Abstract
Raccoons (Procyon lotor) are abundant in urban/wildland interfaces and are key sources of canine distemper virus (CDV) outbreaks in domestic, zoo, and free-ranging wildlife species. CDV is pantropic, which provides multiple potential routes of transmission (urine, respiratory secretions, feces), but the specific role of skin as a target of infection, as a diagnostic sample, or as a potential source of environmental persistence and transmission is unknown. We have characterized the distribution of CDV and its known receptor, nectin-4, in skin samples of 36 raccoons. Even with skin samples that were grossly and histologically normal, immunohistochemistry of skin was useful in the diagnosis of CDV infection, which was found in both epithelium and endothelium. Nectin-4 was codistributed with cellular targets of viral infection. Skin secretions, shed keratinocytes, and hair of CDV infected raccoons are all potential environmental fomites.
The expansion of the human/wildland interface of suburbia supports substantial population densities of synanthropic (peridomestic) carnivores, (ie, raccoons, skunks, coyotes, foxes) that benefit from anthropogenically modified ecosystems.2,10 In these areas, viral spillover events of multi-host pathogens, such as canine distemper virus (CDV), are favored by the more likely interaction between these species.21,22,26 In addition to being an impactful, often fatal pathogen in domestic and free-ranging Carnivora spp., potential spillover to nonhuman primates also supports zoonotic concerns. 3 Raccoons are abundant mesocarnivores in urban/rural interfaces 11 and are highly susceptible to CDV infection. 5 Seroprevalence studies demonstrate that clinically healthy survivors from CDV exposure are a potential reservoir for the virus 1 and/or a source for virus spillover. 3
CDV binds the signaling lymphocyte activation molecule (SLAM) to enter respiratory epithelium or mucosal leukocytes. 25 Most research regarding CDV host susceptibility and interactions has, therefore, focused on host SLAM receptor differences. 17 But SLAM is not expressed in epithelium or endothelium, so viral targeting of cells involved in the shedding, transmission, and spillover of the virus most likely relies on binding and entry via the second known receptor for CDV, the adherens junctional molecule nectin-4.7,24 Transmission studies that specifically identify how intra- and interspecies transmission might occur are extremely challenging in natural settings, where (at a minimum) patterns of behavior, nesting, movements, and viral stability in the environment are all at play. While direct contact is effective for transmission, 20 patterns of outbreaks suggest that indirect transmission in natural and laboratory settings does occur and is efficient.6,23 Nectin-4 is the receptor most likely to support the pantropic tissue distribution (urine, feces, respiratory secretions, skin) that provides multiple potential sources of shed virions, although no long-term stability studies of persistence and infectivity from different tissue sources have been performed. 16 Any effort to understand indirect modes of transmission requires a thorough understanding of tissue tropism and viral pathogenesis. Skin and mucosa are among many known potential tissue targets of CDV, but the distribution of the virus in skin and whether it is a consistent target of infection are unknown. We propose that CDV within skin, including shed hair shafts and keratinocytes, would be environmentally stable sources of CDV. 9
The complex pathogenesis of CDV makes it a diagnostic challenge in any species, but especially problematic in wildlife that typically present with no detailed history of disease progression or inception. CDV can persist in tissues, can manifest as neurologic disease days to months after infection, and can be shed long after initial infection. The complex susceptibility of an individual host and the variability in the clinical sequelae of infection make diagnostics challenging. Polymerase chain reaction (PCR), including for raccoons, is highly sensitive and specific, and reverse transcription real-time polymerase chain reaction (RT-rtPCR) well correlates with immunohistochemistry (IHC) findings; however, histologic evaluation and IHC are commonly used for the detection of CDV infection 19 in most diagnostic laboratories. Reports using skin as a diagnostic sample include 2 studies in the dog, one examining the footpad 15 and the other a study reporting viral distribution in haired skin 12 which concluded that the skin is a useful sample in that species. We hypothesized that if the highly conserved nectin-4 receptor is widely distributed in epidermis, follicular epithelia, adnexal structures, and endothelium of raccoons, as it is in the dog, 8 skin would be a useful diagnostic sample for analysis of CDV infection (antemortem or postmortem), and that skin (hair, secretions, or keratinocytes) could be a source of inter- or intraspecies transmission.
We used a series of naturally occurring cases of distemper virus infection in raccoons: (1) to analyze the presence and distribution of CDV in skin, (2) to characterize the distribution of nectin-4 in haired and nonhaired skin, and (3) to determine whether skin is a reliable sample for the diagnosis of infection in raccoons. We analyzed CDV by IHC in the skin of 36 raccoons with a confirmed diagnosis of CDV. In 32 cases, IHC was positive in a sample of brain, lung, or both. Specifically, of the samples tested from 36 cases, CDV was detected by IHC in the brain in 9 (82%) of 11 cases, in lung in 27 (84%) of 32 cases, in lip skin in 29 (81%) of 36 cases, and in other skin in 17 (74%) of 23 cases (Supplemental Table S1).
Animals for this study were adults or young adult raccoons (estimated >6 months) submitted for routine necropsy during the years 2012–2020 to the anatomic pathology services at the Veterinary Medicine Teaching Hospital or the California Animal Health and Food Safety laboratory at the University of California, Davis (20 animals) or to the Michigan State University Veterinary Diagnostic Laboratory (16 animals). The inclusion criteria for this cohort were positivity by IHC using lung, brain, or skin. Necropsy (Supplemental Methods for details) included routine tissue sampling, along with sampling of lip (mucocutaneous junction and vibrissae in all cases) and additional haired skin samples (dorsal cervical and/or ventral inguinal skin in 23 of 36 cases).
By gross examination alone, the skin of CDV-positive animals was normal except in 4 of 36 cases that had exudative or ulcerative dermatitis (Fig. 1a). In those, plus 4 additional cases (total 8/36, 22%), there was histologic evidence of multifocal dermatitis or folliculitis, and pinpoint to segmental ulceration (Fig. 1b). We speculate, given the endotheliotropism and epitheliotropism of the virus, that ulceration was caused by a combination of direct cytolysis and infarction. Among the remaining 28 cases, no gross or histologic evidence of inflammation (dermatitis or folliculitis) was present.
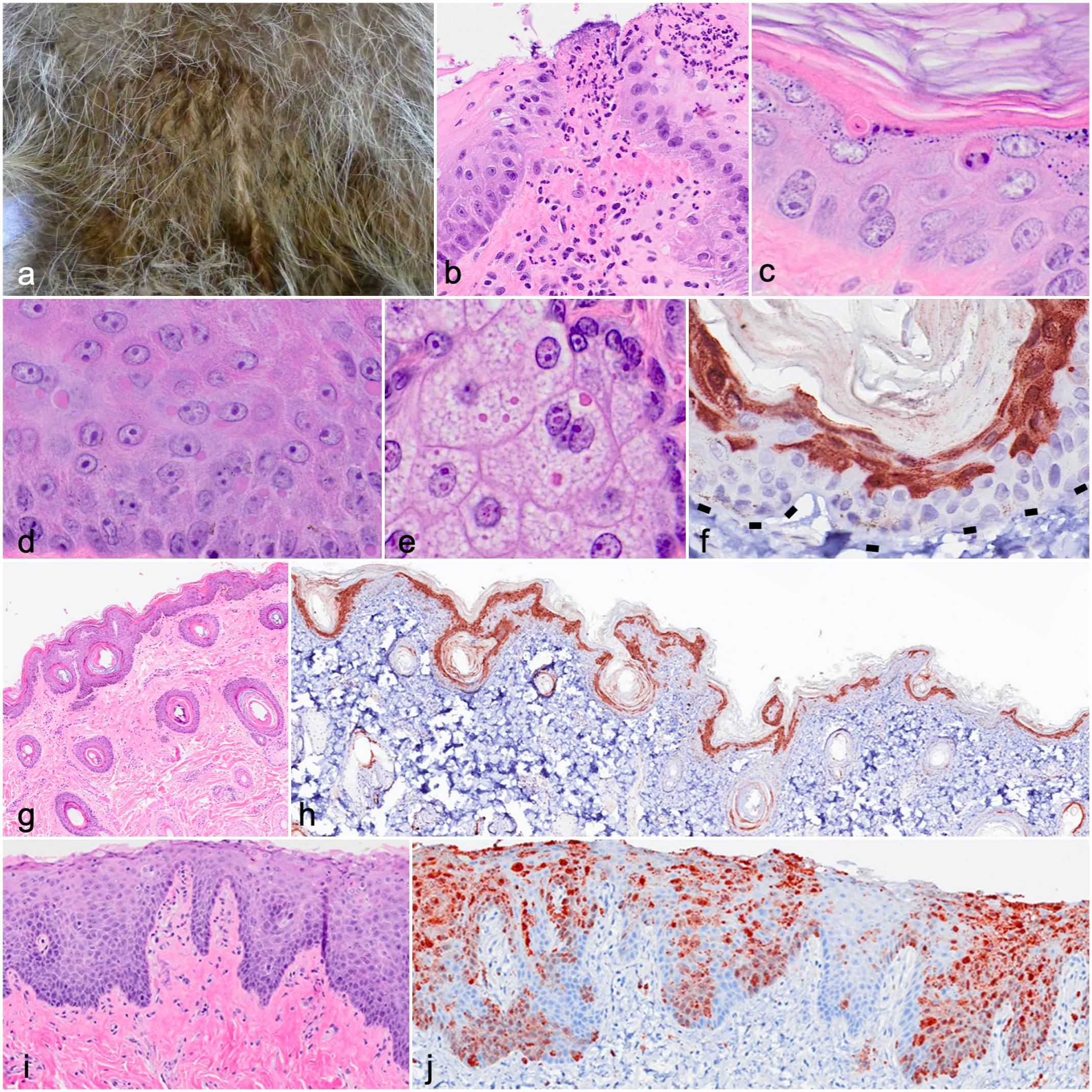
Distribution of canine distemper virus (CDV) in raccoon skin and adnexa. (a) Exudative dermatitis, ventral thorax. Case 5. (b) Ulcer, lip mucosa. Neutrophilic stomatitis is limited to the region subjacent to the ulcer. Case 7. Hematoxylin and eosin (HE). (c) Individual cell death, haired skin. Case 3. HE. (d) Intracytoplasmic inclusions, lip mucosa. Case 12. HE. (e) Intranuclear and intracytoplasmic inclusions in sebaceous glandular epithelium. Case 12. HE. (f–h) Distribution of immunolabeling for CDV in the epidermis of haired skin. Immunohistochemistry (IHC). Case 13. (f) Immunolabeling in the epidermis is limited to the superficial epithelial strata and spares the basal layer. Dashed lines represent the epidermal-dermal junction. (g, h) In infected but histologically normal haired skin, IHC labels CDV antigen in the superficial epidermis and most follicles. At the isthmus of a hair follicle, CDV is present within the outer root sheath and is contiguous with the upper strata of the interfollicular epidermis. (i, j) Mucosa (lip). Case 24. There is minimal inflammation (i, HE) but extensive immunolabeling for CDV in the mucosa (j, IHC), where CDV antigen is multifocally contiguous from the basal to the surface epithelium.
More subtle histologic findings were detected in many cases: Single epithelial cell death characterized by pyknotic or fragmented nuclei (Fig. 1c) was present in 16 (44%) of 36 cases but was considered nonspecific. Epithelial syncytia formation, while highly suggestive of CDV infection, was rare (2/36). Cytoplasmic and/or nuclear inclusions were present in skin epithelium in 14 (38%) of 36 cases (apocrine glands [13/14] > follicular epithelium [10/14] > epidermis [8/14] and/or sebaceous glands [6/14] (Fig.1d, e)).
Immunohistochemistry with a mouse monoclonal antibody to virus nucleoprotein (clone DV2-12; Bio-Rad, Hercules, California; Supplemental Methods) was performed on the skin (lip) sample in all cases and was positive in the lip of 29 of 36 cases. For comparison, detection of CDV by IHC on the lung was positive in 27 of 32 cases, and 9 of 11 animals were positive by IHC of the brain. The selection of the lung and/or brain, and the site within those tissues, was chosen by the on-site pathologist and was not consistent. As has been demonstrated in studies of other tissues (lung, brain), 20 virus-specific microscopic lesions of the skin detectable by H&E were not consistently associated with infection. Specifically, of the 36 cases of CDV infection confirmed by IHC, 14 (39%) of 36 had inclusions, 8 (22%) of 36 had dermatitis, and 2 (6%) of 36 had syncytia detected by histopathology (Supplemental Table S1).
In the skin, CDV antigen was detected by IHC in the endothelium (Supplemental Fig. S2) and the epidermal, follicular, and adnexal epithelia (Fig. 1g, h). In haired surfaces, CDV immunolabeling of surface epidermis was segmental to expansive (Fig. 1f, h) but often completely spared the basilar layer (Fig. 1f, h). Where CDV antigen was detectable in the epidermis, it was often contiguous with the follicular ostia and infundibula and coalesced along the surface layer (Fig. 1h). In hair, CDV antigen was identified in the isthmus, outer root sheath, hair bulb, and the endothelium of the vascular bed of the ring (blood) sinus in the lip vibrissae (Fig. 2g–i). In lip mucosal epithelium, CDV antigen was present segmentally in the basilar layer and more extensive in progressively more superficial keratinocytes (Fig. 1i, j). Distribution of viral antigen among dermal structures (glands, follicles) was similar in all cases. By both histopathology (Fig. 1e) and ultrastructural pathology (Supplemental Fig. S1, case 7), inclusions of the sebaceous glands and/or hair follicular epithelium were both cytoplasmic and nuclear. If viral stability and infectivity are preserved in shed keratinocytes, hair shafts, and the secretions that coat these structures, these could be a source of environmental transmission and persistence.
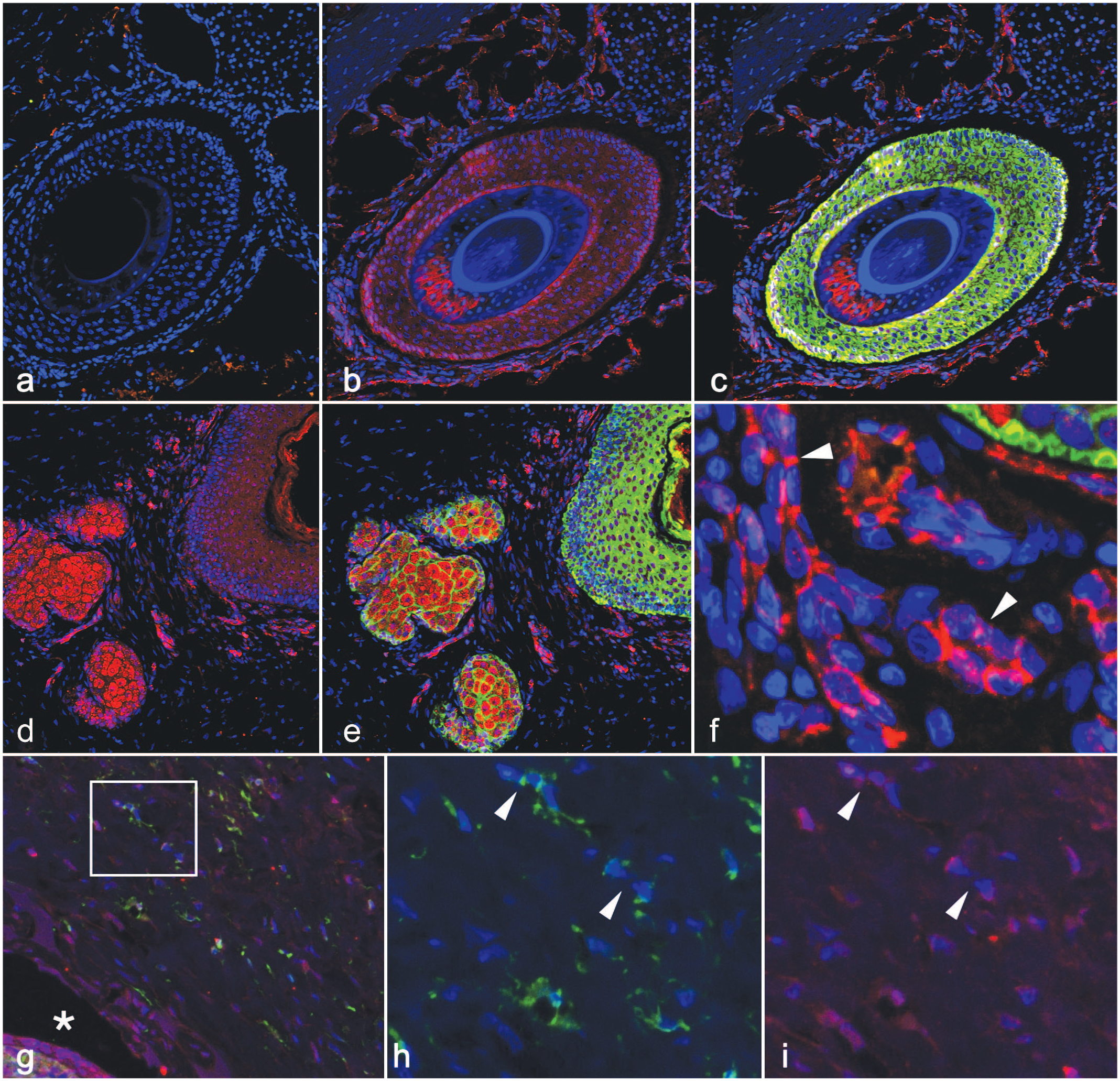
Distribution of nectin-4 and canine distemper virus (CDV) antigens in normal and infected epidermis, follicles, and adnexa. (a–f) Normal (uninfected) raccoon, sinus hair. (a) Using a negative control antibody for signaling lymphocyte activation molecule (SLAM, green), there is a lack of labeling in the hair follicle. Nuclei are visible in all sections by counterstain (DAPI, blue). (b, c) In serial sections of uninfected sinus hair, using an antibody specific to nectin-4 (red, b, c) and to cytokeratin (green, c) there is abundant expression of nectin-4 in the follicular epithelium of the outer root sheath. Nectin-4 is also found in cell-cell junctions of endothelial cells of the vascular bed surrounding the sinus hair (b and c, red). Cytokeratin (green, c) is expressed in outer root sheath follicular epithelium. (d, e) Nectin-4 (d, red) and cytokeratin (e, green) immunofluorescence shows abundant nectin-4 in follicular epithelium and sebaceous glands. (f) Nectin-4 (red) is expressed in the endothelium of the capillary bed in the ring sinus of the vibrissae (arrowheads). (g–i) Sinus hair from a raccoon infected with CDV. Case 7. (g) CDV (green) and nectin-4 (red) labeling overlap (merged photo) in endothelium (inset) of cavernous sinus vessels and the hair follicle wall (asterisk). (h, i) Squared area in (g). Endothelial cells (arrowheads) from the sinus hairs are infected with CDV (h, green) and coexpress nectin-4 (i, red).
To establish whether the distribution of a known, well-conserved CDV receptor (nectin-4)7,24 co-localizes with the viral cellular targets we identified by IHC, we analyzed the nectin-4 distribution in normal and infected raccoon skin in 4 animals (2 uninfected and 2 infected) by immunofluorescence using a rabbit polyclonal antibody (Biorbyt, St Louis, Missouri; catalog number orb5809, RRID: AB_10924278; Supplemental Methods). A BLAST search identified a 312-kb scaffold, JAABKC0 10002608.1, that contained the raccoon nectin-4 sequence, with high similarity (96.7%) to the protein in dogs and humans, suggesting that this antibody recognizes the highly conserved nectin-4 protein. Consistent with distribution seen in those species, immunofluorescence of normal raccoon haired skin demonstrated nectin-4 widely distributed within the follicular surface epithelium (Fig. 2b), glandular (sebaceous) epithelia (Figs. 2e), and the endothelium of the capillary beds of the dermis (Fig. 2f), as well as the capillaries within the ring sinus of the vibrissae (Fig. 2b, i). In infected animals, CDV distribution in endothelium (Fig. 2g–i and Supplemental Fig. S2) and in epithelia overlapped with that of nectin-4 (Fig. 2g–i).
The epidermis, follicular epithelia, and adnexal structures, as well as the capillary endothelia of raccoons, are expected targets of CDV infection (based on prior literature), and the distribution of the known receptor nectin-4 overlaps with the viral distribution in these skin structures. Nectin-4 is part of the junctional complex 24 of both endothelium and epithelium, so infection of skin epithelium could be similar to the proposed route of infection in the brain, where endothelium serves as the route of viral spread to neuronal and glial cells after systemic infection. 4
Neither inflammation nor virus-associated histologic changes (inclusions, syncytia, apoptosis) were correlated with the presence of CDV antigen, so the value of most skin samples, like those of the lung and brain, requires appropriate testing with immunohistochemistry to establish a diagnosis. In this study, the brain and/or lung site tested was by the choice of the diagnostician, so a direct comparison of the diagnostic usefulness of any single tissue was not possible. Reports of skin infections in the domestic dog 12 have shown the diagnostic usefulness of skin samples for detection of CDV antigen. Comparative studies of viral distribution in other CDV-susceptible species13,14,18,21 could benefit from including skin as a convenient and potentially consistent target of infection.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221102598 – Supplemental material for Distribution of canine distemper virus and nectin-4 in raccoon (Procyon lotor) skin
Supplemental material, sj-pdf-1-vet-10.1177_03009858221102598 for Distribution of canine distemper virus and nectin-4 in raccoon (Procyon lotor) skin by Pedro A. Triana Garcia, Andrew Cartoceti, Verena K. Affolter, Kenneth Jackson, M. Kevin Keel, Dalen Agnew, Thomas Cooley, Julie Melotti, Scott D. Fitzgerald and Patricia A. Pesavento in Veterinary Pathology
Footnotes
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Center for Companion Animal Health, University of California, Davis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
