Abstract
A 9-year-old female spayed Domestic Medium Hair cat presented to the referring veterinarian with a 2-week history of sneezing, which progressed to swelling over the nasal planum. The cat had been under veterinary care for inflammatory bowel disease and had been treated with 1.25 mg/kg prednisolone once a day for approximately 1 year. On physical examination, an approximately 2–3 mm diameter, round polypoid pink soft-tissue mass was protruding slightly from the right nostril. Through histologic examination of representative sections from the mass, there was a severe diffuse infiltrate of epithelioid macrophages and neutrophils that surrounded frequent 15–20 µm yeast organisms. A Grocott methenamine silver stain revealed the presence of pseudohyphae in addition to the previously noted yeast forms. Real-time polymerase chain reaction (PCR) for Cryptococcus neoformans, Ajellomyces dermatitidis (syn. Blastomyces dermatitidis), Coccidioides immitis, Ajellomyces capsulatus (syn. Histoplasma capsulatum), Malassezia spp., and Candida spp. was performed on the paraffin-embedded sample. The PCR for Candida spp. was positive; the product was then sequenced and was determined to be consistent with Candida parapsilosis. Following the PCR diagnosis and prior to treatment of the infection, C. parapsilosis was cultured from a nasal swab. The infection in the cat in the current report was considered opportunistic and secondary to immunosuppression, following treatment for the inflammatory bowel disease.
A 9-year-old female spayed Domestic Medium Hair cat presented to the referring veterinarian for a 2-week history of sneezing. The cat had been routinely seen for treatment of inflammatory bowel disease and had been on 1.25 mg/kg prednisolone once a day for approximately 1 year. The cat was Feline leukemia virus (FeLV) and Feline immunodeficiency virus (FIV) negative as determined by a SNAP FIV/FeLV Combo Test. a The physical exam was otherwise within normal limits with no other abnormalities. The cat was prescribed a 10-day course of amoxicillin–clavulanic acid 15 mg/kg twice a day and the prednisolone was continued. The cat returned to the referring veterinarian 2 weeks later for continued sneezing. At the second visit, the cat had developed a swelling over the nasal planum. Radiographs showed no bony involvement. No further changes in treatment were initiated. A month later, the cat returned to the referring veterinarian and a small 2–3 mm soft, pink, fleshy mass was protruding from the right nostril. The swelling noted previously had increased in size and now extended toward the eye. Clinical differentials at that time included Cryptococcus neoformans infection and neoplasia, including nasal carcinoma and nasal lymphoma.
A representative sample of the nasal mass was taken and immersion fixed in 10% formalin. The tissue samples were routinely sectioned, paraffin embedded, and 4–5-μm sections were prepared and stained with hematoxylin and eosin. Sections were also routinely stained with Grocott methenamine silver (GMS).
On histologic examination, the submucosa was diffusely and markedly expanded by sheets of epithelioid macrophages admixed with neutrophils and small numbers of lymphocytes and plasma cells. There were occasional multinucleated Langhans giant cells. Embedded within the inflammation were numerous 15–20 µm yeast organisms that rarely had a dematiaceous outer wall and a pink round central structure (Fig. 1). The presence of the yeast forms was confirmed with a GMS stain. In addition, small numbers of pseudohyphae were also evident with the GMS stain (Fig. 2). The top differentials based on the morphologic appearance of the organisms included Candida spp. and Malassezia spp.
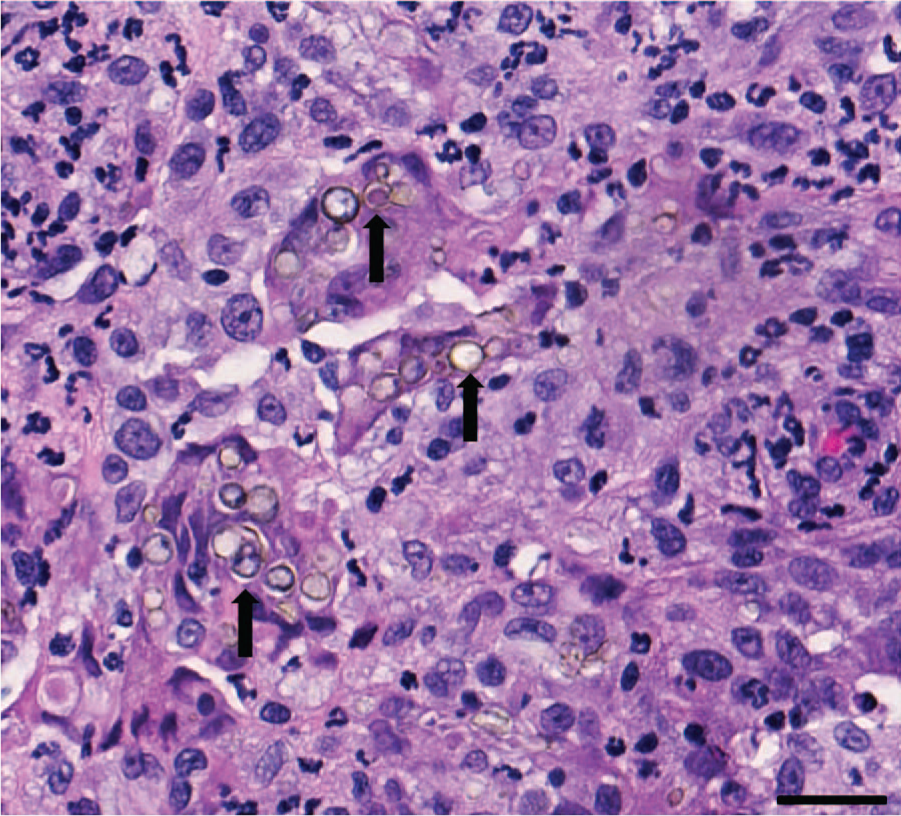
Fungal rhinitis in a cat due to Candida parapsilosis from a rostral nasal mass. There is a marked infiltrate of macrophages and neutrophils that surround variably pigmented yeast organisms (arrows). Hematoxylin and eosin. Bar = 50 µm.
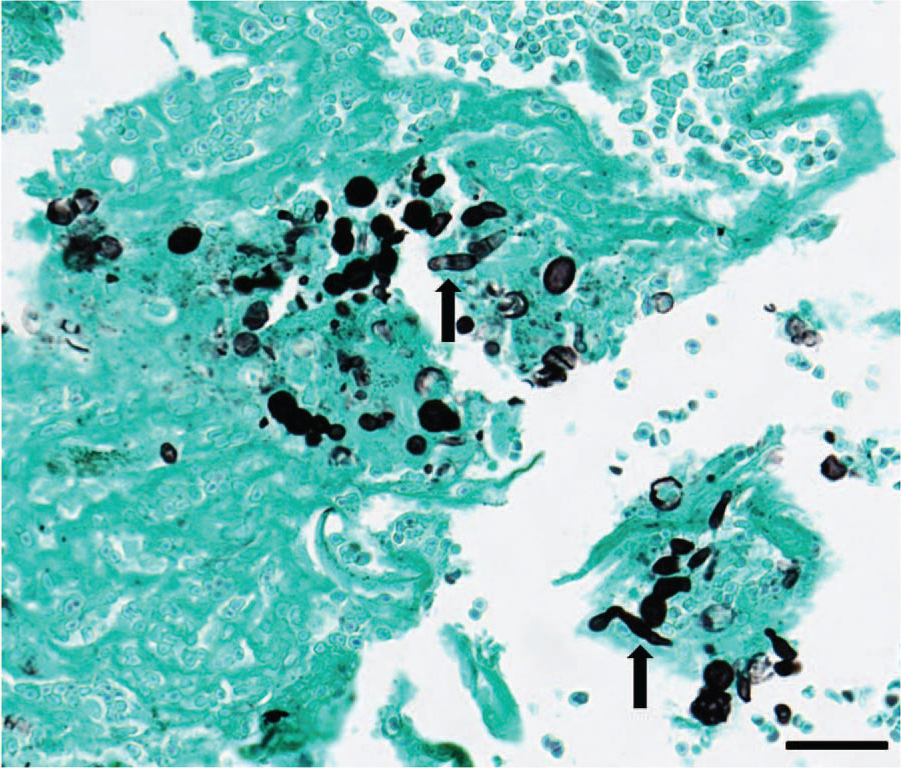
Fungal rhinitis in a cat due to Candida parapsilosis from a rostral nasal mass. Yeast and pseudohypheal forms (arrows) are highlighted with a Grocott methenamine silver stain. Bar = 50 µm.
Three 20-µm scrolls from the paraffin-embedded tissue were used to extract total nucleic acid. Briefly, paraffin was removed with xylene and 2 ethanol wash steps followed by digestion of the tissue with a lysis solution and proteinase K. Subsequently, total nucleic acid was extracted using a column-based extraction system. b Real-time polymerase chain reaction (PCR) was performed with a commercially available panel of PCR tests c including Ajellomyces dermatitidis (syn. Blastomyces dermatitidis), Coccidioides spp., Cryptococcus spp., Ajellomyces capsulatus (syn. Histoplasma capsulatum), Candida spp., and Malassezia pachydermatis. The positive Candida spp. real-time PCR test (based on internal transcribed spacer 2 sequences, GenBank accession nos. EF568101 [C. albicans] and EU564209 [C. parapsilosis] producing PCR products of 79 and 91 base pairs, respectively), was speciated by using outside primers that amplify both C. albicans and C. parapsilosis and direct PCR product sequencing using conventional Sanger sequencing protocols. The obtained sequence showed 99% sequence identity to reported C. parapsilosis sequences in GenBank.
Following initial diagnosis with real-time PCR and prior to treatment, swabs of the cat’s nose were obtained and submitted for fungal culture. The swab was plated onto inhibitory mold agar (IMA), d an enriched selective medium containing chloramphenicol and gentamicin. White colonies were observed growing in the 1+ area of the IMA plates 3 days later. The colonies were routinely Gram-stained and identified as yeast based on size and shape. Colonies were subcultured onto Sabouraud dextrose agar, Emmons formula. d Identification of colonies from subculture was obtained using an automated system and a yeast identification card. e The PCR and culture findings confirmed C. parapsilosis as the etiology of the fungal rhinitis in the current case.
The most common cause of fungal rhinitis in the cat is Cryptococcus neoformans, which results in regional destruction and swelling of the nasal cavity and sinuses. 2 The infection can spread into the cranial vault as well as systemically. Cryptococcus spp. are 4–8 µm in diameter yeast organisms with narrow based budding, a thick nonstaining capsule (soap-bubble appearance), and lack pseudohypheal forms. 2 Cryptococcal infection was ruled out in the current case based on the histologic appearance of the organisms and PCR results. The morphology of the organisms in this case was more suggestive of Candida spp. or Malassezia spp. Unlike some other species of Candida that have multiple forms, C. parapsilosis only has the yeast and pseudohypheal forms. 17 Polymerase chain reaction and culture was useful in confirming C. parapsilosis infection as the cause of the fungal rhinitis in the present case.
Candida parapsilosis is a common commensal organism of a variety of species and can also be isolated from soil. 17 Candida parapsilosis is one of the leading causative agents of fungal disease in people.15,17 It is the second most isolated Candida spp. from blood cultures in adults and third most common organism in general isolated from blood cultures in underweight neonates with late-onset sepsis.4,12,15,17 Infections occur secondary to immunosuppression and are also reported in surgery patients postoperatively, associated with biofilms that have formed on solutions, prosthetics, and indwelling catheters.3,12,17 Candida parapsilosis has been associated with cases of endocarditis, meningitis, septicemia, peritonitis, arthritis, endophthalmitis, keratitis, otitis, cystitis, and skin infections, particularly the nail beds, in people.15,17 Immunosuppression is the major predisposing factor for development of C. parapsilosis infection.
Similar to people, immunosuppression is a predisposing factor for unusual fungal infections in cats. Candida spp. can be a normal commensal organism in cats and has been rarely isolated from the skin of immunosuppressed cats with FIV or FeLV that are otherwise clinically healthy.9,14 Candida albicans is the most commonly isolated Candida spp. in domestic animals with clinical disease. 13 In cats, Candida spp. infection is most often associated with urinary tract infections.5,8,13 Sporadic cases of disseminated candidiasis, intestinal candidiasis, and pyothorax due to C. albicans infection have also been reported in separate cats.6,7,10
Candida spp. are sensitive to itraconazole and voriconazole. 11 Clotrimazole has also been shown to be effective in the treatment of urinary candidiasis. 16 Lufenuron has also been successfully used in the treatment of fungal infections in cats. 1 The cat in the present case was treated with 3.5 mg/kg of itraconazole once a day for 1 month. Two weeks following the start of antifungal treatment, the swelling had completely disappeared and the amount of sneezing had significantly reduced. The findings in this case underscore the importance of including unusual organisms on the differential list for infectious diseases in immunosuppressed patients and highlight the usefulness of PCR and culture in identification of unusual organisms in an effort to direct appropriate treatment.
Footnotes
Acknowledgements
The authors would like to thank the staff of the histology, microbiology, and molecular diagnostic laboratories at IDEXX Laboratories. The authors would also like to thank Dr. Steve Smith for guidance on this case.
a.
IDEXX Laboratories Inc., Westbrook, ME.
b.
DNeasy blood and tissue kit, Qiagen Inc., Valencia, CA.
c.
Test code 3104, IDEXX Laboratories Inc., West Sacramento, CA.
d.
BD, Franklin Lakes, NJ.
e.
Vitek 2, bioMérieux Inc., Durham, NC.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lamm, Estrada, McIlwain, and Leutenegger are employees of IDEXX Laboratories Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
