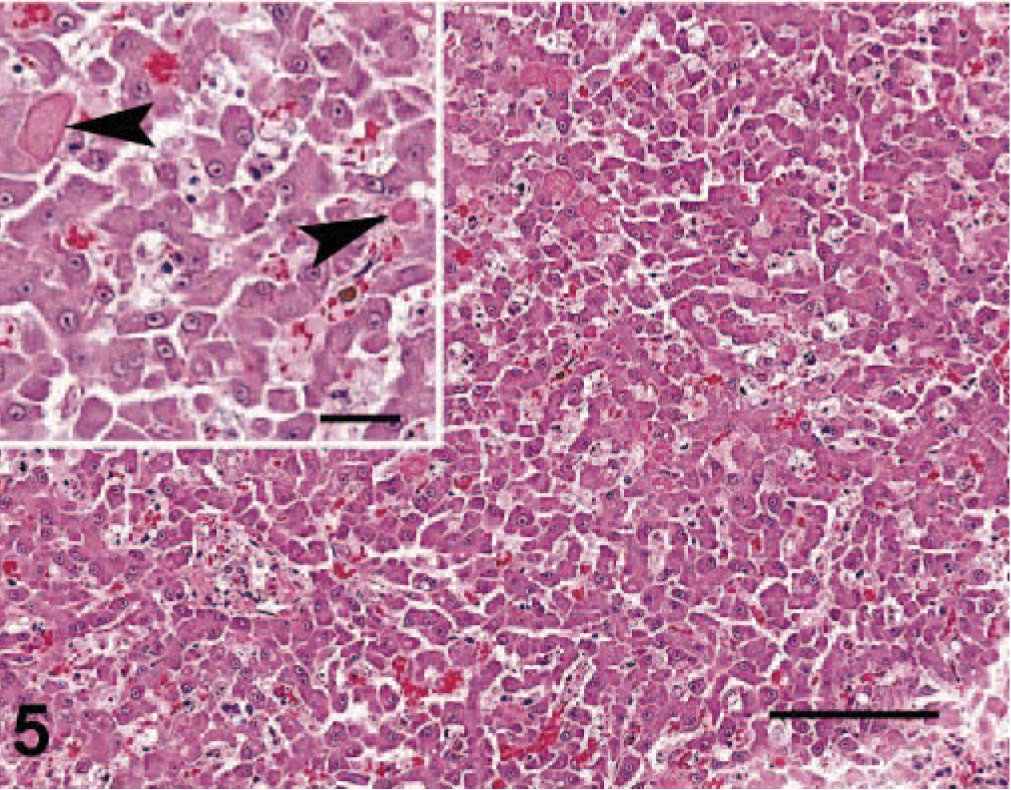
Liver; cat No. 4. There is individualization of hepatocytes, scattered hepatocyte necrosis, and fibrin deposits (inset, arrowheads). HE. Bar = 72 µm. Inset bar = 40 µm.
Skin; cat No. 3. Strong immunoreactivity of epithelial cells in an early lesion of epithelial necrosis. Envision HRP immunohistochemical method for feline calici antibody. Bar = 50 µm.
Skin; cat No. 2. Immunoreactivity is limited to segmental regions of necrotic epidermal (arrow) and follicular (inset) epithelium. Envision HRP immunohistochemical method for feline calici antibody. Bar = 72 µm.
Immunohistochemistry
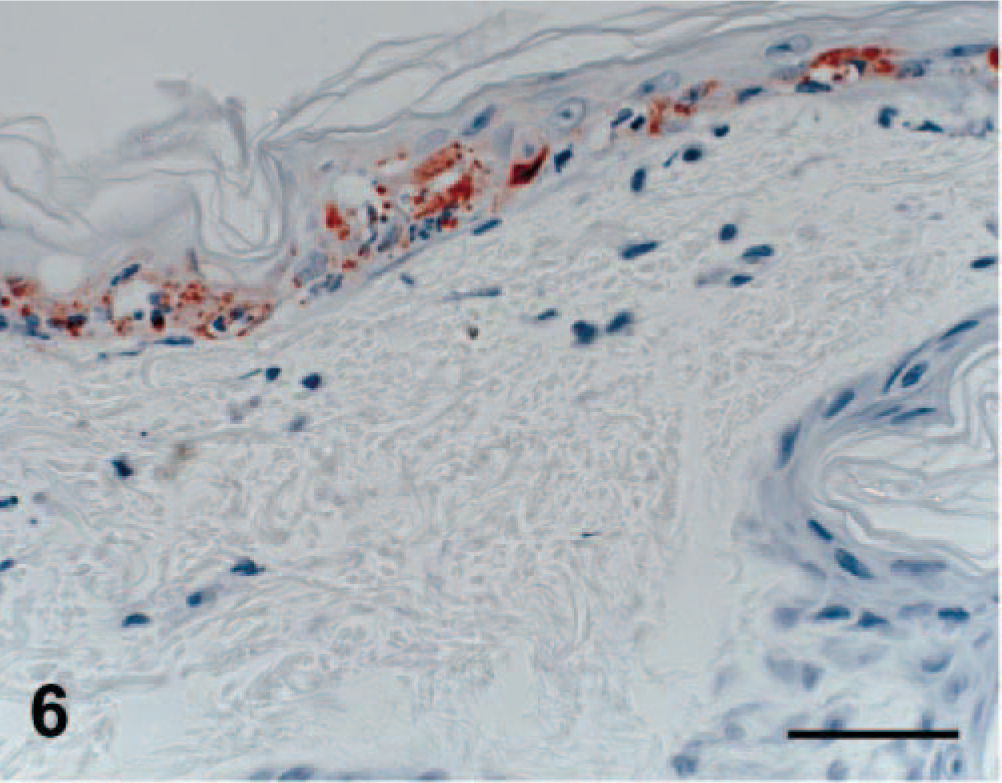
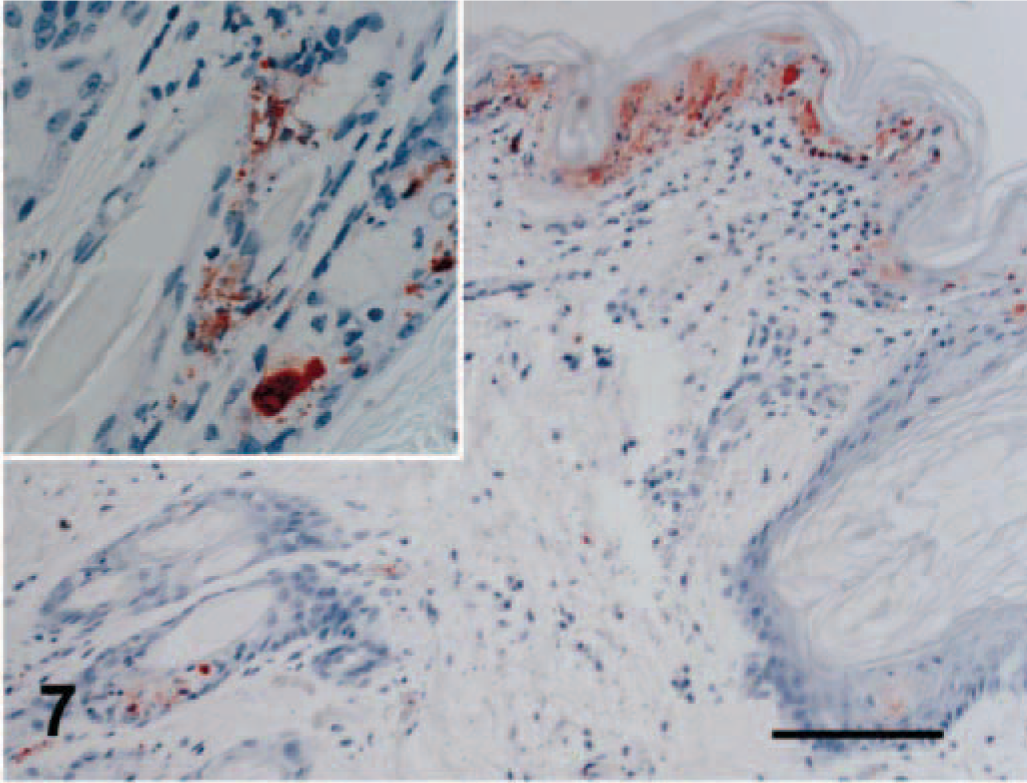
Viral antigen was detected in affected regions of skin, nasal mucosa, tongue, buccal mucosa, pinna, pawpad, lung, and pancreas. Staining intensity was proportional to the severity of the lesion. In early skin, mucosal, or pawpad lesions, antigen was limited to areas of individual or segmental epithelial necrosis (Fig. 6). In more chronic cases, FCV antibody reactivity included follicular epithelium (Fig. 7). Endothelial cells in small vessels of the dermis contained viral antigen (Fig. 8). Antigen was present in exocrine pancreatic cells within and bordering the regions of necrosis (Fig. 9). In minimally affected or unaffected lungs (by histologic evaluation), scattered individual cells within alveolar septae contained antigen. In more chronic lesions of bronchointerstitial pneumonia, viral antigen was limited to lining cells of small bronchioles and necrotic cells within the bronchiolar lumen. No antigen was detected in affected liver, spleen, or lymph nodes.
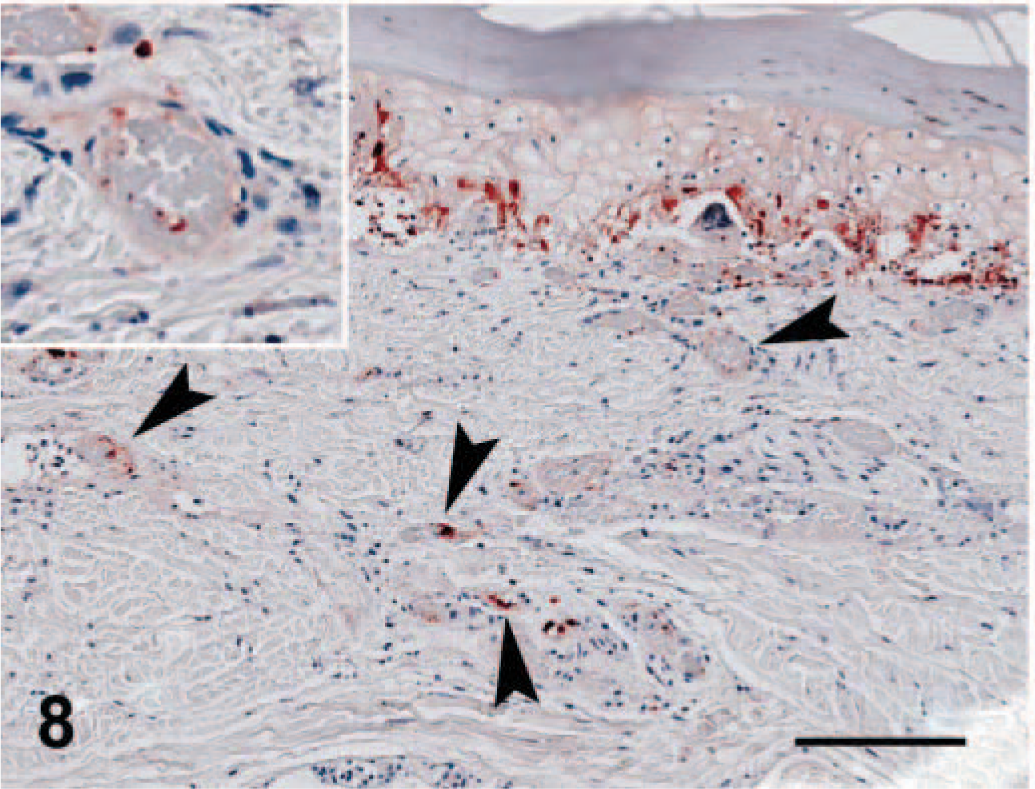
Pawpad; cat No. 3. In more severe lesions, reactivity is strongest in the basal layer; numerous scattered individual endothelial cells of the dermis strongly label with FCV antibody (arrowheads and inset). Envision HRP immunohistochemical method for feline calici antibody. Bar = 124 µm.
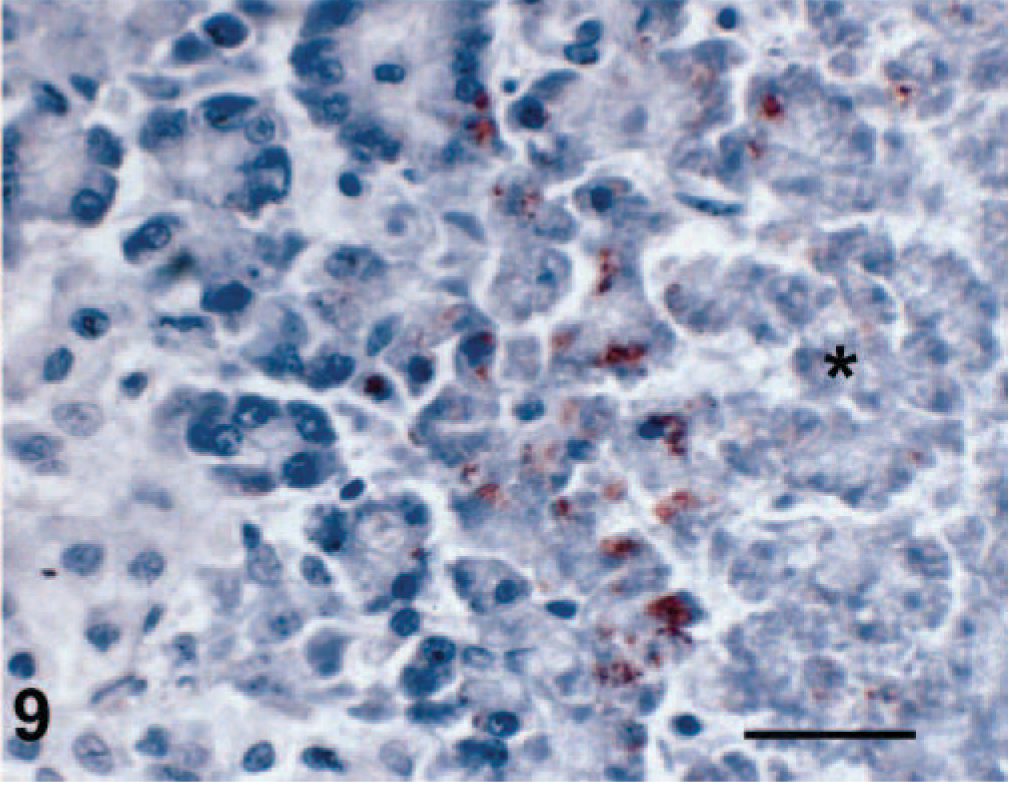
Pancreas; cat No. 4. Immunostaining of necrotic exocrine pancreatic acinar cells within and bordering a region of necrosis (asterisk). Envision HRP immunohistochemical method for feline calici antibody. Bar = 40 µm.
Electron microscopy
Virus particles of approximately 33.5 nm diameter, with scalloped borders and surface indentations that were typical of Caliciviridae, were identified by DEM on purified VS-FCV (Fig. 10). Multiple affected epithelial tissues (pawpad, nasal cavity, tongue) from two cases were examined (cat Nos. 1 and 3) by TEM. The VS-FCV within these lesions was identified by its consistent size (32–35 µm), central, electron-dense core 20 nm in diameter, and less electron-dense periphery. There was a substantial variation in the epithelial integrity between affected regions. In sections from pawpad, virus, sometimes in paracrystalline arrays, was detected in the superficial epithelium and associated with cells undergoing lysis or vacuolar degeneration (Figs. 11, 12). In both cases, mature virions were present within epithelial nuclei (Figs. 12, 13). In one case, paracrystalline arrayed virus was adjacent to protofilaments interpreted to be associated with viral assembly (Fig. 12). The liver of one VS-FCV–infected cat (cat No. 4) was examined by TEM. There was extensive disorganization of hepatic architecture, and most hepatocytes were necrotic with swollen mitochondria, fragmentation of nuclei, and disruption of plasma membranes. Scattered within the parenchyma were masses of fibrils with striations and a periodicity characteristic of fibrin.
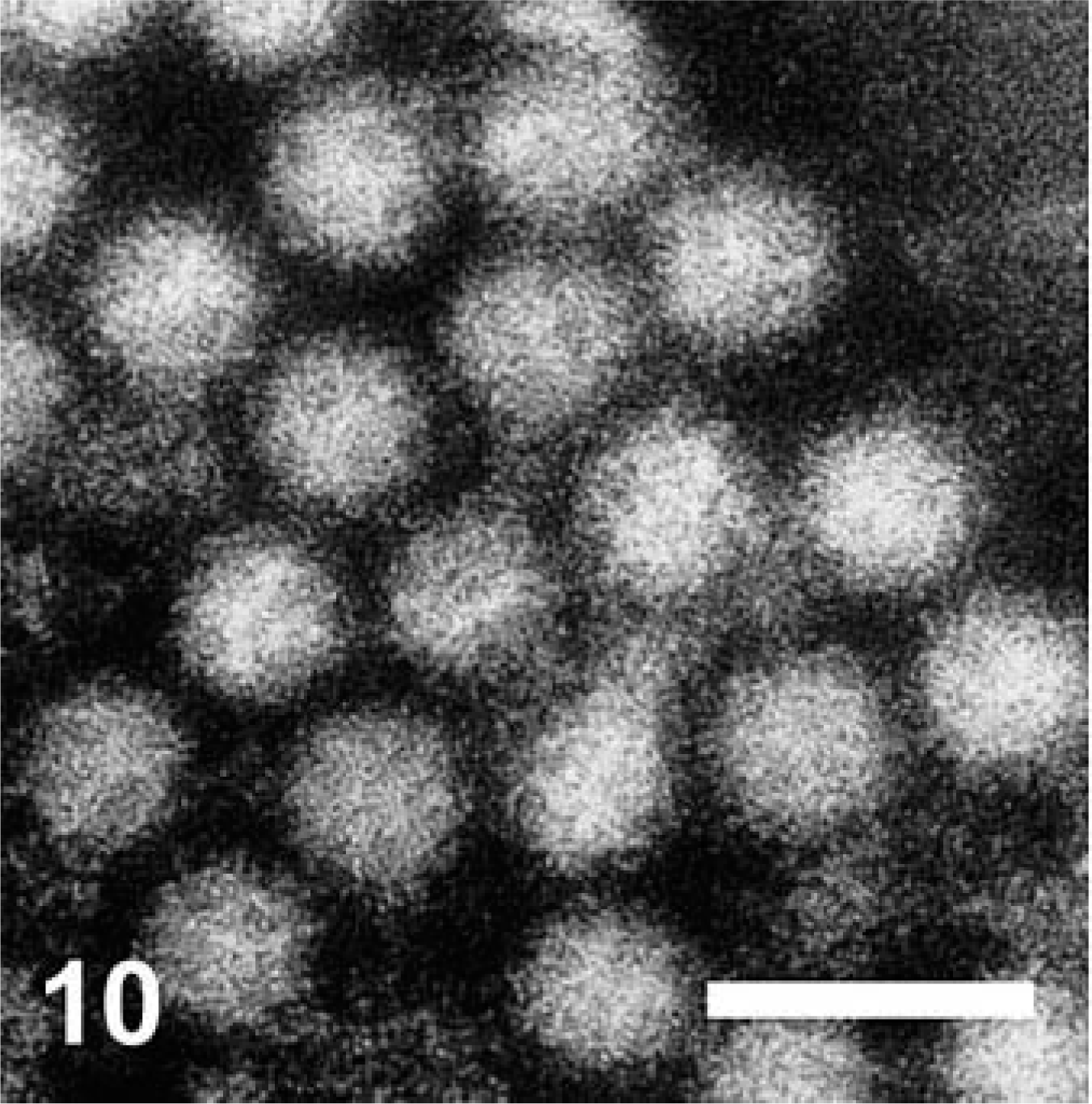
Cat No. 1. Purified VS-FCV. Particles are 33.5 nm in diameter and have the scalloped borders and surface indentations typical of Caliciviridae. Direct electron micrograph. Bar = 64 nm.
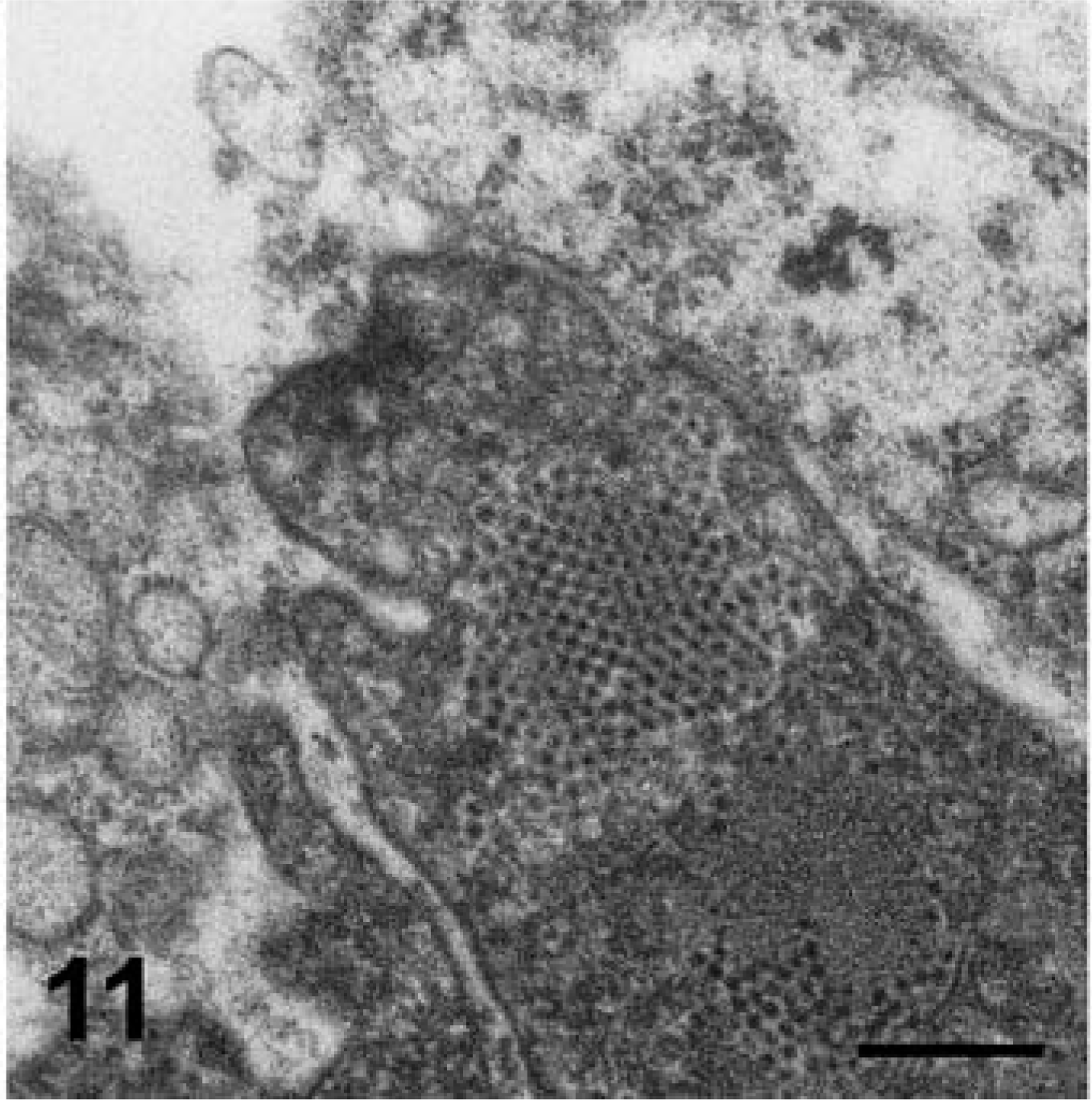
Skin; cat No. 1. A membrane-bound fragment contains paracrystalline arrays of virions. Transmission electron micrograph. Bar = 332 nm.
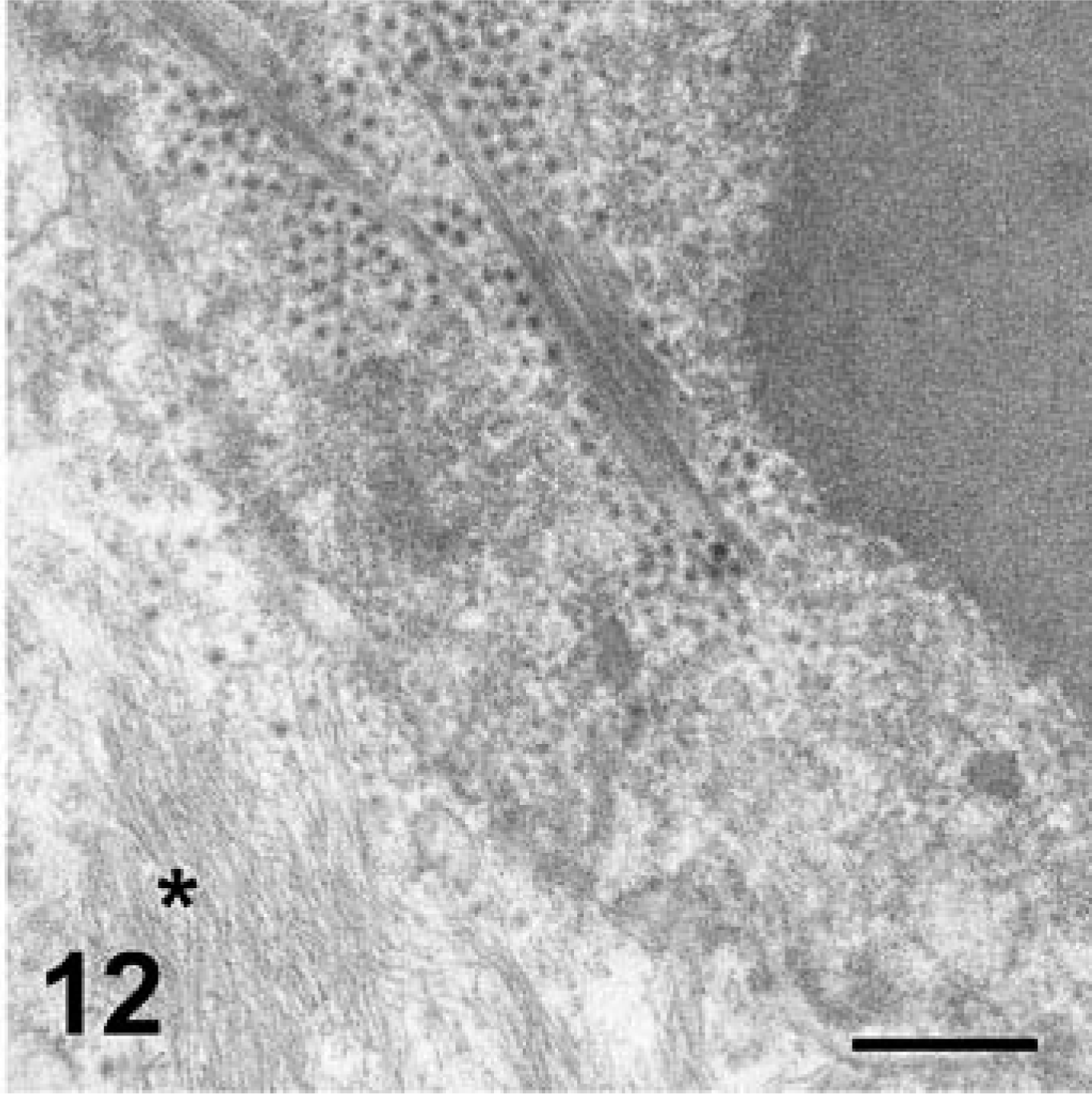
Skin; cat No. 3. Paracrystalline arrays of virus particles within the nucleus of an epithelial cell are associated with protofilaments. Asterisk indicates cytoplasmic cytokeratin bundles. Transmission electron micrograph. Bar = 456 nm.
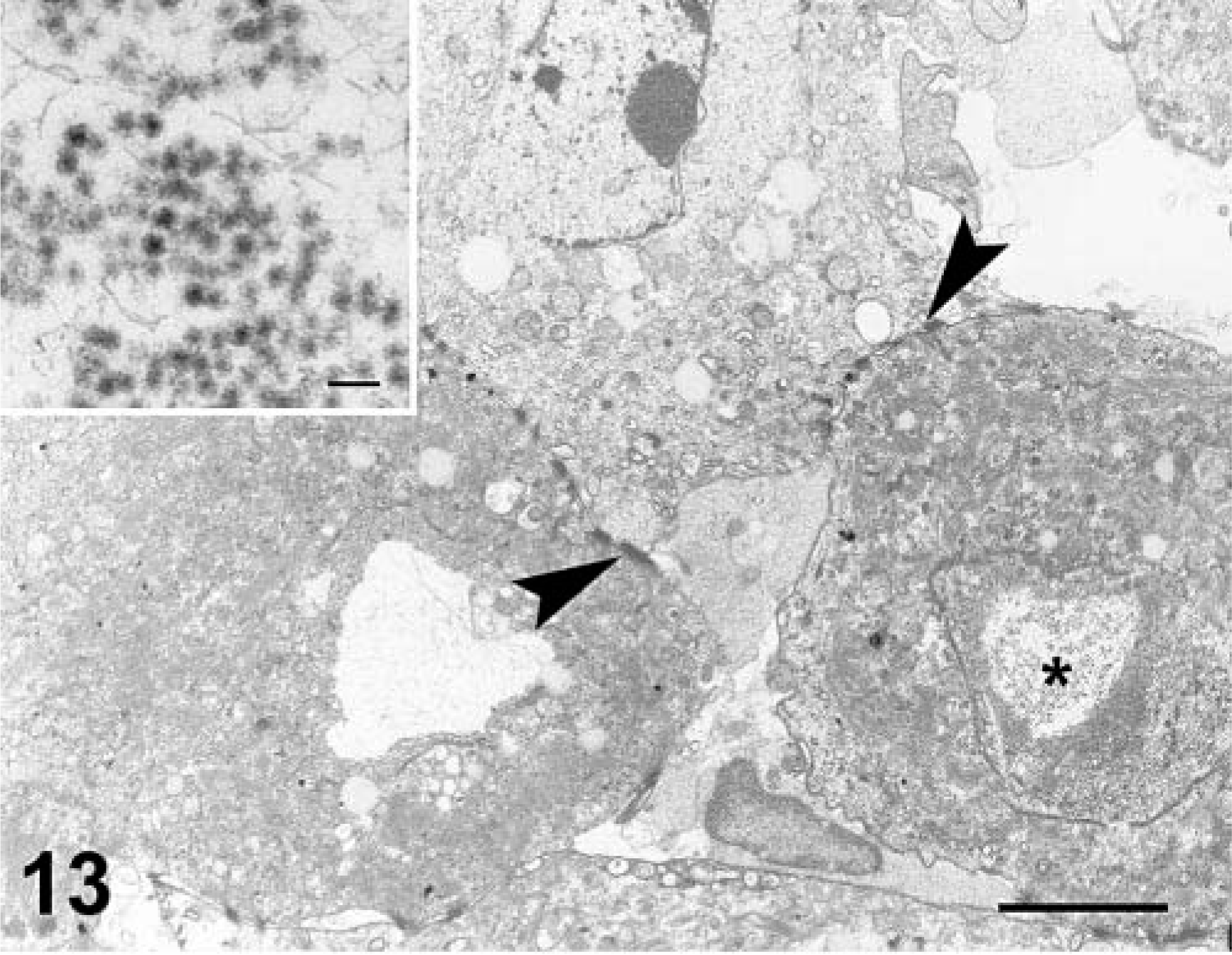
Skin; cat No. 1. There is vacuolation of epithelial cells. Multiple, remnant desmosomes are indicated by arrowheads. Nucleus of one cell (asterisk) contains viral particles and fine filamentous material (inset). Transmission electron micrograph. Bar = 20 µm. Inset bar = 96 nm.
Discussion
This article describes the gross, immunohistochemical, and ultrastructural features of spontaneous VS-FCV infection of cats. Lesions associated with infection included extensive facial and limb edema, ulcers of the skin, footpads, mouth, nares, tongue, and pinnae, and variable involvement of visceral and internal organ systems, including lungs, liver, pancreas, spleen, and lymph nodes.
Clinical signs and lesions of VS-FCV infection are likely to result from a combination of epithelial (cytolytic) and endothelial injury. Viral antigen was intimately associated with epithelial injury. Antigen was present in necrotic epithelial cells of follicles, skin, mucosa, and of affected bronchioles and alveolar septae. VS-FCV virus was also present within the cytoplasm of pancreatic exocrine cells, where it was limited to regions of necrosis. Although field strains of calicivirus had been previously detected in sites of oral epithelial injury (K. West, personal communication). VS-FCV was also identified within endothelial cells in the present study. Virus-induced vascular injury was reflected by edema, microthrombi, and accumulations of fibrin. Viral antigen was not detected within hepatocytes, thus the hepatic lesions that occur in VS-FCV–infected cats may reflect either ischemic injury because of virus-induced destruction of sinusoidal endothelial cells or the effect of circulating mediators produced in response to the infection.
Previous ultrastructural studies on FCV are limited to evaluation of the virus in infected cell cultures that demonstrated FCV within the cytoplasm of cultured cells. 7,13
Definitive identification of virus in these tissues relied on the presence of paracrystalline arrays of 32- to 35-nm viral particles, sometimes associated with proviral fibrillar material. In our studies, VS-FCV was associated with marked degeneration and cytolysis of follicular, mucosal, and keratinized epithelium. An unexpected finding for a positive-stranded RNA virus was the presence of mature viral particles within the nucleus of epithelial cells. Immunohistochemical analyses of rabbit calici viruses (RVHDV and EBHV) revealed that antigen was localized to both the nucleus and cytoplasm of hepatocytes. 9
Ultrastructural studies (DEM) of RVHDV have been accomplished with liver homogenates, however, not in ultrathin sections of tissue (TEM), although this has been attempted. 2,15 Although individual proteins of other positive-stranded RNA viruses have been demonstrated within the nuclei of infected cells, mature viral particles are limited to the cytoplasm. 14–16 The significance of intranuclear VS-FCV is unclear. It is possible that there is a physiologic breakdown of the nuclear envelope, however, the concurrent presence of viral filaments and paracrystalline arrays suggest that viral replication is occurring within the nucleus. Moreover, cells that contained virus within their nucleus contained no detectable cytoplasmic virus.
Very little is known about the pathogenesis of virulent strains of FCV. Caliciviridae exhibit a remarkable genetic diversity. 3,8 VS-FCV strains of FCV are genetically distinct, which likely explains the very striking and characteristic lesions that these viruses induce in susceptible cats.
Footnotes
Acknowledgements
We thank Drs. J. Foley, V. Affolter, H. DeCock, J. Sykes, and D. Wilson for advice and reviewing the cases, Diane Naydan for preliminary immunohistochemical studies, and Bob Nordhausen for his excellent electron microscopy expertise. Support was provided by Maddie's Fund, the Winn Feline Foundation, and the UC Davis Center for Companion Animal Health.
