Abstract
There have been unpublished reports of a follicular dysplastic syndrome in adult white-tailed deer (Odocoileus virginianus; WTD), known colloquially as “toothpaste hair disease.” The current report aims to describe the gross and histologic lesions in skin samples from 2 adult WTDs that presented to the Wisconsin Department of Natural Resources and the Wisconsin Veterinary Diagnostic Laboratory with reports of hair loss in 2018. Both cases were grossly alopecic with sparing of the distal extremities and variably the head and neck. Histologic features included hair follicles and adnexa present in relatively normal numbers, dilated and misshapen follicles, and dysplastic hair bulbs. Hair follicles were empty, contained fragmented and irregular hair shafts, or contained concretions of keratin. Hair bulbs were rarely infiltrated by small lymphocytes, suggestive of alopecia areata as a cause of the gross appearance. This condition does not appear to be directly responsible for WTD mortality but presumably would predispose affected animals to greater environmental exposure. Evaluation of additional affected individuals is warranted to further evaluate for features of alopecia areata.
Causes of alopecia in animals are numerous and relatively well characterized when related to infectious, inflammatory, immune-mediated, endocrine, nutritional, and traumatic etiologies.7,12 Alopecic conditions in the absence of such underlying conditions are recognized although frequently not well understood. These less-understood alopecic conditions are best described and reported in domesticated species, especially in dogs and cattle,6,7,11–15,18 while rarely documented in free-ranging or captive wildlife species.1–3,14,21
Follicular dysplasia is an uncommon cause of alopecia reported to occur in several animal species and in humans.7,11,12,14,15,17,18 Few reports of follicular dysplasia in wild animal species exist in the literature.1–3,8,9 The aim of this report is to present and document two cases of alopecia in free-ranging, wild white-tailed deer (Odocoileus virginianus; WTD) in Wisconsin with follicular dysplasia, which has been referred to as “toothpaste hair disease” in the vernacular of the Great Lakes and Mississippi Valley regions.9,10
In the fall of 2018, previously frozen, formalin-fixed skin samples from 2 hunter-harvested, free-ranging and variably alopecic, adult male WTDs were submitted by the Wisconsin Department of Natural Resources (WDNR) to the Wisconsin Veterinary Diagnostic Laboratory (WVDL) for histologic evaluation. Harvest dates for these submissions occurred in late October in Polk County on the west side of the state bordering the Mississippi river (case 1) and in mid-November in Sheboygan county on the state’s eastern side bordering Lake Michigan (case 2). Neither carcass was received by the WVDL, and due to the free-ranging nature of these wild WTDs, clinical histories were minimal for both cases. Case 1 was believed to be seen alive by trail camera prior to being hunter-harvested with alopecia noted at that time. Additional samples of grossly normal deer skin were frozen and processed similarly as a histologic control (Fig. 1). Gross images of these adult male deer were provided by the hunters and WDNR (Fig. 2a, b).
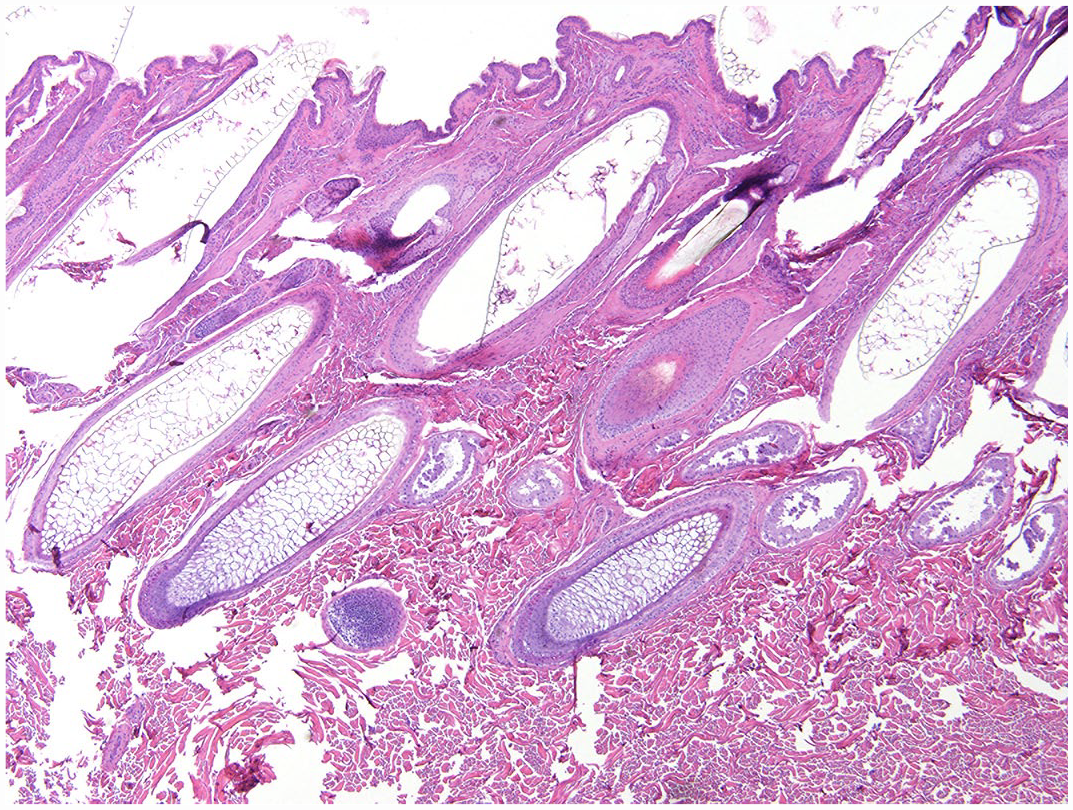
Skin from the hindquarters of a grossly normal haired white-tailed deer buck. Sections were frozen prior to processing to approximate artifactual changes for comparison. Note rounded, fish-scale-like cuticular pattern and uniform diameters of hair shafts. Hematoxylin and eosin.
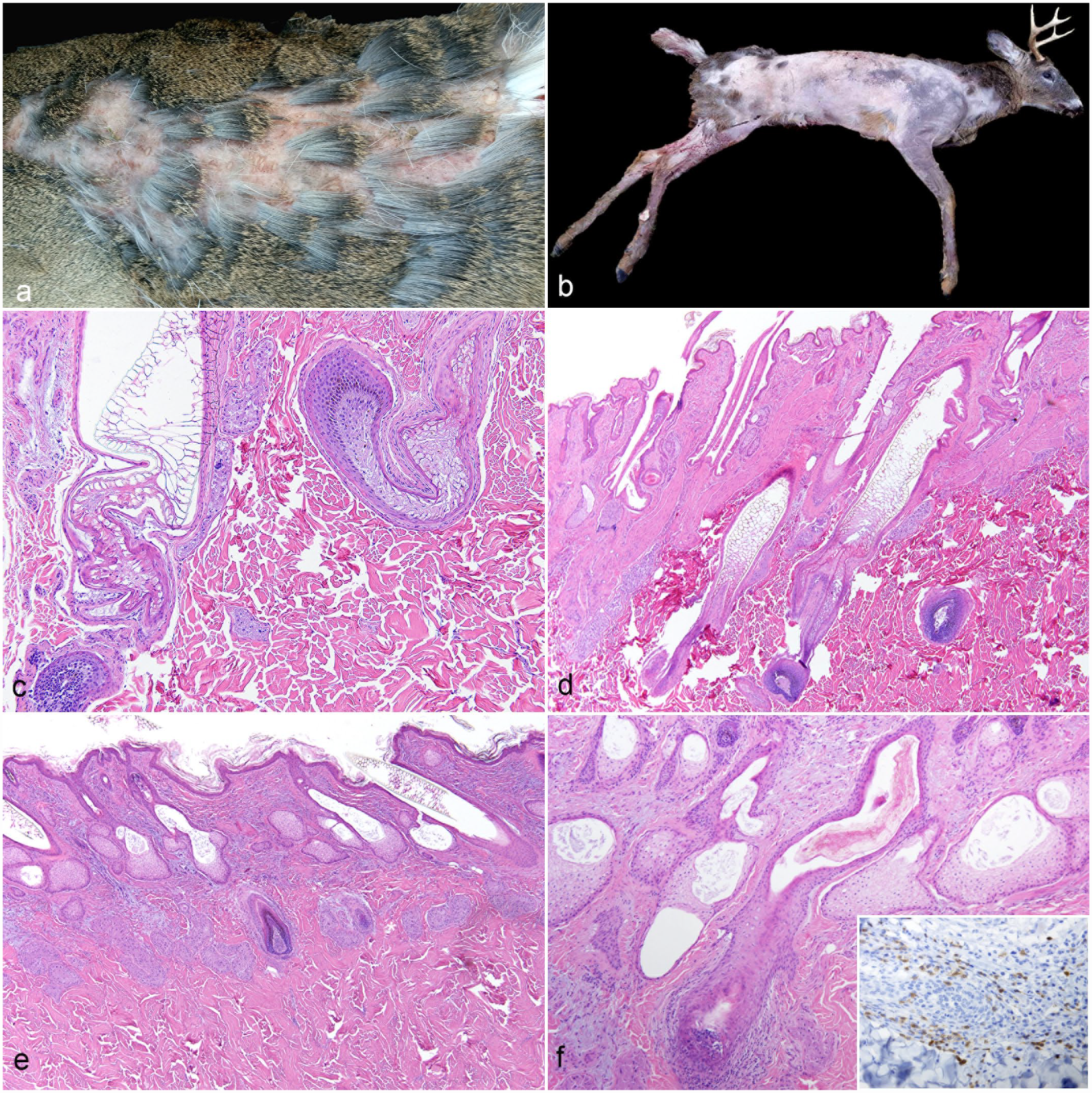
Follicular dysplasia in 2 adult white-tailed deer: (a) Case 1, deer rump skin. Patchy alopecia. (b) Case 2, deer carcass. Severe and extensive alopecia of the trunk and limbs with relative sparing of the head, neck, rump, and tail. (c) Case 1, haired skin. Undulation and contortion of hair follicles with conforming hair shafts. Marked infundibular lumen and hair shaft dilation with vacuolation and distortion of the hair shaft scale pattern. Hematoxylin and eosin (HE). (d) Case 1, haired skin. Follicle density is within normal limits, but with frequent dilation of follicles, hair bulb malformation, and arrector pili muscle prominence. HE. (e) Case 2, haired skin. Atrophic follicles with superficial dermal hair bulbs (*). HE. (f) Case 2, haired skin. Wavy hair follicles with infundibular hyperkeratosis and prominent and hyperplastic sebaceous glands with dilated ducts. A hair bulb is surrounded and infiltrated by small numbers of lymphocytes and plasma cells. HE. Inset: Immunohistochemistry for CD3. Positively staining round cells infiltrate the affected hair bulb.
Case 1 was a 3-antler-point buck observed to be moving slowly prior to harvest and was subsequently found to have regionally extensive alopecia over the rump. Grossly, the rump region was sparsely haired, with retention of patches of paintbrush-like tufts of hair (Fig. 2a). Histologically, hair follicles and sebaceous and apocrine glands were present in normal numbers beneath a normal overlying epidermis (Fig. 2c, d). Hair follicles were variably sized and formed with undulating to contorted, uneven walls and frequent infundibular dilations that ranged from 500 to 900 µm in diameter. Occasionally, isthmus segments were also significantly dilated. Hair shafts assumed the forms of the aberrantly shaped, attenuated, and dilated follicular lumina, with those within dilated segments exhibiting distorted, lattice-like cuticular scale profiles (similar to normal shafts in Figure 1 and previous descriptions4,5). Multiple hair bulbs also exhibited variability in shape and organization. Overall, relatively few histomorphologically normal hair follicles and hair shafts were observed in the rump skin sections for this animal. Arrector pili muscles were prominent throughout the sections. Few lymphocytes and plasma cells multifocally infiltrated the dermal interstitium.
Case 2 was a 5-antler-point, 2.5-year-old buck that was nearly hairless except for the head, neck, and areas over the rump (Fig. 2b). Histologically, hair follicles and other adnexal structures were present in normal numbers (Fig. 2e, f). The epidermis was hyperkeratotic, and the basal epithelial layer was mildly pigmented. Hyperkeratosis extended into the follicular ostia. Infundibular segments were dilated with accumulation of intraluminal laminated keratin and dense aggregates of keratinaceous debris (infundibular hyperkeratosis). Hair follicles exhibited variable contours with empty lumina or irregular, sometimes attenuated, intraluminal hair shafts or hair shaft fragments. The normal cortical-medullary structure of the hair shafts was disrupted and distorted. Hair follicles were often atrophied or in telogen phase and were situated within the superficial dermis. Occasional hair bulbs were surrounded and sparsely infiltrated by predominantly small numbers of lymphocytes, which immunolabeled for CD3 (Fig. 2f, inset), and fewer plasma cells. Sebaceous glands were mildly hyperplastic with ectatic ducts containing keratin fragments. Apocrine glands were ectatic and contained eosinophilic, acellular, amorphous material. In a few areas, minimal pigmentary incontinence was present within the superficial dermis and surrounding apocrine and sebaceous glands. Multifocally, few lymphocytes and plasma cells infiltrated the dermal interstitium. Tissue Gram and Grocott’s methenamine silver stains did not identify microorganisms. Immunohistochemistry for bovine viral diarrhea virus was negative (Supplemental Methods).
In mice (eg, nude mice) and humans, numerous genetic factors associated with noninflammatory follicular dysplastic and congenital alopecic conditions have been identified and studied.15,17,20 The inheritance patterns of such alopecic disorders have also been studied in domestic animal species, chiefly dogs, cattle, and cats,6,7,11–14,18 with few genes being definitively identified as being responsible,6,10,11 although many conditions exhibit strong breed and/or coat color predilections.7,12,13,18 Color coat follicular dysplasias are associated with melanin pigment accumulations within hair shafts, follicular lumina and walls, and/or bulbs, with structural weakening and loss of affected hairs. Published reports of follicular dysplasia in adult domesticated ruminant species are few in comparison, with some cases exhibiting strong associations with coat color.11,12 Congenital hypotrichosis conditions are among the most widely recognized alopecic conditions in cattle, often exhibiting other ectodermally derived defects in conjunction with follicular dysplasia, with some being lethal in effect.6,10,12,21
Causes and presentations of follicular dysplasia in WTDs are also likely to be diverse but are far less well characterized and documented due to fewer opportunities to observe, capture, fund, and study individuals affected by this alopecic condition over extended periods. Summer hair coats of upper-Midwest WTDs are thinner in hair shaft density and reddish in color. By late summer to early fall, it has been shed and replaced by a thicker, brownish-gray winter hair coat. 16 In one study, winter coats of yearling deer were reported to have the greatest density of hair follicles and sebaceous glands in the skin over the dorsal tail and the neck. 4 Proportionately, guard hair to wooly hair follicles and shafts were present in a 1:2 ratio in all regions except the tail. Primary guard hairs were straight in conformation with maximum diameters of 260–310 µm over the shoulder and back regions. 4 Deer guard hairs are additionally described as having fluted-type wine glass–shaped root profiles, with the proximal root segment being thin (stem portion) which then flares distally (bottom of the glass portion) to maximum diameters in the neighborhood of 300 µm. 5 Most of the dysplastic hair shafts in case 1 of this report exceeded this value, frequently exhibiting diameters of 500–900 µm.
Two previous cases describing the presence and pathology of follicular dysplasia as “toothpaste deer disease” in WTDs are present in the Joint Pathology Center Wednesday Slide Conference archives.9,10 Some of the histologic features common to both the Wednesday Slide Conference cases and the two Wisconsin deer in this report are highlighted in Table 1 and include hair shaft attenuation, hair shaft coiling and fragmentation, and mononuclear dermatitis. All four cases had some degree of mononuclear inflammation, occasionally infiltrating hair bulbs in some animals. Alopecia areata is characterized by nonscarring, presumably immune-mediated, alopecia with involvement of cytotoxic T lymphocytes and helper T cells, similar to findings described here.7,12,19 Immunofluorescence assays indicate targeting of trichohyalin, a structural protein necessary for inner-root-sheath differentiation, in some cases of canine alopecia areata. Clinical severity of affected individuals and their responses to immunosuppressive therapy are variable, suggesting the existence of subsets of alopecia areata, which differ in antigen targeting. 7 The variation in morphologic findings in deer cases detailed here may be attributed to similar individual variation, as well as presentation during different stages of the disease.
Histologic findings for cases of alopecic white-tailed deer described with “toothpaste hair disease.”

Abbreviations: WVDL, Wisconsin Veterinary Diagnostic Laboratory; JPC WSC, Joint Pathology Center Wednesday Slide Conference.
A single report of congenital alopecia characterized as hypotrichosis was reported in a wild 4-month-old, male WTD fawn in South Dakota, with some histologic features similar to those observed in these follicular dysplasia cases. 21 The most obvious of those include broad areas of alopecia despite normal hair follicle density, follicular ectasia, and intraluminal follicular keratin. Variable follicular densities are described in characterizations of hypotrichosis in other species,6,7,12,13,18 presumably referring to either a lack of grossly evident hair and/or histologically absent follicles. A relatively normal number of follicles are described in these Wisconsin bucks; however, the degrees of alopecia exhibited by these individuals suggest unlikely survival until the age of harvesting if their conditions were congenital in nature. If truly congenital, the possibility exists that other factors (nutrition, stress, environment, and so on) may influence progression of disease with aging.
The number of other wild animal species reported to have been diagnosed with follicular dysplasia is minimal. Follicular dysplasia was documented in Alaskan gray wolves (Canis lupus) in 2012, chiefly involving the guard hairs over the trunk and caudal thigh regions of affected individuals, with the condition colloquially referred to as “poodle coat.” 3 Since that time, follicular dysplasia in gray wolves in and around the Denali National Park appears to continue to be an issue. 1 As of 2018, the majority of the Denali wolf packs regularly surveilled exhibit bilaterally symmetric truncal alopecia due to follicular dysplasia, with the tail and ventral neck more recently and intermittently involved.
Bilateral follicular dysplasia has also been reported in polar bears (Ursus maritimus) inhabiting the southern region of the Beaufort Sea of Alaska. 2 The alopecic areas were not symmetrical in distribution, chiefly affecting the lateral and ventral areas of the neck, head, and proximal shoulders. Reduced density of undercoat hairs was grossly present in addition to the loss of guard hairs. Histologically, reduced numbers of hair follicles were evident, extant follicles were in telogen phase, and irregular follicular and epidermal hyperkeratosis were present.
Hair serves numerous important functions, such as thermoregulation; protection from environmental elements, ultraviolet radiation, and trauma; conspecific communication; and camouflage.4,8,9 Unhealthy and/or alopecic hair coats may be detrimental to the general health and longevity of affected individuals and, specifically in humans, can have major impacts on psychosocial well-being.
Opportunities for the observation and health status surveillance of clinically affected wildlife are significantly restricted and primarily limited to field biologists, conservation and natural resource professionals, and the hunting public. These observations are typically only a snapshot in time with little light shed on long-term health impacts and survivability. Captive and farmed deer could provide a more accessible population to advance the understanding of noninfectious alopecic disease in free-ranging WTD populations. This report documents “toothpaste hair disease” in 2 free-ranging WTDs in Wisconsin with characteristics suggestive of alopecia areata, but evaluation of more cases is warranted. Recognition, reporting, and characterization of additional cases can ultimately lead to a better understanding of the epidemiology, seasonality and/or coat color influences, and potential genetic predispositions of this disorder.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231185106 – Supplemental material for Follicular dysplasia and hair loss in white-tailed deer (Odocoileus virginianus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231185106 for Follicular dysplasia and hair loss in white-tailed deer (Odocoileus virginianus) by Kathleen M. Deering, Lorelei L. Clarke, Shawn V. Lennix, Elizabeth Layne, Lindsey Long and Nancy Businga in Veterinary Pathology
Footnotes
Acknowledgements
We thank the Wisconsin Veterinary Diagnostic Laboratory Histology staff for processing the slides and all submitters.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
