Abstract
Neural stem cells (NSCs) produce neuron intermediate progenitor cells (nIPC), oligodendrocyte precursor cells (OPCs), and immature astrocytes. To confirm NSC lineages in the normal canine brain and the association of these cells with gliomas, an immunohistochemical study was conducted on fetal and adult canine brains, gliomas, and a glioma cell line. In fetal brains, glial fibrillary acidic protein (GFAP)- and nestin-immunolabeled NSC were observed in the ventricular zone, β-3 tubulin- and/or neuronal nuclei (NeuN)-immunolabeled nIPC in the subventricular zone (SVZ), and platelet-derived growth factor receptor-α (PDGFR-α)- and OLIG2-immunolabeled OPC and GFAP- and OLIG2-immunolabeled immature astrocytes in the SVZ and intermediate zone. Ki-67 immunohistochemistry revealed that nIPC exhibited high proliferative activity. Quiescent nIPC and OPC were observed in adult brains. Among 58 glioma cases including 4 low-grade oligodendrogliomas (LGOGs), 48 high-grade oligodendrogliomas (HGOGs), 1 low-grade astrocytoma, and 5 high-grade astrocytomas (HGACs), immunohistochemical analyses revealed that oligodendrogliomas expressed PDGFR-α and OLIG2, whereas astrocytomas expressed GFAP and OLIG2. HGOG showed significantly higher immunohistochemical scores for NeuN and β-3 tubulin than LGOG. The Ki-67 labeling index was high in PDGFR-α and NeuN-immunolabeled tumor cells, and low in β-3 tubulin- and synaptophysin-immunolabeled cells. A HGOG cell line possessed the same immunohistochemical characteristics as HGOG. In this study, glioma cells with the OPC and IPC immunophenotypes had a higher Ki-67 labeling index, indicating their high proliferative activity. Furthermore, high-grade gliomas showed the characteristics of nIPC and neurons, which may suggest the pluripotent NSC lineage nature of these tumors.
Keywords
Gliomas are the second most common intracranial tumor in dogs.10,22 Immunohistochemical markers for astrocytic differentiation, such as GFAP, and oligodendroglial differentiation, including OLIG2 and 2’, 3’-cyclic nucleotide 3’-phosphodiesterase (CNPase), are useful to support a histopathological diagnosis of astrocytoma and oligodendroglioma, respectively.8,15 However, astrocytic and oligodendroglial markers are often both positive in canine gliomas, 13 suggesting that pluripotent stem cells are the cell of origin in these cases. Human gliomas originate from NSC, OPC, or astrocytes. 33 Although the origin of canine gliomas has not yet been elucidated, we previously demonstrated that OPC markers, such as PDGFR-α, SRY-box transcription factor 10 (SOX10), and neuron-glial antigen 2 (NG2), were expressed in canine HGOG. 11 Other reports describe the presence of cancer stem cell (CSC) which expresses stem cell markers (nestin, CD133, and activating transcription factor 5) in canine gliomas.4,29,32
In mice, NSC produce intermediate progenitor cells (IPC) for neurons (nIPC), oligodendrocytes, and astrocytes. 14 OPC appear to correspond to IPC for oligodendrocyte, which are committed to an oligodendroglial lineage. Various combinations of immunohistochemical markers have been used to characterize different types of NSC. Nestin is an intermediate filament that was discovered in neuroepithelial cells and is expressed in NSC.14,18 β-3 tubulin, a structural element of microtubules, is expressed in a subpopulation of neuroepithelial cells and during neuronal cell differentiation. 9 The NeuN antigen is an immunohistochemical marker for postmitotic neuroblasts and neurons. 6 PDGFR-α, a receptor for PDGFA, is the best-characterized marker for OPC. 15 Astrocytes and neurons produce PDGFA and promote the proliferation of OPC. 15 OLIG2 is needed for IPC to develop into OPC, and is expressed by oligodendroglial lineage cells and developmental astrocytes, but not by mature astrocytes.15,21
Previous studies have shown that multipotent cells can be cultured from the canine olfactory bulb, SVZ, subgranular zone, mesencephalon, and the spinal cord.3,19,20,23,28,31 These stem cells form spheres and express NSC markers, such as nestin, SOX2, and GFAP.3,19,20,23,28,31 These cells can differentiate into neurons (expressing β-3 tubulin, microtubule-associated protein 2, and neurofilament), astrocytes (expressing GFAP), and oligodendrocytes (expressing CNPase and oligodendrocyte marker o4).3,19,20,23,31 Also, canine glial restricted progenitor cells, which differentiated into astrocytes, have been isolated. 12 Immunohistochemically, nIPC (expressing doublecortin and NeuN) and NSC (expressing Sox2 and GFAP) are found in the subgranular zone. 20 Doublecortin-immunolabeled cells are also reported in canine SVZ. 2 However, canine NSC, IPC, and OPC are not sufficiently characterized immunohistochemically.
Therefore, this study investigated whether NSC, IPC, and OPC were present in the canine fetal brain using immunohistochemical markers. Canine glioma tissues and the colonies of a cell line derived from HGOG were immunohistochemically examined using NSC, IPC, and OPC markers to reveal tumor cell heterogeneity and characterize their immunophenotypes. Furthermore, the Ki-67 labeling index (LI) was assessed in each tumor cell population to show their different proliferative activities.
Materials and Methods
Tissue Samples
Formalin-fixed paraffin-embedded tissues of 58 canine gliomas (46 biopsies and 12 necropsies) were retrieved from the archives of the Laboratory of Veterinary Pathology, University of Tokyo. Case details are summarized in Supplemental Table 1. Tissues were sectioned at a thickness of 4 µm and stained with hematoxylin and eosin. Each case was diagnosed and graded according to the classification scheme described by the National Cancer Institute-led Multidisciplinary Comparative Brain Tumor Consortium. 13
Average immunohistochemical scores and statics analysis for canine gliomas and immunocytochemical scores in an AOFB-01 cell line.
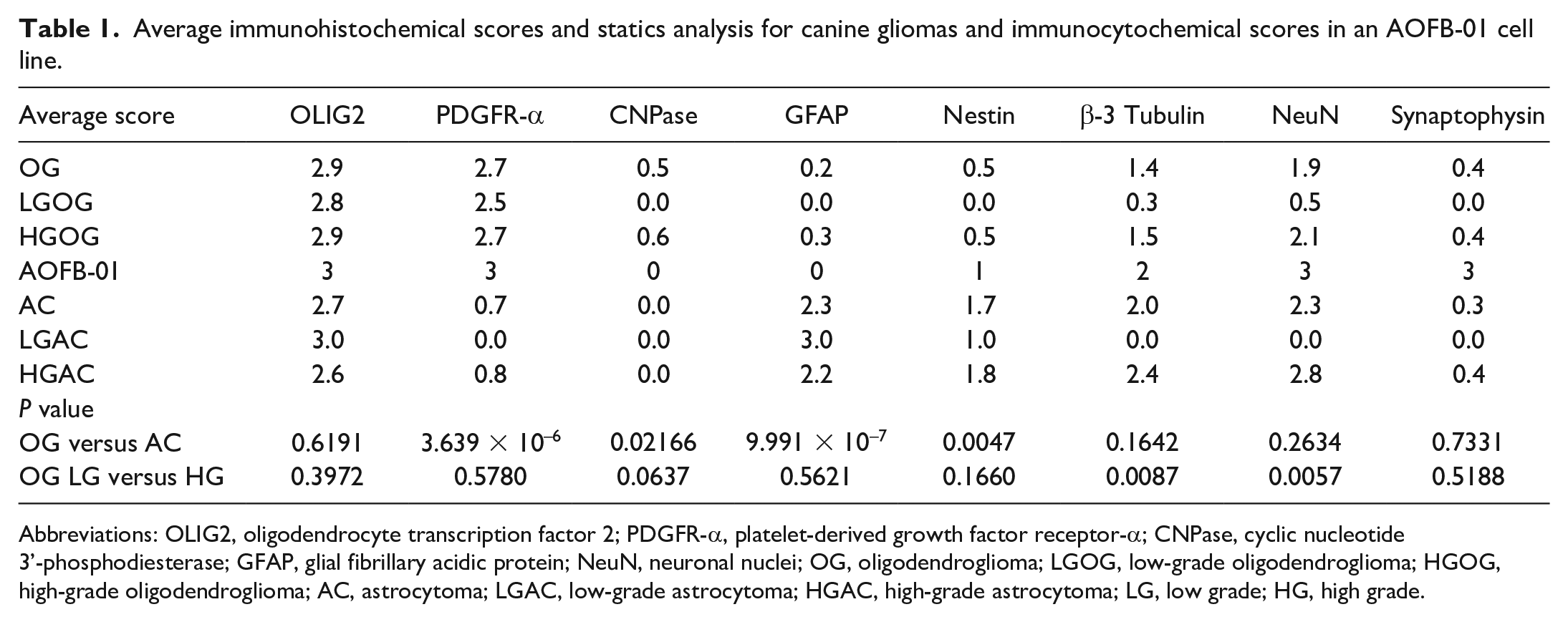
Abbreviations: OLIG2, oligodendrocyte transcription factor 2; PDGFR-α, platelet-derived growth factor receptor-α; CNPase, cyclic nucleotide 3’-phosphodiesterase; GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei; OG, oligodendroglioma; LGOG, low-grade oligodendroglioma; HGOG, high-grade oligodendroglioma; AC, astrocytoma; LGAC, low-grade astrocytoma; HGAC, high-grade astrocytoma; LG, low grade; HG, high grade.
The brains of 3 canine fetuses (2 Chihuahuas and 1 Toy poodle) with an approximate gestation age of 6–8 weeks that were collected through routine necropsy at the laboratory were examined. The contralateral cerebral hemispheres of 3 glioma cases (cases 5, 7, and 9) were used as adult controls.
Immunohistochemistry
Immunohistochemistry was performed using the primary antibodies summarized in Supplemental Table 2. Deparaffinized tissue sections were incubated with 10% hydrogen peroxide in methanol at room temperature for 5 minutes to block endogenous peroxidase. Antigen retrieval conditions were summarized in Supplemental Table 2. Tissue sections were incubated with 8% skim milk-Tris-buffered saline (pH 7.6) at 37°C for 40 minutes to prevent nonspecific reactions. Then, these sections were incubated with primary antibodies at 4°C overnight. Dilution of primary antibodies was summarized in Supplemental Table 2. A horseradish peroxidase-labeled anti-mouse or anti-rabbit IgG polymer (EnVision+, Dako) was used as the secondary antibody and applied at 37°C for 40 minutes. 3-3’-diaminobenzidine (DAB) was used as the chromogen. Counterstaining was performed using hematoxylin. As negative controls, each primary antibody was replaced by an irrelevant antibody. Primary antibodies were validated by Western blotting (Supplemental File 1).
Average Ki-67 LI in tumor cells and in dual-labeling immunochemistry.
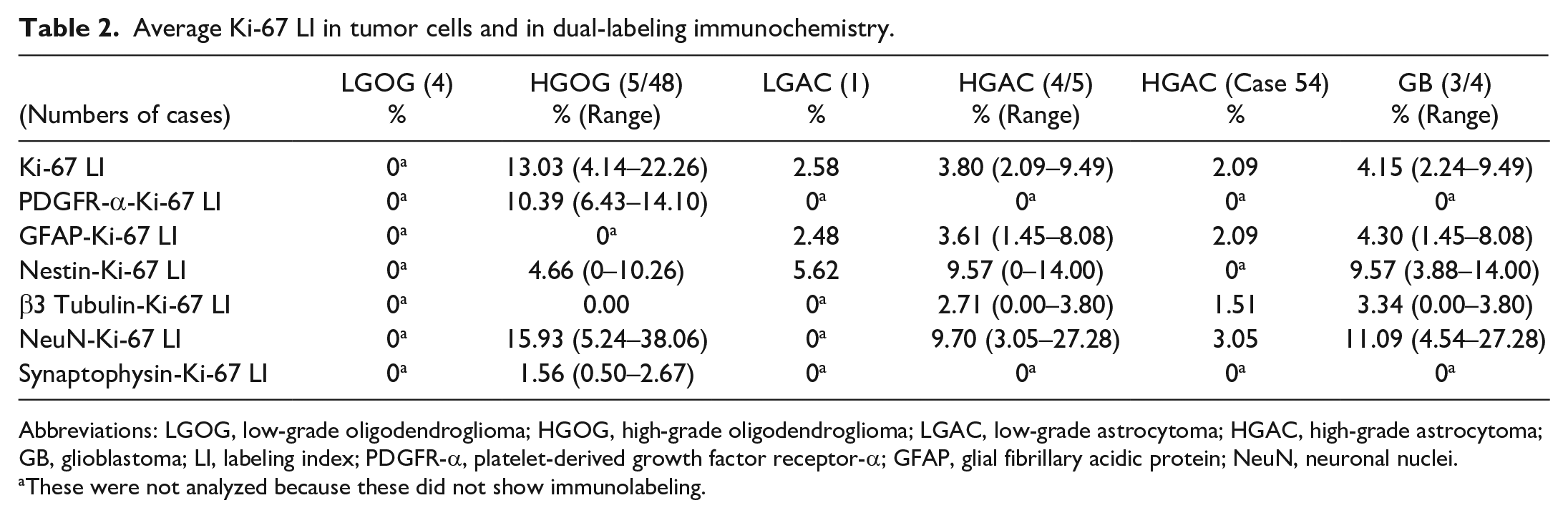
Abbreviations: LGOG, low-grade oligodendroglioma; HGOG, high-grade oligodendroglioma; LGAC, low-grade astrocytoma; HGAC, high-grade astrocytoma; GB, glioblastoma; LI, labeling index; PDGFR-α, platelet-derived growth factor receptor-α; GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei.
These were not analyzed because these did not show immunolabeling.
Dual-labeling Immunohistochemistry and Immunofluorescence
Dual-labeling immunohistochemistry was conducted using 2 different primary antibodies with the combinations of rabbit polyclonal anti-OLIG2 antibody/rabbit polyclonal anti-PDGFR-α antibody, mouse monoclonal (MIB-1) anti-Ki-67 antibody/rabbit polyclonal anti-PDGFR-α antibody, mouse monoclonal (MIB-1) anti-Ki-67 antibody/mouse monoclonal (5G8) anti-β-3 tubulin antibody, mouse monoclonal (MIB-1) anti-Ki-67 antibody/mouse monoclonal (A60) anti-NeuN antibody, mouse monoclonal (MIB-1) anti-Ki-67 antibody/mouse monoclonal (DAK-SYNAP) anti-synaptophysin antibody, rabbit polyclonal anti-GFAP antibody/mouse monoclonal (MIB-1) anti-Ki-67 antibody, and rabbit monoclonal (N1602) anti-nestin antibody/mouse monoclonal (MIB-1) anti-Ki-67 antibody (antibody for first target/antibody for second target). Procedures for blocking, antigen retrieval, and primary antibody incubation were the same as single-labeling immunohistochemistry. A horseradish peroxidase-labeled anti-mouse or anti-rabbit IgG polymer (EnVision+, Dako) and alkaline phosphatase-labeled anti-mouse or anti-rabbit IgG polymer (Histofine, Simple Stain, Nichirei Bioscience) were used as the secondary antibody and applied at 37°C for 40 minutes. DAB, New Fuchsin (Histofine, Nichirei Bioscience), and HIGHDEF blue (Enzo Life sciences) were used as chromogens. After visualizing the first target, immunohistochemistry for the second target was performed following heat-induced epitope retrieval. Counterstaining was conducted using hematoxylin or none.
Dual-labeling immunofluorescence was performed for GFAP/nestin, OLIG2/PDGFR-α, OLIG2/GFAP, and NeuN/β-3 tubulin. Antigen retrieval was conducted as follows: none for GFAP/nestin, heat-induced epitope retrieval in target retrieval solution (Dako; pH 9) for OLIG2/PDGFR-α, and heat-induced epitope retrieval in citrate buffer (pH 6) for OLIG2/GFAP and NeuN/β-3 tubulin. Procedures for blocking and primary antibody incubation were the same as single-labeling immunohistochemistry. Dilution of a rabbit monoclonal (N1602) anti-nestin antibody was 1:10, and dilution of other primary antibodies was the same as single-labeling immunohistochemistry. Sections were incubated with a rabbit polyclonal anti-GFAP antibody at 4°C overnight and then with an Alexa Fluor 594-labeled donkey anti-rabbit IgG (Thermo Fisher Scientific) secondary antibody at 37°C for 1 hour. After washing with Tris-buffered saline, sections were incubated with a rabbit monoclonal (N1602) anti-nestin antibody labeled with Alexa Fluor 488 (rabbit IgG labeling kit, Zenon, Invitrogen) at 37°C for 2 hours. Sections were then postfixed by 4% formalin in Tris-buffered saline at room temperature for 15 minutes. Regarding OLIG2/PDGFR-α, mouse monoclonal (211F1.1) anti-OLIG2 and rabbit polyclonal anti-PDGFR-α antibodies were used as the primary antibodies and applied at 4°C overnight. Alexa Fluor 594-labeled goat anti-mouse IgG (Thermo Fisher Scientific) and Alexa Fluor 488-labeled goat anti-rabbit IgG (Thermo Fisher Scientific) were used as the secondary antibodies and applied at 37°C for an hour. Concerning OLIG2/GFAP, mouse monoclonal (211F1.1) anti-OLIG2 and rabbit polyclonal anti-GFAP antibodies were used as the primary antibodies, and Alexa Fluor 488-labeled goat anti-mouse IgG (Thermo Fisher Scientific) and Alexa Fluor 594-labeled donkey anti-rabbit IgG as the secondary antibodies. For NeuN/β-3 tubulin, rabbit monoclonal (EPR12763) anti-NeuN and mouse monoclonal (5G8) anti-β-3 tubulin antibodies were used as the primary antibodies. Alexa Fluor 488-labeled goat anti-rabbit IgG and Alexa Fluor 594-labeled goat anti-mouse IgG were used as the secondary antibodies. Incubation conditions for OLIG2/GFAP and NeuN/β-3 tubulin were the same as the OLIG2/PDGFR-α. Dilution of secondary antibodies was 1:400. Sections were counterstained with DAPI (Vectashield; Vector Laboratory) and observed under a laser scanning confocal microscope (LSM700, Zeiss). All images were captured in single region of interest with × 40 objective and presented as a single z-plane. In OLIG2/GFAP image, z-stack images were shown as the column on the right and bottom.
Immunohistochemical Scoring and Statistical Analysis
The immunohistochemical results of gliomas and the immunocytochemical results of the cell line were scored as follows: 0: no positive cells; 1: <25% positive cells; 2: 25%–75% positive cells; and 3: >75% positive cells. OLIG2 and NeuN were scored using rabbit polyclonal anti-OLIG2 and mouse monoclonal (A60) anti-NeuN antibodies, respectively. Scores were tested by the Mann-Whitney U test for oligodendrogliomas versus astrocytomas and LGOG versus HGOG. Statistical analyses were performed using R version 4.2.0 (The R Foundation for Statistical Computing). A P-value <.01 was considered to be significant. The number of astrocytoma cases was insufficient to perform a statistical analysis between low-grade astrocytomas (LGAC) and HGAC. For evaluation of β-3 tubulin-, NeuN-, and synaptophysin-immunolabeled cells, tumor cells were distinguished from entrapped neurons based on their cellular atypia and diffuse distribution.
Ki-67 LI
Ki-67 LI was calculated by the number of Ki-67-immunolabeled cells divided by the number of tumor cells. Cells were counted using 5 high-power fields (40 × objective, 0.11 mm2) images taken at hot spots defined as the area with the highest number of immunolabeled cells on the slide. Similarly, Ki-67 LI in PDGFR-α-, β-3 tubulin-, NeuN-, synaptophysin-, GFAP-, and nestin-immunolabeled cells were calculated using dual-labeled slides. Five HGOG cases were selected (cases 14, 16, 24, 28, and 46) to evaluate Ki-67 LI in PDGFR-α-, β-3 tubulin-, and NeuN-immunolabeled cells. These cases were selected based on the amount of tumor tissue. Five HGOG cases with immunohistochemical scores 2 or 3 for synaptophysin were selected (cases 28, 30, 35, 41, and 48) to assess Ki-67 LI in synaptophysin-immunolabeled cells. Since LGOG cases were negative for Ki-67, Ki-67 LI was not evaluated in dual-labeled slides. Ki-67 LI was evaluated in GFAP- and nestin-immunolabeled cells for LGAC and in GFAP-, nestin-, β-3 tubulin-, and NeuN-immunolabeled cells for HGAC. Because 1 HGAC (case 56) showed very weak nuclear immunolabeling for Ki-67, this case was not analyzed for Ki-67 LI. NeuN-immunolabeled cells were counted using mouse monoclonal (A60) anti-NeuN antibody. Similar to immunohistochemical scoring, entrapped neurons were distinguished from tumor cells based on their cellular atypia and diffuse distribution.
In vitro Colony Formation by the Glioma Cell Line and Immunocytochemistry
We previously established a cell line (AOFB-01) from an HGOG case (case 9 in this case). 8 Cells were maintained in Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12, Gibco), 10% fetal bovine serum (Cytiva or Serana), and 1% antibiotics (penicillin streptomycin, Fujifilm) in a humidified atmosphere of 5% CO2 and 95% air at 37°C, and medium was changed every 2 days. Subconfluent cultured cells were washed with sterile phosphate-buffered saline (pH 7.4), dispersed with 0.05 w/v% trypsin-0.53 mmol/L EDTA・4Na solution with Phenol Red (Fujifilm), and collected by centrifugation at 1000 rpm at 4°C for 5 minutes. After rinsing with phosphate-buffered saline, cells were suspended in Matrigel (Corning) and pipetted onto a 30-mm plastic dish or 24-well plate, incubated upside down for gelatinization, and cultured in DMEM/F-12 with 10% fetal bovine serum. After colonies had formed, gels were fixed in 10% neutral-buffered formalin or 4% paraformaldehyde, embedded in paraffin, sectioned at a thickness of 4 μm, and stained with HE. Immunocytochemistry was conducted using the same method for immunohistochemistry. Immunocytochemical results were assessed by the same scoring system in immunohistochemistry.
Results
Immunohistochemistry of Fetal and Adult Canine Brains
In fetal canine brains, single-labeled immunohistochemistry revealed cytoplasmic immunolabeling for GFAP and nestin in the ventricular zone (VZ) (Fig. 1a, b). GFAP- and nestin-immunolabeled cells lined the surface of the ventricle and had radial projections into the SVZ, consistent with radial glial cells. 14 In the VZ, SVZ, and cortical plate (CP), numerous cells showed cytoplasmic and nuclear immunolabeling for β-3 tubulin and NeuN, respectively (Fig. 1c, d). These cells in the VZ and SVZ, and cells in the CP were consistent with nIPC and immature neuron, respectively.6,9,14 Nuclear immunolabeling for OLIG2- and cytoplasmic and membrane immunolabeling for PDGFR-α were observed in the SVZ and intermediate zone (IZ) (Fig. 1e, f), consist with OPC. 15 The neuropils of IZ and CP had immunolabeling for synaptophysin and β-3 tubulin. The fetal brain lacked immunolabeling for CNPase.
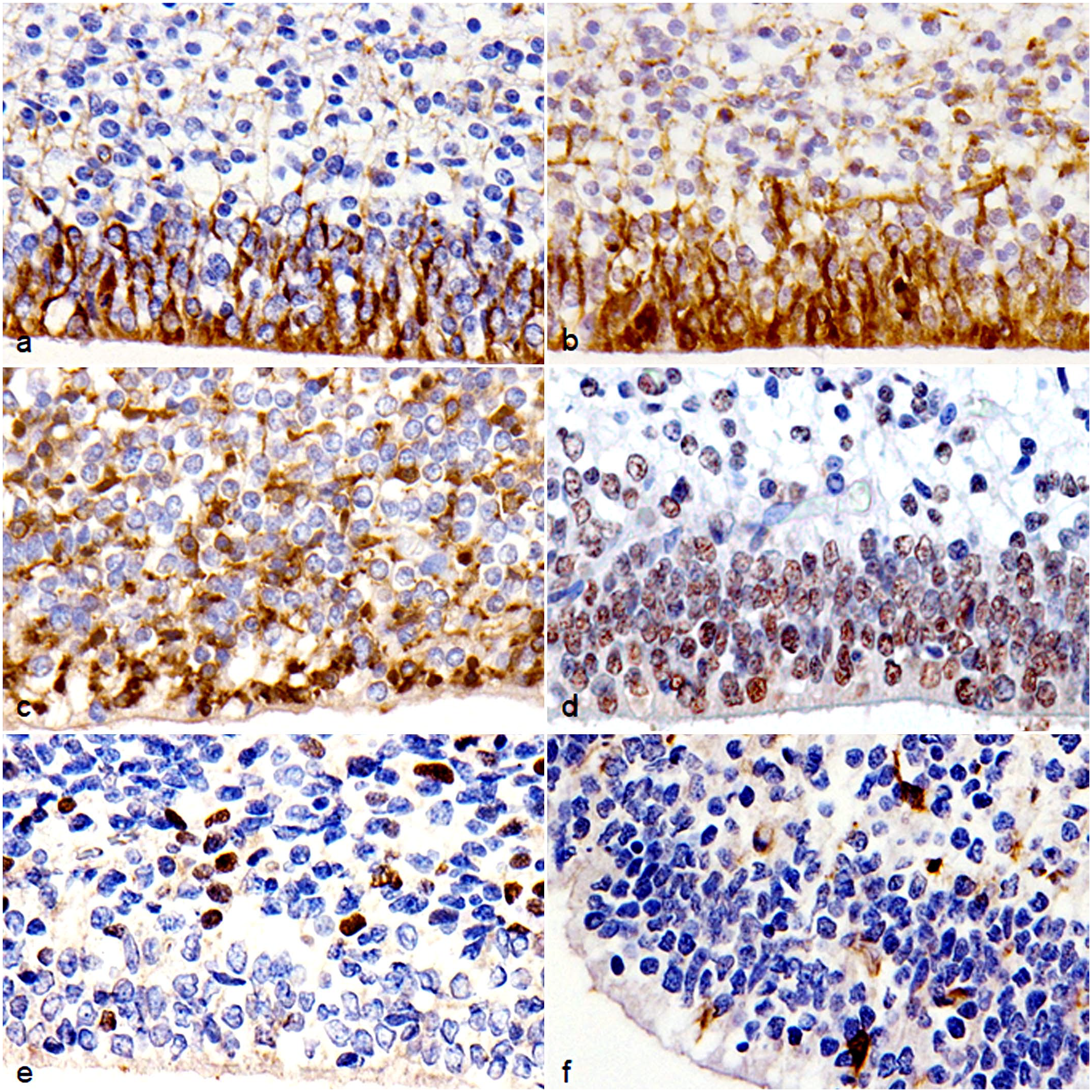
Brain, dog (Chihuahua), fetus. Immunohistochemistry. (a) Cells lining the surface of the VZ are immunolabeled for GFAP. (b) Cells lining the surface of the VZ are immunolabeled for nestin. (c) Cells in the VZ and SVZ are immunolabeled for β-3 tubulin. (d) Cells in the VZ and SVZ are immunolabeled for NeuN. (e) Cells in the SVZ are immunolabeled for OLIG2. (f) Cells in the SVZ are immunolabeled for PDGFR-α. Abbreviations: GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei; OLIG2, oligodendrocyte transcription factor 2; PDGFR-α, platelet-derived growth factor receptor-α.
Dual-labeling confirmed that most cells lining the surface of the ventricle suspected to be radial glial cells coexpressed GFAP and nestin (Fig. 2a). 14 β-3 tubulin- and/or NeuN-immunolabeled cells, consistent with nIPCs, were observed in the VZ and SVZ (Fig. 2b).6,9,14 Numerous β-3 tubulin- and NeuN-dual-immunolabeled cells, consistent with immature neurons, were also found in the CP.6,9,14 OLIG2- and PDGFR-α-dual-immunolabeled OPCs were detected in the SVZ and IZ (Fig. 2c). 15 Few OLIG2- and GFAP-dual-immunolabeled cells, likely representing immature astrocytes, were also observed in the SVZ and IZ (Fig. 2d). 21 GFAP- and nestin-immunolabeled radial glial cells in the VZ sporadically had nuclear Ki-67 immunolabeling (Fig. 3a, b). β-3 tubulin- and NeuN-immunolabeled nIPCs in the SVZ frequently had immunolabeling for Ki-67 (Fig. 3c, d), whereas β-3 tubulin- and NeuN-immunolabeled immature neurons in the CP lacked Ki-67 immunolabeling. PDGFR-α-immunolabeled OPCs and GFAP-immunolabeled immature astrocytes in the SVZ and IZ had sporadic immunolabeling for Ki-67 (Fig. 3e, f).
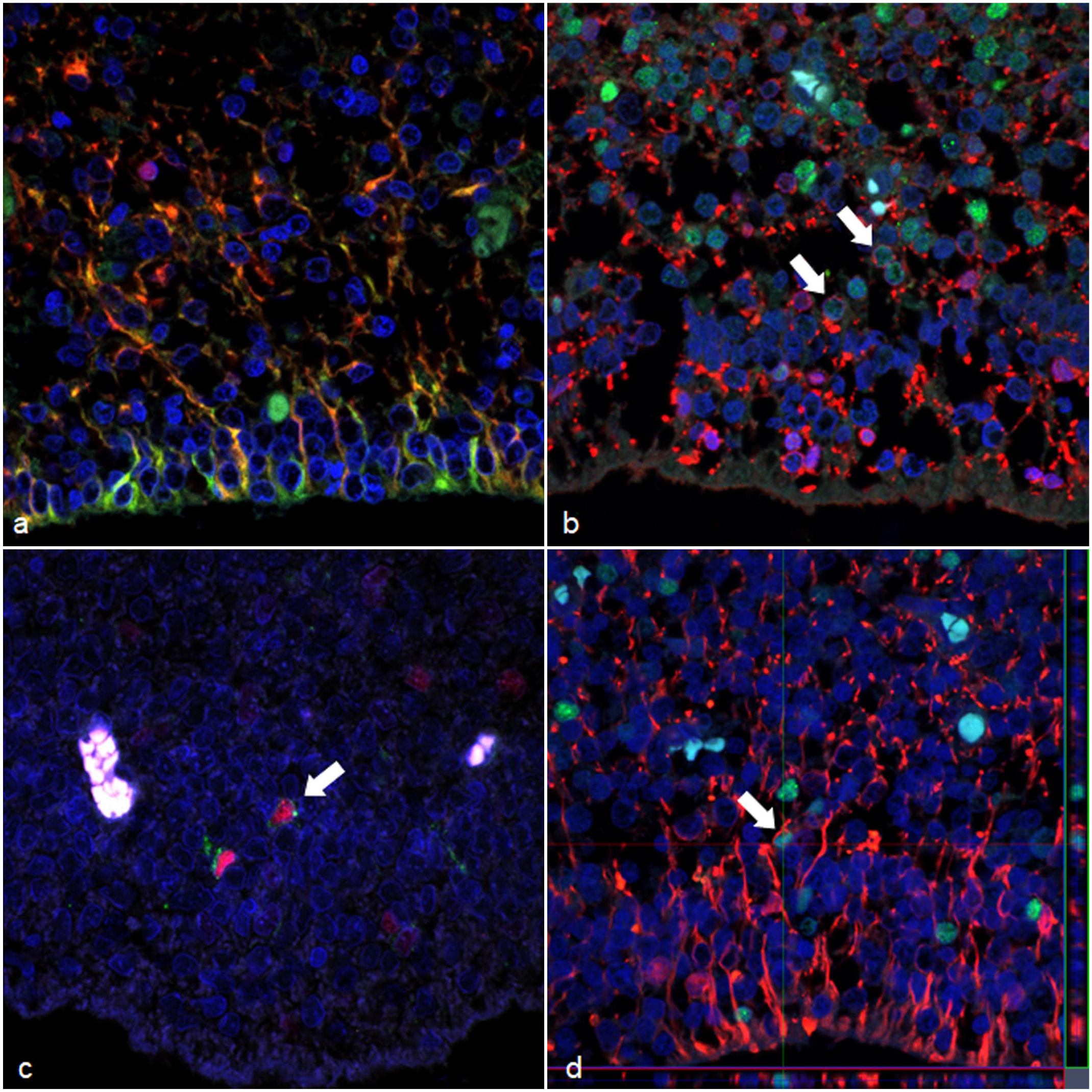
Brain, dog (Chihuahua), fetus. Dual-labeling immunofluorescence. (a) Radial glial in the VZ are immunolabeled for GFAP (red) and nestin (green) with coexpression demonstrated in yellow. (b) nIPC in the SVZ are immunolabeled for NeuN (green) and/or β-3 tubulin (red). Cells dual-labeled for NeuN and β-3 tubulin are shown by arrows. (c) OPC in the SVZ are immunolabeled for PDGFR-α (green) and OLIG2 (red) (arrow). (d) Immature astrocytes in the SVZ are immunolabeled for GFAP (red) and OLIG2 (green) (arrow). Red or green lines show positions for z-stack images under or right side of the figure, respectively. Z-stack images show that a pointed cell with arrow has cytoplasmic immunolabeling for GFAP. Abbreviations: GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei; PDGFR-α, platelet-derived growth factor receptor-α; OLIG2, oligodendrocyte transcription factor 2.
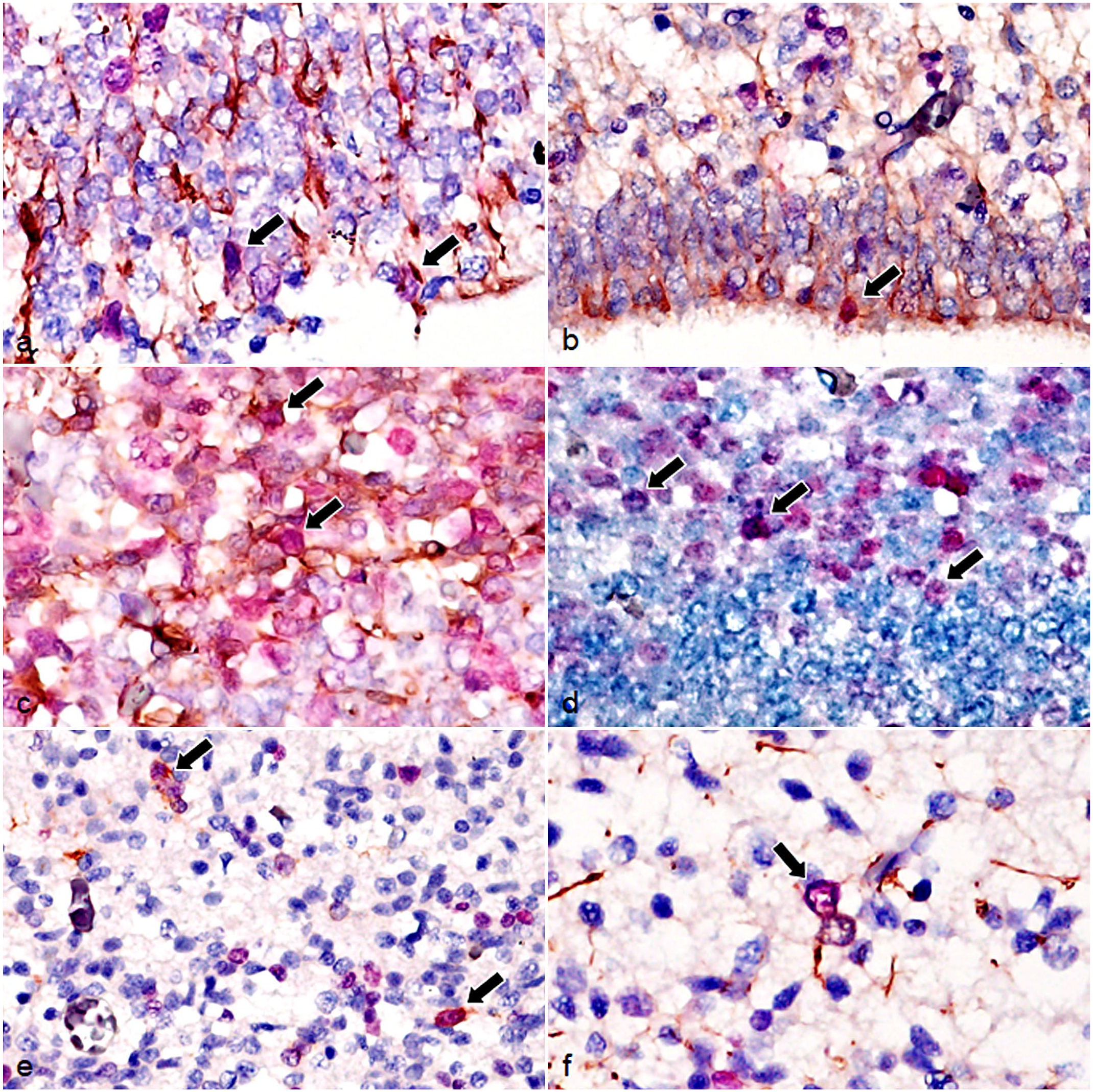
Brain, dog (Chihuahua), fetus. Dual-labeling immunohistochemistry for Ki-67 and other cell markers. (a) GFAP-immunolabeled cells (brown) in the VZ are immunolabeled for Ki-67 (red) (arrows). (b) Nestin-immunolabeled cells (brown) in the VZ are immunolabeled for Ki-67 (red) (arrow). (c) β-3 tubulin-immunolabeled cells (brown) in the SVZ are immunolabeled for Ki-67 (red) (arrows). (d) NeuN-immunolabeled cells (blue) in the SVZ are immunolabeled for Ki-67 (red) (arrows). (e) PDGFR-α-immunolabeled cells (brown) in the IZ are immunolabeled for Ki-67 (red) (arrows). (f) GFAP-immunolabeled cells (brown) in the IZ are immunolabeled for Ki-67 (red) (arrow). Abbreviations: GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei; PDGFR-α, platelet-derived growth factor receptor-α.
In the SVZ of adult canine brains, single-labeled immunohistochemistry revealed a small number of NeuN- and β-3 tubulin-immunolabeled cells, consistent with nIPCs.6,9,14 GFAP-immunolabeled astrocytes were also detected in the SVZ. PDGFR-α-immunolabeled OPCs and CNPase-immunolabeled oligodendrocytes were observed in the white matter. 15 Furthermore, astrocytes and oligodendrocytes in the cortex and white matter were immunolabeled for GFAP and OLIG2, respectively; neurons in the cortex were immunolabeled for β-3 tubulin and NeuN, while the neuropil of the cortex was immunolabeled for β-3 tubulin and synaptophysin. Immunolabeling for nestin was only observed in the vascular walls. Immunolabeling for PDGFR-α was also observed in the vascular walls.
Using dual-immunolabeling, nIPCs that coexpressed NeuN- and β-3 tubulin were found in the SVZ (Supplemental Fig. S1a). OLIG2- and PDGFR-α-dual-immunolabeled OPCs were detected in the white matter (Supplemental Fig. S1b). Only a few NeuN-immunolabeled nIPCs in the SVZ expressed Ki-67 (Supplemental Fig. S1c).
Canine Glioma Cases
The histopathological diagnoses of the 58 cases were as follows: 4 LGOG (6.9%), 48 HGOG (82.8%), 1 LGAC (1.7%), and 5 HGAC (8.6%) (Supplemental Fig. S2a–e). Four HGAC cases (cases 55–58) were glioblastoma (Supplemental Fig. S8). Most HGOG cases were brachycephalic breeds (39/48, 81.3%: 31 French bulldogs, 7 Boston terriers, and 1 English bulldog). Case details are summarized in Supplemental Table S1.
Immunohistochemistry and Scoring Results of Canine Glioma Cases
Immunohistochemical scoring and statistical results are summarized in Table 1 and Supplemental Table 1. In all glioma cases, tumor cells had nuclear immunolabeling for OLIG2 (Fig. 4a, b). Oligodendrogliomas (average score, 2.9) and astrocytomas (average score, 2.7) both had high scores for OLIG2. In all oligodendroglioma cases, tumor cells expressed cytoplasmic and membranous PDGFR-α (Fig. 4c). The score of PDGFR-α was significantly higher in oligodendrogliomas (average score, 2.7) than in astrocytomas (average score, 0.7; P < 0.01). In all astrocytoma cases, tumor cells had cytoplasmic GFAP expression (Fig. 4d). The score of GFAP was significantly higher in astrocytomas (average score, 2.3) than in oligodendrogliomas (average score, 0.2; P < 0.01). β-3 tubulin was frequently expressed in high-grade gliomas (LGOG 1/4, 25%; HGOG 41/48, 85.4%; LGAC 0/1, 0%; and HGAC 5/5, 100%) (Fig. 4e, f), and was significantly higher in HGOG (average score, 1.5) than in LGOG (average score, 0.3; P < 0.01). Furthermore, high-grade gliomas had frequent nuclear immunolabeling for NeuN (LGOG 1/4, 25%; HGOG 44/48, 91.7%; LGAC 0/1, 0%; and HGAC 5/5, 100%) (Fig. 4g, h), which was significantly higher in HGOG (average score, 2.1) than in LGOG (average score, 0.5; P < 0.01). Some HGOG cases had variable cytoplasmic immunolabeling for synaptophysin (12/48, 25%; scores 0–3) (Fig. 4g), while LGOG lacked immunolabeling for synaptophysin (0/4). Two glioblastomas (cases 57 and 58) had minimal synaptophysin immunolabeling (HGAC 2/5; scores 0–1). Various degrees of cytoplasmic immunolabeling for nestin were observed in oligodendrogliomas and astrocytomas (Fig. 4j, k). The score of nestin was higher in astrocytomas (average score, 1.7; P < .01) than in oligodendroglioma (average score, 0.5). All glioblastoma cases (HGAC 4/5) had nestin scores of 2 or 3. Cytoplasmic CNPase expression was observed in approximately 50% of HGOG (25/48, 52.1%) (Fig. 4l), while LGOG and astrocytoma cases lacked immunolabeling for CNPase. The score of CNPase was low in HGOG (average score, 0.6), but tended to be higher in oligodendrogliomas (average score, 0.5; P = .02166) than in astrocytomas (average score, 0.0).
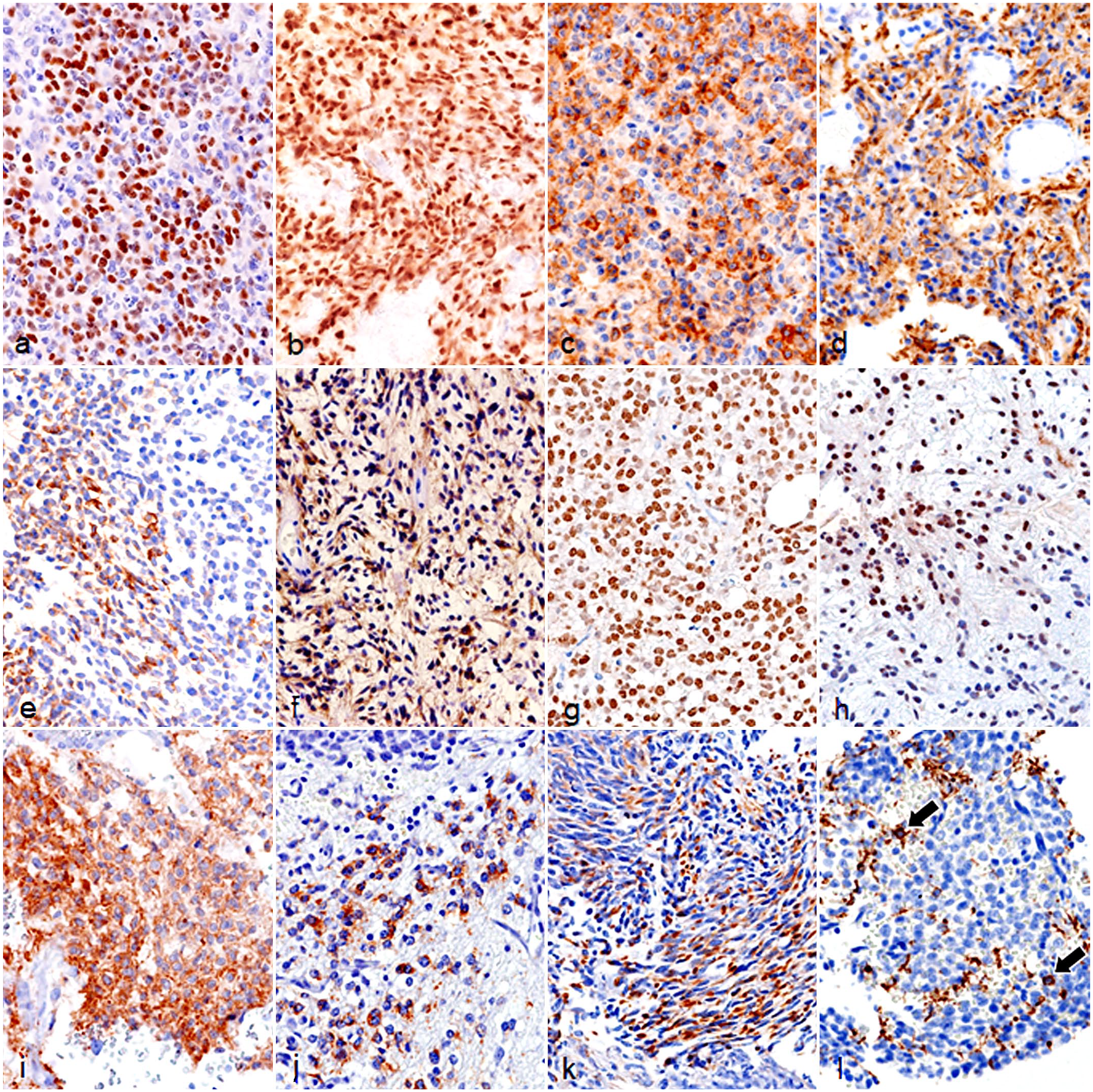
Glioma, brain, dog. Immunohistochemistry. (a
Ki-67 LI in Canine Glioma
The results for Ki-67 LI are summarized in Table 2. Average Ki-67 LI was higher in HGOG (13.03%; range: 4.14%–22.26%) than in LGOG (0%). Average Ki-67 LI was higher in HGAC (3.80%; range: 2.09%–9.49%) than in LGAC (2.58%). In HGAC, average Ki-67 LI was higher in glioblastoma cases (4.15%; range: 2.24%–9.49%) than in a non-glioblastoma case (case 54; 2.09%).
Regarding Ki-67 dual-labeling immunohistochemistry, immunolabeling for Ki-67 was observed in the PDGFR-α-, NeuN-, nestin-, and synaptophysin-immunolabeled cells of HGOG, but not β-3 tubulin-immunolabeled cells (Supplemental Fig. S3a–e). In HGOG, average Ki-67 LI were 10.39% in PDGFR-α- (range: 6.43%–14.10%), 15.93% in NeuN- (range: 5.24%–38.06%), 4.66% in nestin- (range: 0%–10.26%), 1.56% in synaptophysin- (range: 0.50%–2.67%), and 0% in β-3 tubulin-immunolabeled cells.
In astrocytomas, immunolabeling for Ki-67 was observed in GFAP-immunolabeled cells and nestin-immunolabeled cells (Supplemental Fig. S3f, g). Average Ki-67 LI in GFAP-immunolabeled cells and nestin-immunolabeled cells was higher in HGAC (GFAP 3.61%; range: 1.45%–8.08% and nestin 9.57%; range: 0%–14.00%) than in LGAC (GFAP 2.48% and nestin 5.62%). In HGAC, immunolabeling for Ki-67 was also observed in β-3 tubulin- (LI 2.71%; range: 0.00%–3.80%) and NeuN-immunolabeled (LI 9.70%; range: 3.05%–27.28%) cells (Supplemental Fig. S3h, i). In LGAC, β-3 tubulin- and NeuN-immunolabeled cells lacked immunolabeling for Ki-67. In HGAC, the average Ki-67 LI in dual-labeling immunohistochemistry was higher in glioblastoma cases (GFAP 4.30%; range: 1.45%–8.08%, β-3 tubulin 3.34%; range: 0.00%–3.80%, NeuN 11.09%; range: 4.54%–27.28%, and nestin 9.57%; range: 3.88%–14.00%) than in a non-glioblastoma case (case 54, GFAP 2.09%; β-3 tubulin 1.51%; NeuN 3.05%; and nestin 0%).
Morphology and Immunocytochemistry of AOFB-01 Cell Colonies
Colony-forming AOFB-01 cells were round to elongated in shape with a baso-eosinophilic cytoplasm (Fig. 5a). These cells showed a high N/C ratio and many mitotic figures. These features were similar to those of the original tumor tissue (case 9). Immunocytochemically, AOFB-01 cells had immunolabeling for Olig2 (score 3), PDGFR-α (score 3), β-3 tubulin (score 2), NeuN (score 3), synaptophysin (score 3), and nestin (score 1) (Fig. 5b–g and Table 1).
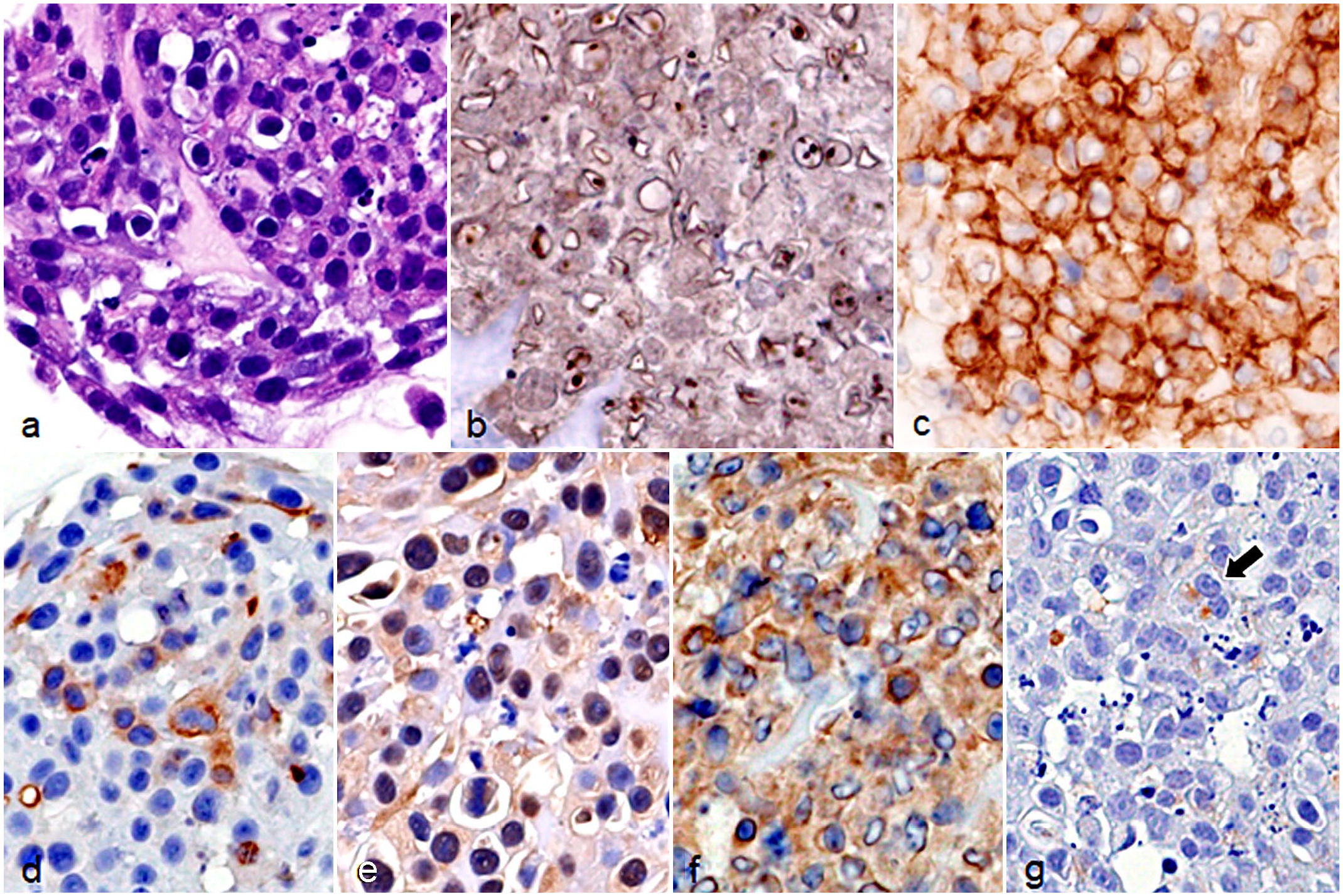
Cultured AOFB-01 cells. (a) Round to elongated cells with a high N/C ratio showing solid growth. Hematoxylin and eosin. (b) Nuclei are immunolabeled for OLIG2. Immunocytochemistry (ICC). (c) Cell membranes are immunolabeled for PDGFR-α. ICC. (d) The cytoplasm of some cells is immunolabeled for β-3 tubulin. ICC. (e) Nuclei are immunolabeled for NeuN. ICC. (f) The cytoplasm is immunolabeled for synaptophysin. ICC. (g) The cytoplasm of some cells is immunolabeled for nestin (arrow). ICC. Abbreviations: OLIG2, oligodendrocyte transcription factor 2; PDGFR-α, platelet-derived growth factor receptor-α; NeuN, neuronal nuclei.
Discussion
In this study, developing cells in the fetal canine brain were immunohistochemically characterized using markers for the NSC lineage (Fig. 6). Radial glial cells in the VZ differentiate from neuroepithelial progenitors and give rise to mature neurons or NSC, which re-enter the cell cycle as IPC. As previously described in the mice, 14 GFAP- and nestin-immunolabeled radial glial cells with elongated processes were detected in the VZ of the fetal canine brain. nIPC have been shown to proliferate in the SVZ of the fetal brain and directly generate neurons. 14 In the fetal canine brain, immunolabeled cells for the neuronal markers, β-3 tubulin, and NeuN in the SVZ were frequently immunolabeled for Ki-67, reflecting the features of nIPC. On the contrary, β-3 tubulin- and NeuN-immunolabeled cells in the CP lacked immunolabeling for Ki-67, and thus, were likely to be immature neurons. OLIG2 is an oligodendroglial lineage marker that is expressed during glial cell development.15,21 OLIG2- and PDGFR-α-immunolabeled cells in the SVZ and IZ of canine fetal brains were considered to be OPC. A few immature astrocytes that were immunolabeled for OLIG2 and GFAP were also observed.
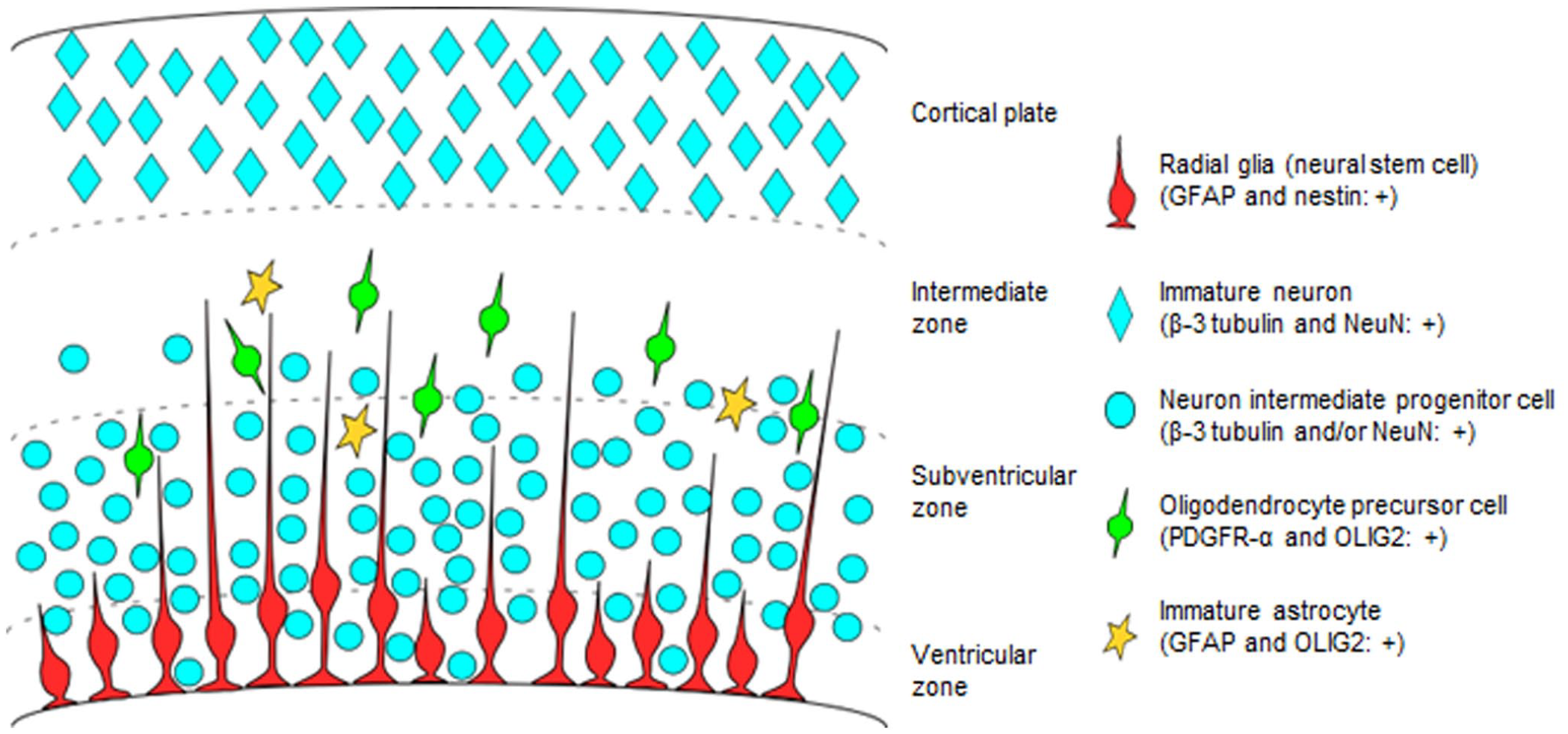
Drawing of a neural stem cell lineage in the ventricular zone, subventricular zone, intermediate zone, and cortical plate of canine fetal brain. Distribution and immunolabeling of the cells. Abbreviations: GFAP, glial fibrillary acidic protein; NeuN, neuronal nuclei; PDGFR-α, platelet-derived growth factor receptor-α; OLIG2, oligodendrocyte transcription factor 2.
Adult NSC are quiesccent, express GFAP and nestin, and produce IPC. 14 Experimental studies on rodents demonstrated that adult OPC accounted for approximately 6% of all CNS cells and were mainly distributed in the white matter. 15 In adult canine brains, OLIG2- and PDGFR-α-immunolabeled OPCs were found in the white matter. A small number of β-3 tubulin- and/or NeuN-immunolabeled nIPCs was detected in the SVZ of adult canine brains. The low immunolabeling for Ki-67 of NeuN-immunolabeled cells in the SVZ and lack of immunolabeling for Ki-67 in other cells may reflect the quiescent state of the adult NSC lineage in the adult canine brain.
Consistent with previous findings,11,13 canine oligodendrogliomas were highly immunolabeled for PDGFR-α and Olig2, while astrocytomas were highly immunolabeled for GFAP and Olig2. Immunolabeling for CNPase was low in oligodendrogliomas and astrocytomas. These immunohistochemical characteristics of oligodendroglioma and astrocytoma cells are similar to those of OPC and immature astrocytes, respectively, which were observed in fetal and adult canine brains, and may suggest their cells of origin. Canine oligodendrogliomas often develop adjacent to the ventricle and invade the cerebral white matter, which also supports the NSC lineage origin.22,27
Human gliomas exhibit tumor cell heterogeneity, which is influenced by genetics and the microenvironment. 24 Based on mRNA expression patterns, human glioma cells may be divided into several different populations, including OPC-like, astrocyte-like, oligodendrocyte-like, and neural-progenitor-like (nIPC-like) cells.24,30 Neural differentiation in human gliomas has been characterized by the expression of NeuN, synaptophysin, and neurofilament in tumor cells. 17 Moreover, the expression of β-3 tubulin and neuron-specific enolase was found to be up-regulated in higher grade gliomas.9,26 Immunolabeling for β-3 tubulin, neuron-specific enolase, microtubule-associated proteins 2, and synaptophysin has been reported in canine oligodendroglioma cases, suggesting the neural differentiation of tumor cells.1,7 In this study, HGOG immunohistochemical scores for β-3 tubulin and NeuN were significantly higher than those of LGOG, while the scores of HGAC were higher than those of LGAC. These results may suggest that canine gliomas consist of heterogeneous tumor cells showing the immunophenotypes of different stem cell lineages, and populations with neural lineage differentiation may be larger in high-grade gliomas.
Ki-67 LI was higher in high-grade gliomas than in low-grade gliomas, which is consistent with previous findings. 5 In this study, Ki-67 dual-labeling revealed high Ki-67 LI in the PDGFR-α- and NeuN-immunolabeled cells of HGOG, suggesting their OPC and nIPC natures, respectively. NeuN-immunolabeled cells in HGAC also had high Ki-67 LI. Moreover, high immunohistochemical scores for NeuN and PDGFR-α in HGOG and for NeuN and GFAP in HGAC may suggest that high-grade glioma cells have the features of both nIPC and OPC/immature astrocytes. In contrast, low Ki-67 LI in β-3 tubulin- and synaptophysin-immunolabeled cells may be associated with their differentiated neuron nature.
CSCs are a minor population within tumors that generate tumor cells through self-renewal and differentiation into multiple cell types, and contribute to persistent tumor proliferation with low proliferative activity. 16 Nestin has been suggested as a CSC marker of neurogenic tumors, including gliomas, in humans. 25 A subpopulation of nestin- and CD113-immunolabeled tumor cells was previously reported in canine gliomas.4,29 The present results showed that Ki-67 LI was low in the nestin-immunolabeled cell population in HGOG, which may represent the self-renewal potential of CSCs and maintenance of the stem cell pool. On the contrary, cases of glioblastoma showed higher immunohistochemical scores for nestin and higher average Ki-67 LI in nestin-immunolabeled tumor cells than other astrocytomas, which may suggest that tumor cells of glioblastoma show NSC nature rather than CSC nature and these NSC-like cells mainly proliferate in glioblastoma.
Colony-forming AOFB-01 cells showed similar immunohistochemical results to formalin-fixed paraffin-embedded sections of the original tumor (case 9), except for synaptophysin and nestin, which were not immunolabeled in the original tissue, but were expressed in vitro. Two explanations for these differences are as follows: CSC was selected during the cloning process, which resulted in the expression of a stem cell marker (ie, nestin) and a wider differentiation potential including the neuronal phenotype (ie, synaptophysin), and/or different microenvironments in vivo and in vitro are associated with differences in cell differentiation. In either case, the immunohistochemical results for the AOFB-01 colony suggest that HGOG cells may include a cellular population that has the potential to exhibit neural lineage differentiation (nIPC to neurons).
In this study, the immunophenotypes of NSC lineage cells were characterized in fetal and adult canine brains. Furthermore, Ki-67 LI was higher in the cell population showing the OPC and IPC immunophenotypes in glioma tissue, which may indicate their high proliferative activity and their possible cells of origin in the NSC lineage. In addition, the tumor cells of high-grade gliomas and the colony-forming glioma cell line showed nIPC and neuron characteristics, which may suggest that canine glioma cells include pluripotent stem cells that exhibit neuronal and glial differentiation. However, a limitation of this study is that the cellular markers used in this study label both pluripotent and mature cells. Further studies are needed to elucidate the relationship between molecular changes in the NSC lineage during tumorigenesis and the genetic background of brachycephalic breeds that are predisposed to gliomas. 10
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221136297 – Supplemental material for Immunohistochemical study of neural stem cell lineage markers in canine brains, gliomas, and a glioma cell line
Supplemental material, sj-pdf-1-vet-10.1177_03009858221136297 for Immunohistochemical study of neural stem cell lineage markers in canine brains, gliomas, and a glioma cell line by Kio Yoshida, James K. Chambers and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
