Abstract
Freshwater mussels are one of the most endangered groups of animals in Indiana, with nearly half of the native species either extirpated or listed as “state endangered” or of “special concern.” Nationally, numerous freshwater mussel species are considered threatened. Freshwater mussel diseases are not well understood and few published accounts of freshwater mussel diseases with detailed histological descriptions exist. Mass mortality events within mussel populations are increasingly recognized, often with undetermined etiology. Our objective was to determine baseline histopathology in free-living populations of freshwater mussels. One-hundred twenty individual mussels representing 2 species—plain pocketbook (Lampsilis cardium) and fatmucket (Lampsilis siliquoidea)—were collected from 3 different locations within the Wildcat Creek watershed in central Indiana during June and July 2019. A cross-section through the visceral mass was obtained and immersed in 10% neutral-buffered formalin, with routine processing and hematoxylin and eosin staining. Branchial acariasis occurred in 43/60 fatmuckets and 22/60 plain pocketbooks. Infection with a bucephalid trematode was recognized in 18/60 fatmuckets, while infection of the gonadal duct with an unidentified trematode species was identified in 4/60 fatmuckets and 18/59 plain pocketbooks. Additional changes associated with unidentified trematodes, bacteria, fungi or oomycetes, and ciliates were observed. Other miscellaneous changes included mineralization, neuronal lipofuscinosis, and gonadal atrophy/atresia. A range of histological changes were observed. These changes likely represented background lesions: incidental findings, spontaneous infectious or endosymbiotic conditions, or normal physiological changes that routinely occur in free-living wild populations. Awareness of baseline lesions should inform future diagnostic investigations of mussel mortality events.
Native freshwater mussels (Order: Unionida) are known to be highly imperiled in North America.21,28,35 As an example, over half of the nearly 80 native species known to historically inhabit the waters of Indiana are already extirpated or currently listed as a species of greatest conservation need. 16 While the underlying causes often seem obvious—extreme habitat loss/degradation, competition with or direct predation by non-native species, major water quality issues, and overharvest—in other situations, the reasons are far more inconspicuous or enigmatic.11,13 Mass mortality events, or “die-offs,” presenting threats to freshwater mussel populations are increasingly recognized throughout their range, with etiologies often undetermined. Microbiota and pathogens have been understudied and may play an important role. 34 For example, a novel densovirus was recently determined to be epidemiologically linked to morbidity in a die-off of freshwater mussels in the Clinch River in Tennessee and Virginia, suggesting viral infections could be a direct cause, indirect consequence, or contributing factor in mass mortality events. 29 As another example, the microbial composition of the digestive gland was found to shift dramatically when moving Pleurobema cordatum to artificial rearing conditions, a possible mechanism to explain wasting and mortality in altered environmental conditions. 2 More work characterizing infectious diseases and commensal microbiota of freshwater mussels, as well as other causes of mortality, is needed to better understand possible reasons for decline.
Histopathology can be an effective tool for health assessment and study of disease in mollusk populations. Marine bivalves are more well studied than their freshwater counterparts, with several recent studies incorporating histopathologic endpoints for population health assessment. Histopathology was used as the primary endpoint in a broad health assessment of the marine mussel Mytilus chilensis in southern Chile, with the authors concluding that the population was relatively healthy while also characterizing infection with various pathogens and parasites as well as the presence of degenerative conditions and neoplasia. 19 Similarly, histology was used to identify several baseline parasitic infections and neoplastic conditions in razor clams (Ensis arcuatus) in coastal Spain. 10 Beyond descriptive histology studies, quantitative histomorphometry endpoints have also been used to determine health status in different populations of Mytilus sp. in the Tamar estuary, United Kingdom, with correlation to level of pollutants likely present at each location. 4 When used in combination with other techniques, histology has also been shown to be a useful component of multidisciplinary studies assessing marine mussel health. 7 In one study, histology was crucial in identifying a reportable infectious disease—marteiliosis—as well as reproductive and degenerative disorders likely indicating poor health status of the sampled populations. Histological findings nicely supported molecular data indicating stress in the study populations. 7
Fewer broad, population-based health assessments of freshwater mussels with associated histological findings have been published. One relevant study was published in 2001 and evaluated the health of the eastern elliptio (Elliptio complanata) in North Carolina, with hopes of identifying risks to a sympatric species, the Carolina heelsplitter (Lasmigona decorata). 9 The study incorporated bacterial culture in addition to histology, with important findings including digestive gland atrophy and inflammation as well as multiple parasites/endosymbionts. In these broad, population-based health assessments, potentially novel findings may surface, as the study may be among the relatively few published works in which a large number of a particular species was examined histologically. 9 Histological examination alone, or especially as part of a multidisciplinary approach to mussel health, can yield important information about infectious disease, parasitic infestation, and degenerative changes.
The concept of background lesions has been most thoroughly explored in the discipline of toxicological pathology. Specifically, background lesions are important in the context of differentiating spontaneous lesions from those induced by test articles. 20 Thus, entire textbooks have been written, detailing the background findings in those species used in the pharmaceutical industry. Varied definitions of what constitutes a background lesion have been formulated and include incidental or individual variations of normal, a spontaneous change in tissue morphology outside normal variation, physiological or hormonal changes, expected aging changes, and even normal histological findings unique to a particular species.20,24 Recent research trends have seen the incorporation of less-traditional laboratory animals as models, including swine, sheep, and goats, sourced from less biosecure sources. 14 Thus, some investigators have broadened the scope of background lesions to include those spontaneous infectious conditions that commonly affect livestock species. 14 By the broadest definition, in the research realm, background lesions can be understood to be any changes not associated with experimental manipulation. In the context of wild populations, specifically freshwater mussels experiencing mass mortality events, the broad concept of background lesions can be useful in that these changes may be considered as any lesion not directly associated with a mass mortality event. Undoubtedly these mortality events can be complex, with possible underlying conditions that prime a population to experience mortality. However, the first step to unraveling these complex events may very well be understanding what the baseline is and what conditions affect free-living populations of mussels not undergoing mortality events.
The objective of the current study was to determine baseline histological findings in free-living populations of freshwater mussels in Indiana. This work was part of a larger study designed to use the same specimens to determine microbial populations, including viral, bacterial, and fungal organisms; presence of parasitism; antibiotic resistance patterns of cultured bacteria; hemolymph and soft tissue analyte levels, including glycogen, stable isotopes, and contaminants; and water and habitat assessments. While the health assessment project included several components, histopathologic findings will be the focus of this article.
Materials and Methods
Study Sites
All mussels were collected from 3 sites within the Wildcat Creek drainage in west-central Indiana in June and July of 2019. The watershed generally flows in a westerly direction, entering the Wabash River upstream of West Lafayette, IN, in Tippecanoe County. Total watershed size is 805 miles2 (2085 km2). Location 1 (Wildcat Creek) was located in Carroll County, between the Adams Mill dam and CR 50 East bridge (40.484836/–86.50501), and animals were collected here on June 18, 2019. Total drainage area at the site is 373 miles2 (966 km2). While the land use is dominated by cultivated crops (79.6%), developed lands are also present at 13.2%, which includes the city of Kokomo, Howard County, about 48 km upstream of the site. Location 2 (Kilmore Creek) was located in Clinton County, near the N. Hamilton Road bridge (40.335962/–86.616708), and animals were collected here on July 2, 2019. Total drainage area at the site is 77 miles2 (199 km2) and is within the South Fork Wildcat Creek watershed, and the land use is dominated by cultivated crops (88.3%). Location 3 (Mud Creek) was located in Tipton County, downstream of the State Road 213 bridge (40.405322/–85.951402), and animals were collected here on July 9, 2019. Total drainage area at the site is 74.5 miles2 (193 km2) and is within the Wildcat Creek site’s watershed, but is situated about 32 km upstream of Kokomo. The land use is dominated by cultivated crops (90.7%). No recent or historic mass mortality events involving mussels have been recorded in these streams. No active surveillance for mussel mortality events exists at these locations.
Animals, Sample Collection, and Sample Processing
Twenty mussels from each of 2 species, plain pocketbook (Lampsilis cardium) and fatmucket (Lampsilis siliquoidea), were collected from each of the 3 previously described locations within the Wildcat Creek watershed in central Indiana between June 18 and July 9, 2019 (120 total individuals). Both species included in this study are native to Indiana and widely distributed throughout the Midwestern United States. Both species have no current conservation concerns. All mussels were collected using visual and tactile searches (hand-grabbing or toe-picking), depending on water clarity. After obtaining the weight, length, and a hemolymph sample from each mussel, the shell was opened and a visual evaluation of each specimen was performed, with the presence of any gross lesions or macroscopic parasites recorded. Using a sterile scalpel or scissors, a piece of gill tissue and a cross-section of the visceral mass, intended to incorporate portions of kidney, digestive gland, gonad, foot, and mantle, were excised and placed immediately into 10% neutral-buffered formalin at approximately a 1:10 tissue to formalin ratio. Duration of fixation prior to trimming varied, but was at least 48 hours in all cases. Fixed specimens were trimmed appropriately, with the goal of encompassing at least gills, mantle, foot, digestive tissue, and reproductive tissue, and processed routinely for histology. Sections were stained with hematoxylin and eosin and, in some cases, periodic acid-Schiff, Von Kossa, and/or Brown and Hopps, and examined by a board-certified veterinary pathologist (G.N.B.).
Sex and Reproductive Status Determination
Sex was determined based on the presence of ovarian or spermatogenic tissue. Hermaphroditic individuals were categorized as being “male-female hermaphrodites,” in which ovarian and spermatogenic tissue were roughly equal; “male hermaphrodites,” in which spermatogenic elements comprised the majority of the visible gonad; and “female hermaphrodites,” in which ovarian tissue comprised the majority of the visible gonad. Mussels were determined to be gravid based on the presence of glochidia within gills.
The data analyzed in this study are not available as supplemental materials.
Results
Of the 120 individual animals sampled, the visceral mass was not available for review in 1 plain pocketbook from location 3. Gills were available for review in all individuals. Clinical signs associated with severe disease, such as gaping, weakness in closing valves, or laying on substrate, were not observed in any of the individuals collected. Significant gross lesions were not recorded. Sex data were collected based on the histological appearance of the gonad, and reproductive status was determined based on the presence of glochidia within the gills (Table 1). Hermaphroditism was observed in a subset of both Lampsilis sp. examined (Fig. 1a, b).
Sex data and reproductive status from the study population.
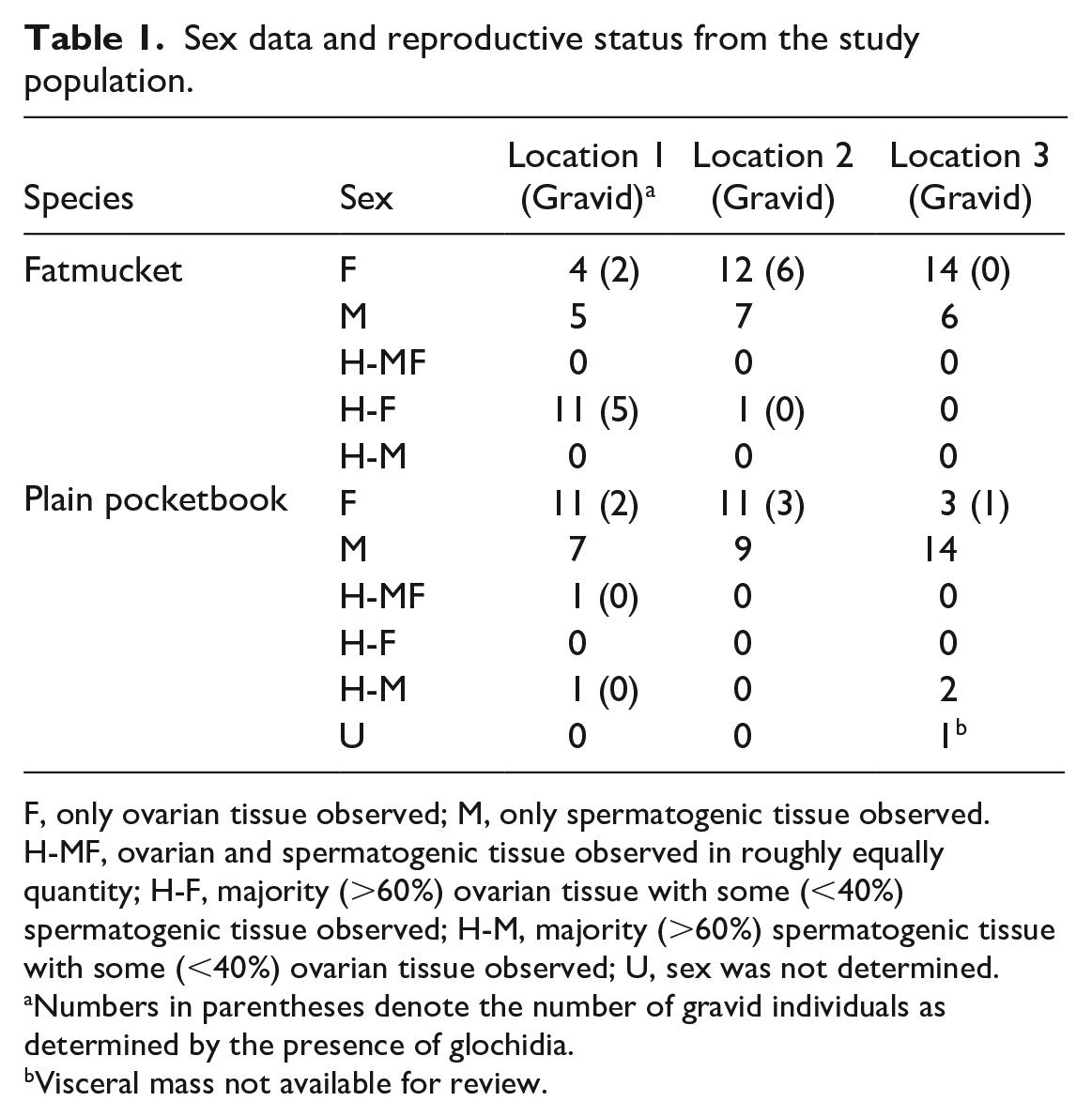
F, only ovarian tissue observed; M, only spermatogenic tissue observed. H-MF, ovarian and spermatogenic tissue observed in roughly equally quantity; H-F, majority (>60%) ovarian tissue with some (<40%) spermatogenic tissue observed; H-M, majority (>60%) spermatogenic tissue with some (<40%) ovarian tissue observed; U, sex was not determined.
Numbers in parentheses denote the number of gravid individuals as determined by the presence of glochidia.
Visceral mass not available for review.
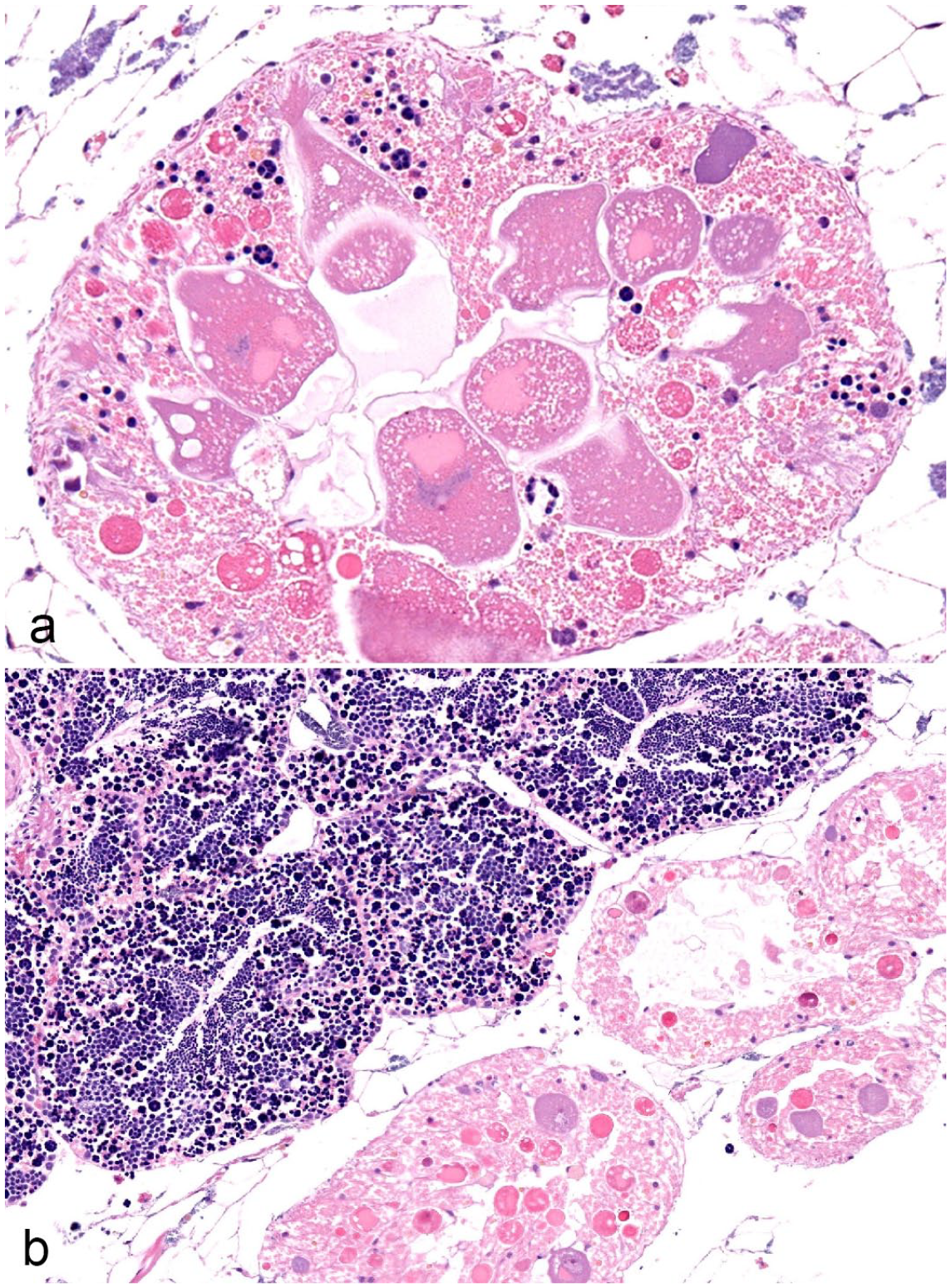
Hermaphroditism in native freshwater mussels. Hematoxylin and eosin. (a) Female gonad with small numbers of spermatogenic elements. Fatmucket (Lampsilis siliquoidea), location 1. (b) Male gonad intermixed with small numbers of ovarian acini. Male and female elements remained separate in this individual, as compared with the individual in (a). Plain pocketbook (Lampsilis cardium), location 3.
Parasitism
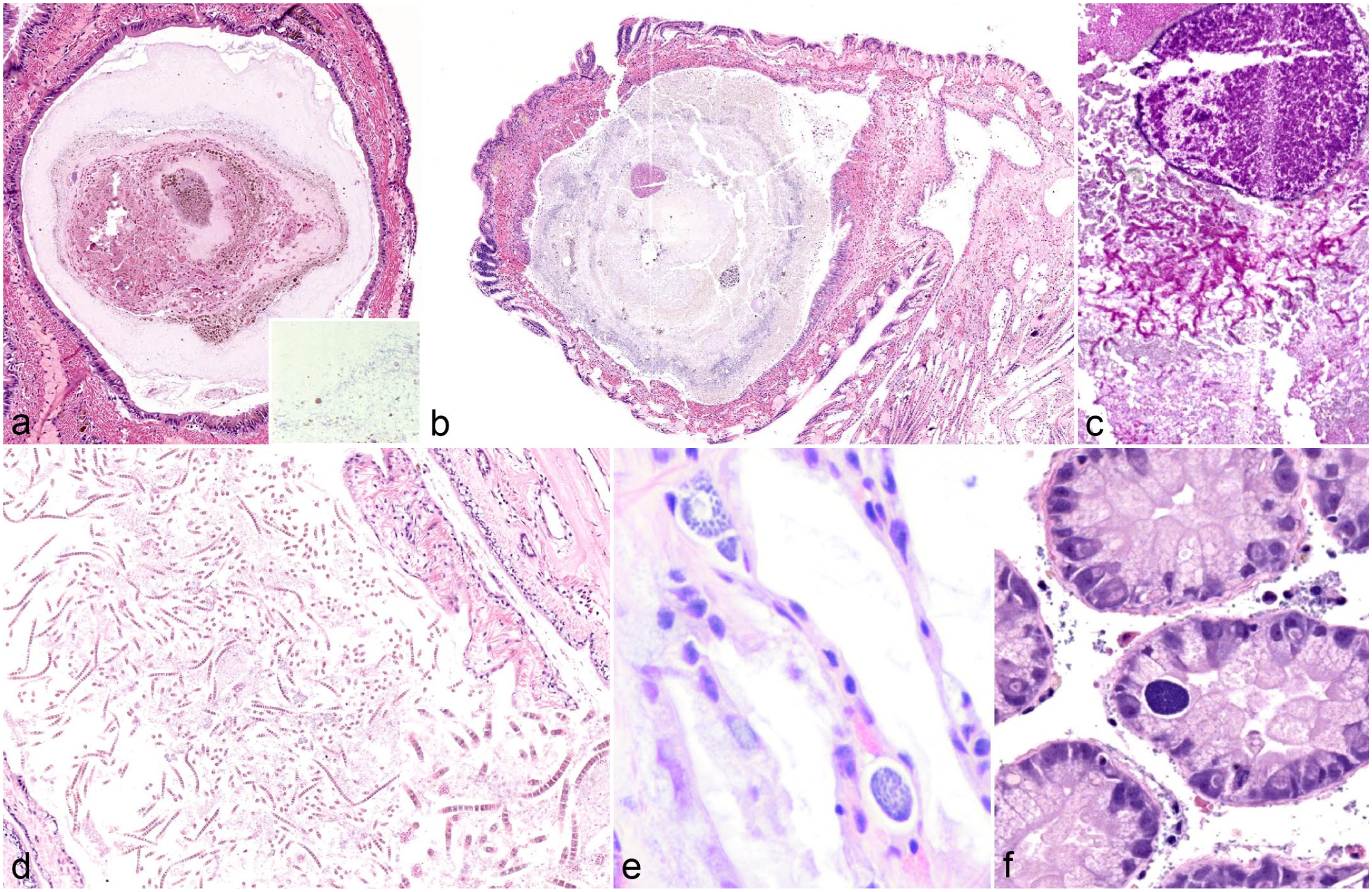
Infection with various parasitic organisms was the most common finding throughout the study population. Five fatmuckets collected from location 2 contained mites free within the mantle cavity that were collected in the field and later identified as Unionicola sp. by the parasitology laboratory at the Animal Disease Diagnostic Laboratory. During histological examination, 43 of 60 fatmuckets had intact mites, mite fragments, and/or mite ova embedded within the gills. Of 60 plain pocketbooks, 22 were similarly affected. Mites, mite ova, and mite fragments were also observed embedded within the mantle or integument of mussels, with 19 of 60 fatmuckets and 13 of 59 plain pocketbooks affected. Intact mites and ova elicited little hemocyte infiltrate in gills or mantle (Fig. 2a). Mite fragments or ova fragments in either location were surrounded by granular and agranular hemocytes and, in some cases, fibrosis (Fig. 2a–e). Numbers of mites or mite-associated material per section were relatively low in most individuals, ranging from 3 or 4 mites to no more than 10. The notable exception was a single fatmucket with a mass of mite ova and mites embedded within the integument (Fig. 2d, e).
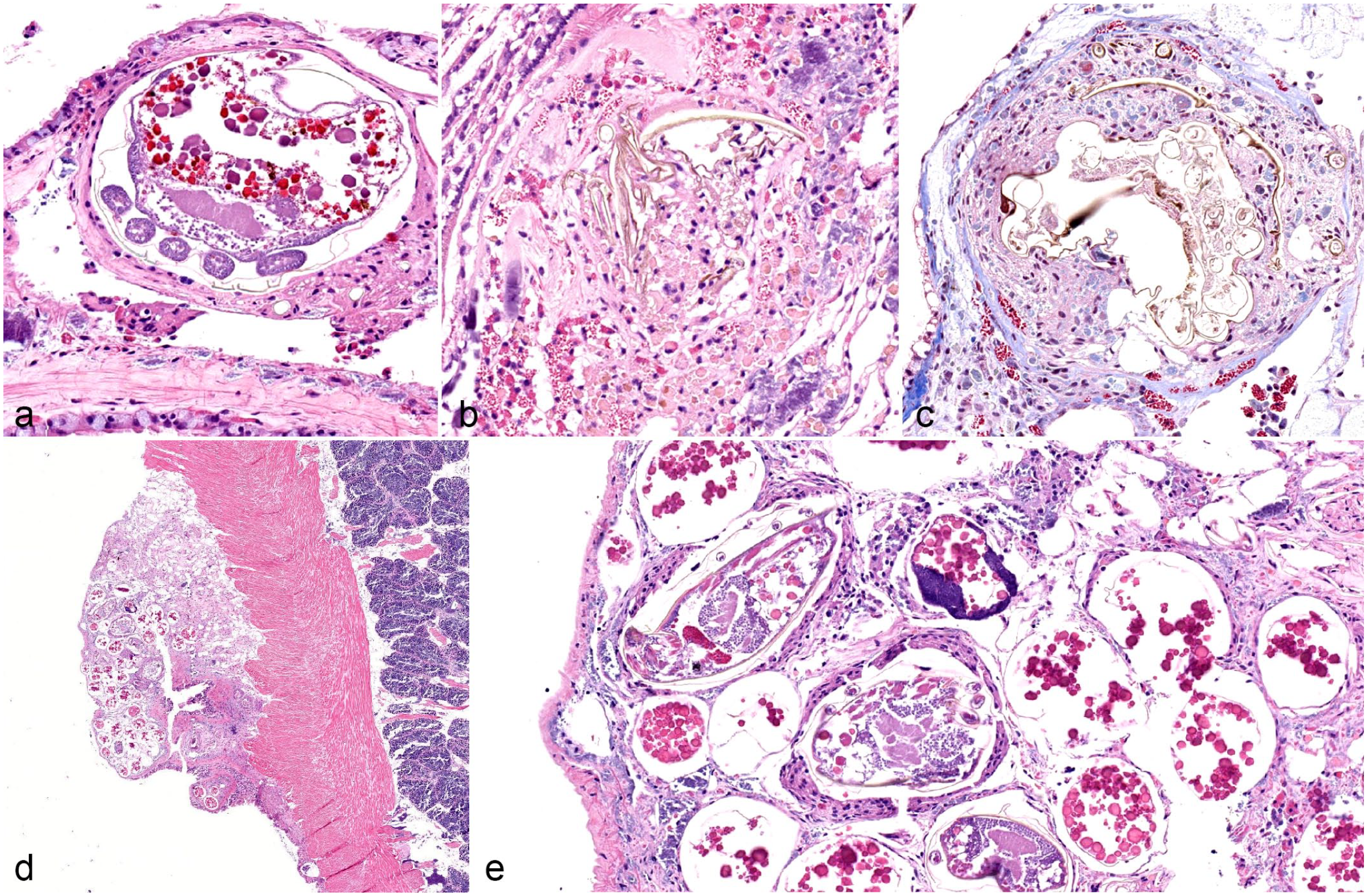
Acariasis in native freshwater mussels. (a) Viable unionicolid mite in the gills of a fatmucket (Lampsilis siliquoidea), location 1. Note the mild encapsulation reaction surrounding the mite. Hematoxylin and eosin (HE). (b) Refractile remnants of unionicolid mite surrounded by hemocytes in the gills of a plain pocketbook (Lampsilis cardium), location 1. HE. (c) Refractile remnants of unionicolid mite surrounded by hemocytes and early fibrosis in the gills of a plain pocketbook (L. cardium), location 1. Masson’s trichrome. The structures in (b) and (c) are definitively identified as mite remnants and differentiated from other parasites by the refractile nature of the foreign material. (d) Mass of unionicolid mite ova and larvae or nymphs protruding from the integument of a fatmucket (Lampsilis siliquoidea), location 2. HE. (e) Higher magnification of various life stages of unionicolid mites in the integument of a fatmucket (Lampsilis siliquoidea), location 2. Some hemocytes and fibrosis surround mite larvae or nymphs and ova. HE.
Trematode infection was the next most common finding in the Lampsilis sp., with 4 types or patterns of trematode infection recognized. First, in fatmuckets, 18 of 60 animals contained sporocysts filled with trematode cercariae that exhibited bifurcated tails (Fig. 3a). The presence of forked tails in larval forms identified these organisms as digenean trematodes in the family Bucephalidae. Bucephalid infections were extensive in some individuals, occupying most of the organs of the visceral mass, notably the gonads (Fig. 3b). Usually, trematode sporocysts did not elicit any reaction; however, in some individuals, sporocysts had ruptured and fragments of cercariae were surrounded by hemocyte infiltration (Fig. 3c). Bucephalid infection was not recognized in plain pocketbooks.
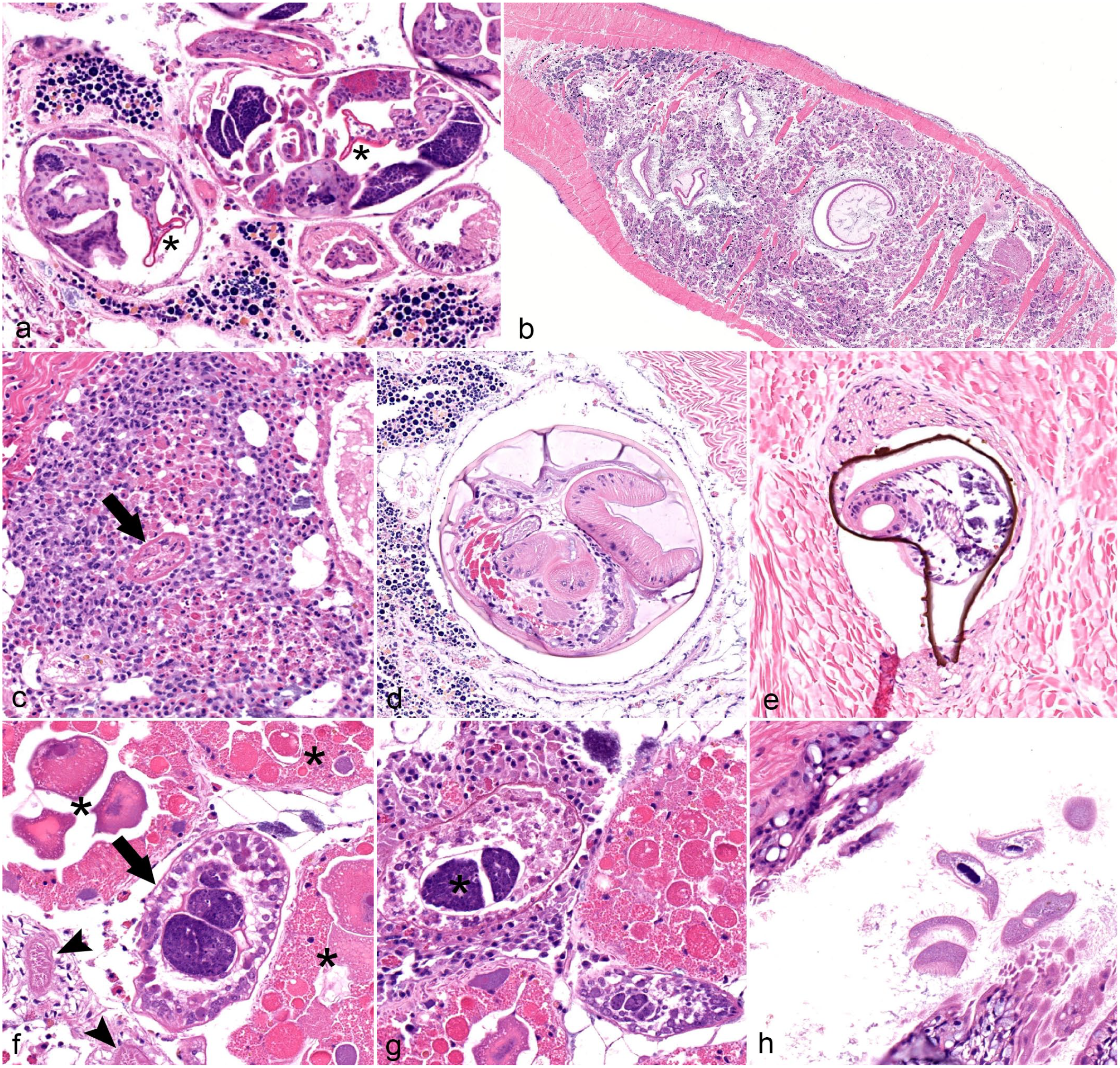
Parasitism in native freshwater mussels. Hematoxylin and eosin. (a) Buchephalid trematode infection in a fatmucket (Lampsilis siliquoidea), location 1. Multiple trematode sporocysts are present in the image, each containing multiple cercariae. Two examples of the bucephalid bifurcated tail are denoted with asterisks. Sporocysts are flanked by spermatogenic tissue. (b) Bucephalid trematode infection in a fatmucket (L. siliquoidea), location 1. Trematode sporocysts (dark eosinophilic elements) are throughout the visceral mass, displacing and replacing almost all normal tissue. (c) Ruptured trematode sporocysts and free cercariae incite extensive hemocyte infiltration within the visceral mass of a fatmucket (L. siliquoidea), location 2. A degenerate bucephalid cercaria (denoted by an arrow) is surrounded by both granulocytic and agranulocytic hemocytes. (d) Gonadal duct that is dilated and filled with an unidentified trematode cercaria. Parasite sections include an outer cyst, tegument with ridges, paired ceca, and a prominent sucker. More than one of these cysts is in each section, giving the indication of a branching sporocyst throughout the gonadal duct system. Plain pocketbook (Lampsilis cardium), location 1. (e) Mesocercariae or metacercariae of an unidentified trematode that is encysted within the foot muscle of a plain pocketbook (L. cardium), location 1. An encapsulation-type inflammatory response partially surrounds the refractile outer layer. Refractile outer layer is slightly out of place, indicating microtome drag when encountering an object of differing density than surrounding tissue (suspected mineral). (f) Unidentified trematode cercaria within a sporocyst (denoted by arrow) located in hemolymph sinus of a plain pocketbook (L. cardium), location 2. The trematode cercaria is located between ovarian tissue (upper left, right; asterisks) and gonadal ducts (lower left, arrowheads). (g) Unidentified trematode cercariae (asterisk) from a ruptured sporocyst. Note surrounding hemocyte infiltration. Plain pocketbook (L. cardium), location 1. (h) Multiple ciliates, measuring about 100 µm in length and with a prominent ovoid macronucleus are in the water tubes of the marsupial gill in a fatmucket (L. siliquoidea), location 1. There is no apparent hemocyte infiltration in adjacent tissue.
The second pattern of trematode parasitism involved an unidentified trematode species that was fully contained within a cyst-like structure and was present within gonadal ducts of 4 of 60 fatmuckets and 18 of 59 plain pocketbooks (Fig. 3d). These structures were thought to represent larval forms (putative cercariae) of digenean trematodes, but a specific taxonomy was not identified. Muscular suckers, intestines, and suspected reproductive structures were recognized (Fig. 3d).
The third type of trematode parasitism included multiple larval trematodes contained within a suspected mineralized outer coating that were embedded within the muscle of 1 fatmucket and the muscle or visceral mass of 26 plain pocketbooks (Fig. 3e). These structures were suspected metacercariae or mesocercariae of an unknown trematode species, or multiple species. Muscular suckers identified these organisms as trematodes. Many of these structures were surrounded by mild hemocytic encapsulation (Fig. 3e).
A fourth type of trematode colonization was recognized in 1 fatmucket and 5 plain pocketbooks. In this type, trematode sporocysts with multiple trematode cercariae were present within hemolymph sinuses of the gills and throughout the visceral mass (Fig. 3f). Internal structures were rudimentary and not easily identifiable, although no bifurcated tails compatible with bucephalid trematodes were observed. As in the case of bucephalid trematodes in fatmuckets, granular and agranular hemocytic infiltration was recognized in some cases in which the sporocysts had likely ruptured (Fig. 3g).
Oval-shaped ciliates with a prominent macronucleus were identified within spaces throughout the gills in 9 of 60 fatmuckets and 1 of 60 plain pocketbooks (Fig. 3h). Ciliates were not associated with inflammation or tissue destruction and were not recognized within host tissue.
Lesions Associated With Bacteria, Fungi or Oomycetes, Rickettsia-Like or Chlamydia-Like Organisms, and Cyanobacteria
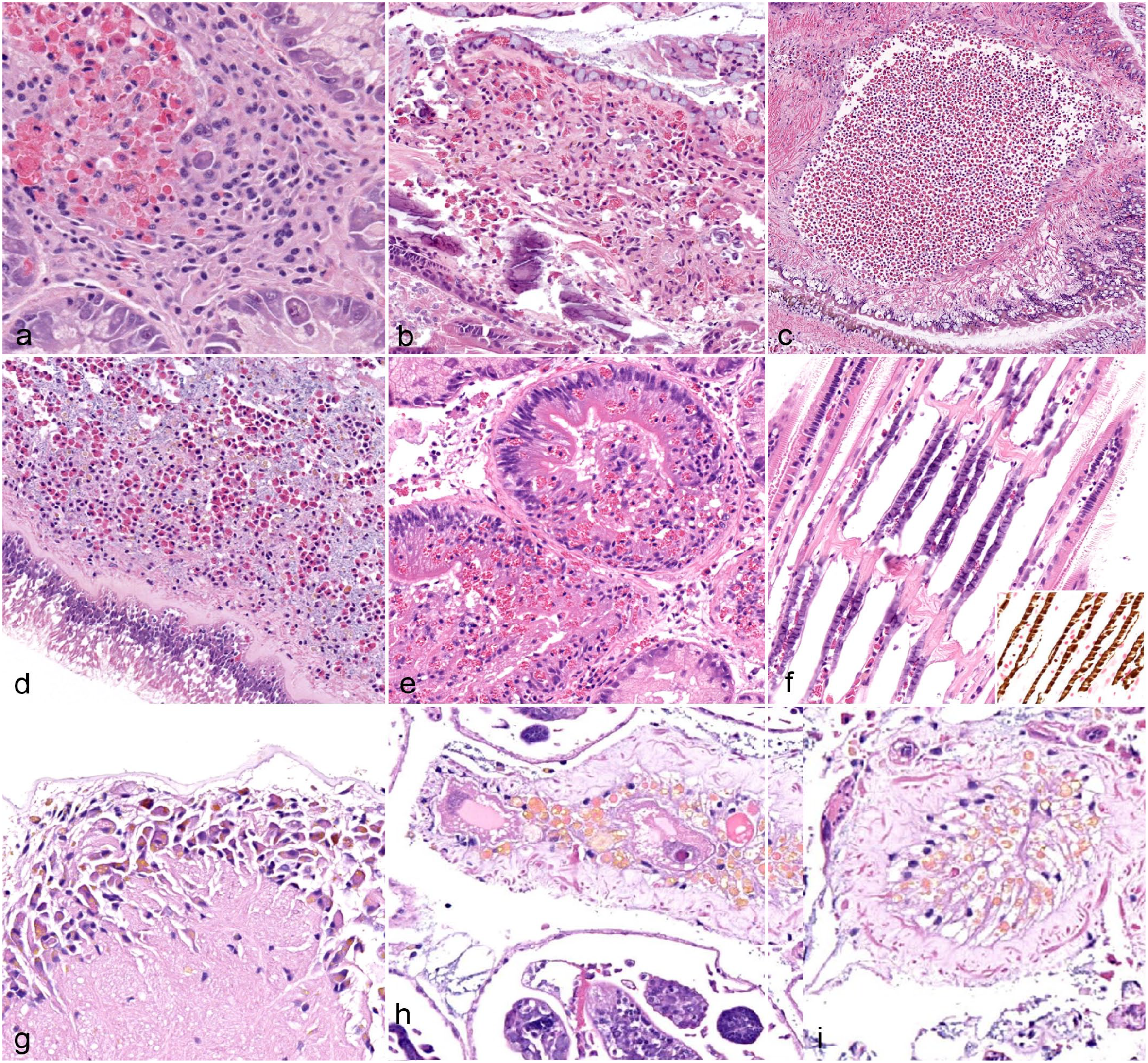
Lesions associated with organisms other than mites or trematodes were rare, and usually only observed in a single individual. Bacterial infection of the gills was observed in a single fatmucket. In this case, a single water tube was dilated and filled with an aggregate of hemocytes and debris (Fig. 4a). Surrounding tissue was mostly devoid of inflammation, even though hemocytes were a prominent feature of the intratubular mass. Mats of thin, filamentous bacteria were present within the debris (Fig. 4a, inset). With a tissue Gram stain, organisms were best characterized as gram negative.
Prokaryotic and single-celled or otherwise simple eukaryotic infections or colonization in native freshwater mussels. (a) Bacterial branchitis in a fatmucket (Lampsilis siliquoidea), location 1. A single water tube is dilated and filled with hemocytes and debris. Surrounding tissue is mostly devoid of hemocytic infiltration. Inset: masses of thin, filamentous bacteria are present within the debris. Hematoxylin and eosin (HE). (b) The distal aspect of a single water tube is dilated and filled with debris. Surrounding gill filament tissue is infiltrated by hemocytes. Plain pocketbook (Lampsilis cardium), location 1. HE. (c) A mat of Periodic acid-Schiff (PAS)–positive hyphae that are septate and irregularly branching is within the debris seen in (b). A round mass of unidentified PAS-positive material suggestive of spores is present (upper right). PAS. (d) Multiple marsupial water tubes are dilated and filled with mats of organisms forming chains composed of small, 5–7 µm, pigmented square cells suggestive of cyanobacteria. Hemocyte infiltration is not appreciated in the surrounding tissue. Inset: higher magnification of chains of square cells suggestive of cyanobacteria. Fatmucket (L. siliquoidea), location 3. HE. (e) Two, round to ovoid cysts composed of 1–2 µm, basophilic structures suspected to be Rickettsia-like or Chlamydia-like organisms are embedded within gill filament of a fatmucket (L. siliquoidea), location 2. No surrounding tissue reaction is observed. HE. (f) A single round to ovoid cyst composed of numerous, deeply basophilic structures suspected to be Rickettsia-like or Chlamydia-like organisms within the cytoplasm of epithelial cells of a digestive gland acinus in a fatmucket (L. siliquoidea), location 3. No surrounding tissue reaction was noted. HE.
Branchitis associated with fungus- or oomycete-like organisms was observed in a single plain pocketbook. As in the case of the bacterial infection, a single water tube was dilated and filled with debris, hemocytes, and organisms (Fig. 4b). Unlike the bacterial infection, though, surrounding gill tissue was heavily infiltrated with granulocytic hemocytes. Organisms were septate, irregularly branching, and periodic acid-Schiff reaction positive (Fig. 4c). A large, round cyst containing individual periodic acid Schiff reaction–positive spore-like structures was also present within the debris, adjacent to hyphae (Fig. 4c).
A mat of organisms compatible with cyanobacteria was present in the spaces surrounding the gills in 1 fatmucket. Cyanobacteria were composed of square cells that formed long, filamentous chains (Fig. 4d). No inflammation or tissue destruction was associated with the presence of cyanobacteria in the examined section.
Round to ovoid structures composed of multiple, regularly shaped, round to ovoid components were observed within the gill tissue of 1 fatmucket and the digestive gland of 5 fatmuckets (Fig. 4e, f). These structures were not associated with inflammation or degeneration, and were morphologically compatible with organisms traditionally reported as Rickettsia-like or Chlamydia-like organisms in marine bivalves.
Inflammatory Lesions Without Visible Etiologic Agents
Hemocyte nodulation was present in the visceral mass in 4 fatmuckets and 8 plain pocketbooks. The composition of hemocytes within these nodules varied, but nodules could include enlarged hemocytes with abundant, eosinophilic cytoplasm and clear vacuoles; hemocytes with cytoplasmic, eosinophilic granules; or agranulocytic hemocytes with scant cytoplasm (Fig. 5a). These hemocytic nodules did not contain identifiable etiologic agents when examined with hematoxylin and eosin or additional histochemical stains (tissue Gram stain, acid fast stain, or periodic acid-Schiff reaction). Hemocytic nodulation occupied a relatively small percentage of overall tissue mass—less than 5% of the total tissue area in all individuals.
Inflammatory lesions not associated infectious agents and miscellaneous changes in native freshwater mussels. (a) A nodular aggregate of both granulocytic and nongranulocytic hemocytes within the interstitium of the digestive gland of a plain pocketbook (Lampsilis cardium), location 1. Histochemical stains for infectious agents (not pictured) failed to yield an etiology. Hematoxylin and eosin (HE). (b) Gill filaments are moderately infiltrated by various hemocytes in a plain pocketbook (L. cardium), location 1. No etiologic agents are associated with inflammation; unionicolid mites were not observed in this individual. HE. (c) A hemolymph sinus within a gill filament is markedly dilated and filled with granulocytic hemocytes. Plain pocketbook (L. cardium), location 2. HE. (d) Increased numbers of hemocytes are within hemolymph sinuses surrounding the intestine in a plain pocketbook (L. cardium), location 1. HE. (e) Large-caliber ducts within the digestive gland are infiltrated by granulocytic hemocytes. Ductal epithelium is partially disrupted. Plain pocketbook (L. cardium), location 3. HE. (f) Interstitium of gill filaments is multifocally mineralized in a plain pocketbook (L. cardium), location 1. HE. Inset: basophilic material in gill filaments stains brown with a Von Kossa stain, indicating calcium deposition. Von Kossa. (g) Neuronal soma within a ganglion contains golden brown pigment compatible with lipofusin. Fatmucket (Lampsilis siliquoidea), location 2. HE. (h) Ovarian atrophy or atresia in a fatmucket (L. siliquoidea), location 2. The ovarian acinus contains 2 mature oocytes that have extensive cytoplasmic vacuolation. The acinus contains numerous smaller cells filled with golden brown pigment compatible with lipofusin. A bucephalid trematode sporocyst is visible in the lower half of the image. HE. (i) Ovarian atrophy or atresia in a fatmucket (L. siliquoidea), location 2. Ovarian acini are devoid of mature oocytes and contained cells with intracytoplasmic lipofusin. HE.
Nonspecific hemocyte infiltration was present in the gills of 1 fatmucket and 4 plain pocketbooks. Again, the hemocyte composition varied, but usually included both granulocytic and agranulocytic hemocytes (Fig. 5b). No etiologic agents were observed. A lesion best characterized as branchial hemocytosis was observed in a single plain pocketbook. In this lesion, hemocytes were observed in abundance in dilated hemolymph sinuses of the gills, but, did not infiltrate tissue (Fig. 5c).
Hemocytes surrounded the intestine in 7 plain pocketbooks. Hemocytes were usually granulocytic and mixed with hemolymph (Fig. 5d). Thus, this change may best have been characterized as per-intestinal hemocytosis and was not considered an infiltrative-type inflammatory response. No inciting cause in or around the intestine was recognized. In contrast, in a single plain pocketbook, granulocytic hemocytes infiltrated between epithelial cells lining large-caliber ducts located within the digestive gland, leading to a diagnosis of dochitis or infiltrative-type inflammation of the digestive glands (Fig. 5e). Inflammation was severe and restricted to the ducts of the gland, sparing gland acini. In some affected ducts, epithelium was disrupted (Fig. 5e). Again, no inciting cause of inflammation was observed.
Miscellaneous Changes
Three additional changes were recognized: mineralization, neuronal lipofuscinosis, and gonadal atrophy. Mineralization was the most common change recognized in the study. Mineralization was present most commonly in the gills, with 33 of 60 fatmuckets and 39 of 60 plain pocketbooks affected with some level of branchial mineralization (Fig. 5f). Mineralization was less commonly present in other tissues, including the gonad, mantle, and visceral mass. Mineralization of the visceral mass or mantle was present in 8 of 60 fatmuckets and 4 of 59 plain pocketbooks. Gonadal mineralization was present in 1 of 60 fatmuckets and no plain pocketbooks. Mineralization was confirmed with Von Kossa stain (Fig. 5f, inset).
Neuronal soma in ganglia of 9 of 60 fatmuckets and 9 of 59 plain pocketbooks contained fine, brown, intracytoplasmic pigment interpreted as lipofuscin accumulation (Fig. 5g).
Eight of 60 fatmuckets and 2 of 59 plain pocketbooks had a constellation of histological changes to the gonadal tissue that were most compatible with atrophy or atresia. In these individuals, mature germ cells, either oocytes or spermatids, were rare or completely absent (Fig. 5h, i). Degenerative changes, such as cytoplasmic vacuolation, were observed in oocytes (Fig. 5h). Small cells with brown intracytoplasmic pigment interpreted as lipofuscin were present in high numbers within gonadal tissue (Fig. 5h, i). This change was present in 3 fatmuckets with spermatogenic tissue, 4 fatmuckets with ovarian tissue, 1 plain pocketbook with spermatogenic tissue, 1 plain pocketbook with both ovarian and spermatogenic tissue, and 1 fatmucket in which the change was severe enough to preclude histological determination of sex via examination of the visceral mass. The individual with unidentifiable gonads histologically was identified as male via shell morphology and lack of marsupia in available gill sections. All individuals with degenerative gonadal changes were confirmed to be sexually mature via shell characteristics. All affected fatmuckets had moderate to severe infestation with bucephalid trematode larvae. The 2 affected plain pocketbooks had moderate infestation with trematode larvae in the gonadal ducts.
Discussion
The objective of this study was to determine baseline histological findings in free-living populations of 2 common species of freshwater mussels, as an aid in the event of any future population decline or mortality event. The study documented a range of incidental findings, spontaneous and endosymbiotic infections, and common histological changes that, taken together, are likely best described as background lesions in stable populations of freshwater mussels in the Midwestern United States.
Sex ratios and gravid status varied between the 3 collection sites. Unionid species of mussels are typically dioecious, with a few exceptions. Interestingly, hermaphroditic individuals were found in both populations of plain pocketbook and fatmucket, with a majority of fatmuckets at location 1 displaying hermaphroditism (Table 1). A previous histological survey of a broad taxonomic range of North American freshwater mussels found that “sporadic” or “accidental” hermaphroditism was fairly common in the family Unionidae, with the phenomenon observed in 21 of 95 species. 33 The referenced work recorded 7 species within the Lampsilini tribe, referred to as the Lampsilinae subfamily in the referenced text, with “sporadic” hermaphroditism, in which a majority of gonadal tissue was classified as one sex, with a small amount belonging to the other sex. Individuals belonging to the fatmucket and plain pocketbook species were analyzed in the referenced study, but no hermaphroditic individuals were noted in either species. The author’s description of “sporadic” hermaphroditism in this reference fits well with the observations of hermaphroditism in the unionid species examined in the current study. A single plain pocketbook collected from location 1 displayed roughly equal amounts of gonadal tissue from both sexes, but evaluation of this individual was complicated by concurrent gonadal atrophy, and this observation was the outlier. While no specific reference to hermaphroditism in fatmuckets and plain pocketbooks was found in the literature, this phenomenon has been reported in related species, including one member of the Lampsilis genus, L. cariosa. 33
Gravid individuals were observed for each species at each location, with the exception of no gravid fatmuckets at location 3 (Table 1). This location also contained the lowest number of gravid plain pocketbooks, as compared with sites 1 and 2, possibly indicating less ideal conditions for spawning for all species at location 3.
Parasitism was the most common finding in the 2 native Lampsilis spp. Branchial and/or mantle acariasis was common in both species. Mites collected from the mantle cavity in 5 fatmuckets were identified as Unionicola sp., suggesting that the mites, mite ova, and mite fragments observed histologically were likely members of this genus. A larger water mite, Najadicola ingens, has also been described as a parasite/endosymbiont of freshwater mussels in North America and could be considered as a possible etiology for mite material observed histologically in this case; however, given the prevalence of Unionicola spp. (69 Unionicola spp.), Unionicola infection seems most likely. 12 The histopathology of Unionicola infections in freshwater mussels has been detailed in 2 recent publications.1,23 In the McElwain et al 23 study, mites and mite ova were observed in mantle tissues and gills, while mite remnants were present in a host of various tissues. In the Abdel-Gaber et al 1 work, mites were observed in gills, gonads, and the visceral mass. In the current work, mites or mite fragments were not recognized in locations other than the mantle or gills.
Aside from mite infections, trematodiasis was the next most common histological change noted. In this study, taxonomic identification of trematodes was limited to the evaluation of histological sections of parasites. This method of identification presents major challenges, which likely accounted for lack of specific conclusions concerning trematode classification. 9 The first type of trematode infection could be identified to the broad group known as bucephalid trematodes, given the bifurcated tail observed in some cercarial stages. 18 The bucephalid Rhipidocotyle septpapillata has been described in L. siliquoidea, and the trematode in the current study may have been this species. However, without further evidence (eg, molecular identification), the authors of the current study were unwilling to be so precise, as Rhipidocotyle papillosa is also known to infect North American unionids.12,17
Three other histological patterns associated with trematodes were described; however, a definitive taxonomic group was not assigned to these infections. In a review of literature concerning endosymbionts in freshwater mussels (specifically Appendix A of the referenced manuscript), both L. cardium and L. siliquoidea have been reported to be hosts for the aspidogaster trematodes Aspidogaster conchiola, Cotylaspis insignis, Cotylogaster occidentalis, and Cotylogasteroides barrowi. 6 Adult A. conchiola occurs in the peri-renal and pericardial spaces, adult C. insignis occurs in the gill chambers, and both C. occidentalils and C. barrow adults occur in the intestinal lumen.12,15,26,30 However, the findings in the current study were limited to immature trematodes in the gonadal duct, foot muscle, visceral mass connective tissue, and hemolymph sinuses; thus, the previously described aspidogaster species were not considered probable candidates for these infections based largely on the location of infection. Several studies have described encysted trematode cercariae in muscle and other tissues, but most authors named these infections Cercaria spp., knowing they were immature forms of another, possibly already-described species. 6 Those that have attempted speciation via histological observation include caveats about the difficulty of this method. 9 Should investigators attempting broad, baseline health assessments in mussels require precise taxonomic classification of recovered parasites, specialized expertise in parasites of freshwater mussels must be sought during the planning stages of any project. Additional techniques such as parasite dissection and wet mounts, which are unfamiliar to many researchers, may be required. In the current study, the authors decided to leave the trematodes unnamed with hopes of molecular identification at a later date.
Ciliates were observed within the gills of 9 fatmuckets and 1 plain pocketbook. Ciliates are common symbionts in freshwater mussels and are even considered ubiquitous by some authors. 12 The most likely genus in the current study was considered Conchophthirus given the histological characteristics and prevalence of the genus. Other genera that infect freshwater unionid mussels include Trichodina, Heterocinetopsis, and Mantoscyphidia; however, all other genera are reported to be smaller (70–100 µm, 40.5 µm, and 11–29 µm, respectively) than Conchophthirius (62–140 µm) and were deemed less likely. 12 A relatively newly described ciliate, Tetrahymena glochidiophila, has also been documented in fatmuckets, with documented deleterious effects to developing glochidia. 27 This species may also be considered as a possible identity for the observed ciliates, based on histological characteristics.
Other microorganisms observed in the study were rare, including one case each exhibiting bacteria, fungus-, or oomycte-like organisms, and cyanobacteria. Of these, the bacterial and fungal/oomycete infections were associated with hemocytes, while cyanobacteria growth was devoid of hemocytes. All 3 organisms were located within the gills. Unequivocal evidence that harmful bacterial infections occur in freshwater mussels is currently lacking.8,12 Given that the bacterial infection observed in the current study seemed relatively localized and lacked substantial inflammation of surrounding tissues, the observed lesion was likely subclinical. Even less has been published regarding fungi or oomycetes in freshwater mussels, with mentions coming from a group in Finland that has observed fungal hyphae within marsupial gills, a study in Michigan that found a fungal mat growing on the mantle of a single L. siliquoidea by scanning electron microscopy, and the recent description of a novel microsporidian in pheasant shell mussels.5,12,22,25 In the current study, the presence of organism was localized, but elicited inflammation in surrounding tissue. The single case of suspected cyanobacteria within the gills seems relatively straightforward as a nonclinical finding, as no inflammation or tissue destruction was noted. However, documenting these findings was considered important from a “background lesions” perspective.
Structures suggestive of Rickettsia-like or Chlamydia-like organisms were observed in the gills of a single fatmucket and the digestive gland epithelium in 5 fatmuckets. These structures were not associated with inflammation or necrosis, and further molecular interrogation of these tissues would be needed to confirm their taxonomy. However, these findings are not unprecedented, as other researchers have observed similar structures, especially within the gills and digestive gland epithelium. Chittick et al 9 described a suspected microorganism in digestive gland epithelium in 2 E. complanata that had similar characteristics to those in this study.
Hemocyte infiltrates without a clear infectious etiology were observed in several individuals. Hemocyte nodulation was in the visceral mass, as well as gill interstitium. Histochemical stains failed to identify any specific cause of inflammation in these cases. However, given the numerous individuals with parasitic infection and clear association between mite fragments or ruptured trematode sporocysts and inflammation, the possibility that a previous parasite infection was the initial cause of inflammation should be considered. Similarly, hemocytosis in gill hemolymph sinuses, as well as peri-intestinal hemolymph sinuses, was observed, but a clear cause was not determined. These accumulations of hemocytes were not thought to be infiltrative-type inflammatory reactions within tissue, but simply accumulations of hemocytes within sinuses. One of the most interesting lesions encountered throughout the study was digestive gland dochitis or infiltrative-type inflammation of the digestive gland (Fig. 5e). Granulocytic hemocytes appeared to be specifically targeting ductal structures within the digestive gland. No infectious causes were associated with this lesion. Whether an infectious cause did exist (eg, virus) or this lesion resulted from a noninfectious cause (eg, digestive enzyme stasis) is unknown. Establishing these findings as an existing baseline will help future researchers determine whether these or other inflammatory lesions warrant further investigation into possible, unidentified infectious causes of disease.
Mineralization was the most common change encountered throughout the study. Mineralization occurred in the gills as well as other tissues throughout the body. This change likely does not raise to the level of “lesion” status, as freshwater mussels are known to store calcium in gill connective tissue for use as a calcium reservoir and for possible metal detoxification. 8 Considering a broad definition of background lesions, variations on normal may be included, especially if the changes represent unique histological features of certain species.20,24 Mineralization likely falls into this category for freshwater mussels. Given that there was a range of mineralization observed in this study, with some individuals displaying little or no mineralization, pathologists may want to be aware of mineralization levels histologically, as shifts within populations could indicate underlying issues. Similarly, neuronal lipofuscinosis, as observed in the current study, likely represented a change associated with aging and, thus, a variation of normal. Lipofuscin accumulation has been shown to correlate with age and has been used as an accurate marker of age in other invertebrates. 31 As with mineralization, noting increased lipofuscin accumulation in younger animals could indicate possible altered physiology or molecular pathology.
Another miscellaneous change noted during the study was gonadal atrophy or atresia, which was observed in both male and female gonads. Similar decrease is size of ovarian acini and in size and number of oocytes within acini has been documented in the freshwater pearl mussel Hyriopsis myersiana in the suspected post-spawn period, but other histological changes, such as oocyte vacuolation and accumulation of lipofuscin-laden cells, were not discussed. 32 Whether histological differences exist between pathological gonadal atrophy and physiological atresia in freshwater mussels is not apparent in the literature; thus, a distinction was not made in this case. Specimen collection occurred from June 18 to July 9, within the known spawning season of both species. Of 8 affected fatmuckets, 3 were collected on July 2, 5 of 8 affected fatmuckets were collected on July 9, and 2 of 2 affected plain pocketbooks were collected on July 9. Thus, a trend toward later collection dates and gonadal atrophy does exist and may indicate that those individuals with gonadal atrophy were simply post-spawn. However, other causes of gonadal atrophy were considered as well. Parasitism with bucephalid trematodes is well known as a cause of loss of gonadal tissue in freshwater mussels, likely via replacement of gonadal tissue by parasitic structures. The lesion described as gonadal atrophy in this study was not replacement of gonad by the parasite, although replacement of gonad tissue by parasite structures was documented in this study. Instead, gonadal atrophy in the current study was defined as degeneration or atrophy of existing gonadal structures without replacement by another structure (eg, parasites). This atrophy occurs in the face of moderate to severe parasitism elsewhere in all the individuals with this diagnosis, so, the idea that one lesion follows the other was considered. In addition, certain pollutants could result in gonadal atrophy, as occurs with exposure to tar in the males of the marine mussel Mytilus galloprovincialis. 3 Further studies on normal seasonal gonadal histology in fatmuckets and plain pocketbooks would be needed to begin to elucidate whether the observed changes were pathological (atrophy) or physiological (atresia).
This study provided a preliminary assessment of background lesions found in seemingly healthy populations of 2 native unionid mussels. Although no current or historic mass mortality events have been recorded through passive surveillance at the studied locations, a lack of population-level data for the studied species should be considered an important limitation of this data set. Further work could improve the information gleaned from this data set. For example, quantification of parasite load has been suggested as a tool for measuring disease burden in freshwater mussels and could be useful in the event that subsequent health assessments are performed at these sites. 12 Quantification of parasite loads could also become a useful tool when comparing disease burden between sites or between this seemingly healthy population and mussels collected from a mass mortality event. In addition, molecular methods could be applied to formalin-fixed paraffin-embedded tissues of trematode-infected mussels or mussels with suspected Rickettsia-like or Chlamydia-like organisms to further classify the observed parasites. This tool may lead to range expansion for certain parasites or, potentially, discovery of new species.
A range of lesions was observed in the 2 species of freshwater mussels that were collected. The populations were considered relatively healthy; thus, the conditions that were observed likely formed a suite of background lesions: those incidental findings, spontaneous infectious or endosymbiotic conditions, or normal physiological changes that routinely occur in free-living wild populations. These lesions should be considered when pathologists examine tissue from mussels collected at sites of mass mortality events or when investigating freshwater mussel disease.
Footnotes
Acknowledgements
The authors would like to express their appreciation to Diane Waller (US Geological Survey, Upper Midwest Environmental Science Center), Jordan Richard (US Fish and Wildlife Service, Southwestern Virginia field office), and Susan Knowles (US Geological Survey, National Wildlife Health Center) for their invaluable input on sampling techniques. They are also grateful to Casey Maynard (Indiana Department of Natural Resources) for her dedication to the project and her expertise in logistics, organization, and sample management. They also thank Julie Kempf, Cassie Hudson, Jacob Adams (Indiana Department of Natural Resources), Cassie Hauswald (The Nature Conservancy), Audrey Rupple, and Lauren Dorsey (Purdue University College of Veterinary Medicine) for assistance with sample collection. Finally, the authors would like to thank the histology technicians at the Animal Disease Diagnostic Laboratory (Purdue University) for excellent tissue processing and slide preparation, parasitology technicians at the Animal Disease Diagnostic Laboratory for mite identification, and MacKenzie McIntosh and Abigail Cox (Comparative Pathobiology, Purdue University) for slide scanning assistance.
Authors’ Note
Portions of this work were presented as abstracts at the Biennial Symposium of the Freshwater Mollusk Conservation Society, which was held virtually in 2021, and the Annual Meeting of the American College of Veterinary Pathologists, held virtually in 2020. Limited portions of the work were also presented at the International Wildlife Disease Association conference in 2019 and as part of the US Fish and Wildlife Service Freshwater Mussel Conservation webinar series held virtually in 2020.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the Indiana Department of Natural Resources State Wildlife Grant T-3-S-9 and public contributions to the Indiana Nongame Wildlife Fund.
