Abstract
The genus Edwardsiella is composed of a diverse group of facultative anaerobic, gram-negative bacteria that can produce disease in a wide variety of hosts, including birds, reptiles, mammals, and fish. Our report describes the isolation and identification of Edwardsiella piscicida associated with chronic mortality events in 2 separate captive largemouth bass (Micropterus salmoides) populations in New York and Florida. Wet-mount biopsies of skin mucus, gill, kidney, and spleen from several affected largemouth bass contained significant numbers of motile bacteria. Histologic examination revealed multifocal areas of necrosis scattered throughout the heart, liver, anterior kidney, posterior kidney, and spleen. Many of the necrotic foci were encapsulated or replaced by discrete granulomas and associated with colonies of gram-negative bacteria. Initial phenotypic and matrix-assisted laser desorption ionization–time of flight mass spectrometric analysis against existing spectral databases of recovered isolates identified these bacteria as Edwardsiella tarda. Subsequent molecular analysis using repetitive sequence mediated and species-specific PCR, as well as 16S rRNA, rpoB, and gyrB sequences, classified these isolates as E. piscicida. As a newly designated taxon, E. piscicida should be considered as a differential for multiorgan necrosis and granulomas in largemouth bass.
Keywords
Classified within the family Enterobacteriaceae, the genus Edwardsiella is composed of a diverse group of motile, facultative anaerobic, gram-negative rods. 21 Until recently, the genus consisted of 3 species: Edwardsiella ictaluri, Edwardsiella hoshinae, and Edwardsiella tarda.5,11,12 However, significant genetic differences have been identified within the group of organisms classified as E. tarda,2,9,30 which has led to the adoption of Edwardsiella piscicida as a new taxon. 1
Newly derived molecular methods have made delineation between closely related species of pathogenic bacteria such as E. tarda and E. piscicida possible. 9 As such, isolates once identified as E. tarda are now being reclassified as E. piscicida.1,10 Edwardsiella spp. have the ability to cause disease in a variety of host species, including 17 species of fish as well as humans.16,18 Many of the affected fish species described to date are of high economic importance such as channel catfish (Ictalurus punctatus), 23 tilapia (Oreochromis spp.), 22 Japanese flounder (Paralichthys olivaceus), 14 and Japanese eel (Anguilla japonica). 19 The presentation of disease varies widely and is often dependent on the species affected. Lesions range from focal subdermal fistulas in catfish to multiorgan necrosis and abscessation in tilapia. 17 We describe the histopathology, isolation, and genetic characterization of E. piscicida from largemouth bass (Micropterus salmoides).
E. piscicida was recovered from diseased largemouth bass residing in 2 separate facilities. The first facility was a public display exhibit in Florida, and the second facility was a closed recirculation system in New York. The first report of largemouth bass disease was submitted in June 2014 by an aquarist of a public exhibit containing Florida native temperate fishes. The aquarist reported several largemouth bass that had multiple, thick, white patches of mucus on their skin. Wet-mount biopsies were performed of skin mucus, fin, and gill; however, the results were unremarkable. Two weeks later, 1 largemouth bass was found dead in the enclosure. The next day a second largemouth bass was found dead in the enclosure and was promptly submitted for autopsy (fish 1). Several days later, 3 more largemouth bass died, the last of which was also submitted for autopsy (fish 2). Fish 1 and fish 2 were submitted to North Florida Aquatic Veterinary Services (NFAVS; Fort White, Florida) as representative samples from the diseased population (Table 1).
Fish sources and identification codes.

Similar to the previously described cases from Florida, a closed recirculation aquaculture facility in New York that contained cultured largemouth bass experienced chronic, low level mortality, of as many as 40 fish per day out of a total population of 7,500 fish. Mortalities occurred in June, August, and October of 2014. On 3 separate occasions during the mortality events, moribund largemouth bass were submitted to the Fish Disease Diagnostic Laboratory of the Aquatic Animal Health Program at Cornell University (CFDDL; Ithaca, New York). A total of 28 moribund largemouth bass from the population were evaluated by autopsy, histopathology, and bacterial culture with subsequent isolate identification.
Representative fish from the Florida outbreak, fish 1 (1.6 kg, 45.7 cm) and fish 2 (1.4 kg, 43.8 cm), were male and in good nutritional status with minimal postmortem autolysis. External examination of fish 1 was unremarkable; however, external examination of fish 2 revealed mild, multifocal erosion of the pelvic fins and diffuse necrosis of the gill epithelium involving the entire filament. Both fish had numerous motile and nonmotile bacteria that were observed on wet-mount biopsies of the skin mucus, gill, and fin. Internal examination of both fish revealed fibrinous adhesions extending from the celomic viscera to the body wall. Both fish had pale areas scattered throughout the epicardium and myocardium. In addition to the pale areas, fish 1 also had numerous, multifocal, small white nodules, up to 1 mm in diameter, protruding from the epicardium and extending into the myocardium of the atrium and ventricle (Fig. 1). Livers of both fish were moderately enlarged, and fish 1 had numerous multifocal digenetic trematode metacercarial cysts scattered throughout the hepatic parenchyma.
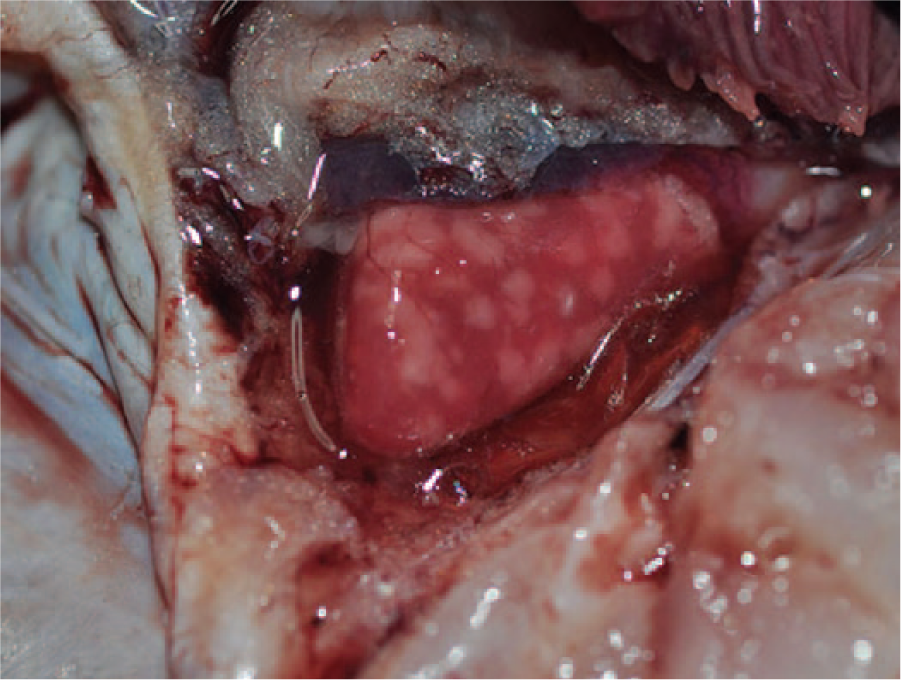
Gross photograph of the heart of fish 1 containing numerous, well-demarcated granulomas in the epicardium and myocardium.
Wet-mount evaluation of the splenic and anterior renal parenchyma revealed motile bacteria within the tissue of fish 1 and many small granulomas in the liver, anterior kidney, and posterior kidney of fish 2. The stomachs of both fish were empty, and normal-appearing digesta was present throughout the length of the intestine.
Fresh samples of spleen and posterior kidney from fish 1 and fish 2 were inoculated on trypticase soy agar (TSA) with 5% sheep’s blood and incubated at 25°C. Small, round, white bacterial colonies were present after 24 h of incubation. Three separate bacterial isolates (designated for this report as NFAVS-1, -2, and -3 [Table 1]) were submitted to the University of Florida’s Veterinary Diagnostic Laboratory (Gainesville, FL) for identification; all isolates were identified phenotypically as E. tarda.
The 28 fish received from the recirculation aquaculture facility in New York ranged in length from 113 to 157 mm, weighed 14–45 g, and were in good to fair body condition. The fish were immature, so sex could not be determined. External gross abnormalities were variable and included epidermal petechial hemorrhage at the base of the fins, around the vent, and around the eye. A subset of fish had swollen celoms that bulged. Wet-mount evaluation of skin mucus and gills was unremarkable. Within the celomic cavities of numerous fish, there was extensive lipid deposition in the mesentery and visceral organs. The liver, anterior kidney, and posterior kidney were swollen and pale in some fish. Fresh samples of posterior kidney were aseptically collected from several representative fish for bacterial culture.
Evaluation of bacterial isolates from the New York fish was performed at the Animal Health Diagnostic Center at Cornell University (Ithaca, New York). After 48–72 h of incubation on TSA with 5% sheep’s blood at 25°C, small, white bacterial colonies were grown from the posterior kidney of several fish from the recirculation aquaculture facility. On wet-mount evaluation, these colonies were composed of highly motile bacterial rods. Initial characterization of 3 isolates was performed by direct colony transfer to a matrix-assisted laser desorption ionization time of flight mass spectrometer (MALDI-TOF MS) automated identification system,a,b and a conventional biochemical screen. Isolates evaluated by MALDI-TOF were identified as E. tarda (HL23_1 EGS,3,4 score = 2.449 and score = 2.268). Other choices with a score >2.0 included E. tarda ACC 35.1 EGS 4 and E. tarda ACC 36.1 EGS. 3 One commercial system b designated these isolates as E. tarda with 100% confidence and no other identification choices. One representative isolate (designated as CFDDL-1 for this report; Table 1) was allocated for subsequent molecular characterization at IDEXX BioResearch (Columbia, Missouri).
All tissues collected from fish 1 and fish 2 (Florida) were fixed in 10% neutral buffered formalin for at least 24 h, infiltrated with paraffin, sectioned at 3–5 µm, mounted on glass slides, and stained with routine hematoxylin and eosin stain or Brown and Brenn Gram stain. Histologic examination of the anterior kidney, posterior kidney, heart, liver, spleen, intestine, and gill of both fish revealed similar lesions. Several organs had multifocal parenchymal necrosis progressing to granulomatous inflammation or discrete granulomas. Areas of necrosis and inflammation were often associated with colonies of gram-negative bacilli. The lesions in fish 2 were more chronic in nature than those in fish 1, and areas of parenchymal necrosis were more often surrounded by epithelioid macrophages rimmed by concentric layers of flattened macrophages or fibrous connective tissue, which was consistent with granuloma formation (Fig. 2A). Renal tubules in the posterior kidney were multifocally necrotic and characterized by hypereosinophilic epithelial cells with pyknotic or karyorrhectic nuclei that often sloughed into the tubule lumina. In fish 2, tubules were also multifocally replaced by granulomas containing bacteria. Anterior kidney and spleen had similar scattered, necrotizing to granulomatous foci throughout the parenchyma that were associated with variably sized colonies of gram-negative bacilli (Fig. 2B). In the heart, most notably at the junction of the ventricular compact and spongy layers, cardiac myocytes were multifocally necrotic with hypereosinophilic hyalinized cytoplasm, loss of cross striations, and pyknotic or karyorrhectic nuclei (Fig. 2C). Within the atrial and ventricular lumina as well as in the small- to medium-sized vessels of the majority of organs evaluated, eosinophilic lamellated fibrin thrombi adhered to the endothelium. Many of the thrombi contained small colonies of bacilli (Fig. 2A, 2B, 2D). In addition, bacterial emboli were observed in numerous small vessels of the heart, liver, intestine, and gill.
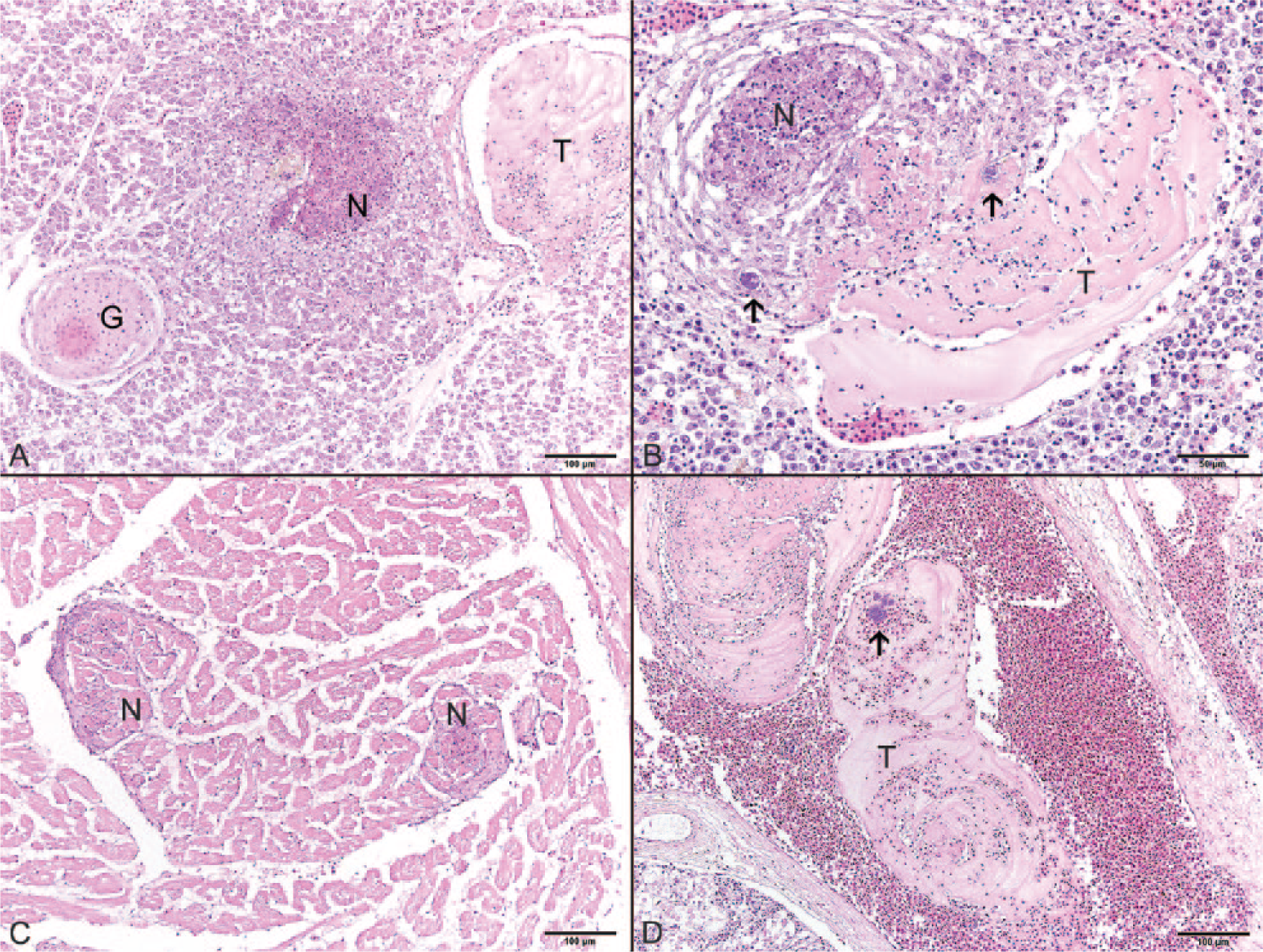
Characteristic histologic lesions observed in disseminated Edwardsiella piscicida infection.
Several other incidental lesions not associated with the disseminated bacterial process were present within these fish. Scattered throughout multiple organs, including the mesentery, liver, spleen, anterior kidney, and posterior kidney, were numerous granulomas in various stages of chronicity. Some of the granulomas contained intact metazoans; others had a core of eosinophilic necrotic cellular debris surrounded by epithelioid macrophages rimmed by concentric layers of fibrous connective tissue. The gills of fish 2 were mildly hyperplastic with synechiae of the lamellar tips and an accumulation of edema and necrotic cellular debris that multifocally expanded the intralamellar troughs.
Similarly, all major organs were collected from a representative fish (fish 3) from the New York outbreak and processed for histopathology. Microscopic lesions in fish 3 were similar to those found in the 2 fish reported above. The liver, anterior kidney, posterior kidney, and spleen had multifocal areas of necrosis as well as well-formed granulomas. Ziehl–Neelsen acid-fast staining was negative for acid-fast organisms; Brown and Brenn Gram stain revealed gram-negative rod-shaped bacteria within some lesions. All evaluated organs, particularly the liver, contained abundant lipid deposition. As with fish 1 and 2, incidental lesions were observed in fish 3, including myxozoan cysts in the posterior kidney.
Bacterial isolates from all 3 fish (fish 1–3) were subjected to further molecular analysis. Primers used in this molecular analysis are listed in Table 2. Initial sequence analysis of the fish 3 isolate (CFDDL-1) was performed at IDEXX BioResearch. The isolate was deemed most closely related to E. piscicida based on initial genetic sequence analysis of the bacterial RNA polymerase β-subunit gene (rpoB) and the 16S ribosomal RNA gene (16S rRNA), and confirmed by sequence analysis of the DNA gyrase subunit B gene (gyrB). Across the analyzed rpoB segment, isolate CFDDL-1 most closely matched E. piscicida isolate C07-087 (GenBank CP004141) from channel catfish in Mississippi 25 and E. tarda isolate EIB202 (GenBank CP001135) from turbot (Scophthalmus maximus) in China, 28 with each sharing 352/355 bp (99.2% identity). Similarly, the 16S rRNA segment shared 100% identity (673/673 bp) with E. piscicida isolate C07-087, whereas the gyrB segment shared 100% identity (1,064/1,064 bp) with E. piscicida isolate LADL 99-462 (GenBank JX866997) recovered from a channel catfish in Louisiana.9,10
PCR primers used in the present study.*

F = forward; R = reverse; P = probe.
These results were supported by more complete 16S rRNA and gyrB sequencing performed at the National Warmwater Aquaculture Center (Stoneville, Mississippi), which identified all 4 bacterial isolates in this study (CFDDL-1, NFAVS-1, NFAVS-2, NFAVS-3) as E. piscicida. The isolates were a 100% match (1,294/1,294 bp) to partial 16S rRNA sequences within an E. piscicida population set (GenBank JX866988–JX867005) previously characterized,9,10 yet also demonstrated >99% similarity to E. ictaluri and E. tarda isolates from the same study. Consistent with analysis at IDEXX, 3 of the isolates (CFDDL-1, NFAVS-2, NFAVS-3) shared 100% identity (1,894/1,894 bp) at the gyrB sequence with E. piscicida isolate LADL 99-462. Alternatively, E. piscicida isolate NFAVS-1 possessed a unique gyrB sequence compared with other published gyrB sequences available in GenBank, being most similar (1,891/1,894 bp; 99.8%) to E. piscicida isolate S07-1019 (GenBank JX867002) from a blue catfish (Ictalurus furcatus) in Mississippi. This identification was further supported using a validated end-point and real-time PCR targeting the E. piscicida fimbrial subunit,
23
which identified all 4 isolates as E. piscicida. Genetic fingerprints using the BOX and ERIC (
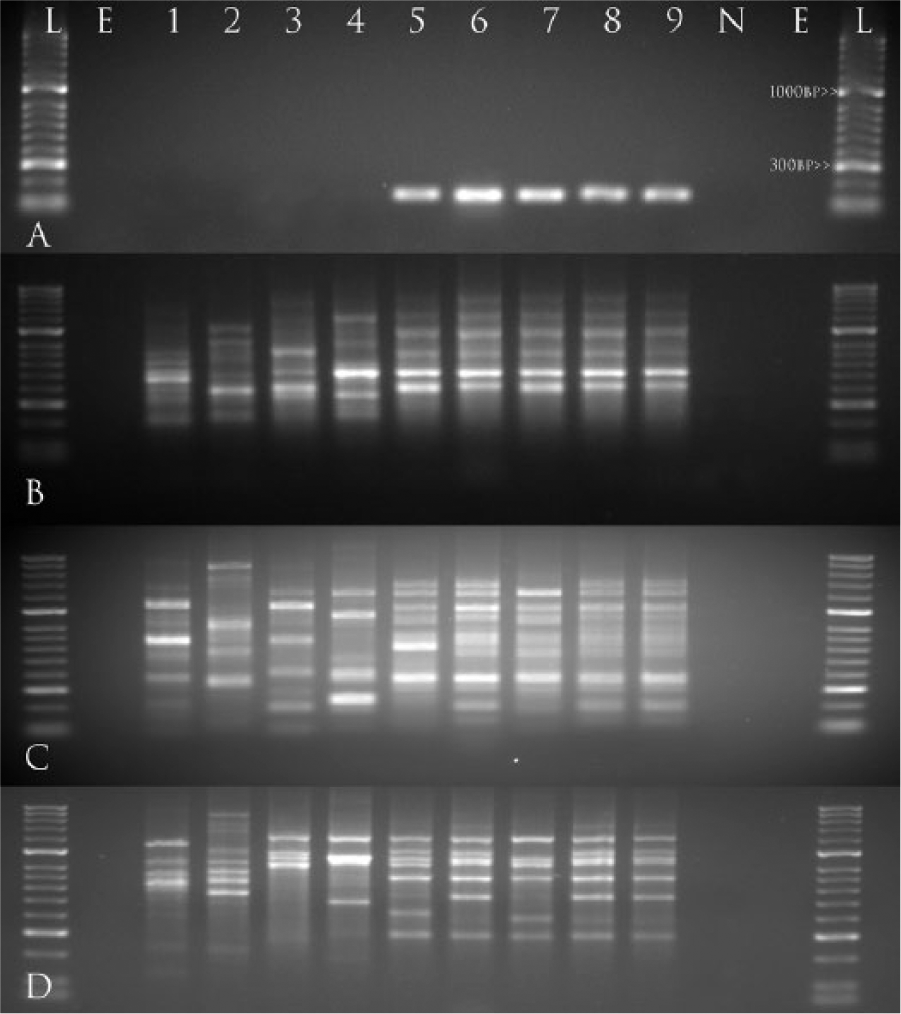
Gel images representing (
Numerous reports of disease in fish, including largemouth bass, catfish, eel, tilapia, and seabream, have been attributed to Edwardsiella tarda. The described clinical cases in Florida and New York are reports of E. piscicida in largemouth bass. It is likely that this organism has been associated with infections in largemouth bass in the past but was classified as E. tarda. The initial identification of largemouth bass isolates in this case was complicated by the recent designation of 2 new species of Edwardsiella (E. piscicida and E. anguillarum).1,24 Further complicating the issue, several strains of Edwardsiella have been described, classified, and designated as E. tarda in GenBank and other nucleotide databases prior to the adoption of E. piscicida and E. anguillarum. As such, many sequences are misidentified.
E. piscicida was first characterized as a pathogen of European and Asian fishes, predominantly eel and turbot. 1 A genetic assessment of 44 archived, phenotypically identified E. tarda isolates from diseased catfish in the southeastern United States has revealed E. piscicida is present in the United States.9,10
In largemouth bass, gross and histologic lesions associated with E. piscicida can be similar to lesions caused by other bacterial species within the genus Edwardsiella, 18 as well as other gram-negative, motile bacilli such as Aeromonas hydrophila (Camus A, et al. Aeromonas bacterial infections: motile aeromonad septicemia. SRAC Publication No. 478. Southern Regional Aquaculture Center, 1998. Available at: www.aces.edu/dept/fisheries/aquaculture/pdf/478fs.pdf). Although external gross examination can vary from normal to multifocal cutaneous petechiation, abdominal swelling, and fin or skin erosion, the histopathological changes associated with disseminated disease are consistent. Similar to other bacterial septicemias, there is multiorgan necrosis and granuloma formation with the presence of gram-negative bacteria within lesions. Gross and histologic evaluation of moribund fish in concert with molecular evaluation of bacterial isolates can optimize proper diagnosis of these phenotypically ambiguous organisms. 20 The near complete 16S rRNA sequence in combination with rpoB or gyrB can be employed as molecular markers to differentiate between aquatic gram-negative pathogens such as Edwardsiella spp. Our report highlights the challenges of identifying closely related bacterial species that can cause disease in largemouth bass and suggests caution must be used when consulting reference databases, as they may not support the most contemporary bacterial identification methodologies.
Footnotes
Authors’ contributions
SB Fogelson contributed to conception and design of the study; contributed to analysis and interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. BD Petty contributed to conception of the study; contributed to analysis of data; critically revised the manuscript; and gave final approval. SR Reichley contributed to design of the study; contributed to analysis and interpretation of data; critically revised the manuscript; and gave final approval. C Ware contributed to analysis and interpretation of data; critically revised the manuscript; and gave final approval. PR Bowser and MJ Griffin contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. MJ Crim contributed to analysis and interpretation of data and critically revised the manuscript. RG Getchell and H Marquis contributed to analysis and interpretation of data; critically revised the manuscript; and gave final approval. KL Sams contributed to analysis of data; critically revised the manuscript; and gave final approval.
a.
Bruker Microflex mass spectrometer with flexControl and MALDI BioTyper software; Bruker Daltronics Inc., Billerica, MA.
b.
Sensititre, Thermo Fisher Scientific, Waltham, MA.
c.
Hyperladder II, BIOLINE, Taunton, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
