Abstract
Neural stem cell (NSC) lineage cells have not been fully identified in feline brains, and the NSC-like nature of feline glial tumors has not been determined. In this study, 6 normal cat brains (3 newborn and 3 older cats) and 13 feline glial tumors were analyzed using immunohistochemical NSC lineage markers. The feline glial tumors were subjected to immunohistochemical scoring followed by hierarchical cluster analysis. In newborn brains, glial acidic fibrillary protein (GFAP)/nestin/sex-determining region Y-box transcription factor 2 (SOX2)-immunopositive NSCs, SOX2-immunopositive intermediate progenitor cells, oligodendrocyte transcription factor 2 (OLIG2)/platelet-derived growth factor receptor-α (PDGFR-α)-immunopositive oligodendrocyte precursor cells (OPCs), OLIG2/GFAP-immunopositive immature astrocytes, and neuronal nuclear (NeuN)/β-3 tubulin-immunopositive mature neuronal cells were observed. The apical membrane of NSCs was also immunopositive for Na+/H+ exchanger regulatory factor 1 (NHERF1). In mature brains, the NSC lineage cells were similar to those of the newborn brains. A total of 13 glial tumors consisted of 2 oligodendrogliomas, 4 astrocytomas, 3 subependymomas, and 4 ependymomas. Astrocytomas, subependymomas, and ependymomas were immunopositive for GFAP, nestin, and SOX2. Subependymomas and ependymomas showed dot-like or apical membrane immunolabeling for NHERF1, respectively. Astrocytomas were immunopositive for OLIG2. Oligodendrogliomas and subependymomas were immunopositive for OLIG2 and PDGFR-α. Feline glial tumors also showed variable immunolabeling for β-3 tubulin, NeuN, and synaptophysin. Based on these results, feline astrocytomas, subependymomas, and ependymomas appear to have an NSC-like immunophenotype. In addition, astrocytomas, subependymomas, and ependymomas have the characteristics of glial, oligodendrocyte precursor, and ependymal cells, respectively. Feline oligodendrogliomas likely have an OPC-like immunophenotype. In addition, feline glial tumors may have multipotential stemness for differentiation into neuronal cells. These preliminary results should be validated by gene expression analyses in future studies with larger case numbers.
Glial tumors are broadly classified into astrocytic, oligodendroglial, ependymal, and subependymal tumors based on their morphological and immunohistochemical features.18,25,55 In dogs, undefined glioma is also classified as having high proportions of oligodendroglial and astrocytic phenotypes (>30%–40% each). 24 The biological natures of canine glial tumors have been described in detail, whereas limited information is currently available on feline glial tumors.18,47 In cats, oligodendroglioma and astrocytoma have immunohistochemical features that correspond to normal oligodendrocytes and astrocytes, namely immunolabeling for OLIG2 and GFAP, respectively.17,20,48 The expression of doublecortin, a neuronal precursor marker, has also been reported in feline gliomas, suggesting the multipotential differentiation potential of glioma cells. 10
Feline ependymomas are immunopositive for GFAP and OLIG2, suggesting a stem-like phenotype of the tumor cells.8,59 Feline subependymomas have not been as extensively examined as gliomas and ependymomas.18,51,59 Human subependymomas have ultrastructural features of astrocytic and/or ependymal cells. 37 Subependymomas in humans and cats have an eosinophilic fibrillary matrix and are immunopositive for GFAP.7,51,59 In addition, the microvilli of human subependymomas express NHERF1, similar to those of ependymomas.7,15,37 These findings indicate that subependymoma is an intermediate tumor between astrocytoma and ependymoma. Moreover, human subependymoma is considered to originate from a subependymal multipotential stem cell. 7
NSC lineage cells are proposed as a candidate for the origin of glial tumors in rodents, dogs, and humans38,44,56,60,63; however, characteristics of stem cells in feline glial tumors, particularly ependymoma and subependymoma, remain unclear. In rodents, stem cells in the brain are considered to originate from neuroepithelial cells. 26 Neuroepithelial cells differentiate into NSCs, called radial glia, in the ventricular zone (VZ). 26 Radial glia are characterized by immunopositivity for GFAP and nestin. NSCs then produce intermediate precursor cells (IPCs) in the subventricular zone (SVZ) and ependymal cells in the VZ. 26 NSCs and IPCs express SOX2.11,19 IPCs may differentiate into neurons, oligodendrocytes, and astrocytes, and IPCs for oligodendrocytes are also known as OPCs characterized by expression of PDGFR-α.26,27 Neuronal differentiation is characterized by the expression of β-3 tubulin and NeuN protein.16,21
In the adult brain of rodents, quiescent NSCs, known as B cells, remain in the SVZ and extend elongate processes to the ependymal layer.26,29 B cells express astroglial markers, such as GFAP, and activated B cells express nestin.26,29 B cells also produce IPCs, known as C cells, which may differentiate into oligodendrocytes or A cells, which are immature neurons.26,29 A cells have been found to be immunopositive for β-3 tubulin. 9 SOX2 is widely expressed in NSC lineage cells. 11 Fetal or adult NSC lineage cells have been detected in humans,41,45 dogs,12,13,36,40,57,60 cows 50 , sheep, 31 pigs, 6 and rabbits. 2 However, feline NSC lineage cells have only been examined in in vitro assays and the rostral migratory stream.23,32,58
This study revealed the presence of feline NSC lineage cells in normal newborn and adult brains using immunohistochemistry. The immunophenotypes of feline oligodendroglioma, astrocytoma, and ependymal tumors were then compared with those of NSC lineage cells to reveal the NSC lineage cell characteristics of each tumor type.
Materials and Methods
Sample Collection
The histopathological database of the Laboratory of Veterinary Pathology, the University of Tokyo was searched for the term “brain tumors” in cats between April 2013 and December 2022. Records were reviewed, and then, oligodendrogliomas, astrocytomas, ependymomas, and subependymomas were selected. Accordingly, 13 cases were used in the analysis.
Brain tissues from 3 newborn cats (less than 30 days), 1 kitten (3 months), 1 young adult cat (2 years), and 1 senior cat (13 years 4 months) were used as normal controls. These cases had no neurological signs or histopathological lesions in the brain and were collected through routine necropsies at our laboratory.
Histopathological and Immunohistochemical Analyses
The formalin-fixed paraffin-embedded blocks of 13 oligodendroglioma, astrocytoma, ependymoma, and subependymoma cases and 6 normal brains were sectioned at a thickness of 4 µm and stained with hematoxylin and eosin (HE). The histopathological diagnosis was based on the World Health Organization (WHO) International Classification of Tumors of Domestic Animals 25 and the human WHO Classification of Tumors of the Central Nervous System 2021. 55 Mitotic activity was counted in 10, nonoverlapping 40× high-power fields (2.37 mm2) in areas with the most mitotic activity. Immunohistochemistry was performed on the tumors and normal brains using primary antibodies for OLIG2, PDGFR-α, GFAP, nestin, SOX2, β-3 tubulin, NeuN, synaptophysin, NHERF1, Ki-67, and cytokeratin AE1/AE3. Details on the primary antibodies and immunohistochemistry conditions are summarized in Supplemental Table S1. Deparaffinized tissue sections were incubated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes to block endogenous peroxidase. After heat-induced epitope retrieval, sections were incubated with 4% skim milk-Tris-buffered saline (pH: 7.6) at 37°C for 40 minutes to prevent nonspecific reactions. Each section was treated with the primary antibodies at 4°C overnight. A horseradish peroxidase-labeled antimouse or antirabbit IgG polymer (Envision+, Dako) was used as the secondary antibody at 37°C for 40 minutes. 3-3’-diaminobenzidine was used as the chromogen. Counterstaining was performed using hematoxylin. As negative controls, each primary antibody was replaced by an irrelevant antibody. The primary antibodies were validated by western blotting. To confirm the specificity of the primary antibodies, brain tissues from an adult (12 years) and a fetal cat (unknown age), and a kidney from a cat (20 years) frozen at −80°C were used for western blotting. Western blotting was conducted according to a previously reported method. 60 The incubation conditions of the primary antibodies are summarized in Supplemental Table S1. The antibodies for Ki-67 (clone MIB-1, Dako) and cytokeratin AE1/AE3 (clone AE1/AE3, Dako) were not validated because they are frequently used in veterinary literature. The results of western blotting are shown in Supplemental Fig. S1. Immunohistochemical results for tumor cases were scored as follows: 0, no immunopositive cells; 1, <25% immunopositive cells; 2, 25%–75% immunopositive cells; and 3, >75% immunopositive cells. Immunopositivity for tumor cells was assessed in the nucleus for OLIG2, SOX2, and NeuN; in the cytoplasm for GFAP, nestin, β-3 tubulin, synaptophysin, NHERF1, and cytokeratin AE1/AE3; and in the membrane for PDGFR-α.
Dual Immunofluorescence
Dual immunofluorescence was performed with the antibody combinations of GFAP/nestin, OLIG2/PDGFR-α, OLIG2/GFAP, OLIG2/SOX2, GFAP/SOX2, β-3 tubulin/SOX2, NHERF1/GFAP, and cytokeratin AE1/AE3/GFAP. Details on the primary antibodies and antigen retrieval conditions are summarized in Supplemental Table S2. After the heat-induced epitope retrieval of deparaffinized sections, blocking by 4% skim milk-Tris-buffered saline (pH: 7.6) at 37°C for 40 minutes was conducted to prevent nonspecific reactions. A primary antibody cocktail was applied to sections at 4°C overnight. Alexa Fluor 488-labeled goat antimouse IgG (Thermo Fisher Scientific), Alexa Fluor 488-labeled goat antirabbit IgG, Alexa Fluor 594-labeled donkey antirabbit IgG, and Alexa Fluor 594-labeled goat antimouse IgG were used as the secondary antibodies at 37°C for 1 hour. Sections were enclosed with 4',6-diamidino-2-phenylindole (Vectashield; Vector Laboratory). Images were captured in a single region of interest with a ×60 objective (0.0103 mm2) by a laser scanning confocal microscope (LSM700, Zeiss).
Dual Immunohistochemistry
Dual immunohistochemistry for the combinations of OLIG2/GFAP, GFAP/SOX2, and SOX2/NeuN was conducted on samples. OLIG2/GFAP and GFAP/SOX2 dual immunohistochemistry were performed to verify the results of dual immunofluorescence. SOX2/NeuN dual immunohistochemistry was performed because the hosts of these antibodies were the same (rabbit antibody). Deparaffinized sections were incubated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes to block endogenous peroxidase. After heat-induced epitope retrieval with citrate buffer (pH: 6.0), sections were incubated with 4% skim milk-Tris-buffered saline (pH: 7.6) at 37°C for 40 minutes to prevent nonspecific reactions. A polyclonal rabbit anti-GFAP antibody was used for the combination of OLIG2/GFAP. A monoclonal mouse anti-GFAP antibody was used for the combination of GFAP/SOX2. The primary antibody cocktail used for the combination of OLIG2/GFAP and GFAP/SOX2 was applied to sections at 4°C overnight. The anti-SOX2 antibody used for the combination of SOX2/NeuN was applied at 4°C overnight as a first immunohistochemical reaction. After visualizing SOX2, sections were heated by citrate buffer (pH: 6.0) at 121°C for 15 minutes in an autoclave to denature the primary and secondary antibody complexes. Accordingly, the anti-NeuN antibody was applied at 4°C overnight. The secondary antibodies were applied at 37°C for 40 minutes, and a horseradish peroxidase-labeled antimouse or antirabbit IgG polymer (EnVision+, Dako) and alkaline phosphatase-labeled antimouse or antirabbit IgG polymer (Histofine, Simple Stain, Nichirei Bioscience) were used as the secondary antibody. The primary and secondary antibody complexes were visualized using New Fuchsin (Histofine, Nichirei Bioscience) and HIGHDEF blue (Enzo Life Sciences). Counterstaining was omitted.
Ki-67 Labeling Index
Ki-67 Labeling Index (LI) was calculated as the number of Ki-67-immunolabeled tumor cells divided by the total number of tumor cells. Cell counting was conducted using 5 high-power field (40× objective, 0.11 mm2) images taken in the area with the highest number of immunolabeled cells on the slide (hot spots). Inflammatory cells were morphologically distinguished based on their nuclear and cytoplasmic shape. Ki-67 LI was not assessed in cases for which Ki-67 immunopositivity was not observed in the slides.
Cluster Analysis of Immunohistochemical Scores
To compare the immunohistochemical features of feline oligodendroglioma, astrocytoma, and ependymal tumors, immunohistochemical scores were analyzed by the Ward method using R version 4.2.0 (The R Foundation for Statistical Computing). Euclidian distances between the clusters were calculated and used to make a dendrogram.
Results
Immunohistochemistry of Newborn and Mature Brains
In feline newborn brains, the cytoplasm of VZ cells and their long processes to the SVZ were immunopositive for GFAP and nestin (Fig. 1a, b). These GFAP-immunopositive cells were mostly located on the septum side of the ventricular wall. The nuclei of VZ and SVZ cells were positive for SOX2 (Fig. 1c). A small number of cells in the intermediate zone (IZ) and cortical plate (CP) were also immunopositive for SOX2. Nuclear OLIG2-immunopositive cells and membranous PDGFR-α-immunopositive cells were sporadically distributed in the SVZ to IZ (Fig. 1d, e). Nuclear OLIG2-immunopositive cells were also observed in the CP. Cytoplasmic β-3 tubulin-immunopositive cells were detected in the VZ and SVZ (Fig. 1f). A few cells in the SVZ showed nuclear immunolabeling for NeuN (Fig. 1g). Neuronal cells in the CP were also immunopositive for β-3 tubulin and NeuN, and the neuroparenchyma was immunopositive for β-3 tubulin and synaptophysin. The apical membrane of the VZ and choroid plexus cells was immunopositive for NHERF1 (Fig. 1h). No cells were immunolabeled for cytokeratin AE1/AE3.
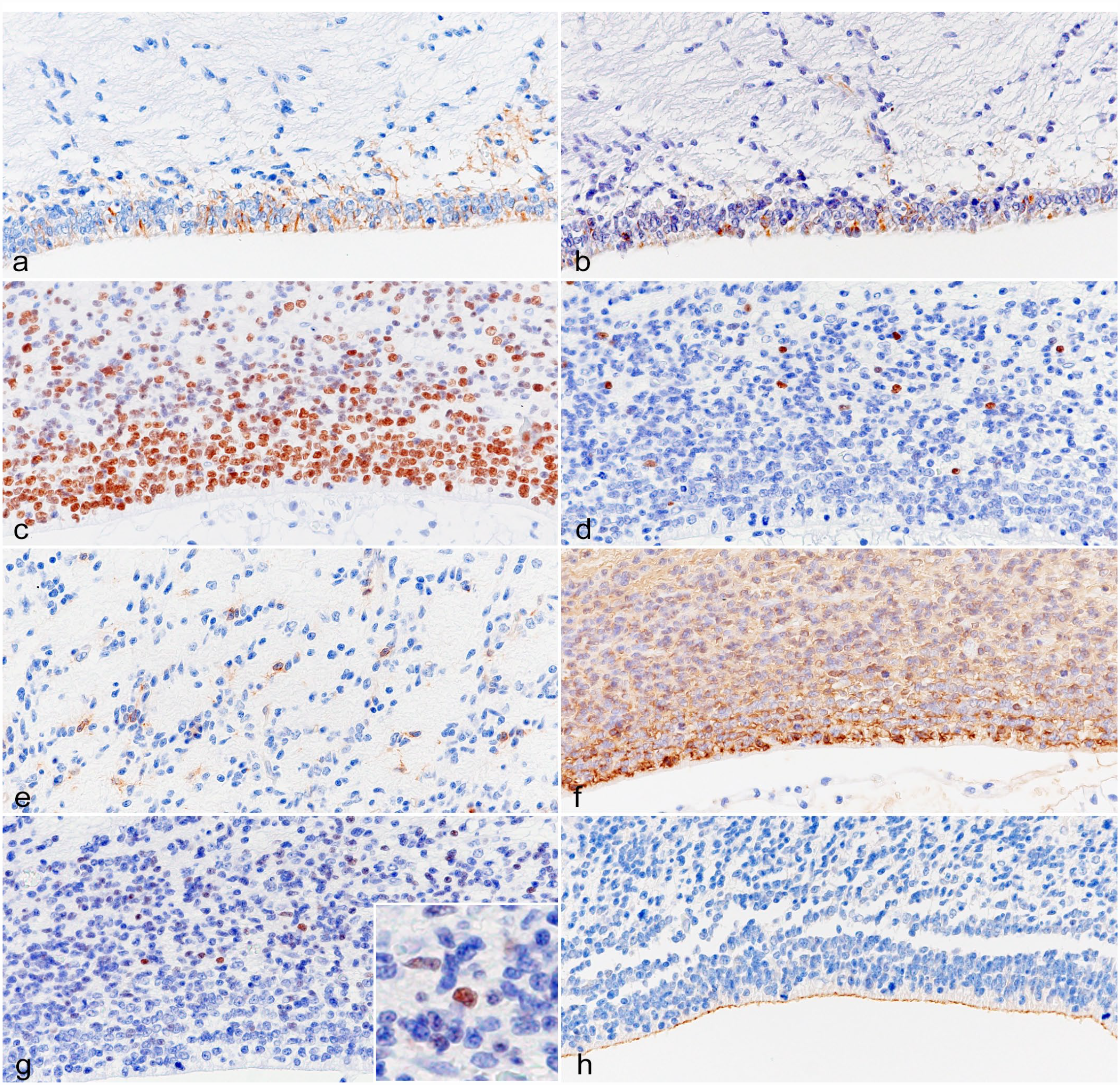
Brain, cat, less than 30 days. Immunohistochemistry. (a) The cytoplasm of the VZ cells is immunopositive for GFAP. (b) The cytoplasm of the VZ cells is immunopositive for nestin. (c) The nuclei of the VZ and SVZ cells are immunopositive for SOX2. (d) The nuclei of the SVZ cells are immunopositive for OLIG2. (e) The membrane of the IZ cells is immunopositive for PDGFR-α. (f) The cytoplasm of the VZ and SVZ cells is immunopositive for β-3 tubulin. (g) Nuclei in the SVZ cells are immunopositive for NeuN. (h) The apical membrane of cells in the VZ is immunopositive for NHERF1. VZ, ventricular zone; GFAP, glial fibrillary acidic protein; SVZ, subventricular zone; SOX2, sex-determining region Y-box transcription factor 2; OLIG2, oligodendrocyte transcription factor 2; IZ, intermediate zone; PDGFR-α, platelet-derived growth factor receptor-α; NeuN, neuronal nuclear; NHERF1, Na+/H+ exchanger regulatory factor 1.
Dual immunofluorescence showed that a few cells in the VZ were double-immunopositive for GFAP and nestin (Supplemental Fig. S2a). Cells double-immunopositive for OLIG2 and PDGFR-α were distributed in the SVZ to IZ (Supplemental Fig. S2b). A few double-immunopositive cells for OLIG2 and GFAP were observed in the SVZ to IZ in dual immunofluorescence and immunohistochemistry (Supplemental Figs. S2c and S3a). SOX2-immunopositive cells were also immunopositive for GFAP in the VZ to SVZ (Supplemental Fig. S2d), OLIG2 in the SVZ to CP (Supplemental Fig. S2e), and β-3 tubulin in the SVZ (Supplemental Fig. S2f). Dual immunohistochemistry revealed that SOX2-immunopositive cells were also immunopositive for NeuN in the SVZ to CP (Supplemental Fig. S3b).
In mature feline brains, GFAP-immunopositive cells were observed in the SVZ (Fig. 2a). A few nestin-immunopositive cells in the SVZ were detected in 2 cats (13 years 4 months, and 3 months) (Fig. 2b). Pericytes around blood vessels were also immunopositive for nestin. Ependymal cells and SVZ cells were immunopositive for SOX2 (Fig. 2c), and a few cells in the white and gray matter were also immunopositive for SOX2 (Fig. 2d). OLIG2-immunopositive cells were widely distributed in the brain. PDGFR-α-immunopositive cells were observed in the white matter of the 3-month-old cat brain. A few cells in SVZ were immunopositive for β-3 tubulin and NeuN (Fig. 2e, f). Neuronal cells in the cortex were also immunopositive for β-3 tubulin and NeuN, and the neuroparenchyma was immunopositive for β-3 tubulin and synaptophysin. The apical membrane of ependymal cells and choroid plexus cells was immunopositive for NHERF1 (Fig. 2g). The glia limitans near the pia matter and blood vessels exhibited mild immunolabeling for NHERF1. Cytoplasm and processes of astrocyte-like cells were immunolabeled for cytokeratin AE1/AE3 (Supplemental Fig. S4a).
Brain, cat, 13 years 4 months. Immunohistochemistry. (a) The cytoplasm of the SVZ cells is immunopositive for GFAP (arrowheads). (b) The cytoplasm of the SVZ cells is immunopositive for nestin (arrowhead). (c) Nuclei of the ependymal and SVZ cells are immunopositive for SOX2 (arrowhead). Aggregation of SOX2-immunopositive cells is also observed. (d) Nuclei of cells in the cortex are immunopositive for SOX2. (e) The cytoplasm of the SVZ cells is immunopositive for β-3 tubulin (arrowhead). (f) Nuclei of the SVZ cells are immunopositive for NeuN (arrowhead). (g) The apical membrane of the ependymal cells is immunopositive for NHERF1. SVZ, subventricular zone; GFAP, glial fibrillary acidic protein; SOX2, sex-determining region Y-box transcription factor 2; NeuN, neuronal nuclear; NHERF1, Na+/H+ exchanger regulatory factor 1.
Dual immunofluorescence revealed the presence of SOX2/GFAP-double-immunopositive cells or SOX2/β-3 tubulin double-immunopositive cells in the SVZ (Supplemental Fig. S5a, b). Nestin-immunopositive cells were also immunopositive for GFAP (Supplemental Fig. S5c). Cells double-immunopositive for OLIG2 and PDGFR-α were observed in the white matter of a 3-month-old cat brain. In the white and gray matter, some cells were double-immunopositive for SOX2 and OLIG2 or GFAP (Supplemental Fig. S5d, e). Dual immunohistochemistry also showed that SOX2-immunopositive cells were immunopositive for GFAP and NeuN in the white and gray matter (Supplemental Fig S6a, b). Furthermore, SOX2 and NeuN double-immunopositive cells were detected in the SVZ. The GFAP-immunopositive glia limitans were also immunopositive for NHERF1 (Supplemental Fig. S5f). GFAP-immunolabeled astrocytes were immunolabeled for cytokeratin AE1/AE3 (Supplemental Fig. S4b).
Histopathological Features of Feline Glial Tumors
The details of cats with glial tumors, including breed, sex, age, and tumor location, are summarized in Table 1. All cases were mixed-breed cats. The sex of affected cats consisted of 7 neutered males, 1 intact male, 4 neutered females, and 1 intact female. The ages of 12 cats ranged between 7 years 2 months, and 14 years 5 months, and information on the age at disease onset was not available in 1 case. Average and median ages were 11.06 and 11.00 years, respectively. Oligodendrogliomas and astrocytomas were frequently localized to the cerebral parenchyma (4/6 cases; olfactory bulb to temporal lobe). Ependymomas and subependymomas formed in the ventricle, except for 1 case diagnosed as parenchymal ependymoma that formed in the cerebral temporal lobe (case 13). Histopathologically, 13 feline glial tumor cases consisted of 2 oligodendrogliomas, 4 astrocytomas, 3 subependymomas, and 4 ependymomas.
Details of the 13 feline gliomas.
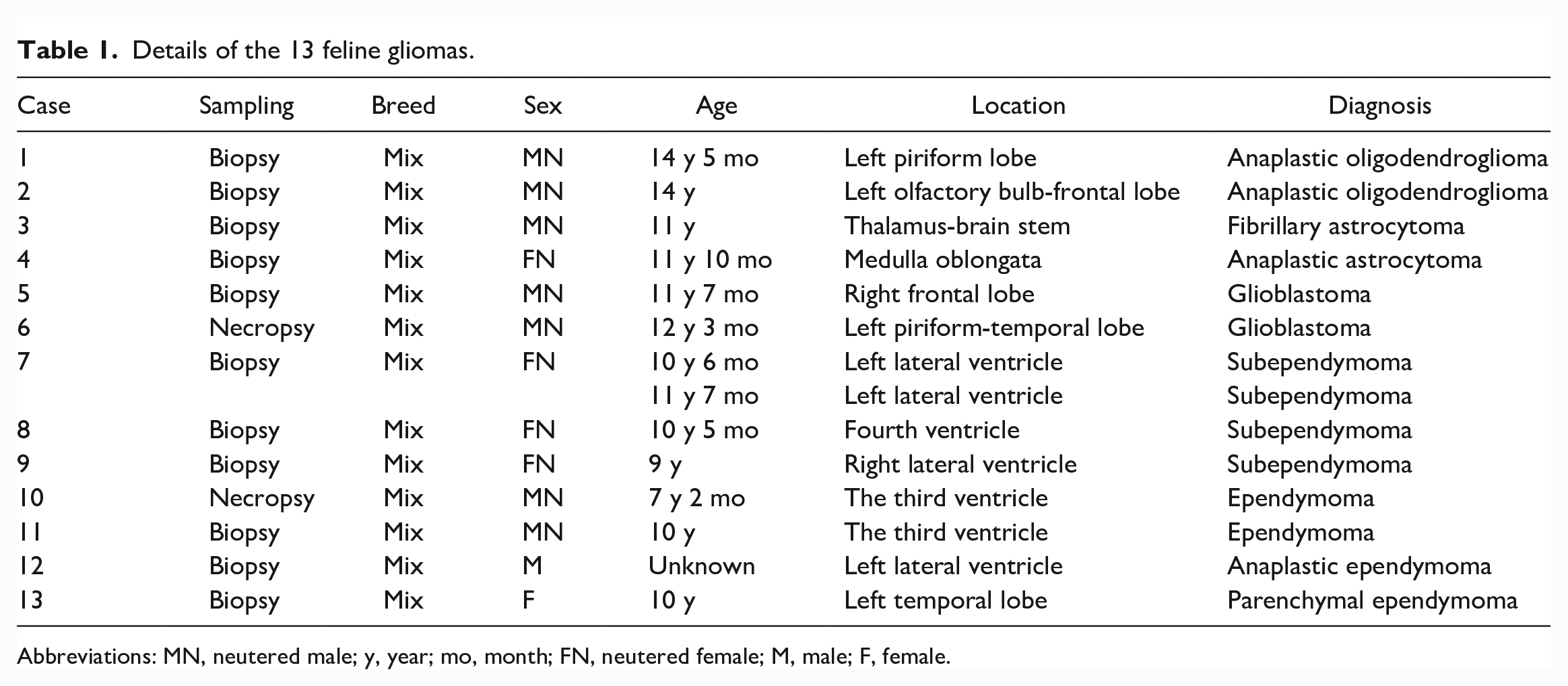
Abbreviations: MN, neutered male; y, year; mo, month; FN, neutered female; M, male; F, female.
Oligodendrogliomas (cases 1 and 2): Both cases were diagnosed as anaplastic oligodendrogliomas based on high cellularity and moderate nuclear atypia. Tumor cells were round and had clear cytoplasm and round nuclei with coarse chromatin (Fig. 3a). Mitotic activity was not observed in either case.
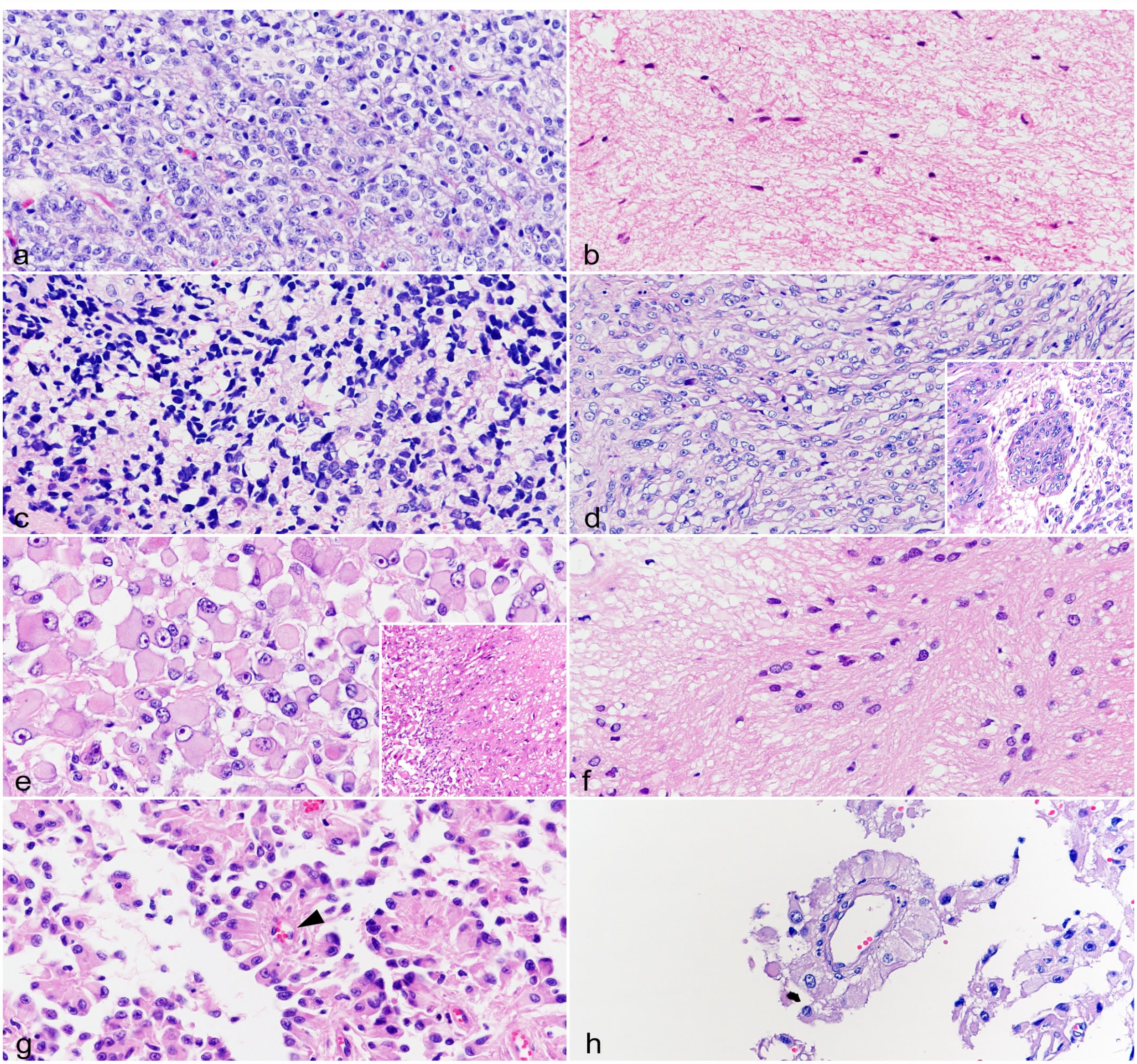
Glial tumor, brain, cat. Hematoxylin and eosin. (a) Anaplastic oligodendroglioma (case 1). Tumor cells are round and have clear cytoplasm and round nuclei with coarse chromatin. (b) Fibrillary astrocytoma (case 3). The tumor has abundant eosinophilic fibrillary matrix, and the cytoplasm of the tumor cells is poorly defined. Tumor cells have oval nuclei with dense chromatin. (c) Anaplastic astrocytoma (case 4). Tumor cells are pleomorphic, and have eosinophilic cytoplasm and round to oval nuclei with coarse to dense chromatin. (d) Glioblastoma (case 5). Tumor cells are spindle shaped, and have eosinophilic cytoplasm and oval nuclei with coarse chromatin and distinct nucleoli. Microvascular proliferation is observed (inset). (e) Glioblastoma (case 6). Tumor cells are polygonal (gemistocytic) shaped and have marked anisocytosis. The cytoplasm of tumor cells is eosinophilic. Nuclei have marked anisokaryosis and coarse chromatin with a distinct nucleolus. Necrosis is observed (inset). (f) Subependymoma (case 7). The tumor has uneven cell distribution and consists of cluster of tumor cells and an eosinophilic process-rich fibrillary matrix. Tumor cells are spindle to polygonal shaped, and have eosinophilic cytoplasm and round to oval nuclei with coarse chromatin. (g) Ependymoma (case 10). The tumor has a papillary growth pattern with vascular stroma. Tumor cells are columnar to pleomorphic, and have eosinophilic cytoplasm and round to oval nuclei with coarse to dense chromatin. Pseudorosettes are observed (arrowhead). (h) Parenchymal ependymoma (case 13). The tumor has papillary and perivascular growth patterns. Tumor cells are columnar to polygonal shaped, and have eosinophilic cytoplasm and round nuclei with coarse chromatin and a nucleolus.
Astrocytomas (cases 3–6): Astrocytomas commonly had eosinophilic fibrillary to abundant cytoplasm and were histopathologically diagnosed as a fibrillary astrocytoma (case 3), an anaplastic astrocytoma (case 4), and glioblastomas (cases 5 and 6). The fibrillary astrocytoma was a sparsely cellular tumor with mild atypia and had abundant eosinophilic fibrillary matrix. Tumor cells had poorly defined cell borders and oval nuclei with dense chromatin (Fig. 3b). Mitotic figures were not observed. The anaplastic astrocytoma was a highly cellular tumor with moderate atypia. Tumor cells were pleomorphic and had eosinophilic cytoplasm and round to oval nuclei with coarse to dense chromatin (Fig. 3c). One mitotic figure per 2.37 mm2 was observed. Glioblastomas were highly cellular with severe atypia. Tumor cells were spindle shaped in case 5 and polygonal (gemistocytic) in case 6 and had eosinophilic cytoplasm, round to oval nuclei with coarse chromatin, and a distinct nucleolus (Fig. 3d, e). Anisokaryosis was moderate in case 5 and severe in case 6. Based on extensive microvascular proliferation in case 5 and geographic necrosis in case 6, these cases were diagnosed as glioblastomas. Focal tumor necrosis was also present in case 5. The mitotic counts in cases 5 and 6 were 5 and 4, respectively.
Subependymomas (cases 7–9): Subependymomas had uneven cell distribution and consisted of clusters of tumor cells separated by eosinophilic process-rich fibrillary matrix. These features are similar to those previously reported. 59 Tumor cells were spindle to polygonal and had eosinophilic cytoplasm and round to oval nuclei with coarse chromatin (Fig. 3f). Moderate anisokaryosis was observed. Mitotic figures were not observed in any of the subependymoma cases.
Ependymomas (cases 10–13): Ependymomas commonly had papillary and perivascular growth patterns. Ependymomas in the ventricle (cases 10–12) were highly cellular tumors and had predominantly papillary growth patterns with vascular stroma. Tumor cells were columnar to pleomorphic and had eosinophilic cytoplasm and round to oval nuclei with coarse to dense chromatin (Fig. 3g). Moderate anisokaryosis and pseudorosettes were observed, but true rosettes were not. Among them, 1 case (case 12) was diagnosed as anaplastic ependymoma based on marked anisokaryosis of tumor cells. The parenchymal ependymoma (case 13) was also a highly cellular tumor with moderate atypia. Tumor tissue was arranged in solid, papillary, and perivascular, similar to pseudorosettes, growth patterns. Tumor cells were columnar to polygonal and had eosinophilic cytoplasm and round nuclei with coarse chromatin and a nucleolus (Fig. 3h). Mitotic figures were not observed in any of the ependymoma cases.
Immunohistochemical Features of Glial Tumors
The results of the immunohistochemical scoring of glial tumors are summarized in Supplemental Table S3.
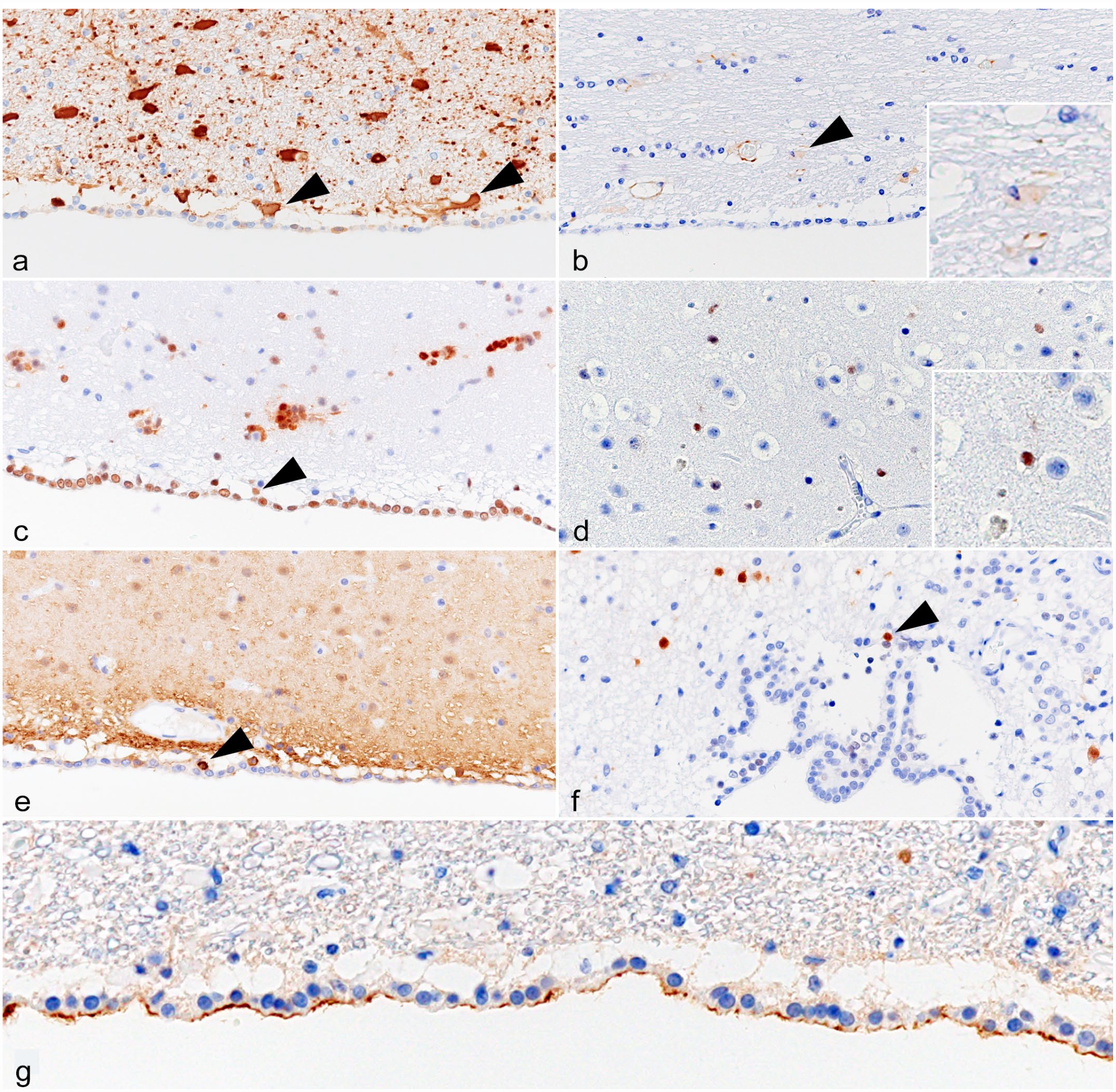
Oligodendrogliomas (cases 1 and 2): The nuclei of tumor cells were immunopositive for OLIG2 (scores 2 and 3) and SOX2 (score 2) (Fig. 4a, b). The cytoplasm and membrane of tumor cells were immunopositive for β-3 tubulin (score 3) and PDGFR-α (score 3) (Fig. 4c, d). In case 2, nuclear NeuN immunolabeling was observed in tumor cells (score 2) (Fig. 4e).
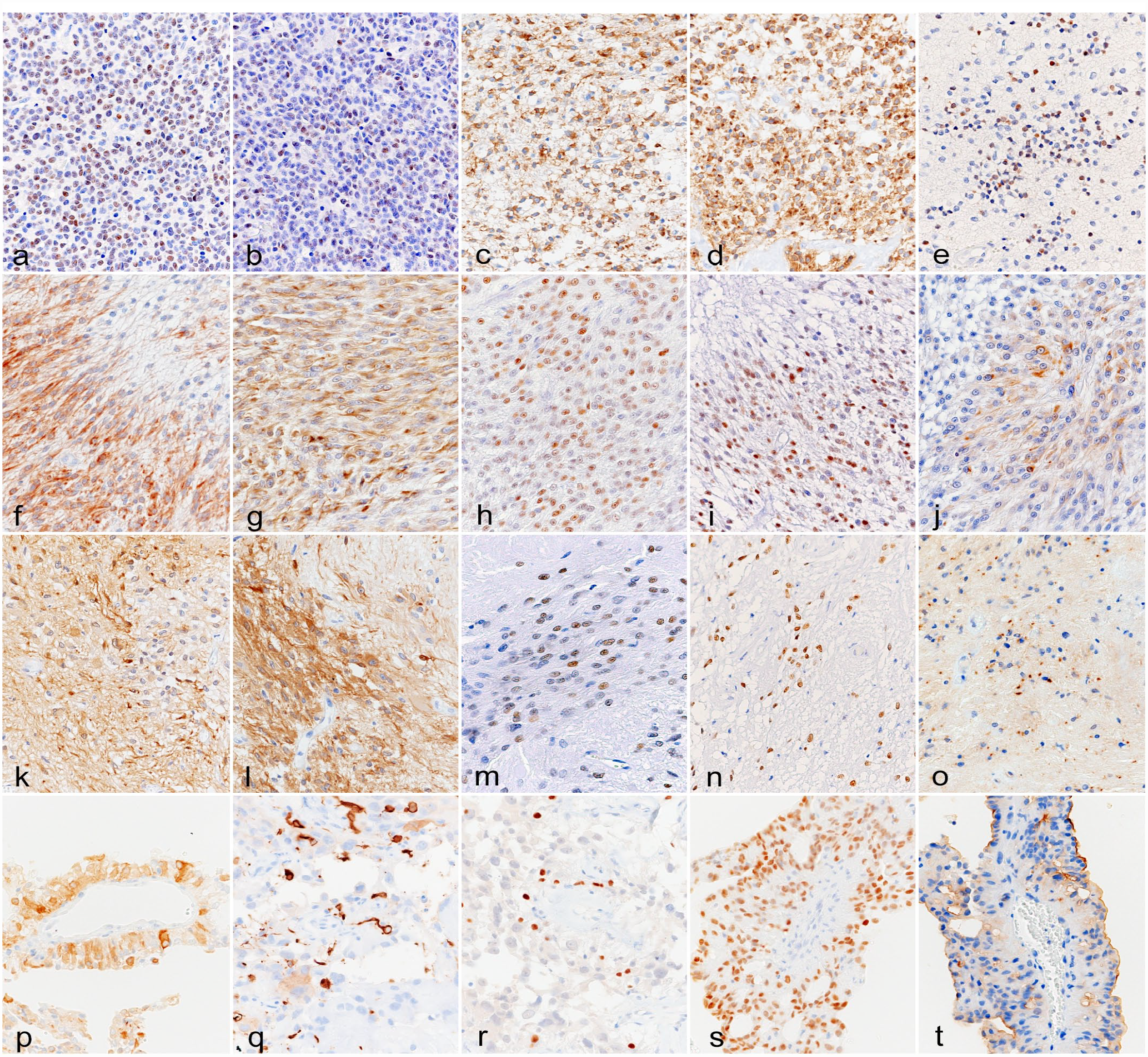
Glial tumor, brain, cat. Immunohistochemistry. (a) Anaplastic oligodendroglioma (case 2). Tumor cells are immunopositive for OLIG2 (score 2). (b) Anaplastic oligodendroglioma (case 2). Tumor cells are immunopositive for SOX2 (score 2). (c) Anaplastic oligodendroglioma (case 1). Tumor cells are immunopositive for β-3 tubulin (score 3). (d) Anaplastic oligodendroglioma (case 1). Tumor cells are immunopositive for PDGFR-α (score 3). (e) Anaplastic oligodendroglioma (case 2). Tumor cells are immunopositive for NeuN (score 2). (f) Glioblastoma (case 5). Tumor cells are immunopositive for GFAP (score 2). (g) Glioblastoma (case 5). Tumor cells are immunopositive for β-3 tubulin (score 3). (h) Glioblastoma (case 5). Tumor cells are immunopositive for OLIG2 (score 2). (i) Glioblastoma (case 5). Tumor cells are immunopositive for SOX2 (score 2). (j) Glioblastoma (case 5). Tumor cells are immunopositive for nestin (score 2). (k) Subependymoma (case 9). Tumor cells are immunopositive for GFAP (score 3). (l) Subependymoma (case 9). Tumor cells are immunopositive for β-3 tubulin (score 3). (m) Subependymoma (case 7). Tumor cells are immunopositive for OLIG2 (score 2). (n) Subependymoma (case 8). Tumor cells are immunopositive for SOX2 (score 3). (o) Subependymoma (case 7). Tumor cells show dot-like NHERF1-immunolabeling. (p) Ependymoma (case 11). Tumor cells are immunopositive for GFAP (score 2). (q) Ependymoma (case 12). Tumor cells are immunopositive for β-3 tubulin (score 1). (r) Ependymoma (case 12). Tumor cells are immunopositive for OLIG2 (score 1). (s) Ependymoma (case 11). Tumor cells are immunopositive for SOX2 (score 3). (t) Ependymoma (case 11). The apical membrane of tumor cells is immunopositive for NHERF1. OLIG2, oligodendrocyte transcription factor 2; SOX2, sex-determining region Y-box transcription factor 2; PDGFR-α, platelet-derived growth factor receptor-α; NeuN, neuronal nuclear; GFAP, glial fibrillary acidic protein; NHERF1, Na+/H+ exchanger regulatory factor 1.
Astrocytomas (cases 3–6): The cytoplasm of astrocytomas was immunopositive for GFAP (scores 1–3), NHERF1 (score 0 or 2), and β-3 tubulin (score 0 or 3) (Fig. 4f, g and Supplemental Fig. S7a, b). Nuclear OLIG2 immunolabeling was also observed in tumor cells (scores 1–3) (Fig. 4h). The nucleus and cytoplasm of tumor cells were immunopositive for SOX2 (scores 0, 2, and 3) and nestin (scores 0–2), respectively (Fig. 4i, j). The tumor cells of the anaplastic astrocytoma (case 4) were immunopositive for PDGFR-α (score 2), NeuN (score 3), and synaptophysin (score 2) (Supplemental Fig. S7c, d). The cytoplasm of tumor cells was immunopositive for cytokeratin AE1/AE3 (scores 1 and 2) (Supplemental Fig. S4c).
Subependymomas (cases 7–9): The cytoplasm of the subependymomas was immunopositive for GFAP (scores 2 and 3), β-3 tubulin (score 3), and nestin (scores 1–3) (Fig. 4k, l). Nuclear immunolabeling for OLIG2 (scores 1–3) and SOX2 (scores 2 and 3) was also observed in tumor cells (Fig. 4m, n). The tumor cells of cases 7 and 9 were immunopositive for PDGFR-α (score 1) and NeuN (scores 1 and 2) (Supplemental Fig. S7e). The tumor cells of case 9 were immunopositive for synaptophysin (score 2). All cases showed dot-like immunolabeling for NHERF1, while the number of dots was smaller in cases 8 and 9 than in case 7 (Fig. 4o). The cytoplasm of tumor cells was immunopositive for NHERF1 in all subependymomas (score 3). The cytoplasm of tumor cells was immunopositive for cytokeratin AE1/AE3 (scores 2 and 3) (Supplemental Fig. S4d).
Ependymomas (cases 10–13): The cytoplasm of ependymomas was immunopositive for GFAP (scores 2 and 3) (Fig. 4p) and nestin (scores 0–2). The cytoplasmic immunolabeling for β-3 tubulin was limited in ependymomas (scores 0 and 1) (Fig. 4q), excluding a parenchymal ependymoma (case 13, score 3). Nuclear immunolabeling for OLIG2 (scores 0–2) and SOX2 (scores 1–3) was also detected in tumor cells (Fig. 4r, s). The tumor cells of all cases showed apical membranous immunolabeling for NHERF1, and the cytoplasm of tumor cells was also immunopositive for NHERF1 in all ependymomas (scores 2 and 3) (Fig. 4t and Supplemental Fig. S7f). The cytoplasm of tumor cells was immunopositive for cytokeratin AE1/AE3 (scores 1 and 2) (Supplemental Fig. S4e, f).
Ki-67 Labeling Index
The results of Ki-67 LI are summarized in Supplemental Table S3. High-grade astrocytomas (cases 4–6) had a high Ki-67 LI (mean, 23.31; range, 17.66–27.79). Subependymomas had a low Ki-67 LI (mean, 4.78; range, 3.35–6.93), while an increase in Ki-67 LI was observed in a recurrent subependymoma (13.93, case 7 [recurrent]). Ki-67 LI was low in ependymomas (mean, 2.35; range, 1.23–3.47) and high in the anaplastic ependymoma (12.99, case 12). Immunolabeling for Ki-67 was not detected in 2 oligodendrogliomas (cases 1 and 2), 1 fibrillary astrocytoma (case 4), and 1 ependymoma (case 10).
Cluster Analysis of Immunohistochemical Scoring
The results of the cluster analysis are shown in Fig. 5. The dendrogram was constructed through a hierarchical clustering approach. Feline glial tumors were roughly divided into 2 groups. One group consisted of oligodendrogliomas and 1 astrocytoma (case 4), which showed immunolabeling for PDGFR-α. Another group consisted of astrocytomas, subependymomas, and ependymomas.
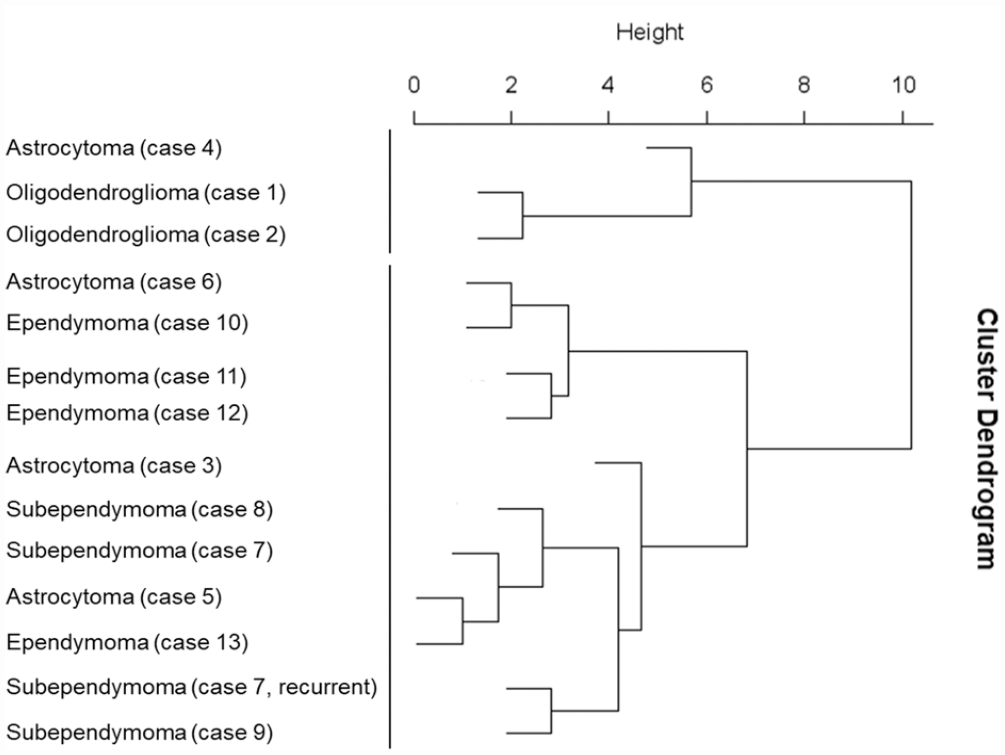
Dendrogram based on hierarchical cluster analysis of immunohistochemical scoring in feline glial tumors constructed using the Ward method shows that feline glial tumors are roughly divided into 2 groups. One group consisted of oligodendrogliomas and 1 astrocytoma (case 4), which is PDGFR-α immunopositive. Another group consisted of astrocytomas, subependymomas, and ependymomas. PDGFR-α, platelet-derived growth factor receptor-α.
Discussion
This study revealed the immunohistochemical characteristics of NSC lineage cells in feline newborn and mature brains. Similar to rodents, NSCs were observed in the VZ of newborn cats and were immunopositive for GFAP, nestin, and SOX2.11,14,19,26 SOX2-immunopositive IPCs in the SVZ were also observed and may differentiate into OLIG2-immunopositive glial cells and β-3 tubulin- and NeuN-immunopositive neurons in the developing brain.11,19,26 OLIG2- and PDGFR-α- or GFAP-immunopositive cells in the SVZ to IZ are considered OPCs and immature astrocytes, respectively.26,27,33 NHERF1 was previously reported to be expressed in the microvilli of ependymal cells in mice and humans.15,52 Therefore, apical membranous immunolabeling for NHERF1 in the VZ cells of newborn cats suggested that radial glia possessed microvilli, similar to human fetal brains. 61
NSC lineage cells are also present in adult rodent brains.26,29 GFAP- and SOX2-immunopositive cells in the VZ and SVZ, suggestive of B cells, and nestin-immunopositive cells in the SVZ, suggestive of activated B cells, were detected in adult cat brains.14,26,29 SOX2-immunopositive cells were identified in the adult feline SVZ, and a few of these cells were also positive for β-3 tubulin or NeuN, which likely represent C cells as adult IPCs and A cells as premature neurons, respectively.9,11,26,29 SOX2- and OLIG2- or GFAP-immunopositive cells in feline brains may be adult glial progenitor cells based on previous reports.1 –3,14,34,39,53,62 Murine ependymal cells are immunopositive for SOX2, and apical membranous immunolabeling for NHERF1 has been demonstrated.14,15 These findings were confirmed in feline adult brains. NHERF1 is associated with GFAP and is expressed in the astrocytic process. 54 NHERF1 expression in feline astrocytic processes was also observed in our study.
Feline astrocytomas, subependymomas, and ependymomas were classified into the same group by cluster analysis of the immunohistochemical results, excluding 1 astrocytoma (case 4). These tumors commonly expressed GFAP, nestin, and SOX2, which were also expressed in feline NSCs, suggesting an NSC-like immunophenotype for these tumor types. In support of this, the characteristics of NSCs have been reported in human astrocytoma, subependymoma, and ependymoma, and stem cell markers, including SOX2, nestin, CD133, Musashi, CD15, and CD44, were expressed in human glial tumors.7,38,43,44,46
There were several immunohistochemical differences between feline astrocytomas, subependymomas, and ependymomas. The most significant difference was NHERF1 immunolabeling patterns. Subependymomas had dot-like immunolabeling, while ependymomas had apical membranous immunolabeling, which is consistent with human subependymomas and ependymomas.7,15,37 Histologically, subependymomas have long processes similar to radial glia, and ependymomas mimic the polarized lining of the ventricles. Therefore, immunolabeling for NHERF1 in subependymomas and ependymomas may represent radial glial and ependymal cell characteristics, respectively. The cytoplasm of feline astrocytomas was positive for NHERF1, consistent with astrocytic differentiation, as normal feline astrocytes expressed NHERF1.
Immunolabeling for PDGFR-α was observed in the feline anaplastic oligodendroglioma. In human, murine, and canine oligodendrogliomas, an OPC immunophenotype was characterized by the expression of PDGFR-α, neural/glial antigen 2, and SOX10.22,30,42,60 In addition, feline subependymomas contained a small number of PDGFR-α-immunopositive cells, which have also been reported for human subependymomas. 7 These results suggest that OPC-like cells are present in varying proportions in feline anaplastic oligodendrogliomas and subependymomas. However, further study with more cases is needed to conclude this. In this study, oligodendrogliomas and 1 astrocytoma (case 4) were also immunopositive for PDGFR-α. This may be the reason why the PDGFR-α-immunopositive astrocytoma (case 4) was included in the oligodendroglioma group by cluster analysis. Moreover, OLIG2-positive cells were detected in feline oligodendrogliomas, astrocytomas, and subependymomas, whereas ependymomas had a limited population of OLIG2-positive cells. OLIG2 expression is observed in developing oligodendrocytes and astrocytes in humans and rodents.27,33 Based on these findings, evidence of oligodendroglial and astrocytic differentiation is observed in oligodendrogliomas and astrocytomas, and may be present in subependymomas in cats, but may be a limited event in feline ependymomas. However, a previous study reported that feline ependymomas were widely immunopositive for OLIG2. 8 This difference may be attributed to an insufficient number of cases or differing methods used in this study.
β-3 tubulin-positive cells were frequently detected in feline oligodendrogliomas, astrocytomas, and subependymomas. β-3 tubulin expression has also been reported in human and canine astrocytomas and oligodendrogliomas.21,60 On the contrary, the feline ependymomas of this study contained limited numbers of β-3 tubulin-positive cells, except for the parenchymal ependymoma (case 13). Consistent with a few neoplastic cells, immunolabeling for β-3 tubulin in our feline ependymomas, neuronal differentiation characterized by NeuN, neurofilament protein, chromogranin, and synaptophysin is an uncommon feature in human ependymomas. 49 In this study, NeuN and synaptophysin were also used as neuronal differentiation markers. Immunolabeling for NeuN, synaptophysin, and neuron-specific enolase has been reported in human, canine, and feline glial tumors.4,5,7,18,28,48,60 Although a previous study showed the lack of NeuN immunolabeling in feline oligodendrogliomas and astrocyomas, 10 the present results indicated that the tumor cells of feline oligodendrogliomas, astrocytomas, and subependymomas possessed some ability for neuronal differentiation and for multipotential stemness. Further analyses of a larger number of cases are needed to support the hypothesis.
Parenchymal ependymoma is a rare tumor in humans, and to the best of our knowledge, parenchymal ependymoma in other species has not yet been reported.18,25,55 In humans, supratentorial ependymoma may or may not have an obvious connection to the ventricular system. 55 Case 13 in this study was diagnosed as parenchymal ependymoma based on the papillary growth pattern and perivascular arrangement of tumor cells; however, the tumor was located in the left temporal lobe without a clear connection to the ventricle.
All ependymomas in this report had cytokeratin immunolabeling as in the previous reports in cats and dogs.20,35,59 However, cytokeratin immunolabeling was also observed in astrocytomas and subependymomas. Therefore, ependymomas cannot be distinguished from astrocytomas and subependymomas solely by cytokeratin expression.
In conclusion, this study supports the presence of feline NSC lineage cells in feline newborn and mature brains. Feline astrocytoma, subependymoma, and ependymoma cells commonly exhibited the NSC-like immunophenotype, and each of these tumors showed the characteristics of glial cells, OPCs, and ependymal cells, respectively. On the contrary, feline anaplastic oligodendroglioma cells showed a similar immunophenotype to OPCs. These results suggest that feline glioma and ependymal tumors may originate from NSC lineage cells in the mature brain. A limitation of this study was the usage of antibodies that labeled both immature and mature cells. These preliminary results should be validated by gene expression analyses in future studies with larger case numbers.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231182337 – Supplemental material for Immunohistochemical analyses of neural stem cell lineage markers in normal feline brains and glial tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858231182337 for Immunohistochemical analyses of neural stem cell lineage markers in normal feline brains and glial tumors by Kio Yoshida, James K. Chambers, Kazumi Nibe, Yumiko Kagawa and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
