Abstract
Histiocytic sarcoma is a tumor of the hematopoietic system considered to be derived from macrophages. Although rare in humans, it occurs frequently in mice. Histiocytic sarcoma can be a difficult tumor to diagnose due to its diverse cellular morphologies, growth patterns, and organ distributions. The varying morphology of histiocytic sarcomas makes it easy to confuse them with other types of neoplasia, including hepatic hemangiosarcoma, uterine schwannoma, leiomyosarcoma, uterine stromal cell tumor, intramedullary osteosarcoma, and myeloid leukemia. As such, immunohistochemistry (IHC) is often needed to differentiate histiocytic sarcomas from other common tumors in mice that they can morphologically mimic. The goal of this article is to present a broader perspective of the diverse cellular morphologies, growth patterns, organ distributions, and IHC labeling of histiocytic sarcomas encountered by the authors. This article describes these features in a set of 62 mouse histiocytic sarcomas, including the IHC characterization of the tumors using a panel of markers for the macrophage antigens F4/80, IBA1, MAC2, CD163, CD68, and lysozyme, and describes differentiating features of histiocytic sarcomas from other morphologically similar tumors. The genetic changes underlying the pathogenesis of histiocytic sarcoma in humans are beginning to be elucidated, but this is difficult due to its rarity. The higher prevalence of this tumor in mice provides opportunities to investigate mechanisms of its development and to test potential treatments.
Histiocytic sarcoma is a tumor of the hematopoietic system and is considered to be one of the tumor types derived from cells of the macrophage lineage. It is rare in humans, accounting for less than 1% of all hematopoietic tumors. 22 A study based on cases reported in the Surveillance, Epidemiology, and End Results (SEER) database between 2000 and 2014 found an overall incidence of 0.17 per 1,000,000 individuals. 24 However, the tumor is much more common in mice, and its incidence varies between strains and with sex, and increases with age. One of the earliest studies on histiocytic sarcoma in mice (then called histiocytic lymphoma) found incidences of 1%–2% in aged C57BL/6 mice and 0.3% in aged BALB/c mice. 16 In 1995, 2 studies examined the effect of caloric restriction on the lifetime incidence of neoplasia, one in C57BL/6 mice 7 and the other in B3C3F1 mice. 40 In mice fed ad libitum, the combined incidence in male and female C57BL/6 mice was 34.7%, representing 67.5% of all hematopoietic tumors in these animals, 7 while the study in B6C3F1 mice found an overall incidence of 7.2% (male and female combined), which accounted for 17% of all hematopoietic tumors in these animals. 40 However, another study that evaluated only female C57BL/6 mice found an incidence of only 12.6%. 26 The reason for the variation in incidence data is not clear, although substrain differences may be partially responsible.
Diagnosing histiocytic sarcoma can be difficult, as the tumor presents with myriad morphologies. When the morphology is predominantly round cells, the tumor can appear classically hematopoietic. Accordingly, it was termed “reticulum cell neoplasm Type A” in Dunn’s classic paper on mouse hematopoietic tumors 12 and “malignant lymphoma” by Frith et al 16 in 1980. However, in its more spindle cell form, the tumor does not have a typical hematopoietic morphology, and tumors that were diagnosed as endometrial cell sarcoma9,11 or malignant schwannoma 42 in the 1960s and 1970s are now believed to have actually been histiocytic sarcoma. 45 Subsequent to the initial designation of this tumor as malignant lymphoma, further investigation by Pattengale and Frith 33 led to its redesignation as histiocytic sarcoma in 1983. The advent of immunohistochemical (IHC) detection of cells of the macrophage/monocyte lineage has greatly facilitated the diagnosis of these tumors, and detection of these markers is frequently necessary when the tumor morphology mimics that of other tumor types.35,45
The International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (INHAND) guide to hematolymphoid lesions provides a guide to the general diagnostic features of histiocytic sarcoma. 46 The goal of this article is to present a broader perspective of the diverse cellular morphologies, growth patterns, organ distributions, and IHC labeling of histiocytic sarcomas encountered by the authors, based on a retrospective review of 62 cases. It is intended to help veterinary pathologists and other researchers who do not often encounter histiocytic sarcomas to be aware of this extensive variation in the appearance and properties of the tumors and, therefore, to be able to recognize the tumors as such when presented with them.
Materials and Methods
Case Material
Archived materials from the Division of Comparative Pathology at St. Jude Children’s Research Hospital in Memphis, Tennessee, were reviewed for the 62 cases described in this report. The frequency of features described below is reported as both the number and the percentage of these 62 cases. Tissues were collected from mice as part of studies approved by the St. Jude Institutional Animal Care and Use Committee. Of these mice, 33 (53%) were female, 20 (32%) were male, and the sex in 9 (16%) mice was unknown. The ages ranged from 4 to 24 months, with a mean age of 13.3 months and a median age of 11.5 months. The strain background was C57BL/6J for 61 mice and B6/129 for one mouse. The mice had various genetic manipulations present; 10 were wild type, 28 were null for exon 1β of the Cdkn2a gene (known as Arf KO), 13 were heterozygous for the Xaf1 gene and 2 were null for it, 5 had a loss of function mutation in Nras, 3 were heterozygous for the Pax5 gene, and 1 was heterozygous for Trp53. There were not enough mice present in each group of genotypes to allow for the analysis of genetic manipulations on the various features of histiocytic sarcoma described herein.
Histology and IHC Methods
Tissues submitted to the laboratory were routinely fixed for a minimum of 48 hours and a maximum of 2 weeks in 10% neutral-buffered formalin. Bone specimens were decalcified in 10% formic acid. Tissues were embedded in paraffin and sectioned at 4-µm thickness. Sections were mounted on slides and either stained with routine hematoxylin and eosin (HE) or used for IHC analysis. The antibodies used for IHC analysis are listed in Table 1. IHC for F4/80, MAC2, IBA1, CD68, CD163, lysozyme, and S100 were performed on most cases, only being omitted when tissue blocks had been depleted. The other antibodies listed were used as needed to rule out differential diagnoses. Antibodies were evaluated using appropriate internal and external positive and negative tissue controls.
Immunohistochemistry methods for antibodies used in the study.
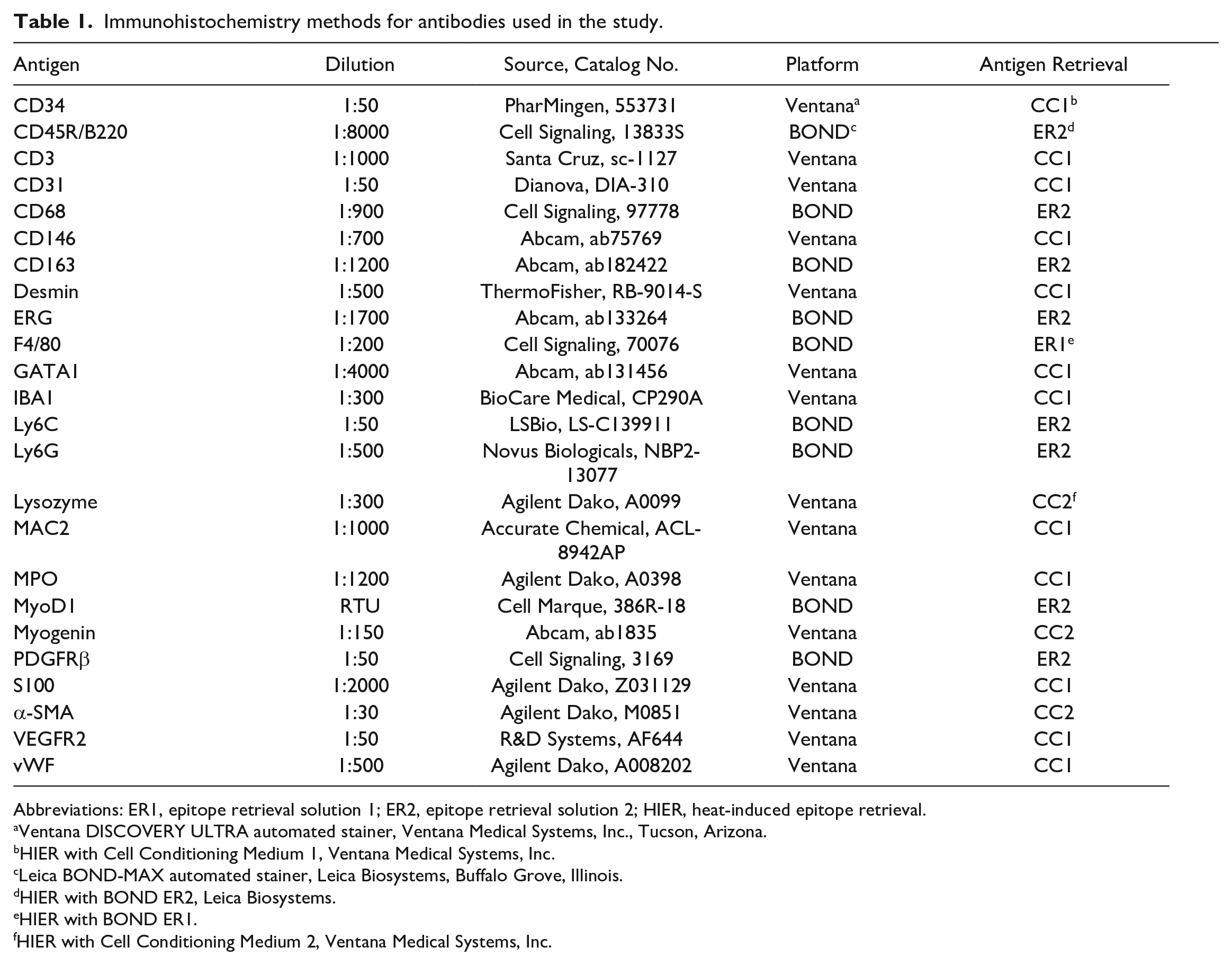
Abbreviations: ER1, epitope retrieval solution 1; ER2, epitope retrieval solution 2; HIER, heat-induced epitope retrieval.
Ventana DISCOVERY ULTRA automated stainer, Ventana Medical Systems, Inc., Tucson, Arizona.
HIER with Cell Conditioning Medium 1, Ventana Medical Systems, Inc.
Leica BOND-MAX automated stainer, Leica Biosystems, Buffalo Grove, Illinois.
HIER with BOND ER2, Leica Biosystems.
HIER with BOND ER1.
HIER with Cell Conditioning Medium 2, Ventana Medical Systems, Inc.
Results
Cellular Morphology
The morphology of histiocytic sarcoma cells can vary widely in terms of cell size and shape, nuclear size and shape, and the amount and character of the cytoplasm. In this study, 2 cellular morphologies were most common: small round cells and spindle cells. These 2 cell types often occurred within the same tumor. A single population of round cells was present in 9 (15%) cases, spindle cells in 3 (5%), and 50 (80%) cases had both types. The borders of the small round cell type were often indistinct (Fig. 1a). The cytoplasm varied in the degree of eosinophilia and in amount. The nuclei were small (1.5–2 red blood cell diameters) and round to oval with one or more small nucleoli (Fig. 1a, inset). The spindle cell type often had more distinct cell borders imparting an elongated shape to the cells (Fig. 1b). Their cytoplasm was brightly eosinophilic and variable in quantity, and their nuclei were oval to elongated. In both cell types, the cytoplasm was homogeneous, foamy, or vacuolated, and cellular pleomorphism was observed in 24 of 62 (39%) tumors (Fig. 1c, d). Multinucleated giant cells (Fig. 1c) were present in 13 of 62 (21%) histiocytic sarcomas and were focal or scattered throughout the tumor.
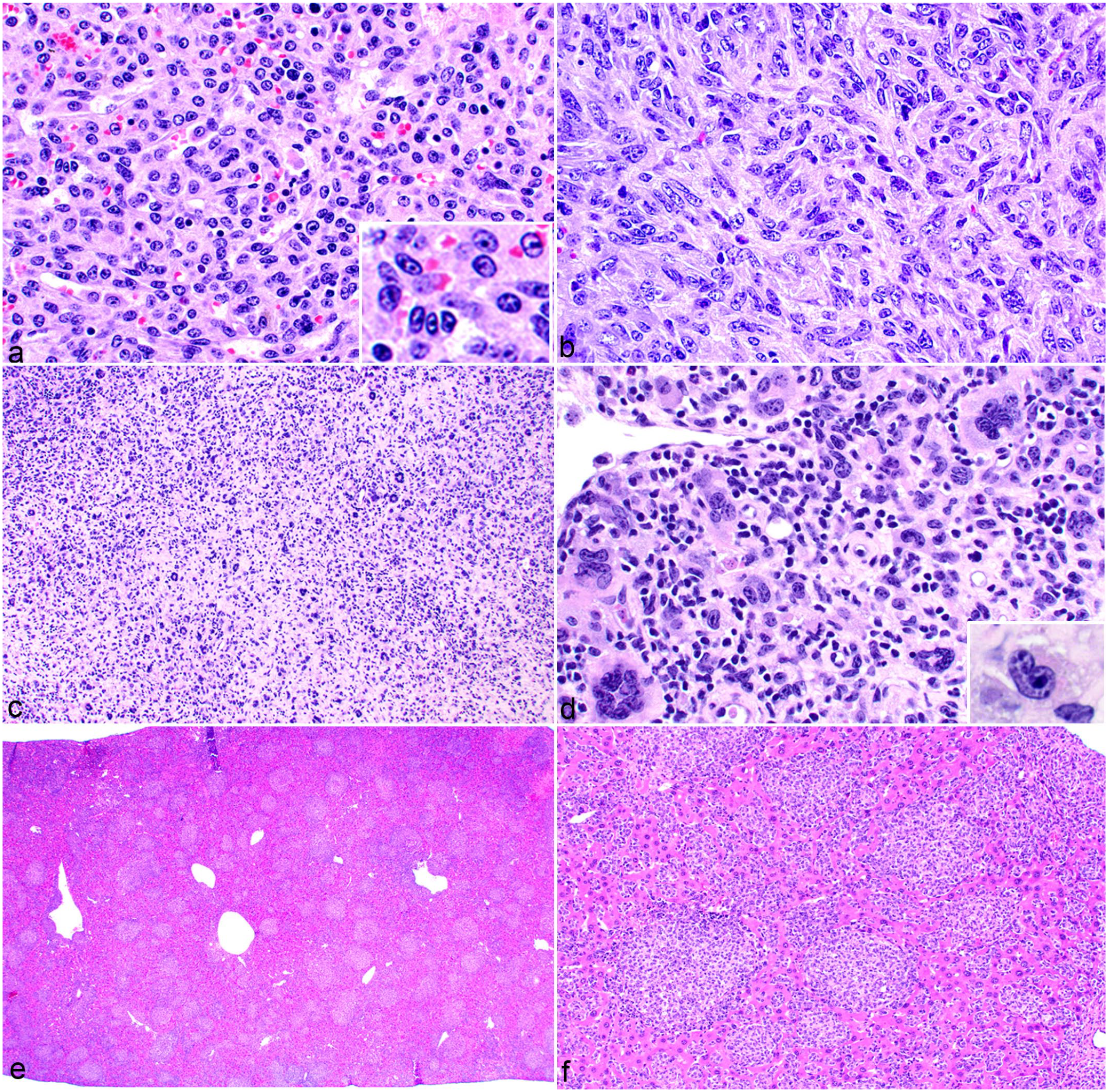
Cellular morphology and tissue distribution of histiocytic sarcoma in the mouse. Hematoxylin and eosin. (a) Liver. Histiocytic sarcoma with round cell morphology. Cells have small, round to oval nuclei with one or more nucleoli (inset) and a small to moderate amount of amphophilic to brightly eosinophilic cytoplasm. The distinctness of cell borders is variable. (b) Lymph node. Histiocytic sarcoma with spindle cell morphology and distinct cell borders. The cytoplasm is moderately eosinophilic and abundant. The nuclei are oval to elongated. (c, d) Lymph node. Histiocytic sarcoma with diffuse pleomorphism and multinucleated cells. (c) At low magnification, numerous large multinucleated cells are evident throughout the tumor. (d) At high magnification, the nuclear pleomorphism can be appreciated (the inset shows embryoid nuclear morphology). (e) Liver. Low magnification of a histiocytic sarcoma with a multinodular growth pattern. (f) High magnification of the tumor in (e).
Cellular Patterns
Within individual organs, the cells formed discrete nodules (32/62 tumors, 52%) or diffuse sheets (28/62 tumors, 45%), and occasionally, both patterns were present (2/62 tumors, 3%). These patterns were often apparent in the liver. A nodular pattern was often discernible at low magnification (Fig. 1e, f) and was not associated with any particular cellular morphology. The diffuse sheet growth pattern was often difficult to discern at low magnification in the liver (Fig. 2a), but the infiltrative nature of the cells within the sinusoids was evident at higher magnification (Fig. 2b). Tumor cells were associated with variable numbers of lymphocytes (35/62, 56%). B and/or T lymphocytes surrounded the nodules or were admixed among the sheets of tumor cells (Fig. 2c, d).
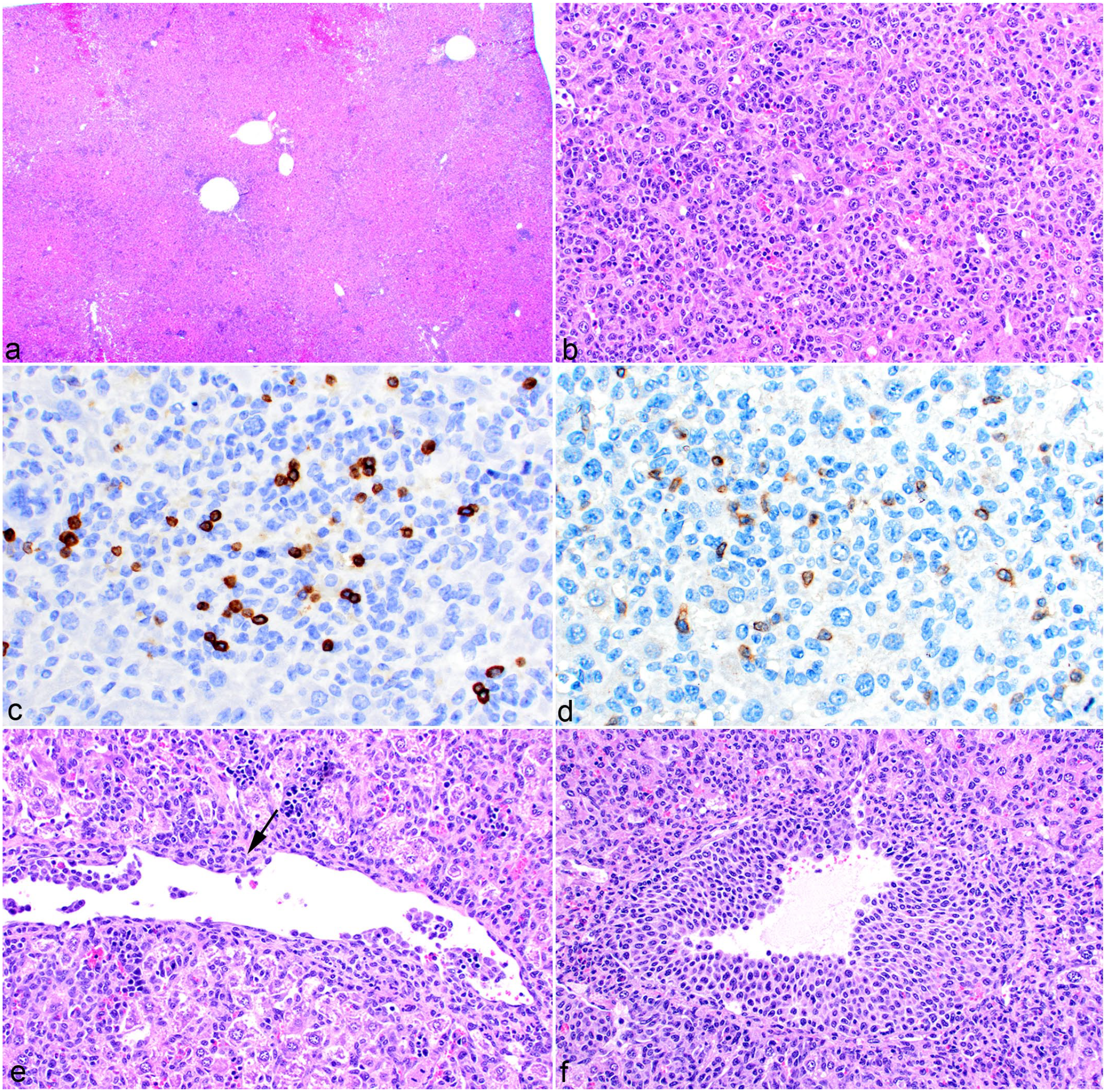
Histiocytic sarcoma, liver, mouse. (a) At low magnification, the presence of histiocytic sarcoma cells is not as evident as in Fig. 1e because of their diffuse sinusoidal growth pattern. Hematoxylin and eosin (HE). (b) At high magnification, the histiocytic sarcoma cells are readily apparent throughout the hepatic sinusoids. HE. (c) Variable numbers of B220-positive B lymphocytes and (d) CD3-positive T lymphocytes are scattered throughout histiocytic sarcoma tumors. (e) Histiocytic sarcoma cells line the luminal surface of the vascular endothelium and multifocally invade the vascular wall (arrow). HE. (f) Multiple layers of histiocytic sarcoma cells line the endothelium, almost causing vascular occlusion. HE.
Organ Distribution
Histiocytic sarcoma often occurred in multiple organs in the same mouse. The liver was the most frequently affected tissue, followed by the lung, bone marrow, spleen, lymph nodes, kidney, uterus, intestine, and the central nervous system (CNS; Table 2), although it should be noted that not all these tissues were submitted for all cases. The CNS was commonly involved when the histiocytic sarcoma was present in the bone marrow of the skull, which then infiltrated out of the cranial marrow into the meninges.
Organ distribution of histiocytic sarcoma.
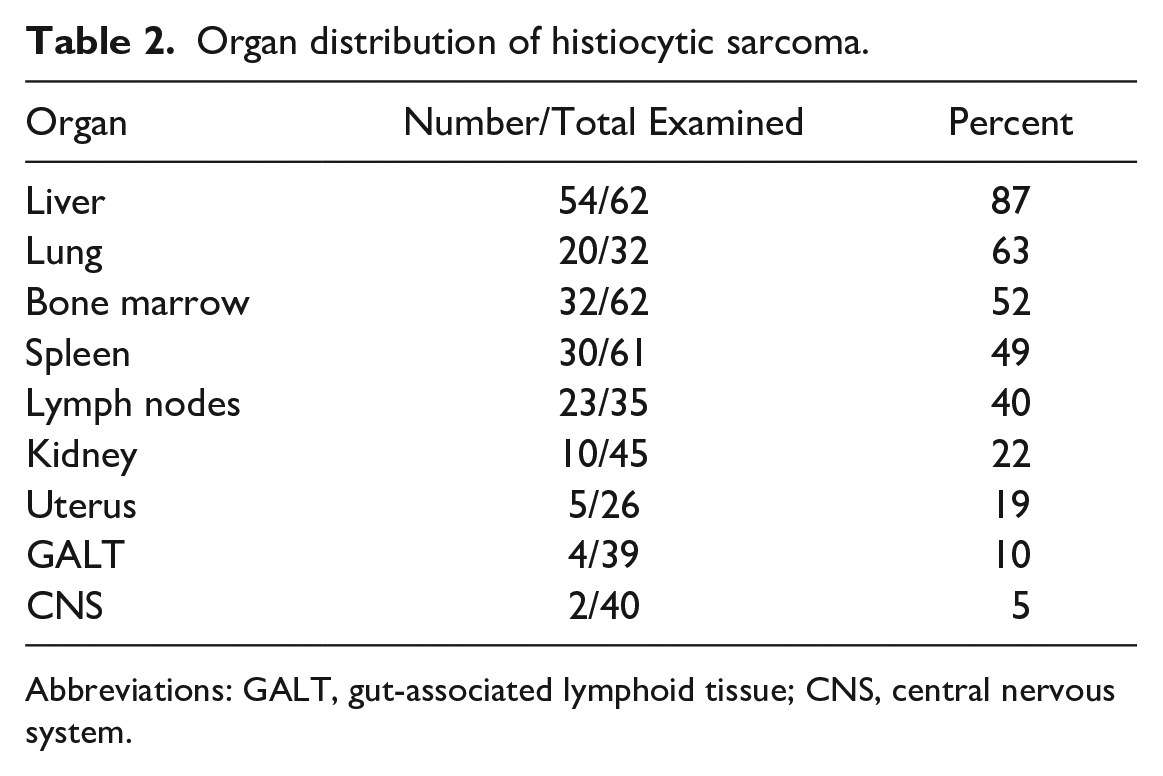
Abbreviations: GALT, gut-associated lymphoid tissue; CNS, central nervous system.
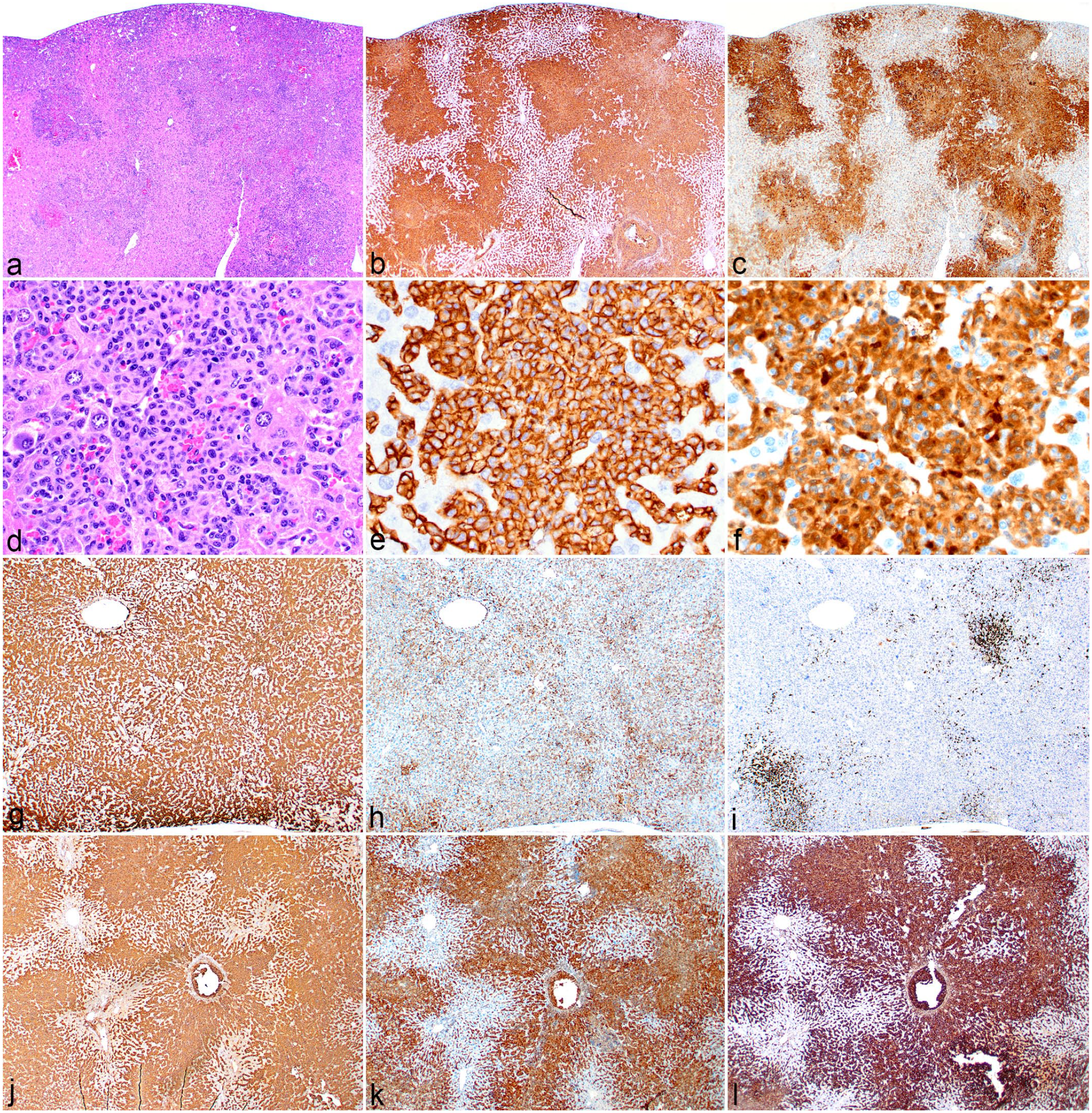
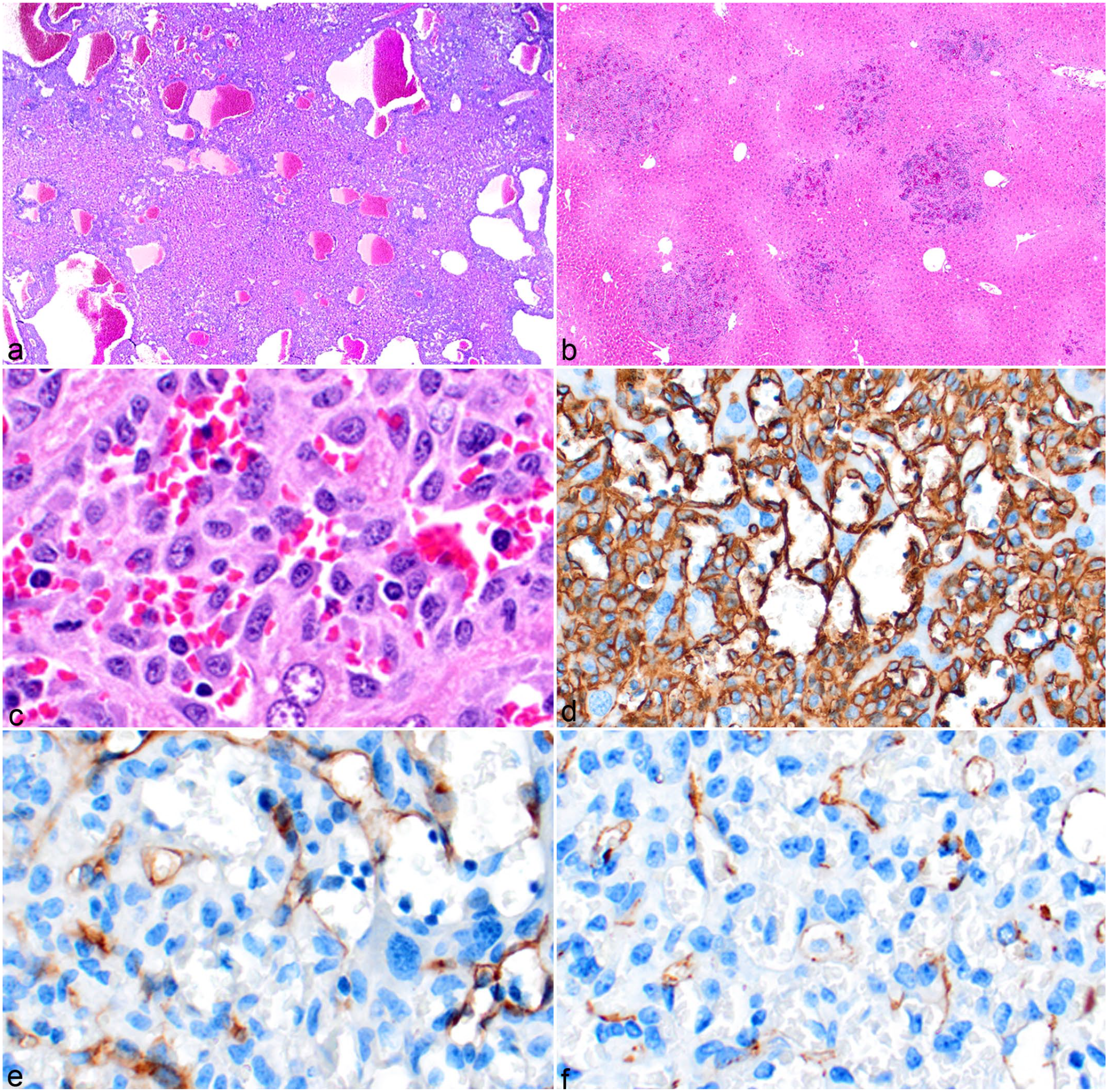
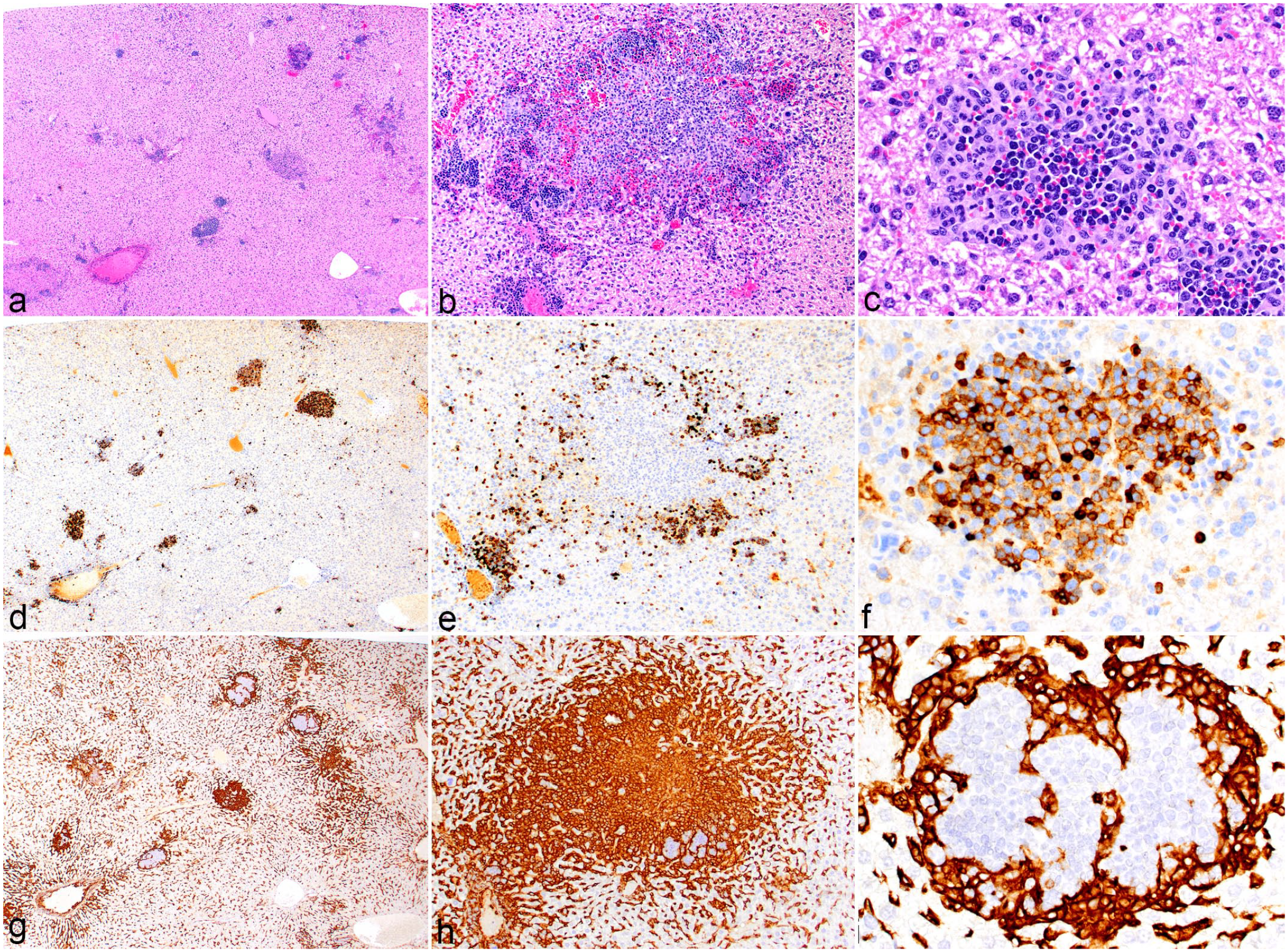
Examples of histiocytic sarcoma in the liver are shown in Figs. 1e, f and 2a, b. Features associated with histiocytic sarcoma in the liver included intravascular involvement with and without vascular occlusion (38/62 tumors, 61%), necrosis (33/62 tumors, 53%), hemorrhage (26/62 tumors, 42%), and extramedullary hematopoiesis (EMH; 46/62 tumors, 74%). Intravascular involvement consisted of tumor cells either lining the luminal surface of the endothelium or migrating through the vascular wall (Fig. 2e). The quantity of tumor cells lining the endothelium varied from a few cells to a thick band of cells occluding the vessel (Fig. 2f). Intravascular involvement was also present in the lung. Liver tumors were sometimes associated with necrosis, hemorrhage, and EMH. At low magnification, large areas of hemorrhage up to 1 mm in diameter were visible (Fig. 3a, b), whereas at higher magnification, necrotic areas ranging from small foci (approximately 50 µm in diameter) with EMH (Fig. 3c) to larger areas with hemorrhage (up to 0.5 mm) were apparent. Occasional ectatic blood vessels were evident, presumably resulting from compromised vascular flow (Fig. 3a). EMH not associated with necrosis and hemorrhage occurred in tumors to varying degrees and was associated with the tumor cells both individually and as colonies (Fig. 3d, e). Erythropoiesis was often prominent, but the EMH was usually more polymorphous, with megakaryocytes and granulocytes also present (Fig. 3f, g). Histiocytic sarcoma cell erythrophagocytosis was present in the liver and spleen of 9/62 tumors (15%). At low magnification, areas of the liver resembled dilated sinusoids with congestion (Fig. 4a); however, at higher magnification, it was apparent the intact red blood cells (RBCs) were within neoplastic histiocytes (Fig. 4b). Labeling of the histiocytes with F4/80 antibody demonstrated the RBCs within the tumor cells’ cytoplasm (Fig. 4c).
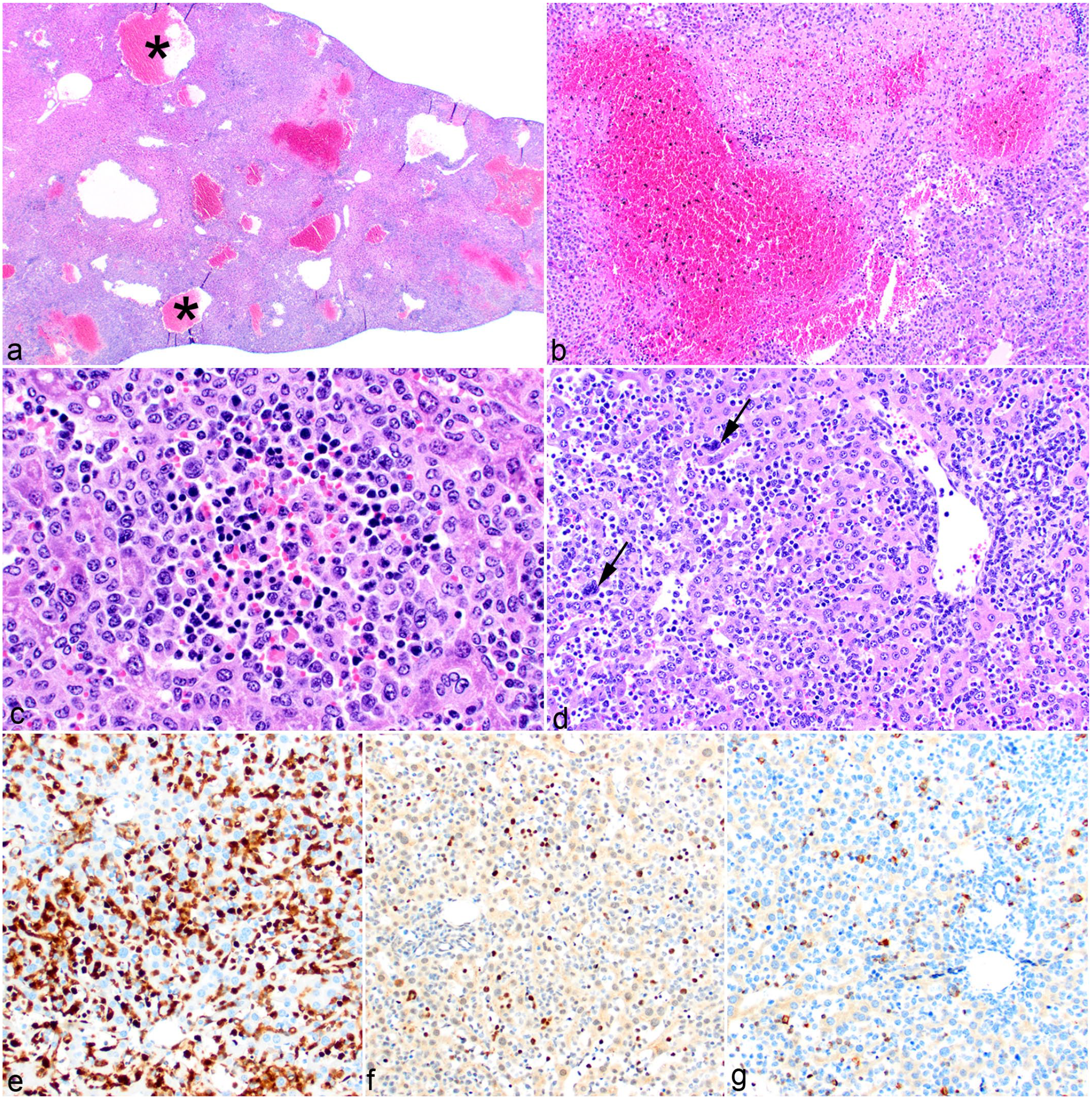
Histiocytic sarcoma, liver, mouse. (a) Areas of necrosis, hemorrhage, and vascular ectasia (asterisk) are present in the liver. Hematoxylin and eosin (HE). (b) Aggregates of histiocytic sarcoma cells have central areas of necrosis that are replaced by hemorrhage. HE. (c) Aggregates of extramedullary hematopoiesis (EMH) are interspersed with tumor cells. HE. (d) Histiocytic sarcoma cells are diffusely present throughout the sinusoids and are admixed with EMH. Megakaryocytes can be identified at this level (arrows). HE. (e) Labeling for MAC2 shows a diffuse distribution of histiocytic sarcoma cells with associated EMH illustrated in Fig. 2f, g. (f) GATA1 immunohistochemistry highlights the erythroid and megakaryocytic progenitors. (g) Myeloperoxidase labels myeloid precursors intermixed with tumor cells.
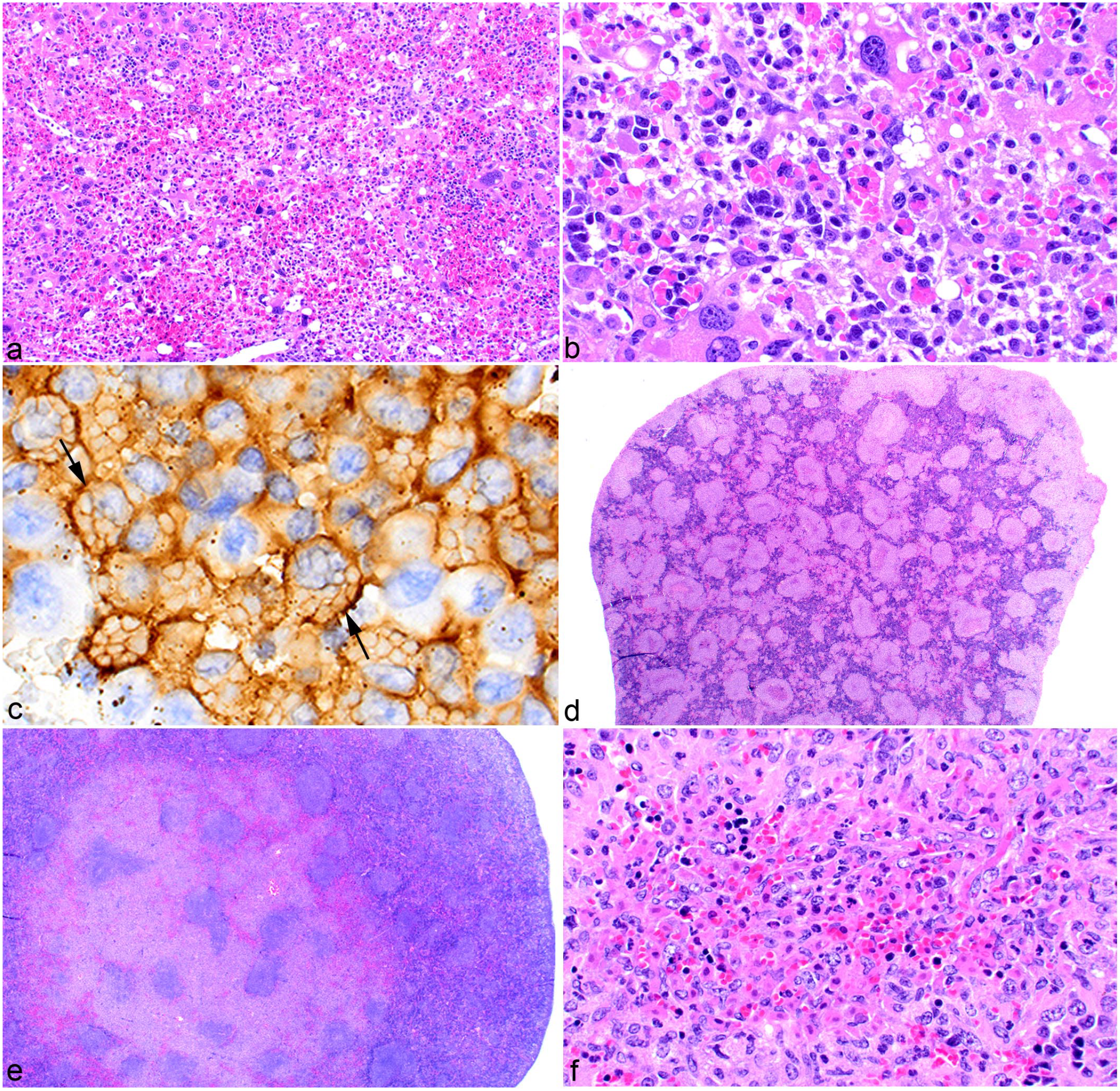
Histiocytic sarcoma in the mouse. (a) Liver. Histiocytic sarcoma with erythrophagocytosis. Distended blood-filled sinusoids are evident at low magnification. Hematoxylin and eosin (HE). (b) At higher magnification, individual histiocytic sarcoma cells with cytoplasmic, engulfed red blood cells (RBCs) can be seen. HE. (c) Immunohistochemistry confirms that the RBCs (arrows) are within the cytoplasm of the histiocytic sarcoma cells expressing F4/80. (d) Spleen. Coalescing nodules of histiocytic sarcoma cells affect the entire spleen. HE. (e) Spleen. Coalescing nodules of histiocytic sarcoma affect a localized area. HE. (f) Spleen. Nodules of histiocytic sarcoma nodules with central areas of necrosis and hemorrhage. HE.
In the spleen, histiocytic sarcoma cells formed focal, multifocal, or coalescing nodules. Multifocal or coalescing tumor nodules in some cases affected the entire spleen (Fig. 4d) and in ohters only a portion of it (Fig. 4e). The nodules sometimes had central areas of necrosis (Fig. 4f). As in the liver, cellular morphologies included both round and spindle cells. In the bone marrow, the tumor cells formed diffuse sheets (Fig. 5a) or nodules and were either round (Fig. 5b) or spindle (Fig. 5c).
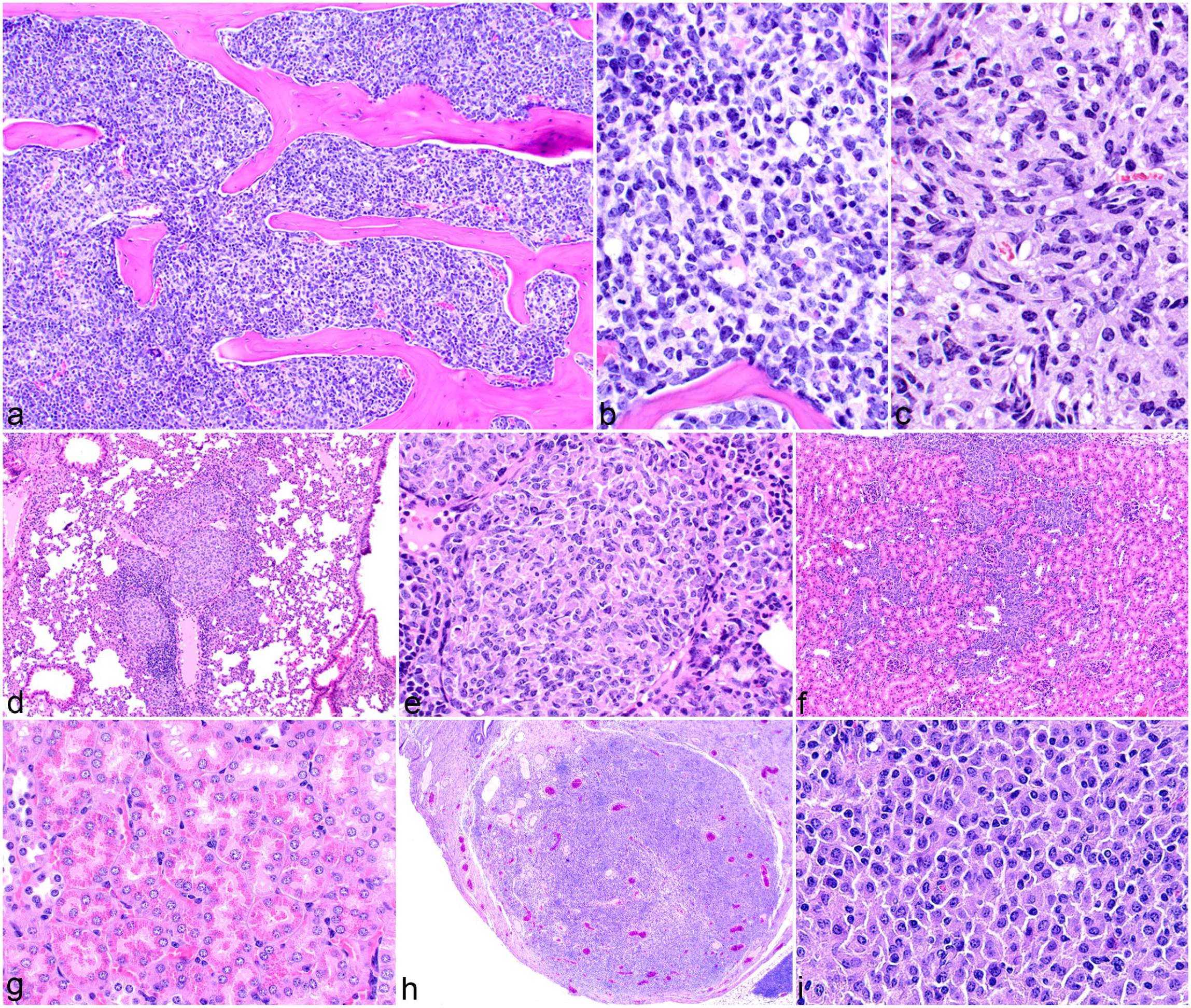
Histiocytic sarcoma in the bone marrow, lungs, kidney, and uterus; mouse. Hematoxylin and eosin (HE). (a) Histiocytic sarcoma cells replace normal bone marrow hematopoietic cells. (b) Round cell morphology of neoplastic cells in the bone marrow. (c) Spindle cell morphology of neoplastic cells in the bone marrow. (d, e) Lung. Nodules of histiocytic sarcoma cells located intravascularly and surrounding pulmonary blood vessels. (f, g) Kidney. (f) Histiocytic sarcoma accumulation around blood vessels and interstitially. (g) Hyaline droplets in the kidney proximal tubular epithelial cells. (h, i) Uterus. (h) Histiocytic sarcoma invading and replacing the normal parenchyma. (i) Neoplastic histiocytic sarcoma cells in the uterus have a round cell morphology.
Histiocytic sarcoma in the lung was located perivascular (Fig. 5d, e), intravascular, or lining the vascular endothelium, sometimes to the point of occlusion. Neoplastic cells had the variable morphologies as previously described. Other tissues not uncommonly affected were the kidney and uterus. In the kidney, the tumor cells were around blood vessels, in the interstitium, or in infrequently formed nodules (Fig. 5f). Intracytoplasmic eosinophilic hyaline granules, which were positive for lysozyme, were sometimes present in the proximal renal tubules (Fig. 5g). Tubular lysosomal granules were seen irrespective of renal tumor cell presence. In the uterus (Fig. 5h, i), tumor cells infiltrated and expanded the myometrium.
Immunohistochemistry
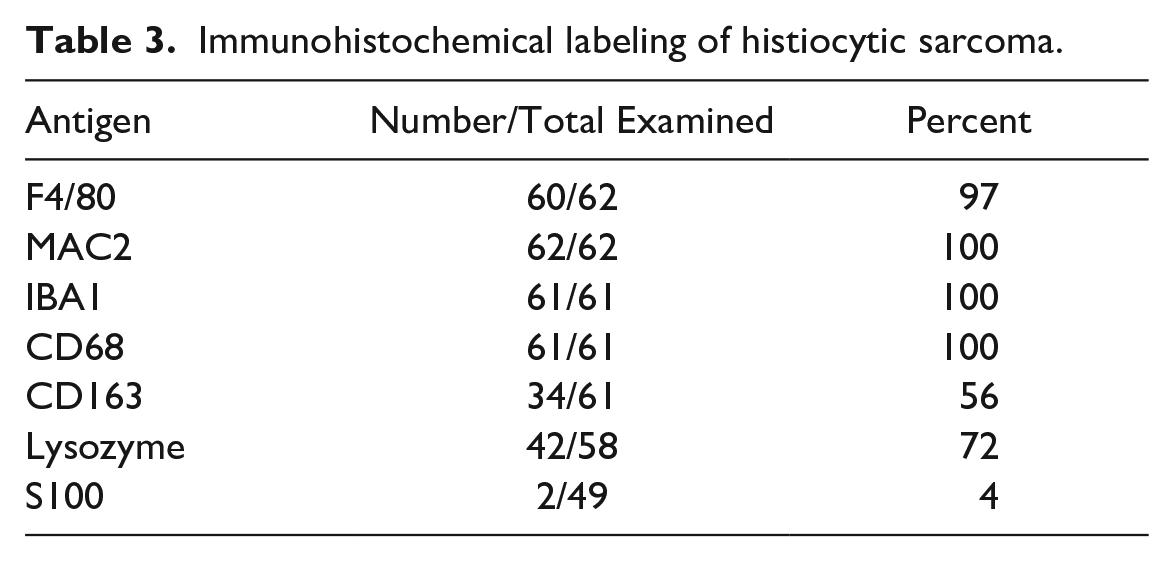
A summary of the IHC findings for the 62 cases is listed in Table 3. In all, 97% to 100% of the 62 tumors expressed F4/80, MAC2, IBA1, and CD68. In the literature, F4/80 and MAC2 are the 2 most used markers for diagnosing histiocytic sarcoma in the mouse. They generally have similar labeling patterns (Fig. 6a–f); however, in some tumors, labeling for 1 marker was stronger or weaker than for the other. This is demonstrated in Fig. 6g, h, in which labeling for F4/80 is strong and that for MAC2 is weaker, respectively. CD163 is the most common marker used to diagnose histiocytic sarcoma in humans. However, CD163 was expressed in only 34 of 61 (56%) mouse tumors. Therefore, this antigen may or may not be expressed in a mouse histiocytic sarcoma. Furthermore, the degree and intensity of expression of F4/80, MAC2, and CD163 may vary within the same histiocytic sarcoma (Fig. 6g–i), or they may be very similar (Fig. 6j–l). Other somewhat commonly used markers are IBA1, lysozyme, and CD68. Although IBA1 is often thought of as a marker for microglia, it can be considered a pan-macrophage marker and the anti-IBA1 antibody labeled all histiocytic sarcomas (Fig. 7a) in this study. IBA1 is expressed in both the cytoplasm and the nucleus of the cells. Lysozyme was used as a marker before more specific macrophage markers were discovered. In this study, only 42 of 58 (72%) tumors labeled for lysozyme. Lysozyme is located in the cytoplasm (Fig. 7b). CD68 is a glycoprotein that is a component of lysosomes and phagosomes and is, therefore, expressed in the cytoplasm. It is present in most monocytes and macrophages (Fig. 7c), but, like lysozyme, it is nonspecific and an anti-CD68 antibody will label any cells with a high lysosomal content.
Immunohistochemical labeling of histiocytic sarcoma.
Immunohistochemical (IHC) labeling of histiocytic sarcomas, liver, mouse. (a–f) Low and high magnification hematoxylin and eosin (HE) and IHC of a histiocytic sarcoma case. (a, d) Histiocytic sarcoma cells (HE) labeled as positive for (b, e) F4/80 and (c, f) MAC2. (g) Histiocytic sarcoma case showing strong labeling for F4/80, (h) moderate labeling for MAC2, and (i) foci of cells labeling for CD163; these may represent reactive Kupffer cell hyperplasia or a subset of tumor cells positive for CD163. (j–l) Histiocytic sarcoma case in which, in addition to positive labeling for (j) F4/80 and (k) MAC2, histiocytic sarcoma cells are positive for (l) CD163.
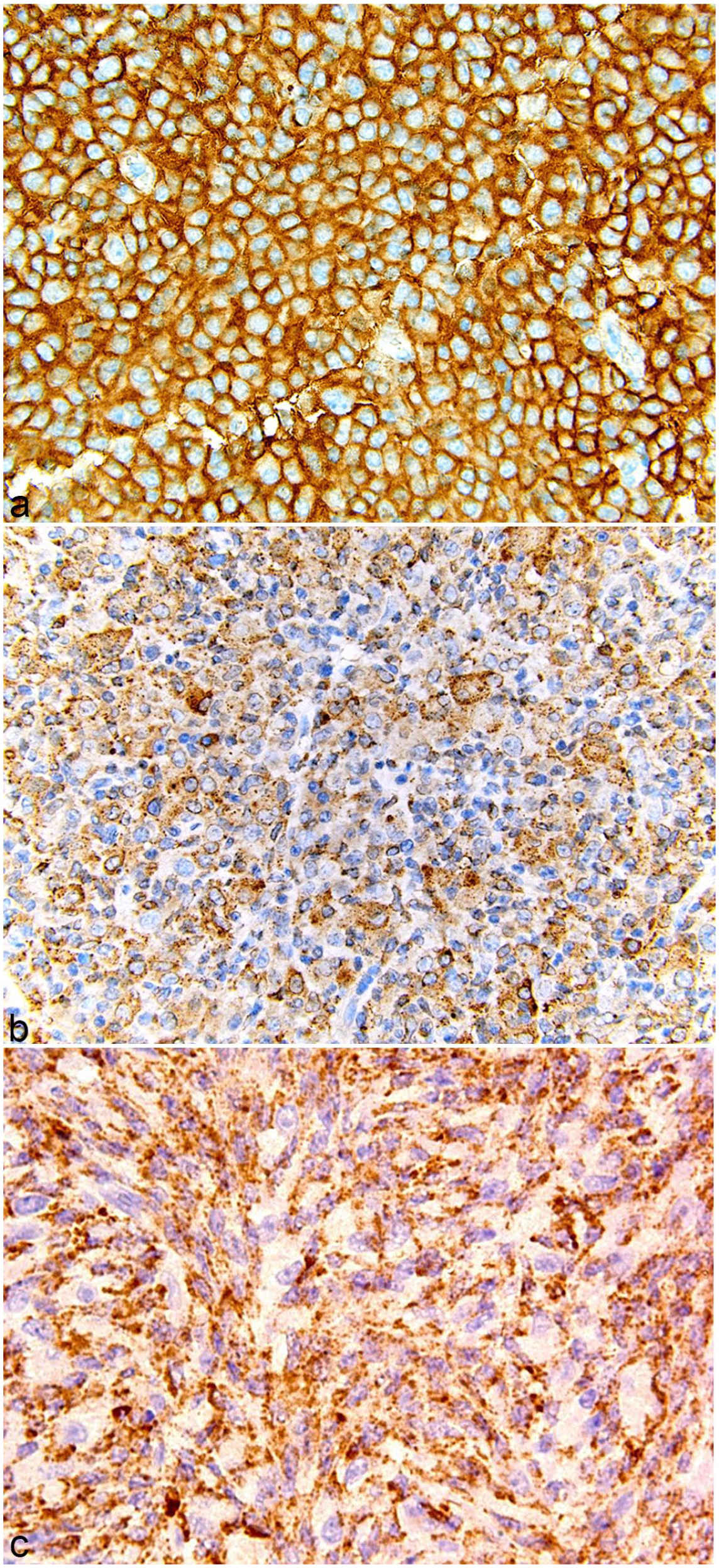
Other immunohistochemical markers for histiocytic sarcoma, mouse. (a) Uterus, positive for IBA1. (b) Lymph node, positive for lysozyme. (c) Mesentery, positive for CD68.
Common Morphologic Mimics
The varying morphology of histiocytic sarcomas makes it easy to confuse them with other types of neoplasia, including solid tumors and leukemia. Histiocytic sarcomas can sometimes have a morphology similar to that of hemangiosarcoma, especially when present in the liver. Of the 62 histiocytic sarcomas evaluated, 13 had large blood-filled spaces lined by a few layers of neoplastic cells, resembling peliosis (Fig. 8a), or had hypercellular foci with prominent blood-filled slits or channels (Fig. 8b) with features suggestive of a hemangiosarcoma (Fig. 8c). Such cases had prominent EMH, especially of the erythroid lineage. The tumor cells lining the blood-filled spaces labeled with IBA1 (Fig. 8d), F4/80, and MAC2, consistent with their being of histiocytic lineage. In hemangiosarcoma, the neoplastic endothelial cells will be labeled for VEGFR2, CD31, or other endothelial antigens. In our histiocytic sarcoma cases, sinusoidal endothelial cells labeled with VEGFR2 (Fig. 8e) and CD31 (Fig. 8f), but the tumor cells forming the blood-filled spaces did not label with antibody to these 2 markers. To rule out the possibility that the tumor cells were pericytes, the tumors were labeled with antibodies for the pericyte markers CD146, platelet-derived growth factor receptor-beta, and alpha-smooth muscle actin (α-SMA), which were negative (data not shown). While it should be noted that these 3 markers are not specific for pericytes, 49 the fact that the cells did not express any of the 3 markers strongly suggests the tumor cells are not pericytes.
Histiocytic sarcoma cell distribution in the liver mimicking the appearance of hemangiosarcoma, mouse. (a) Histiocytic sarcoma nodules forming large blood-filled spaces. Hematoxylin and eosin (HE). (b) Solid nodules with small blood-filled channels. HE. (c) At high magnification, it is evident that the blood-filled channels are not lined by endothelial cells but are instead loosely admixed with individual and clusters of histiocytic sarcoma cells. HE. (d) The histiocytic sarcoma cells are positive for IBA1, and hemangiosarcoma is ruled out as a differential by the negative labeling for (e) vascular endothelial growth factor receptor 2 and (f) CD31.
Histiocytic sarcoma was diagnosed in 5 of the 26 cases in which uterus was available, and 2 of these 5 had cellular features commonly associated with schwannomas. In the uterus, histiocytic sarcoma cells formed nuclear palisades with intervening eosinophilic cytoplasmic processes, resembling Verocay bodies (Fig. 9a), or arranged themselves around a central core to form rosettes (Fig. 9b). Uterine histiocytic sarcomas can replace a large portion of the normal tissue (Fig. 9c). Both of these examples labeled for IBA1 (Fig. 9d, e) and other macrophage markers listed in Table 3, but neither expressed S100 (Fig. 9f), thus confirming the diagnosis of histiocytic sarcoma and not schwannoma. Uterine histiocytic sarcomas may also need to be differentiated from uterine smooth muscle cell tumors and endometrial stromal cell tumors. Smooth muscle cell tumors will express desmin or α-SMA, whereas histiocytic sarcomas will be negative for desmin (Fig. 9g, h) and/or α-SMA, but will label for IBA1 (Fig. 9d, e) and other macrophage antigens. Another uterine tumor that histiocytic sarcoma may be confused with is an endometrial stromal tumor (Fig. 9i). These tumors uniquely express either desmin (Fig. 9j) or α-SMA and S100. These tumors may have scattered tumor-associated macrophages making it difficult to interpret IHC for macrophage markers. The case shown here has many macrophages, and labeling for IBA1 (Fig. 9k) highlights that the macrophages are between and distinct from the endometrial stromal tumor cells.
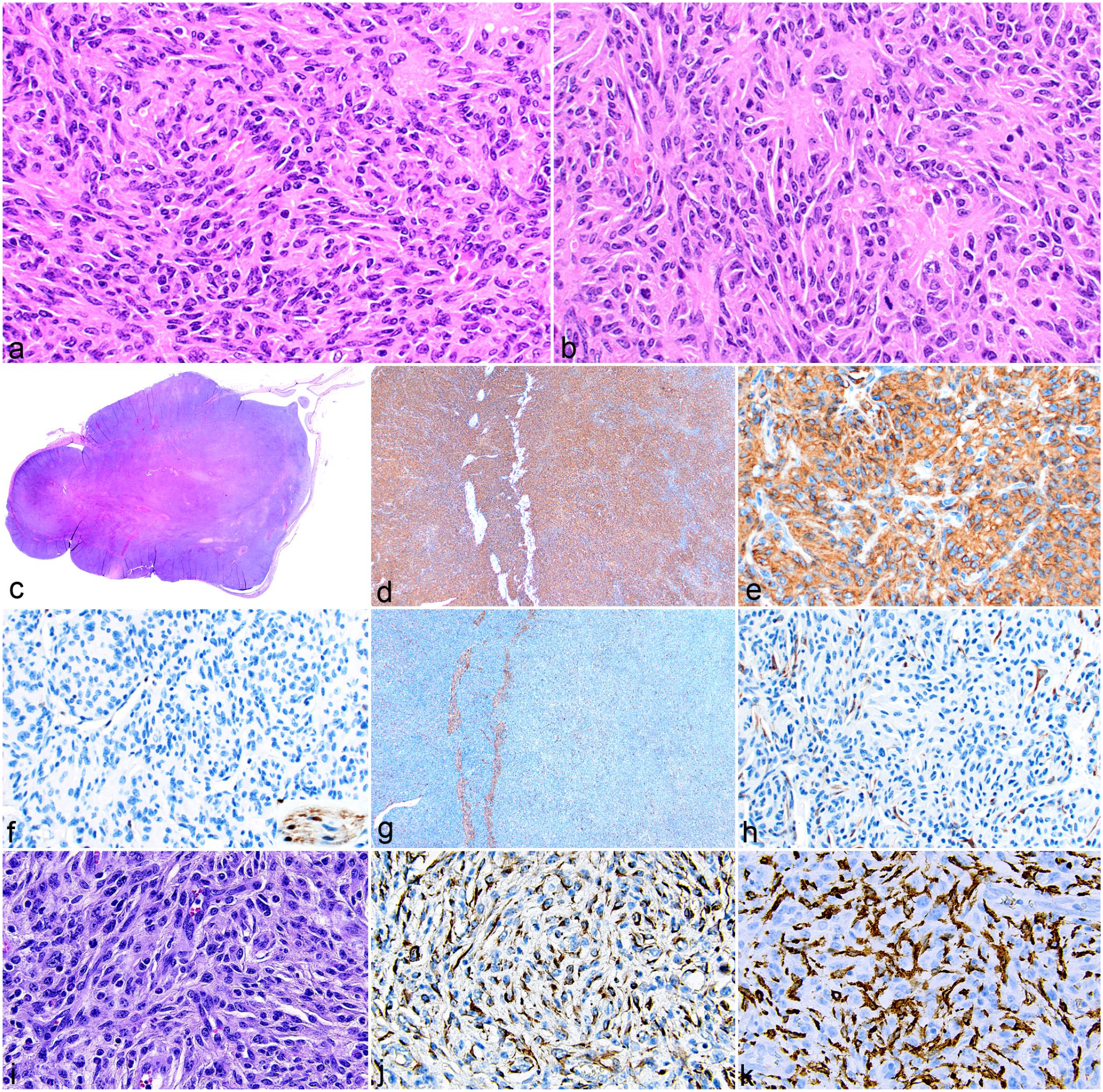
Histiocytic sarcoma and morphologic mimics in the mouse uterus. (a, b) Histiocytic sarcoma in the uterus with a spindle cell morphology mimicking schwannoma. Patterns resembling (a) Verocay bodies and (b) cells arranged around a central core, forming rosettes. Hematoxylin and eosin (HE). (c) Uterine tumor (HE) with differential diagnosis of schwannoma confirmed to be histiocytic sarcoma by (d, e) positive labeling for IBA1. Schwannoma was ruled out by (f) negative labeling for S100 (inset shows an internal positive control of nerve). Similarly, the diagnosis of leiomyosarcoma was ruled out by (g, h) negative labeling for desmin, with only residual uterine smooth muscle labeling as positive for desmin. (i) Uterine stromal cell tumor resembling a spindle cell histiocytic sarcoma (HE) is (j) positive for desmin and negative for (k) IBA1. Note the numerous macrophages residing in the tumor that label with IBA1.
The morphology of histiocytic sarcomas can be confused with that of other tumors commonly occurring in the bone and bone marrow. Histiocytic sarcomas were detected in the bone marrow of 32 of the 62 mice. Nine of the 32 histiocytic sarcomas involving the bone marrow were associated with trabecular bone proliferation (osteosclerosis, Fig. 10a). In these cases, the proliferations could be confused with osteosarcoma. The appearance of similar neoplastic cells in other tissues and IHC for F4/80 (Fig. 10b) can aid in differentiating histiocytic sarcomas from osteosarcomas. F4/80 will not label osteoblasts, 21 whereas some other macrophage markers such as MAC2 will. 3 Histiocytic sarcomas can also appear like monocytic leukemia and poorly differentiated myeloid leukemia when it is in the bone marrow. One of the 32 histiocytic sarcomas involving the bone marrow had nuclear features similar to features that are observed in these 2 leukemias. Monocyte and myeloid precursors and their leukemias in the mouse have the unique nuclear feature of being occasionally ring (donut)-shaped. The nuclei of mouse histiocytic sarcomas can also have this morphology. Therefore, IHC is necessary in some of these cases to differentiate histiocytic sarcoma and these leukemias. As there is no IHC marker that specifically labels monocytes but not macrophages, a diagnosis of monocytic leukemia (Fig. 10c) is based on the tumor cells being positive for CD68 (Fig. 10d) and/or Ly6C, and negative by IHC for markers of macrophage differentiation such as IBA1 (Fig. 10e), F4/80, and CD163. Myeloid leukemia is ruled out by the absence of the histiocytic sarcoma cells labeling for myeloid cell marker myeloperoxidase (Fig. 10f). Myeloid leukemia cells label for myeloperoxidase and Ly6G.
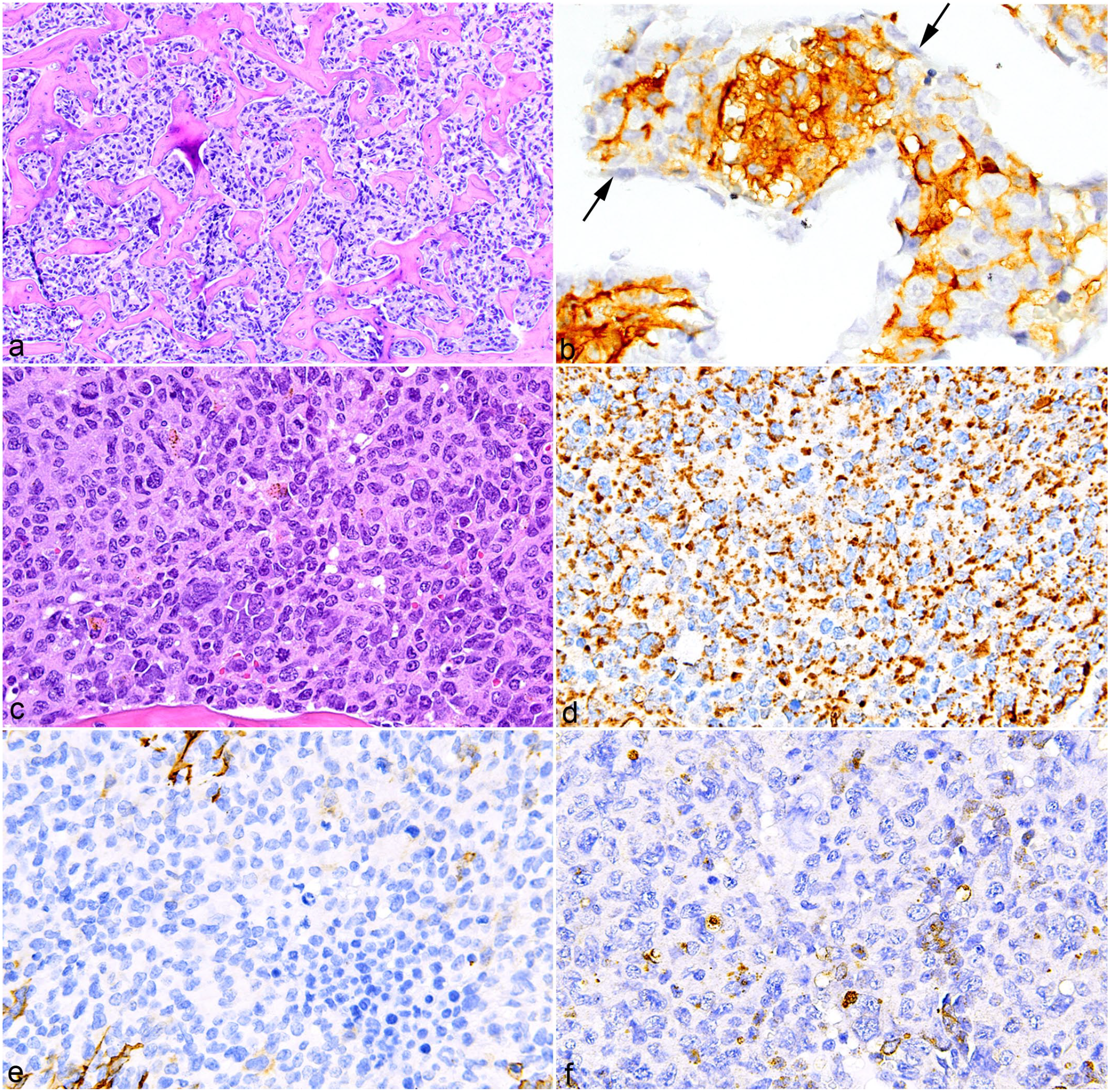
Bone marrow lesions associated with histiocytic sarcoma and monocytic leukemia mimicking histiocytic sarcoma in the bone marrow, mouse. (a) Osteosclerosis in the bone marrow associated with histiocytic sarcoma resembling osteosarcoma. Hematoxylin and eosin (HE). (b) Histiocytic sarcoma cells label for F4/80. Osteoblasts negative for F4/80 are indicated by arrows. (c) Monocytic leukemia that resembles the round cell type of histiocytic sarcoma. HE. (d) Monocytic leukemia positive for CD68 and negative for (e) IBA1 and (f) myeloperoxidase.
Combined Histiocytic Sarcoma and Lymphoma/Leukemia
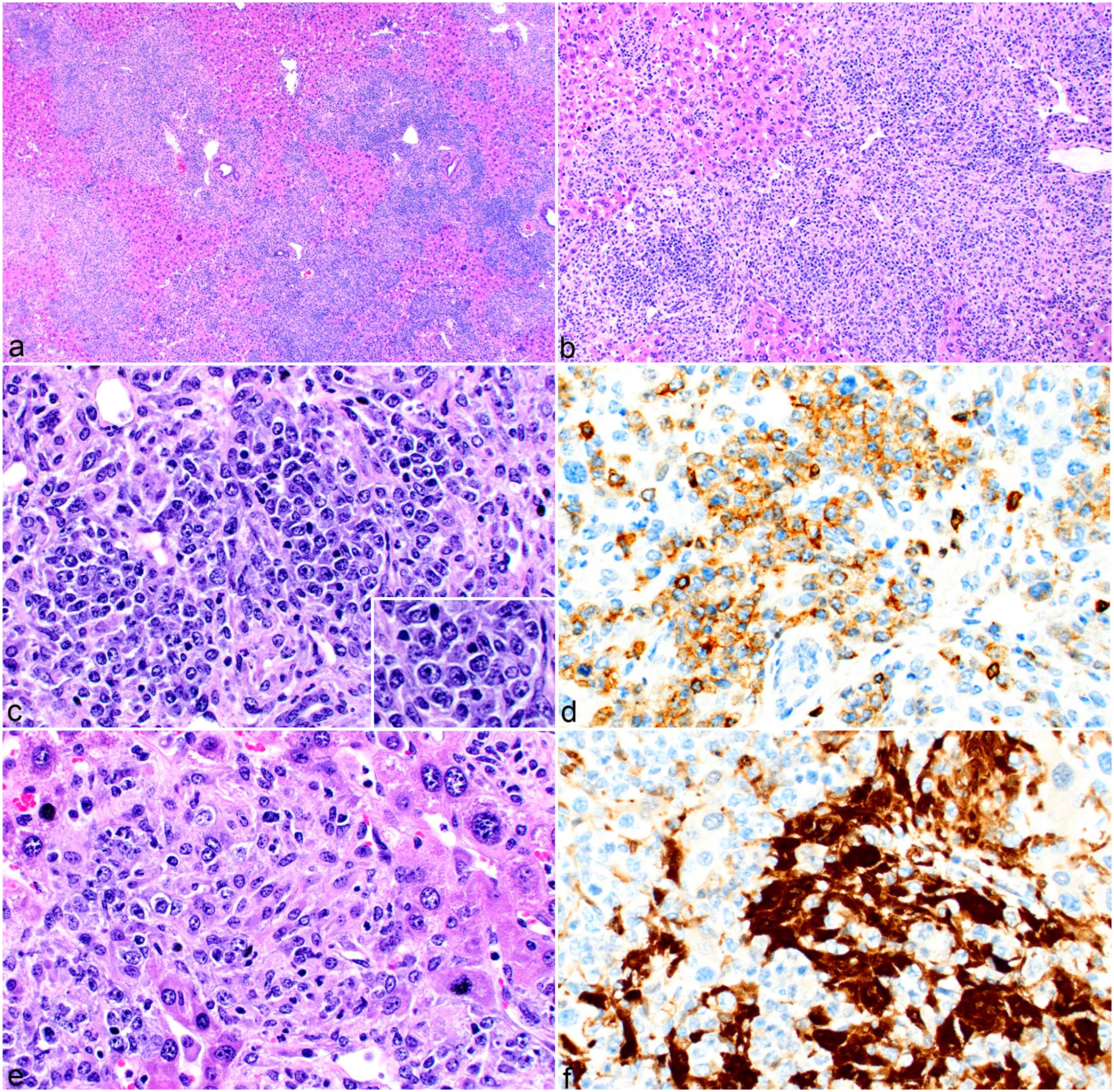
Mouse histiocytic sarcomas can be associated with normal, mature lymphocytes. However, there are cases in which the lymphocytes are not reactive inflammatory cells but are indeed a second population of neoplastic cells. In addition to histiocytic sarcomas, 8 of the 62 mice had a concurrent lymphoma/leukemia within the same mouse. Seven of the 8 lymphoma/leukemias were of B-cell lineage and 1 was of T-cell lineage. Two of the B-cell lymphomas had morphologic features and an immunophenotype consistent with B-cell acute lymphoblastic lymphoma (B-ALL). The T-cell lymphoma had morphologic features and an immunophenotype (TdT-negative) of a mature T cell. The histiocytic sarcoma and lymphoma populations were distributed as either separate nodules of homogeneous histiocytic sarcoma cells or lymphoid tumor cells, or as admixed nodules with various proportions of both neoplastic cells. Figure 11 shows B-ALL and a histiocytic sarcoma in the same liver. At low magnification, nodules of infiltrating cells with varying morphologies are evident (Fig. 11a). The 2 tumor cell populations formed homogeneous nodules of either histiocytic sarcoma or lymphoma. In other instances, the 2 populations appeared as diffusely scattered, admixed cellular aggregates or as mixed nodules. The mixed nodules commonly had a central area of one population surrounded by a border of the other population, that is, a core of histiocytic sarcoma cells surrounded by B-ALL cells (Fig. 11b, e, h) or a core of B-ALL cells surrounded by histiocytic sarcoma cells (Fig. 11c, f, i). Similar patterns were seen with a mixed T-cell lymphoma and histiocytic sarcoma (Fig. 12a–f).
Combination of histiocytic sarcoma and B-cell acute lymphoblastic leukemia (B-ALL), liver, mouse. (a) At low magnification, the liver contains mixed foci of neoplastic cells with different morphologies. Hematoxylin and eosin (HE). (b) Nodule with a central area of histiocytic sarcoma cells surrounded by small foci of B-ALL cells. HE. (c) Nodule with a central area of B-ALL cells surrounded by histiocytic cells. HE. (d) B220 labeling of the B-ALL subset of neoplastic cells. (e) B220 labeling of nodule depicted in (b), B220-positive B-ALL cells surround a focus of negative histiocytic sarcoma cells. (f) B220 labeling of nodule depicted in (c), central core of B-ALL cells surrounded by negative histiocytic sarcoma cells. (g) Low magnification F4/80 labeling of the histiocytic sarcoma subset of neoplastic cells. (h) Positive F4/80 labeling of core of nodule depicted in (b, e), surrounded by negative B-ALL cells. (i) F4/80 labeling of nodule depicted in (c, f), where positive histiocytic sarcoma cells surround a core of negative B-ALL cells.
Combination of histiocytic sarcoma and T-cell lymphoma/leukemia, liver, mouse. (a, b) There are coalescing nodules of neoplastic cells with different morphologies that are admixed with each other. Hematoxylin and eosin (HE). (c) Foci of predominantly T cell lymphoma/leukemia (with inset, HE) label for (d) CD3. (e) Foci of predominantly histiocytic sarcoma cells (HE) label for f) MAC2.
Discussion
The diagnosis of histiocytic sarcoma in humans is guided by the World Health Organization (WHO) Classification of Tumors of Haematopoietic and Lymphoid Tissues, 34 which classifies histiocytic sarcoma under the category of histiocytic and dendritic cell neoplasms. Histiocytic sarcoma is the only tumor of macrophage origin in this classification. The dendritic cell tumors include Langerhans cell histiocytosis, Langerhans cell sarcoma, indeterminate dendritic cell tumor, interdigitating dendritic cell sarcoma, follicular dendritic cell sarcoma, fibroblastic reticular cell tumor, disseminated juvenile xantho-granuloma, and Erdheim-Chester disease. The numerous types of dendritic cell tumors have not yet been described in the mouse. This is partly because of the paucity of suitable cell markers in the mouse, that are specific for macrophages versus dendritic cells and are specific for dendritic cell subsets.
Because of its many morphologic presentations, histiocytic sarcoma in the mouse can be problematic to diagnose. It must be kept in mind as a differential diagnosis for small round cell, spindle cell, and pleomorphic tumors, especially when present in the more common locations of the liver, uterus, spleen, and bone marrow. IHC may be necessary to definitively diagnose this tumor. F4/80, MAC2, IBA1, and CD68 were expressed in almost all tumors examined. Some considerations should be kept in mind when using these markers. Depending on the density and distribution of the cells, one marker may lend itself better to interpretation than the others. For example, the distribution of F4/80 labeling is such that 80% is in the membrane and 20% in the cytoplasm. Consequently, in tumor cells with long processes, the F4/80 labeling will be interwoven among normal resident cells, making the labeling appear more diffuse. However, MAC2 is predominately cytoplasmic, so the labeling for that marker tends to be more localized to the body of histiocytic sarcoma cells, enabling better differentiation between tumor cells and resident nontumor cells. Labeling for lysozyme must be interpreted with caution because other hematopoietic cells, some epithelial cells, melanocytes, and tumors thereof will also express lysozyme. In this study, lysozyme was variably expressed in histiocytic sarcoma; this may be associated with lysozyme being expressed only in activated macrophages. Lysozyme along with CD68 and alpha-1 antitrypsin were historically used as markers for histiocytic sarcoma, but they are less specific and are expressed in other tumors featuring high numbers of cytoplasmic lysosomes, such as melanoma. Similarly, MAC2 is expressed in a highly specific pattern on numerous types of epithelial cells; therefore, it is not specific for histiocytic cells. 35 CD163 is the most specific histiocytic marker, and therefore, the anti-CD163 antibody is the one most often used for diagnosis in human patients.27,43 However, as shown here, CD163 is inconsistently expressed by mouse histiocytic sarcoma; therefore, the present authors more commonly use F4/80 and IBA1 as markers for histiocytic sarcoma.
This tumor may similarly be difficult to diagnose in humans, especially given its rarity. In its more round cell form, it can be confused with anaplastic large-cell lymphoma and diffuse large B-cell lymphoma, as shown in a study in which previously diagnosed cases of histiocytic sarcoma were subsequently identified as different forms of lymphoid neoplasia.1,47 Not only can histiocytic sarcoma and lymphoma resemble one another, in the current study they were found to occur together in the same mouse. In these cases, the diagnosis of the lymphoma population was based on usual characteristics such as cellular morphology and its presence in other tissues. 31 IHC helped to differentiate the histiocytic and lymphoid populations and highlight their proportions. When the distribution is entirely in the form of admixed nodules, it may be difficult to detect the presence of neoplastic lymphocytes by using HE stain alone, and IHC is needed to highlight these cells. Also, in these circumstances, the differential diagnosis of histiocyte-associated lymphoma, which consists of a large population of non-neoplastic histiocytes admixed with neoplastic B cells, needs to be considered. Diagnosing a true mixed histiocytic sarcoma and lymphoid neoplasia is not always straightforward, but the presence of numerous histioblasts and mitotic figures has been suggested to indicate malignancy. 19 A diagnosis can be aided with the use of IHC for the macrophage markers previously indicated, and the lymphoid population will be positive for either the B-cell markers CD45R/B220, PAX5, and/or IRF4 or the T-cell marker CD3. Descriptions of the diagnostic features of lymphoid neoplasia are reported in several references.31,35,44
When a histiocytic sarcoma presents with a more anaplastic morphology, it must be differentiated from the anaplastic variants of common soft tissue sarcomas such as hemangiosarcoma, leiomyosarcoma, and rhabdomyosarcoma, as well as from the less common undifferentiated pleomorphic sarcoma. Histiocytic sarcoma will be positive for the markers noted above, whereas these other sarcomas will be negative for them. However, antibodies to MAC2 can nonspecifically label some types of epithelial cells and fibroblasts, and the endothelium and/or pericytes of newly formed blood vessels may be positive for F4/80. 35 In the mouse, hemangiosarcomas will express CD31, VEGFR2, CD34, ERG, and/or von Willebrand factor, whereas histiocytic sarcomas will not express these markers. Interestingly, human histiocytes have been reported to express CD31, but to date mouse histiocytes have not.20,30 Therefore, CD31 probably should not be used as the sole diagnostic marker for differentiating histiocytic sarcoma from hemangiosarcoma in the mouse. Differentiation of leiomyosarcoma and rhabdomyosarcoma from histiocytic sarcoma is aided by the fact that unlike histiocytic sarcoma, both leiomyosarcomas and rhabdomyosarcomas express desmin and leiomyosarcomas are usually positive for α-SMA, while rhabdomyosarcomas may or may not express α-SMA. Furthermore, rhabdomyosarcomas will be positive for myogenic antigens such as MyoD1 and/or myogenin, whereas leiomyosarcomas will lack expression of these antigens. Specifically in the uterus, endometrial stromal sarcomas can be morphologically similar to histiocytic sarcomas, but they will be positive for S100, along with desmin and/or α-SMA, and negative for histiocytic markers. Similarly, anaplastic epithelial tumors and melanomas can be confused with histiocytic sarcoma; they are labeled with antibodies to keratins and HMB-45, respectively, whereas histiocytic sarcomas are negative for these antigens.
Given the rarity of histiocytic sarcoma in humans, there has been little research in animal models for this neoplasm. The tumor has been incidentally found to occur with increased incidence in numerous genetically engineered mouse lines, such as those with mutations in Ing2, 37 p21Waf1/Cip1/Sdi1, 5 and the Trp53 family, 15 and in mice lacking the combination of Dok1, Dok2, and Dok3. 29 However, the most important factor contributing to the development of mouse histiocytic sarcoma appears to be the genetic background of the mouse. The pathogenic mechanism underlying this strain-dependent development of the neoplasm is yet to be elucidated, but genome-wide analysis studies of aging mice may provide an opportunity to do so. The tumor is more common in C57BL/6 mice than in BALB/c mice,16,17 leading one group to postulate that because BALB/c mice are more susceptible than C57BL/6 mice to radiation-induced cancer, genomic instability is not a risk factor for developing histiocytic sarcoma. 26 The mouse model of Hertwig’s anemia, which is associated with the an/an allele (later identified as an allele of the Cdk5rap2a gene), was reported in 1979 to have a 98% incidence of histiocytic sarcoma. 32 A larger study in 2005 found an incidence of 90% in WBB6F1(F1)-an/an mice. 5 In that study, there was a correlation between myelopoiesis in the liver and the incidence of histiocytic sarcoma. This led the investigators to postulate that the increased proliferation of myeloid precursors in conjunction with the demonstrated genomic instability of hematopoietic precursors in the model led to the formation of histiocytic sarcoma.
A 2014 study attempted to delineate the genetic changes underlying histiocytic sarcoma in the mouse. 6 They used the Sleeping Beauty transposon as a mutagen, using the Lyz2 (lysozyme 2) promoter. One of the top 4 candidate genes was Raf1, which is part of the MAP kinase pathway. Also, more than 50% of the tumors had transposon insertions near mitogen-activated protein kinase (MAPK) pathway genes. Interestingly, studies using next-generation sequencing of human histiocytic sarcoma cases identified recurrent mutations involving the MAPK pathway in 57%–60% of cases.28,39
Additional insight into the genomic origin of histiocytic sarcoma in humans has been derived from studies undertaken to identify the specific mutations that cause histiocytic sarcoma secondary to lymphoid malignancy. A study on 16 such cases secondary to various lymphoid tumors (follicular lymphoma, chronic/small lymphocytic leukemia (CLL/SLL), B-ALL, or T-cell acute lymphoblastic leukemia) identified mutations in the RAS/MAPK (mitogen activated protein kinase) pathway in 14 of the cases. 13 This is particularly interesting given that the Sleeping Beauty transposon mouse study outlined above also found genes within the MAPK pathway that might underlie the development of histiocytic sarcoma. 6 One mutation within the MAPK, BRAFV600E, has gained particular attention in regard to tumors of histiocytic and Langerhans cell origin. This mutation is common in many diverse tumor types,2,14,25,41,48 and the availability of specific BRAF inhibitors10,23,38 prompted investigations of its occurrence in histiocytic and Langerhans cell tumors. The mutation was first demonstrated to be present in Langerhans cell histiocytosis at a rate of 38%–57%.4,36 The first reported case of Langerhans cell sarcoma with this mutation occurred subsequent to CLL/SLL. 8 In a later series analyzing 129 cases of various types of histiocytic and Langerhans cell tumors, 62.5% of the histiocytic sarcomas had the BRAFV600E mutation. 18
The studies outlined above indicate that there are probably similarities in the origins of mouse and human histiocytic sarcoma. Therefore, studies of naturally occurring and induced histiocytic sarcoma in mice may contribute to the identification of underlying genetic mutations involved in the pathogenesis of the tumor, as well as suggesting targetable therapeutic pathways. Genetically engineered mouse models featuring the more common mutations present in human histiocytic sarcoma, such as BRAFV600E, may provide opportunities to test possible treatments. This is especially important, because the rarity of these tumors in humans makes large clinical trials difficult. Whether any of the mouse histiocytic sarcoma morphologies are associated with mutations in specific gene pathways or with specific mouse strains remains to be resolved.
Identifying histiocytic sarcoma in the mouse can be challenging because of its varied morphologic presentations and its similarity to other hematopoietic tumors and sarcomas commonly seen in mice. IHC is of great value for diagnosing tumors definitively as histiocytic sarcoma. Because this tumor is rare in humans, investigators and pathologists who are only familiar with human tissues will not readily consider a diagnosis of histiocytic sarcoma in mouse studies. Therefore, it is incumbent upon veterinary pathologists to know the multiple faces of the tumor and to have it among their differentials when evaluating tumors in mice.
Footnotes
Acknowledgements
The authors thank the St. Jude Animal Resource Center and the Comparative Pathology Core for technical assistance. The authors thank Keith A. Laycock, PhD, ELS, for scientific editing of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was supported, in part, by the American Lebanese Syrian Associated Charities (ALSAC).
