Abstract
Primary central nervous system (CNS) neoplasms are uncommonly diagnosed in cats. The majority of primary feline CNS neoplasms described in the veterinary literature consist of meningioma and glioma occurring mainly in the brain and less often in the spinal cord. Although most neoplasms can be diagnosed based on routine histologic evaluation, less typical tumors need to be further characterized using immunohistochemistry. This review compiles the relevant information about the most common primary CNS neoplasms of cats available in the veterinary literature, aiming to serve as a converging source of information for the topic.
In veterinary medicine, most primary central nervous system (CNS) neoplasms are diagnosed in dogs and cats. 36 Although numbers vary across institutions, the incidence rate of primary CNS neoplasia in cats is estimated to be around 3.5 per 100 000 cases. 1 In autopsied cats, nearly 2% of all neoplasms are intracranial, with 70% consisting of primary brain tumors.113,120 Primary spinal cord tumors are less common and consist of approximately 16% of all spinal cord and vertebral neoplasms.4,66,67 However, these numbers may not reflect the true frequency of spinal cord tumors since spinal cord examination is not part of a routine autopsy in most institutions. Regardless of the affected neuroanatomic site (brain or spinal cord), the most commonly diagnosed primary CNS neoplasms of cats are meningioma and glioma (astrocytoma, oligodendroglioma, and less often ependymoma),32,91,119 followed by other less common to rare tumor types, such as choroid plexus neoplasms, olfactory neuroblastoma, embryonal tumors, and round cell tumors (particularly lymphoma and histiocytic sarcoma).90,113
The diagnosis, classification, and grading of CNS neoplasms in domestic animals relies on their neuroanatomic localization, histology, and immunohistochemistry (IHC), and is heavily based on the Histological Classification of Tumors of the Nervous System of Domestic Animals and the human World Health Organization (WHO) Classification of Tumors of the Central Nervous System (Table 1).47,57,58 Although revised guidelines for classification and grading of canine glioma and meningioma are available, further investigations are needed to correlate tumor type and grade with tumor behavior and clinical outcome, allowing for subsequent modifications or validation of these species-specific guidelines.5,46 No such comprehensive clinical studies have been conducted for canine and feline CNS neoplasms since most affected patients are euthanized following a diagnosis of a brain or spinal cord neoplasm. In addition, starting in its 2016 edition, the WHO classification system has adopted an integrated diagnostic approach that uses tumor morphology and IHC as the basis for diagnosis, but relies on molecular diagnostic tools to provide a more objective tumor diagnosis that is not currently available for companion animals.58,118 Efforts to understand the molecular mechanisms involved in CNS tumorigenesis and tumor progression is an evolving topic in veterinary neuro-oncology and neuropathology and may help in the development of similar tools to diagnose these neoplasms in the future. 53
Morphologic features and tumor grading for the main primary central nervous system neoplasms of cats. a

Abbreviations: CNS, central nervous system; IHC, immunohistochemistry; NA, not applicable.
Modified from the Histological Classification of Tumors of the Nervous System of Domestic Animals 47 and the World Health Organization (WHO) Classification of Tumors of the Central Nervous System.57,58
Tumor subtypes reported in cats.
A WHO grade 1 meningioma should be classified as atypical (WHO grade 2) if the necessary criteria are fulfilled.
No specific cut-off point for mitoses has been described for gliomas in cats.
Extrapolated from the human classification system, not validated in cats.
No specific cut-off point for mitoses has been described for ependymomas in cats.
This review compiles the most relevant information about commonly diagnosed primary CNS neoplasms of cats available in the veterinary literature and aims to serve as a comprehensive and converging source of information for the topic.
Meningioma
General Features
Feline meningioma is the most common primary CNS neoplasm of cats and has been described since the early 1960s.10,25–27,34,41,42,52,60,67,69,75,84,93,100,112,113,120 Meningiomas account for nearly 60% of all intracranial and 85% of primary brain neoplasms of cats. 113 Spinal cord meningiomas are less frequently reported and account for about 7% of all spinal and vertebral neoplasms.66,67 Meningioma is a slow-growing neoplasm that arises from arachnoid cells that line the meningeal arachnoid villi. 74 Arachnoid cells share a mesenchymal and neural crest origin, which results in the wide morphologic spectrum and IHC profile characteristic of meningiomas. 36 The common occurrence of meningiomas at sites with a higher density of arachnoid villi supports their association with these structures. 115
Meningioma affects mainly adult to older cats (3–18 years).9,75,86,112,113 Although a slight predominance of male cats has been occasionally reported,2,27,75 no evidence for a sex predisposition has been confirmed in large population studies.9,74,113 Similarly, domestic shorthaired cats were the predominant breed in some studies, 9 but that is likely a reflection of the large number of domestic shorthaired cats in the overall feline population and not a true breed predisposition. 113 Meningiomas have been reported in a group of 4 young cats affected by mucopolysaccharidosis type 1, but a relationship, if any, between tumor development and the metabolic disorder remains unproven; 33 all cats were less than 3-years-old and had multiple, clinically silent meningiomas. 33
Clinical Signs and Clinical Diagnosis
The slow growth of meningiomas leads to insidious and progressive clinical signs that can be directly associated with the neurolocalization of the neoplasm or with the secondary effects of the tumor mass (peritumoral edema and cerebellar herniation).27,75,85 Clinical signs in most cases consist of altered consciousness, seizures, ataxia, impaired vision, circling, vestibular deficits, and paresis.9,27,74,113 The median duration of the neurologic signs until diagnosis is approximately 1 month.9,27,113 Nearly 20% of meningiomas in cats are clinically silent, and 18% are reported as an incidental finding during autopsy.36,113
A clinical diagnosis can be achieved in most cases by the detection of an extraparenchymal tumor by computed tomography (CT) or magnetic resonance imaging (MRI).9,74,112 Although the diagnostic accuracy of CT is not well established for feline meningioma, MRI has been shown to correctly identify intracranial meningiomas in 96% of cases. 112 Tumors typically have distinct and sharp margins, are isointense or hypointense and homogeneous on T1-weighted images, hyperintense and heterogeneous on T2-weighted images, and have moderate to marked post-contrast enhancement (Fig. 1a). 112 Some meningiomas have a dural tail sign, ring enhancement, and peritumoral edema, but these are nonspecific features and can be observed in other CNS lesions. 112 After a meningioma is clinically suspected, diagnostic confirmation relies on histologic evaluation of biopsy or autopsy samples. 40
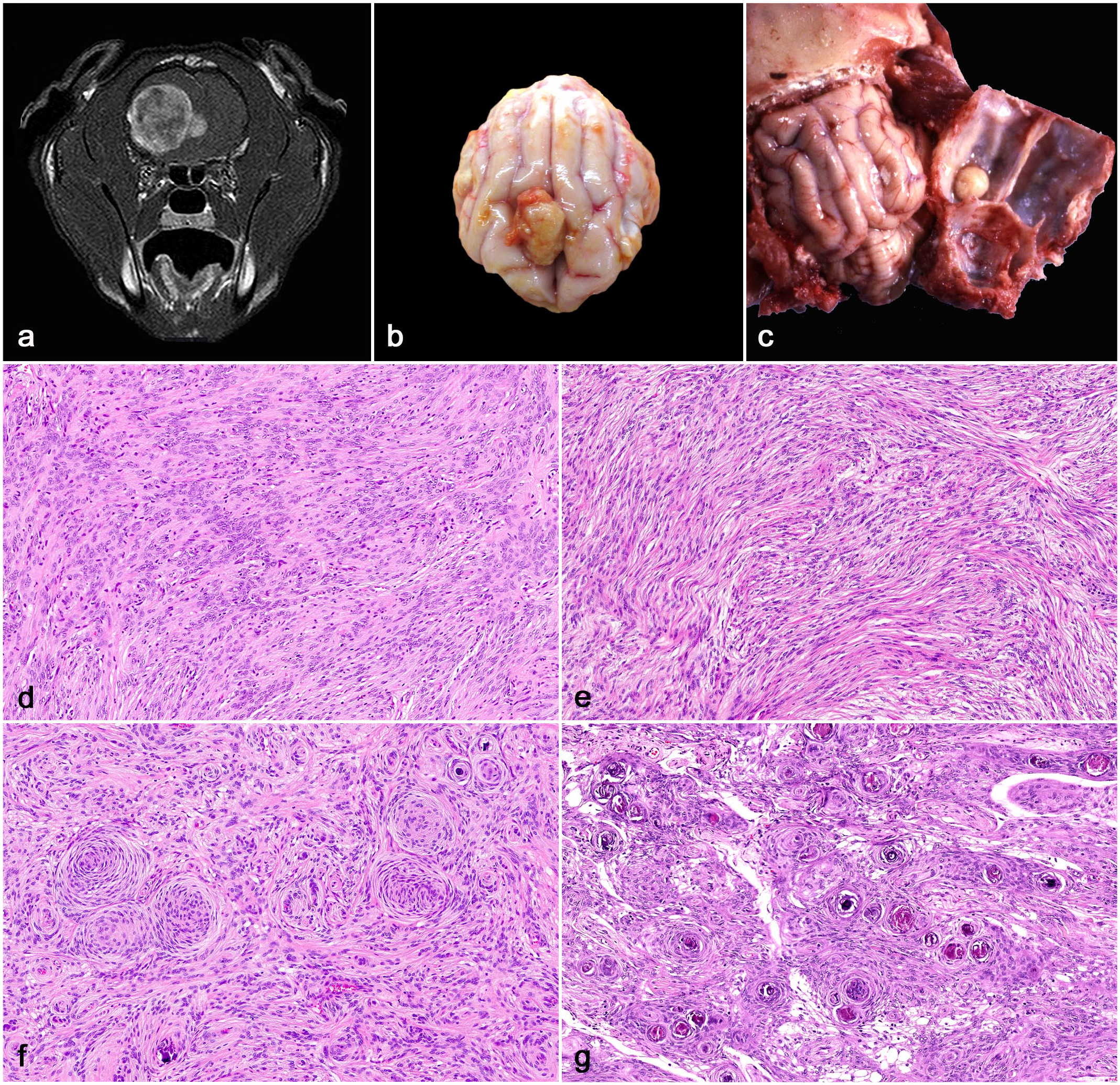
Meningioma, brain, cat. (a) Magnetic resonance imaging, T1-weighted post-contrast image with fat saturation in transverse plane. At the level of the right parietal lobe, there is a well-demarcated mass with broad dural contact that compresses and laterally displaces the right telencephalic hemisphere. (b) A well-demarcated, pale-yellow extraparenchymal meningioma compresses the right and to a lesser extend the left frontal telencephalic lobe. (c) A well-demarcated, pale-yellow meningioma is attached to the dura mater overlying the right occipital telencephalic lobe. Tumor compression has led to a focal depression in the neuroparenchyma. (d) A meningothelial meningioma consisting of solid sheets and lobules of neoplastic cells with eosinophilic cytoplasm (hematoxylin and eosin, HE). (e) A fibrous meningioma with the characteristic interweaving bundles and fascicles of spindle cells associated with a variable amount of collagen stroma (HE). (f) A transitional meningioma with scattered concentric whorls of meningothelial cells admixed with spindle cells (HE). (g) Psammomatous meningioma with the concentric whorls of meningothelial cells containing numerous central mineralized structures (HE).
Pathology
Grossly, meningiomas are typically well-demarcated, white or pale yellow, firm extraparenchymal tumors that typically exhibit no invasion into the adjacent neuroparenchyma (Fig. 1b).42,84,113 The dura mater underlying the skull bones should always be inspected for meningiomas after the skull cap is removed during autopsy (Fig. 1c). Ignoring the inner skull examination may lead to a failure in the diagnosis as many tumors are attached to the dura. Compression by the tumor often leads to atrophy of the underlying neuroparenchyma. Nearly 75% of meningiomas are supratentorial,
9
with only a small number (approximately 10% of tumors) occurring at infratentorial locations.9,21,25,27,75,86,109 Supratentorial neoplasms occur mainly in the parietal lobe, temporal lobe, frontal lobe, occipital lobe, diencephalon, and cerebral falx.75,109,113 Infratentorial neoplasms mainly affect the cerebellum.42,84,113 Intraventricular meningiomas arising from the
The most prevalent meningiomas in cats are WHO grade 1 transitional,2,9,10,21,23,26,31,43,50,62,63,65,68,82,86,94,95 fibrous,2,9,21,22,30,50,62,65,68,86,94,95 psammomatous,2,9,21,45,50,62,65,68,86,95 and meningothelial subtypes.9,23,28,33,56,62 Angiomatous (WHO grade 1), 86 papillary (WHO grade 3), 42 and anaplastic (WHO grade 3) meningiomas9,62,65,95 are rarely reported. Other WHO grade 1 meningiomas (microcystic and secretory) and WHO grade 2 meningiomas (chordoid and clear cell) have not been yet described in cats. Histologically, neoplastic meningothelial cells (meningothelial meningioma) are typically arranged in closely apposed lobules supported by a fine fibrovascular stroma and have abundant, polygonal, eosinophilic cytoplasm with indistinct cell margins (Fig. 1d). Nuclei are round to oval and have finely stippled chromatin. Neoplastic meningothelial cells occasionally form concentric whorls that surround mineral concretions (psammoma bodies). Fibrous meningiomas consist of spindle neoplastic cells arranged in interweaving bundles and fascicles supported by a fine collagen stroma (Fig. 1e). Neoplastic cells have elongated eosinophilic cytoplasm and oval nuclei with finely stippled chromatin and indistinct nucleoli. Meningothelial and fibrous meningiomas consist of a dominant population of meningothelial or spindle neoplastic cells, respectively, whereas transitional meningiomas (Fig. 1f) are characterized by a combination of both neoplastic cell populations. Psammomatous meningiomas consist of meningothelial and spindle neoplastic cells with widespread concentric whorls surrounding psammoma bodies (Fig. 1g). A diagnosis of psammomatous meningioma should apply to meningiomas consisting of widespread whorls of neoplastic cells surrounding psammoma bodies throughout the neoplasm. Occasional psammoma bodies (as observed in many transitional meningiomas) or areas of mineralization (which are distinct form psammoma bodies) do not qualify for such a diagnosis.36,94 Angiomatous meningiomas are characterized by nests of neoplastic meningothelial cells intercalated with numerous dilated blood vessels. 86 WHO grade 1 meningiomas have low-to-moderate cellularity, low cell and nuclear pleomorphism, and less than 4 mitoses in 2.37 mm2 (10 FN22/40× fields).36,47 A WHO grade 1 meningioma with 4 or more mitoses in 2.37 mm2, CNS invasion, or at least 3 atypical features (hypercellularity, neoplastic cell sheeting, small cell formation, macronucleoli, and geographic necrosis) should be classified as a WHO grade 2 atypical meningioma.36,47 WHO grade 3 papillary meningiomas consist of neoplastic meningothelial cells arranged in widespread papillary projections supported by cores of fibrovascular tissue. 42 According to the available grading guidelines, WHO grade 3 meningiomas (papillary, rhabdoid, and anaplastic) are highly cellular and pleomorphic neoplasms with 20 or more mitoses in 2.37 mm2.9,62,65,95 Anaplastic meningiomas have been rarely reported in cats, with no mention of other WHO grade 3 subtypes in cats in the veterinary literature. 108 It is common for meningiomas classified as one particular morphologic type to have areas with distinct morphology. In such cases, the tumor should be diagnosed based on the dominant morphologic features.
The classification and grading guidelines for meningiomas in veterinary medicine are adapted from human medicine.5,36 However, the clinical significance of these grading schemes has not been evaluated in any animal species other than humans. The Histological Classification of Tumors of the Nervous System of Domestic Animals was published in 1999 and classifies meningiomas as slow-growing or anaplastic tumors. 47 However, these guidelines are outdated. For instance, granular cell meningioma, previously classified as a slow-growing meningioma, 47 is no longer considered a meningioma subtype (see “Granular Cell Tumors” section). 36 Papillary meningioma, previously classified as a slow-growing meningioma, 47 is currently classified as a WHO grade 3 tumor, together with anaplastic meningioma. 36 For these reasons, further investigations should be conducted to evaluate whether the veterinary CNS tumor classification and grading systems should be updated (as has the human WHO grading system) and standardized to species-specific neoplasms.
Diagnosis
Morphologic features typically suffice for a routine histologic diagnosis of meningioma, but IHC may be necessary in less differentiated neoplasms or less common subtypes of meningioma. 86 Multiple immunomarkers, including vimentin, cytokeratin, glial fibrillary acidic protein (GFAP), claudin-1, glucose transporter 1, laminin, progesterone receptor, protein gene product 9.5, E-cadherin, and S100, have been evaluated in feline meningiomas, with variable positive immunolabeling among tumors.86,94 An IHC panel consisting of concurrent immunolabeling for vimentin, CD34, and E-cadherin has been proposed as supportive of a diagnosis of feline meningioma, but results may vary among tumors. 86 In a study evaluating 45 feline meningiomas, nearly 92% of neoplasms exhibited immunolabeling for E-cadherin, suggesting this is a useful immunomarker for the diagnosis of meningiomas in cats. 94 However, the relative low number of IHC studies on feline meningiomas still warrants further research to assess and validate a diagnostic IHC panel for the diagnosis and prognosis of these neoplasms.
Immunohistochemical evaluation of the tumor microenvironment in a group of feline meningiomas revealed that all tumors had a moderate number of CD3+ lymphocytes and Iba1+ histiocytic cells and that nearly half of the cases had Pax5+ B lymphocytes. 68 The significance of these findings remains unknown, but the characterization and understanding of the immunologic cell population within the tumor microenvironment may be useful in future CNS tumor studies focusing on immunotherapy.
Biologic Behavior, Treatment, and Survival
Proliferative activity and other potential prognostic parameters have been assessed by a few studies, but the factors that may have a role in the oncogenesis and biologic behavior of feline meningiomas remain largely unknown. 68 The prognostic value of Ki67 immunolabeling has not been well established in feline meningioma, 68 but no significant differences in Ki67 immunolabeling has been detected among distinct tumor types and grades in cats. 94 Although limited data indicate that feline meningiomas with a dense intratumoral CD3+ lymphocytic population have lower Ki67 immunolabeling, further research is needed to elucidate the relationship between immune cell infiltration and tumor proliferation. 68 No associations between nuclear immunolabeling with β-catenin and N-cadherin and anaplastic meningiomas have been demonstrated in cats.37,94
In humans, meningiomas are more than twice as likely to affect adult females than males. 106 Radiation exposure, advanced age, genetic predisposition, and the effects of sex hormones have all been associated with a higher incidence of meningiomas in females. 106 In veterinary medicine, gonadal steroid receptors were first demonstrated in feline meningiomas in the early 1990s, 104 but no associations between immunolabeling and tumor morphology and grade have been confirmed to date.2,63 In one study, progesterone receptor immunolabeling was inversely associated with proliferative activity expressed by Ki67 immunolabeling, 63 suggesting that progesterone receptor expression could be used to assess the biologic behavior of feline meningiomas and that anti-progesterone therapy could be used in cases in which surgery is not a viable option. 63 However, the low number of assessed cases requires further research to determine the clinical significance and effect of these sex hormone receptors in tumor behavior and therapy.2,63
Associations between cyclooxygenase-2 (COX-2) immunolabeling with cell proliferation and tumor grade have been reported in human meningioma. 55 In cats, COX-2 immunolabeling has been associated with tumor necrosis, but no differences were observed between COX-2 immunolabeling and tumor grade. 95 Matrix metalloproteinase-2 and 9 (MMP-2 and MMP-9) expression predicts meningioma recurrence independent of its proliferative index in humans. 78 In veterinary medicine, MMP-2 and MMP-9 immunolabeling in feline meningiomas did not correlate with meningioma type or proliferative activity. 62 Expression of telomerase reverse transcriptase (TERT) has been evaluated as a potential prognostic tool in the assessment of feline meningiomas, but no associations among TERT expression and tumor subtype and grade were found. 65
Most meningiomas in cats do not infiltrate the neuroparenchyma and have a benign clinical behavior.2,67,109,113 Metastases are exceedingly rare and have been reported in the lungs of one cat. 13 Consequently, the most common treatment for feline meningiomas is surgical resection combined with radiation therapy, chemotherapy, and palliative care.9,23,27,49,50,113 Surgical excision is achievable in most cases,21,23,25,27,50,77,113 but the procedure can be challenging in more infiltrative tumors or in tumors involving areas of difficult access, especially the skull base. 2 Tumor excision has been associated with a longer survival time after the diagnosis when compared with other treatment modalities.9,113 In one study assessing 34 patients, the median survival time for cats that were not treated with surgery was 18 days compared with 685 days for cats that had the tumors surgically resected. 113 Other investigations reported postsurgical median survival times of 485 days (4 cats), 77 27 months (11 cats), 25 612 days (7 cats), 50 and approximately 4 years (42 cats). 27 Clinical complications (such as seizures, nystagmus, head tilt, and visual deficits) during the postoperative period have been reported in nearly 20% of the cases.25,27,77 Most patients do not experience tumor recurrence, with overall survival rates of nearly 70% at 6 months post-surgery and 50% at 2 years post-surgery.25,27 The age of the patient and the neurolocalization of the neoplasm does not affect the survival time. 27 Local tumor recurrence has been recorded in up to 30% of cats in a period of 3 to 69 days after surgery.21,25,27,77,113 Recurrence at distant CNS sites can also occur, and it remains unknown whether these represent de novo meningiomas or spread of the original tumor. 27 Radiation therapy is a viable treatment for cats with inoperable meningiomas or for those with high peri- or post-surgical risks. 49
Glioma
General Features
Glioma is the second most common primary CNS neoplasm of cats,36,91,112,113 accounting for about 8% of all intracranial neoplasms, 12% of all primary brain neoplasms, and 8% of all spinal and vertebral neoplasms.66,67,113 Although their cell of origin remains undetermined, current evidence suggests that gliomas likely arise from subependymal stem cells or immature glial cells, which have the potential to differentiate into cells with glial and neuronal phenotypes. 36 In cats, the most common type of glioma is astrocytoma,3,12,16,18,32,48,67,89,96–98,105,108,112,113,120 followed by ependymoma,6,24,35,39,71,102,111,113,119,120 oligodendroglioma,17,32,67,103,107,112,113,116 and other rare glioma subtypes, such as unclassified glioma, diffusely infiltrating gliomas (gliomatosis cerebri),12,32,67 and leptomeningeal gliomatosis. 121 However, the overall prevalence of different types of glioma would likely vary depending on different classification systems utilized at the time of diagnosis and on the availability of specific IHC antibodies for diagnostic confirmation of each neoplasm. 91 Gliomas affect mainly adult to older cats (8–12 years), and no sex or breed predispositions have been reported to date.16,32,67,91,113,119 Domestic shorthaired cats are overrepresented in most studies, but that is likely a reflection of the frequency of this breed in pet cat populations.91,113
Gliomas are typically intraparenchymal neoplasms, with rare reports of leptomeningeal gliomatosis, in which there is extensive neoplastic cell infiltration in the leptomeninges. 121 Astrocytomas and oligodendrogliomas occur mainly in the telencephalon,16,18,48,89,91,96–98,107,112,113,116 followed by spinal cord,3,16,32,35,67,91,105,108 brainstem,12,16,17,91,113 and cerebellum.103,113 Intraventricular astrocytomas and oligodendrogliomas have been rarely reported.17,113 Spinal cord gliomas occur most frequently in the cervical segment, but the reason for this anatomic predilection remains undetermined.32,67
Ependymomas arise from radial glial cells, are typically intraventricular, and occur mainly in the lateral ventricles,6,111,119 third ventricle,6,102,119 and fourth ventricle.39,71 Spinal cord ependymomas and extraventricular encephalic ependymomas are rarely reported. 119
Clinical Signs and Clinical Diagnosis
Given that the majority of gliomas occur in the telencephalon, clinical signs are typically characterized by seizures, depression, altered consciousness, and circling91,97 and tend to be progressive and have been reported to occur from 1 to 90 days prior to diagnosis. 113 In addition, tumors affecting the brainstem or cerebellum can cause vestibular signs.91,103 Spinal cord gliomas lead to neurologic signs that occur from 2 to 12 weeks prior to diagnosis and are characterized mainly by paraparesis, paraplegia, and pain associated with the spinal cord lesion.32,67 A clinical diagnosis can be suspected using MRI, which reveals an intraparenchymal mass that is typically hypointense on T1-weighted images and hypointense to hyperintense on T2-weighted images, with variable post-contrast enhancement (Fig. 2a, b).32,112,113 In addition, tumors can have ring enhancement and peritumoral edema, but these are nonspecific findings that can be associated with other neoplastic and nonneoplastic masses in the CNS.36,112
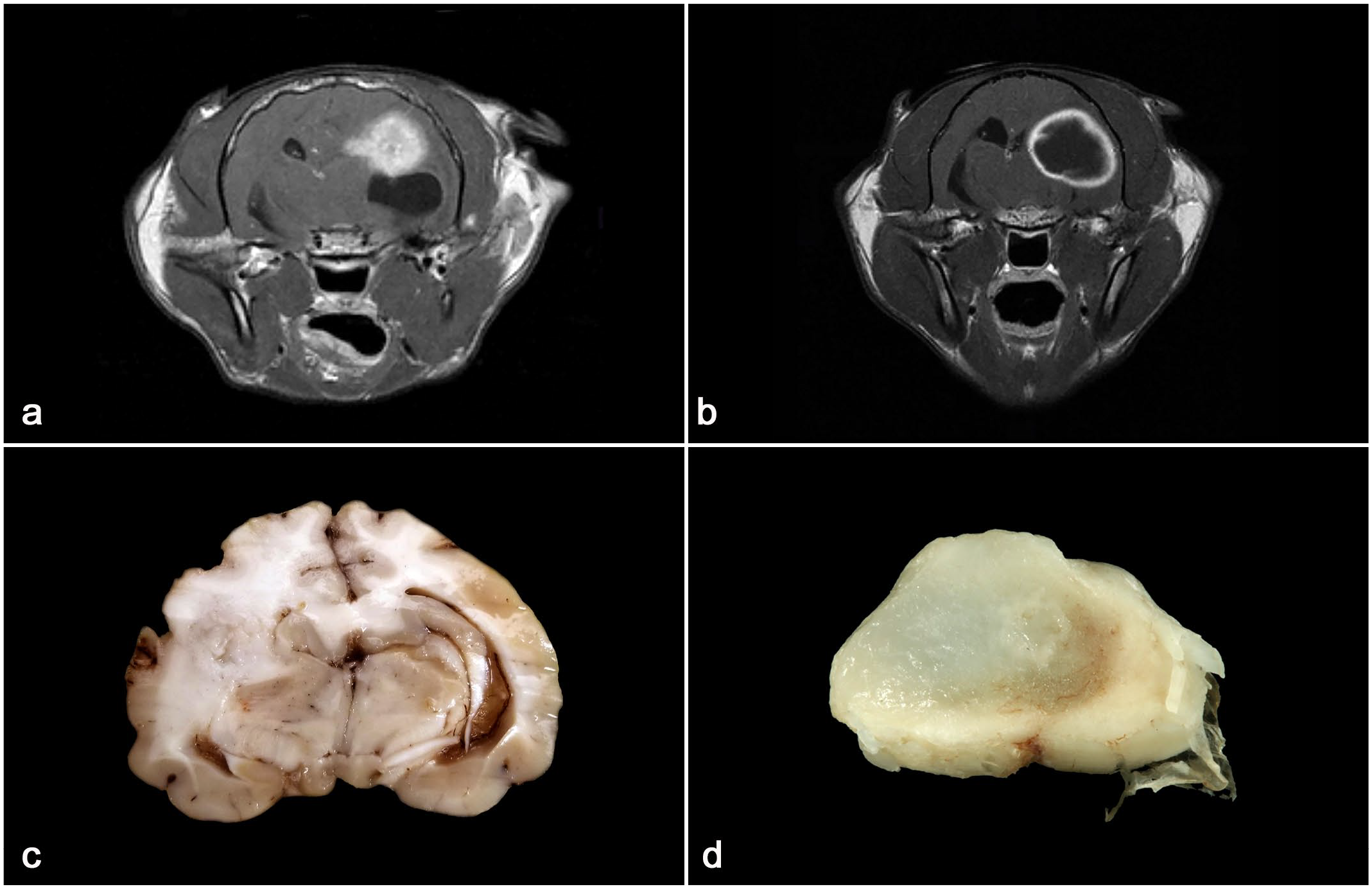
Glioma, brain and spinal cord, cat. (a) Magnetic resonance imaging, T1-weighted post-contrast image in transverse plane at the level of the thalamus. There is a poorly demarcated, strongly enhancing intraparenchymal mass (astrocytoma) in the left telencephalic hemisphere and a fluid collection (left lateral hydrocephalus) ventral to the mass. (b) Magnetic resonance imaging, T1-weighted post-contrast image with fat saturation in transverse plane at the level of the thalamus. There is a well-demarcated intraparenchymal mass (oligodendroglioma) with a hypointense, non-enhancing core and strongly enhancing rim in the left telencephalic hemisphere. (c) A poorly demarcated, pale white, intraparenchymal astrocytoma expands and effaces the subcortical white matter (right parietal lobe) and dorsal thalamus. There is compression of the lateral ventricle and midline shift to the left secondary to the tumor. (d) A well-demarcated, pale-white to light-brown oligodendroglioma expands and effaces most of the spinal cord neuroparenchyma.
Pathology
Grossly, gliomas are poorly or well-demarcated, white to pale yellow to gray, soft neoplasms that can exhibit dark red areas of hemorrhage. There may be variations among cases, but astrocytomas tend to be pale white to tan and poorly demarcated (Fig. 2c),18,48,96–98 whereas oligodendrogliomas are typically soft, gelatinous, and well demarcated (Fig. 2d).17,91,103,116
As is the case for meningioma, the histologic classification and grading of feline gliomas is mostly based on the veterinary guidelines and on the WHO classification of human CNS tumors.47,58 Histologically, low-grade astrocytomas (WHO grades 1 and 2) are characterized by low-to-moderate cellularity and low-to-moderate cellular and nuclear pleomorphism (Fig. 3a). Neoplastic cells are arranged in sheets or streams embedded in an eosinophilic, neuroparenchyma-like stroma and have elongate eosinophilic cytoplasm. Nuclei are round to elongate and have dense to finely stippled chromatin with 1 to 2 nucleoli. The rare subependymal giant cell astrocytoma, although a WHO grade 1 glioma, consists of pleomorphic, elongate to epithelioid neoplastic cells in a fibrovascular stroma. 18 Anaplastic astrocytoma (WHO grade 3) and glioblastoma (WHO grade 4) are highly cellular and pleomorphic tumors consisting of sheets or bundles of round or elongate neoplastic cells that have variable-sized, irregular, small-to-large nuclei and frequent mitoses (Fig. 3b, c). A diagnosis of glioblastoma is dependent on the presence of neoplastic cell palisading around geographic areas of necrosis (Fig. 3d) and/or microvascular proliferation (Fig. 3e).16,36,91,97,98,115
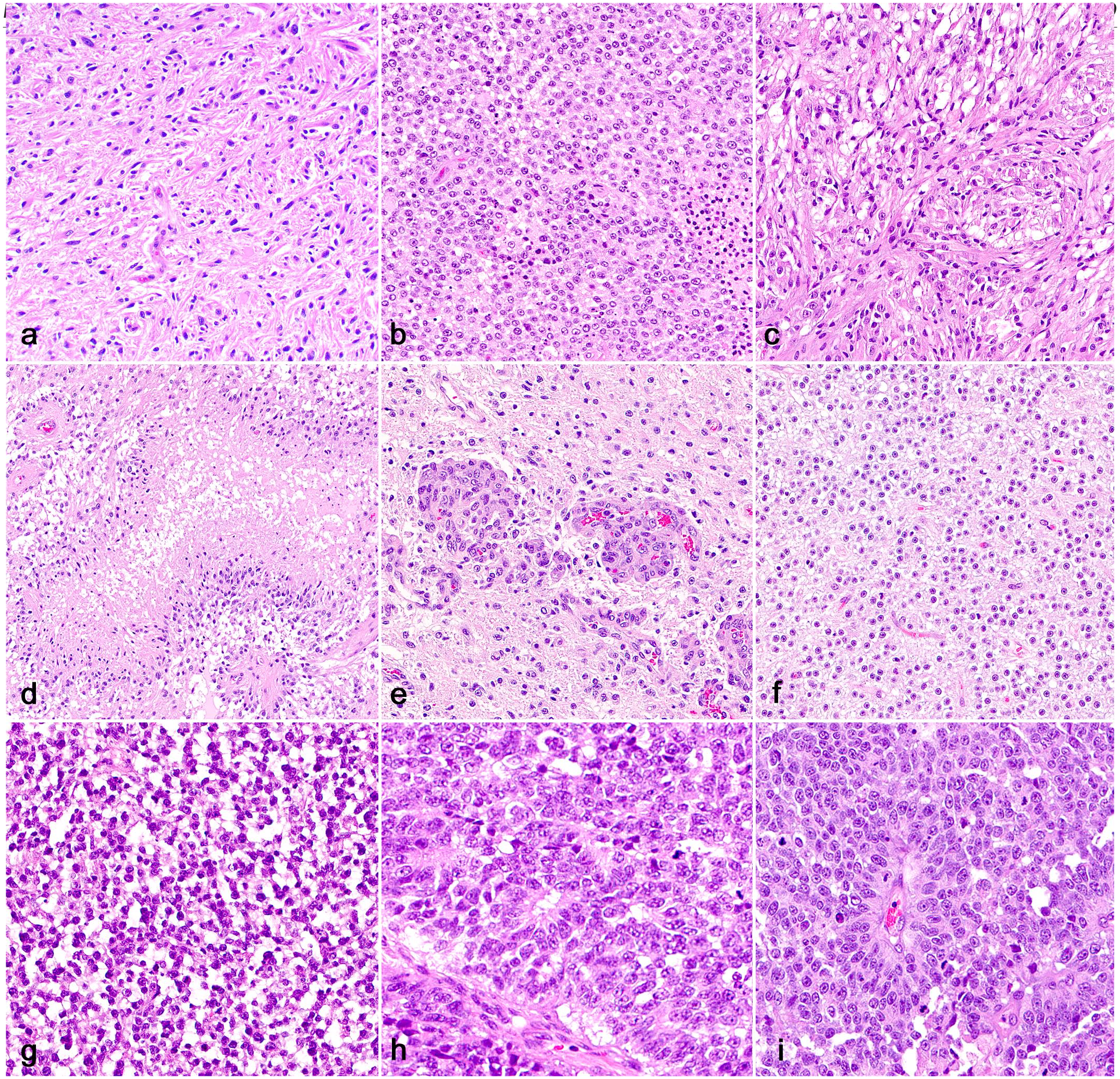
Glioma, brain, cat. (a) Low-grade astrocytoma characterized by neoplastic cells with elongate nuclei arranged in sheets or streams embedded in an eosinophilic, neuropil-like stroma (hematoxylin and eosin, HE). (b) Highly cellular anaplastic astrocytoma with prominent cellular and nuclear pleomorphism (HE). (c) Glioblastoma with increased cellular and nuclear pleomorphism (HE). (d) Glioblastoma with the characteristic neoplastic cell palisading around extensive geographic necrosis (HE). (e) Areas of microvascular proliferation in a glioblastoma (HE). (f) Low-grade oligodendroglioma characterized by low-to-moderate cellularity and low cellular and nuclear atypia. Neoplastic cells are embedded on a fine mucinous stroma and have round, eosinophilic, or clear cytoplasm that creates a perinuclear halo (HE). (g) Anaplastic oligodendrogliomas are densely cellular and have increased cellular and nuclear pleomorphism (HE). (h) Neoplastic cells form true rosettes (center) supported by a fine fibrovascular stroma in a feline ependymoma (HE). (i) Neoplastic cells palisade around capillaries (pseudorosettes) in a feline ependymoma (HE).
Low-grade oligodendrogliomas (WHO grade 2) have low-to-moderate cellularity and low cellular and nuclear atypia (Fig. 3f). Neoplastic cells are typically embedded on a fine fibrovascular or mucinous stroma and have round, eosinophilic, or clear cytoplasm that creates a perinuclear halo (fried egg or honeycomb feature). Nuclei are round and have dense chromatin with 1 to 2 nucleoli. Mitoses are absent or rarely observed, and rare areas of necrosis can be present. Anaplastic (WHO grade 3) oligodendrogliomas are densely cellular gliomas (Fig. 3g) with marked cellular and nuclear pleomorphism, high mitotic activity (6 or more mitoses in 2.37 mm2), and extensive areas of geographic necrosis and/or microvascular proliferation.36,91,115 However, this mitotic cut-off point is based on the human CNS classification system and has not been standardized for oligodendrogliomas or any other gliomas in cats.
Morphologic features of ependymomas can vary considerably among tumors and within the same tumor. 119 The most common ependymoma subtype reported in cats is classic ependymoma (WHO grade 2),15,119 with rare reports of papillary and tanycytic ependymoma (WHO grade 2),15,119 subependymoma (WHO grade 1), 119 and anaplastic ependymoma (WHO grade 3). 91 No information about the relationship between ependymoma subtype and prognosis is available. Classic ependymomas are moderately cellular, with low-to-moderate cellular and nuclear pleomorphism. Neoplastic cells typically palisade around capillaries (pseudorosettes) or can form true rosettes or ependymal canals and are supported by a fine fibrovascular stroma (Fig. 3h).15,119 In papillary ependymomas, neoplastic cells arrange in papillary projections with occasional rosettes and pseudorosettes. 119 Neoplastic cells are round to polygonal and have eosinophilic cytoplasm. Nuclei are round to oval and have finely stippled chromatin with 1 to 3 nucleoli. 119 In tanycytic ependymoma, neoplastic cells are arranged in bundles with elongate processes. 119 Subependymomas attach to a ventricular wall and are characterized by clusters of neoplastic glial cells embedded in a fibrillary stroma. 119 One feline anaplastic ependymoma has been reported. 91 The neoplasm was highly cellular and consisted of polygonal neoplastic cells randomly organized in sheets or pseudorosettes (Fig. 3i), with high mitotic activity (18 mitoses in 2.37 mm2) and necrosis.
Diagnosis
The diagnosis of glioma relies on histologic evaluation of biopsy or autopsy samples.40,91 Although most neoplasms can be diagnosed based on routine histology, less typical gliomas may require IHC for a final diagnosis.40,91 For diagnostic confirmation, astrocytomas have variable immunolabeling for GFAP (Fig. 4a) and oligodendrocyte transcription factor 2 (OLIG2). In contrast, oligodendrogliomas have robust immunolabeling for OLIG2 (Fig. 4b) and variable to no immunolabeling for GFAP.32,40,91 Presumed gliomas in the brain or spinal cord of cats with no immunolabeling for OLIG2 should be differentiated from a round cell tumor, especially lymphoma. 91 Such tumors should be subjected to IHC for lymphocytic markers for further characterization and diagnostic confirmation. 91 Synaptophysin and neuron-specific enolase immunolabeling have been reported in feline oligodendrogliomas and likely indicate, as in dogs and humans, that these tumors may share a common progenitor cell origin with other CNS neoplasms and exhibit neuronal features.83,88,91 Immunolabeling for doublecortin, an immunomarker for neuronal progenitor cells, has also been detected in feline gliomas, which supports this hypothesis.19,91 Doublecortin immunolabeling is reportedly more predominant in astrocytomas, which are more invasive and have stronger immunolabeling at the tumor margins, similar to what has been reported for invasive human gliomas and anaplastic canine meningiomas. 19
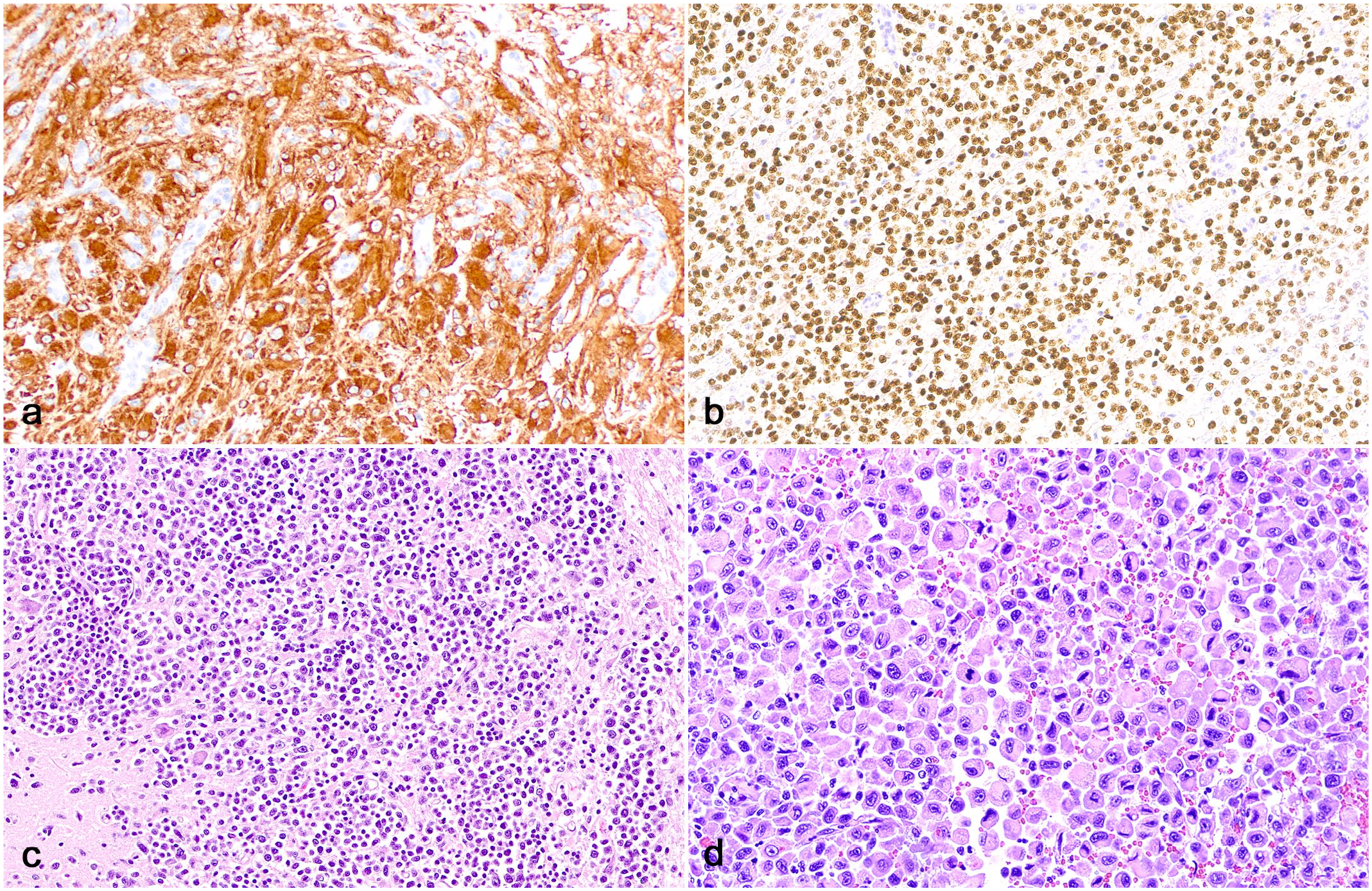
Glioma and other central nervous system neoplasms, brain, cat. (a) Astrocytomas have robust cytoplasmic immunolabeling for glial fibrillary acidic protein (GFAP). (b) Oligodendrogliomas have robust nuclear immunolabeling for oligodendrocyte transcription factor 2 (OLIG2). (c) An intraparenchymal lymphoma expands and partially effaces the cerebellar peduncle (hematoxylin and eosin, HE). (d) A histiocytic sarcoma effaces the brainstem (HE).
Feline ependymomas have variable immunolabeling for GFAP and pancytokeratin, which is typically predominant in the cells forming pseudorosettes, and widespread immunolabeling for OLIG2. 15
The assessment of the immune cell population within feline glioma has revealed comparable results to the meningioma microenvironment, with intratumoral and peritumoral CD3+ lymphocytes primarily within perivascular spaces and rarely around areas of necrosis or leptomeningeal spread, fewer perivascular CD20+ lymphocytes, and a large number of Iba1+ histiocytic cells that were distributed particularly around areas of necrosis and leptomeningeal spread. 92
Biologic Behavior, Treatment, and Survival
Information about the biologic behavior and treatment of feline glioma is not as abundant as for meningioma. 49 Although increased Ki67 immunolabeling was reported in high-grade feline gliomas, suggesting Ki67 could be useful to support a diagnosis of more anaplastic glial tumors, further research using a larger number of cases is needed to corroborate or refute these results. 91
As opposed to feline meningiomas, which are typically well-demarcated and extraparenchymal neoplasms, gliomas are intraparenchymal and can be infiltrative, which makes surgical excision more challenging. Surgical debulking with radiation therapy and palliative corticosteroid therapy have been attempted in a small number of feline gliomas, with post-surgical survival times varying from 1 to 179 days. 113 Surgical resection of an ependymoma with complete clinical recovery has been reported. 102 However, given the low number of investigations assessing these parameters, conclusions from most therapeutic studies evaluating feline glioma are limited and should be interpreted with caution.
Other Neoplasms
Neoplasms described in this section are rare in cats and have been published as single case reports or small case series. For this reason, not enough information is available to determine potential sex and breed predispositions or particular pathologic features (neuroanatomic localization) of different tumor types.
Choroid Plexus Neoplasms
Choroid plexus neoplasms are exceedingly rare in cats, with one choroid plexus cystadenoma reported in the left ventricle and one choroid plexus oncocytoma in the cerebellopontine angle.11,113 Neoplasms arise from the choroid plexus epithelium and thus appear as intraventricular tumors. It remains to be determined whether choroid plexus tumors in cats exhibit similar histologic and IHC features to those described in dogs and other animal species. 14
Embryonal Neoplasms
Embryonal neoplasms are currently classified as medulloblastomas or embryonal tumors of difficult diagnostic characterization (also known as primitive neuroectodermal tumors).58,70 In cats, there have been descriptions of a medulloblastoma44,51 and multiple olfactory neuroblastomas.7,80,99,113 Medulloblastoma occurs in young individuals and is typically characterized as white mass that affects the cerebellum and dorsal brainstem. Histologically, tumors are densely cellular and consist of small round cells arranged in sheets or forming rosettes. Neoplastic cells have eosinophil cytoplasm and dense, round to elongate nuclei.44,51 Immunohistochemistry for neuron-specific enolase and GFAP was demonstrated in the feline medulloblastoma. 44 Olfactory neuroblastoma (esthesioneuroblastoma) arises from the olfactory neuroepithelium and thus occurs within the caudal nasal cavity (cribriform plate) and olfactory bulbs.7,80,99,113 In a published case series of 9 cats, 7 tumors affected adult cats (3–10 years) of both sexes and led to upper respiratory signs, such as rhinitis and hemorrhagic nasal discharge, and aggressive behavior. Olfactory neuroblastomas are highly cellular and composed of uniform, round to oval neoplastic cells arranged in lobules supported by a fibrovascular stroma. Nuclei are round and have finely stippled chromatin and inconspicuous nucleoli. Rosettes and pseudorosettes are common throughout the neoplasm. A grading system has been applied to olfactory neuroblastomas in cats, with high-grade tumors exhibiting increased cell and nuclear pleomorphism, increased mitotic activity, and areas of necrosis. 7 However, too few cases have been published and evaluated to draw any conclusions between tumor grade and behavior. Neoplastic cells exhibit immunolabeling for microtubule-associated protein-2 (MAP2), neuron-specific enolase, and less frequently synaptophysin, neurofilament, chromogranin, GFAP, and cytokeratin.7,80 Although a possible role of feline leukemia virus infection in the development of olfactory neuroblastomas has been proposed, no further confirmation of that hypothesis has been published to date. 99
Granular Cell Tumors
Granular cell tumors (GCTs) are rare tumors of uncertain histogenesis that affect many animal species and organ systems. 81 In cats, CNS GCTs have been described in the brain and spinal cord.64,114 Grossly, GCTs are well demarcated, extraparenchymal, and typically attached to the meninges. 36 Histologically, tumors are characterized by nests or sheets of round neoplastic cells with abundant cytoplasm containing periodic acid-Schiff (PAS)-positive, diastase-resistant granules and eccentric nuclei with dense chromatin.64,114 Neoplastic cells exhibit diffuse immunolabeling for vimentin, S-100, neuron-specific enolase, and desmin. 114 Although GCTs were classified as a type of meningioma (granular cell meningioma), 47 electron microscopy studies revealed that neoplastic cells lack desmosomes or gap junctions, ruling out a meningeal origin. 36
Round Cell Neoplasms
Although CNS lymphoma occurring as part of a multisystemic lymphoma is the second most common intracranial tumor and the most common spinal cord tumor of cats,61,67,113 primary CNS lymphoma is rare and accounts for less than 3% of all primary CNS neoplasms of cats.20,29,72,73,76,90,101 The diagnosis of a primary CNS lymphoma may be challenging, as it requires a thorough examination of extraneural tissues to exclude other potential sites affected by the tumor.61,90 Primary CNS lymphoma typically affects adult cats, and clinical signs vary according to the neurolocalization of neoplastic cells. Although gross lesions may be absent in most cases, tumors can appear as white to gray masses within the neuroparenchyma or meninges.73,101 In the spinal cord, lymphomas occur mainly within the epidural spaces, but are less often intraparenchymal.8,120 Histologically (Fig. 4c), CNS lymphomas occur as either a distinct neuroparenchymal neoplasm or as widespread perivascular infiltration throughout the neuroparenchyma and leptomeninges.8,20,29,72,73,76,101,109,112,113,120 Cases with perivascular distribution may mimic meningoencephalitis and can difficult the diagnosis.29,72 Neoplastic lymphocytes vary from small to intermediate cells with round nuclei and coarse chromatin with 1 to 2 nucleoli. Mitotic activity is variable, but most reported cases have low mitotic activity.20,29,72,73,76,101 Most primary CNS lymphomas are reportedly T-cell lymphomas.20,29,47,72
Primary histiocytic sarcoma of the CNS is exceedingly rare, with only a few confirmed cases in the literature.38,87,117 Similar to lymphoma, cases with tumors in the CNS and extraneural tissues do not fulfill the criteria to be classified as primary CNS histiocytic sarcomas. 117 Tumors typically occur in adult cats and lead to progressive clinical signs. 87 A clinical diagnosis of an intraparenchymal mass can be achieved with MRI.38,117 Published tumors were described in the brain.38,117 Grossly, neoplasms are nodular and yellow to gray. 87 Histologically (Fig. 4d), neoplasms consist of sheets of highly pleomorphic, round to elongate histiocytic cells with abundant eosinophilic and often vacuolated cytoplasm and round to indented nuclei with dispersed chromatin and 1 to 4 nucleoli. Giant multinucleated neoplastic cells are usually present throughout. Mitotic activity is variable.38,117 Neoplastic cells exhibit cytoplasmic immunolabeling for histiocytic markers such as Iba1. 38
Conclusions
Unlike the WHO classification of human CNS neoplasms, which utilizes tumor morphology and IHC aided by molecular diagnostic tools to provide a more objective tumor diagnosis, the diagnosis of CNS neoplasms in domestic animals relies heavily on histology and IHC. Until molecular data becomes available in veterinary medicine, investigations are needed to standardize species-specific CNS tumor classification and grading systems and to correlate distinct tumor types and grades with tumor behavior, prognosis, treatment modalities, and clinical outcome. These studies need to objectively define the histologic hallmarks of each neoplasm (tumor cellularity, cell pleomorphism, nuclear pleomorphism, mitotic activity, necrosis, etc.) as a means to provide reproducible methods for the evaluation of these neoplasms, increasing interobserver agreement and diagnostic accuracy among pathologists.
Footnotes
Acknowledgements
I thank Dr Chad West (Figs. 1a, 2a, 2b), Dr Kaori Sakamoto and Dr Chloe Goodwin (Fig. 1b), Dr Andrew D. Miller (Fig. 1c), Dr Uriel Blas-Machado (Fig. 2c), and Dr Aline Rodrigues Hoffmann (Fig. 2d) for allowing the use of their clinical and autopsy images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
