Abstract
Phocine distemper virus (PDV) is a significant cause of mortality for phocid seals; however, the susceptibility of otariids to this virus is poorly understood. The authors used a lymph-node explant culture system from California sea lions (Zalophus californianus, CSL) to investigate: (1) the role of signaling lymphocyte activation molecule (SLAM) and nectin-4 in PDV infection and their cellular expression patterns, (2) if PDV induces transcriptional regulation of cell-entry receptors, and (3) the involvement of apoptosis in PDV infection. PDV replicated in the lymph-node explants with peak replication 3 days post-infection (dpi), but the replication was not sustained 4 to 5 dpi. The PDV+ cells co-localized SLAM and nectin-4. These cells expressed IBA1, indicating a histiocytic lineage. Comparison of receptor expression between infected and mock-infected lymph nodes suggested transcriptional downregulation of both receptors during the initial stage of infection and upregulation during the late stage of infection, but the values lack of statistical significance. Cleaved caspase-3+ cells were slightly increased in the infected lymph nodes compared with the mock-infected lymph node from 1 to 4 dpi, but without statistical significance, and a few apoptotic cells co-expressed PDV. The results suggest that lymph-node explants might be an important model to study PDV pathogenesis. CSLs have the potential to be infected with PDV, as they express both cell-entry receptors in histiocytes. The lack of statistical significance in the PDV replication, transcriptional regulation of viral receptors, and changes in apoptosis suggest that although CSL might be infected by PDV, they might be less susceptible than phocid species.
Phocine distemper virus (PDV) is a member of the genus Morbillivirus and it has caused important mortality events in harbor seal populations in Europe and North America.14,15,22 The first mass mortality event due to PDV occurred in 1988, when thousands of harbor seals (Phoca vitulina) and lower numbers of gray seals (Halichoerus grypus) died in Northern Europe.8,36 In 1992, PDV was associated with the mortality of harp seals (Pagophilus groenlandicus) and gray seals on the North American portion of the North Atlantic coast.13,14 In 2002, a second PDV outbreak affected harbor seals in Denmark, Sweden, the Netherlands, the UK, and Germany.23,30,40 In 2006, an unusual mortality event caused by PDV occurred in harbor seals, gray seals, and hooded seals (Cystophora cristata) off the North Atlantic coast of the USA.15,47 Nucleic acids of PDV were also detected in live and dead northern sea otters (Enhydra lutris kenyoni) and pinnipeds in the North Pacific, suggesting that PDV may have gained entry to this region by migration of marine mammals facilitated by loss of Arctic Sea ice in recent decades.17,50 However, to date, no case of PDV mortality has been documented in pinnipeds from British Columbia, Washington, Oregon, and California, where a number of phocids and otariids, such as harbor seals and California sea lions (Zalophus californianus, CSLs) are sympatric. 14
Most PDV studies have focused on the negative population effects of the virus during outbreak events, and little is known about the host susceptibility to PDV in many pinnipeds, especially in otariids. Cellular receptors are the critical initial step for cell entry in viral infections. Competence of a cell type for viral replication depends on many factors, but ultimately cell adhesion and internalization are the critical first steps for infection of a given cell type. Morbilliviruses display a strong affinity for their natural cell-entry receptors.12,41 SLAMF1 (synonym, CD150 or SLAM) 49 and poliovirus receptor-like 4 (PVRL4; synonym, nectin-4)29,32 were initially discovered as the receptors for measles virus cell entry. The expression of SLAM was thought to be limited to leukocytes, whereas nectin-4 was thought to be present in epithelial cells. The identification of the receptors aided in the understanding of the tropism of morbilliviruses for leukocytes and epithelial cells, as well as the development of the immunosuppression and pneumonia associated with infection.10,24 Due to substantial evidence linking the pathogenesis of the measles virus to host cell receptors, other researchers have sought to demonstrate similar data for other members of the genus Morbillivirus, including canine distemper virus (CDV),33,46 rinderpest virus, 3 peste des petits ruminant virus, 5 and PDV. 27 In marine mammals, the SLAM and nectin-4 amino acid sequences indicate the potential for interaction with the hemagglutinin protein of morbilliviruses, including PDV, to facilitate infection.24,35 It was previously shown that PDV preferentially bound cell lines overexpressing SLAM in their membrane. 31 Another study showed that PDV is able to use SLAM and nectin-4 receptors, as well as the additional receptor pro-heparin-binding epidermal growth factor (HB-EGF), for cell entry in Vero cells. 27
Our lab has utilized canine lymph-node explants to study the pathogenesis of CDV infection in dogs, 18 and we hypothesized that lymph-node explants from CSLs would be equally effective for the detection of PDV receptors. The aims of this study were to demonstrate (1) the involvement of histiocytic cells expressing SLAM and nectin-4 receptors in the PDV lymphoid infection; (2) that both receptors might be transcriptionally regulated during experimental PDV infection, and (3) the possible role of PDV in lymphoid apoptosis in CSL lymph-node explants.
Material and Methods
Cell Culture
VerodogSLAMtag cells were seeded in T75 flasks and incubated with 1× Dulbecco’s Modification of Eagle’s Medium (DMEM) (CORNING, cellgro) supplemented with 10% Fetal Bovine Serum (Corning, cellgro), 0.008% G418 (VWR, Life Science) and 0.01% of 100× antibiotic-antimycotic solution (Anti-Anti, Gibco, Life Technologies). The cells were incubated at 37°C in an atmosphere containing 5% CO2 until the monolayer reached 95% confluency.
Phocine Distemper Virus
We used the PDV strain that caused the unusual mortality event off the East Coast of USA in 2006 (PDV/USA2006). The virus was grown in VerodogSLAMtag cells. When the monolayer was more than 95% confluent, the medium was removed, and cells were rinsed twice with 1× Dulbecco’s phosphate-buffered saline (DPBS) (Gibco, Life Technologies). One milliliter of the PDV inoculum was added and incubated for 1 hour at 37°C in 5% CO2, after which the inoculum was removed and 10 ml of cell culture medium was added. The inoculated explants were incubated for 5 days. Cell culture medium and monolayers with cytopathic effect were harvested, cleared by brief centrifugation, and aliquots were stored at –80°C until infection.
Animals
Axillary lymph nodes, spleen, and brain samples from 5 juvenile CSLs admitted for rehabilitation to The Marine Mammal Center, Sausalito, California, were collected at necropsy. The lymphoid system was not compromised by disease in any of the animals based on clinical, gross, and microscopic examination. Necropsies and sample collection were performed within 4 hours after euthanasia, and carcasses were stored at 4°C until necropsy. Lymph node, spleen, and brain samples from each animal were tested by real-time polymerase chain reaction (RT-PCR) (described below) to assure the samples were free of PDV.
Collection, Culture, and Infection of Lymph-Node Explants
One axillary lymph node from each animal was aseptically removed and immersed in transport media with 10 parts per liter of 100× antibiotic-antimycotic solution (Gibco, life technologies) and 1× phosphate-buffered saline (PBS) (Gibco, life technologies). Lymph nodes were transported on ice packs and processed within 6 hours of collection. The samples were vigorously washed with the transport media, and the pericapsular fat and connective tissues were removed. Approximately, 2-mm thick transverse sections were made, and explants were placed in 6-well plates (1 per well) and incubated overnight at 37°C in 5% CO2. The explant medium contained 5 ml of Roswell Park Memorial Institute (RPMI) 160 medium with
After overnight incubation, the explant culture medium was removed, and the explants were inoculated with 200 µl of cell culture media containing 1.5 × 1010 PDV viral copies/µl. Both cut surfaces of the explants were exposed to the inoculum and were incubated for 1 hour at 37°C in 5% CO2. The explants were then washed twice with cell culture media, submerged in fresh culture medium, and maintained for 5 days at 37°C in 5% CO2. Control lymph-node explants were maintained as described above, but the PDV inoculum was replaced by cell culture media without PDV. Infected and control explants were sampled every day after inoculation. A subsample from each time point was stored at –80°C, and another was fixed in 10% buffered formalin.
Immunohistochemistry and Image Analysis
Formalin-fixed lymph-node explants were dehydrated in graded alcohol and embedded in paraffin. Serial, 5-µm thick sections of each explant placed on positively charged glass slides that were deparaffinized and rehydrated after passage through xylene and graded alcohol. Heat-induced epitope retrieval was performed using a vegetable steamer and a commercial target antigen retrieval solution (DAKO, pH: 6.0). The endogenous peroxidase was blocked with a commercial blocking solution (BLOXXAL™, Vector Laboratories). Endogenous avidin and biotin were also blocked with a commercial blocking system (Thermo Scientific). Non-specific antigens were blocked with 5% normal goat or 5% normal rabbit serum in tris-buffered saline-tween 20 buffer. Mouse monoclonal antibodies specific for anti-CDV-Nucleoprotein (VMRD, Inc., Pullman, WA, USA) that detects PDV 14 and rabbit anti-cleaved caspase-3 (Asp175, Cell Signaling) were diluted to 1:400 and 1:800, respectively, then incubated overnight. A biotinylated goat anti-rabbit immunoglobulin (Ig) G was used as secondary antibody followed by incubation with the avidin-horseradish peroxidase (HRP) complex (Thermo Scientific). Binding was visualized with a commercial red substrate (ImmPACT™ NovaRED™, Vector Lab), and slides were counterstained with modified Mayer’s hematoxylin (Thermo Scientific). After each step, the slides were washed twice with tris-buffered saline-tween 20 for 10 minutes. Slides of canine brain infected by morbillivirus (CDV) were used as positive controls, and CSL spleen from a pup was used as positive control for cleaved caspase-3. For negative control, the primary antibody was replaced by an antibody with identical isotype.
Ten fields for each infected and mock lymph-node explant (capsule, subcapsular sinus, lymphoid follicle, and medullary sinus) were photographed at 400x (field of view: 450 × 450 micrometers) and the PDV+ or cleaved caspase-3+ cells were quantified using the software, Fiji (NIH, Bethesda, MD), with the cell counter plugin.
Double Immunofluorescence
Slides were deparaffinized, rehydrated, and pretreated with heat-induced epitope retrieval. Non-specific, antigen blocking was performed with a commercial blocking solution (Universal Blocker™ in TBS, Thermo Scientific). Combined pairs of primary antibodies were incubated at 4°C overnight as follows: mouse monoclonal CDV-NP antibody with rabbit polyclonal SLAMF1 (Invitrogen, 1:100 dilution), nectin-4 (Invitrogen, 1:100 dilution), IBA-1 (Ionized Binding Calcium Adapter Molecule-1) (Wako, 1:800 dilution), or anti-cleaved caspase-3 (Cell Signaling, 1:400) antibody. Two fluorescent conjugated secondary antibodies, Alexa Fluor 594-red conjugated goat anti-rabbit (Molecular probes, Life Technologies) and Alexa Fluor 488-green conjugated rabbit anti-mouse antibody (Invitrogen), were used. Slides were mounted with medium containing 4',6-diamidino-2-phenylindole (DAPI) (DAPI Fluoromount-G Mounting Medium, Southern Biotech) for nuclear staining. After each step, the slides were washed twice with tris-buffered saline-tween 20 for 10 minutes. Samples of tonsil and spleen from CSL pups were used as positive controls for SLAM, nectin-4, and IBA1, and cleaved caspase-3.
RT-PCR for PDV Quantification
Viral replication in the lymph-node explants was measured using tissues sampled at 24-hour intervals after infection. To develop a standard curve, RNA extracted (QIAamp® Viral RNA, QIAGEN) from the PDV/USA2006 strain grown in VerodogSLAMtag cells was used. A 169 bp amplicon corresponding to the PDV nucleoprotein (N) gene was amplified using the primers, PDV-N-Forward (5′-GCACCTAACCAAAGACTCCCTC-3′) and PDV-N-Reverse (5′-GGTCCCCTTCCTGTGTCAATTGT-3′) 19 and one-step reverse transcriptase PCR kit (QIAGEN), following the manufacturers guidelines, in a 50-µl reaction volume containing enzyme mix, 10 mM dNTP, 5× RT-PCR buffer, forward and reverse primers, RNase-free water, and viral RNA. The PCR amplicon was cloned into the pCR4-TOPO vector (TOPO TA cloning kit, Invitrogen), and it was chemically transformed into competent Escherichia coli (One shot TOPO 10, Invitrogen). The plasmids were extracted from the E. coli colonies utilizing the quick plasmid miniprep kit (PureLink, Invitrogen). Plasmids were linearized with the PmeI restriction enzyme (New England BioLabs) and purified with a commercial spin column system (Wizard® SV Gel and PCR clean-up system, Promega). Ten-fold dilutions of the cDNA were made to create a standard curve. Samples were reverse transcribed with a commercial reverse transcription kit (Quanti Tect®, QIAGEN) in a reaction volume of 20 µl using random primers. The Taqman RT-PCR used the PDV primers above and the probe 5′-/56-FAM-TTCCAATCAGCTCATT/36-TAMRA-Sp/-3′. 19 The reaction volume of cDNA, primers, probe, ROX dye solution, and master mix was used according to the manufacture’s guidelines (QuantiFast, QIAGEN), and the fluorogenic RT-PCR was carried out in a 7500 Fast Real-Time PCR system (Applied Biosystems). The cycling conditions started with a PCR initial heat activation at 95°C for 3 minutes and a total of 40 cycles consisting of denaturation at 95°C for 3 seconds and a combined annealing/extension step at 60°C for 30 seconds. The Ct values were converted into log10 viral RNA copies/µl based on the 10-fold standard dilutions. 53
RT-PCR for SLAM and Nectin-4 Receptor Expression
Infected and control lymph-node explants were sampled at 24-hour intervals after infection, and these samples were processed using a 2-step RT-PCR SYBR® green assay. Total RNA was extracted with a commercial kit (RNeasy® Plus, QIAGEN) and the first-strand cDNA was synthesized (qScript™ XLT cDNA SuperMix, Quanta, Biosciences) in a total volume of 20 µl with Moloney Murine Leukemia Virus (M-MLV) reverse transcriptase. We used the PerfeCta™ SYBR® Green FastMix (Quanta, Biosciences) kit as the RT-PCR master mix that contains 2× reaction buffer, dNTPs, Taq DNA Polymerase, SYBR Green I dye, ROX reference dye, and stabilizers. The primer pairs used were specific for GAPDH, the housekeeping gene, 19 CSL SLAM (F-5′-GAACAGGTCAGAGCTTGATG-3′ and R-5′CGCTACCTTCTTCTTGACAC-3′), and CSL nectin-4, (F-5′-GACCACGGTGAGGGAGATTG-3′ and R-5′-ATGTAGATGCCATTGCCGGT-3′), These produced amplicon sizes of 197, 167, and 165 bp, respectively. The final volume of the reaction mixture was 20 µl per reaction with the following cycling protocol: initial denaturation at 95°C for 3 minutes and 40 cycles at 95°C for 15 seconds with an annealing and extension step at 60°C for 60 seconds. The ΔCt of the infected and control lymph-node explant per day were used to obtain the ΔΔCt value and the expression-fold change was calculated based upon the 2–ΔΔCt formula. 26
Statistical Analysis
The plots of the PDV replication rate per day post-infection, microscopic localization of PDV+ cells in the lymph node, SLAM and nectin-4 receptor expression, and cleaved caspase-3+ cells in lymph-node explants are presented using the mean and bars representing standard error of the mean (SEM). A P value < .05 was considered statistically significant. The normality of the data obtained from the above experiments was determined by Shapiro-Wilk test, and statistical differences were determined with either 1-way analysis of variance (ANOVA) or Kruskal-Wallis 1-way ANOVA according to the parametric or non-parametric nature, respectively. Statistical analyses and graphics were designed with the 7.0 GraphPad software (La Jolla, California).
Results
Infection and Replication of PDV in Lymph-Node Explants From CSLs
Infection with PDV in lymph-node explants was detected by immunohistochemistry. Most of the PDV+ cells had a morphology consistent with macrophages. Scattered multinucleate cells were also observed, mainly from 3 to 5 dpi, and these were interpreted to be syncytia (Fig. 1a). Replication of PDV was detected by 1 dpi and viral-copy numbers gradually increased until 3 dpi with > 1-log-unit increase 1 to 3 dpi (Fig. 1b). The titers on the 4 and 5 dpi were similar to the 3 dpi, but they were greater than the first 2 dpi. Although, the replication tended to increase after viral exposure, the differences in the titers were not statistically significant (P > .05). These results prove that PDV is able to infect, replicate, and generate cytopathic effects in the lymphoid cells of CSLs, but the viral-copy numbers did not significantly increase over time.
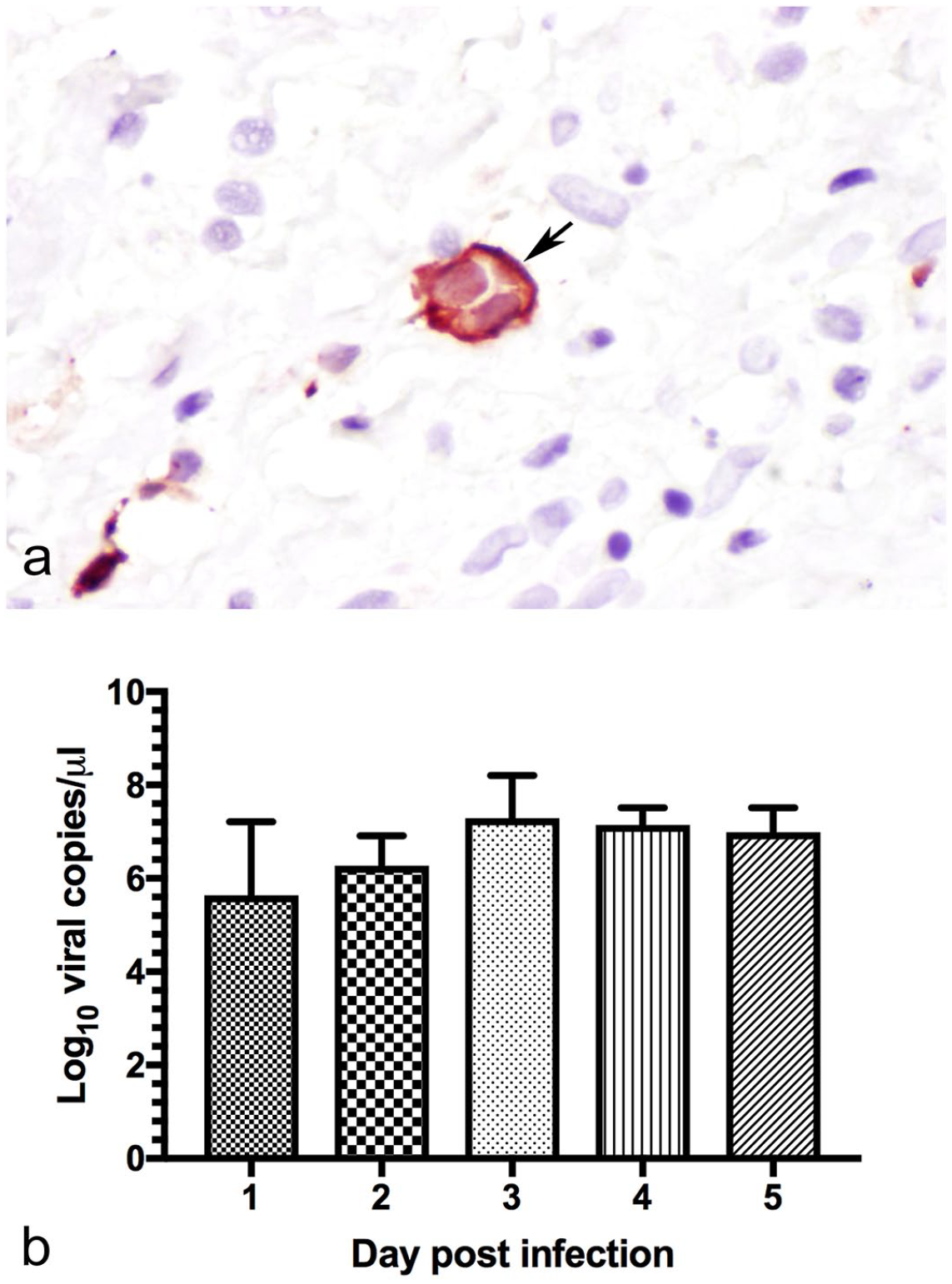
Experimental infection and replication of phocine distemper virus (PDV) in California sea lion lymph-node explants in culture. (a) A PDV immunolabeled multinucleated cell resembling syncytium (arrow) is shown in the capsule, with abundant cytoplasm and multiple nuclei. Anti-nucleoprotein-canine distemper virus immunohistochemistry. (b) PDV replication with the highest titers detected at 3 days post-infection (dpi) with a slight decrease between 4 and 5 dpi.
SLAM and Nectin-4 Receptors are Present in CSL Tissues and Could Facilitate PDV Infection
To determine whether the cells of CSLs express competent receptors for PDV, we assessed the expression of SLAM and nectin-4 in lymph nodes using double immunofluorescence. The PDV+ cells also had cytoplasmic labeling for either SLAM (Fig. 2a–d) or nectin-4 (Fig. 2e–h), but not both simultaneously (Fig. 2a–h). To identify the potential target cell for PDV cell entry in lymph nodes, we co-localized the PDV-infected cells with IBA1 (Fig. 2i–l) and demonstrated that a population of resident histiocytes have receptors needed for cell entry. Within the lymph nodes, quantification of PDV+ cells showed that subcapsular sinuses contained more positive cells than other regions, the capsule and medullary sinus had the second and third greatest concentration of positive cells, respectively, while the smallest number of PDV+ cells was in the lymphoid follicles (Fig. 3a–c). Cells with viral antigen in the cytoplasm were large, round to polygonal, and had a single prominent nucleus; all features consistent with macrophages (Fig. 3a–c). The differences in the number of PDV+ cells in the capsule, the subcapsular sinuses, and the medullary sinuses were statistically significant when compared with the number of PDV+ cells in the lymphoid follicles (P < .05) (Fig. 3d).
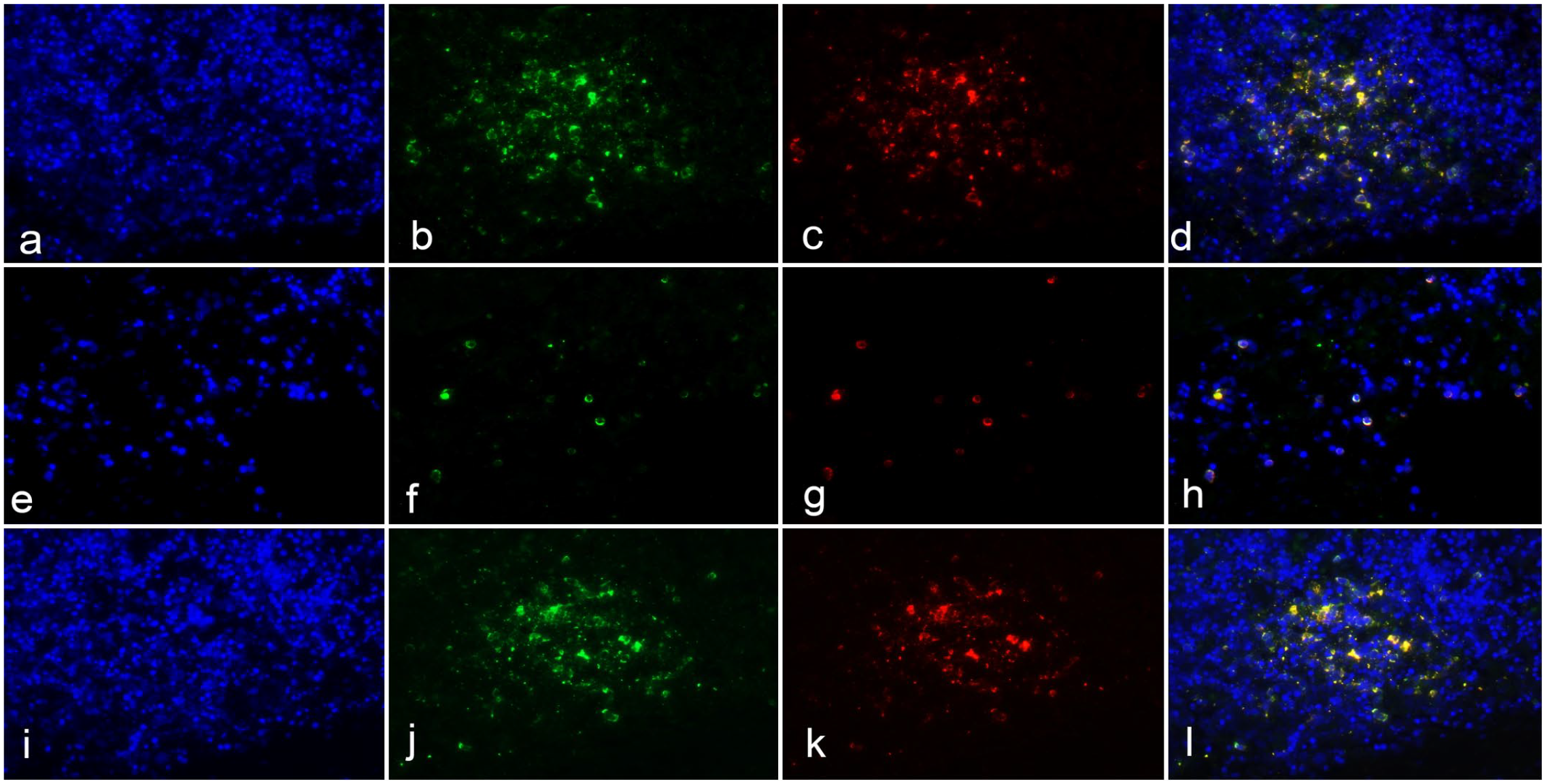
Double immunofluorescence of phocine distemper virus (PDV) with SLAM, nectin-4, and IBA1 receptors in lymph-node explants with DAPI nuclear counterstaining (a, e, and i, blue). (a–d) Clusters of cells in the cortex show cytoplasmic labeling for PDV (
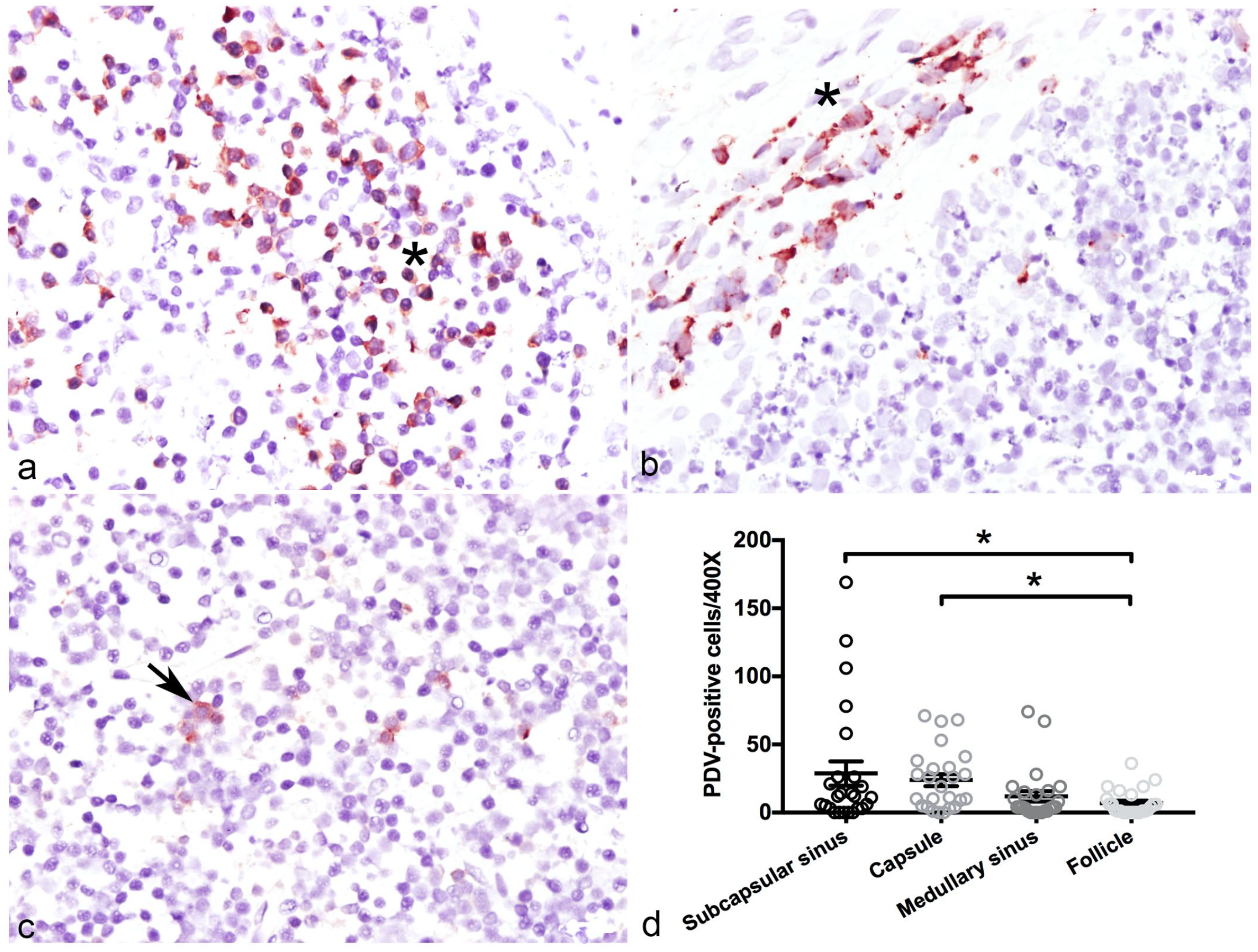
Localization of phocine distemper virus (PDV)-infected cells in lymph-node explants in culture with immunohistochemistry (IHC). (a) Subcapsular sinus (asterisk) with numerous histiocytic cells showing red cytoplasmic labeling. PDV IHC. (b) Lymph-node capsule (asterisk) containing multiple round to fusiform cells with red, granular labeling in the cytoplasm. PDV IHC (c) Lymphoid follicle with a few PDV+ cells (arrow). PDV IHC. (
SLAM and Nectin-4 Might be Transcriptionally Regulated in CSL Lymph-Node Explants Infected by PDV
Since PDV was able to replicate in the lymph-node explants, we investigated the transcriptional regulation of the cell-entry receptors in the PDV-infected lymph nodes, in comparison with the non-infected controls. Transcription of both SLAM and nectin-4 was downregulated in the first 2 dpi (Fig. 4a, b) and varied by 0.35-log-unit for SLAM and 0.15-log-unit for nectin-4 at 1 dpi. However, only changes in SLAM were statistically significant (P < .05) compared with the mock-infected lymph node at 1 dpi (Fig. 4a). Even though expression of both receptors was decreased by 2 dpi, decreases were not statistically significant (P > .05). By 3 dpi, SLAM was slightly downregulated (0.1 log units) but nectin-4 was upregulated with a log unit of 0.2. Minimal upregulation of SLAM and nectin-4 was evident in samples collected at 4 and 5 dpi. In general, SLAM and nectin-4 gene-expression were downregulated in the first 2 days after inoculation, and they were slightly upregulated in the last 2 days of the experiment. However, these results are trends and not supported by statistical significance.
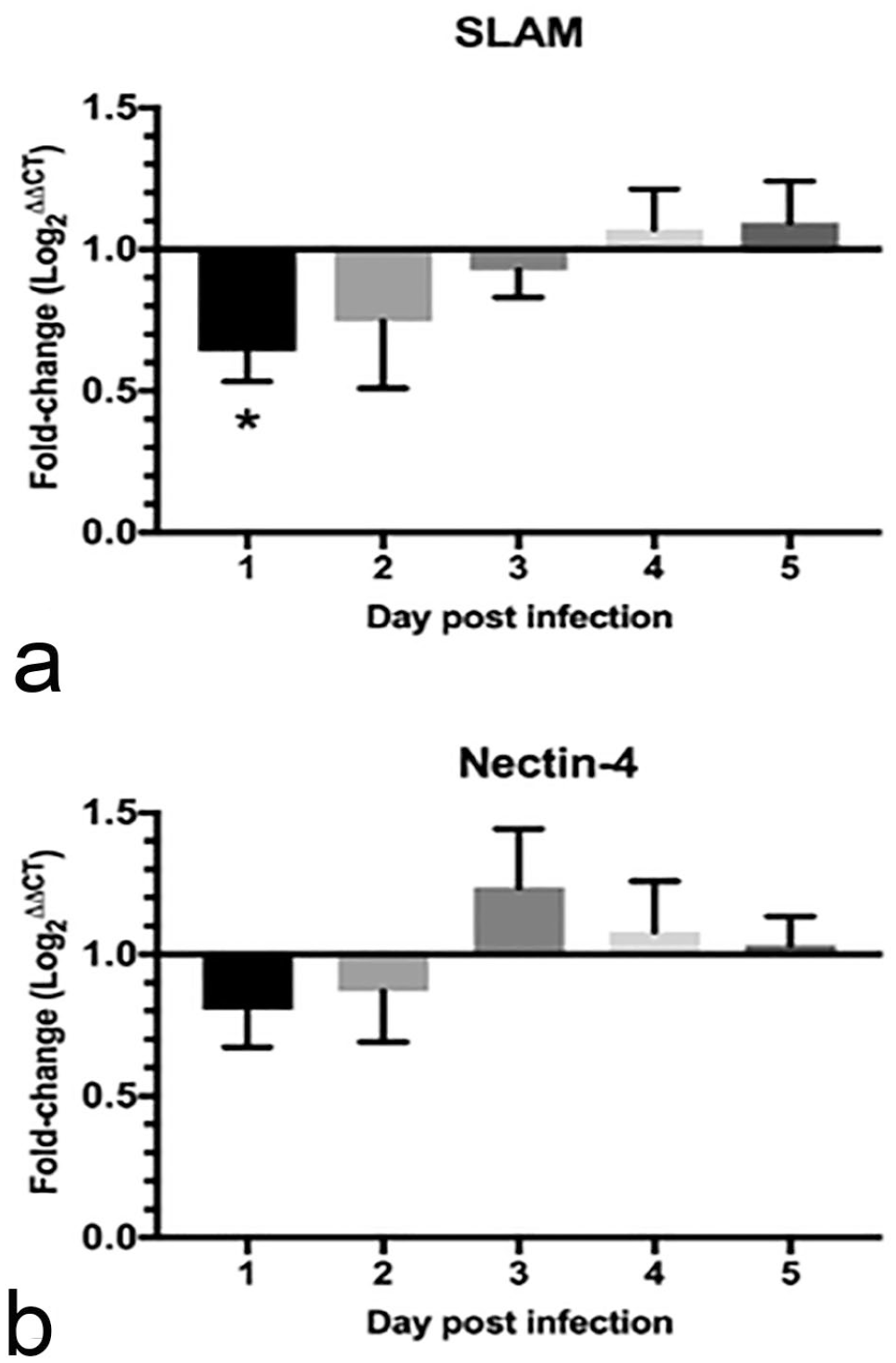
Fold-change difference of SLAM and nectin-4 receptor expression in infected compared with mock-infected lymph nodes. (a) SLAM was downregulated from 1 to 3 days post-infection (dpi) and was slightly upregulated at 4 and 5 dpi. (b) Nectin-4 expression was downregulated at 1 and 2 dpi and upregulated in the remaining dpi. Asterisk (*) indicates statistically significant difference when P < .05.
Caspase-3-Based Apoptosis in CSL Lymph-Node Explants Infected by PDV
We found slightly increased cleaved caspase-3+ cells in PDV-infected lymph-node explants compared with uninfected explants during the first 4 dpi (Fig. 5), but this difference was not statistically significant (P > .05). To determine whether PDV directly induces apoptosis in leukocytes, we performed double immunofluorescence in which a few apoptotic cells positive for cleaved caspase-3 (Fig. 6b) co-expressed the viral PDV antigen (Fig. 6c, d). This result suggests the possible selective inhibition of apoptosis by PDV during lymph-node explant infection in CSLs.
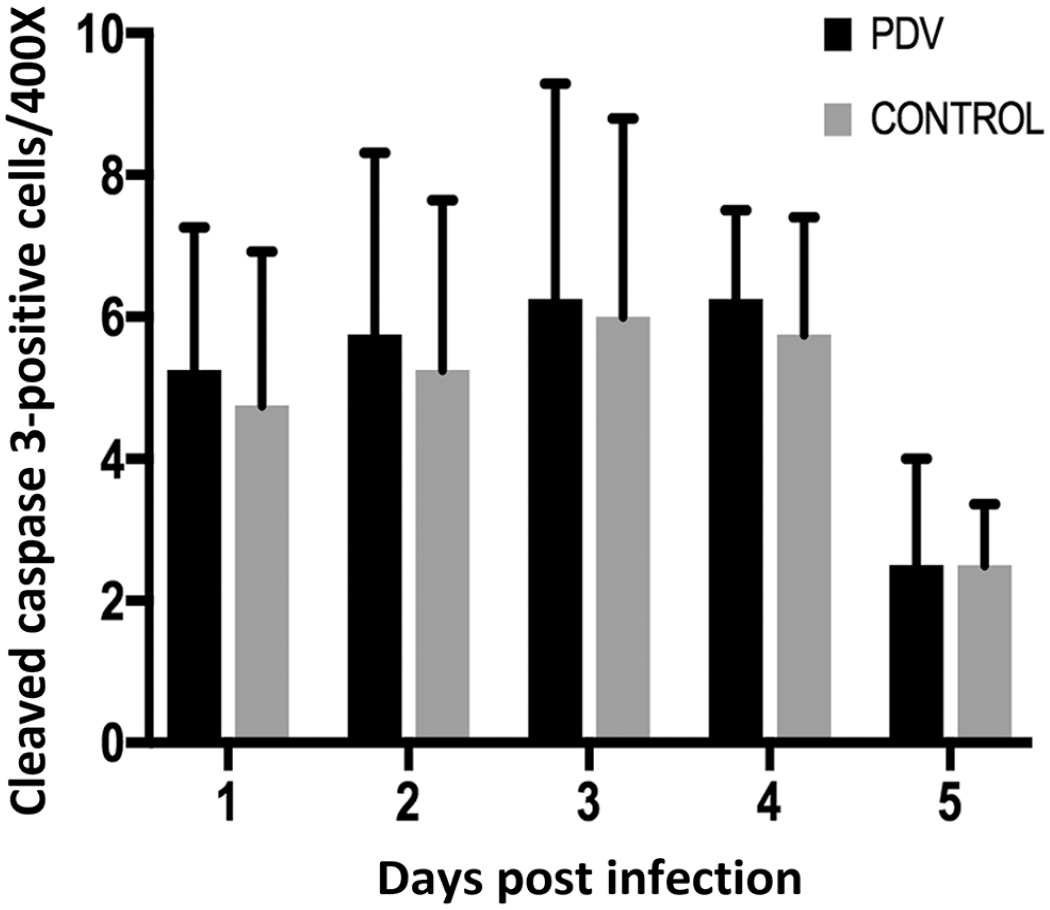
Quantification of cleaved caspase-3+ cells in lymph-node explants in culture. There was a slight increase in the number of apoptotic cells in the infected lymph nodes, in comparison with negative controls, from days 1 to 4.
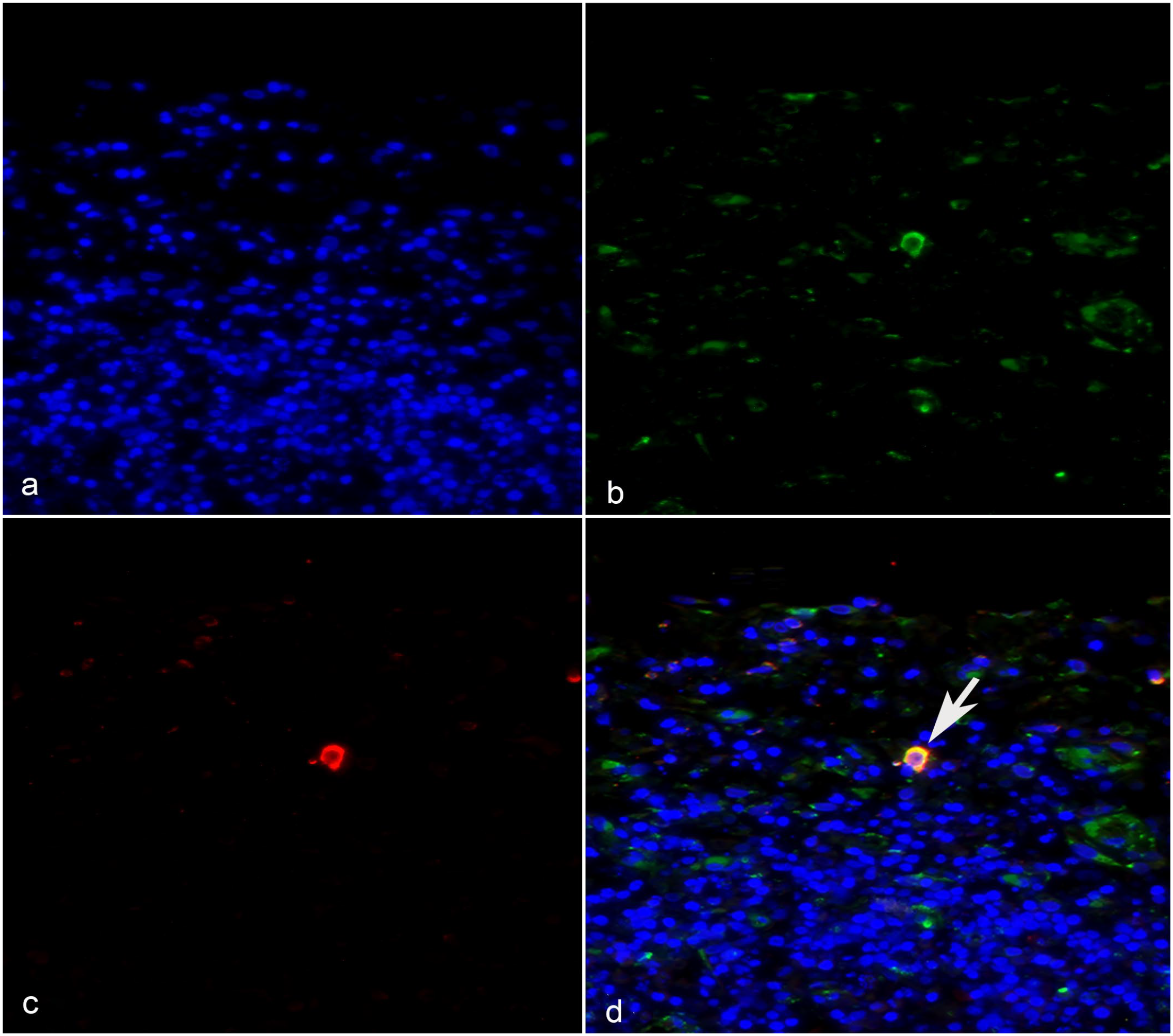
A few cells expressing phocine distemper virus (PDV) antigens co-localized with cleaved caspase-3 (arrow), whereas the majority of PDV+ cells did not co-label with the apoptotic marker. (a) DAPI nuclear counterstaining (blue). (b) PDV+ cells (green). (c) Cleaved caspase-3+ cytoplasm (red). (
Discussion
PDV was capable of cell entry and replication in CSL lymph-node explants in culture, confirming that they have the receptors necessary for PDV viral entry and infection. To date, no evidence has been documented to suggest that free-living CSLs may be infected by PDV, 14 but there is one report of a fatal infection of a captive CSL by the related morbillivirus, CDV. 4 However, PDV has been detected in ice-associated seals, Steller sea lions, and northern sea otters in the Artic North Pacific. 50 In our study, expression of the SLAM and nectin-4 receptors in the lymph-node explants, which are needed for morbilliviral cell entry, suggest that these cells are susceptible to infection by PDV in culture. Our results suggest that if CSLs are exposed to PDV, they may have the potential to become infected.
PDV was able to replicate in CSL lymph-node explants, demonstrating the potential of this model for the study of PDV infection in pinnipeds. In vitro methods to study the PDV pathogenesis have mainly used stable and transformed Vero and CHO cell lines;27,45 however, those cells have limitations since they were originally obtained from species other than pinnipeds. Experiments investigating replication of PDV in culture have used gray seal and harbor seal peripheral-blood mononuclear cells6,7 and primary seal kidney cells. 31 Although, both types of cell lines are from pinnipeds, they also have some limitations. Peripheral blood mononuclear cell cultures are challenging to use and need to be activated with exogenous compounds that may change the expression of cell membrane molecules, as well as the intracellular machinery.2,21 PDV does not propagate efficiently in primary seal kidney cells, and the cells do not show typical cytopathic effects. 31 Our study demonstrates that PDV is able to infect, replicate, and form syncytia in the infected lymph-node explants from CSLs. The syncytial cells have been observed in PDV-infected lymph nodes and spleens during natural infection in seals, but are smaller and not as abundant as the equivalent in cetaceans infected with cetacean morbillivirus. 14 Another advantage of using lymph-node explants is the ability to co-localize viral antigens with host receptors, such as SLAM and nectin-4, and cell markers, such as IBA1.
SLAM and nectin-4+ cells are involved in the infection of lymph nodes by PDV. Both cell-entry receptors are important for morbillivirus pathogenesis and determine tissue tropism within the host. 10 The expression of SLAM is exclusive to activated T and B lymphocytes, mature dendritic cells, macrophages, thymocytes, and platelets in humans and mice.41,49 It serves as a co-stimulatory molecule to initiate signal transduction networks in natural killer cells, T cells, and antigen-presenting cells.10,41 A set of transfected cell lines were previously used to show that PDV is able to use SLAM as a cell-entry receptor; however, this has not been shown in pinniped tissues. 27 In our study, numerous cells expressed SLAM, and SLAM was co-localized with PDV in the cytoplasm, suggesting a role of the receptor in the ability of PDV to infect pinniped lymph-node explants.
Nectin-4 is a member of the nectin family that contains Ig-like domains similar to SLAM. It is essential in cell-to-cell adhesions and in the regulation of cell polarization, differentiation, movement, proliferation, and survival.34,41 Human nectin-4 is exclusively expressed on the basolateral side of epithelial cells.29,32 This specific receptor localization is crucial for measles virus and CDV cell entry, as the virus binds to nectin-4 receptors on the basolateral surface to enter epithelial cells.25,34,42 In apparent contradiction, we detected nectin-4 in the CSL lymph-node explants by immunofluorescence in their cytoplasm of cells that are morphologically suspected to be macrophages. This suggests nectin-4 could also serve as a cell-entry receptor for PDV in lymph nodes.
The PDV-infected cells in our experiments were identified as histiocytes based on their IBA1 expression. This marker is used to identify cells from the monocyte/macrophage lineage in dogs and cats 37 and has been used to visualize macrophages in bacterial and protozoal inflammatory processes in CSLs.43,44 Another important factor to determine the histiocytic lineage was the cell morphology and the predominant histological distribution in the capsule and subcapsular sinus, which are the primary regions containing macrophages in lymph nodes. 20 Similar results have been shown with measles virus infection, where macrophages and dendritic cells are the predominant infected cell type in humans in different stages of infection. 1
The transcriptional regulation of SLAM and nectin-4 receptors was minimally influenced by PDV in the infected lymph-node explants from CSLs, and the transcription difference between SLAM and nectin-4 was statistically significant only in the first-day post-infection. Virus-induced receptor downregulation has been demonstrated in association with some natural viral infections, including human immunodeficiency virus (HIV), simian immunodeficiency virus (SIV),9,38 and measles virus.32,48,51 For measles virus, 2 major mechanisms have been proposed to explain this phenomenon. One involves the retention of SLAM in the endoplasmic reticulum by the measles hemagglutinin, thereby blocking its efficient transport to the cellular surface. The second involves the direct hemagglutinin-SLAM interaction in the membrane that promotes receptor internationalization by endocytosis.48,51 Both mechanisms involved post-translational influence of the hemagglutinin protein in receptor downregulation. In our study, we demonstrated that PDV was able to downregulate SLAM and nectin-4 during the first 2 dpi, but only SLAM was statistically significant at 1 dpi. This finding suggests PDV may suppress the transcriptional factors that induce the synthesis of the viral receptors. Since the morbillivirus receptors, such as SLAM, are major co-stimulatory molecules, downregulation may affect lymphocyte activation and promote immunosuppression,48,51 or could enhance total virus replication by preventing the lethal superinfection of individual cells. 38 The studies demonstrating measles-induced receptor downregulation have been performed following 48 hours post-infection.32,48,51 In contrast, we carried out infections up to 5 days and found on the third day the receptors behaved differently, with a slight downregulation of SLAM and an upregulation of nectin-4. Both receptors were upregulated at 4 to 5 dpi. Similarly, SLAM is upregulated by CDV when dogs show clinical signs of disease. 52 Another possibility for not having seen exclusive receptor upregulation during the 5 days post-PDV infection is the potential for reduced susceptibility in CSLs to PDV infection. More studies examining this possibility are needed.
Lymphocyte apoptosis induced by caspase-3 is a feature of PDV replication, and other members of the morbillivirus family are known to induce lymphocyte depletion by apoptosis during infection.11,16,28 In our study, the number of cleaved caspase-3+ cells were highest at 3 dpi in the control and PDV-infected explants. Furthermore, a few cells expressing PDV antigens had cytoplasmic cleaved caspase-3 labeling, which suggests the possible induction of apoptosis by PDV in cells in infected lymph-node explants, but the quantification of apoptotic cells was not statistically significant. In CDV-infected dogs, lymphocyte apoptosis is induced by cleaved caspase-3 through the caspase-8 initiator pathway. 11 In our explants, most leukocytes expressing PDV antigens were not positive for cleaved caspase-3. This finding suggests that PDV-induced apoptosis may not be universal during PDV infection of lymph nodes of CSLs. In CDV-infected ferrets, the virus may selectively inhibit lymphoid cell apoptosis as a probable mechanism of immune evasion. 39 This finding suggests that PDV may induce apoptosis through the caspase-3 pathway, but it is not increasing apoptosis in CSL lymph-node explants.
In summary, lymph-node explants were used to investigate if PDV could infect lymphoid tissues in CSLs. Results show that CSLs have the cell-entry receptors SLAM and nectin-4 in the cells of the histiocytic lineages in lymphoid tissues and have the potential to become infected if exposure to the virus were to occur. Given that replication and apoptotic cells were not increasing on time in the CSL tissue explants in culture, and that down and up regulation of the receptors was not significant compared with controls, results could indicate that CSLs may not be as susceptible to PDV infection compared with some phocid seal species. This article highlights the value of studying viruses not only in most susceptible species, but in less susceptible to know disease mechanisms and factors that influence this susceptibility.
Footnotes
Acknowledgements
We thank Magdalena Plancarte and the staff at The Marine Mammal Center for sample collection from the California sea lions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding support to OGV was provided by Innovate Peru and the graduate student support program, School of Veterinary Medicine, UC Davis. The Wild Animal Health Fund of the American Association of Zoo Veterinarians funded the majority of this project.
