Abstract
Freshwater mussels are one of the most imperiled groups of organisms in the world, and more than 30 species have gone extinct in the last century. While habitat alteration and destruction have contributed to the declines, the role of disease in mortality events is unclear. In an effort to involve veterinary pathologists in disease surveillance and the investigation of freshwater mussel mortality events, we provide information on the conservation status of unionids, sample collection and processing techniques, and unique and confounding anatomical and physiological differences. We review the published accounts of pathology and infectious agents described in freshwater mussels including neoplasms, viruses, bacteria, fungi, fungal-like agents, ciliated protists, Aspidogastrea, Digenea, Nematoda, Acari, Diptera, and Odonata. Of the identified infectious agents, a single viral disease, Hyriopsis cumingii plague disease, that occurs only in cultured mussels is known to cause high mortality. Parasites including ciliates, trematodes, nematodes, mites, and insects may decrease host fitness, but are not known to cause mortality. Many of the published reports identify infectious agents at the light or ultrastructural microscopy level with no lesion or molecular characterization. Although metagenomic analyses provide sequence information for infectious agents, studies often fail to link the agents to tissue changes at the light or ultrastructural level or confirm their role in disease. Pathologists can bridge this gap between identification of infectious agents and confirmation of disease, participate in disease surveillance to ensure successful propagation programs necessary to restore decimated populations, and investigate mussel mortality events to document pathology and identify causality.
Keywords
Freshwater bivalves have a worldwide, albeit uneven, distribution with 19 families and over 1200 species.28,122 They are among the most threatened organisms in the world with nearly 40% of the species assessed as near threatened, threatened, or extinct. 122 Within Unionoida, 6 families have at least 800 species with a biodiversity hotspot in North America that is home to over 300 of the recognized species.28,78 Declines in North American freshwater mussels have been considerable and began over 100 years ago. 76 Sixty-five percent of North American freshwater mussels are considered endangered, threatened, or vulnerable, and more than 30 have gone extinct in the last century, 78 with 8 declared extinct in 2021. 52
Many causes of freshwater mussel declines are postulated in the literature, but most lack supporting evidence. 54 Habitat alteration and destruction are the most commonly cited causes with strong evidence, particularly earlier this century following widespread placement of dams and impoundments, deforestation, and release of industrial effluents. 54 However, more recent large-scale freshwater mussel declines in North America in the central and southeastern United States were “enigmatic,” 78 with epidemic mussel mortality events occurring in otherwise healthy ecosystems with minimal anthropogenic effects and no known cause. 76 Infectious disease is often not considered as a possible cause of mortality events 78 or mentioned in mussel conservation literature. 202 Much of the literature addresses mussel declines in the context of environmental contamination, often without methods to measure or describe health parameters, apart from growth and survival. More recently, diagnostic investigations of mussel mortality epidemics have documented a variety of putative infectious agents in dying mussels. These findings emphasize the need for a more systematic approach to disease investigation in declining mussel populations, with the goal of identifying causative factors that can be mitigated. Knowledge of the normal physiology of healthy individuals and pathology of diseased mussels is limited, making it difficult for pathologists to identify and interpret the significance of pathological findings during health surveys or mortality events.
In this work, we synthesize the current knowledge of pathology and disease in unionids to aid the diagnostician, and to encourage the involvement of pathologists in this arena.
Assessing Body Condition and Sex
Condition index (CI), a measure of overall health, has not been widely used in freshwater mussels, but can be calculated using soft body wet weight (SBWW), where CI = SBWW / (shell length * shell height * shell width) * 100. 102 Some studies use dry body weight, 169 which is less confounded by the variable water content in bivalves, 156 but cannot be performed when tissues are to be sampled for diagnostic tests. Gonadosomatic index (GSI), a ratio between gonadal weight and body weight used to assess sexual maturity, is problematic in bivalves because the gonad is fused with other organs. Linear measurements of the gonad region have been used as an estimate for gonad volume to emulate the GSI. 195 Other studies focus on female fecundity and instead use brood (ie, the number of gill marsupia [specialized brood pouch derived from the demibranchs] containing larvae), 66 or glochidia (larval) counts as a representation of reproductive fitness. 140 Freshwater mussels can be sexed by assessing shell shape in sexually dimorphic species, visual inspection for pallor and inflation of the marsupium to assess gravidity, collection of gonadal fluid and staining to evaluate microscopically for developing gametes, and histopathologic identification of ovaries or testes.47,88,134,171
Sample Collection and Processing
Nonlethal Sample Collection
Nonlethal sample collection techniques include hemolymph collection and tissue biopsies that may be especially useful for the study of endangered or threatened freshwater mussel populations. Anesthesia techniques for freshwater mussels can be found in Lellis et al. 115 Hemolymph can be collected from an adductor mussel sinus, 74 and used for hematologic and biochemical analyses.32,74 However, reference standards for hematologic and biochemical parameters are not available for all species, and developing reference ranges for healthy freshwater mussels is complicated because ranges are likely to vary by species, time of year, water quality, and physical habitat.75,187 To collect hemolymph, mussel valves are opened slightly, for example, using a pediatric nasal speculum until a rubber stopper can be placed in a relatively anterior location in the opening between the shells. Up to 0.5 mL was drawn over 30 seconds using a small (28 gauge) needle and 1 ml syringe from 4 to 7 cm (50 g) Elliptio complanata (eastern elliptio) without negative impact on growth or survival. 74 Up to 1 ml was safely collected from Quadrula sp., Anodonta cygnea (swan mussel), and Anodonta anatina (duck mussel).32,92 Hemolymph has been used for the detection of vitellins, 65 genetic identification, 131 flow cytometry, 92 culture, 114 transcriptomics, 25 proteomics, 117 and metagenomics. 165 Nonlethal biopsies can be collected from the mantle,24,184 foot, 141 or gonad, 171 and can be used for histologic, genetic, contaminant, and biochemical analyses. Swabs have been used to obtain DNA from tissues. 86
Lethal Sample Collection and Tissue Processing
For the investigation of mortality events, tissues for histologic evaluation should be collected during outbreaks of acute mortality (ie, “die-offs”). Mussels found dead are not suitable for histology because they autolyze quickly and are rapidly colonized by bacteria and fungi. 202 Moribund mussels paired with a few healthy control mussels from the same site or ideally a nearby unaffected site are the typical sample for disease investigation, but moribund mussels may be secondarily colonized with opportunists. 202 The assessment of “healthy” versus “diseased” can be difficult. The best indicators of morbidity include diminishment of behavioral responses such as valve closure, righting, horizontal movement, and burrowing.48,203
After collection, mussels can be stored or shipped on ice, or transported to the laboratory in an aerated water tank, before depuration, euthanasia, 115 sample collection, fixation, and processing.93,108,118 Depuration involves holding mussels in clean water for at least 24 hours to remove biological contaminants and particulates such as sand or silt that may be attached to the surface of the tissues. Failure to complete depuration can lead to damage of the microtome blade during sectioning and cause histological tissue artifacts such as chatter. 134 A single oblique cross-section through the middle of the visceral mass, mantle, and gills may sufficiently encompass most of the major organs for histology, apart from heart, kidney, and adductor muscle. 93 Any remaining tissue can be stored frozen for subsequent analyses such as culture, PCR, metagenomics, and metabolomics.
Gross Anatomy and Histopathology
Knowledge of the anatomy of freshwater mussels can be helpful with processing cross-sections for histopathology. Details of the dissection and anatomy of a freshwater mussel can be found in Lőw. 124 Likewise, understanding the normal histology of freshwater mussels is necessary before evaluating the pathology. A review of normal histology of several unionid species can be found in McElwain and Bullard, 134 and a broader review of bivalve histology is provided in Smolowitz. 181
Unique or Confounding Anatomical or Physiological Differences
Hermaphroditism
Most unionids are dioecious; 196 however, hermaphroditic individuals occur at low prevalence, but are poorly understood. 103 Microscopically, they have either both ovarian and testicular gametogenic acini (Fig. 1a), or hermaphroditic acini, which make both ova and sperm. 134 They may have lower reproductive success, and gravid individuals tend to have dominantly female gonad. 53 However, the possibility for self-fertilization may be advantageous where population density is low. 20 In some species, sequential hermaphroditism (ie, protandry or sex reversal) occurs, where the higher energy stores required for oogenesis can be reserved for larger individuals whereas small individuals are capable of spermatogenesis. 53 Hermaphroditism can be pathologic, such as a developmental anomaly or response to larval digenean infection.103,194 Certain species may be facultative hermaphrodites, potentially influenced by environmental conditions. 82 The association of hermaphroditism with environmental endocrine disruptors has not been investigated in unionids, as in other bivalves.9,111
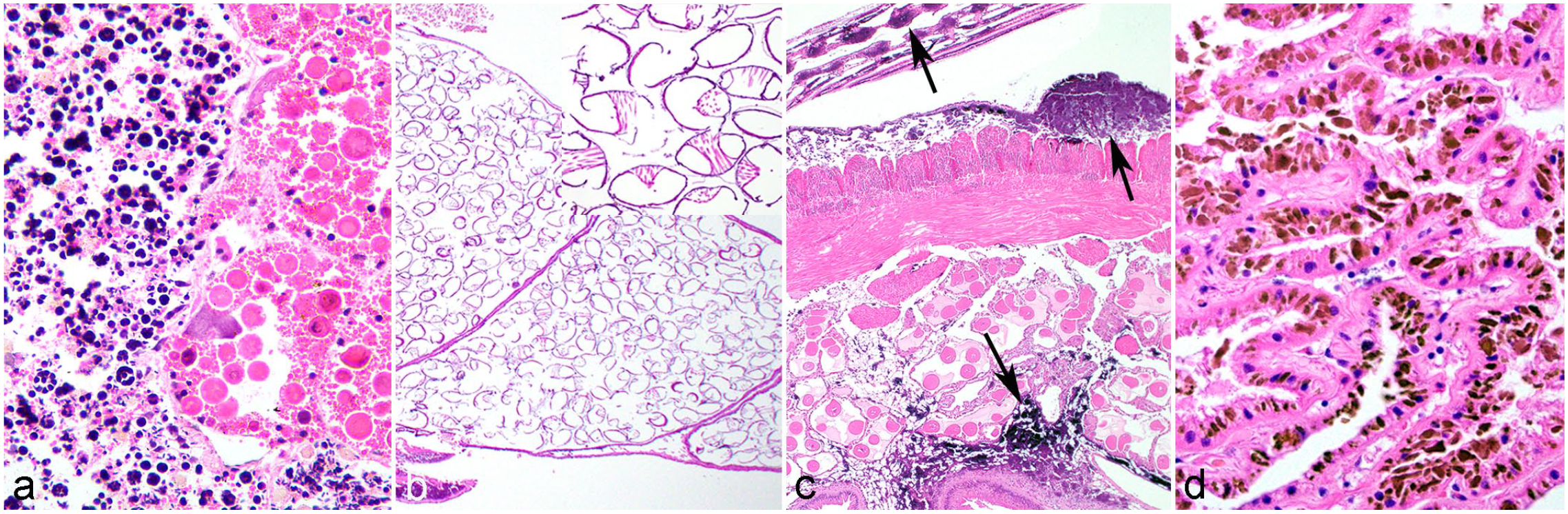
Unique or confounding anatomical or physiological differences in freshwater mussels. Hematoxylin and eosin.
Marsupium
Reproduction in freshwater mussels involves parental care and an obligate parasitic stage on a fish or amphibian. 37 Fertilization in freshwater mussels occurs in the dorsal exhalant chamber, or “suprabranchial chamber,” of the female. 190 The gills, composed of 2 pairs of demibranchs, divide the mantle cavity into the ventral inhalant chamber and the dorsal exhalant chamber and serve as the roof and floor of the chambers, respectively. Embryos develop in the marsupium, a specialized brood pouch derived from the demibranchs. 47 Grossly, gills normally appear as thin flaps hanging from each side of the body, but in gravid mussels, the demibranchs fill with eggs and glochidia (larvae) and are greatly thickened. 44 Histologically, the water tubes of the marsupium are filled with eggs or glochidia (Fig. 1b). 134
Concretions
Pathologists unfamiliar with freshwater mussels may not be aware of the abundant concretions present throughout mussel tissues. Calcium is scarce in freshwater environments and freshwater mussels have evolved an ability to sequester calcium within granular concretions in their tissues. 34 Large calcium concretions can be found in the interstitial connective tissues of the gills, mantle, labial palps, digestive gland, gonad, kidney, pericardial gland, heart, muscle, and visceral mass.4,34,91,157 They can account for more than half of the dry tissue weight of the gills. 178 Although the metal-ion content of the concretions can differ among species and tissues in an individual animal, calcium-phosphate often predominates.4,34
Calcium concretions, when combined with iron, have been observed grossly in Hyridella depressa (depressed mussel) as a bright orange covering on the mantle and palps and within the visceral mass. 34 On histopathology, basophilic, refractile, round to irregular granular concretions of varying sizes that stain positive for calcium occur within interstitial connective tissues (Fig. 1c).34,91 On transmission electron microscopy with fixed tissues, concretions have annulations with an electron-dense core surrounded by electron-dense (organic material) and electron-lucent material (inorganic material), but annulations were not observed in frozen tissue indicating the annulations may be a fixation artifact.34,91
The function of the calcium stores is not completely understood, but theories include roles in shell formation, detoxification of metals, calcium homeostasis, and biomineralization. 34 Although some authors suggest concretions in the gills serve as a reservoir for shell development in glochidia, 178 in some species concretions are scarce in the gills, indicating they are not a calcium source for glochidia shell formation.34,157 The role that concretions play in detoxification of metals is unclear. If a mussel binds divalent cations for detoxification in gill concretions and then transfers those calcium stores to larvae, this would be disadvantageous; however, concretions in different tissues could have different functions. 177
Renal Lipofuscin
Yellow-brown acid-fast granules ascribed to lipofuscin are common within the cytoplasm of renal epithelial cells among marine and freshwater bivalves (Fig. 1d).26,134,168 Lipofuscin accumulation is considered a consequence of lysosomal lipid peroxidation associated with oxidative stress, 85 and is often attributed to pollution with inorganic or organic contaminants.26,168,174 It may have a role in renal excretion of metals, where granules of metal-binding lipofuscin are excreted by exocytosis. 26 Indices for renal lipofuscin have been used as a histopathological biomarker of health,26,85,168 although the effect of pigment accumulation on renal function is unknown. Lipofuscin similarly accumulates in the digestive gland of marine bivalves. 36
Neural Pigment
As in other mollusks, neuroglia and extracellular space of unionid ganglia may contain yellow-brown granules, 134 considered a lipochrome-containing organelle sometimes referred to as cytosomes. 199 These pigmented granules have not been characterized in unionids and are of uncertain function or pathological significance. In marine bivalves, they are ultrastructurally diverse, 145 and are hypothesized to play a role in survival during anoxic conditions. 224
Immune System
A review of immune responses by bivalves to infectious disease can be found in Allam and Raftos. 7 Bivalves lack an adaptive immune system in the traditional sense as they do not have lymphocytes and antibodies, 138 and rely entirely on the physical, humoral, and cellular components of the innate immune system for defense against pathogens. 182 The shell, mucus, and epithelial cells provide a physical barrier against insults.6,7 Proteins with bacteriolytic and opsonic functions in bivalve hemolymph help to coordinate the humoral response. 182 Hemocytes are the only circulating cell of freshwater mussels and provide the primary cellular response against pathogens. 182 They are capable of phagocytosis, encapsulation, 182 melanization, mineralization, 7 and wound repair. 153
Although invertebrates lack the machinery of the adaptive immune system, recent evidence shows that invertebrates are capable of specificity and immunological memory or “priming.” 138 With immune priming in invertebrates, the immune response can fade and then be recalled with a secondary challenge, implying “memory,” or induce a sustained response after the primary response with upregulation of immune effectors which are protective in a secondary challenge. 215 In shellfish, the evidence for immune priming is mostly phenomenological showing a stronger immune response upon reinfection, and more work is needed to understand the underlying mechanisms. 215 As these mechanisms become better understood, the potential for vaccine development for use in propagation efforts increases.
Pathology and Infectious Agents of Unionids
Although there are several reviews of the known infectious agents, diseases, and disorders of freshwater mussels,37,73,133,184 most focus on identification of infectious agents, rather than comprehensive pathological descriptions that encompass associated lesions. This may reflect the scarcity of diagnostic investigation of mussel mortality, lack of involvement of pathologists, absence of lesions accompanying various infections, poor understanding of mussel response to injury and therefore interpretation of observed lesions, or uncertain pathogenicity of infectious agents. The lack of descriptions makes definitive diagnosis of any disease challenging because they are typically an essential component of a case definition, 43 emphasizing the scientific value of greater involvement of the discipline of veterinary pathology in investigating mussel mortality. This section will focus on gross and microscopic pathology in freshwater mussels and the known infectious agents that occur in these species, including viruses, bacteria, fungi or fungus-like agents, and parasites.
Gross Pathology
Few publications addressing freshwater mussel mortality events or pathology describe or illustrate gross lesions. Reasons for this could include lack of diagnostic investigation of mussel declines or mortality events, prioritizing field samples or tissue collection for diagnostic testing rather than macroscopic examination, focusing on identification of etiologic agents rather than lesions, and difficulty with identifying subtle gross lesions due to small host size and anatomic conglomeration of organs into a visceral mass.
Shell Abnormalities
Shell abnormalities are poorly described in unionids, and their recognition depends on a firm understanding of what is normal for a particular species. Considerable environment-associated intraspecific variability can occur in shell dimensions, thickness, and surface erosions that also need to be taken into account by the diagnostician. 169 For example, shell mineralization can be influenced by water acidity, calcium availability (hardness), substrate, exposure, nutritional state, and pollution.90,123,192 Shell erosion has been attributed to water chemistry and turbulence as well as primary shell weakness due to endolithic organisms (ie, shell-boring algae or cyanobacteria) or unidentified causes.44,90,169,193 Protuberances of excessive nacre (conchiolin and calcium carbonate) deposition on the inner surface of the shell (ie, “blister pearls” or “pustules”) can result from foreign body or ectoparasites between the shell and mantle, or from previous shell injury. 42 Discoloration of inner shell (“nacre staining”) can represent a shell damage response, 170 similar to the discoloration seen with Vibrio tapetis infection of the periostracal lamina at the mantle edge in marine clams. 147 In marine bivalves, mantle retraction from the pallial line (ie, mantle recession) can lead to befouling at the shell margin, and can indicate underlying systemic infection as seen with haplosporidiosis in oysters. 57 Malformed shells are poorly understood but have been circumstantially linked to anthropogenic land use and trematode infections.73,188
Gill Pigment
Brown-black areas within the marsupium have been attributed to unfertilized oocyte decay, 101 as well as unspecified glochidial disease or fungal or ciliate infection. 156 Stress of the female mussel can result in eggs or glochidia being prematurely expelled from the marsupia, a process termed miscarriage. 217
Nodules
Grossly evident nodules often reflect foci of hemocyte infiltration, particularly instances of nodulation or encapsulation, but must be differentiated grossly from rarely seen neoplasms and digenean cysts, which may be yellow or orange.159,175 Irritants in the mantle can result in the deposition of layers of nacre that form a grossly visible nodule with similar consistency to the inner shell (ie, “pearls”). Nacrezation is a process similar to shell repair that occurs when mantle injury or irritation by infectious or noninfectious agents (ie, foreign particles) results in excessive deposition of nacre matrix on the inner shell or within associated soft tissues. In mussels, nacrezation is often induced by parasites, including digenean and insect larvae, and mites. 133 Microscopic examination of pearls postdecalcification can demonstrate the presence of parasites centrally. 68
Microscopic Pathology
Inflammation and Wound Repair
Most studies classify bivalve hemocytes as either granulocytes (eosinophilic or basophilic) or hyalinocytes (or agranulocytes).32,50 Granulocytes have abundant cytoplasmic granules and a low nuclear to cytoplasmic ratio, and hyalinocytes have few or no cytoplasmic granules and a higher nuclear to cytoplasmic ratio (Fig. 2a).32,51,92 Hemocytes are capable of recognizing and phagocytosing foreign agent molecules and destroy them by phagolysosomal fusion and the release of lysosomal enzymes, reactive oxygen species, nitric oxide, and antimicrobial factors. 182 Both types of hemocytes are phagocytic, but granulocytes are considered the primary phagocytic cell responding to infectious agents. 36 Hemocytic infiltrates are confirmed microscopically and typically interpreted as a cellular response to injury (ie, inflammation). In other bivalves, hemocytic defensive responses are classified according to 3 microscopic morphologies: infiltrative hemocytosis, nodulation, and encapsulation.36,51 Infiltrative hemocytosis can be focal when the inciting agents are localized to one area or diffuse with systemic infection, and is distinguished by the increased presence of hemocytes without the formation of nodules (Fig. 2b). Diffuse infiltrative hemocytosis presents a greater interpretive challenge because hemocytes normally move through tissues of animals with open circulatory systems. For example, granulocytes are often present in hemolymph sinuses in the gill and within the epithelium and connective tissues of the alimentary tract where they may play a role in digestion and nutrient transport.39,134 Defining the normal extent of hemocyte presence is challenging and may vary by species, individual, tissue, mussel size, and environmental factors. Hemocytosis can be induced by a variety of stimuli including infectious, chemical, and physical injury. Severe diffuse hemocytosis is particularly suspicious for systemic infection, whereas gill hemocytosis is often associated with a polluted environment in marine bivalves. 51
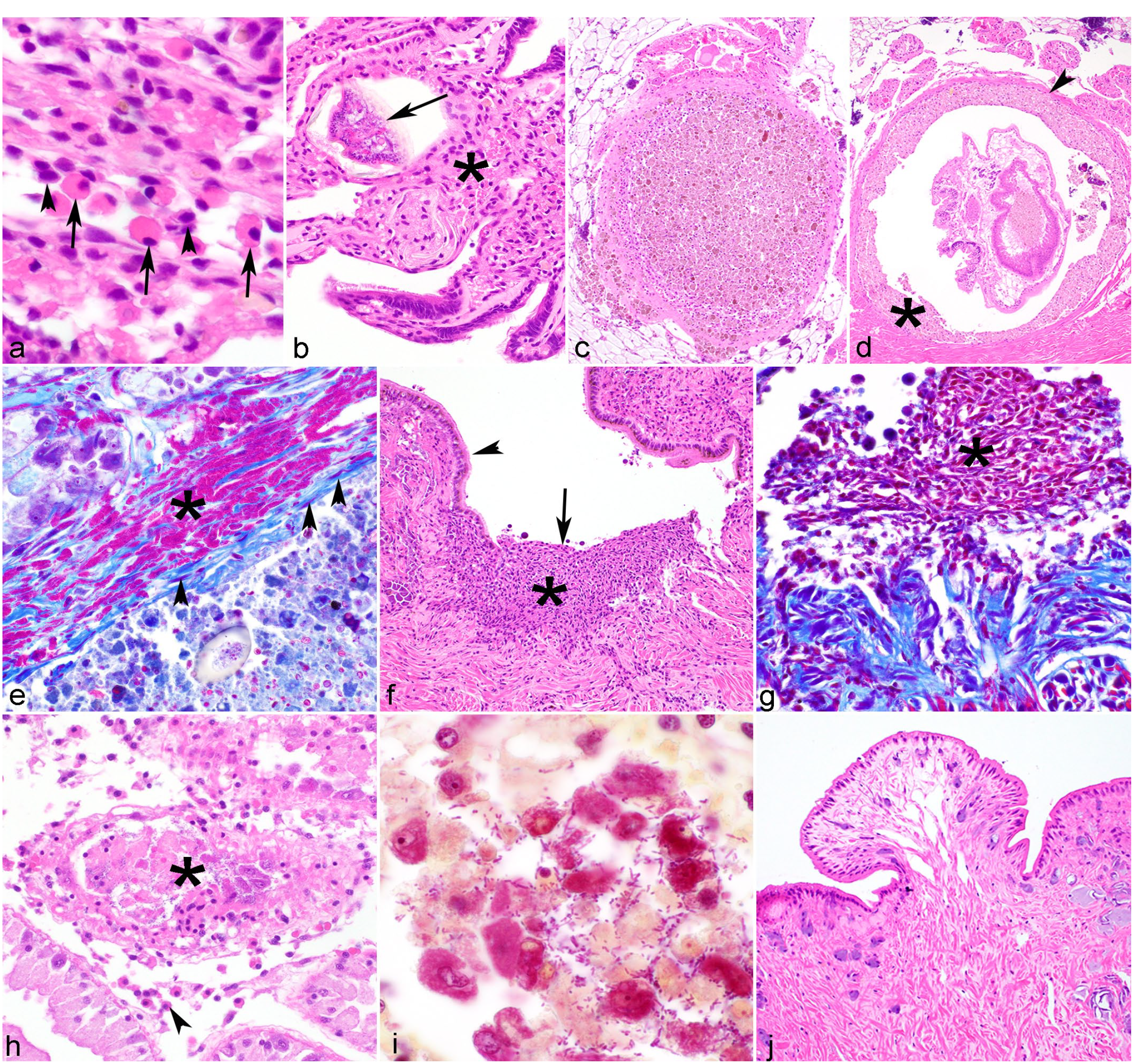
Microscopic lesions in freshwater mussels.
Nodulation consists of hemocyte aggregation, primarily involves granulocytes (sometimes termed granulocytoma), and may have a central area of necrosis, particularly when associated with bacteria (also variously referred to as an abscess, pustule, or blister when involving an organ surface). Nodulation occurs either in a tissue or in a hemolymphatic vessel and is often ascribed to phagocytic response (Fig. 2c). It is usually associated with bacterial infections, 51 but has been associated with protist infections, presumptively attributed to contaminant exposure, or otherwise unexplained.37,41,85 Encapsulation occurs when phagocytosis is ineffective (ie, with large organisms such as metazoan parasites, foreign material, fungi, protists, and bacteria that evade phagocytosis), and acts to confine injurious agents by encapsulating the target and releasing cytotoxic products in an attempt to destroy the foreign body.37,182 Encapsulation occurs when layers of hemocytes surround foreign material, and is sometimes accompanied by melanization, mineralization, or bordered by fibrosis (Fig. 2d, e). 7
Hemocytes are also involved in wound repair. After tissue injury, hemocytes accumulate at the site of the wound causing a “clot,” then line the wound before fibroblasts and collagen repair the injury (Fig. 2f, g). 153
Degeneration and Necrosis
Most descriptions of degeneration and necrosis in Unionidae involve the digestive gland,85,87,211,222 where they may be grossly evident as pale green-brown discoloration of the gland. 205 Microscopically, degeneration manifests as cell swelling, cytoplasmic vacuolation (indicating lipidosis or lysosomal swelling), or lipofuscin accumulation. 37 Necrosis is microscopically characterized by cell sloughing, leaving a denuded basement membrane and luminal cell debris (Fig. 2h, i). Degeneration and necrosis may be accompanied or followed by epithelial atrophy,41,168 comprising reduction in cell height and condensation of cytoplasm. In marine bivalves, digestive gland degeneration and atrophy are considered sensitive indicators of environmental stress, encompassing poor nutrition, saline or temperature derangements, and contaminant exposure.37,107,125
Disturbances of Growth
Only a handful of tumors are described in freshwater mussels. The classification of these as true neoplasms is often uncertain and will remain so until more of these abnormal growths are studied and their relationship to injury and infection are clearly understood.149,150 A pedunculated, hazlenut-sized, ovoid mass, suggestive of adenomyoma, occurred on the internal aspect of the left pallial lobe of A. cygnea. 206 Two tumors were observed grossly in the mantle of A. cygnea, 1 in a previously injured mussel with a pea-sized mass beneath the mantle and another connected to the mantle and outer gill associated with dysfunction of nacreous production. 45 Histologic descriptions were not provided.
A polypoid connective tissue tumor from the labial palp was described in Utterbackiana implicata (alewife floater). 33 The tumor was covered by small, irregular columnar epithelial cells with abundant goblet cells. The core of the mass was composed of large connective tissue cells with granular cytoplasm that were scant centrally with numerous hemolymph sinuses and few mitoses.
A hyperplastic growth composed of nervous and glandular tissue with frequent mitoses was observed in the marsupial gill of a hermaphroditic, weak, watery, and thin Pseudanodonta complanata (depressed river mussel). 155 Chironomid larvae were present on the inner shell, mites were in the mantle, and Periodic acid-Schiff-positive, germinating suspect fungal spores were in multiple tissues. The growth was admixed with mites, fungal hyphae, and host cells (glochidia), and the author suggested that irritation from these might have induced the growth.
Two tumors were described in the feet of Anodonta californiensis (California floater).150,151 The foot of freshwater mussels is covered by a ciliated epithelium with an underlying layer of dense connective tissue with muscle fibers and mucous glands, and a core of muscle bundles permeated by sinusoids. 181 In one report, a 3 mm × 2 mm well-demarcated, firm papilliform growth extended from the lateral edge of the ventral foot of a mature male. 150 The mass was composed of irregularly arranged glandular cells and small myofibers covered by convoluted, vacuolated epithelium that formed deep crypts. Described as an adenomyoma, the author cautioned it may not be a true neoplasm, but rather a hyperplastic response to irritation or injury. Pedal papilliform neoplasms were found in 40% of A. californiensis. 151 Fourteen neoplasms were observed in the 4 affected mussels, with each mussel having 2 to 6 masses resembling polyps or pedunculated adenomas found in human colon. The author suggested the neoplasm is similar to the adenomyoma observed in A. cygnea. 206 Harshbarger reviewed a specimen with the pedal lesion and concluded that the growth was an extension of normal pedal tissues, and was a trauma-induced polyp. 81
We observed a 0.37 mm × 0.26 mm exophytic polyp (Fig. 2j) on the integument adjacent to the foot of an Actinonaias pectorosa (pheasantshell). Although similar to previously described pedal growths, this mass occurred in the integument adjacent to the foot and thus lacked pedal glandular cells.
Toxins
Because they reside in the benthos and have the propensity to filter large volumes of water and bioaccumulate chemicals, freshwater mussels are indicators of ecosystem health72,77 and may be vulnerable to toxins acquired from the water column or sediments at different points in their life cycle. 46 Toxins can be inorganic (eg, heavy metals, chlorine, ammonia, nitrates, and phosphates) or organic (eg, polycyclic aromatic hydrocarbons, pesticides, and fecal matter) and may originate from agricultural, industrial, municipal, or natural sources (eg, algal toxins). 77 Toxins have been implicated in mortality events in freshwater mussels. 58 For example, a chemical spill of an accelerant used in the manufacture of foam rubber resulted in the death of 18,000 mussels. 77 To improve the quality of mussel toxicity data, American Society for Testing and Materials International developed a standard guide for conducting toxicology tests in freshwater mussels. 8 These standards use mortality as the main indicator of toxicity, yet sublethal effects of toxins on mussel physiology and reproduction are often suspected as a driving factor for mortality and population declines. 60 Although there are textbooks on freshwater bivalve ecotoxicology, 58 knowledge is lacking on microscopic lesions instigated by various toxins, which is in stark contrast to marine mussel literature. Assessing the response of mussels to contaminants has been identified as a conservation priority. 60
Infectious Agents
Viruses
Viruses are the most abundant biological entities on Earth, with an estimated 10 31 individuals. 83 In recent decades, the number of novel viral genomes discovered has greatly increased, with aquatic environments and invertebrate samples proving to be particularly rich sources of viral diversity.5,176,208,219 These advances are primarily due to rapid progress in viral metagenomic techniques, which allow for the detection of viruses from a sample without the need for cell cultures or prior knowledge of the target virus genomes.
Hyriopsis cumingii plague disease
Hyriopsis cumingii plague disease (HcPD) is currently the only viral disease of freshwater mussels for which detailed pathological observation is available.220,221 HcPD is caused by an arenavirus, H. cumingii plague virus (HcPV), and infects cultured H. cumingii (Chinese pearl mussel), which are used for commercial pearl production in China. 222 HcPV has been documented to cause “explosive epidemics” in aquaculture settings since the 1980s, although in some cases of high acute mortality, other etiological agents have been proposed (eg, the bacterium Aeromonas veronii). 223
H. cumingii artificially infected with HcPV exhibit lesions in multiple organs. 222 The digestive gland, stomach, and intestine are the primary targets of the virus. Digestive gland epithelial cells exhibit vacuolation, such that the lumen appears shrunken, nuclear peripheralization with basophilic cytoplasm, and detachment or erosion. Within the stomach, there is loss of cilia or detachment of columnar epithelial cells. Cells in the mucosa are irregularly arranged and there is goblet cell hyperplasia. Similarly, the intestines exhibit loss of cilia and shrunken, eroded, or absent villi with irregularly arranged nuclei.
Other tissues affected by HcPV include the mantle, foot, and gills. In diseased animals, the mantle has epithelial loss with irregularly arranged myofibers and vacuolated or fragmented connective tissue. In the foot, myofibers are fragmented and vacuolated and goblet cells are enlarged and basophilic. Gill filaments are irregularly arranged with epithelial cells that are enlarged or detached and connective tissue is fragmented with vacuoles.
On transmission electron microscopy, 80–120 nm spherical, enveloped virions covered with 12-nm-long club-shaped projections with moderately electron-dense nucleocapsids were present in the cytoplasm of cells in the digestive gland, stomach, mid-intestine, gills, mantle, and foot.73,222 Virions were primarily found in the digestive gland, gill, intestinal epithelium, and connective tissues between muscle fibers. Few virions were observed in the mantle and foot of infected mussels, despite extensive pathologic changes to these tissues. 222 Intranuclear icosahedral, enveloped herpes-like virus particles have also been observed in the gonad and digestive gland of diseased H. cumingii, but the relationship between these 2 viruses and disease attributed to HcPV is unclear. 73
Challenges and considerations for the diagnosis of viral diseases in freshwater mussels
The lack of described viruses in freshwater mussels compared to marine bivalves may be due to a difference in economic importance, and thus, effort.15,163 Limited methodology for virus detection also likely has played a role. 73 Historically, diagnosis of viral infection or disease in mollusks has relied on the observation of histologic changes suggestive of viral infection and the subsequent identification of virus particles by transmission electron microscopy and putative assignment to viral families, but neither technique provides a definitive diagnosis. 15 Serologic techniques such as hemagglutination inhibition, virus neutralization, enzyme-linked immunosorbent assays, western blots, and immunohistochemistry often used to detect viruses are not possible in freshwater mussels as they lack antibody production. 154 Viruses identified by light microscopy and described by ultrastructure in freshwater mussels have lacked molecular characterization.73,222
In past years, the lack of bivalve cell lines for virus isolation has hindered additional virus characterization. 137 The only immortal molluskan cell line is that of the gastropod Biomphalaria glabrata (bloodfluke planorb) (American Type Culture Collection number CRL-1494). 80 Primary cell lines have been established for few mollusks and most often marine bivalves with commercial value. 216 Although primary cell cultures have been established for the freshwater mussels Lamellidens rhadinaeus and Dreissena polymorpha (zebra mussel; Dreissenidae),19,160 they were only maintained for 45 and 15 days, respectively, and neither proliferation nor reproduction was achieved. 160 One of the main difficulties in establishing freshwater mussel primary cell lines is a lack of knowledge about host nutritional requirements and metabolism; therefore, identifying the most suitable growth media to promote cell proliferation is challenging. 160 Other difficulties include damage to tissues during isolation, cellular disaggregation, contamination, and maintaining the appropriate physical and chemical conditions for the cells. 216 Although virus purification and genome sequencing has been completed for ostreid herpesvirus type 1 infecting larval Pacific oysters, no viral genomes for freshwater mussel viruses exist. 49
Often, detection of viruses relies upon the more complex metagenomic viral discovery methods in which samples are deep sequenced and genes resembling those of known viruses are selected for further examination to determine if they constitute new virus species. A few studies using metagenomics have begun to illustrate patterns of viral abundance and diversity in freshwater mussels.69,165,172 Viruses detected via metagenomics may represent environmental contaminants, 106 belong to parasites of the mussels, 98 or represent endogenized viruses, 120 and this needs to be considered when evaluating metagenomic data. For these reasons, to describe novel viral diseases of freshwater mussels, a combined approach of virus discovery and traditional visualization of viral particles associated with host cell pathology along with ancillary and confirmatory diagnostic techniques and sound epidemiological study designs is ideal.
Bacteria
Although bacteria are often suspected to be involved in mortality events of wild freshwater mussels, none have been confirmed as pathogens.37,73,142 Published reports of lesions associated with bacterial infections of wild mussels are scant, although histologic observation of hemocyte nodulation due to a bacterial infection in wild unionids in Italy has been noted. 37 Pathogenic bacteria have been associated with mortality in cultured mussels, especially in intensive aquaculture facilities. 213 Stenotrophomonas maltophilia and A. veronii were isolated from cultured H. cumingii from a large mortality event, and subsequent experimental infections resulted in myofiber degeneration and gill necrosis in infected mussels; intralesional bacteria were not reported. 214 Another study to investigate the immune response to bacterial infection at the transcriptomic level showed 5957 differentially expressed genes in H. cumingii challenged with A. veronii. 213
The study of bacteria in freshwater mussels is complicated by the fact that freshwater mussels constantly interact with bacteria in the aquatic ecosystem, use bacteria as a food source, 198 and host diverse bacterial taxa in health.73,185 Additionally, bacteria may be transient rather than endosymbiotic; 144 can vary by season, habitat, and in response to environmental change; 185 and can shift rapidly during mortality events. 164
Sampling methods also complicate our understanding of bacterial diseases in mussels. Many studies used lethal sampling methods to culture bacteria from drained fluids or homogenized tissues, which sometimes underwent an external disinfection step.73,186 Interpretation of results was complicated by a lack of tissue specificity as well as potential issues with sample integrity due to disinfection techniques or lack thereof. 114 Selection of appropriate culture medium and incubation temperature is also an important consideration for bacterial isolation. 73
Recent studies have analyzed the microbiomes of healthy mussels,40,89,113,132,204 and a few have compared the bacteria from moribund and healthy mussels,114,164,173,183,186 with some investigations focusing on hemolymph samples,114,164,183 which allow for nonlethal sampling, 74 and provide a better sample for identifying bacteremia. Although primary pathogens have not been confirmed, some patterns have been noted. Bacillus spp. have been isolated more frequently from healthy mussels. 114 Aeromonas spp., generally considered an opportunistic pathogen, has been identified with relatively high prevalence from both healthy and diseased populations.114,164,183,186 However, the prevalence of Aeromonas spp. is higher during mortality events in some cases,114,164,186 but not others. 183 Yokenella regensburgei was consistently isolated with high prevalence during periods of active pheasantshell mortality, but rarely before or after these events. 114 Despite the lack of evidence for pathogenicity associated with Y. regensburgei, this bacterium may be a bioindicator of a contaminant that could play a role in the mortalities. 114
Pathologists can play a role in these investigations by assessing any hematologic or tissue changes caused by bacteria, and further identifying bacteria with special stains, immunohistochemistry, or transmission electron microscopy.
Fungal and Fungal-Like Pathogens
An estimated 5 million species of fungi are on earth and occupy a wide range of niches. 162 However, in marine bivalves, as is the case in freshwater mussels, few true fungal infections have been identified, and whether the fungi are primary or secondary invaders is not always clear. 71 The fungus Ostracoblabe implexa (incertae sedis) is known to be pathogenic, causing severe shell disease in several species of oysters. 71 Unidentified fungi, and several fungus-like pathogens including oomycetes (Chromalveolata) and microsporidia (Opisthosporidia), have been seen in freshwater unionid mussels. A single, thin, moribund P. complanata with a deformed shell exhibited growths in its marsupial gill that microscopically contained fungal hyphae and suspect Periodic acid-Schiff-positive, germinating fungal spores. 155 Phagocytosis and encapsulation of the spores was observed in many tissues including the mantle. Brown streaks were observed in the marsupia of Unio pictorum (painter’s mussel), and fungal hyphae were observed among normal and diseased embryonic glochidia. 156 It has been suggested that hyphae from this case were likely oomycetes. 73 There is a brief mention of “Oomycetes saprofites” infecting unionids, but no gross or histologic descriptions are provided. 37
The most thoroughly described fungus-like infection of mussels comes from a recently described microsporidian. Microsporidia are unicellular intracellular organisms once classified as protists, but are now considered to be more closely related to fungi. A single species, Knowlespora clinchi, infects A. pectorosa.30,108 Spores of K. clinchi occur in the cytoplasm of oocytes in ovarian acini (Fig. 3a), in lumina of ciliated gonadal ducts, in water tubes of the gills, and free within the ovary. Often no inflammatory response is associated with the spores. Spores can be accentuated with Giemsa stain and are Gram-positive and acid fast. Transmission electron microscopy shows proliferative and sporulation stages of the microsporidia, and identification of their unique organelle, polar filaments, in mature spores is useful in the diagnosis. Sequencing of the small subunit rRNA gene using ovarian tissue is also diagnostic. The significance of the microsporidia at the individual and population level is undetermined, but heavy infections appear to contribute to decreased fecundity and recruitment. If vertical transmission is possible, infected broodstock may transmit microsporidia to progeny used in restoration efforts. Future work to identify fungal pathogens in mussels could utilize a combination of traditional culture based methods, histology, and molecular techniques. 162
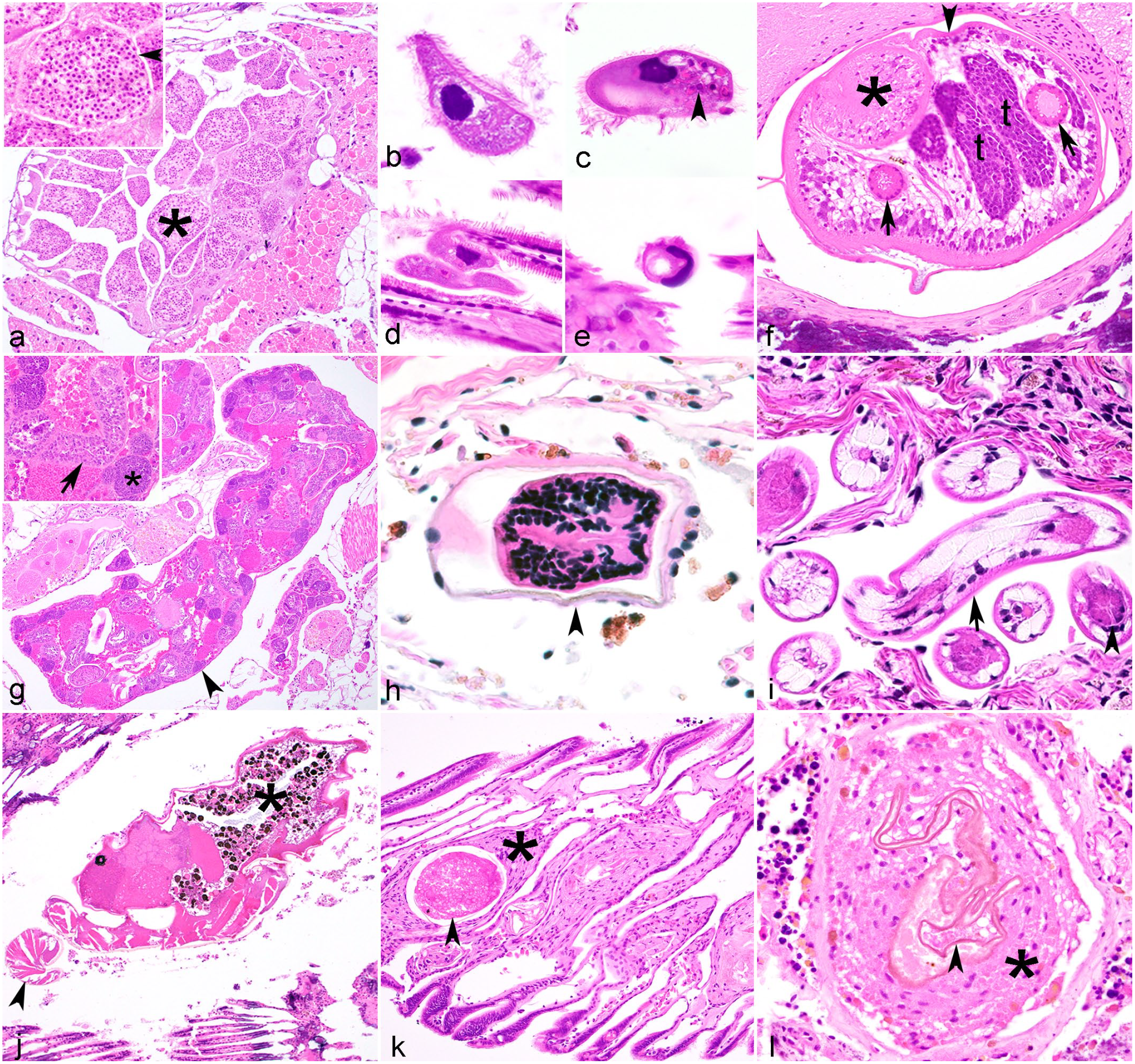
Infectious agents in freshwater mussels. Hematoxylin and eosin.
Ciliated Protists
Worldwide, over 8000 species of ciliated protists (Phylum Ciliophora) occupy a wide range of aqueous habitats. 126 Although most species are free-living some are symbionts with relationships varying from mutualistic, commensalistic, parasitic, to pathogenic. Ciliates are covered by a cell membrane and are easily recognized by the presence of cilia or kineties on their cell surface, a cystosome surrounded by cilia, a contractile vacuole, and nuclear dualism with both a macronucleus and micronucleus (Fig. 3b–e). 126 Although light microscopy is useful in the initial identification of ciliates, further identification requires the use of traditional description by morphology and morphometrics, and molecular methods. 126 A detailed review of techniques for the speciation of ciliates has been previously published. 3 In freshwater mussels, scuticociliates, peritrichs, and hymenostomes have been described from the class Oligohymenophorea and rhynchodids from the class Phyllopharyngea.
Scuticociliates of the family Conchophthiridae genus Conchophthirus have only been reported from freshwater bivalves. 37 They were first discovered in 1829 in the mantle cavity of unionid mussels and placed in the genus Leucophrys and species anodontae. 12 In 1861, they were placed in the new genus Conchophthirus. 12 Scuticociliates are the most commonly reported ciliate in freshwater mussels with a worldwide distribution. 12 Prevalence at some sites were reportedly 100% with more than 1000 ciliates observed in an individual.23,158 Seasonal changes in prevalence with Conchopthirus spp. have been documented with ciliates more plentiful in warmer months. 104 Ciliate prevalence may be reduced in habitats with poor water quality and contaminants. 11
Conchophthirus spp. are approximately 100 µm in length, laterally flattened, and have an elliptical body covered in dense cilia with a mouth located in the middle of the body. 37 Conchophthirus curtus, C. anodontae, and C. magna vary in size and have differences in host specificity. 105 Mussel behavior may make hosts more suitable to some species as is observed with “shell gape” for C. curtus. 12 Coinfections can occur with both C. magna and C. anodontae documented in infected mussels. 14 Conchophthirus discophorus, usually found in fingernail clams (Veneroida, Sphaeriidae), has been observed in unionids. 14 Location on the host varies with ciliates found in the mantle cavity, gills, palps, 14 and foot in multiple species. 13 All 3 ciliate species are considered to be commensals, 105 evidenced by the presence of sloughed epithelial cells, algae or bacteria in food vacuoles, with no detrimental effect on hosts. 13 A review of the molecular phylogeny of Conchophthirus has been recently published. 14
Peritrich ciliates have a rich species diversity and are successful symbionts likely due to their ability to attach to a variety of substrates using either an adhesive disk or a scopula. 128 Members of the orders Mobilida and Sessilida have been described in freshwater mussels. Members of Mobilida are mobile with a skeleton of denticles and attach to their host by an aboral adhesive disk. 128 Trichodina unionis was described in the labial palps and less often gills of Unio crassus batavus (thick shelled river mussel), A. cygnea, 79 and U. pictorum. 161 Trichodina sp. was reported in different mussels species, but no description was provided. 13 A possible trichodinid was seen in the gills of E. complanata, but it differed in size and shape from T. unionis. 41 Sessile peritrichs attach to the host using a stalk or scopula. 128 A scyphidiid peritrich of the genus Mantoscyphidia was tentatively described in the gill arch of E. complanata with no associated inflammatory response. 41
Few species of hymenostomes have been reported in habitat surveys, indicating a patchy distribution. 128 Veterinarians are probably most familiar with the hymenostome Ichthyophthirius multifiliis, a fish parasite. A hymenostome, possibly Tetrahymena rostrata, was reported from a unionid mussel, 13 but no description was provided.
There are only a few reports of ciliates from the class Phyllopharyngea in freshwater mussels. The rhynchodid, Heterocinetopsis unionidarum, was identified on the gill of E. complanata, 38 A. cygnea L. and U. pictorum L., 99 and gill and palp of Pyganodon grandis (giant floater) and Lasmigona complanata (white heelsplitter) with no ill effects. 13 Rhynchodids possess a suctorial tentacle used to attach to the host and are small with an average length of 20–50 µm. 127
Aspidogastrea and Digenea
Trematodes infecting unionids include Aspidogastrea and Digenea. Aspidogastrea are considered primitive trematodes, which have a simple life cycle (many have a direct life cycle). In contrast to Digenea, Aspidogastrea have direct development and do not develop from multiple larval stages and, depending on species, may complete development in a single mollusk host or self-fertilize. Aspidogastrids infecting unionids include Aspidogaster conchicola, Cotylaspis insignis, Cotylogaster occidentalis, and Lophatspis interiora.73,84,94,112 Digenean trematodes have complex life cycles usually involving 1 or more molluskan intermediate hosts and a vertebrate definitive host. Unionids are first or second intermediate hosts for several families of digeneans including Bucephalidae, Allocreadiidae, Gorgoderidae, Echinostomatidae, and Apocreadiidae. 73
Diagnosis of trematode infection is accomplished during dissection or needle biopsy, with subgross examination or histopathology. Superficial trematode larvae may be large enough to discern grossly as small nodules, and some are light orange or yellow.159,175 Other associated gross lesions include shell deformation, nacre deposits (ie, pearls), or nacre staining of the inner shell surface.68,73,146 More typically, larvae are observed histologically, where presumptive taxonomical diagnosis is based on host species, larval stage, size and shape of larvae, and tissue infected. 112 Definitive diagnosis requires parallel subgross morphological examination and molecular techniques for identification. 27
Aspidogastridae adults are distinguished by the presence of well-developed gonads, ova, or egg capsules and a ventral adhesive disk with rugae featuring alveoli (ie, suckerlets) rather than ventral or posterior suckers (Fig. 3f). Aspidogaster adults can graze on host epithelium or feed on hemolymph.67,94 Cotylaspis insignis typically infests the gill and mantle cavity and C. occidentalis parasitizes the intestine; associated lesions have been reported for neither.64,67 In contrast, A. conchicola is usually within the nephridium or pericardial cavity. Associated pathology is variable but may include erosion of adjacent epithelium or hemocytic to fibrous encapsulation around the adults or eggs.94,152
Digenean sporocysts and rediae are asexually reproducing stages observed in the tissues of a first intermediate host (mollusks may be first or second intermediate hosts or paratenic hosts). They are typically invasive and occupy a large percentage of a tissue, usually the gonad and surrounding tissues. Sporocysts may be spherical, ovoid, elongate, or branched, and have an outer tegument of simple cuboidal to columnar epithelial cells with clear to granular cytoplasm (Fig. 3g). In contrast, rediae are elongate and have 2 to 4 ambulatory buds that facilitate movement of larvae within its host, as well as a mouth, pharynx, and cecum. 29 Centrally, the sporocysts and rediae have a brood chamber containing cercariae in various stages of development ranging from germinal balls to mature cercaria. The mature cercaria usually has a tail, mouth, pharynx, intestine, pigmented eyespots, and various types of glands. The anterior end may have a stylet or minute spines, ventral suckers, genital primordium, and caeca, although these structures may be difficult to discern in a tissue section. In contrast, metacercariae, typically encysted within the tissues of the second intermediate host, are round, and are contained within a hyaline capsule (Fig. 3h). Less commonly, and when forming in the first intermediate host, metacercaria may develop within sporocysts.
Pathogenicity of larval digeneans vary by trematode species, but largely reflect tissue tropism and developmental stage. The sporocysts of Bucephalidae, particularly Rhipidocotyle spp., localize to gonad, where heavy infections may significantly reduce gametogenic tissue (ie, “parasitic castration”), but can also involve the gill, digestive gland, and kidney.61,191,209 Bucephalid cercariae have bifurcate tails and infections involving the gonads have been putatively associated with hermaphroditism.103,112,194 Phyllodistomum spp. form C-shaped metacecariae within elongate sporocysts often in the gill of the first intermediate host, but may also involve gonad, digestive gland, and kidney.73,112,140 The sporocysts may be grossly evident as yellow to white stripes in the gill. 159 Echinostomatidae metacercariae uncommonly infect unionids, 130 but are histologically identified by double wall cysts and collar spines. 112 They more commonly infect D. polymorpha and are considered benign, encysting within mantle epithelium. 110 Metacercariae of Polylekithum spp. also tend to encyst in the mantle.68,175 Homalometron spp. sporocysts and metacercariae infect a wide variety of tissues.41,75 Microscopic lesions associated with larval digeneans may be absent or include parenchymal compression or atrophy, variable hemocyte encapsulation, and nacrezation when involving the mantle.41,68,140 The variation in severity and cellular composition of hemocyte response induced by different trematode larvae species has not been well studied in unionids.
Although trematode infections are common among unionids and can negatively affect their growth and reproduction,66,75 they have not been documented to have a primary role in mass mortality events and are considered unlikely to be responsible for declines. 133 However, environmental stress is associated with increased virulence from trematode infections, 100 and digenean infections may instigate host immunosuppression, 37 which could explain relatively high prevalence or intensity of trematode infections in declining mussel populations. 85
Nematoda
Five publications have documented putatively parasitic nematodes from members of Unionoida, mostly from the United States. Gross observations of Ascaris-like nematodes or Ascaris sp. were reported in the digestive tracts of unspecified mussels42,44 and stomach of P. grandis. 207
Adult Hysterothylacium sp. (Anisakidae) are parasites of fish, and their larvae use fish and invertebrates as intermediate hosts. Third-stage larvae of Hysterothylacium sp., found in the pericardial cavity of Diplodon suavidicus, were conspicuous through the translucent dorsal region of the integument based on a gross examination of de-shelled mussels. 121 Although prevalence (82%) and intensity (4.71) were high, there was no necrosis, cellular response, or any physiological impairment to the infected hosts.
Second-stage larvae of Ascaridomorpha sp. were observed amongst pedal myofibers, in intestinal epithelium, and in the mantle edge of Cambarunio nebulosa (Alabama rainbow mussel). 136 The prevalence was high (74%) and nematodes were most frequently observed in the ventral margin of the foot and intestinal epithelium. Two individuals were infected with more than 100 nematodes. Histologically, nematodes were approximately 25 µm wide with a nonrefractile cuticle, pseudocoelom, and alimentary canal (Fig. 3i). No inflammatory response was associated with the nematodes. Sequencing of the 18S rDNA showed 99% similarity to ascarids from aquatic or semiaquatic vertebrates. C. nebulosa is likely an intermediate or paratenic host for this nematode.
Acari
Commensalistic and parasitic mites, Najadicola (Najadicolidae) and Unionicola (Unionicolidae) spp., commonly occur in unionids. 56 Najadicola ingens, the only described species, infects at least 30 species of mussels in North America and Asia. 56 Unionicola has over 200 species that infect numerous species of mussels worldwide. 135 The prevalence may reach 100%, but the number of mites in an individual can vary greatly with N. ingens infected species only containing a few mites. 73 The life stages consist of an egg, larva, nymphochrysalis, nymph, teleochrysalis, and adult. 56 Mites may injure mussels through feeding, oviposition, or chrysalis formation. 135
Najadicola ingens occur in the suprabranchial chambers, gills, and pericardial chambers of unionids. 179 Eggs are deposited in gelatinous masses between host gill lamellae, 179 and may be grossly evident as nodules or “papillae,” up to 5 mm in diameter, near the base of the anterior gills.95–97 N. ingens may lower the fecundity of its hosts because male and female mites, and their egg masses, can occupy suprabranchial chambers of the gills and potentially obstruct passage of host embryos into the marsupia.96,97
Unionicola spp. deposit eggs into superficial tissues of the mantle cavity, and nymphochrysalises and teleochrysalises are often encysted in the superficial tissues of the mantle, foot, labial palps, and gill, whereas other stages may be predatory in the environment, tending to reside on host tissue surfaces within the mantle cavity.59,200 Adults can be discerned grossly as pin-point black foci on the gill and mantle. Gross lesions are often attributed to eggs and chrysalises encysted in connective tissue, and include cuplike depressions of the epithelium, “hypertrophy of the mantle and visceral mass tissues,” mottled white-to-yellow-to-brown pigmentation of the mantle, or minute foci of nacrezation (ie, blister pearls).56,61,135,139 Whether adult mites may also induce foci of nacrezation is unclear.22,56,62
Histologically, adult mites (Fig. 3j) and embedded subadult stages have a well-defined, eosinophilic refractile cuticle surrounding an oval idiosoma with appendages, striated muscle, nervous ganglia, ceca, and visceral eosinophilic granules. 135 Some are surrounded by a thin hyaline membrane. Unionicolid eggs are oval, consisting of a series of eosinophilic yolk globules surrounded by a thin shell (Fig. 3k).2,135,212
Encysted Unionicola spp. stages may not have any associated microscopic lesions, or may locally compress adjacent tissues, be associated with mild hyperplasia of unspecified adjacent tissues, or have associated focal hemocytosis or hemocytic encapsulation.135,139,212 Portions of adult Unionicola spp., especially pedipalps, may be embedded in gill or mantle tissues, causing mechanical injury or focal hemocytosis, and may be surrounded by a hyaline membrane.2,16,17 Cuticular remnants may be derived from exuvia or portions of mites that died in the host. They are refractile to hyaline, and when not clearly representing an anatomic structure are curvilinear to lamellar (Fig. 3l).2,135 Mite remnants are typically associated with a host cellular response ranging from focal hemocytosis to hemocytic encapsulation, and occur in various tissues including the ventricular myofibers, pericardial gland, serosa of the intestine, gonad, and muscle of the foot, indicating larval or adult mites sometimes burrow deeper into the visceral mass.2,135
Diptera
Chironomids (Diptera: Chironomidae), or “non-biting midges,” are a diverse group of freshwater insects that usually have aquatic larvae. 116 They are holometabolic with a larva hatching from an egg that goes through 4 larval instars before becoming a pupa and finally becomes a winged adult. 201 Five species have been observed in mussels where they are described as symbionts or parasites.22,62,63,70,166
In the first record of chironomid larvae inhabiting a bivalve, Glyptotendipes (possibly Glyptotendipes paripes), measuring 12 mm × 14 mm × 1 mm, were observed in the extrapallial cavity (between the nacre and mantle), or embedded in the mantle tissue of A. cygnea. 22 The point of entry of the chironomids is not known, but a point of shell damage created for the study is a possibility. 21 Evidence of the movement of larvae in the extrapallial cavity was observed grossly as whitish tracks and scars on the mantle surface. Histologically, the white tracks had large collections of hemocytes (or “amoebocytes”) in subepithelial tissues. Although G. paripes feeds on phytoplankton, the gut of the embedded larvae contained material similar in appearance to mantle epithelial cells and hemocytes, indicating utilization of the mantle as a food source and a parasitic relationship.
Baeoctenus bicolor and a second species, possibly related to Phycoidella, were found in the gills of Pyganodon cataracta (eastern floater) and U. implicata 70 As much as 50% of gill tissue was absent in some mussels, demonstrating that larvae (instars III and IV) were actively feeding on gill tissue. Infections were seasonal with invading instar III first observed in the fall, pupae not seen until late spring, and emergence occurring in summer. No chironomids were observed in mussels collected from late June until early November.
Larvae and pupae of Xenochironomus canterburyensis were found in Echyridella menziesii (kākahi).62,63 Infection with these commensals is also seasonal, with the chironomids relying on seasonal growth of the mussel shell for successful pupation and emergence. In summer, instars I and II were found in the extrapallial cavity and by winter had migrated as instars III and IV to the posterior margin of the valve near the inhalant siphon. In the spring, growth of the periostracum leaves instar IV outside of the extrapallial cavity where it pupates and later emerges as an adult. 62 The change in location of the larval stages was mirrored by a change in diet. The digestive tracts of second- and third-stage larvae appeared to contain sloughed mantle cells, mucus, and detritus indicating these larvae feed on mantle tissue while in the extrapallial cavity. Fourth-stage larvae, positioned in the inhalant siphon groove, contained epiphytic diatoms in their digestive tract consistent with what would be growing on mussels, aquatic plants, and rocks in that area. 62
Ablabesmyia sp. were observed between the anterior gills of Potamilus purpuratus (bleufer) and other unionids, and detailed morphologic descriptions are provided. 166 Grossly, instars were bold orange-pink, which made them easy to identify against the off-white tissues of the host. Tissue damage caused by the insect larvae was evident, but no mussel tissues were found in the digestive tracts of instar IV. It was unclear whether larvae pupate within the mussel or in the environment. Chironomids were most common in habitats with little or no flow and a fine sandy bottom. Of note, Ablabesmyia sp. and Unionicola sp. were never colocated on a demibranch, indicative of a mutually exclusive relationship. Additional chironomid larvae observed included Orthocladius dorensus in Actinonaias ligamentina (mucket), Psectrocladius sp. in Lampsilis hydiana (Louisiana fat mucket), Pseudochironomus sp. in Cyclonaias pustulosa (pimpleback) and Glyptotendipes sp. nr. lobiferus in Obliquaria reflexa (threehorn wartyback).
Odonata
Gomphidae, a member of Odonata (dragonflies and damselflies), includes clubtails, which like all dragonflies, have an aquatic larval period. 1 There is a single report of a Gomphus miliataris nymph in the mantle cavity of Popenaias popeii (Texas hornshell). 119 Grossly, gills had ragged notches and semicircular portions of tissue missing. Gill damage was caused by nymphs feeding on the gills evidenced by glochidia and gill tissue in their gut. Gill damage was often extensive with some mussels losing 1 or more demibranchs. Gomphids preferentially infected outer gills and gravid females, while fewer nongravid mussels were infected.
Discussion
Freshwater mussel die-offs have been observed in the United States for several decades. The need for their biomedical investigation was emphasized over 30 years ago, 142 yet little progress has been made in the understanding of potential pathogens that may affect freshwater mussel health. Continued declines of freshwater mussel populations highlight an ongoing need to develop a more coordinated and systematic approach to health investigations. Although well-established systems exist in the United States for the monitoring, surveillance, and reporting of diseases in domestic livestock, avian, lagomorph, and aquaculture species, 10 fewer networks exist for reporting mortality events of wildlife species. The development of a national system for reporting freshwater mussel mortality events complete with detailed event history forms would aid in the coordination and response to these events. Even with rapid reporting, biologists often arrive to the mortality site to find dead mussels, which are not suitable specimens for diagnostic investigations. Visually distinguishing between healthy and sick mussels remains problematic and can result in the sampling of healthy animals from mortality events. Establishing baselines for biomarkers and using them to distinguish between healthy and diseased mussels could aid in the investigation and characterization of unionid disease.143,202 A comprehensive guidebook for the clinical, gross, and histopathologic examination of freshwater mussels and appropriate diagnostic tests would help to standardize data collected by biologists and pathologists.
Currently, the World Organization for Animal Health (founded as the Office International des Epizooties) lists no diseases of international concern for freshwater mollusks. 210 This lies in contrast to marine bivalves for which 7 diseases, including viral, protozoal, and bacterial diseases, are listed. 210 The disparity may be in part due to the market value of these species. Although freshwater bivalves were once sought after for the manufacturing of buttons and later for use of their shell in the Japanese pearl industry, 129 their market value is small compared with the current billion-dollar marine bivalve market. 189 However, as filter and deposit feeders, mussels provide crucial ecosystems services including nutrient cycling and water purification, 197 and their loss can result in long-term deterioration of ecosystem functions. 55 For these reasons, hatcheries in many U.S. states have programs to restore imperiled mussels through restocking programs. Diseases in marine bivalves can have a significant economic impact so more is done to investigate, identify, document, and control disease spread and introductions.31,35,71,148,167,218 For example, Perkinsus marinus, ostreid herpesvirus, and disseminated neoplasia, diseases for which no counterparts are known in freshwater mussels, can all cause significant mortality in commercially important bivalve species and have been extensively studied.18,49,180
With light and electron microscopy as the best tools to characterize host–pathogen interactions, pathologists have a large role in documenting new pathogens and diseases. With the increased use of metagenomics, pathologists can play a vital role in bridging the gap between detection of an infectious agent and confirmation of the role of that agent in disease. Once a pathogen is discovered and its sublethal or lethal impact on the individual is known, investigating the effect of the pathogen on the population is important. Although some authors provide prevalence and intensity data for infections in freshwater mussels, often no additional studies are available to compare data over time or in different locations. Such “baseline data” are crucial for determining the effect of disease on mussel populations. Disease surveillance of mussel populations may allow for the collection of prevalence and intensity data, the evaluation of temporal and spatial trends, and the determination if disease is epizootic or endemic in that population. A better understanding of potential disease effects may be obtained through further examination of relationships between hosts, pathogens, and the environment. For the marine protistan parasite P. marinus, it is well established that the parasite thrives in high temperature and high salinity, and that a combination of low temperature and low salinity has a synergistic effect on low cell viability. 109 These data are useful in the prediction of epizootics and in the management of oyster populations. The evaluation of diseased populations over time has also led to the detection of disease resistant oysters, which can be used in propagation efforts. 180
Finally, translocated freshwater mussels are typically not screened for pathogens in part due to our poor understanding of mussel disease. 78 Pathologists can participate in disease surveillance in freshwater mussel hatcheries to ensure successful propagation programs and the translocation of healthy progeny to natural watersheds in an effort to restore decimated populations and prevent introduction of pathogens into naïve ecosystems.
Footnotes
Acknowledgements
We thank Tim Lane, Brian Watson, Sarah Colletti, Tiffany Leach, Brett Ostby, Jeronimo Silva, Gerry Dinkins, Augustin Engman, and Caitlin Carey for assistance with sample collection, and Stephen A. Bullard and Rachael Hoch for providing specimens.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. The findings and conclusions in this article are those of the authors and the U.S. Geological Survey and do not necessarily represent the views of the U.S. Fish and Wildlife Service.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the U.S. Fish and Wildlife Service (grants F20AC10617 and F21AC00707) and the U.S. Geological Survey, Ecosystem Mission Area.
