Abstract
Degus (Octodon degus) that were kept at a breeding facility presented with neurological or respiratory symptoms and died. Necropsies were performed on 9 individuals, and no significant gross lesions were found. Histologically, spinal cord necrosis was observed in all 9 cases and granulomatous myelitis in 5 of the 9 cases. Locally extensive necrosis of the brain and encephalitis were observed in 7 of the 9 cases. Acid-fast bacteria were found in the spinal cords, brains, and lungs from all 9 cases. Immunohistochemically, Mycobacterium tuberculosis antigen was observed in the spinal cords, brains, and lungs from all 9 cases. Double-labeling immunofluorescence revealed M. tuberculosis antigen in IBA1- and myeloperoxidase-immunopositive cells. Extracted genomic DNA from 8 of the 9 cases was successfully amplified with the primers for Mycobacterium genavense ITS1 and hypothetical 21 kDa protein genes, and the polymerase chain reaction products were identified as M. genavense by DNA sequencing. This report highlights the susceptibility of degus to M. genavense infection in the central nervous system.
Mycobacterium genavense is a nontuberculous mycobacterium that causes granulomatous pneumonia in various animal species. 18 Some reports suggest M. genavense is a potential zoonotic pathogen.30,31 In humans, abdominal lymphadenopathy associated with M. genavense infection has been reported in immunocompromised patients, especially in patients with acquired immunodeficiency syndrome (AIDS). 6 In birds, M. genavense is one of the most common causes of mycobacteriosis, along with M. avium. 37 M. genavense infection has been commonly identified in psittacines and other pet birds11,27,28,30,37 and is associated with granulomatous pneumonia and splenomegaly.30,31 Clinical signs include sudden death, weight loss, diarrhea, neurological changes, and respiratory disorders.30,31 M. genavense has also been detected in the feces of clinically healthy parrots. 20 Thus, birds are considered to be a possible reservoir of M. genavense.
M. genavense infection has caused generalized lymphadenopathy in dogs, cats, horse, small mammals, birds, and immunocompromised humans.4,5,7,9,11–13,18,21,24,28 In small mammals, M. genavense infections have been reported in a dwarf rabbit, a grizzled giant squirrel, a chinchilla, a guinea pig, and two ferrets.14,21,22,25,34 Systemic granulomatous inflammation was observed in these cases. Here, we report an outbreak of M. genavense infection resulting in spinal cord necrosis in degus (Octodon degus) that were kept at a breeding facility.
Materials and Methods
Cases and Pathological Examinations
Forty degus were kept in the facility (at least 22.5% of the degus were affected). No predisposing factors were identified. Eight degus showed neurological or respiratory signs; one degu died suddenly without any neurological signs. The degus were kept in the same room, and their cages were situated under birdcages. The age, sex, and clinical signs of the degus are provided in Table 1. Necropsies were performed on all 9 degus, and tissue samples from the heart, lung, gastrointestinal tract, liver, pancreas, spleen, kidney, brain, and spinal cord were collected. Tissues were fixed in 10% neutral-buffered formalin and embedded in paraffin. Formalin-fixed, paraffin-embedded tissues were sectioned to a thickness of 3 µm and stained with hematoxylin and eosin (HE) and Ziehl-Neelsen staining.
Clinical information of degus.
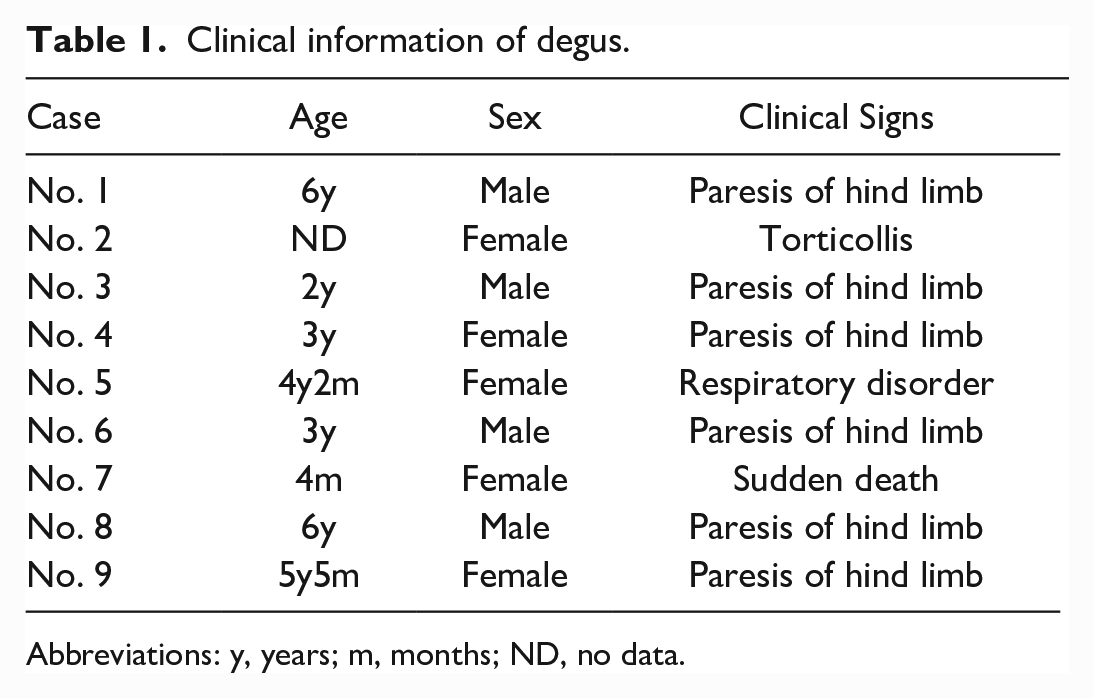
Abbreviations: y, years; m, months; ND, no data.
Immunohistochemistry
Serial sections were subjected to immunohistochemical analysis using primary antibodies including anti-Mycobacterium tuberculosis antigen, Iba1, glial fibrillary acidic protein (GFAP), neurofilament, and myeloperoxidase antibodies. Detailed information about the primary antibodies is listed in Supplemental Table S1. A previous report has shown that anti-M. tuberculosis antibody cross-reacts with nontuberculous mycobacteria. 32 Deparaffinized tissue sections were immersed in 3% hydrogen peroxide (H2O2) in methanol at room temperature for 5 minutes and then incubated in 8% skim milk in Tris-buffered saline (TBS) at 37°C for 40 minutes to avoid nonspecific reactions. After the blocking reaction, the sections were treated with a primary antibody at 4°C overnight. After washing with TBS, the sections were incubated with the Envision+ polymer (Dako, Denmark). Finally, the sections were visualized using diaminobenzidine tetrahydrochloride and counterstained with hematoxylin. Brain and spleen from a degu were used as positive controls for IBA1, glial fibrillary acidic protein (GFAP), neurofilament, and myeloperoxidase. A lymph node from a cat with mycobacterium infection was used as a positive control for M. tuberculosis immunohistochemistry. A negative control was prepared by applying an irrelevant antibody and buffer instead of the primary antibodies.
Double-Labeling Immunofluorescence
Double-labeling immunofluorescence was performed on spinal cord sections to identify M. tuberculosis antigen-positive cells using the anti-IBA1, neurofilament, and myeloperoxidase antibodies. Double-labeling immunofluorescence with primary antibodies from same host was performed using a method previously described. 33 Sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37°C for 40 minutes and then incubated with the anti-M. tuberculosis antibody at 4°C overnight. After washing with TBS, sections were incubated with a fluorescein-conjugated goat anti-rabbit IgG antibody (1:500, Vector Laboratories, United States). Sections were then heated at 121°C for 10 minutes in the citrate buffer (pH 6.0) to inactivate the anti-M. tuberculosis antibody and the anti-rabbit secondary antibody. Sections were blocked with 8% skim milk in TBS at 37°C for 40 minutes and then incubated with each primary antibody (IBA1, GFAP, neurofilament, or myeloperoxidase) at 4°C overnight. After washing with TBS, sections were incubated with an Alexa 594-conjugated goat anti-rabbit IgG antibody (1:500, Invitrogen, Japan).
When primary antibodies were raised in different hosts (anti-neurofilament and anti-M. tuburculosis antibodies), after antigen retrieval, a mixture of 2 primary antibodies was applied to each section and incubated at 4°C overnight. After washing with TBS, sections were incubated with an Alexa 488-conjugated goat anti-mouse IgG antibody (1:500, Invitrogen, Japan) and Alexa 594-conjugated goat anti-rabbit IgG antibody (1:500, Invitrogen, Japan) at 37°C for 40 minutes. After washing with TBS, sections were counterstained with DAPI (Vector Laboratories, USA) and the slides were examined with a laser scanning confocal microscope (LSM700, Carl Zeiss Meditec, Japan).
Polymerase Chain Reaction and Sequencing
Genomic DNA was extracted from formalin-fixed, paraffin-embedded spinal cord tissues using the QIAamp DNA FFPE Tissue Kit (Qiagen, Deutschland). Primer pairs for internal transcribed spacer region 1 (ITS1) and M. genavense hypothetical 21kDa protein gene were used (Table 2).1,8,31 Polymerase chain reaction (PCR) was performed in a thermocycler (C1000 Touch Thermocycler, USA) using KOD FX Neo DNA polymerase (TOYOBO, Japan) by preparing the following reaction mixture: 10 µL 2× PCR Buffer for KOD FX Neo, 4 µL dNTPs (2 mM), 0.4 µL forward primer (10 µM), 0.4 µL reverse primer (10 µM), and 0.4 µL KOD FX Neo (1 U/µL). Template DNA was added between 30 and 40 ng per PCR reaction followed by distilled water to a total volume of 20 µL. A negative control was included by adding 1 µL of ultrapure water (Wako, Japan) instead of DNA extracts. Synthetic genes of M. genavense ITS1 and hypothetical 21 kDa protein genes were used as positive controls (Eurofins, Japan; Supplemental Table S2). PCR using primer pairs 1 and 2 was performed as follows: initial denaturation at 95°C for 3 minutes, followed by 45 cycles of denaturation at 98°C for 10 seconds, annealing at 60°C for 5 seconds, and extension at 68°C for 1 second. After 45 cycles, samples were kept at 72°C for 5 minutes and then stored at 4°C. PCR for primer pair 3 was performed as follows: initial denaturation at 95°C for 3 minutes, followed by 45 cycles of denaturation at 98°C for 10 seconds, annealing at 55°C for 5 seconds, and extension at 68°C for 1 second. After 45 cycles, samples were kept at 72°C for 5 minutes and then stored at 4°C. The PCR products were submitted for sequence analysis (FASMAC, Japan). Sequence analysis was performed using the Basic Local Alignment Search Tool, and multi-sequence alignment was performed using CLUSTALW (https://www.genome.jp/tools-bin/clustalw).
PCR primers used in the present study.
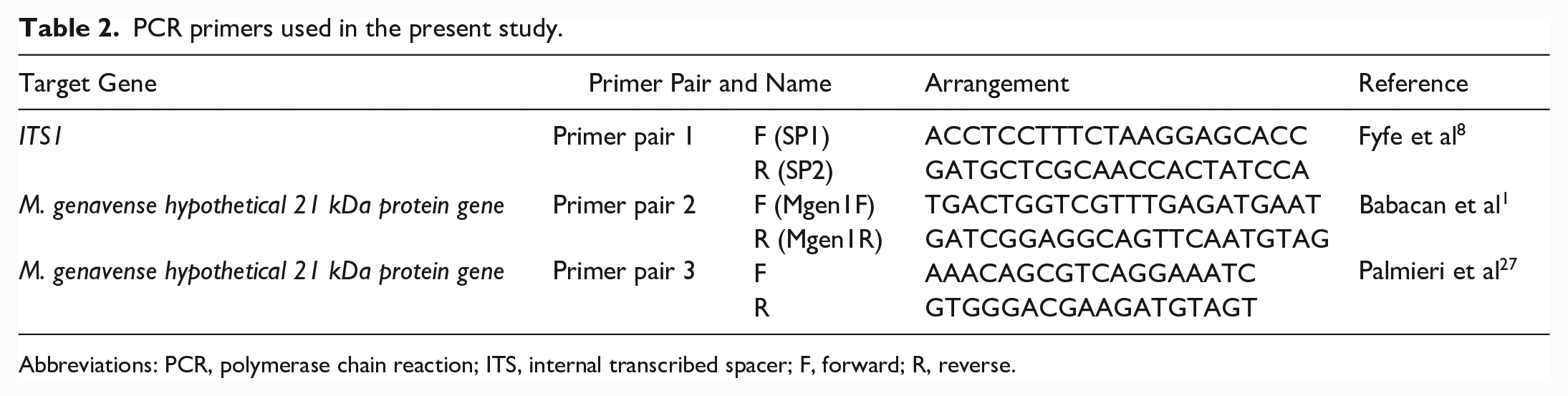
Abbreviations: PCR, polymerase chain reaction; ITS, internal transcribed spacer; F, forward; R, reverse.
Results
Gross and Histological Finding
At necropsy, the animals were in fair to good body condition, and no gross lesions were found. Microscopically, all cases had spinal cord necrosis affecting the white and gray matter of the spinal cord (Fig. 1a). The lesions were characterized by extensive granulomatous inflammation with macrophages, neutrophils, and multinucleated giant cells surrounding the vessels (Fig. 1b, c). The macrophages had moderate pale eosinophilic or foamy cytoplasm. Lesions in the brain were milder in comparison to the spinal cord. Locally extensive necrosis and granulomatous encephalitis were observed in 7 of the 9 cases. Furthermore, diffuse meningitis was observed in the spinal cord and brain. In the lungs, a few macrophages and lymphocytes infiltrated the alveolar walls and bronchioles. Ziehl-Neelsen staining revealed acid-fast bacteria in the cytoplasm of macrophages within the lesions of the spinal cord, brain, and lungs (Fig. 1d). No significant lesions were observed in the heart, spleen, gastrointestinal tract, liver, or kidney.
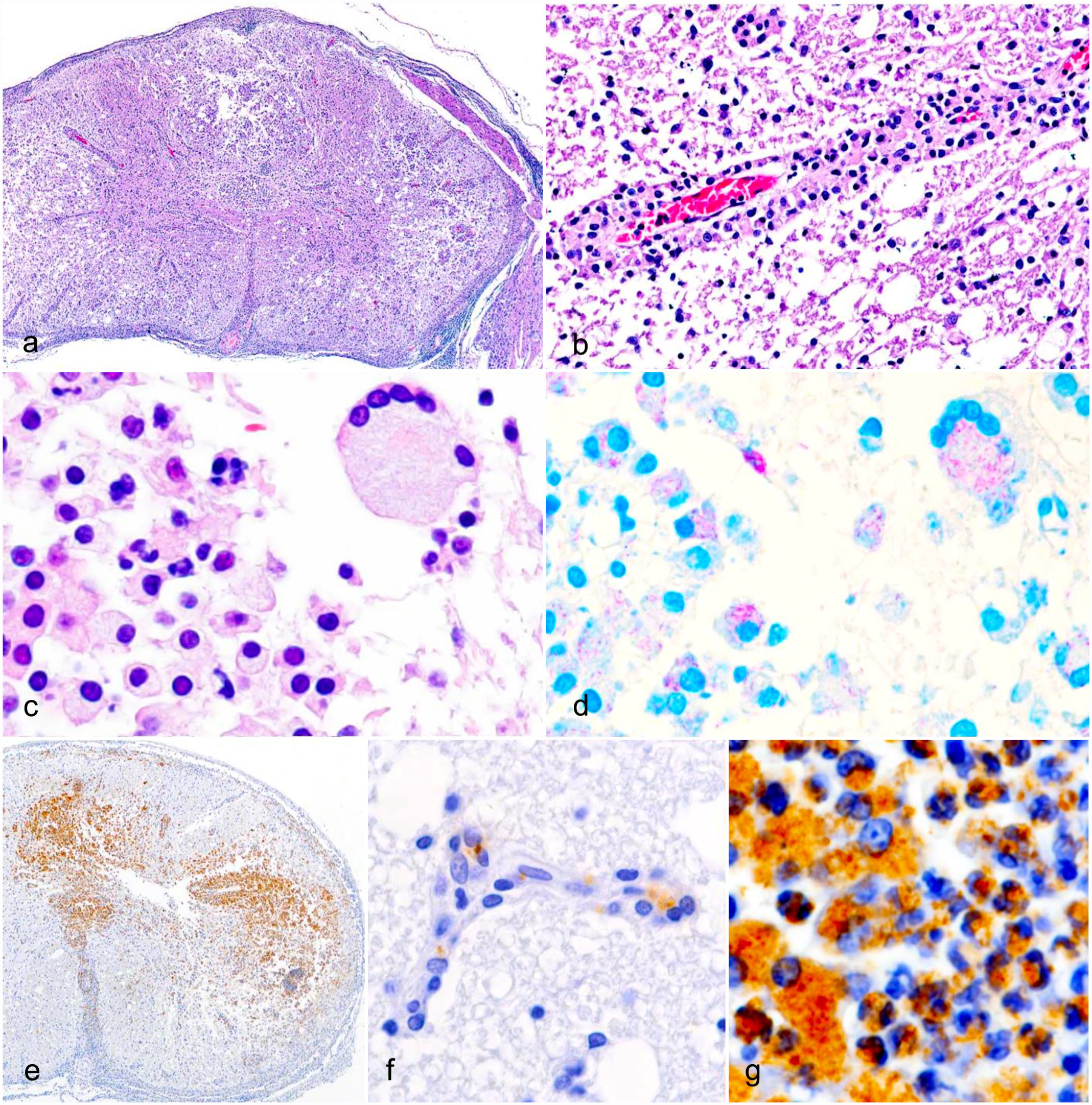
Mycobacteriosis, spinal cord, degu. (a) Diffuse spinal cord necrosis in the white and gray matter. Case 3: Hematoxylin and eosin staining (HE). (b) Macrophage infiltration around a vessel. Case 3: HE. (c) Macrophages, neutrophils, and a multinucleated giant cell in the lesion. Case 3: HE. (d) Macrophages and a multinucleated giant cell contain acid-fast bacteria. Case 3: Ziehl-Neelsen staining. (e) Immunolabelling is present in the white and gray matter. Case 3: Immunohistochemistry (IHC) for Mycobacterium tuberculosis antigen. (f) Immunolabelling is present in microvascular endothelial cells. Case 2: IHC for M. tuberculosis antigen. (g) Immunolabelling is present in macrophages and neutrophils. Case 2: IHC for M. tuberculosis antigen.
Immunohistochemistry and Double-Labeling Immunofluorescence
Positive reactions for M. tuberculosis antigen were observed in the spinal cords of all cases (Fig. 1e). M. tuberculosis antigen was found in the endothelial cells and macrophages of the lesions (Fig. 1f, g). A few positive reactions were observed in the lesions of the brain and lungs. M. tuberculosis antigen was not detected in the heart, spleen, gastrointestinal tract, liver, or kidney from 8 of the 9 cases and was observed in the heart and liver of case 7. Double-labeling immunofluorescence revealed M. tuberculosis antigens in the cytoplasm of myeloperoxidase-positive cells and IBA1-positive cells (Fig. 2), but not in GFAP- or neurofilament-positive cells.
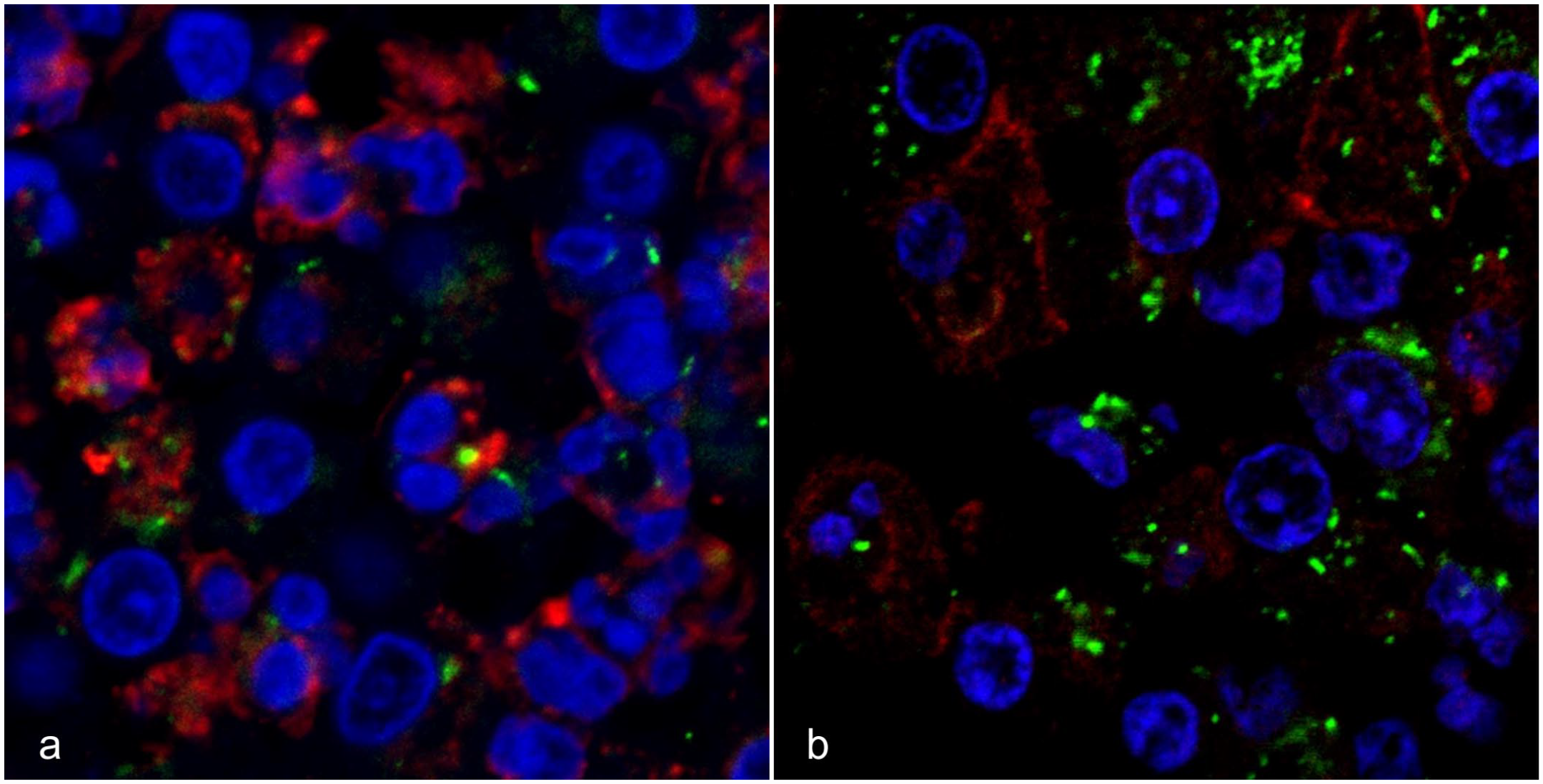
Mycobacteriosis, spinal cord, degu. Case 3: Double-labeling immunofluorescence (IF) for M. tuberculosis antigen (green) and inflammatory cell markers (red: myeloperoxidase and IBA1). Immunolabelling for M. tuberculosis is present in (a) the myeloperoxidase immunolabeled cells and (b) IBA1 immunolabeled cells.
Polymerase Chain Reaction
The extracted genomic DNA samples of 7 cases (cases 1–6, and 9) were successfully amplified with all 3 primer pairs, and that of case 8 was successfully amplified with primer pairs 1 and 3. The sequenced PCR products using primer pair 1 had >98% identity with M. genavense ITS1 in all tissues tested, except for those from case 5. The sequenced PCR products using primer pairs 2 and 3 had >98% identity with M. genavense hypothetical 21 kDa protein gene in all tissues tested. The DNA sequence alignment using CLUSTALW among the amplified DNA and M. genavense gene are shown in Supplemental Tables S3–S5. The quality of the extracted genomic DNA from case 7 was too poor and could not be successfully amplified with the 3 primer pairs.
Discussion
M. genavense is a slow-growing, fastidious mycobacterium that is difficult to culture. 5 In previous reports, M. genavense was detected by PCR.14,21,22,34 In the present study, M. genavense was detected in 8 of the 9 cases by PCR using primer pairs for ITS1 and hypothetical 21 kDa protein genes. The authors hypothesize that the negative PCR result in 1 case was associated with poor DNA quality, possibly related to significant postmortem changes. M. genavense infection of the central nervous system (CNS) is rare. Intracranial infection has been reported in severely immunocompromised humans.3,5,19,35,36 In such cases, multiple intracranial masses were found during imaging and diagnosed by biopsies. Mass lesions were not found in other visceral organs. Granulomatous encephalomyelitis with M. genavense infection has been reported in a spectacled Amazon parrot. 9 The parrot exhibited infection of the CNS with perivascular inflammation. The present study shows that M. genavense infection causes severe lesions in the CNS of degus without significantly affecting other visceral organs. In the present study, spinal cord necrosis with infiltration of macrophages and neutrophils was observed around the blood vessels. Ziehl-Neelsen staining and M. tuberculosis antigen-positive bacilli were detected in macrophages and neutrophils. The animals were in fair to good body condition, which may be associated with the localized lesions in the CNS and that other visceral organs were spared. The perivascular distribution of the lesions suggests a hematogenous invasion of the bacteria. The hematogenous route of infection has also been suggested in the parrot with CNS M. genavense infection and other CNS mycobacterial infections in cattle and humans.9,17,26 In addition to immune cells and endothelial cells, experimental infection studies suggested mycobacterium could infect neurons and astrocytes.10,29 In the present study, mycobacterium was not detected within neurons and astrocytes by double-labeled immunofluorescence.
An in vivo study utilizing CD18 leukocyte adhesion deficient mice suggested that free mycobacteria may traverse the blood–brain barrier independent of leukocytes or macrophages. 38 In addition, in vitro studies demonstrated that free mycobacterium invades brain microvascular endothelial cells.2,15 In the present cases, mycobacterium antigens were detected in endothelial cells by immunohistochemistry. However, we could not confirm it by double-labeling immunofluorescence using endothelial cell markers, such as CD31 and von Willebrand factor, due to the lack of cross-reactive primary antibodies for these samples.
Nontuberculous mycobacteria have been isolated from tap water and feces of both healthy and diseased birds, which acted as reservoirs.1,16,20,23,24,30 Furthermore, the presence of M. genavense DNA in the feces of birds has been reported. 1 In the present cases, the degus and birds were kept in the same room, and the degus’ cages were under the cages of birds. Although the route of infection and M. genavense reservoir in this facility have not been clarified, we suspect that the degus inhaled bird feces containing M. genavense, which caused pulmonary infection, and then M. genavense invaded the blood vessels and spread to the CNS.
In addition to the present report, M. genavense infection has been reported in other mammals, such as chinchilla, ferrets, a dwarf rabbit, a guinea pig, and a grizzled giant squirrel.14,21,22,25,34 In contrast to M. genavense infection in other mammals,14,21,22,25,34 the present cases showed neurological signs and were part of a multi-animal disease outbreak. Immunosuppression can be responsible for the unusual CNS presentation.2,3,6,15,17,19 Although there was no evidence of immunosuppression in the animals examined, immune competence assessments such as blood tests and bone marrow examinations are necessary for more definitive evaluation of the immune status, but were not available in this study. Degus may be susceptible to developing CNS lesions from M. genavense infection, which has not been described in other animal species. The present report describes an outbreak of M. genavense in degus at a breeding facility. Exposure of animals in research settings to other species that can be carriers of infectious diseases should be prevented. This study highlighted the importance of good hygenic conditions in a confined environment for public health.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231179094 – Supplemental material for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231179094 for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus) by Tatsuhito Ii, James K. Chambers, Mizuho Uneyama, Ayumi Sumi, Yutaro Nakayama, Nanae Tsurita, Yasutsugu Miwa and Kazuyuki Uchida in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858231179094 – Supplemental material for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus)
Supplemental material, sj-xlsx-2-vet-10.1177_03009858231179094 for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus) by Tatsuhito Ii, James K. Chambers, Mizuho Uneyama, Ayumi Sumi, Yutaro Nakayama, Nanae Tsurita, Yasutsugu Miwa and Kazuyuki Uchida in Veterinary Pathology
Supplemental Material
sj-xlsx-3-vet-10.1177_03009858231179094 – Supplemental material for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus)
Supplemental material, sj-xlsx-3-vet-10.1177_03009858231179094 for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus) by Tatsuhito Ii, James K. Chambers, Mizuho Uneyama, Ayumi Sumi, Yutaro Nakayama, Nanae Tsurita, Yasutsugu Miwa and Kazuyuki Uchida in Veterinary Pathology
Supplemental Material
sj-xlsx-4-vet-10.1177_03009858231179094 – Supplemental material for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus)
Supplemental material, sj-xlsx-4-vet-10.1177_03009858231179094 for Central nervous system mycobacteriosis caused by Mycobacterium genavense in degus (Octodon degus) by Tatsuhito Ii, James K. Chambers, Mizuho Uneyama, Ayumi Sumi, Yutaro Nakayama, Nanae Tsurita, Yasutsugu Miwa and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
