Abstract
The eastern oyster, Crassostrea virginica (Gmelin), is both an important component of our estuaries and an important farmed food animal along the east and south coasts of the United States. Its populations have been significantly diminished in the wild due to decades of overfishing beginning in the 1890s. Unfortunately, in 1950, a new disease in eastern oysters caused by the protistan agent, Perkinsus marinus, was identified. The disease, resulting from infection with this protozoan, leads to high mortality of both wild and cultured eastern oysters. Current restoration efforts are hampered by the disease, as is the aquaculture of this economically important food. The parasite infects hemocytes and causes hemolytic anemia and general degeneration of the tissues, leading to death. Ongoing research efforts are attempting to develop oysters resistant to the disease. Transport regulations exist in may states. Infection with P. marinus is listed as a reportable disease by the World Health Organization.
Crassostrea virginica (Gmelin), the eastern oyster, has been and continues to be an important member of the coastal environment of the eastern and southeast United States. Historically, over several generations, populations of wild eastern oysters produced “oyster reefs” that provided important ecosystem services, including homes for other organisms. 2 Oysters filter enormous amounts of water, up to 15 liters per animal/d (http://www.sms.si.edu/irlspec/Oyster_reef.htm) and, in the process, remove not only microalgae from the water (their food) but also particulate matter, thus reducing the nitrogen loading in the water column. 18 In the past 120 years, C. virginica populations numbers have been significantly reduced from levels present in the 1890s by overfishing and loss of habitat. 29 Oysters and oyster reefs have almost completely disappeared, resulting in the loss of many of the ecological advantages provided to the estuaries. 2,18,31
C. virginica has been fished extensively for 100 years both by American Indians and later by European immigrants. It was a cheap and abundant food for coastal human populations, but with decreased availability and increased prices, they are now considered a specialty food item, and oyster aquaculture has become an economically profitable business. Oyster culture has long been a farming activity in the world and has been documented during the Roman Empire in Italy (although this was not C. virginica). Oyster culture in the United States began in 1896 and continues to be a major farming activity along the east, south, and northwest coasts of the United States. 28 In the United States, most oyster farms are owned and operated by small families who are not fishermen but rather aquatic farmers. Only recently has aquaculture, including aquaculture of oysters, been recognized by US Department of Agriculture (USDA) as a type of farming. This important step allows the USDA to provide agriculture services to the oyster farmers (as well as other types of aquaculture).
In addition to the overfishing of our natural populations of eastern oysters, an important new disease that continues to cause significant morbidity and mortality, both in wild and cultured animals, was identified in the Gulf of Mexico in 1951 by Mackin 30 and was named Dermocystidium marinum (nicknamed Dermo). In 1978, the genus was renamed as Perkinsus, 26,33 but the nickname “Dermo” is still used by many people in the industry. For many years, P. marinus was classified as a member of the phylum Apicomplexa, but recent molecular evidence shows it is part of a phylum that branched from the phylum Dinoflagellata just after the dinoflagellates themselves branched from the Apicomplexa. P. marinus is now classified as part of the phylum Perkinsozoa of the kingdom Alveolata. 15 This has important implications for understanding the life cycle of the organism and for identification of forms seen in oyster tissues and the water column.
From the 1950s to 1990s, the disease appeared to spread up the US eastern coastline from southern locations and currently causes significant morbidity and mortality in wild and cultured eastern oysters in Massachusetts and all the intervening states. 16 It is unknown if the agent was in these areas without detectible disease for a period before disease was noted. Currently, mortality from this disease varies, 43 but in most locations, it is the primary cause of mortality in cultured C. virginica on the east coast of the United States. 16 The economic impact of the disease has not been calculated but is considered to be high, and because of the possibility of spread of the disease agent to other areas of the world, P. marinus is on the World Health Organization’s list of reportable diseases (http://www.oie.int/animal-health-in-the-world/oie-listed-diseases-2012/).
The Open Vascular System and Hemocytes of Bivalves
P. marinus infects the blood vascular system of C. virginica. Oysters contain an open circulatory system. The vascular system consists of 2 atria, a ventricle, aortas, and arteries that conduct hemolymph (blood) to the various parts of the body. The aortas, as well as the proximal portion of the arteries leading from them, are thick-walled structures inconsistently lined by endothelium and surrounded by connective tissue (Fig. 1). Both endothelium and supporting connective tissue walls are lost as the arteries traverse through the tissues to the sinusoids. At the tissue level, the thin-walled vessels merge with, and empty into, large sinuses that permeate and surround all of the organs of the body (Fig. 2). Blood flow occurs through the sinusoids primarily because of the hydraulic action of the contracting and relaxing muscle bundles found throughout the tissues. Flow is loosely directed through the vascular system into venous sinuses, thin-walled vessels that collect the hemolymph and direct it back to the atria. The body tissues form organs as in vertebrates, but the organs are not surrounded by a serosal membrane, nor do they lie within a peritoneal cavity and thus are not physically separated from each other. As a result, the tissues have been described as “floating” in the sinusoids. 12 Exceptions to this arrangement are the heart, mantle tissue (that lines the inner surface of the shell’s valves), and the gills. The open vascular system of oysters may potentiate the ability of the parasites to move throughout the body from the initial location of infection.
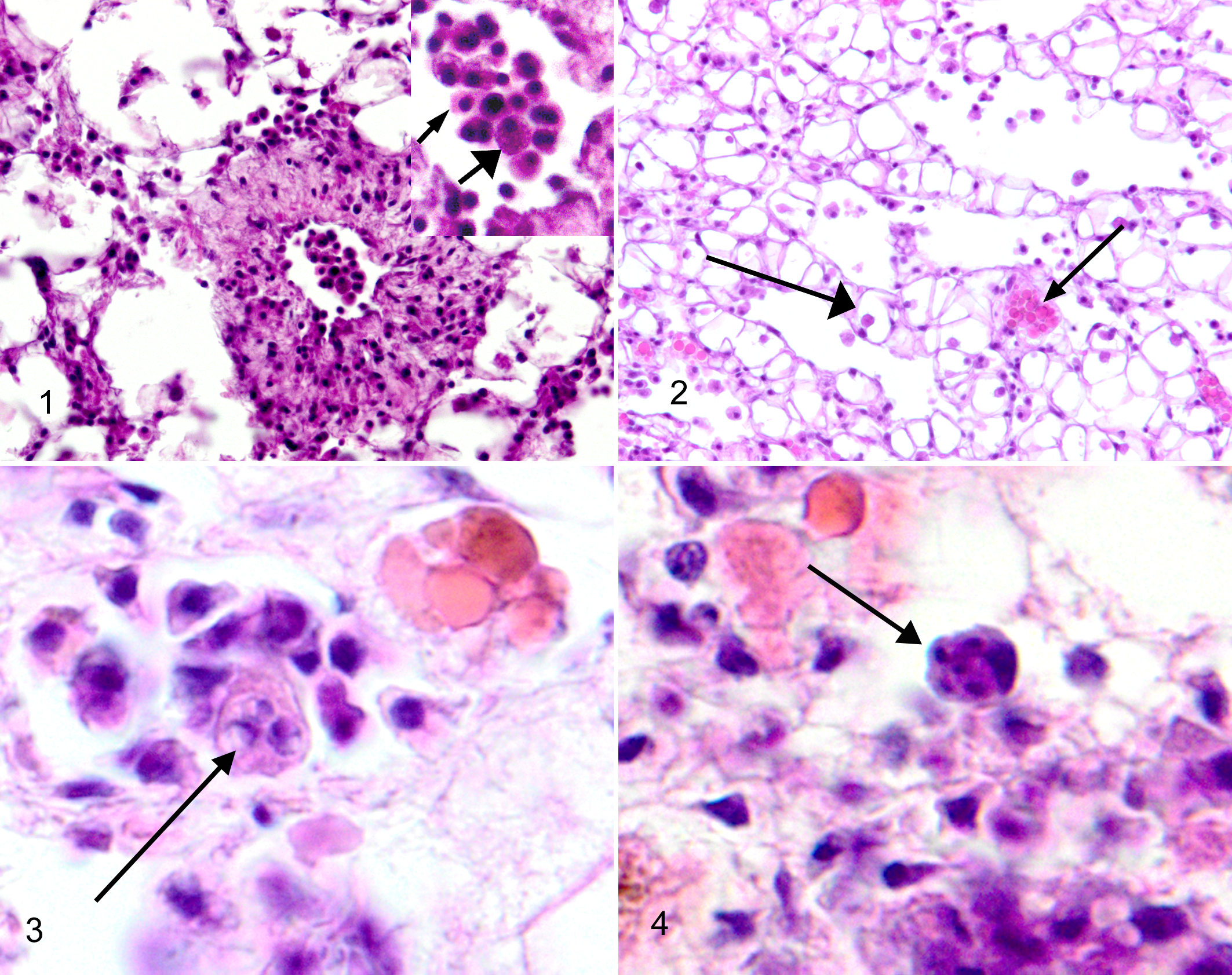
Vascular tissue; Crassostrea virginica. Hemocytes, including an agranulocyte (small arrowhead) and granulocyte (large arrowhead), are shown in the magnified inset of the arterial lumen. Hematoxylin and eosin (HE).
As research continues, scientists are learning more about the innate immune system present in all invertebrates, including oysters, and its capacities. C. virginica do not have lymphocytes or an inducible immune system. C. virginica organisms, as do most all bivalves, contain 2 basic circulating cell types (hemocytes): granulocyte and agranulocyte. 7,40 No circulating oxygen-carrying pigmented cells exist. Hemocytes easily move through most of the epithelial surfaces of the animal, but especially the gastric and digestive gland epithelium. Although both types of cells can migrate to areas of infection, engulf particles, adhere to each other to form aggregates (clots), and form encapsulations (granuloma-like structures), when organisms are too large to phagocytize, the granulocytes appear more readily able to engage in these activities. Granulocytes contain numerous important degrading proteins that can be expelled into the surrounding tissue and hemolymph to kill invading organisms. Recent work has shown that hemocytes exhibit a system of pattern-recognition receptors that provide the means for the cells to determine self from nonself. 40
Infection of Oyster Tissues by P. marinus
Trophozoites found within the water column are considered to be the infectious form of P. marinus. Uninucleate, 2- to 4-μm trophozoites that express specific ligands on their surfaces are complexed with hemocyte surface receptors, resulting in phagocytosis by the hemocytes. In 2007, Tasumi and Vasta 41 identified the hemocyte surface receptor, galectin CvGal, as that used by P. marinus to adhere and gain entry to the hemocytes.
Within a phagosome in the hemocyte, the trophozoite grows to 4 to 8 μm in diameter and forms an eccentric vacuole within itself as it expands. These forms of the parasite are identified as “signet rings” when examined in histological sections of infected tissue (Fig. 3). Sometimes a refringent vaculoplast can be identified in the vacuole, especially if a fresh squash of infected tissue is examined. The mature trophozoite undergoes successive partitioning (karyokinesis followed by cytokinesis) to form a rosette (Fig. 4) that develops into a mature sporangia containing anywhere from 8 to 32 new trophozoites. The mature sporangia (and the hemocyte!) lyses to release the newly formed trophozoites, which are 2 to 4 μm in diameter. Newly formed trophozoites infect naive hemocytes or float free in the hemolymph. 1,15,17
The methods used to evade destruction by hemocytes are currently under investigation. Evidence shows that the parasite is able to prevent the phagocytic respiratory burst that usually accompanies phagocytosis and most likely does so by the production of superoxide dismutases. 14,37,44 Other studies have demonstrated the ability of the parasite to modulate the normal apoptotic reaction within the hemocyte, thus allowing time for replication by the parasite in the hemocyte. 23,45 Effects of iron on the parasite are also identified, including the promoting or inhibiting effects of iron and iron chelators on the proliferation of the parasite in the hemocyte. 27
Oyster Tissue Responses to Infection
Early in infections, a significant inflammatory response is noted in the infected tissues. Hemocytes containing the trophozoite move from epithelial locations into the open vascular system and travel throughout the body in the hemolymph as part of the hemocyte. Although initial infections can occur anywhere in theory, they are predominately identified histologically in the gastric and digestive gland epithelium (Fig. 5). Gills are an important alternate location for initial infections. Hemocytes containing different reproductive stages of P. marinus can be histologically identified between epithelial cells of the initially infected tissues. Migration of moderate numbers of hemocytes to the infected areas is noted in the underlying and surrounding sinusoidal connective tissues. Gastric ulcers associated with P. marinus infections are occasionally identified but are rare. In general, the tall columnar gastric epithelium remains intact but contains numerous infected hemocytes.
Gastric epithelium; Crassostrea virginica. Tall columnar gastric epithelium with numerous infected hemocytes intercalated between the epithelial cells. Increased numbers of hemocytes are noted in the underlying sinusoidal connective tissues. Hematoxylin and eosin (HE).
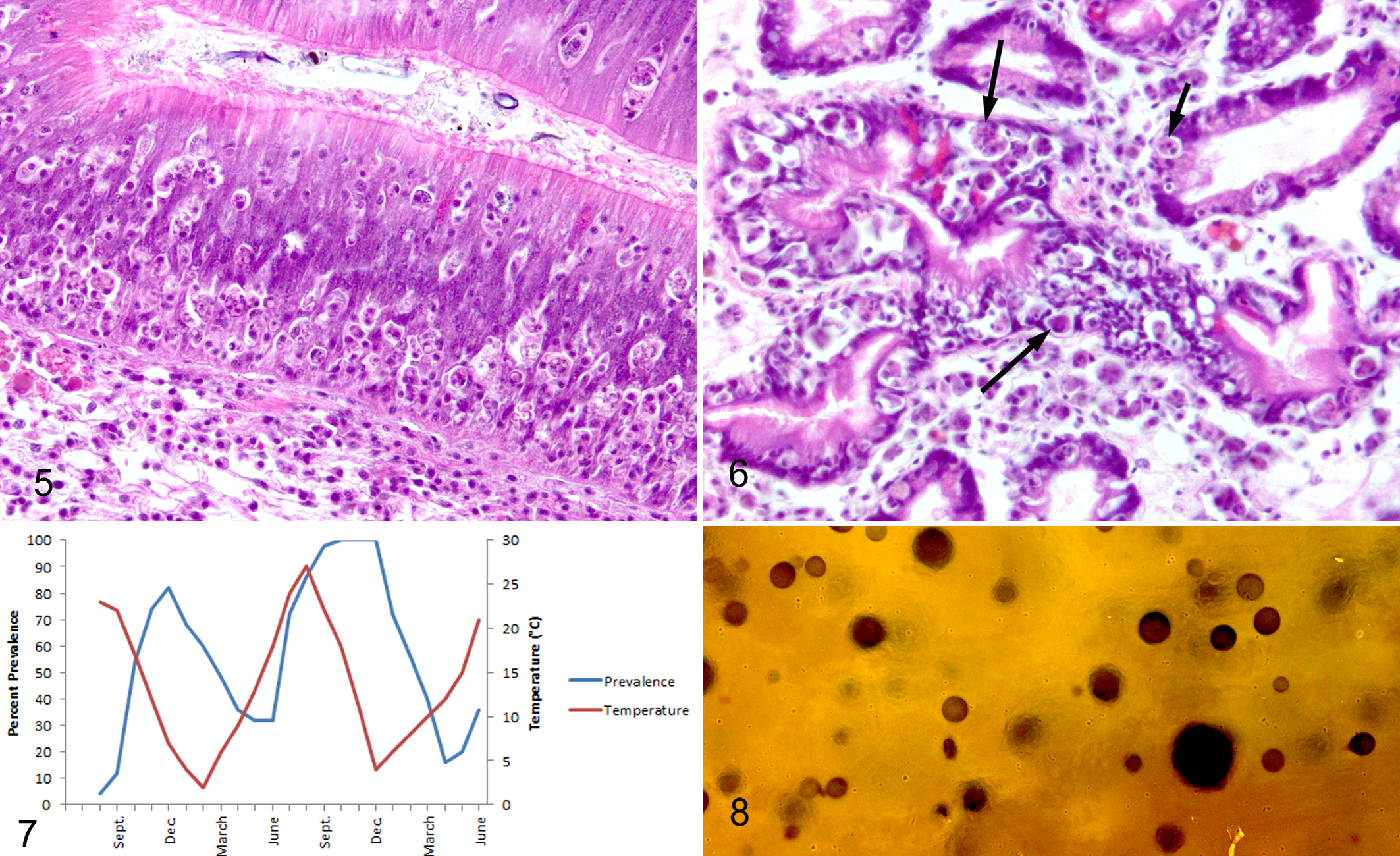
The digestive gland is formed as outpouchings from the gastric lumen that branch or arborize to form a series of ducts, lined by tall cuboidal to columnar epithelium, ultimately leading to blind-ended tubules. These tubules are lined by absorptive, columnar to cuboidal epithelium divided by 4 areas of crypt-like foci of mitotic cells that continuously replace the discarded absorptive cells. The digestive gland provides the majority of the animals’ digestive and absorptive abilities. In P. marinus infections (Fig. 6), variable necrosis of the digestive gland epithelial cells is noted multifocally in the ducts and tubules, which results in low cuboidal to squamous metaplasia of epithelial cells. Peri- and intraepithelial hemocytic inflammation is noted and, in moderate to severe infections, most of the hemocytes contain identifiable P. marinus forms. Obliteration of the infected ducts and tubules sometimes occurs. In the gills, lesions resulting from P. marinus infections consist of aggregates of hemocytes within the sinusoids of the gill, some of which obviously contain the P. marinus forms. Damage to the gill epithelium is not noted, but it is thought that the hemocyte aggregates impede hemolymph flow.
As the disease progresses, fewer hemocytes are noted in the vascular spaces throughout the animal’s body (parasitic anemia), and increased numbers of free sporangia and trophozoites are identified in the sinusoids. In animals severely affected by the disease, most tissues are atrophic and edematous, and few viable hemocytes are noted in the sinusoids. In the early stages of the disease, the animal is not significantly affected, but as the disease progresses, the loss of hemocytes that form the innate immune system, as well as the decrease in available nutrients to the oyster tissues, causes the animals to slowly become dehabilitated 8 (R. Smolowitz, personal communications). Other suggested mechanisms of death include increases in lytic factors in the hemolymph. 11
The gross appearance of C. virginica infected with low numbers of P. marinus is not readily differentiated from uninfected animals. As infections increase in intensity and severity, dehabilitation of the oyster results in watery, thinned soft tissues visible when the oyster valve is shucked (valves are broken open). However, other diseases can cause similar degeneration of the oyster tissues, resulting in a lack of any gross lesion that can be associated with the disease caused by P. marinus. In addition, normal animals that have recently spawned can present a similar gross appearance.
Other bivalves are infected by different species of Perkinsus spp. One, Perkinsus olseni, on the World Health Organization’s list of reportable diseases (http://www.oie.int/animal-health-in-the-world/oie-listed-diseases-2012/), currently causes significant mortality in numerous bivalve species and is widespread in wild bivalves located in the Indo-Pacific region. Tridacna crocea (an ornamental reef clam) imported into the United States from Vietnam was recently diagnosed with P. olseni. 38 Unlike P. marinus, other forms of Perkinsus incite multifocal encapsulation reactions (granuloma-like) in the tissues that can be identified both grossly and microscopically.
Epizoology of Infection by P. marinus
Infection is spread directly between oysters. Trophozoites from dead, rotting oyster tissue have been experimentally shown to directly infect oysters, as have trophozoites from cultured P. marinus. 17 Trophozoites are also released through the digestive epithelium into the intestinal lumen in viable oysters. This is an important second method for release of the infectious agent into the water column. When the oyster dies and the oyster tissues begin to degenerate, trophozoites of P. marinus formed in the oyster tissues will develop into hypnospores (enlarged trophozoites). Replication occurs in the hypnospores, and eventually zoospores (flagellated spores) are produced and then released into the water column from the hypnospores in the rotting oyster tissue. 15,17 Although research shows that trophozoites can easily infect oyster tissue, as yet no one has been able to show that zoospores can infect oysters directly. It is not known if zoospores develop into trophozoites, 3,15 and recent evidence strongly suggests that P. marinus may undergo a sexual reproductive cycle in the sediment/water column. 42 Importantly, evidence points to the probability that different strains of P. marinus exist and that pathogenicity varies with strain. 3,45 This finding has implications for movement of infected oysters.
Severe disease resulting from infection of C. virginica by P. marinus occurs several months after infection. The metabolic system of both the oyster and the parasite is most active at high temperatures and slows down as the winter months approach. It is thought that in the northeastern United States, where winters have historically been long and cold, the disease was moderated or even prevented by this environmental control, but as climate change with warming increases the average year-round temperature and shortens the winters, an increase in the disease is occurring in northeastern coastal areas. 16 In the laboratory, the parasite grows best in culture at 25°C to 35°C and dies at 35°C, and although it does not reproduce, the organism can withstand temperatures of 1°C. In the northeastern United States, disease prevalence cycles annually. Prevalence and severity of the disease rise from a low prevalence and severity in the early spring to a high level in the late fall (Fig. 7). Mortality peaks in the late fall. Mortality has been reported to slow significantly in an oyster populations when the temperature decreases to 20°C, and although it is uncertain why, it may be that the innate immune system may be slightly better at ridding the animal of the less actively proliferating parasite at lower temperatures. 1,17 Such findings have been identified in hard clams (Mercenaria mercenaria) infected with protozoan parasites. 21 In the southeastern United States and Gulf of Mexico, the disease does fluctuate, but the change is not as dramatic, although it is linked with an annual change in temperature. 17 In southern areas, prevalence and severity are lowest between January and April and highest between August and November.
Both the oyster and the parasite are also affected by the salinity of the water in which they are found. Eastern oysters can live at very low salinities, 1.2 parts per thousand (ppt), up to 36.6 ppt, but they grow best between approximately 14 and 28 ppt. 39 However, significant oyster mortalities due to sudden, excessive freshwater runoff, especially during periods of high temperature, have been identified. 39 P. marinus itself does not survive at salinities below 3 ppt and grows best in salinities of 29 to 35 ppt. 17 Annual environmental changes, such as drought and periods of heavy rain, both considered important changes due to global warming, have a measurable effect on the prevalence and severity of the disease in both wild and cultured eastern oysters 16 by increasing the number of infectious agents and decreasing the potential effectiveness of the oyster’s innate immune system.
The origin of P. marinus is unknown. Interestingly, P. marinus was recently noted in an oyster species considered for culture along the east coast of Mexico (Crassostrea corteziensis). As reported by Hughes et al, 23 P. marinus (confirmed molecularly) has also been identified in natural populations of C. corteziensis (pleasure oyster). 47 In addition, a wild species of oyster, Saccostrea palmula (the mangrove oyster), in the same area on the west coast of Mexico, was later confirmed to be infected with P. marinus. 5 Other oysters, including Crassostrea gigas, is cultured along the west cast of the United States and can become infected with P. marinus but does not exhibit even mild disease. 6 At this time, it is not known where P. marinus originated; because it is identified in various species of oysters, on both coasts of the United States, the possibility of introduction of the parasite along with movement of various species of oysters between coasts needs to be considered.
Diagnostic Methods
Three methods are commonly used to diagnose P. marinus infections. Unfortunately, the small trophozoites are easily missed in traditional histological examination of stained tissue sections, so the use of histological evaluation of specimens, especially young animals with early infections, is not usually conducted. The method of diagnosis used for the past several decades is the thioglycollate culture method. 35 Using this method, rectal and mantle tissue (less often digestive gland tissue) is removed from a shucked oyster (aseptically) and incubated in sterile thioglycollate culture media for 5 to 7 days. During this period, the trophozoites in the tissues form hypnospores. After incubation, the degraded oyster tissue containing the hypnospores is removed from the culture media, minced, and placed on a slide. The tissue is swamped with a dilute iodine solution that stains the thick hypnospore walls (Fig. 8). A coverslip is added and the preparation is examined using a compound microscope so that the number of black/brown-stained hypnospores can be estimated. 22 The prevalence of infected animals as well as the intensity of infection can be determined when several animals in the population are examined in this manner. The thioglycollate culture method of diagnosis is neither sensitive nor specific because only small pieces of each animal’s tissue are examined, and all other Perkinsus species may produce stained hypnospores. However, species of Perkinsus other than P. marinus are exceedingly rare in the eastern oyster (and hopefully will remain so). Some diagnostic and research laboratories have begun to use molecular techniques to identify P. marinus in oyster tissues. 10 This method is specific and sensitive, but most laboratories that provide diagnosis services for the disease still rely on the thioglycollate culture method.
Treatments
There are no practical treatments for large juvenile and adult oysters infected with P. marinus. To acquire the microalgae food needed to support oyster growth, once oysters have grown to a juvenile stage (approximately 600 μm in diameter; 2 months), they must be moved to a culture environment that will allow them to filter plankton from the seawater. Thus, treatment of large numbers of large juvenile and adult oysters would be extremely difficult to accomplish safely because they have been deployed in our estuaries. Treatments for P. marinus infections using environmental modulation (such as transferring animals to low salinity) have been examined, but again, most culturists have thousands of oysters in bags or trays in the intertidal or subtidal environment. Environmental treatment of such large quantities of animals in a location away from the culture environment, and even on board a boat at the site, is in many cases not economically or physically possible. However, treatment with quinine, triclosan, and other chemotherapeutics has been shown to be effective in a laboratory setting. 9,32
Various organizations, both governmental and industrial, have promoted the possible introduction of a nonnative oyster species into the US eastern and southern coastal waters in the hope that a new oyster species could take the place of C. virginica. Examples are Ostrea edulis (the European flat oyster), C. gigas (the Pacific oyster), and, most recently, Crassostrea ariakensis (the Suminoe oyster). Pockets of O. edulis do currently exist along the northeastern coastline because some animals were introduced into Maine waters several years ago, but they do not inhabit the same locations as C. virginica and so have not proliferated and are not used by the culturists. In addition, O. edulis brought along its own diseases. The introduction of C. ariakensis was proposed a few years ago and continues to be debated. Work shows C. ariakensis will probably inhabit the same locations as C. virginica and could possibly serve as a replacement for C. virginica. Most regulators and culturists are not willing to introduce this nonindigenous species of oyster into the environment since it may provide the death blow to the remaining populations of C. virginica by outcompeting or producing infertile hybrids. 19 The use of triploid C. ariakensis in aquaculture has been investigated but currently has not been accepted. The potential reversion to diploidy, although low, is possible and could result in wild populations of C. ariakensis. Culturists and regulators are hesitant to “give up” on C. virginica. Most regulators, culturists, and researchers hope to develop resistant, locally adapted C. virginica organisms in the future. 19 To date, introduction of a nonindigenous species has been determined to be an inappropriate method. But comparisons of the 2 oyster species continue to be investigated with the thought that C. ariakensis may someday be used in aquaculture or released as diploids into the estuaries to form new oyster reefs. 4,24
Current Regulations for Control of P. marinus Spread in the United States
Regulations concerning transport of oysters infected with P. marinus vary from state to state, and regulations controlling inter- and intrastate movement are determined by different agencies in each state. Some states do not regulate movement. Others, such as Massachusetts, do not allow adult animals to move between different water bodies and only allow uninfected seed to be moved and thus used in aquaculture (requiring a health examination before movement occurs). In Rhode Island, movement of seed is allowed if the prevalence and severity of the disease are less in the stock to be moved than in the stocks in the area that will receive the animals. This knowledge requires monitoring, which is not always available and does not at this time take into account the possibility of variable pathogenicity of P. marinus strains. Unfortunately, in all states, animals transported to restaurants or other locations in which the oysters are used as food are not required to be inspected for prevalence and severity of P. marinus infections. Although P. marinus does not infect humans or other animals, the shucked oyster waste, especially when discarded as fresh material from restaurants situated on the shore into the surrounding waters, can be a source of infectious agent. Many states do not permit the placement of oysters, purchased from stores or restaurants in other states, to be put in their own state waters, but it is a hard policy to enforce since tourists often bring along oysters from their home state to hang off of the dock of a vacation house they rented in another area. Since this is a directly infecting organism, transmission of a new strain of P. marinus (or introduction of P. osleni or other species of Perkinsus) is a concern.
The oyster industry, industry regulators, and researchers have recognized that the best way to deal with P. marinus is to prevent it. Several groups of researchers have identified disease resistance, also termed tolerance, in oyster populations that have experienced the disease for several generations in the natural setting. 13,34 Current research is aimed toward understanding the genes involved in the development of resistance so that resistant brood stock can be selected and used for culture or restoration. 20,25,36,46,47 Strains of resistant oysters have been established. The most commonly used, best-recognized resistant oyster strain is NEH, which was developed by Rutgers University, Haskin Shellfish Laboratory (http://hsrl.rutgers.edu/services/HSRLDisResOysterLineOrigins.pdf).
Conclusion
The eastern oyster continues to be both a prized food and an important component of the estuarine environment and the economy along the east and southeast coasts of the United States. Its populations have been devastated in the past few decades both by overfishing and by the introduction of new diseases. The most significant disease affecting these populations is that caused by P. marinus, a protistian organism. Climate change is already associated with increasing prevalence and severity of the disease and accompanying oyster mortality in the mid and northeast Atlantic coastal areas. The industry, state and federal regulators, and researchers continue to examine methods to deal with this problem, and although no one solution is in sight, much progress has been made in understanding the disease, developing resistance in eastern oysters, and establishing new wild populations of oysters along the coast. The direction that is taken by the industry and the regulators to deal with the effects of this disease on the ecological services provided by wild oysters will depend on the research conducted in the next few years.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
