Abstract
Reports of canine ependymoma are generally restricted to single case reports with tumor incidence estimated at 2% to 3% of primary central nervous system (CNS) tumors. While most commonly reported in the lateral ventricle, tumors can occur anywhere in the ventricular system and in extraventricular locations. Rosettes and pseudorosettes are a common histologic feature; however, these features can be mimicked by other CNS neoplasms. Thirty-seven potential ependymoma cases were identified in a retrospective database search of 8 institutions, and a histologic review of all cases was conducted. Of 37 cases, 22 candidate cases were further subjected to a consensus histologic and immunohistochemical review, and only 5 of 37 (13.5%) were conclusively identified as ependymoma. The neuroanatomic locations were the lateral ventricle (3/5), third ventricle (1/5), and mesencephalic aqueduct (1/5). Subtypes were papillary (4/5) and tanycytic (1/5). Histologic features included rosettes (5/5), pseudorosettes (5/5), ependymal canals (2/5), tanycytic differentiation (1/5), blepharoplasts (1/5), ciliated cells (1/5), and high nuclear to cytoplasmic ratio (5/5). Immunolabeling for GFAP (4/4) and CKAE1/3 (3/4) was found in pseudorosettes, rosettes, and scattered individual neoplastic cells. Diffuse but variably intense cytoplasmic S100 immunolabeling was detected in 3 of 4 cases. Olig2 intranuclear immunolabeling was observed in less than 1% of the neoplastic cells (3/3). Tumors that had pseudorosettes and mimicked ependymoma included oligodendroglioma, choroid plexus tumor, pituitary corticotroph adenoma, papillary meningioma, and suprasellar germ cell tumor. These findings indicate that canine ependymoma is an extremely rare neoplasm with histomorphologic features that overlap with other primary CNS neoplasms.
Ependymal cells are specialized ciliated epithelial cells that line the ventricular system. These cells are present throughout the central nervous system and perform a number of functions, including interaction with the subventricular zone during development and regulation of permeability to cerebrospinal fluid components. 3 The ependyma consists of a single layer of cuboidal, ciliated cells that line the ventricles and a smaller population of specialized cells called tanycytes, which are most commonly located in the third ventricle and have long processes that interact with the adjacent neuroparenchyma. 3 In veterinary species, neoplasms with ependymal differentiation (ependymomas) are uncommon to rare, with the incidence highest in cats. 18 In dogs, the incidence of ependymoma is difficult to determine due to limited scientific literature; however, 1 retrospective series reported only 1 case of ependymoma in 435 cases of intracranial neoplasia. 12 Other reports suggest a prevalence of approximately 2% to 3% of all primary intracranial tumors in the dog. 15
Canine ependymoma is reported most frequently in the lateral and third ventricles and rarely in the spinal cord. 15 Regardless of location, tumors typically manifest grossly as a large, intraventricular mass that variably infiltrates and compresses the adjacent parenchyma. 9,14,15,17,19 Histologically, their appearance can be quite diverse, and subtypes include clear cell, papillary, and tanycytic. 9,14,15,17 Diagnosis is aided by the presence of perivascular pseudorosettes with antibasilar nuclei and cytoplasmic processes that abut blood vessels; however, this pattern is not unique to ependymomas and can be easily misinterpreted. Immunohistochemistry and transmission electron microscopy can aid in the diagnosis, the latter used to confirm the presence of desmosomes and cilia. Herein, we describe a case series of canine ependymomas that was derived from participation of multiple board-certified veterinary pathologists in a consensus review of cases previously diagnosed as ependymoma at their respective institutions. Given the challenges in accurately diagnosing ependymoma based solely on histologic appearance, we briefly review the neoplasms that were misdiagnosed as ependymoma and provide diagnostic support for the alternate microscopic interpretations.
Materials and Methods
Biopsy and necropsy records for the New York State Animal Health Diagnostic Center, Cornell University College of Veterinary Medicine (1979–2015); University of Georgia Department of Pathology and Athens Veterinary Diagnostic Laboratory (2000–2015); Auburn University College of Veterinary Medicine, Department of Pathobiology (1996–2016); The Animal Medical Center, Department of Anatomic Pathology (1999–2015); Animal Health Trust (1990–2015); Texas A&M University College of Veterinary Medicine & Biomedical Sciences, Department of Veterinary Pathobiology (2002–2015); and The Royal Veterinary College, Department of Pathobiology and Population Sciences (1997–2017) were searched for cases of canine ependymoma. Additional select cases were provided from the Armed Forces Institute of Pathology archives by 1 coauthor (F.Y.S.). All cases that fit inclusion criteria (ie, coded in the respective institutional computer system as ependymoma) were reviewed by all coauthors, and from this initial review, cases that were clearly not ependymoma based on inconsistent histologic features were removed from consideration. Consensus was based on at least 75% agreement among the reviewers. The following immunohistochemical stains were used: glial fibrillary acidic protein (GFAP; rabbit polyclonal, catalog #Z0334; Agilent, Carpinteria, CA), cytokeratin AE1/AE3 (CKAE1/3; mouse monoclonal, catalog #MS351501-2; Agilent), oligodendrocyte transcription factor 2 (Olig2; rabbit monoclonal, catalog #AB109186; Abcam, Cambridge, MA), S100 (rabbit polyclonal, catalog #IS50430-2; Agilent), and adrenocorticotropic hormone (ACTH; mouse monoclonal, catalog #M350101-2; Agilent). The stains were reviewed by the coauthors before coming to a consensus diagnosis. Phosphotungstic acid–hematoxylin (PTAH) histochemical staining and ACTH immunohistochemistry were performed only on select cases based on the histologic features, whereas GFAP, CKAE1/AE3, Olig2, and S100 were performed on all cases in which tissue was available.
PTAH staining was performed according to standard laboratory protocols. The following protocols were used for immunohistochemistry using the Leica Bond Max automated immunohistochemistry staining system, according to the manufacturer’s instructions (Leica Microsystems, Buffalo Grove, IL). Briefly, tissues were sectioned at 5 μm and deparaffinized with Bond Dewax Solution (catalog #AR9222; Leica Microsystems). Pretreatment with heat-induced antigen retrieval was performed for 30 minutes using Tris/EDTA pH 9 (Bond Epitope Retrieval Solution 2, catalog #AR9640; Leica Microsystems) for CKAE1/3 and Olig2. No pretreatment was used for GFAP, S100, and ACTH. Endogenous peroxidase activity was blocked with a 3% peroxide solution for 5 minutes (catalog #DS9800; Leica Microsystems). The antibodies were diluted at 1:500 for 15 minutes (GFAP), 1:200 for 60 minutes (CKAE1/3 and Olig2), 1:400 for 14 minutes (S100), and 1:200 for 30 minutes (ACTH). Biotin-free PowerVision poly-polymeric horseradish peroxidase antimouse (catalog #PV6114; Leica Microsystems) or antirabbit (catalog #PV6119; Leica Microsystems) IgG reagent was then applied to the slides for 30 minutes followed by incubation with Bond Polymer Refine Detection for 10 minutes (catalog #DS9800; Leica Microsystems). Tissues were developed with 3,3′-diaminobenzidine (DAB) (catalog #DS9800; Leica Microsystems) for 10 minutes. The slides were counterstained with hematoxylin (catalog #DS9800; Leica Microsystems) for 5 minutes, dehydrated, cleared, and mounted. Positive controls consisted of normal canine brain (GFAP, Olig2, and S100), small intestine (CKAE1/3), and pituitary gland (ACTH). When normal brain tissue was present, internal positive staining was noted in all cases for GFAP, Olig2, and S100. Negative controls consisted of an isotype-matched antibody.
Results
Thirty-seven cases were originally identified by computer search, and after initial histologic review, 22 cases (60%) had histologic features potentially consistent with ependymoma and therefore were included for further analysis. Of the 15 cases that were not subject to further review, diagnoses included oligodendroglioma (6/15), pituitary adenoma (3/15), astrocytoma (3/15), nephroblastoma (1/15), meningioma (1/15), and nondiagnostic (1/15). Of the 22 cases reviewed for consensus diagnosis, only 5 of 22 (22.7%) were diagnosed as ependymoma based on the combination of histologic, histochemical, and immunohistochemical features. Breed, sex, age, tumor location, and tumor subtype are described in Table 1.
Signalment and Pathologic Features in 6 Cases of Canine Ependymoma.
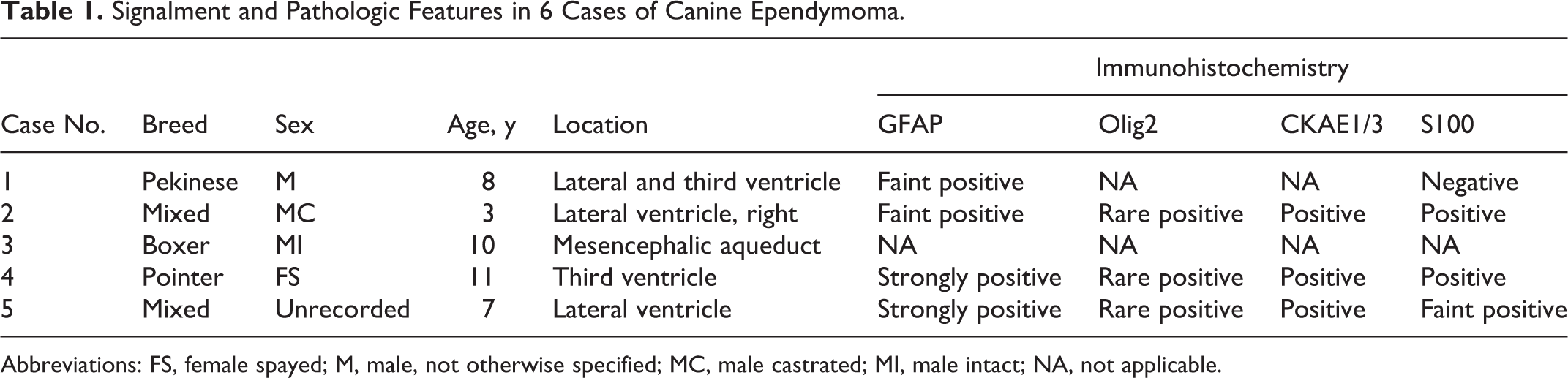
Abbreviations: FS, female spayed; M, male, not otherwise specified; MC, male castrated; MI, male intact; NA, not applicable.
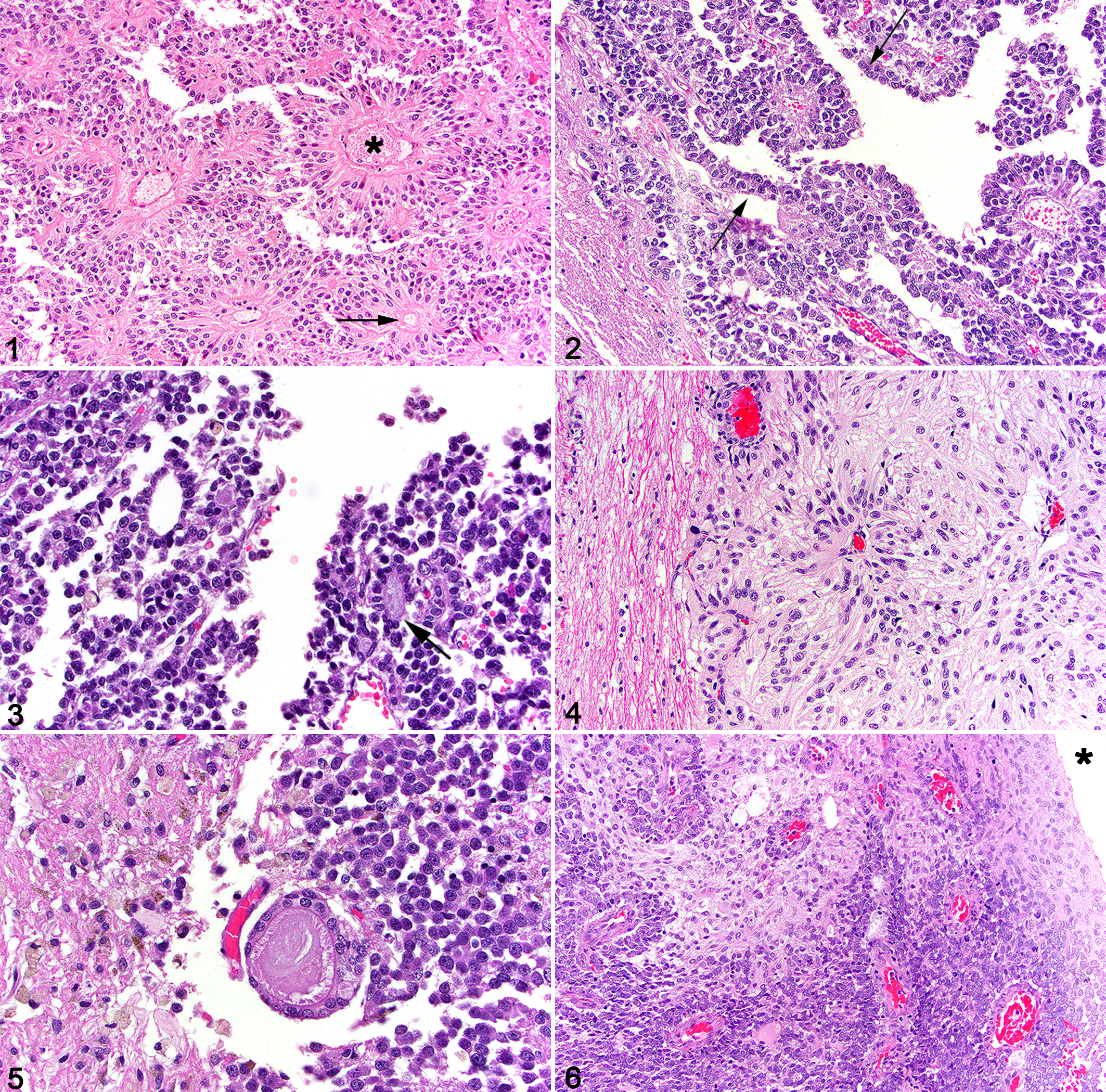
Four of the cases were classified as papillary ependymoma and 1 as a tanycytic ependymoma. Papillary ependymomas were characterized by cords, ribbons, and papilliform projections of neoplastic ependymal cells that had moderate amounts of cytoplasm and round to ovoid nuclei with a dense chromatin pattern (Fig. 1). All ependymomas had rosettes (5/5) and pseudorosettes (5/5) (Figs. 2,3). Rosettes often contained intraluminal, pale basophilic material (Fig. 3). The single case of ependymoma with tanycytic differentiation had elongated cells with flattened, spindled nuclei (Fig. 4). Fewer cases had ependymal canals (2/5), blepharoplasts (1/5), and ciliated cells (1/5) (Fig. 5). One case had clear progression from a thickened, dysplastic ependyma (Fig. 6). A high nuclear to cytoplasmic ratio (5/5) and areas of necrosis (3/5) were features in a majority of cases. Hemorrhage, microvascular proliferation, and areas of calcification were absent in all cases. Atypia was moderate in all cases, and the mitotic rate was less than 1 per 400× field (2.37 mm2) in all cases.
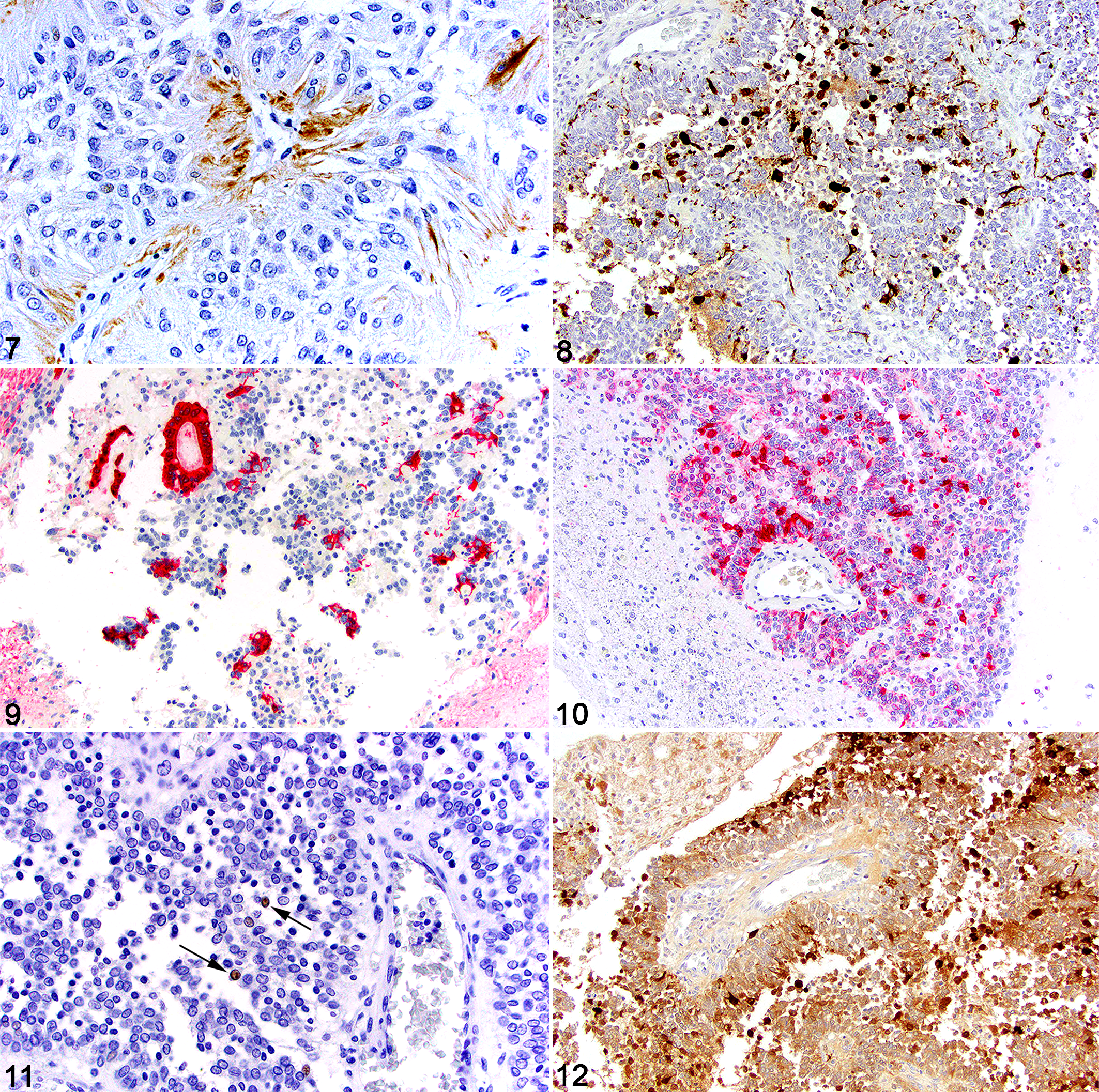
Multifocal cytoplasmic staining for PTAH was present in the 1 case in which this stain was used. Strong cytoplasmic immunolabeling for GFAP (4/4) was found in pseudorosettes (Fig. 7), rosettes, and in up to 60% of the neoplastic cells not forming pseudorosettes or rosettes (Fig. 8). Strong cytoplasmic immunolabeling to CKAE1/3 (3/4) generally mirrored the GFAP staining and was found in pseudorosettes, rosettes (Fig. 9), and scattered individual neoplastic cells (Fig. 10). The staining intensity for CKAE1/3 was greater in the rosettes than the GFAP immunolabeling. Olig2 intranuclear immunolabeling was seen in less than 1% of the neoplastic cells (3/3) (Fig. 11). Diffuse, but variable, cytoplasmic S100 immunolabeling was detected in 3 of 4 cases (Fig. 12).
Of the remaining potential 17 cases, the following diagnoses were made with the addition of immunohistochemical stains coupled with the histologic features: oligodendroglioma (6/17), choroid plexus tumor (5/17), pituitary corticotroph adenoma (4/17), meningioma (1/17), and suprasellar germ cell tumor (1/17). All cases of oligodendroglioma were either primary intraventricular neoplasms or periventricular with extension into the ventricle. Oligodendrogliomas were characterized by sheets of round to ovoid neoplastic cells with a variable amount of cytoplasm, round or sometimes molded nuclei, and a variable chromatin pattern. All cases had a significant number of pseudorosettes (Fig. 13), and diffuse intranuclear Olig2 immunolabeling was present in nearly 100% of the neoplastic cells (Fig. 13, inset). Neoplastic oligodendrocytes lacked immunolabeling for GFAP and CKAE1/3 and had variable cytoplasmic immunolabeling for S100.
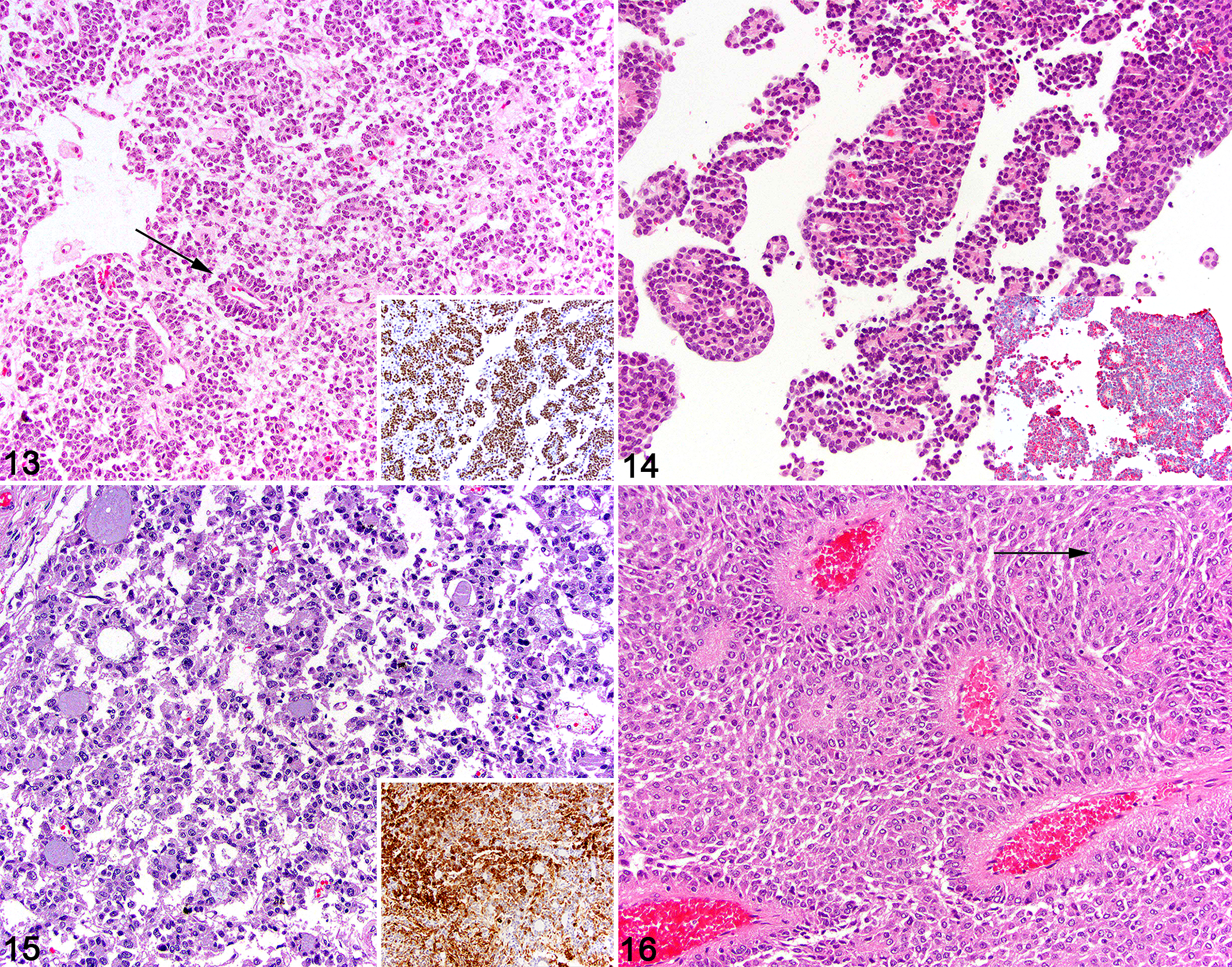
All cases diagnosed as choroid plexus tumors were primary intraventricular masses composed of dense cords, interlacing tubular structures, and pseudorosettes of neoplastic epithelial cells (Fig. 14). Typical papillary and villiform projections common in most canine choroid plexus tumors were lacking in many of the tumors that were included in this study. CKAE1/3 was diffusely positive in all neoplastic cells of the choroid plexus tumors with strong intracytoplasmic immunolabeling (Fig. 14, inset). Choroid plexus tumors lacked immunolabeling for GFAP, Olig2, and S100.
The pituitary tumors were entirely restricted to the third ventricle, bulging into and compressing the thalamus. These tumors were composed of tubules, rosettes, pseudorosettes, and loose clusters of medium to large polygonal epithelial cells with abundant basophilic to amphophilic cytoplasm separated by fine fibrovascular septae (Fig. 15). Cells with abundant eosinophilic cytoplasm were absent. All cases had rosettes containing basophilic material, mimicking the pattern seen in some ependymomas; however, no ciliated cells were present. All cases had robust and diffuse intracytoplasmic immunolabeling for ACTH (Fig. 15, inset). CKAE1/3 immunolabeling in the pituitary tumors was robust and nearly diffuse, and patchy to diffuse cytoplasmic S100 immunolabeling was present. Pituitary tumors lacked immunolabeling for GFAP and Olig2.
For the meningioma case, characteristic meningothelial whorls were present; however, a majority of the neoplasm was composed of prominent pseudorosettes of meningothelial cells typical for the papillary subtype of meningioma (Fig. 16). Last, the germinoma case consisted of a multiphasic neoplasm composed of germ-like cells admixed with a neuroparenchyma-like stroma. While rare pseudorosettes were noted in the germinoma, the histology was distinct from that seen in ependymomas. Immunohistochemistry was not deemed necessary to diagnose the meningioma or germinoma case.
Discussion
Ependymomas are an uncommon to rare glioma in the dog that are most frequently reported in the lateral and third ventricles. 15 While the cell of origin in the dog is not known, studies of human ependymoma reveal features of radial glia, a neural stem cell that resides in the subventricular zone. 4 Ependymomas are typically well-circumscribed intraventricular neoplasms that do not extend extensively into the adjacent parenchyma. Pseudorosettes and rosettes are an important histologic feature of these tumors; however, they are not specific to ependymomas, and using them as the sole criterion for ependymoma diagnosis is ill advised, supported by the findings in this study. Important histologic features that are specific to ependymomas include ependymal canals (linear spaces lined by neoplastic ependymal cells), ciliated epithelial cells lining the rosettes, and blepharoplasts (basal ciliary bodies identified by PTAH histochemical staining). 7 As demonstrated here, the diagnosis can be greatly aided by immunohistochemistry, which typically relies on GFAP immunolabeling in pseudorosettes and other parts of the tumor; multifocal pan-cytokeratin immunolabeling in pseudorosettes, rosettes, and other tumor cells; and sparse Olig2 immunolabeling. Human ependymomas commonly express vimentin and S100 and rarely express neuronal antigens. 5,16 While the underlying molecular abnormalities associated with ependymoma in humans are well described and include mutational activation of MAPK, PDGFR, and VEGF, the rarity of these tumors in the dog has limited their molecular characterization. 1,13
The current case series details the collective efforts and consensus review of 8 board-certified veterinary pathologists, all of whom have extensive experience and expertise with diagnostic veterinary neuropathology. Of the 37 cases originally selected from archival searches, only 5 were confirmed as ependymoma based on histology with or without immunohistochemical features. Based on the small number of cases, no breed or sex predilection can be determined, and the tumors were most common in middle-aged dogs. Three cases involved the lateral ventricle, consistent with the predilection sites recorded in the literature. While pseudorosettes and rosettes were present in all cases, other more specific features of ependymoma (ependymal canals, ciliated cells, and blepharoplasts) were present in a minority of cases. Four of the cases were diagnosed as papillary ependymomas, whereas 1 had tanycytic features. Most reported cases of canine ependymoma are papillary subtypes, with rare reports of clear cell and anaplastic variants. 9,14,17,19
The inclusion of immunohistochemistry can greatly aid in the diagnosis of ependymoma; however, it must be interpreted in context with the expected staining patterns of other intracranial neoplasms in the dog. In the current cases series, GFAP was immunoreactive in all of the ependymoma cases in which tissue was available. Immunolabeling was strong in the cells forming the pseudorosettes and rosettes, as well as in single to small clusters of neoplastic cells throughout the neoplasm. While GFAP is traditionally used to label cells of astrocytic origin and therefore used in the diagnosis of astrocytomas, it is also expressed in normal ependyma and radial glial cells, making its expression another supporting feature used for the diagnosis of ependymoma. 6,19 In these canine ependymomas, GFAP was robustly expressed in pseudorosettes and, to a lesser degree, in rosettes. A similar pattern of immunolabeling was noted for pan-cytokeratin, although immunolabeling was often more robust in the rosettes compared to GFAP. Cytokeratin expression occurs in human and feline ependymomas, but it can be widely variable in intensity, ranging from scant immunolabeling to marked immunolabeling noted in the pseudorosettes and rosettes. 16,18 Importantly, Olig2 immunolabeling was extremely limited in the canine ependymomas in this study. This is consistent with the staining pattern of Olig2 in human ependymomas, in which a minor subset of neoplastic cells has expression of Olig2. 4,5 Since this pattern of Olig2 expression is expected with radial glia, it is often used to support the contention that these tumors arise from a subpopulation of radial glia stem cells. 4,5 S100 immunolabeling was present in 3 of 4 of the cases, and while certainly not specific to ependymoma, the multifocal, intense cytoplasmic staining pattern was consistent with that reported in human ependymoma. 16
The presence of pseudorosettes and rosettes can lead to a diagnostic dilemma, as these are features that can be recapitulated in a number of primary intracranial neoplasms in the dog. In the current consensus series, previously diagnosed ependymomas were reclassified as oligodendroglioma, choroid plexus tumor, pituitary adenoma, meningioma, and suprasellar germinoma.
In the dog, gliomas are the second most common primary intracranial tumor after meningioma, with oligodendrogliomas having an incidence of up to 70%. 8 Oligodendrogliomas have relatively uniform cellularity and are composed of round to polygonal cells with round to ovoid nuclei, finely stippled chromatin, and inapparent nucleoli. The presence of pseudorosettes can be sporadic or abundant in these tumors and can lead to diagnostic confusion when the tumor is located within a ventricle. 8,11 Oligodendrogliomas can have extensive ventricular involvement. The presence of the characteristic “fried-egg” autolytic histologic pattern, the “chicken-wire” vascular pattern, myxoid stroma, nuclear rowing or molding, and coarse nuclear chromatin in oligodendrogliomas can be used to help distinguish them from ependymomas. Importantly, Olig2 immunolabeling is typically present in the majority of neoplastic cells in oligodendrogliomas, in contrast to the pattern observed in ependymomas. 8
Choroid plexus tumors represent approximately 10% of primary intracranial tumors in the dog. 15 They arise from the choroid plexus epithelium, occurring predominately in the lateral ventricles and fourth ventricles with occasional spread to other regions of the ventricular system. Most canine choroid plexus tumors are formed by papillary projections of choroid plexus epithelium supported by a fine fibrovascular stroma, but other patterns include tubule-like differentiation, acinus-like structures, and sheet-like growth of cuboidal epithelial cells. The small acinus-like structures have a central fibrovascular core and mimic a pseudorosette. Unlike ependymomas, these tumors are diffusely immunolabeled with pan-cytokeratin and lack immunolabeling for Olig2 and GFAP. Other more specific antibodies to the choroid plexus, including Kir7.1, E-cadherin, and N-cadherin, can be used to further establish the diagnosis of choroid plexus tumors in the dog. 2,10
Pituitary gland tumors can invade dorsally to involve the third ventricle and the surrounding brain parenchyma. Corticotroph tumors are relatively common in the dog and can either be functional or nonfunctional. Pituitary tumors that include rosettes and pseudorosettes can easily be confused with ependymomas, but while the histologic pattern may be similar in some respects, the immunohistochemical staining patterns are vastly different. Pituitary tumors have abundant immunolabeling for pan-cytokeratin and ACTH and are not expected to have immunolabeling for GFAP and Olig2.
Meningiomas, due to their neuroectodermal lineage, can produce a wide variety of histologic patterns, but meningothelial whorls are a defining feature, even when rarely present within the neoplasm. In addition, the nuclei of meningiomas are large with an open chromatin pattern, which is distinct from other primary intracranial neoplasms. The papillary subtype of meningioma is the most problematic, as it presents with abundant pseudorosettes that can mimic those seen in ependymomas. However, the location of the tumor (extra-axial), as well as the presence of meningothelial whorls, aids in the diagnosis. There are no immunohistochemical stains that are definitive for canine meningioma, but meningiomas lack immunolabeling for Olig2 and can have patchy immunolabeling for pan-cytokeratin. 6
Last, germinomas arising in the suprasellar region can invade the third ventricle and mimic ependymoma. These tumors have a distinct histomorphology composed of clusters, sheets, and islands of germ cells, often with epithelial and hepatocyte-like differentiation. Suprasellar germ cell tumors strongly express α-fetoprotein; however, there have only been limited immunohistochemical characterization of these tumors in the dog.
In conclusion, ependymomas are rare tumors in the dog. While pseudorosettes and rosettes are characteristic features of ependymomas, they are also present in other intracranial neoplasms. The tumor location (intraventricular), other histologic features (ependymal canals, ciliated epithelial cells lining the rosettes, and blepharoplasts), histochemical stains (PTAH), and immunohistochemistry (GFAP, pan-cytokeratin, Olig2) can help differentiate ependymomas from other primary brain tumors.
Footnotes
Acknowledgements
We thank the histopathology technicians at the Cornell University College of Veterinary Medicine, New York State Animal Health Diagnostic Center for assistance with case processing and Jen Patterson for assistance with images. We also thank Dr. Brad Njaa for submitting case No. 5 for review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
