Abstract
A Caribbean spiny lobster, Panulirus argus, was submitted for necropsy after a number of species-specific fatalities in a public aquarium. The hemolymph was opaque and did not clot and the hepatopancreas had multiple foci of necrosis centered on gram-negative bacteria. Pure cultures of Vibrio harveyi were isolated, identified initially by matrix laser desorption ionization-time of flight mass spectrometry, and confirmed by multilocus sequencing of the gyrB, recA, rpoA, and pyrH genes. As Caribbean spiny lobsters continue to be used for consumption and displayed in public aquariums, chronicling potential pathogens is warranted to inform differential diagnoses and to develop management strategies to reduce incidence of infectious disease in captive populations.
Caribbean spiny lobsters (Panulirus argus), named for the protective spines that span their body, are a valuable aquaculture resource and a popular exhibit animal in large-scale public aquaria. Panulirus spp. are primarily native to the Gulf of Mexico and the Caribbean, but some populations have settled throughout the Atlantic Ocean, as far east as the coast of Europe and as far south as Africa. 20 Increased consumer demand, high market value, and increased fishing pressure have made the Caribbean spiny lobster one of the most heavily fished Panulirus spp. worldwide. 7 As such, the lobster is exceptionally valuable and is one of the most significant species caught in Florida, USA, offering a multitude of employment opportunities throughout the region.6,30 Historically, research has primarily focused on the economic and environmental impacts of commercial and recreational fisheries. 30 Until recently, infectious diseases in Caribbean spiny lobsters have not received the same level of research investment. 30
Given their limited ability to respond to infection, lobsters present similar gross and microscopic findings for multiple disease processes. In response to infection, cells in the hemolymph known as hemocytes function in aggregation, coagulation, and phagocytosis. 15 Recognized agents of disease in P. argus include bacteria, fungi, protozoans, and helminths, as well as the viral disease Panulirus argus virus 1 (PaV1).29,30 PaV1 is thought to be specific to P. argus and has a propensity to infect juveniles. 16 Clinical signs include lethargy, with the individual struggling to maintain their position, and watery cloudy hemolymph that does not clot.16,30 However, crustaceans with bacterial sepsis can also exhibit lethargy, decreased physical activity, inability to maintain positioning, and milky/cloudy hemolymph with loss of clotting activity. 18 Bacteria, including Vibrio spp., are commonly associated with marine aquarium species and public health. 17 Vibrio spp. have a variety of virulence mechanisms. For example, Vibrio cholerae, which is often associated with human disease, produces cholera toxin. 1 In this study, we report the findings of a Caribbean spiny lobster submitted for necropsy following a spate of species-specific fatalities in a public aquarium exhibit.
Materials and Methods
Unexplained mortality occurred in a captive population of Caribbean spiny lobsters in a public aquarium with no outward clinical signs or obvious indications of disease. Mortalities occurred in a 3000-gallon multispecies tank connected to the general life support system spanning a combined 300,000 gallons, but fatalities were only observed in P. argus. The tank was stocked with stone crabs (Menippe mercenaria), soldier fish (Myripristis berndti), squirrel fish (Holocentrus adscensionis), and approximately 10 Caribbean spiny lobsters. The rate of loss was 1 to 2 lobsters per month. Water quality parameters were routinely monitored, including total ammonia nitrogen, nitrite, nitrate, dissolved oxygen, temperature, and pH, all of which were within normal levels according to the husbandry staff. There was no history of increased stress, such as recent handling, overcrowding, or failure of the life support system. Carcasses were collected, but the advanced state of decomposition precluded diagnostic testing. Therefore, a lobster was arbitrarily selected for euthanasia and submitted immediately for necropsy. This lobster displayed no clinical signs of disease. The spiny lobster, weighing 586 g, was euthanized by injection with saturated potassium chloride solution (KCl) at the base of the second pleopod. 23 Heart contractions were monitored via ultrasound. An initial dose of 100 mg KCl/100 g of lobster (total 4 mL) was administered, resulting in reduced heart rate. Another 3 mL of 2 mEq/mL KCl was administered at the base of the second pleopod on the other side and visualized heart contractions ceased. The lobster was subsequently placed on ice and transported to the local laboratory for postmortem diagnostic workup, which took place within 2 to 3 hours of euthanasia.
The lobster was visually examined for external lesions and sectioned dorsally along the carapace. All organs were removed and examined. Hemolymph pooled in the specimen and was collected using a needle-less syringe. The hemolymph was assessed for clotting, and cytology was attempted; however, results were inconclusive. All organs were fixed in 10% neutral buffered formalin for a minimum of 24 hours and processed routinely for microscopic examination.2,4 Slides were stained with hematoxylin and eosin (H&E). Additional sections of hepatopancreas containing lesions were stained with Ziehl-Neelsen acid-fast and Gram stain. Small sections of hepatopancreas were collected for aerobic bacterial culture on a general chocolate agar plate, a MacConkey salt agar plate (for gram-negative bacteria), and a phenylethyl alcohol agar plate, which selects for gram-positive bacteria. A Thermo Scientific Sensititre Aquatic Panel (CMV1FVGP) with 96-well broth dilution technique was performed according to the Clinical and Laboratory Standards Institute to determine antibiotic sensitivity breakpoints. 34 Isolated bacteria were initially identified using matrix laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) (Bruker Corporation, Billerica, MA).
The MALDI-TOF MS identification was molecularly confirmed by Sanger sequencing of the gyrB, recA, rpoA, and pyrH genes (Supplemental Table S1). Briefly, the isolate was expanded in porcine brain heart infusion broth and cultured overnight with shaking (200 rpm) at 28°C. An aliquot (3 mL) of expanded culture was concentrated by centrifugation (20,000g) and genomic DNA was isolated from the pellet using the PuroSPIN Genomic DNA Purification Kit (Luna Nanotech, Markham, Ontario, Canada). Polymerase chain reaction (PCR) was performed for each gene loci following previously published protocols.10,32 Amplicons were visualized with UV light after electrophoresis through a 0.8% agarose gel containing ethidium bromide (0.5 μg mL−1). Amplicons were excised, purified using the PuroSPIN Gel Extraction Kit, and sequenced commercially using the same primers employed to generate the amplicons (Eurofins Genomics, Louisville, KY). Contiguous sequences were assembled, and ambiguous base calls were manually annotated from corresponding chromatograms using Geneious Prime 2022.0.1 (Biomatters, Ltd., Auckland, New Zealand). Megablast searches for highly similar sequences for each locus were performed against the National Center for Biotechnology Information’s nonredundant nucleotide (nr/nt) database. Sequences from representative V. harveyi genomes (n = 45) were downloaded for each locus and concatenates of gyrB, recA, rpoA, and pyrH fragments were aligned, similarity matrices were produced using multiple alignment using fast Fourier transform (MAFFT) in Geneious Prime. Phylogenetic analysis of the 20 most similar V. harveyi isolates was performed using the Kimura 2-parameter model with Gamma Distribution and Invariant Sites (K2+G+I), which was determined as the best model fit for the concatenated alignments; phylogenetic relatedness was inferred using Maximum Likelihood with 500 bootstraps in MEGA-X.14,31 Two representative V. campbellii isolates were included as an outgroup. In addition, isolates were tested for the presence of the cholera toxin producing gene ctxAB, following V. cholerae-duplex PCR procedures outlined by Madhusudana and Surendran. 19
Frozen hepatopancreas was prepared for laser capture microdissection (LCMD) following fixation in 95% ethanol. Tissue processing excluded formalin contact, but otherwise followed standard methods. 9 Sectioning at 5 µm was conducted under DNAase-free conditions and focal necrotizing lesions containing bacteria (melanized hemocytic nodules) were excised using an MMI CellCut plus LCMD system (Molecular Machines and Industries, Haslett, MI). DNA was extracted from the sample tissue and a V. harveyi positive control using a Qiagen DNA Micro Kit (Qiagen, Hilden, Germany) and PCR performed using primers PYRH04F and PYRH02R directed against a fragment of the pyrH gene. 32 Last, a section of the hepatopancreas was sent to an outside lab for PaV1 PCR. 30
Results
At necropsy, there were no external signs of disease, shell damage, or discoloration. The gonad was small and immature (Fig. 1a). The hemolymph was opaque, pooled abnormally throughout the body, (most notably around the undersized hepatopancreas), and never clotted (Fig. 1a). Cytologic examination of the hemolymph was inconclusive. The atrophic hepatopancreas was tan to green with multifocal, random, 1 to 2 mm, firm white nodules (Fig. 1a, b). Histologically examined organs included muscle, brain, eye, digestive tract, gills, ovary, hepatopancreas, and stomach, but only the hepatopancreas had lesions. A normal hepatopancreas is composed of arborizing tubular glands. Approximately 30% to 40% of this lobster’s hepatopancreas was effaced by multifocal to coalescing, variably sized foci of necrosis and encapsulation (Fig. 1c–e). Hyaline cells and granulocytes were scattered within foci of necrosis and formed rims and streams in adjacent tissues, but not systemically. Hyaline cells or hyalinocytes refer to hemocytes that contain minimal to no cytoplasmic granules, whereas granulocytes contain abundant granules. 15 Melanization was also identified (Fig. 1d–e). Gram stain revealed the presence of gram-negative bacilli within the center of the necrotic foci (Fig. 1f). Hepatopancreas tissues were acid-fast negative, and no viral inclusions were observed.
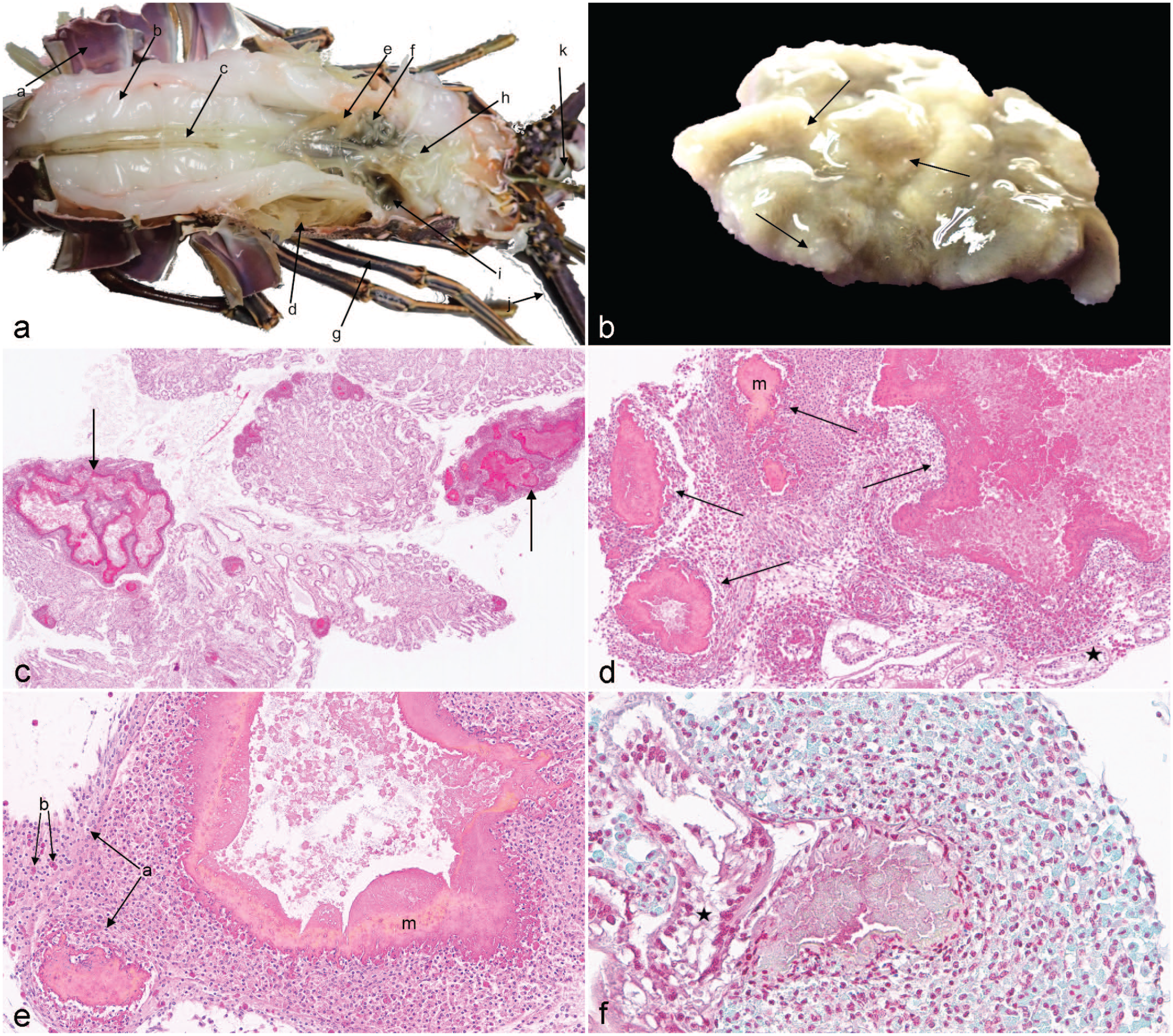
(a) Dorsal view of Panulirus argus. (a) carapace, (b) muscle, (c) digestive tract, (d) gills, (e) ovary, (f) hepatopancreas, (g) pereiopod, (h) stomach, (i) pooled hemolymph, (j) antennae, (k) eye stalk. (b) Hepatopancreas, lobster. Removed atrophied hepatopancreas with multifocal, random, 1-mm, white foci of necrosis throughout the parenchyma (arrows). (c) Hepatopancreas. Approximately 30% of the hepatopancreas parenchyma with tubules was destroyed by multifocal to coalescing, random, variably sized foci of necrosis and encapsulation (arrows). Normal tubular architecture also visible between abnormal foci. Hematoxylin and eosin (HE). (d) Hepatopancreas. Higher magnification of parenchymal necrosis and hemocyte response with encapsulation (arrows) and melanization (m). Normal tubule (black star). HE. (e) Hepatopancreas. Hemocyte response with encapsulation and melanization (m). Hyalinocytes (a) and granulocytes (b) result in aggregation and phagocytosis with rims and streams of hemocytes. HE. (f) Hepatopancreas. Foci of necrosis and encapsulation surrounding colonies of 1 × 2-µm gram-negative bacilli. Normal tubule (black star). Gram stain.
Bacterial colonies isolated from the hepatopancreas grew best on chocolate agar and were identified by MALDI-TOF MS as V. harveyi. The 4 sequenced housekeeping genes confirmed these results showing a high degree (99%-100%) of similarity for gyrB (588 bp), recA (795 bp), rpoA (702 bp), and pyrH (667 bp) to other V. harveyi isolates in GenBank. Phylogenies derived from a concatenate of these 4 loci placed the isolate with other V. harveyi sequences archived in GenBank (Fig. 2); however, metadata such as host and geographic location for some of these GenBank sequences from unpublished studies are lacking (Fig. 2). Still, these data cluster the isolate from P. argus with other V. harveyi isolates from marine fish, corals, and invertebrate species. The MLSA data for the case isolate are available in GenBank (Acc. Nos. OQ079550-OQ079553).

Phylogenetic relatedness of Vibrio harveyi isolates (black text) inferred by maximum likelihood using the Kimura 2-parameter model with Gamma distribution and invariable sites (K2+G+I) with 500 bootstraps. Trees were generated from alignments of concatenated gyrB, recA, rpoA, and pyrH fragments and rooted at Vibrio campbellii (blue text). The Vibrio harveyi case isolate K2014767 from hepatopancreas of Caribbean spiny lobster is presented in red text.
Antibiotic sensitivity testing found the isolate to be nonresistant/susceptible to florfenicol, gentamicin, ormetoprim/sulfadimethoxine, oxytetracycline, and trimethoprim/sulfamethoxazole. However, treatment was not initiated due to loss of the population. All tissues were PCR-negative for PaV1 and the cultured isolate was PCR-negative for ctxAB. Attempts to associate bacterial sequences to the hepatopancreatic lesions using LCMD and PCR for the pyrH gene were unsuccessful.
Discussion
Increased mortality and hemolymph that failed to immediately clot alluded to a viral or bacterial infection in the lobster. Microscopic findings were most consistent with a bacterial infection with a lack of viral inclusions, hemocyte response, necrosis, and encapsulation with melanization centered on gram-negative bacteria in the hepatopancreas, and V. harveyi was cultured from infected hepatopancreatic tissue. In invertebrates, encapsulation is a cell-mediated immune defense mechanism where hemocyte responses result in capsule formation and melanization. 15 Encapsulation is semi-analogous to the formation of vertebrate granulomas. 15
Inoculation of a live lobster was not possible due to unavailability of pathogen-free individuals. So, as an alternative to fulfilling Koch’s postulates, LCMD was attempted to localize V. harveyi genetic material to the hepatopancreatic lesions. However, the PCR failed to amplify DNA from the excised sample hepatopancreas. Possible reasons for failure of the PCR are multifold. Vibrio harveyi may have been present but below the level of detection for the conventional end-point PCR method in combination with the single-copy pyrH gene, which together make the minimum detectable limit for the bacterium relatively high. Additional causes include sample degradation due to autolysis or repeated freeze-thaw cycles, insufficient sample size and excision of infected tissue using the LCMD, failure of the DNA extraction method, and the potential presence of PCR inhibitors in the hepatopancreatic tissue.
Several bacteria including Aerococcus viridans, Vibrio alginolyticus, and Leucothrix mucor have been reported to cause systemic infections in P. argus. 29 Aerococcus viridans is the cause of Gaffkemia red-tail disease, which presents as discolored red carapaces, lethargy, and pink hemolymph that does not coagulate. 29 Another common disease generically referred to as “shell disease,” due to erosion of the exoskeleton (chitinolysis), is often attributed to gram-negative bacteria including Vibrio spp., Shewanella spp., and Aeromonas hydrophila. 29 There is another form of shell disease that primarily affects spiny lobsters known as tail fan necrosis, which presents as eroded areas on the tail.29,35 Tail fan necrosis is thought to be associated with trauma and suboptimal water quality, thus lobsters maintained in culture and aquarium settings are highly susceptible if the water quality is not properly managed.29,35 Multiple bacterial phylotypes including Vibrio spp. have been isolated from the damaged cuticle of infected lobsters.29,35
Vibrio spp. are common in saltwater and freshwater environments, with a predilection for alkaline environments, and can withstand a wide temperature range.22,25 The genus includes multiple halophilic/non-halophilic, facultative anaerobic, curved, rod-shaped, gram-negative bacteria, which can be significant threats to aquariums and public health due to their ubiquity in aquatic environments. 17 While Vibrio spp. are commonly associated with human illness, only 12 of the 100 Vibrio spp. have been confirmed as human pathogens. 13 Vibrio spp. use species-specific virulence mechanisms, but even within Vibrio species there is variation in the mechanisms and manifestation of virulence. 36 Vibrio cholerae produces cholera toxin, a protein that is encoded by the ctxAB gene. 1 Superoxide dismutases, including sodB found in Vibrio alginolyticus, have been identified as playing a role in regulation of reactive oxygen species. 5 Toxin-like PirA and PirB are encoded by a gene of a plasmid in certain strains of Vibrio parahaemolyticus produce hepatopancreatic necrosis disease in aquaculture shrimp. 11 Vibrio parahaemolyticus utilizes toxins as well as adhesions and a type 3 secretion system.12,33,36
Vibrio harveyi is primarily an opportunistic halophilic bacterium associated with disease in warmwater wild and cultured fish, shellfish, coral, and mammals. 37 The majority of V. harveyi isolates are nonpathogenic, but are occasionally associated with disease. 24 Vibrio harveyi is a prominent bacterial pathogen in cultured penaeid shrimp causing luminous vibriosis and Bolitas nigricans, resulting in shedding of tissue into the digestive tract.27,28,37 The first record of luminous vibriosis in phyllosoma larvae of spiny lobster, Jasus verreauxi, infected with V. harveyi found that the hepatopancreas in infected larvae had foci of necrosis and hemocyte response in areas of infection in the gut. 8 Specific pathogenicity mechanisms are not well documented for V. harveyi and further research into the virulence mechanisms of V. harveyi is warranted. In shrimp, endotoxin lipopolysaccharides, extracellular proteases, and bacteriophages are of concern and it has been reported that V. harveyi isolates from marine organisms demonstrate virulence-variation and the genetic machinery for the production of toxins such as hemolysin and protease.21,37 A previous study concluded that V. harveyi was a diverse taxa with no distinct strains demonstrating innate virulence, but rather virulence was opportunistic and driven by mobile genetic elements and horizontal gene transfer. 26
To our knowledge, this is the first report of V. harveyi associated with a species-specific morbidity event in captive Caribbean spiny lobsters. Given the importance of P. argus as a food animal, not to mention its popularity in public aquariums, a detailed census of potential pathogens is warranted to facilitate accurate diagnostics and develop management strategies to reduce the incidence of infectious disease. In a study outlining complete postmortem and histological assessment of more than 300 Caribbean spiny lobsters caught by fisheries, there were no detailed findings of Vibrio harveyi in apparently healthy wild lobsters. 3 This suggests that research is lacking, and future research investment should focus on the risk V. harveyi poses to wild and captive populations of P. argus, as well as risks to other wild and cultured aquatic animal populations.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231164754 – Supplemental material for Vibrio harveyi in a Caribbean spiny lobster (Panulirus argus) with hepatopancreas necrosis
Supplemental material, sj-pdf-1-vet-10.1177_03009858231164754 for Vibrio harveyi in a Caribbean spiny lobster (Panulirus argus) with hepatopancreas necrosis by Hannah M. Bennett, Matt Griffin, Ruth Francis-Floyd, Shirley Baker, Alvin Camus, Craig Pelton and Jennifer Dill-Okubo in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
