Abstract
A juvenile female hooded seal (Cystophora cristata) and a juvenile male harp seal (Phoca groenlandica) stranded separately on the New Jersey (USA) coast and were taken to a marine mammal rehabilitation center. Both were lethargic and emaciated, had dermatitis, and died. Histologic skin lesions in the seals were similar and consisted of epidermal and follicular epithelial hyperplasia, hyperkeratosis, degeneration, and necrosis. The most distinctive finding was extensive syncytial zones bounded superficially by hyperkeratosis and deeply by hyperplastic basal cells. Eosinophilic intracytoplasmic inclusion bodies were present in epithelial cells. Morbilliviral antigen was demonstrated in the skin lesions by immunohistochemistry. Phocine distemper virus was detected in the skin by reverse transcription polymerase chain reaction and a phocine distemper virus-specific probe using the Southern blot technique. This is the first report of morbilliviral dermatitis in marine mammals.
Since 1987, morbilliviruses have emerged as causes of epizootics that killed tens of thousands of pinnipeds and cetaceans. 1,3,4,6,8 When stranded marine mammals are brought into facilities for rehabilitation, there is a risk of transmission of morbillivirus to resident marine mammals, with potentially catastrophic results. The purpose of this report is to describe the clinicopathologic findings and results of immunohistochemical and reverse transcription polymerase chain reaction (RT-PCR) testing of two seals with morbilliviral dermatitis.
During February and March 1998, rehabilitation of a juvenile female hooded seal (Cystophora cristata; seal No. 1) and a juvenile male harp seal (Phoca groenlandica; seal No. 2) that stranded separately on the New Jersey (USA) coast was attempted. The two seals were at the rehabilitation facility at different times. Clinically, both were lethargic and emaciated and had dermatitis. The hooded seal became dyspneic and died 16 days after stranding. The harp seal died 10 days after stranding.
Necropsy findings in the hooded seal included emaciation, pneumonia, rhinitis, and gastric nematodiasis; those in the harp seal included emaciation and gastrointestinal nematodiasis. Skin lesions in the hooded seal affected the front flippers, the abdominal area, and a rear flipper. The gross appearance was not described. The harp seal had areas of alopecia and crusting that affected the dorsal surface of a flipper, cranial and dorsal tail, caudal and dorsal sacrum, and areas just dorsal to the flippers.
Tissues were fixed in 10% neutral buffered formalin and processed for histopathologic evaluation by standard methods. For immunohistochemical analysis, 5-µm thick paraffin-embedded tissue sections were mounted on Esco Superfrost Plus glass slides (Erie Scientific, Portsmouth, NH), deparaffinized and rehydrated. Endogenous peroxidase activity was blocked by incubation with Peroxide Block (Biogenex, San Ramon, CA) for 20 minutes at room temperature. After washing in a phosphate-buffered saline solution (PBS), nonspecific binding of secondary reagent was blocked by incubating specimens in Power Block (Biogenex, San Ramon, CA) for 20 minutes at room temperature. Antisera used as a source for primary antibody was a mouse origin anti-canine distemper virus IgG2b. Tissue sections were incubated with the primary antibody for 1 hour at room temperature. Following a PBS wash, the secondary antibody, biotinylated goat anti-mouse immunoglobulin Mouse Link (Biogenex) was applied for 30 minutes. The slides were washed with PBS and treated with peroxidase-conjugated Label (Biogenex) for 20 minutes, washed with PBS, visualized with aminoethyl carbazole substrate (Biogenex), and counterstained with hematoxylin for 1 minute. Formalin-fixed, paraffin-embedded tissues were tested for morbillivirus by an RT-PCR technique and by hybridization of the amplified products with a phocine distemper virus (PDV)-specific probe as previously described. 5,9
Similar highly distinctive histologic skin lesions were present in both seals. Affected skin had markedly thickened epidermis with three distinct layers: a deeply eosinophilic superficial layer of orthokeratotic hyperkeratosis, a basophilic deep zone of hyperplastic basal cells, and a lightly eosinophilic middle band composed of numerous viable, degenerate, and necrotic syncytial cells (Fig. 1). In addition to typical syncytial cells with 2–10 nuclei, there were extensive areas in which cell boundaries were absent and nuclei, often in clusters of 3–40, were scattered in a background of lightly eosinophilic cytoplasm. These areas gave the appearance of syncytial zones rather than discrete syncytial cells. Hair follicle infundibula had similar changes, with increased amounts of central lamellated keratin, a middle syncytial zone, and peripheral hyperplastic basal cells (Fig. 2). Nuclei in syncytial zones and syncytial cells were sometimes hyperchromatic or pyknotic. In some areas, 3–8-µm-diameter, round to oval, homogeneous, eosinophilic intracytoplasmic inclusion bodies were observed in epidermal, follicular (Fig. 3), and sebaceous cells. Similar intranuclear inclusion bodies were rare. Syncytial cells were also present in sebaceous glands (Fig. 4). There were small numbers of neutrophils and lymphocytes in the epidermis, follicular epithelium, and periadnexal dermis. In some skin samples from both seals, there was a thick layer of bacterial and fungal overgrowth that covered the hyperkeratotic crust.
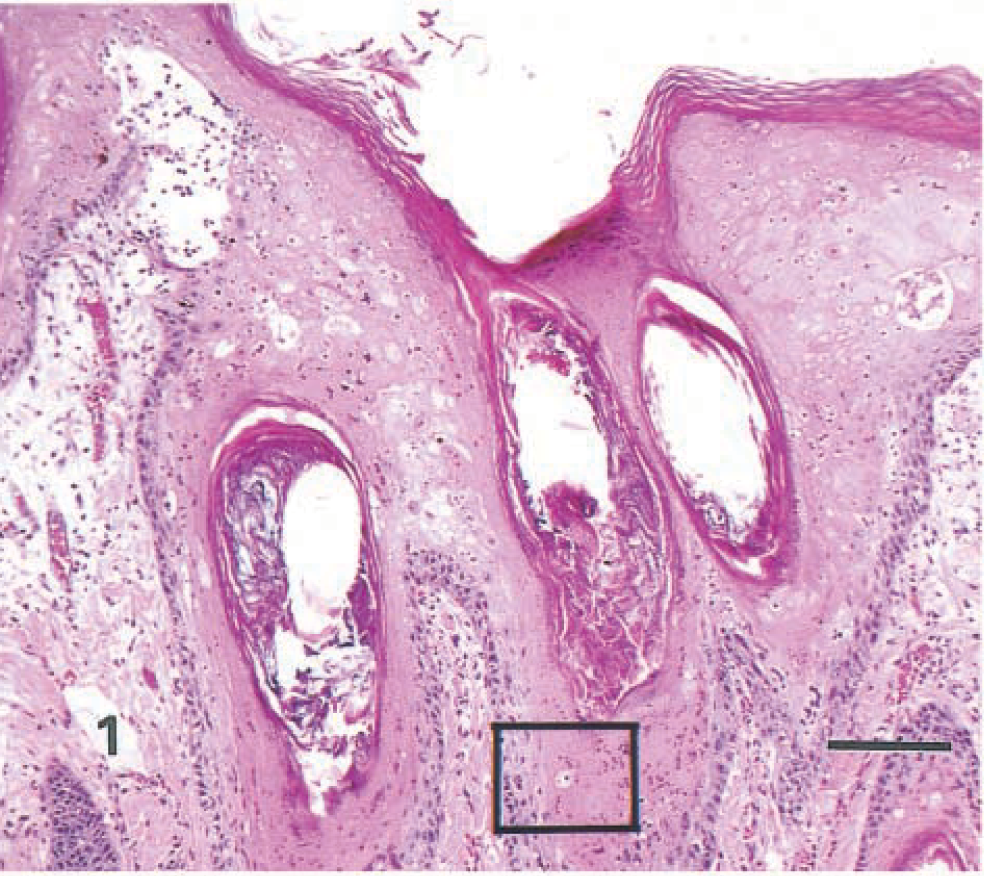
Haired skin; seal No. 1. Epidermis and follicular infundibula are thickened by a deeply eosinophilic layer of hyperkeratosis, a lightly eosinophilic, sparsely cellular region (syncytial zone) and a basophilic layer of hyperplastic basal cells. Area in box is shown at higher magnification in Fig. 3. HE. Bar = 125 µm.
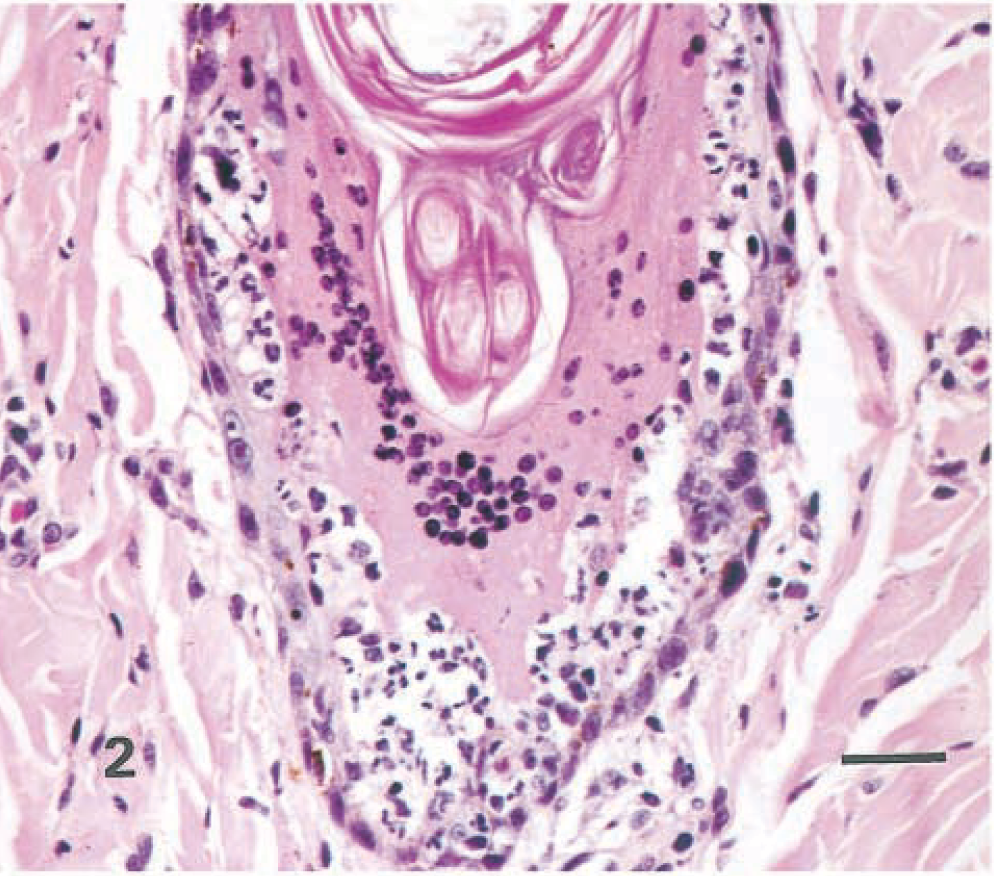
Haired skin, hair follicle; seal No. 2. Syncytial zone separates luminal hyperkeratosis and hyperplastic basal cells. Note clusters of neutrophils within the follicular epithelium. HE. Bar = 25 µm.
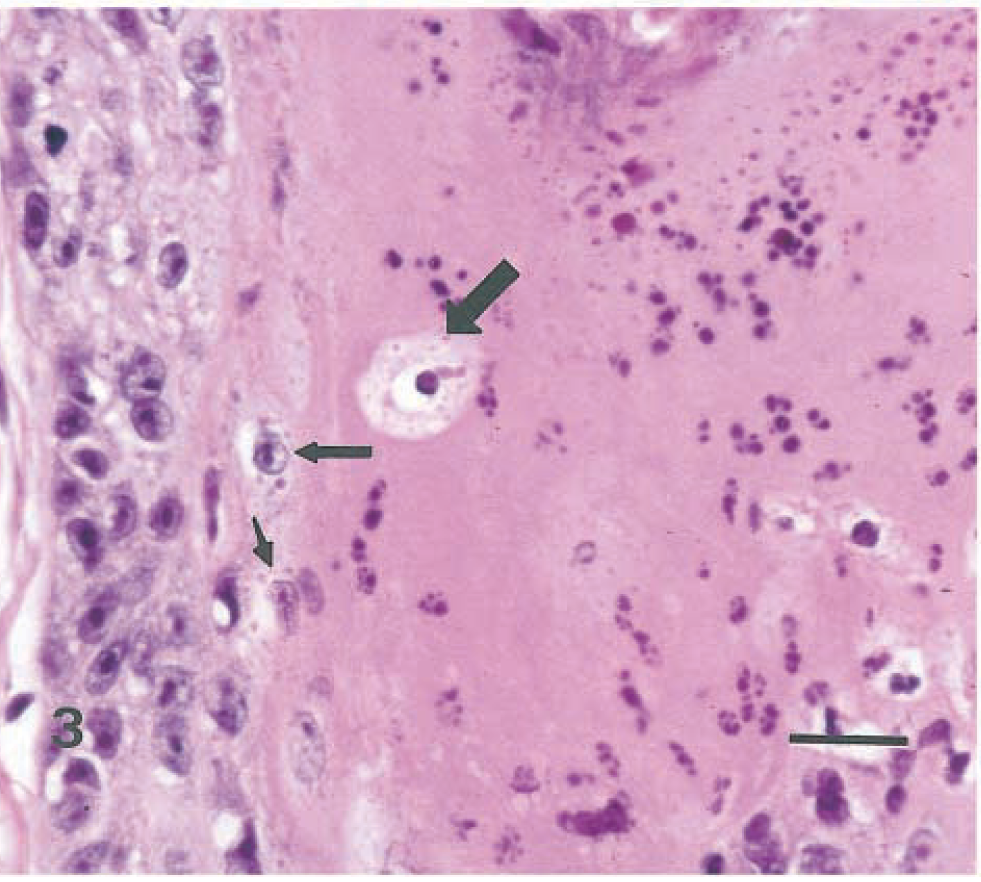
Haired skin, follicular wall; seal No. 1. Higher magnification of area in box in Fig. 1. Hyperplastic basal cells are at left. Syncytial zone contains pyknotic and fragmented nuclei. Note eosinophilic intracytoplasmic inclusion bodies in apparently viable (small arrows) and necrotic (large arrow) epithelial cells. HE. Bar = 20 µm.
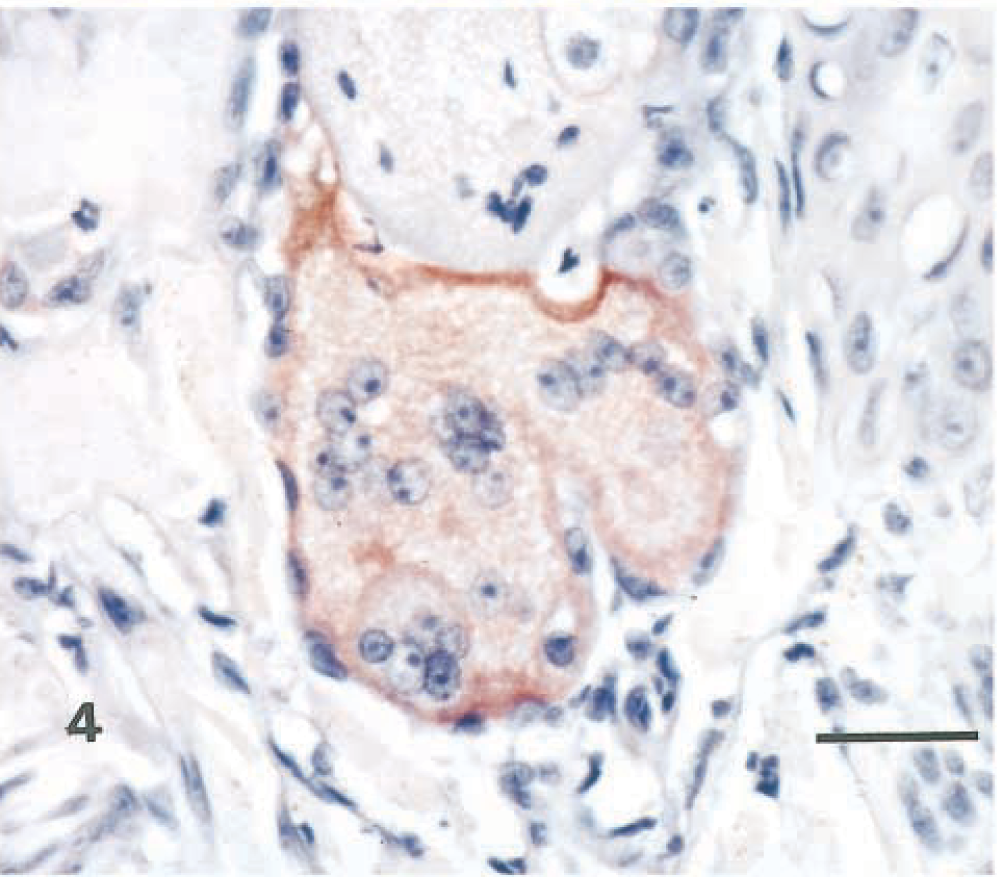
Haired skin, sebaceous gland; seal No. 1. Sebaceous syncytia with positive immunohistochemical staining for morbillivirus antigen. Avidin-biotin-peroxidase complex method, Harris' hematoxylin counterstain. Bar = 25 µm.
The hooded seal also had lymphoid depletion of spleen and lymph nodes and suppurative bronchopneumonia from which Bordetella bronchiseptica was cultured. Round 4–6-µm-diameter, homogeneous, eosinophilic intracytoplasmic inclusion bodies were present in bronchiolar epithelium. The harp seal had lymphoid depletion of lymph nodes but no pneumonia.
Morbillivirus antigen was demonstrated in the skin lesions of both seals by immunohistochemistry (Fig. 4). PDV was detected in the skin of both seals by RT-PCR and the PDV-specific probe using the Southern blot technique.
The combination of highly characteristic histopathologic findings such as syncytial cells and eosinophilic intracytoplasmic inclusion bodies, positive immunohistochemical staining for morbilliviral antigen, and detection of PDV by RT-PCR and PDV-specific probe provides strong evidence that PDV was the cause of the skin lesions. This is the first report of morbilliviral dermatitis in marine mammals. Similar lesions were not observed in harbor seals (Phoca vitulina) or gray seals (Halichoerus grypus) that died during the Northwestern Europe morbillivirus epizootic of 1988 (S. Kennedy, personal communication). Hyperkeratotic skin lesions with syncytial cells and inclusion bodies are well recognized in other morbilliviral diseases, such as canine distemper in dogs 7 and measles in primates, 2 but the syncytial zone observed in these seals is not typically present. However, red pandas (Ailurus fulgens) with canine distemper may have skin lesions remarkably similar to those present in the seals (R. J. Montali, personal communication).
The findings in these seals have practical implications for those involved in medical rehabilitation of seals. Harp and hooded seals with alopecia and hyperkeratotic dermatitis should be considered potentially infected with PDV. PDV-infected harp seals may not have respiratory disease. Skin biopsy has the potential to provide rapid, definitive diagnosis of morbilliviral disease in some harp and hooded seals. Additional research is needed to determine how frequently this distinctive skin lesion is present in morbillivirus-infected seals.
Footnotes
Acknowledgements
This study was partially funded by the National Marine Fisheries Service. We thank R.-A. V. Ferris for photomicroscopy. T. P. Lipscomb and M. G. Mense are lieutenant colonels in the US Army. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense.
