Abstract
Disease outbreaks in several ecologically or commercially important invertebrate marine species have been reported in recent years all over the world. Mass mortality events (MMEs) have affected the noble pen shell (Pinna nobilis), causing its near extinction. Our knowledge of the dynamics of diseases affecting this species is still unclear. Early studies investigating the causative etiological agent focused on a novel protozoan parasite, Haplosporidium pinnae, although further investigations suggested that concurrent polymicrobial infections could have been pivotal in some MMEs, even in the absence of H. pinnae. Indeed, moribund specimens collected during MMEs in Italy, Greece, and Spain demonstrated the presence of a bacteria from within the Mycobacterium simiae complex and, in some cases, species similar to Vibrio mediterranei. The diagnostic processes used for investigation of MMEs are still not standardized and require the expertise of veterinary and para-veterinary pathologists, who could simultaneously evaluate a variety of factors, from clinical signs to environmental conditions. Here, we review the available literature on mortality events in P. nobilis and discuss approaches to define MMEs in P. nobilis. The proposed consensus approach should form the basis for establishing a foundation for future studies aimed at preserving populations in the wild.
Epizootics caused by emerging infectious diseases often result in mass mortality events (MMEs), particularly in cases involving newly introduced pathogens for which the host in question has had no previous exposure. These epizootics have become more frequent in shellfish over the last 30 years. Infectious diseases of fish, including their etiological agents and transmission mechanisms, have been extensively studied, but much less is known about pathogens of shellfish and other benthic fauna, particularly bivalves. 35 The lack of information about the causes of disease in marine invertebrates is severely limiting our understanding of the etiology and environmental factors contributing to MMEs. 18 In aquatic animals, complex interactions between heterogenous bacteria, viruses, and parasites are further complicated by an array of noninfectious environmental factors, resulting in polymicrobial infections with pathologic outcomes that might be difficult to predict and control.19,94 Novel cross-disciplinary approaches, involving simultaneous epidemiological and ecological studies at various levels of biological organization (molecular to population), are promising to provide a deeper understanding of host-pathogen interactions.22,44
The pen shell, also called the fan mussel, Pinna nobilis is one of the largest bivalves of the Mediterranean Sea, inhabiting coastal areas and deeper areas in the range of 0.5–60 meters. In the last decade, extensive mass mortalities of the species in many Mediterranean countries (Italy, Spain, Greece, Turkey, and France) resulted in its inclusion in the Annex II Barcelona Convention (1992), Annex IV of the EU habitats directive (2007), and redefining the species as “critically endangered” by the International Union for Conservation of Nature (IUCN) Red List for Threatened Species. The phenomenon began in 2016 and continues to devastate the populations of fan mussels to the present day. MMEs can reduce a population in a short period of time, in some instances, due to specific environmental conditions or weather events, which can be the underlying and triggering causes. 67
These phenomena have been reported all over the world in different marine taxa. 36 In the Mediterranean Sea, large-scale temperature anomalies, corresponding to increases in the frequency and intensity of marine heatwaves (MHWs), have been reported since 1999 and occurred over 5 consecutive years, between 2015 and 2019. These temperature anomalies were associated with MMEs of microbenthic species belonging to different phyla in Italy, France, and Spain.1,18,22 The most affected species reported during these MMEs belong to the coralligenous community that includes gorgonians, echinoderms, and sponges, which are present along thousands of kilometers of coastline from the surface to 45 meters. Reported data refer mostly to temperature and noninfectious factors, but no pathologic data at the level of the individual or species have been reported for most of these events. 46
A clear understanding of the etiology of the ongoing MMEs affecting native P. nobilis populations in the Mediterranean basin is necessary. Recent diagnostic investigations repeatedly confirmed the simultaneous presence of several pathogens in the diseased bivalves. Mycobacterium spp. was identified frequently, followed by H. pinnae. Vibrio mediterranei and Perkinsus spp. were also detected in some cases, suggesting that exposure to different pathogens could increase the complexity of the disease pathogenesis (Fig. 1). The phylogenetic relationships of Mycobacteria spp. isolated from moribund noble pen shells demonstrated a close relation to the Mycobacterium simiae complex, which includes important zoonotic agents responsible for the cause of emerging human and mammalian diseases.28,46,64
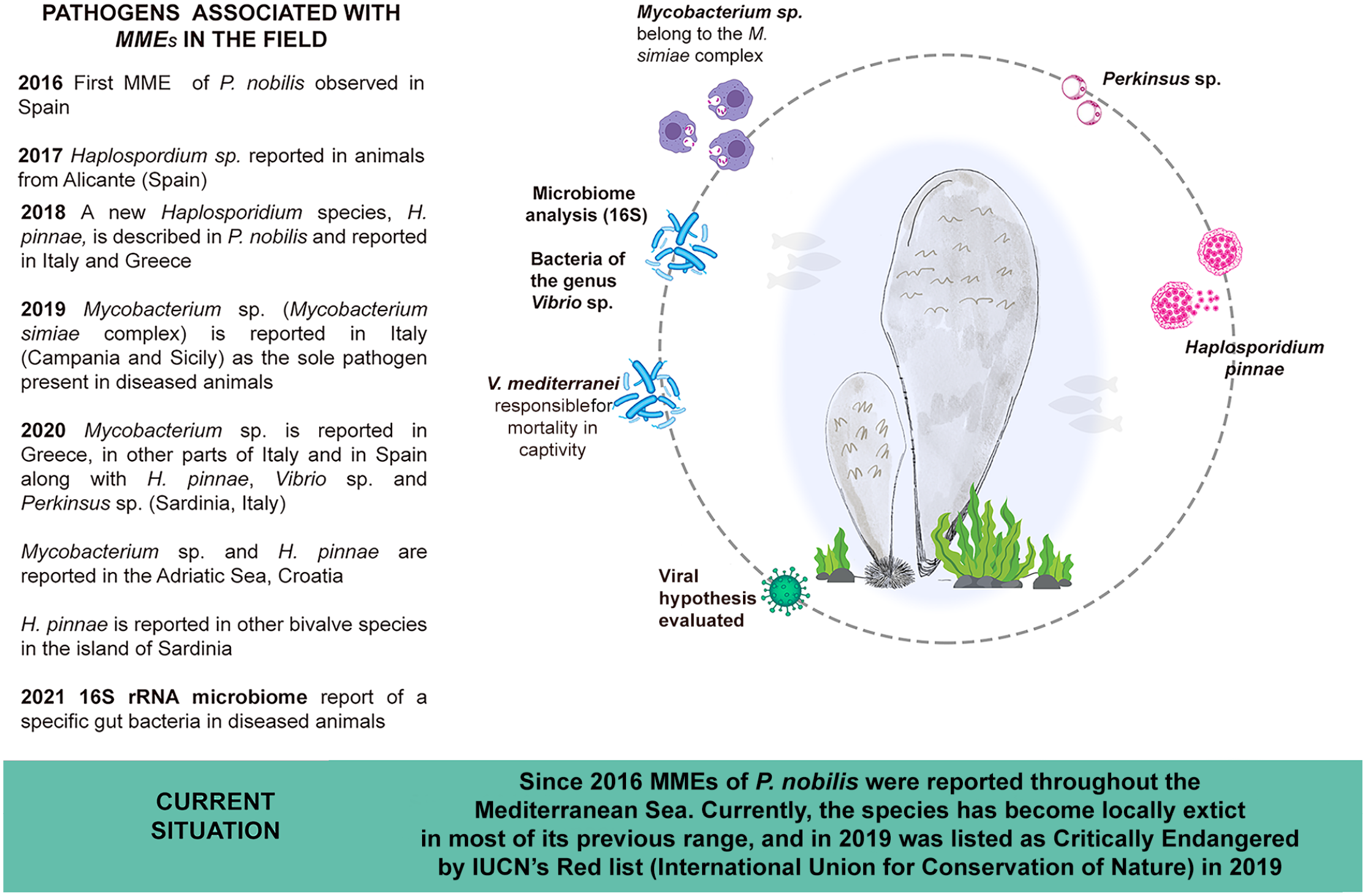
Pathogens reported to be associated with mass mortality events of noble pen shell (Pinna nobilis) since 2016. MME, mass mortality event.
Therefore, there is a strong need for implementing active surveillance programs focusing on new emerging pathogens with zoonotic potential. This is especially relevant for the interface of the human/marine habitat in the Mediterranean basin, which is currently experiencing a unique ecological transition represented by biological disturbances, climate change, and modifications of the deep-sea ecosystems. Altogether, these factors can drive the emergence of new pathogens in unpredictable ways, as already reported.1,96 Improving the available diagnostic protocols for other species is mandatory for updating the surveillance capabilities for disease outbreaks and MMEs and to support healthy coastal ecosystems. Based on the available scientific evidence, we discuss how the MMEs of P. nobilis are likely a consequence of a complex interplay between infections and noninfectious factors. In this review, we stress the urgent need for future research to refrain from simplified disease hypotheses and take into consideration multiple additional elements that may have an impact on the physiology of the largest bivalve in the Mediterranean basin. At events of increased mortality in the marine environment, professionals investigate the episodes through qualitative clinical and pathological examinations and submit samples to laboratories for histopathological assessment and diagnostics for infectious agents. However, the investigation into the pathogenesis of MMEs in marine environment has its limitations in revealing the causality of a disease without standardized diagnostic protocols.
Overview of the MMEs
The Pathogens Involved in P. nobilis MMEs and Their Interplay
Several pathogens have been associated with the MMEs of the pen shell and are related with MHWs. The first report on MMEs of P. nobilis described the presence of a haplosporidian parasite in the epithelium of digestive gland tubules in the autumn of 2016 although other pathogens have since been reported (Fig. 1). 30
Haplosporidians are endoparasites of invertebrates have a wide host range including bivalves, crustaceans, tunicates, and polychaetes. 21 The phylum Haplosporidia has been associated with epizootic mortalities of farmed bivalves due to the Office International des Epizooties–listed pathogens Bonamia ostreae and Bonamia exitiosa and by the multinucleated sphere unknown (MSX) disease caused by Haplosporidium nelsoni. An outbreak due to H. nelsoni along the mid-Atlantic coast of the USA devastated local oyster populations and was responsible for significant economic losses.35,48 In the case of the noble pen shell, the species in question is a member of the genus Haplosporidium, belongs to a large clade of species in the order Haplosporidia, and is closely related to a Haplosporidium sp. that infects cultured shrimp (Penaeus vannamei) in the Caribbean Sea and Indonesia.27,70,91 The parasite has been reported in Greece, Italy, Spain, Croatia, and Turkey24,55,61,80 and is considered to be one of the major causative agents of P. nobilis MMEs.
Haplospordian life cycle stages include 2 main phases: a unimultinucleate plasmodium and a sporulation phase producing resistant spores with a typical opening covered by an external lid. 26 The plasmodia stage divides by plasmotomy and can undergo sporulation. Spores are released into the environment after the death of the host, but they do not seem to be infective to the host in which they are produced and may require a different host for completion of their life cycle. 26 The morphological description of H. pinnae was first reported by Darriba et al 30 and afterward by Catanese et al. 27
Gross examination of affected animals can reveal, in some cases, the presence of fluid-filled cavities in the digestive system (Fig. 2), resulting in atrophy of the underlying digestive epithelium, and haplosporidium sporocyst production within digestive gland tubules. 24
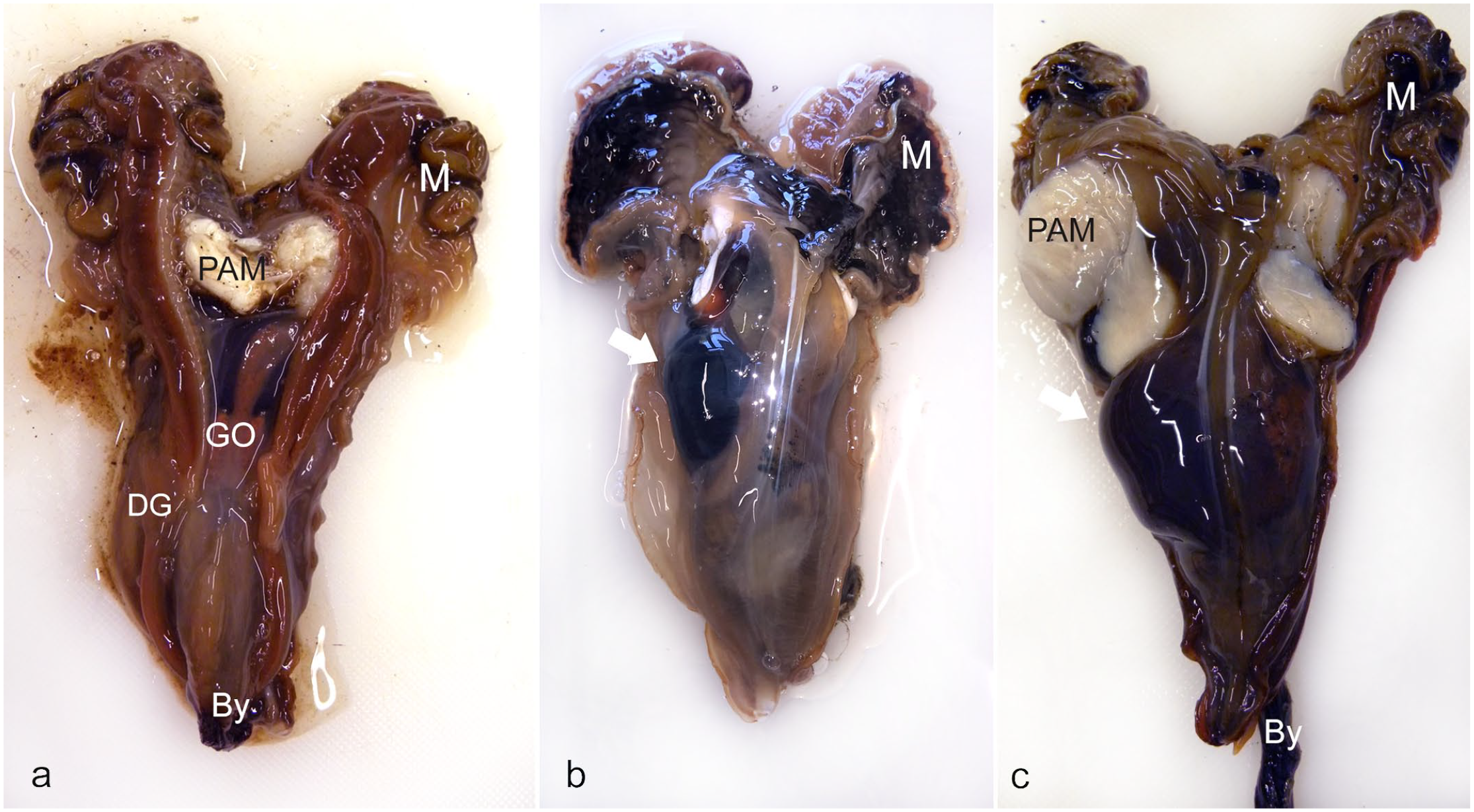
Macroscopic pathology in the digestive gland of noble pen shell (Pinna nobilis). (a) An individual with a normal-size digestive gland (DG). (b, c) Digestive gland displaying a fluid-filled cavity (arrow), containing hemocytes and associated with Haplosporidium pinnae, sampled in Croatia (b) and Italy (c) but classified as “healthy” (b) and “diseased” (c) from the promptness in closing the valves. PAM, posterior adductor muscle; GO, gonad; By, byssus; M, mantle; DG, digestive gland.
The H. pinnae plasmodial phase is mainly uninucleate and measures 2–3 μm in length with a central or slightly eccentric dense nucleus. The plasmodia are generally observed within the hemocytes and in the connective tissue of the mantle and of the digestive gland (Fig. 3a–c). Sporulation occurs in the epithelium of the digestive gland tubules, occasionally in the gut epithelium, with transformed plasmodia having a lighter eosinophilic cytoplasm. Epithelial distention by sporocysts, which are 30 μm in diameter, impinges the lumina of digestive tubules. When mature, sporocysts are released into the lumen (Fig. 3d). In this phase, the host inflammatory (hemocytic) response can appear mild, with few hemocytes present, or absent.24,25,30 However, severe inflammation of the connective tissue of the digestive gland, with hypertrophy and hyperplasia of brown cells (fixed phagocytes of connective tissues of certain bivalves), has been reported (Fig. 3e, f).27,61
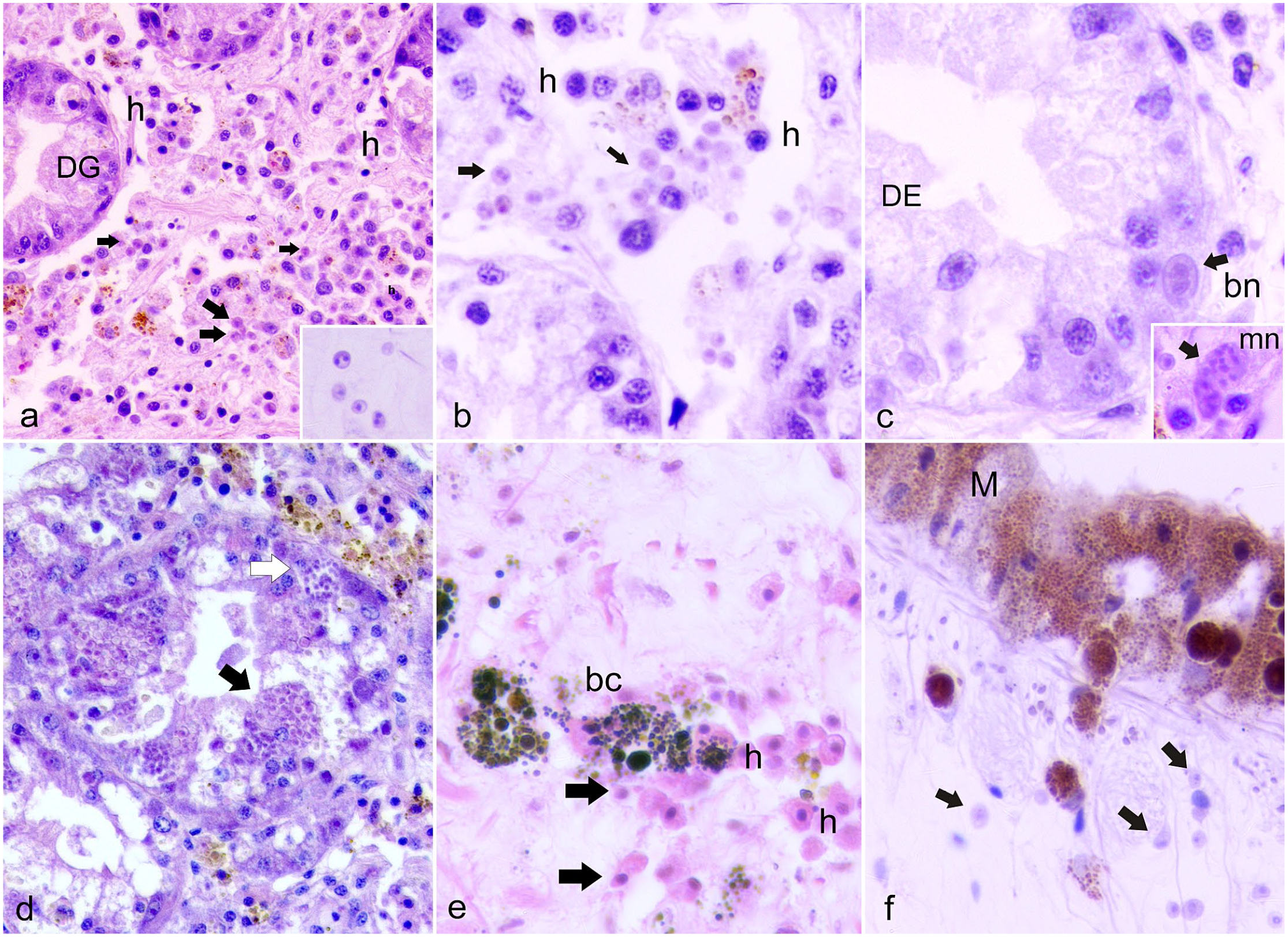
Histopathology of the digestive gland (a–d) and mantle (e, f) of fan mussels, Pinna nobilis infected with Haplsoporidium pinnae. Hematoxylin and eosin. (a, b) Uninucleate and binucleate plasmodial stages (arrows and inset) in the interstitium of the digestive gland (DG) with hemocytes (h). (c) Binucleate phase (bn) and multinucleated (mn, inset) plasmodium within the digestive tubule epithelium (DE) (arrow and inset). (d) Digestive gland tubular epithelium occupied by parasite sporocysts enclosing sporoblasts (black arrow) and sporocysts enclosing variably mature spores (white arrow). (e) Hemocyte (h) clusters with brown cells (bc) and uninucleate plasmodia (arrows). (f) Mantle epithelial cells (M) filled with melanin migrating to plasmodial cells (arrows).
In terms of pathogen prevalence and intensity, the dynamics of haplosporidians in their hosts can be seasonal and dependent on environmental parameters like water currents and salinity. 21 In particular, as a pivotal step toward understanding mortality of P. nobilis, model simulations of particle drift dictated by regional surface currents have been performed. 43 Reported data showed that oceanic currents constitute a potential factor driving the expansion of these type of parasites.23,43 Unfortunately, the distribution of the disease pattern cannot be conclusively ascribed solely to H. pinnae because many of the mortality reports were collected by “citizen scientists” who did not perform pathological analyses to identify pathogens.6,23
Indeed, H. pinnae is not the only pathogen causing MMEs. Mortality of P. nobilis occurs also in the absence of H. pinnae and can be due to an infection by an incompletely classified Mycobacterium. The first report of a mycobacterial MME occurred in 2 southern Italian regions in 2018, Campania and Sicily. 24 Subsequently, a diagnostic survey was conducted on moribund P. nobilis specimens retrieved from other Italian regions, such as Tuscany, Sardinia, and Apulia, and then extended to Spain, in Catalonia. These studies showed that in the early phases of MMEs, dying specimens had a systemic disease associated with an infection by a Mycobacterium species, belonging to the simiae complex. A further molecular study of the Mycobacterium in P. nobilis used the genetic marker hsp65 and the internal transcribed spacer ribosomal DNA for differentiating the strain. The study supported that the Mycobacterium infecting P. nobilis is close to Mycobacterium triplex, within the M. simiae complex with Mycobacterium sherrisii, and is phylogenetically distant to other mycobacteria reported from other mollusk species and other marine organisms. In fact, the M. simiae complex comprises slow-growing nontuberculous mycobacteria (NTM), which were initially identified from Rhesus monkeys in 1965.52,92 The complex has also been reported in humans in the southern United States, Cuba, Palestine, Iran, Israel, Turkey, Japan, and more recently in Sri Lanka.47,54 The species M. sherrisii was recently described and characterized 82 and is now formally validated as a novel species. 93
In most published cases, this Mycobacterium is reported to be an opportunistic pathogen, affecting immunocompromised individuals, such as HIV patients or those with pre-existing pulmonary diseases. Recent 16S rRNA gene data obtained from the American University of Beirut Medical Center in Lebanon revealed that M. simiae complex is the most common NTM isolated in human patients. 8
In P. nobilis, we still have scarce information on the pathogenetic mechanisms of this Mycobacterium.
Upon opening an infected pen shell, gross lesions are nonspecific and include generalized tissue edema, characterized by diffuse tissue swelling due to fluids collecting in the interstitial spaces of mantle and gill. 24 Microscopically, the bacteria seem to localize mainly within the eosinophilic granulocytes and, in some cases, in the brown cells, which has also been shown by ultrastructure. 24 They apparently escape from the phagosome, establishing themselves in the cytoplasm of the host cell in a manner similar to other Mycobacteria (Fig. 4). 24 Generally, three different patterns of infection are described based on the morphology of the inflammatory response involving granulocytes and brown cells and its distribution, classified as focal, multifocal, and diffuse. Diffuse inflammation is differentiated from focal inflammation when the affected area does not have multiple centers of hemocyte infiltration and hemocytes are abundant and distributed broadly over a large section of tissue. Multifocal inflammation forms enodules, and granulocytes can be admixed with aggregates of brown cells located in connective tissue of the digestive gland and gonads (Fig. 4a–c). Numerous intracytoplasmic acid-fast bacteria positive to Ziehl-Neelsen stain are within the nodules of granulocytes. Granulocytes, admixed with brown cells, may be degenerate within the center of the nodules (Fig. 4d). In reactive connective tissue, proliferation of fibrous tissue is frequent with hyalinocytes infiltration and acid-fast bacteria within brown cells (Fig. 3c, d).
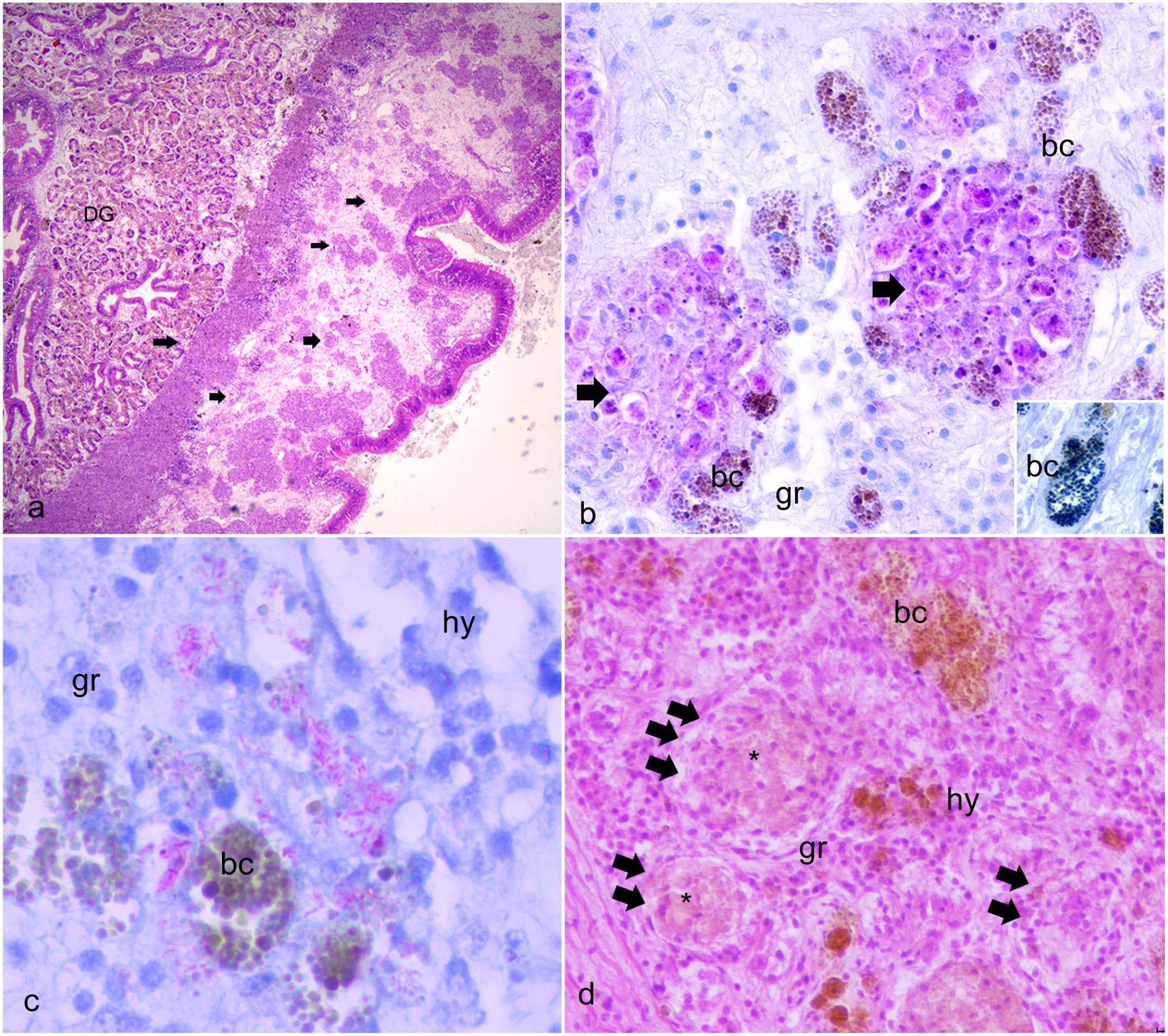
Histopathology of fan mussels, Pinna nobilis infected with Mycobacterium sp. (a) Multifocal nodulation in the connective capsules (arrows) enclosing the digestive gland (DG). Hematoxylin and eosin (HE). (b) Multifocal nodular type granulocytic (gr) inflammation (arrows), including aggregates of brown cells (bc) positive for lipofuscin with Schmorl stain (inset), located in connective tissue of digestive gland. Ziehl Neelsen stain. (c) Aggregated granulocytes (gr) filled with acid-fast bacteria in the connective tissue of visceral mass fusing with brown cells (bc); hy, hyalinocytes. Ziehl Neelsen stain. (d) Recruitment of granulocytes (gr), hyalinocytes (hy), and brown cells creating multifocal inflammatory nodules (arrows) and possibly necrotic centers (asterisks).
Studies before 2019 did not investigate the presence of this Mycobacterium.20,23,62 However, other researchers reported the same mycobacterial species in specimens involved in MMEs from many locations around the entire Mediterranean basin. Both Mycobacterium spp. and H. pinnae have frequently been detected.25,61,74,80
Further phylogenetic analysis of the pathogens isolated from moribund animals showed that strains of Mycobacterium and H. pinnae had high similarity to samples previously reported from Greece, Spain, and Italy.25,59,61,73 In Catalunya, H. pinnae was observed in 36% of the examined specimens, always in association with Mycobacterium sp. 25 Histopathological studies in specimens from Thermaikos Gulf, Greece, showed the presence of Mycobacterium spp. with H. pinnae. 61 Interestingly, monitoring of a P. nobilis population from the Thermaikos Gulf, an estuary of extremely high importance for bivalve production, revealed the presence of both pathogens in a few specimens without clinical symptoms of the disease. 61 Mass mortality of the population in the Thermaikos Gulf occurred during a prolonged period of increased seawater temperature in the autumn, causing the collapse of all populations in shallow waters (4–10 meters). In the Aegean Sea, the infection spread to all habitats by the late spring of the same year, limiting the distribution of surviving fan mussel populations to only the Kalloni Gulf Lesvos Island and the Maliakos Gulf, Greece. Despite the temperature drop in the winter of 2019, mortality of the species continued, albeit at a rate lower than that in the summer months of the previous year. 59 The histopathology of moribund animals showed more severe lesions in specimens that showed concurrent infections of both Mycobacterium spp. and H. pinnae. 80 Moderate to severe inflammatory lesions were linked to the unique presence of Mycobacterium spp., while absent-to-moderate inflammatory foci were seen when H. pinnae was found alone. In the latter case, moderate inflammatory lesions were associated with the sporulation phases of the parasite. Lesions were absent, or of mild intensity, in the presence of plasmodia. This suggests that the detection of different developmental stages of this parasite could have diagnostic and pathogenic relevance during Haplosporidium sp. infections. The simultaneous detection of both pathogens, and their presence within the inflammatory lesions, was observed in most of the examined sick/moribund animals. The absence of MMEs in areas where these pathogens were not detected 80 supports the hypothesis that both Mycobacterium and Haplosporidium are cooperating in the progression of the disease, thus synergistically leading to MMEs affecting P. nobilis in the Mediterranean Sea. Given the lack of clarity of Haplosporidium life cycles, there is also the possibility that additional biotic factors, such as the presence or absence of alternate hosts, may contribute to the severity of MMEs. 36 The lack of such knowledge clouds the interpretation of the available data.
Further molecular diagnostic analyses on P. nobilis, P. rudis, and other bivalve species were performed in Sardinia, Italy, 81 and demonstrated that H. pinnae was present in other bivalves at least 3 years before the first reported occurrence of a P. nobilis MME. H. pinnae was reported in 27 of the 48 (56.3%) P. nobilis analyzed by polymerase chain reaction (PCR). The protozoan was found in 71.4% of individuals with clinical signs, such as weakness in closing the valves, but also in 44.4% of individuals without clinical signs. In a group of asymptomatic individuals, PCR testing for the presence of H. pinnae revealed a total of 12 positive and 15 negative specimens. PCR assays targeting the mycobacteria 16S rRNA were negative in 46 of the 48 individuals tested. The only exceptions were a P. nobilis from the north-western coast of Sardinia and a P. nobilis from the northern coast of Sardinia, which were found positive for a Mycobacterium sp. that had 98% identity with the Mycobacterium sp. formerly described. 25
Other typical bivalve pathogens such as Vibrio spp. and the dinoflagellate parasite Perkinsus sp. were detected in a few cases, suggesting that exposure to multiple pathogens could increase the complexity of the disease pathogenesis.25,62 In Greece, 16S rRNA metagenomic sequencing was used to assess the bacterial diversity within the digestive glands of diseased individuals. 62 Thirty moribund animals were collected in two different marine areas in the Aegean Sea. Sampling was carried out between February and April of 2020. Along with the presence of pathogenic strains of V. mediterranei, multiple bacterial genera were detected including Aliivibrio spp., Photobacterium spp., Pseudoalteromonas spp., Psychrilyobacter spp., and Mycoplasma spp., with the latter found even more abundantly.
Interestingly, in 8%–10% of the cases analyzed from Italy (2018–2019) and Croatia (2019), animals displayed different types of lesions, and the aforementioned common pathogens were not detected in their tissues. 80 In early August of 2019, in an area of Croatia where no mortality was detected (Seline), pen shells were negative for Mycobacterium and H. pinnae, using both PCR and microscopic examination. However, histopathology revealed the presence of extensive inflammatory nodules associated with brown cell hyperplasia in 40% of the samples (2/5 animals). Necrosis of the digestive gland was also recorded in a single individual, along with the presence of intraepithelial gram-negative bacteria in the digestive tract. In 60% of the animals (3/5), an unidentified ciliated protozoan infested the gills. 80 Negative molecular diagnostic (PCR) results were reported from Catalonia 25 for 1 moribund animal, 1 dead animal (although still with turgid flesh), and 3 animals in an advanced state of autolysis, thus excluding the presence of Mycobacterium, Haplosporidium, and V. mediterranei.
V. mediterranei Is Associated With P. nobilis Mortality Under Predisposing Conditions
The influence of pre-existing noninfectious factors on a bivalve’s microbial communities, leading to the establishment of disease conditions, is still not well understood. It is known that to cause disease, pathogens must infect and invade the host’s body and subsequently dominate its microbial community. 2 V. mediterranei (synonym name of Vibrio shiloi or Vibrio shilonii) has been found in various marine invertebrates.79,83,89,90 The type strain (CECT 621T) was originally isolated from marine sediments, 75 indicating that V. mediterranei is a cosmopolitan bacterial species.3,10,73,89 Virulence genes were found in association with temperature/salinity-stressed animals and include chitinases, proteases, and genes involved in an array of secretion and iron sequestration pathways. 10 The pathogenicity of V. mediterranei was first described in association with MMEs of the scleractinian coral Oculina patagonica, under the synonymous name of V. shiloi or V. shilonii.56–58,79 Interestingly, in the case of coral bleaching by V. shiloi in O. patagonica, the major effect of increasing temperature was reported to be the expression of virulence genes by the pathogen, 78 which enables the pathogen to adhere to a β-galactoside-containing receptor produced by zooxanthellae in the mucus on the surface of the coral host. 78
The first detection of V. mediterranei associated with MME in P. nobilis was reported during one of the early MMEs, which was registered in October 2016 along the southern Mediterranean coast of Alicante, Spain. This multidisciplinary study used various methods including histopathology, bacteriology, virology, parasitology, and PCR procedures. 77 A saccharose-positive bacterium was isolated in a thiosulfate citrate bile sucrose medium from organs and tissues of 2 affected specimens and was absent in a third apparently healthy individual. The analysis of the 16s rRNA gene sequence indicated the etiological agent was V. mediterranei. This information, together with experimental challenges of juvenile Manila clams (Ruditapes philippinarum) at 17°C and 24°C,76,77 was used as the basis for further testing Koch’s postulates in juvenile P. nobilis. 3 Similarly, in the Aegean Sea, the investigation of mortality events in P. nobilis populations conducted during the winter months along the Greek coastal zones revealed the first detection of V. mediterranei although concurrently with another Vibrio sp. including a member of the Vibrio splendidus clade. 59 In that study, apart from the presence of Vibrio spp., Mycobacterium sp. was detected in all examined individuals together with H. pinnae, which was present in 3 of 17 specimens studied.
Prado et al 73 recorded over 90% in cumulative mortality over a 19-month period, peaking in summer and early fall and coinciding with water temperatures above 25°C. This temperature effect was also observed in a challenge experiment in which P. nobilis individuals with a mean shell length of 24 cm were injected with a strain of V. mediterranei (IRTA18-108) and held in open flow tanks for 23 days. 3 At the start of the experiment, the water temperature was 18°C. Mortalities started at 22°C on day 6 after injection and sharply increased after water temperature rose above 24°C. Even though mortality rates were not correlated with the bacterial doses injected, pathogenicity of the strain used for challenge was confirmed in P. nobilis through PCR with specific primers that detect virulence factors.
Field samples of P. nobilis collected in Alfacs Bay in the Ebro Delta were also infected with V. mediterranei in different tissues (particularly muscle and kidney), with 60% of the individuals having positive PCR results in at least one tissue (adductor muscle, gonad, kidney, digestive gland, or branchia), without displaying any signs of disease. 3 Apart from the aforementioned challenge experiment, additional individuals held in captive conditions suffered mortalities. Lesions included necrosis of kidney and digestive tissues, which might be due to generalized stress and/or poor nutrition due to inappropriate dietary maintenance, leading to potentially reduced immune capacity and vulnerability to infection by V. mediterranei. With individuals subjected to a more balanced diet, lower and later rates of mortality were observed. 73 Yet, the pathogenicity of V. mediterranei in the context of other stressful events, such as previous infections by other pathogens (eg, H. pinnae or Mycobacterium sp.), remains to be properly investigated. In a field study carried out in 2020 in the Thermaikos Gulf on the remaining populations of P. nobilis, V. mediterranei was detected alongside other Vibrio spp. in moribund individuals (unpublished data).
Pen Shell Immune and Stress Responses Associated With the MMEs
The field of pathology is devoted to defining the causes of disease by describing associated changes in cells, tissues, and organs and the resulting clinical signs. 32 Cells actively interact with their environment, constantly adjusting their structure and function to changing demands and extracellular stressors.38,87 Cells can adapt as they face physiological stresses or adverse conditions, achieving a new homeostatic state and preserving viability and function by activating stress-related and immune genes.2,29
Bivalves possess an innate immune system composed of cellular and humoral immune components that are regulated by many immune-related genes.9,87 Expression of some immune genes influences genetic regulatory mechanisms, modifying cellular and organismal responses of ectotherms, such as bivalves. 34 A first attempt to morphologically describe the pen shell immune cells, using cytology and electron microscopy, was performed by Matozzo et al, 66 just before the spreading of the MMEs. Two types of hemocytes were described: granulocytes and hyalinocytes. Granulocytes were further subdivided as basophilic, acidophil, or neutrophilic, which are all capable of phagocytosis. Hemocytes can produce superoxide anion and acid and alkaline phosphatases. Recently, during MMEs, Bunet et al 17 reported a first insight into the genome of P. nobilis showing a large variety of genes related to immunity, ranging from the pattern-recognition receptors to effectors of the immune response and apoptosis, including responses to cellular damage. 95 Pattern-recognition receptors such as Toll-like receptors, peptidoglycan recognition receptors, glucan-binding proteins, and lectins were highly represented in the P. nobilis genome along with members of the Beclin-2 family, caspases, and inhibitors and activators of apoptosis.
A peculiar immune cell type reported in many bivalve species and highly represented in P. nobilis are the so-called brown cells. These fixed phagocytes are abundant in the hemolymph sinuses of connective tissue underlying the intestinal tract and the renal pericardial region. They aggregate in lesions, are found during H. pinnae and Mycobacterium infection in P. nobilis, and are capable of active diapedesis. They may resemble higher vertebrates’ dendritic cells or melanomacrophage centers from reptiles and fish, and they are filled with brown-/yellow-pigmented granules that contain lipofuscin and melanin. In bivalves, they contain lysozyme, glutathione reductase, and acid phosphatase. 31 When activated, they pass from an immature state into mature cells specialized for antigen capture with the initiation of lysosomal function. Specimens of P. nobilis infected with either bacterial or parasitic pathogens generally display hypertrophic and hyperplastic brown cells throughout the vesicular connective tissue in various states of activity and degeneration (Figs. 3, 4).
Studies on stress-response-related genes in the pen shell have identified genes encoding members of the cytochrome P450 family, heat shock proteins, sulfotransferases, and glutathione-transferase. Various triggering stimuli that upregulate these genes have been identified. Previous works showed that P. nobilis colonized by the invasive algae Lophocladia lallemandii as well as individuals affected by anthropogenic activities have increased levels of markers of oxidative stress.15,88 P. nobilis affected by H. pinnae showed a reduction of the antioxidant effectors catalase and superoxide dismutase, as compared to the healthy individuals, while sick individuals also had higher levels of malondialdehyde, an indicator of lipid peroxidation. 14 Other stress indicators like heat shock proteins, immune response pathways, apoptosis, and autophagy were investigated in few affected animals in Greece. 60 An analysis of both Hsp70/Hsp90 demonstrated that individuals coinfected by H. pinnae, Mycobacterium sp., and Vibrio sp. exhibited higher levels of transcripts encoding the stress proteins, indicating an increased cellular stress response in comparison with the individuals infected only with Mycobacterium and Vibrio. Regarding specific immune genes, transcriptional levels of the proinflammatory cytokine, IL-6 and TNFα, did not show any significant differences between individuals infected only with H. pinnae or the three pathogens together.
Considerations on Diagnostic Error
Histology remains a standard assessment tool for disease diagnosis in pathology, providing information on the state of the host tissues, the etiology of disease, the level of infection, and pathological alterations of affected tissues. In the invertebrate pathologists’ community, there is an increasing concern about the quality and veracity of histopathological findings published in peer-reviewed journals in the field.7,21,25,31 Animal pathology training programs encourage students to obtain rigorous, comprehensive education in histology, histopathology, and systemic pathology. The trained and experienced veterinary pathologist must know the studied animals in detail and have the ability to distinguish normal histologic variations from pathologic processes in examined tissues. Historically, histology has mostly been used for the identification of bivalve parasites, with focus on the species of commercial interest such as clams, oysters, and mussels and, to some extent, as an endpoint in toxicological studies, sometimes using sentinel species such as zebra mussels. On the other hand, even if there is an increased interest in publishing results about invertebrate pathology, limited expert reviewers are available with such a specific training. Consequently, inaccurate interpretations of microscopic observations are published in peer-reviewed scientific publications.
The first histological misinterpretation about P. nobilis was reported by Katsanevakis 53 where reported histopathologic findings of H. pinnae in the digestive gland are in fact photomicrographs of a female gonad follicle with evident regressing oocytes surrounded by brown cells and scattered hemocytes. This error is apparently a combination of the authors’ failure to recognize the normal tissue and the failure of the reviewers to reject the incorrect interpretations of the images and their associated findings. Recently Künili et al 55 also misinterpreted a normal tissue for Haplosporidium. Throughout the publication, the authors erroneously describe constitutive immune cells of a pen shell (brown cells) as “sporocysts enclosing more or less mature spores,” and apparent nuclei of digestive epithelia are incorrectly reported as plasmodia or a binucleate phase of the parasite. Furthermore, designated tissue types are hardly recognizable as such in the published low-quality images. These two examples of inaccurate and poorly presented histopathological data emphasize the need for augmentation of the knowledge on microanatomy and pathology of bivalve species (in particular, those without commercial interest) encountered by comparative pathologists. 7
The Role of Noninfectious Factors in MMEs
Factors like temperature, humidity, soil nutrients, and ocean chemistry can all have strong influences on the spatial distributions of pathogens. Studies of relevant noninfectious factors like water temperature and salinity have been reported. 65 These noninfectious factors also affect animals’ physiological processes, such as spawning and the subsequent recruitment of juveniles. An absence of recruitment has been noted previously among the Ebro Delta populations, 5 and investigations using P. nobilis-specific quantitative polymerase chain reaction detected very low levels of P. nobilis eggs/larvae in seawater samples collected in this region from August to September 2016.
Currently, surviving populations are found only in enclosed bays, including the Sea of Marmara and a few scattered lagoons. With such geographical limitations in distribution, recruitment bottlenecks can be a driver of reduced genetic diversity that ultimately may enhance the severity of MMEs regardless of the cause. The last known remaining P. nobilis population from Greece is located near the estuaries of the Spercheios River, Phthiotis in central Greece, in a habitat characterized as having lower salinity levels than the other populations in the same Gulf. This was the last population to survive the rising temperature regime in the spring of 2020 (unpublished data). Mortalities occurred in the populations of Maliakos, Phthiotis in central Greece, during the winter of 2020; however, not at the scale considered to be an MME. The temperature of these sampling sites ranged between 10°C and 15°C. Also, regarding the Greek coastline, despite the numerous diving efforts in different sites, no living individuals were detected in the Ionian Sea in 2020, except for the population originating from Laganas Bay in Zakynthos Island, Ionian Sea. 59 Data on pen shell MMEs and environmental conditions assembled by scientists and citizens across the Spanish Mediterranean coast, south of France, and some more isolated locations in the Tyrrhenian and Adriatic Seas, Crete, and Chypre indicated that disease expression was closely related to temperatures above 13.5°C and to a salinity range between 36.5% and 39.7%. 23 Although no pathological evaluation was conducted to determine the exact cause of mortality, the results indicated a clear influence of salinity and temperature in outbreak patterns. This suggests that the interaction between these factors at the local scale could influence the outcome of MMEs. Similarly, in the outer part of the Alfacs Bay (South Ebro Delta), MMEs in 2018 and 2019 did not occur until the months of July and August, when temperatures rose above 28°C (considerably higher than 13.5°C) and coincided with salinity increases above 35 g/L (36.5–38.5 g/L range). 74 In the case of Alfacs Bay, it is important to note that mortality rates (100% near the mouth, 43% in middle regions, and 13% in inner regions) were significantly associated with the summer salinity gradient across the bay (averages of 37.4–35.7 g/L) caused by freshwater agricultural discharges during the spring-summer season. H. pinnae was detected in individuals from all study zones of Afacs Bay, whereas Mycobacterium spp. was only found in the region near the mouth of the bay, featuring the highest salinity. Interestingly, neither H. pinnae nor Mycobacterium spp. were found in the small population in Fangar Bay (North Ebro Delta) subjected to summer salinity ranges of 30.5–33.5 g/L. 74 Also, importantly, a small contingent of young surviving individuals (3 years of age) were found in the region of Alfacs Bay subjected to MMEs where both H. pinnae and Mycobacterium were present. 74
In addition to those from the Ebro Delta, surviving pen shell populations have also been found in other confined or semiconfined environments featuring higher or lower salinities than the open sea.37,39,42,71,85 All these observations taken together stress the importance of environmental monitoring to assess the mortality risks to populations and to consider possible palliative management actions, such as controlled release of freshwater, where possible, to balance possible increases in salinity above 36.5 g/L during summer.
The Need for Standardized Diagnostic Protocols for Noble Pen Shell MMEs
Diagnostic procedures have a key role in disease control and infection prevention in the case of captive animals. Medical care, risk evaluation, management, and mitigation, as well as the development of government policies in a framework of One Health, all rely on diagnostic tools to guide further decisions.11,12,16,49,98 However, diagnosing any infectious disease in aquatic animals requires more than just the result of a laboratory-performed diagnostic tests to identify (known) pathogens. The diagnostic process usually requires the expertise of a veterinarian and/or trained para-veterinary pathologist, who could simultaneously evaluate all known factors, from clinical signs to environmental conditions, that could be associated with the presence of a pathogen to support their diagnosis. Moreover, this process is prone to error and potentially undermines the ability to identify the primary causative agent with certainty. The availability of specific and sensitive diagnostic tests is also of high importance to assist veterinary services in providing correct diagnoses. 99
In the case of bivalves, it is challenging to determine health status considering that there is an absence of observable clinical signs until the very late stages of a disease. Preliminary information on the health status of P. nobilis populations is usually based on field observations of animal behavior, describing animals generally as healthy or sick /moribund, the latter displaying a gaping shell. For both adults and juveniles, assigning a bivalve to one of these categories is based on the presence/absence of signs of adductor muscle weakening, such as gaping, and retraction of the mantle from the edge of the shell, to define it as sick.27,30,72 Moreover, during field examinations of bivalves, the valve closure speed is usually estimated by applying a gentle or stronger touch or by pushing the water in the direction of the valve opening; slower closing responses indicate a sick or moribund animal. However, studies performed on bivalves in Croatia and Italy showed that the valve closing speed may be unreliable (F. Carella, personal communication); thus, the determination of their health status based only on this observation could be incorrect. In fact, bivalves considered healthy in the field for the valve closure promptness during the direct or “hands-on” physical examination may show evident clinical signs such as generalized tissue edema, cysts, or areas or discoloration along with infection with pathogens when subjected to microscopic evaluation. This can be true even with the presence of clearly evident gross lesions (ie, fluid-filled cavities in the digestive gland and areas of discoloration associated with the presence of H. pinnae in apparently healthy animals) (Fig. 2). Variability in clinical signs can often be confusing and misleading due to the lack of knowledge of the physiology and pathology of the studied host animals, with consequent difficulty in the correct interpretations of clinical signs and animal behavior. 97 Such absence of a reliable clinical interpretation can be frustrating to researchers, leading to misinformed and possibly wrong decisions. Indeed, a prerequisite for formulating correct etiopathogenetic hypotheses and for the development of treatment or conservation strategies is that animal’s health status be correctly classified. 41
Gaps in the diagnostic processes applied during investigations of mass mortalities in P. nobilis are clearly emerging from the increasing number of reports (Table 1). There is no comprehensive and methodologically standardized description of the morphology, stage, and grade of the macroscopic and microscopic lesions associated with the presence of specific pathogens (Table 2). Therefore, it is not possible to link a pathogen to a specific response, lesion, or molecular pattern.
Overview on the analysis performed and the pathogens isolated in the Mediterranean Sea since the first description of mass mortality events (MMEs) in the noble pen shell (Pinna nobilis).

Abbreviations: DNA, deoxyribonucleic acid; rRNA, ribosomal ribonucleic acid; qPCR, quantitative polymerase chain reaction; PCR, polymerase chain reaction; TSA, tryptic soy agar; TCBS, thiosulfate citrate bile sucrose; FMM broth, Flexibacter maritimus; VRB, violet red bile; Endo, medium for enteric bacteria; TEM, transmission electron microscopy; ITS, internal transcribed spacer; Hsp65, heat shock protein 65; NGS, next-generation sequencing.
Diagnostic techniques to be used for noble pen shell (Pinna nobilis) mass mortality events.
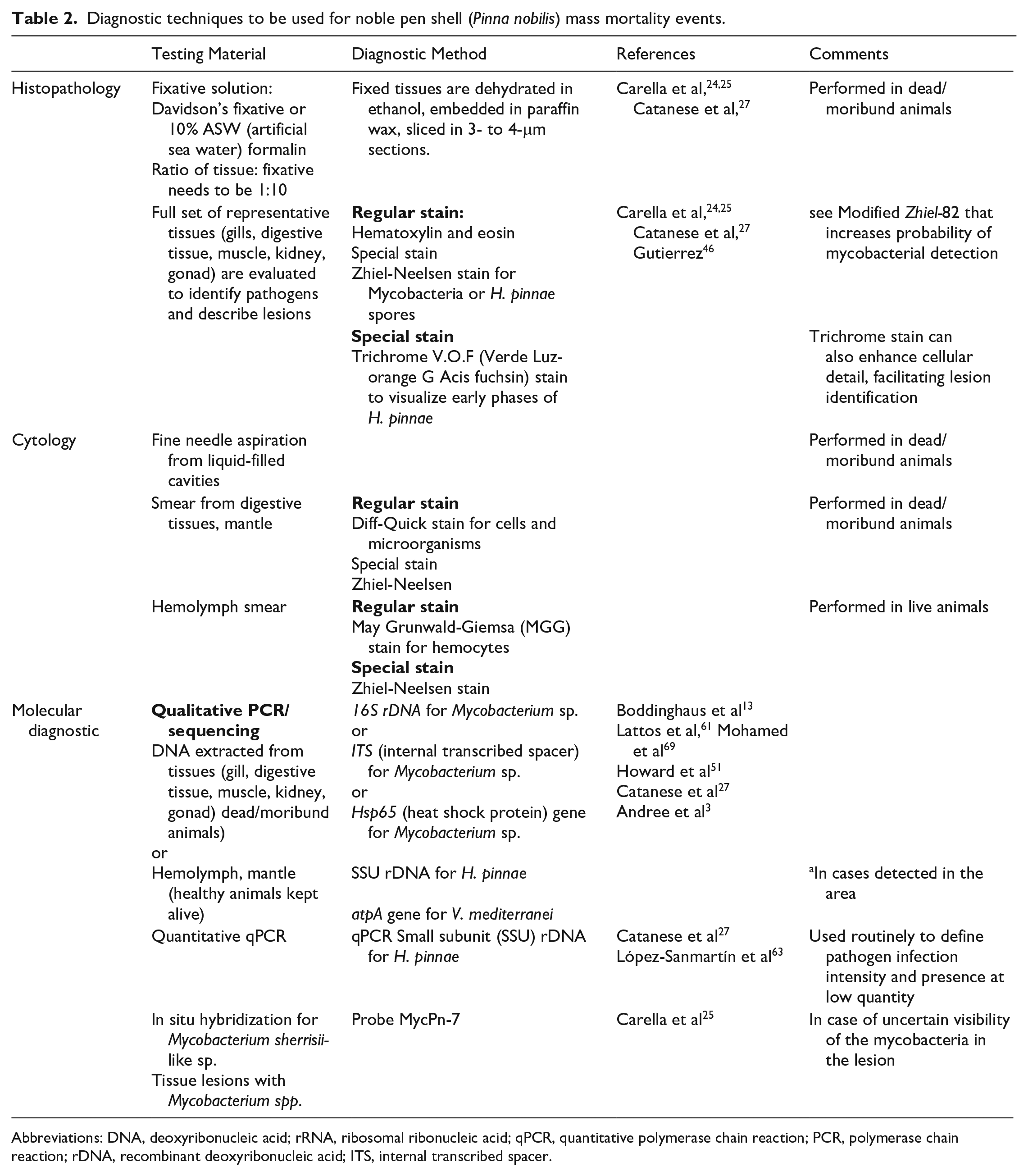
Abbreviations: DNA, deoxyribonucleic acid; rRNA, ribosomal ribonucleic acid; qPCR, quantitative polymerase chain reaction; PCR, polymerase chain reaction; rDNA, recombinant deoxyribonucleic acid; ITS, internal transcribed spacer.
Possibly due to the endangered status of this species, most studies utilize mostly mantle biopsies for molecular diagnostics to define the animal’s health status, which is itself a very limiting approach that can lead to an “overinterpretation” in the pathological diagnosis. 20 Taking into consideration that (1) multiple pathogens are potentially associated with MMEs and that (2) these pathogens show systemic distribution patterns in several organs and tissues during different phases of disease progression, it is obvious that a simplified mantle biopsy/PCR approach cannot work as the optimal diagnostic strategy. Moreover, molecular diagnostics alone do not give enough information to differentiate the occurrence of an infection from its associated disease. Different developmental stages of H. pinnae can elicit very different tissue reactions and lesions; information that cannot be derived from molecular analyses that are qualitative, based on the presence/absence of the pathogen, and possibly quantitative, in the case of quantitative PCR. 25 Furthermore, the absence of detailed histopathology descriptions from tissue lesions linked to the pathogens leads to confusing results.
Nonlethal Assessment
An integrated multifaceted approach needs to be used to collect, identify, and monitor host populations for pathogens and related lesions, along with the associated environmental data. For animals still alive in the field, a mantle biopsy is usually collected to define if an animal is infected by H. pinnae and is coupled with an assessment of the animal’s behavior. This type of sampling cannot be used alone and should be coupled with other nonlethal techniques that allow for parallel testing for other pathogens. For example, hemolymph can be used for microscopic and molecular assessments.
In the field, a mantle biopsy is generally performed while keeping the valves opened with the use a 0.5-cm-diameter wooden stick, placed in proximity of the hinge ligament. A tissue fragment of approximately 0.5 cm2 is taken using sterilized bite forceps and is fixed in absolute alcohol for subsequent molecular analysis. This method incites minimal damage to the sampled animal and is performed by many researchers.45,68,81
A reliable technique for sampling hemolymph is needed to complement mantle biopsies. In bivalves, hemolymph collection is done at the anterior or posterior adductor muscle, depending on the species anatomy and ease of access, and less frequently at the ventricle of the heart.40,50 Similar to blood in vertebrates, it has been demonstrated to be a suitable sample to detect infections in marine invertebrates.49,50,84 Hemolymph sampling in pen shells requires experienced manipulation, as a 21-gauge, 150- to 300-mm needle is inserted in the posterior adductor muscle, which is accessible from the upper part of the valve (F. Carella, personal communication). The needle is inserted into the muscle, visible through gaping valves, allowing collection of approximately 1.5 ml of fluid from a 50-cm-long pen shell. 66
Postmortem Assessment
When moribund animals are collected, a complete microscopic evaluation comprising a hemolymph smear and histologic assessment of all tissues with routine and special staining should be performed (Table 2). After the field examination, animals can be collected directly from their natural habitats and processed as soon as possible (maximum of 3–4 hours). Specimens can survive much better in a chilled environment at 5°C–15°C33,51 and should be placed in damp towels or seaweed and then bagged and transported in refrigerated containers. Care should be taken during collection to prevent damage and stress to specimens, which may affect histological interpretations. Pen shell lengths are measured from the tip of the right valve near the hinge to the longest point on the bill. Few of the published articles on marine bivalve mortality events describe gross lesions. The general appearance and overall body condition of each animal should be assessed as part of the routine examination or the investigation of population declines. External examination must consider shell abnormalities, presence of fouling organisms, parasites, gross abnormalities, predators, and physiologically related conditions as performed for other bivalves. 51 Before animal opening, hemolymph sampling is done as described previously. The pen shells are opened through severance of the adductor muscle and then examined for color, condition (fat, medium, or watery), macroparasites, and shell and tissue abnormalities. The body is removed from the shell by severing the adductor muscle as close to the left valve as possible.
All tissues (digestive tissues, gonad, kidney, gills, muscles) should be fixed in fresh fixative solution (buffered formalin or Davidson’s) in the right amount of volume ratio (1:10 tissue to fixative ratio). Poor fixation can make tissues useless for histological assessment. Formalin and Davidson’s are good general fixatives for bivalves because they have good penetration, prepare the tissue for histological stains, and give superior staining results with hematoxylin and eosin and other staining techniques. Some tissues require special care and handling, like the bivalve’s digestive gland that degrades very rapidly and need to be immediately fixed after sampling, as it is highly sensitive to weak fixation. 33
Development of DNA-based diagnostic methods currently offer a broad panel of probes and tests, but for the pathogens related to P. nobilis MMEs, the effort is still scarce. These methods have the theoretical advantages of high sensitivity and high specificity and possible rapid screening for the presence of a targeted pathogen. These methods should include highly sensitive quantitative PCR assays for multiple targets, ideally multiplexed using TaqMan (which are labeled with a quencher and a reporter) or similar fluorochrome probes. Molecular tools are valuable for establishing the presence of a pathogen, but they require care for their interpretation. This is crucial because many laboratories rely heavily on PCR methods, which provide information as to the presence or absence of a pathogen, although in some assays, the primers are not validated for diagnostic accuracy. Primers sets for a PCR assay that are overly specific may not detect novel emerging strains. Molecular tools can detect DNA sequences of the pathogen, which does not imply that the pathogen is visible in the host cell, although with the use of in situ hybridization techniques, its reliability can be determined.
A routine diagnostic approach should at least include a classification of the extent and severity and nature of the disease (ie, pathological process or lesions), based on a standardized quantitative evaluation of tissue lesions at the time of the diagnosis. The lesion severity should be classified using a standardized histological score, based on semi-quantitative estimates of the amount of tissue involved (in percentages) and on the type of lesions seen (inflammation, degeneration, necrosis, and so on) with consistent lesion nomenclature and definitions in relation to the pathogen detection. 80 Such an approach was developed before for salmonid whirling disease 4 by assigning an arbitrary numerical scale to identifiable discrete stages of disease development. However, such disease grading needs to be optimized for each etiological agent and related lesion and will therefore require careful preparation and validation when multiple concomitant etiologies are suspected. Using light microscopy, the lesions can be graded, and the extent of involvement recorded and related to Mycobacterium spp. and H. pinnae. The importance of consistent grading has been reviewed, and many grading schemes have been reported for bivalve pathology, including one instance in P. nobilis.80,95
The sections need to be analyzed blinded to molecular diagnostic results to minimize bias. In the case of Mycobacterium spp. and H. pinnae, the scorings are similar for both the pathogens and must consider the phase of development for H. pinnae and the site of infection. Special stains can facilitate their identification (Table 1). For H. pinnae, a score 1 indicates mild infection: the presence of few plasmodia in mantle or digestive tissue; score 2, mild to moderate infection: the parasite is present in the digestive gland and within digestive tubules in the presporulation phase within digestive epithelium (until 30% of the digestive tubules in a histological section are involved); score 3, marked infection: the parasite is present within digestive tubule epithelium (more than 30% of the digestive tubules filled). 80
For Mycobacterium spp., Ziehl-Neelsen slides are prepared and evaluated using similar criteria; mild infection (score 1): few immune cells filled with acid-fast bacteria in the connective tissue of gonad and digestive tissue capsules; mild to moderate infection (score 2): aggregates of immune cells filled with acid-fast bacteria throughout the connective tissue of the mantle as well as in the digestive gland, infiltrating tubules and hemolymph vessels; marked infection (score 3): aggregates of acid-fast bacteria within nodules of hemocytes throughout all the tissues. This approach, accompanied by a detailed description of the lesions per case, can give us a better and more complete overview of the pathogenesis leading to mortality.
In parallel, standardized molecular diagnoses should also consider the “quantity” of any identified pathogen(s), to better estimate their relative importance when a polymicrobial infection is suspected. Taken together, it has become evident that one of main reasons for our limited progress in understanding the epidemiology and pathobiology of MMEs affecting the noble pen shell is the lack of standardized diagnostic protocols that could be sensitively applied when multiple pathogens are occurring.
Future Directions
Reported investigations of MMEs and other research efforts addressing the spread of P. nobilis disease have used diverse methodological approaches (Table 1), primarily due to lack of standardized diagnostic tools, unknown/complex disease etiology, largely missing information on basic biology of healthy animals, and lack of communication between scientific groups. Furthermore, diagnostic techniques with inherent limitations were used, including clinical signs based on the closure of the valves, mantle biopsies, and reliance mostly on molecular information.7,21,25,31 Such an approach interferes with the comprehensive analysis and interpretation of the rapidly accumulating data about mass mortalities. Further problems in understanding disease arise due to the limited knowledge about P. nobilis physiology, anatomy, histology, nutrition, and microbiology. Taken together, our efforts to understand this disease are currently undermined by limitations in both quality and quantity of empirical knowledge. Apparently, we are facing a crisis of complexity, in which many potential explanatory factors and their interactions intertwine. Mortalities do not appear to be related to a single pathogen, and it is also possible that a primary agent common to all mortalities remains elusive and is yet to be described. It is therefore necessary to clarify the role of each suspected pathogen and their association to inflammatory lesions of each of the affected organs in observed mortalities, their relationships with environmental variables, routes of entry and dispersion (vectors), and multiple other epidemiological characteristics of this disease. As a way forward to further research, the following points should be considered.
Solve the Diagnostic Issue
We have incomplete knowledge of the problem, due in part to variable approaches to analyze the collected samples and the differences among focus areas of the studies that were carried out to date (Table 1). It is therefore necessary to standardize the pathogen-detection protocols, supported by the further development of reliable detection methodology. A manual of such an approach should be created in the context of P. nobilis MMEs involving all the experts of the sector. Moreover, we advise that collection of hemolymph from the adductor sinus can be safe for sampled P. nobilis and should be explored as a relatively noninvasive, and potentially useful, approach to the evaluation of pen shell health. Researchers studying MMEs of P. nobilis should be trained within a Ring test. “Ring” tests are competence tests coordinated among multiple collaborating laboratories, including enough replicates needed for statistical analysis of assay reproducibility (between laboratories) and repeatability (within a laboratory) and facilitates process improvement. 35 Finally, the analysis of environmental DNA raises interesting perspectives to better detect, characterize, and monitor known pathogens. These techniques will not replace but rather complement diagnostic tools currently used, including general methods such as histopathology, which helps to ensure detection of emerging diseases. Once such protocol is generally accepted by the scientific community, the next step will be to use it in a reanalysis of available samples from previous works, including quantitative molecular investigations for multiple suspected pathogens.
Establishing targeted surveillance programs with the participation of multiple stakeholders (ie, governments, nongovernmental agencies, academic partners, veterinary contributors, and the general public) is of high importance, especially due to the pan-Mediterranean distribution of the MMEs and the official IUCN status of P. nobilis as a critically endangered species. Developing highly specific and sensitive nonlethal diagnostic methods, combined with standardized investigation protocols, will be crucial in supporting national veterinary diagnostic laboratories or other diagnostic facilities in fulfilling the requirements of the European Union habitats directive and Barcelona Convention (Anex II). Furthermore, surveillance programs may assist in the discovery and characterization of resistant individuals or populations, allowing further studies on resistance mechanisms and the establishment of a hatchery for production of P. nobilis for the reintroduction and repopulation of previously affected regions. Improved communication and increased awareness of the problem should be a priority to all involved research groups and stakeholders. Timely sharing of research data and surveillance information between stakeholders will accelerate our understanding of the disease epidemiology and will certainly help in conservation efforts, for example, opening a shared data repository with online access to interested parties or setting up a forum/list of research groups, government and non-government agencies, or other entities to facilitate rapid exchange of information related to emerging/ongoing outbreaks. It is also foreseeable that several public/private partnerships can be founded at various levels (from local coastal communities to multinational consortia) with a task, among others, to encourage and support communication, dissemination of information, and conservation actions based on solid scientific evidence.
Database of Full Genome Sequences and Genomic Approach to Pathogenesis
The simplification of genomic methods for acquisition of whole genomes, transcriptomes, and metagenomes offers an opportunity to identify virulence factors, develop better molecular detection methods, gain knowledge of the molecular mechanisms of both the pathogen and the host immune response, and understand pathogens’ and hosts’ phylogeny. In this sense, it is recommendable to build a unique and dedicated database that includes genomes from Mycobacterium spp., Vibrio spp., and Haplosporidium isolates collected from various mortality episodes and geographical locations. Future effort should also be put into the isolation and culture of the pathogens most often involved in the MMEs to define their virulence and pathogenicity for the pen shell. The lack of bivalve molluscan cell lines has greatly limited studying the experimental transmission of pathogens and defining the host-pathogen interaction. Moreover, classic serological methods are not suitable for diagnostic purposes since mollusks do not produce antibodies.
Definition of Optimal Environmental Variables for P. nobilis to Better Understand the Disease Pathogenesis and to Find Areas for Animal Refugia
Ambient conditions can be a source of increasing stress on host populations involved in MMEs and can cause changes in pathogen virulence and infectivity, but the exact role or mechanisms of the noninfectious factors is currently unclear. To understand the epidemiological connections between various locations and differences in the appearance of the disease, it is necessary to collect information about environmental conditions preceding and during MMEs. Enlightening the association between the abiotic factors of the local environment and the occurrence of MMEs is critical to identify potential refugia that could be used for relocation and introduction of (previously confirmed) healthy specimens. Furthermore, additional studies are needed to investigate the effects of environmental pollution and of the contaminant concentrations present in these animals. Considering a typical habitat of fan mussels (eg, coastal embayment, both natural and man-made: bays, gulfs, and so on), increased urban activities and industrial discharge may incrementally increase pollutant concentrations in areas with slow water exchange, causing higher stress and lowering resistance to all pathogens.
Predisposing host factors allowing engagement of opportunistic pathogens need further investigations. Current information about major potential pathogens suspected in MMEs supports the idea of them being opportunistic in nature, rather than exclusively pathogens. Therefore, a remarkable lack of mortalities in other bivalve species that share habitat with P. nobilis suggests there may be an important intrinsic component of the host acting as a species-specific predisposing factor, making P. nobilis vulnerable to opportunistic infections. Although there is only limited information about normal physiology and ecology of P. nobilis, a possible differential factor could be the high incidence of microlacerations in P. nobilis mantle or gills caused by commensal crustaceans like Nepinnotheres pinnotheres and Pontonia pinnophylax, commonly found living inside almost all P. nobilis specimens, but not in other bivalve species sharing the habitat. The extreme size of P. nobilis sets it apart from other bivalves and enables the cavity between the valves to provide shelter for these supposedly commensal crustaceans. Such microlacerations may act as entry points for opportunistic pathogens, specifically predisposing P. nobilis to the development of diseases. 51
Conclusions
This work offers an objective review of the current status of knowledge about possible causes of recent MMEs of the noble pen shells in the Mediterranean basin. We agree that the disease etiology is complex, involving multiple pathogens causing coinfections, and is strongly related to environmental conditions conducive to poor disease outcomes. We strongly advocate that a standardized MME investigation protocol should be jointly developed and adopted by all parties involved in studies of the epidemiology and pathology of these MMEs. Furthermore, we ask all stakeholders that full consideration should be given to an open data-sharing approach, including access to archived samples, gathering molecular genetic information, and other physical or electronic data from both past and ongoing investigations. This will require a timely response on the part of all Mediterranean coastal nations and will require focused funding. Only by working together will we be able to keep ahead of the devastating consequences of this polymicrobial pathogenic and multifactorial syndrome affecting the noble pen shell in the Mediterranean Sea and possibly have a chance to stop their disappearance from native habitats.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
