Abstract
Proliferation of ectopic Schwann cells within the central nervous system (CNS) parenchyma (schwannosis) in early life is most commonly associated with human neurofibromatosis type-2 and has been unrecognized in domestic animals. Three foals and a calf, 5 to 11 weeks old, with progressive neurological signs from birth were studied. Histologically, at multiple levels of the spinal cord, all animals had bilateral plaques of proliferative spindle cells, predominantly affecting the white matter adjacent to dorsal and ventral nerve roots and variably extending into the gray matter. Proliferating cells had strong intracytoplasmic immunoreactivity for the Schwann cell markers myelin protein zero and periaxin, highlighting the formation of peripheral nervous system (PNS) myelin within the spinal cord. In all cases, foci of disorganized neural tissue (glioneuronal hamartomas) were present, which in 2 cases formed a mass effect that resulted in syringohydromyelia. Neonatal presentation suggests a congenital maldevelopment of the nervous system, with spontaneous invasion of PNS-derived Schwann cells into the CNS.
Keywords
In mature vertebrates, the communication between the central and peripheral nervous systems (CNS and PNS) occurs at the transition zones. In these zones, axons are freely allowed to cross, while an astrocytic endfeet barrier (glia limitans) selectively segregates the territories of myelinating glial cells to their respective compartment of the nervous system. 7 Conventionally, oligodendrocytes are responsible for myelinating axons in the CNS, while Schwann cells myelinate peripheral axons, and intermixing among central and peripheral glia is not expected under normal conditions. The developmental origin of oligodendrocytes and Schwann cells are distinct with the former derived from oligodendrocyte precursor cells within the CNS and the latter derived from neural crest origin Schwann cell precursors. 9,19 Oligodendrocyte precursors arise in restricted regions, such as the subventricular zone of the brain. 9 Importantly, peripheral myelin is distinct from central myelin due to the occurrence of a unique set of proteins, including protein zero (P0) and periaxin, that are absent in central myelin. 15 This difference can be exploited to distinguish Schwann cell-derived and oligodendrocyte-derived myelin sheaths. In a number of developmental and other pathological situations, however, Schwann cells migrate into the CNS, ensheath, and selectively myelinate central axons. Myelin formation is dependent on axonal caliber and axonal trophic factors.
Schwannosis is a rare syndrome characterized by spontaneous invasion and benign proliferation of aberrant Schwann cells into the CNS, with simultaneous myelination of central axons, typically without obvious gross tumor formation. 1,12 Schwannosis should not be mistaken with schwannomatosis (also known as neurilemmomatosis), which is the third and more recently identified form of neurofibromatosis in humans, clinically and genetically distinct from neurofibromatosis type-1 (NF1) and type-2 (NF2). 6 Schwannosis is commonly but not exclusively encountered in patients with NF2, often in association with acoustic schwannomas (the hallmark of NF2) or ependymoma. 13 The nature of schwannosis remains unresolved as molecular assays have not yet evaluated whether the Schwann cell proliferation is clonal. Based on its typically benign clinical course, a maldevelopmental, dysplastic, or hamartomatous process has been suggested, 14 and a reactive etiology is favored in most cases. 5
In non-NF2 patients, the terms intramedullary and perivascular schwannosis have been applied to foci of Schwann cell proliferation in the spinal cord, almost always occurring beneath the dorsal root entry zones and along blood vessels. As it is mostly associated with localized disorders, such as trauma, infarction, syringomyelia, or neighboring neoplasm, this likely represents attempted, albeit aberrant, repair by inwardly migrating Schwann cells of peripheral origin. 13,14
To the best of our knowledge, the spontaneous occurrence of schwannosis has not yet been reported in domestic animals. This study aims to evaluate the histologic and immunohistochemical features of 4 animals with hamartomatous myelodysplasias that had features of schwannosis. We hypothesize that spontaneous PNS-derived Schwann cell invasion and proliferation into the CNS is the predominant feature manifesting in this malformation of the nervous system.
Three foals and 1 calf diagnosed with hamartomatous or dysplastic lesions in the spinal cord were retrieved from the necropsy database archives at Cornell University, College of Veterinary Medicine, Section of Anatomic Pathology, and the University of Florida, College of Veterinary Medicine. The case retrieved from the University of Florida was previously published. 17 All available information, including signalment, history, clinical presentation, diagnostic workup, outcome, and pathology findings, were collected. All patients presented with neurological signs compatible with a spinal cord disorder from birth and had complete neurologic examinations. The animals were euthanized, and necropsies were performed within 24 hours after death. Gross lesions included a focal spinal cord mass with syringohydromyelia (cases 2 and 4), scoliosis (case 2), hydrocephalus (case 1), and asymmetry of cerebral and cerebellar gyri and folia (case 3). Tissues were collected, fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Signalment, gross, and histologic findings are summarized in Table 1.
Signalment and Summarized Findings in 4 Cases of Schwannosis.
Abbreviations: F, female; M, male; wo, week-old.
Formalin-fixed paraffin-embedded tissues were used for immunohistochemistry against periaxin, a protein expressed throughout development that is exclusive to Schwann cells and their myelin product 15 (rabbit polyclonal anti-periaxin antibody, Sigma-Aldrich, Missouri, United States, catalog #HPA001868); myelin protein zero (P0), a membrane glycoprotein exclusively synthesized by Schwann cells and a major component of peripheral nerve myelin 3 (rabbit polyclonal anti-P0 antibody, Abcam, Cambridge, United Kingdom, catalog No. AB31851); oligodendrocyte transcription factor 2 (Olig2) (rabbit monoclonal anti-Olig2 antibody, Abcam, catalog No. AB109186); glial fibrillary acidic protein (GFAP) (rabbit polyclonal anti-GFAP antibody, Dako, California, United States, catalog No. Z0334); microtubule-associated protein 2 (MAP2) (mouse monoclonal Anti-MAP2 antibody, Sigma-Aldrich, catalog No. 45-M4403); S100 protein (rabbit polyclonal Anti-S100 antibody, Dako, catalog No. Z0311); and vimentin (mouse monoclonal anti-vimentin antibody, Dako, catalog No. M7020), using the Leica Bond Max Automated Immunohistochemistry Staining System, according to the manufacturer’s instructions (Leica Microsystems, Illinois, United States). Briefly, tissues were sectioned at 5 μm, and deparaffinized with Bond Dewax Solution (Leica, catalog No. AR9222). Pretreatment with heat-induced antigen retrieval was performed for 30 minutes using citrate-based buffer pH 6 for periaxin, MAP2, and vimentin (Bond Epitope Retrieval Solution 1, Leica, catalog No. AR9961) and Tris/EDTA pH 9 for P0 and Olig2 (Bond Epitope Retrieval Solution 2, Leica, catalog No. AR9640). No pretreatment was used for GFAP. Endogenous peroxidase activity was blocked with a 3% peroxide solution for 5 minutes (Leica, catalog No. DS9800). The antibodies were diluted at 1:1000 and applied to the slides for 15 minutes (anti-periaxin), 1:700 and applied for 10 minutes (anti-P0), 1:2000 and applied for 60 minutes (anti-Olig2), 1:5000 and applied for 15 minutes (anti-GFAP), 1:1000 and applied for 60 minutes (anti-MAP2), 1:400 and applied for 15 minutes (anti-S100), and 1:80 and applied for 15 minutes (anti-vimentin). Next, the biotin-free PowerVision poly-polymeric horseradish peroxidase anti-mouse (Leica, catalog No. PV6114) or anti-rabbit (Leica, catalog No. PV6119) IgG reagent was applied to the slides for 30 minutes. Tissues were then incubated with the Bond Polymer Refine Detection for 10 minutes (Leica, catalog No. DS9800). Subsequently, tissues were developed with 3,3-diaminobenzidine (DAB) chromogen (Leica, catalog No. DS9800) for 10 minutes. The slides were counterstained with hematoxylin (Leica, catalog No. DS9800) for 5 minutes, dehydrated, cleared, and mounted. The specificity of the primary antibodies was validated with recognized positive spinal cord and peripheral nerve controls from a horse and a cow. In the negative controls, isotype-matched antibodies replaced the primary antibodies.
At multiple levels of the spinal cord, all cases had bilateral plaques of proliferative spindle cells admixed with myelinated axons, predominantly affecting the peripheral white matter adjacent to the dorsal and ventral nerve roots. These plaques expanded to involve the dorsal, ventral, and lateral funiculi and variably extended into the gray matter (Table 1; Figs. 1–4). In case 1, a similar proliferation also affected the dorsal spinocerebellar tract in the caudal medulla oblongata. In addition, multifocal nests of immature, dysplastic neuronal and/or glial cells (glioneuronal hamartomas) and well-differentiated ependymal cells (ependymal hamartomas) were randomly distributed throughout the CNS, ranging from occasional small clusters to numerous, confluent lesions. Such hamartomas were noted in the medulla oblongata (case 1), mesencephalon (case 3), and cerebrum (cases 1 and 3), predominantly at the periventricular zones, but differed morphologically from the typical cell rests normally found in the subventricular zone (Figs. 5, 6). In cases 2 and 4, the glioneuronal hamartomas formed a mass effect compressing the spinal central canal and leading to syringohydromyelia.
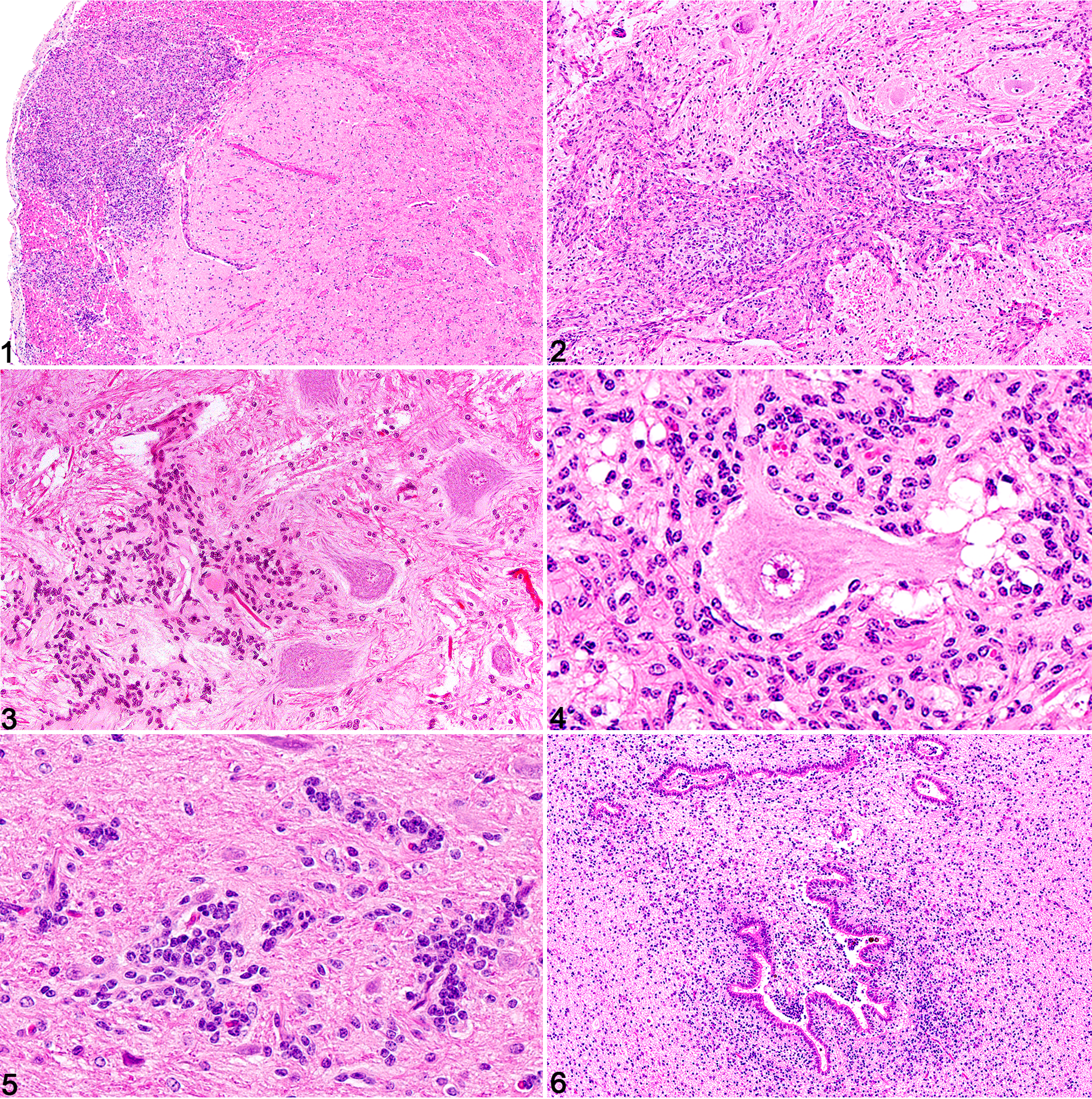
Schwannosis, central nervous system. Hematoxylin and eosin.
In all cases, the proliferating spindle cells and associated myelin exhibited strong immunoreactivity for P0 and periaxin, highlighting the formation of PNS myelin within the spinal cord and normal staining of the spinal roots (Figs. 7–12). Immunohistochemistry directed against periaxin, P0, Olig2, GFAP, MAP2, S100, and vimentin failed to detect immunoreactivity in the glioneuronal hamartomas in cases 1 and 3, suggesting immaturity of this population, but positive immunoreaction for vimentin was detected in the ependymal hamartomas. In cases 2 and 4, the hamartomas were similarly negative for P0 and periaxin but variably positive for Olig2, GFAP, MAP2, S100, and vimentin. Other comorbid histologic findings included widespread neuronal dysplasia involving the cerebellar granule layer in case 3.
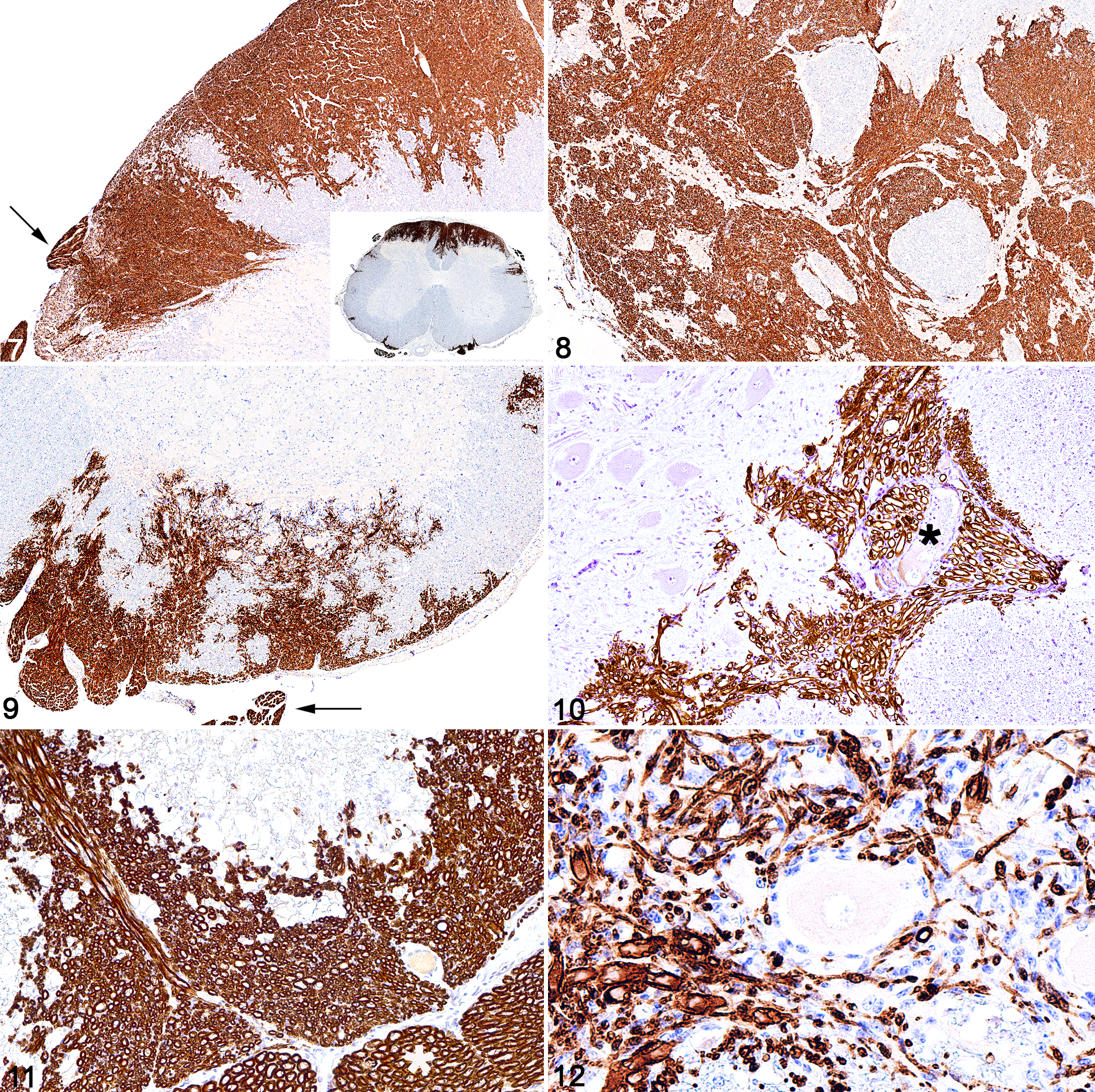
Schwannosis, spinal cord. Immunohistochemistry (IHC).
In humans, dysplastic or hamartomatous lesions of the CNS are well recognized in neurofibromatosis, predominantly in NF2. They may involve a variety of cell types, including neuroglial, meningeal, ependymal, and/or Schwann cells, and usually coexist with florid neoplastic proliferations of the same or different cell of origin, to which they often lie in close proximity. 13 In addition to schwannosis, other dysplastic lesions frequently associated with NF2 include meningiomatosis, angiomatosis, atypical glial cell nests, ependymal ectopias, hypertrophic gliosis of the optic nerve, Chiari malformation of medulla, and sacral spina bifida. Malformative lesions in NF1 are less frequent but most commonly include subependymal glial fibrillary nodules, hyperplastic proliferative gliosis, meningoencephalic cerebellar gliosis, micronodular vascular proliferations, and hydromyelia. 13,14 In an extensive immunohistochemical study of glial microhamartomas in the cerebral cortex and basal nuclei of humans with NF2, except for the S-100 antigen, no markers indicative of glial, neuronal, epithelial, endothelial, mesenchymal, or hematopoietic differentiation could be detected. 18 Despite the occasional appearance of rod-shaped and spherical blepharoblasts indicative of ependymal differentiation, these dysplastic foci in neurofibromatosis were designated glial hamartomas of presumably astrocytic origin. 14,18 It is possible that the spectrum of tissues involved in the hamartomas involves both neural crest- and neuroepithelial-derived elements.
In humans, schwannosis is not limited to a form of neurofibromatosis, and the underlying mechanism that drives the cellular proliferation and invasion has been debated. NF2 patients develop schwannomas, ependymomas, and meningiomas, and schwannnosis may be attributed to a tendency for the proliferation of primitive neural cells. As a consistent and disseminated feature, these proliferative lesions may be a consequence of the same or related genetic alterations associated with neurofibromatosis, allowing abnormally targeted cell invasion and proliferation. However, while in schwannosis patients some of these proliferative lesions presenting early in life can be grouped under the general term of hamartoma, in other instances, schwannosis seems to represent an idiosyncratic response to local injury or other pathological conditions known to stimulate a cellular proliferative reaction. The evidence in favor of a reactive, malformative response, as opposed to a dysplastic condition, has been further discussed, especially in the context of schwannosis in patients without neurofibromatosis, such as occurs in traumatized spinal cords 11 and experimental models of demyelinating diseases. 4
In several natural and experimental conditions in humans and animal models 2,10 and on rare occasions in naturally occurring animal disease, 8 centrally located Schwann cells and Schwann cell repair of CNS demyelination have been observed. This has generated various hypotheses with respect to the origin of the cells involved. For many years, it was assumed that Schwann cells remyelinating CNS axons were ectopic and that they responded to recruitment signals generated by demyelination and migrated from PNS sources into the CNS due to a disruption of the glia limitans. 4 However, genetic fate-mapping studies have revealed that while some CNS remyelinating Schwann cells are of PNS origin, others, perhaps the majority, are derived from the differentiation of intraspinal resident oligodendrocyte progenitor cells. 16,20 This evidence supports the probability that the CNS may normally have a resident Schwann cell population. In our study, the anatomical distribution of schwannosis—consistently in close proximity to PNS sources, such as the dorsal root entry zones and motor exit point— supports PNS-derived Schwann cells, representing ectopic peripheral glia that were centrally displaced during ontogenesis.
In conclusion, in these 3 foals and 1 calf, the morphology, distribution, and immunoreactivity of the proliferative lesions in the spinal cord were typical of Schwann cell infiltration, proliferation, and myelination of CNS axons and indicate that schwannosis was the most predominant feature of hamartomatous myelodysplasias. The neonatal presentation of clinical signs, together with this novel assortment of redundant, disorganized, hamartomatous, dysplastic, and heterotopic tissue, supports a congenital maldevelopment of the nervous system with schwannosis as the underlying primary anomaly. The 3 foals affected were different breeds, suggesting that these equine cases were sporadic and not familial. While neither NF1 nor NF2 are recognized in veterinary medicine, our study describes alterations that closely resemble the dysplastic or hamartomatous lesions associated with NF2 in humans.
Footnotes
Acknowledgements
We thank the histopathology technicians and the anatomic pathology residents and faculty at Cornell University College of Veterinary Medicine. In addition, we wish to acknowledge that Professor Marc Vandevelde at the University of Berne, Switzerland, identified a case of bovine schwannosis, which he discussed with B.A.S. (approximately 30 years ago).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
