Abstract
Invertebrates, including arachnids, are a common taxon in zoological collections. Invertebrate medicine and pathology are emerging subspecialties, but there is limited reference material or published resources describing histologic lesions in arachnids. Histopathology of 26 captive arachnids (20 spiders and 6 scorpions) from institutional collections was reviewed. Most animals were found dead with limited clinical signs. Tissues evaluated included body wall (cuticle and epidermis), skeletal muscle, book lungs, digestive tract (pharynx, esophagus, sucking stomach, midgut tube, midgut diverticula, and stercoral pocket), central and peripheral nervous system, heart, hemolymph vessels and sinuses, Malpighian tubules, coxal glands, and gonads. Inflammation was frequent (24/26, 92%), and seen in multiple organs (18/24, 75%) with the midgut diverticulum most commonly affected (14/24, 58%) followed by the book lungs (13/24 arachnids, 54%), and body wall (8/24 arachnids, 33%). Inflammation comprised hemocyte accumulation, hemocytic coagula, melanization, and nodulation. Infectious agents, including bacteria (11/26, 42%), fungi (10/26, 38%), and parasites (2/26, 8%), were seen within inflammatory aggregates. Coinfection with multiple infectious agents was common (6/24, 25%). No etiologic agent was identified in 7/24 (29%) cases with inflammatory lesions. Lesions suggestive of decreased nutritional status or increased metabolic rate included midgut diverticula atrophy in 11/26 (42%) animals and skeletal muscle atrophy in 6/26 (23%) animals. Atrophic lesions were seen in combination with infection (8/11, 73%), pregnancy (2/11, 18%), male sex (2/11, 18%), or without other lesions (1/11, 9%). Other suspected contributors to death included dysecdysis-associated trauma (2/26, 8%) and uterine intussusception (1/26, 4%). No animals had neoplasia.
The majority of living animals are in the phylum Arthropoda, 31 with animals in the class Arachnida playing a significant role in the pet trade, zoologic collections, and medical research. 24 The diversity of this group is astounding, with over 107,000 arachnid species described. 23 Common arachnids include spiders, scorpions, ticks, mites, and horseshoe crabs, with the latter debated as being true arachnids or subphyla of Chelicerata.1,6,21 There are 44 arachnid species listed under the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), comprising 5 scorpions and 39 spiders. 5 Improvement of medical management, recognition of basic signs of illness (incoordination, weight loss, color changes, and dysecdysis),9–11,24 and development of diagnostic tests to identify common diseases is critical to maintaining arachnid health in captivity.
Arachnids are a diverse group of animals that require specialty knowledge for their care, are important in multiple animal industries, and include multiple endangered species. Despite these factors, relatively little is known about diseases of captive arachnids. 24 While veterinary pathologists’ specialized training in comparative disease pathogenesis provides an ideal background for evaluation of disease, the vast majority of veterinary pathologists lack specialized training in invertebrate pathology, and only recently have resources become available detailing normal arachnid histomorphology. 16 Consequently, histopathologic studies of arachnids are limited in both number and scope. They include a survey of histologic lesions in captive horseshoe crabs, 18 description of midgut atrophy in wild European cave spiders, 19 and characterization of Rickettsiella sp. infection of captive emperor scorpions. 12 Retrospective studies reporting causes of morbidity and mortality in 2 of the more common arachnid genera kept in the pet trade and zoo collections, spiders (Araneae) and scorpions (Scorpiones), are lacking.
We report histologic lesions in captive arachnids in managed care from institutional collections in North Carolina. The objective of this retrospective study is to provide a reference for common histologic lesions affecting captive scorpions and spiders, with the ultimate goal of improving prevention, diagnosis, and treatment of disease in these animals.
Materials and Methods
Archived histopathology reports from North Carolina State University, College of Veterinary Medicine were searched for submissions of arachnid specimens between January 2011 and August 2020. Cases with advanced autolysis and/or missing tissue blocks were excluded from the study. Archival slides were examined to confirm the original diagnoses and further systematically characterize lesions. All cases were reviewed by at least 3 pathologists (CG, MES, and EEBL) and categorized by lesion location and etiology (Table 1, Supplemental Table S1).
Systems and organs evaluated in 26 captive arachnids examined retrospectively after unexpected death for identification of histopathologic lesions.
Dead animals were submitted whole and either fresh or in fixative to North Carolina State University, College of Veterinary Medicine (Supplemental Table S1). Dorsal midline, ventral midline, or parasagittal incisions were made in the opisthosoma to promote formalin perfusion of internal tissues. Cadavers were placed in 10% neutral buffered formalin for at least 24 hours followed by processing in Bouin’s solution for 36–72 hours. The cadavers were then washed for approximately 1 minute with water followed by placement in lithium carbonate for 1 hour. Cadavers were then rinsed in water for 1 hour before placement in 10% formic acid for 24–120 hours, as needed depending on the firmness of the animal’s carapace. Cadavers were then washed for approximately 1 minute with water followed by placement in 70% ethanol for 48 hours. Trimming was performed after incubation in 70% ethanol for at least 24 hours. The majority of cadavers were trimmed in serial transverse sections, though a few were trimmed in serial sagittal sections. A single biopsy specimen was submitted in formalin and routinely trimmed with no additional fixatives used. Trimmed tissues were processed routinely, embedded in paraffin, sectioned at 5 micrometers, and stained with hematoxylin and eosin (HE). Additional standard laboratory histochemical stains (Giemsa, Gram, Periodic acid-Schiff, Grocott’s methenamine silver [GMS], Ziehl-Neelsen, and/or modified Ziehl-Neelson [Fite’s]) were applied to identify or confirm the presence of infectious agents as applicable on a case by case basis (6/24 cases, 25%).
Previously described invertebrate tissue reactions (eg inflammation) were used to guide definitions of inflammation in arachnids in this study.14,16 Briefly, inflammation was characterized by increased numbers of hemocytes in tissues, which can be further delineated into hemocytosis, hemocytic coagula, and hemocytic tissue inflammation with or without nodulation and melanization. For the purposes of this article, hemocytes were defined as any immune cell identified within hemolymph (ie within the aorta or adjacent major vessels, hemocoel, or hemolymph sinuses) and/or foci of tissue inflammation, and for descriptive purposes were further characterized as granulated (containing abundant bright eosinophilic granules) or nongranulated (pale basophilic cytoplasm, Fig. 1a).
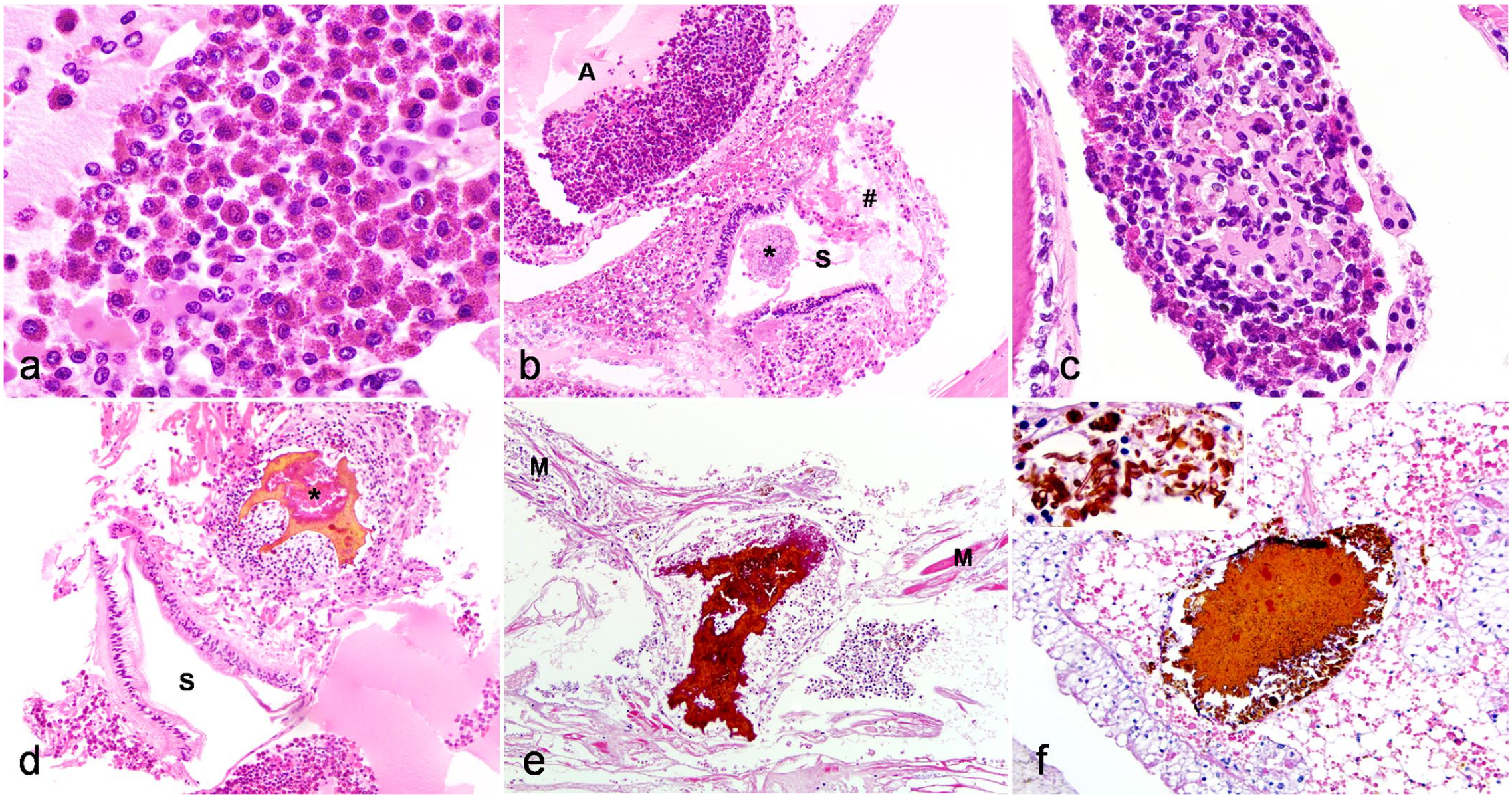
Histomorphology of the inflammatory response in captive arachnids. Hematoxylin and eosin. (a–b) Peripheral hemocytosis, aorta, Chilean rose tarantula (Grammostola rosea, case 6). (a) Granulated hemocytes have oval to round nuclei with condensed chromatin and bright eosinophilic granules. Nongranulated hemocytes have oval to round nuclei with condensed chromatin and scant amphophilic cytoplasm. (b) Peripheral hemocytosis within the aorta is associated with transmural ulcerative gastritis. Within the sucking stomach (S) that is adjacent to the aorta (A), there is ulceration, segmental transmural necrosis, and perforation of the mucosa (#) with adjacent hemocytic inflammation. Luminal enteric bacteria are indicated with an asterisk. The lumen of the aorta contains granulated and nongranulated hemocytes and there is disruption of the vessel wall with transmural hemocytic infiltration and vessel wall necrosis. (c) Hemocytic coagulum, prosomal hemolymph sinus, Costa Rican zebra tarantula (Aphonopelma seemanni, case 14). Granulated and nongranulated hemocytes surround a coagulum of pale eosinophilic fibrin-like hyalinized material. (d–f) Examples of nodulation with melanization. Inflammatory nodules contain central bright eosinophilic to amber extracellular material surrounded by hemocytes. (d) Prosomal nodulation and melanization associated with ulcerative gastritis, Chilean rose tarantula (Grammostola rosea, case 6). Transmural ulcerative and perforating bacterial gastritis with abundant granulated hemocytes (sucking stomach is indicated with an “S”). Bacterial colonies are embedded within melanin, which surrounds a hemocytic coagulum (asterisk). (e) Nodulation with melanization in fungal myocarditis, Haitian brown tarantula (Phormictopus cancerides, case 16). Inflammatory nodules disrupt the myocardium (M). (f) Nodulation with melanization in midgut diverticulitis (Haitian brown tarantula (Phormictopus cancerides, case 16). Inflammatory nodules disrupt the midgut diverticula interstitium and melanized fungal hyphae are present within inflammatory nodules (inset).
Hemocytosis was defined as hemocytes filling more than 10% of the cross-sectional luminal area within hemolymph sinuses, aorta and adjacent vessels, or the heart (Fig. 1a, b). Hemocytic coagula, similar to that previously described in other invertebrates, 18 appeared as a mixed population of granulated and/or nongranulated hemocytes admixed with eosinophilic amorphous fibrin-like material and/or necrotic cellular debris (Fig. 1c). Hemocytic tissue inflammation was characterized by aggregates of granulated or nongranulated hemocytes that were peripherally surrounded by bands of elongated, degranulated hemocytes (Fig. 1d–f). This pattern is consistent with nodulation, an invertebrate inflammatory response that is analogous to vertebrate granuloma formation.2,8,12,14,27 Melanization, an innate humoral immune response shared amongst many arthropods2–4,12,14,16,18,25,27,29 was characterized by deposition of an amorphous mass of dense, amber-yellow, slightly refractile, extracellular material (Fig. 1d–f).
Results
Case Demographics
A total of 31 animals (23 spiders and 8 scorpions) were identified. Animals were submitted from the North Carolina Zoo (n = 1), SEA LIFE Charlotte-Concord Aquarium (n = 1), North Carolina State University College of Veterinary Medicine Laboratory Animal Resource Colony (n = 3), and the North Carolina Museum of Natural Sciences (n = 26). Five animals (3 spiders and 2 scorpions) were excluded due to advanced autolysis and/or missing tissue blocks, resulting in 26 animals included in the study. These included 20 spiders (16 females, 1 male, and 3 undetermined sex) and 6 scorpions (3 females, 1 male, and 2 undetermined sex). Genera and species included in the study are summarized in Table 2. The majority of the cases were whole-body postmortem submissions (25/26, 96%), while tissue from 1 tarantula of unknown species was a postmortem biopsy submission of body wall and book lung (case 25).
Number and species of captive arachnids examined retrospectively after unexpected death for identification of histopathologic lesions.
Either whole-body or selected tissues were submitted for histologic examination after unexpected clinical decline.
Abbreviation: N/A, not applicable.
Clinical information and detailed histologic findings for each animal are summarized in Supplemental Table S1. The majority of animals (22/26, 85%) were found dead. Three animals were euthanized and the type of death was not reported for 1 animal. In addition, the majority of animals (17/26, 65%) had no reported premonitory clinical signs. Clinical signs for the remaining 9 animals included issues with molting/dysecdysis (3/9 arachnids), suspected or witnessed trauma (3/9 arachnids), and lethargy (3/9 arachnids).
Overview of Organs Examined and Frequency of Organ Lesions
A wide range of organs showed histologic lesions that are summarized in Table 3. Three tissues had lesions in 50% or more of examined arachnids. The highest prevalence of lesions was in the midgut diverticula, where inflammation and atrophy were common. The next highest of prevalence lesions was in the book lungs, where pneumonia was most common. The skeletal muscle was the third most commonly affected tissue, where both inflammation and atrophy were identified. All other tissues had lesions in less than 50% of examined arachnids.
Tissue types examined, frequency of tissue identification, and frequency of histological lesions and lesion types in 26 captive arachnids examined retrospectively after unexpected death.
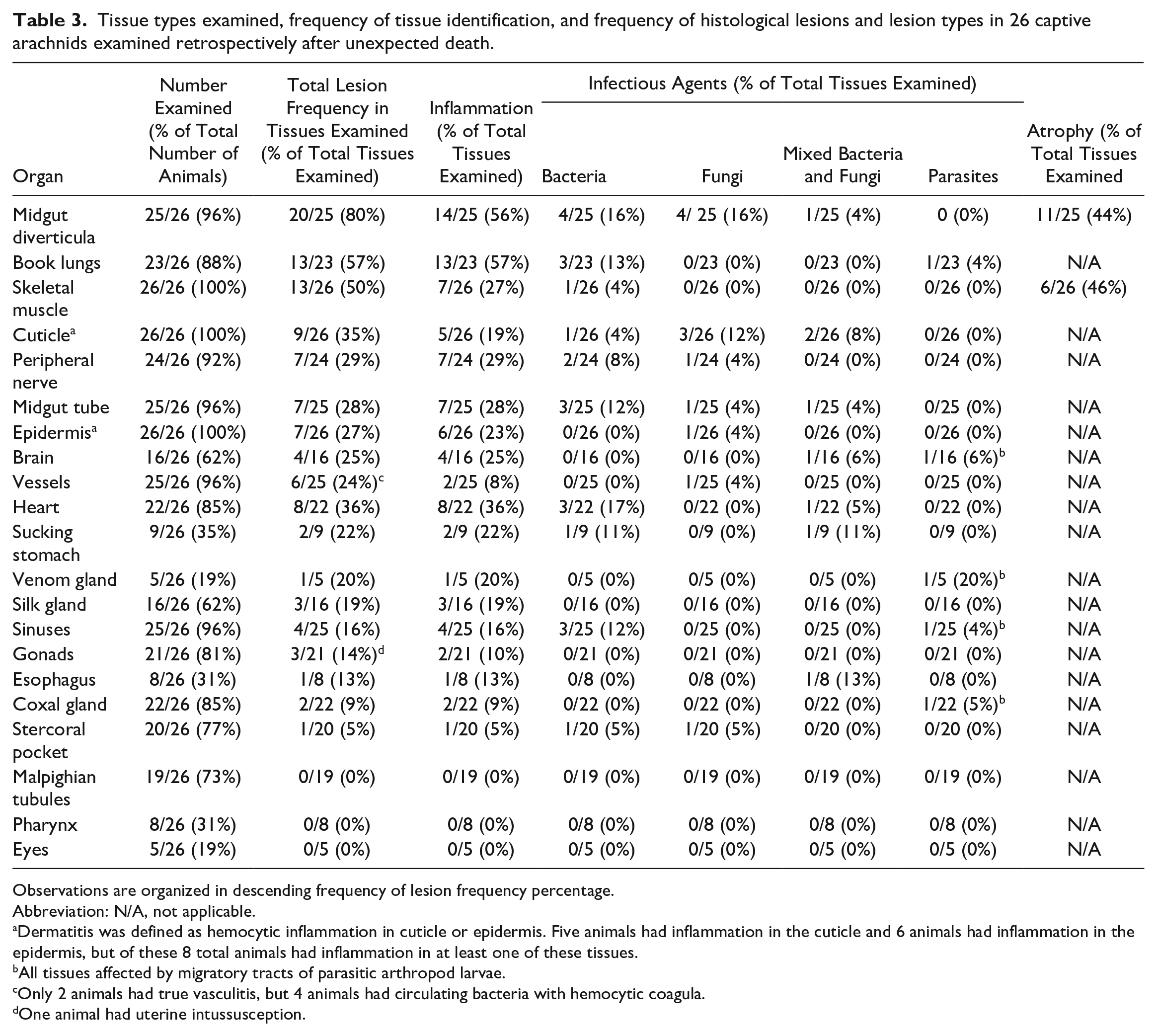
Observations are organized in descending frequency of lesion frequency percentage.
Abbreviation: N/A, not applicable.
Dermatitis was defined as hemocytic inflammation in cuticle or epidermis. Five animals had inflammation in the cuticle and 6 animals had inflammation in the epidermis, but of these 8 total animals had inflammation in at least one of these tissues.
All tissues affected by migratory tracts of parasitic arthropod larvae.
Only 2 animals had true vasculitis, but 4 animals had circulating bacteria with hemocytic coagula.
One animal had uterine intussusception.
Inflammatory Response
Hemocytosis (15/26 arachnids, 58%) was common (Fig. 1a, b). Cases with hemocytosis frequently also had infections (9/15 arachnids, 60%), including fungal (2/9 arachnids), bacterial (2/9 arachnids), or mixed bacterial and fungal infection (5/9 arachnids). Of the 6/15 (40%) of arachnids with hemocytosis and no histologically identified infectious agent, 2/6 had concurrent midgut diverticula and/or skeletal muscle atrophy, and 3/6 had multi-systemic hemocytic tissue inflammation. In cases with hemocytosis and concurrent tissue infection, hemocytes were often seen marginating in the aorta or hemolymph sinuses at the location of infectious organisms (Fig. 1b).
Hemocytic coagula were similar to that previously described in other invertebrates. 18 Hemocytic coagula were seen both within vasculature and hemolymph sinuses accompanying hemocytosis and bacteremia (Fig. 1c) and within the centers of large hemocytic inflammatory aggregates (Fig. 1d).
Hemocytic tissue inflammation and nodulation were similar to that previously described in other invertebrates,2,8,12,14,27 with nodules ranging in size from 30 to 400 µm (Fig. 1d–f). Hemocytic tissue inflammation was often accompanied by melanization, and in cases of nodulation, central melanization with intermixed necrotic nuclear and cellular debris was common (Fig. 1d–f). In some cases, large melanin aggregates comprised the majority of the nodule (Fig. 1e, f), with melanin often coating infectious agents (Fig. 1f). Coagulative to lytic tissue necrosis accompanied more severe hemocytic inflammation (eg, inflammation comprised > 50 hemocytes within a single inflammatory nodule), and was identified in several tissues (skeletal muscle, cardiac muscle, brain, midgut diverticula, epidermis, coxal gland, and silk gland; Supplemental Table S1).
Multi-organ inflammation suggestive of systemic inflammation and/or septicemia was identified in 18/26 (69%) arachnids; this finding was frequently associated with bacterial and/or fungal infection (13/18 arachnids, 72%). For 5 of these animals, no etiologic agent was identified.
Infectious Agents
Infectious agents were commonly identified in our cases, with 17/26 animals (65%) having either bacterial (11/17 arachnids, 65%), fungal (10/17 arachnids, 59%), or parasitic infections (2/17 arachnids, 12%). Six animals had coinfections with multiple categories of these infectious agents, with bacterial and fungal coinfection most common (5/6 arachnids, Table 3, Supplemental Fig. S1). While fungal and bacterial agents were consistently found with hemocytic inflammation and/or hemocytosis (Fig. 2a–d), parasitic infection had minimal to no associated inflammation (Fig. 2e–g).
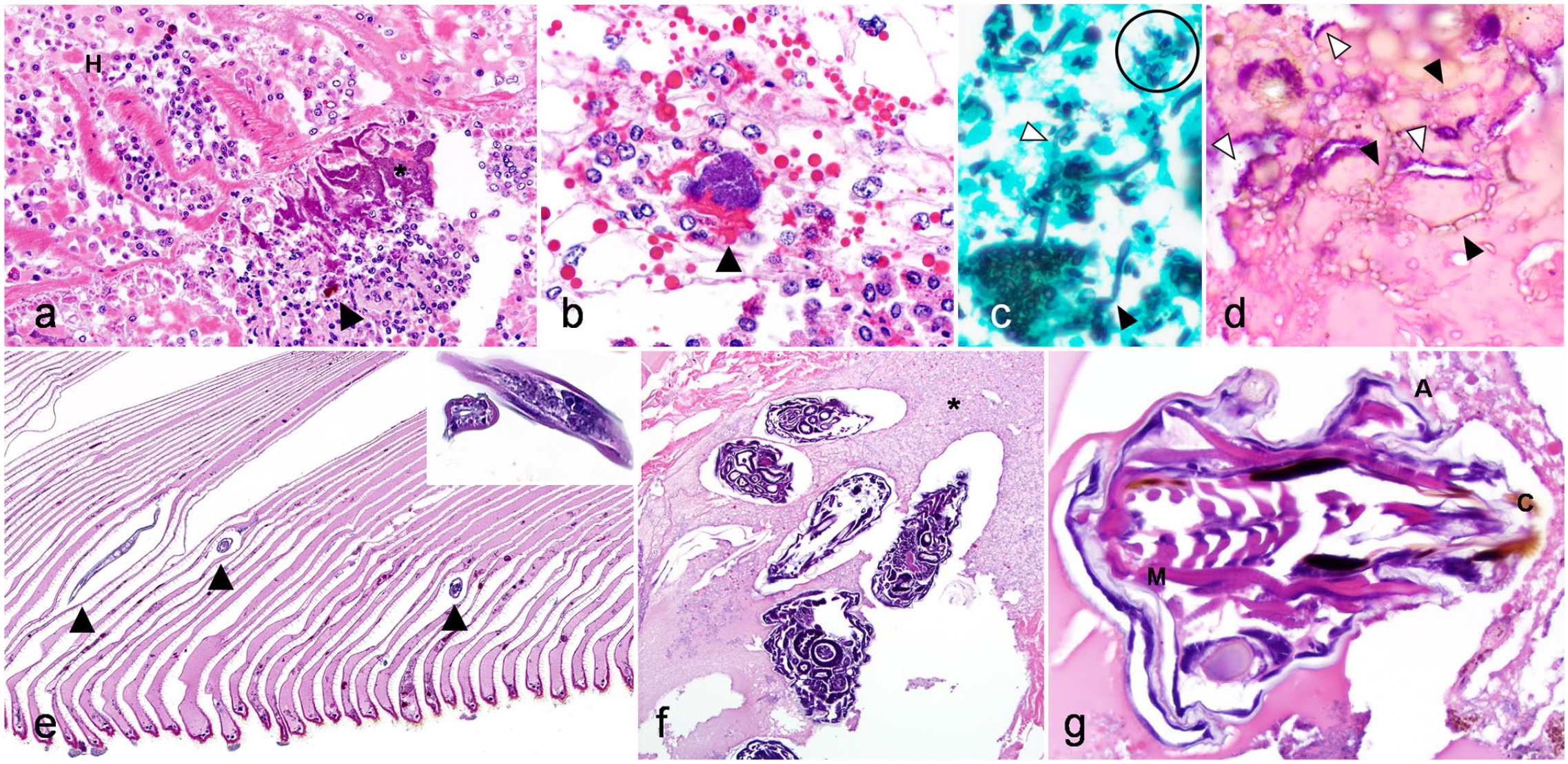
Histomorphology of bacteria, fungi, and parasites infecting captive arachnids. (a) Peripheral hemocytosis and bacterial septicemia, heart, Chaco golden knee tarantula (Grammostola pulchripes, case 15). Increased hemocytes within the heart lumen (H), and an extracellular colony of bacteria (*) in the epicardium and hemolymph of the pericardial sac with associated hemocytic myocarditis, hemocyte necrosis, and foci of melanization (arrowhead). Hematoxylin and eosin (HE). (b) Bacterial midgut diverticulitis, Chaco golden knee tarantula (Grammostola pulchripes, case 15). Granulated and nongranulated hemocytes surround an extracellular colony of bacteria. Bright eosinophilic material associated with bacteria (arrowhead) represents a mixture of hemocytic coagulum and midgut diverticula intracellular protein. HE. (c) Fungal midgut diverticulitis, Haitian brown tarantula (Phormictopus cancerides, case 16). Fungal yeasts (circled), hyphae (black arrowhead), and pseudohyphae(white arrowhead) in an entomopathogenic fungal infection. Hyphae are nonseptate with dichotomous branching and bulbous dilations. Grocott’s methenamine silver. (d) Fungal and bacterial dermatitis, Arizona blond tarantula (Aphonopelma chalcodes, case 1). Cuticular saprophytic fungi have different morphologies from visceral entomopathogenic fungi. No yeast are present and hyphae are prominently septate (black arrowheads). Concurrent bacterial colonization (white arrowheads) is present. HE. (e) Hemocytic pneumonia with interlamellar nematodes (arrowheads), book lungs, Asian forest scorpion (Heterometrus longimanus, case 18). Nematodes are approximately 20 µm in diameter with a pseudocoelom, coelomyarian-polymyarian musculature, lateral alae, and a digestive tract lined by cuboidal cells (inset). HE. (f–g) Larval arthropod infestation of prosoma, cobalt blue tarantula (Cyriopagopus lividum, case 10). (f) Prosoma viscera, including brain, digestive tract, vasculature and hemolymph sinuses, and skeletal muscle are obliterated by arthropod larvae and surrounding tracts of lytic necrosis (asterisk). HE. (g) The parasite has a chitinized cuticle (C), coelom, striated muscles (M), appendages (A), and complicated internal digestive organs, consistent with a larval arthropod. HE.
Eleven arachnids had bacterial infections. In 3 of these, visible bacteria were restricted to the cuticle. The remaining 8 cases showed multi-systemic hemocytic inflammation with bacteria in at least 2 visceral organs. The digestive tract was most commonly affected (6/8 arachnids); followed by the cardiovascular system, lung, and nervous system (3/8 arachnids for each tissue), and the skeletal muscle in 1 arachnid (Supplemental Table S1). Presumptive bacterial septicemia with bacterial colonies within hemolymph vessels or sinuses was seen in 5/8 cases with multi-systemic bacterial infection. Bacterial infection often manifested as large colonies of extracellular coccobacilli surrounded by small deposits of melanin further surrounded by few peripheral hemocytes with variably prominent nodulation (Fig. 2a, b). Notably, bacterial overgrowth was seen in multiple cases, and was distinguished from true bacterial infection by lack of associated hemocytic inflammation, erosion/ulceration, or necrosis.
Ten animals had fungal infections, which were consistently characterized by the presence of hyphal organisms and were always associated with hemocytic inflammation. Hyphal morphologies varied between cases, but generally had nonparallel walls with bulbous dilations, variably prominent septation, and acute angle dichotomous branching (Fig. 2c, d). Rarely, round to ovoid yeast forms, often in chains (“pseudohyphae”) were seen with hyphae and concurrent inflammation, and were interpreted as pathogenic (Fig. 2c). Yeast were often identified in digestive tracts without associated inflammation and were interpreted as normal digestive tract flora. Of the 10 arachnids with fungal infections, 4 cases were limited to the cuticle. The remaining 6 animals all had digestive tract fungal infections. Of these 6 animals with digestive tract fungal infection, 3/6 had fungal infections restricted to the digestive tract, while 1 animal had involvement of the cardiovascular system; 1 animal had involvement of the cardiovascular system and nervous system; and 1 animal had involvement of the nervous system and cuticle/body wall. In contrast to bacterial infections, visceral fungal infections consistently manifested with larger, organized and well-demarcated nodulation and more abundant melanization (Fig. 1e, f).
Parasitic infections by nematode larvae in the book lungs and arthropod larvae in the prosoma were each identified in a single animal. In 1 gravid female Asian forest scorpion (Heterometrus longimanus, case 18), nematode larvae were distributed multifocally in the air spaces of the book lung lamellae and were associated with minimal hemocytic inflammation (Fig. 2e). The nematodes were approximately 20 µm in diameter with a pseudocoelom, coelomyarian-polymyarian musculature, lateral alae, and a digestive tract lined by cuboidal cells. In 1 Cobalt blue tarantula (Cyriopagopus lividum, case 10), there was extensive invasion and colonization of the prosoma by myriad arthropods, which were classified as larvae based on their size and lack of identifiable reproductive tracts. Arthropod larvae were characterized by an outer chitinized cuticle, peripheral skeletal muscle, segmentation, a coelom, and internal nervous system and digestive tracts (Fig. 2f, g). Associated lesions included migratory tracts with extensive lytic necrosis, dissolution of tissues with accumulation of abundant eosinophilic fluid and debris, and minimal inflammation with frequent colonization by mixed bacteria interpreted as postmortem overgrowth.
Digestive System
The midgut diverticula, also known as the digestive ceca and similar to hepatopancreas or fat bodies in other invertebrate species, were present for histologic evaluation in 25 of the 26 arachnids (96%) and were the most commonly affected organs in all animals examined (20/25 arachnids; 80%). Midgut diverticulitis, the most common lesion identified in the digestive system, was identified in 14/25 animals (56%). Midgut diverticulitis was focal (2/14 arachnids, 14%) or multifocal (12/14 arachnids, 86%; Supplemental Table S1). Hemocytic inflammation most often formed discrete aggregates that disrupted/replaced interstitial cells (12/14 arachnids, 86%). Erosion/ulceration of tubular cells was uncommonly identified (2/14 arachnids, 14%; Supplemental Table S1). Melanization was common (12/14 arachnids, 86%) and often associated with nodulation (11/14 arachnids, 79%; Fig. 1f). Midgut diverticulitis was often associated with infectious agents (9/14 arachnids, 64%), including bacteria (4/9 arachnids, 44%), fungi (4/9 arachnids, 44%), or mixed bacteria and fungi (1/9 arachnids, 11%). In 5/14 cases (36%), no etiologic agent was identified. Midgut diverticulitis was often associated with other lesions indicating systemic disease, including multi-organ inflammation (14/14 arachnids, 100%), hemocytosis (12/14 arachnids, 86%), and/or skeletal muscle atrophy (5/14 arachnids, 36%).
Reduction of intracellular eosinophilic globules within midgut diverticula interstitial and tubular cells was commonly seen and was interpreted as atrophy (11/25 arachnids, 44%; Fig. 3a). This lesion was often found with skeletal muscle atrophy (6/11 arachnids, 55%) or systemic inflammatory processes (7/11 arachnids, 64%), including multi-organ inflammation (8/11 arachnids, 73%), hemocytosis (4/11 arachnids; 27%), bacterial infection (2/11 arachnids; 18%), and/or fungal infection (3/11 arachnids; 27%). Two female scorpions were gravid (Fig. 3b, c) and both had lesions suggesting decreased nutritional status (both with midgut diverticula atrophy and 1 with skeletal muscle atrophy). One gravid female had a uterine intussusception characterized by telescoping/inversion of the organ such that layers of mucosa were in intimate contact (Fig. 3c). Cellular inflammation was minimal, but there was multifocal sloughing and ulceration of the mucosa with mild associated melanization (Fig. 3c).
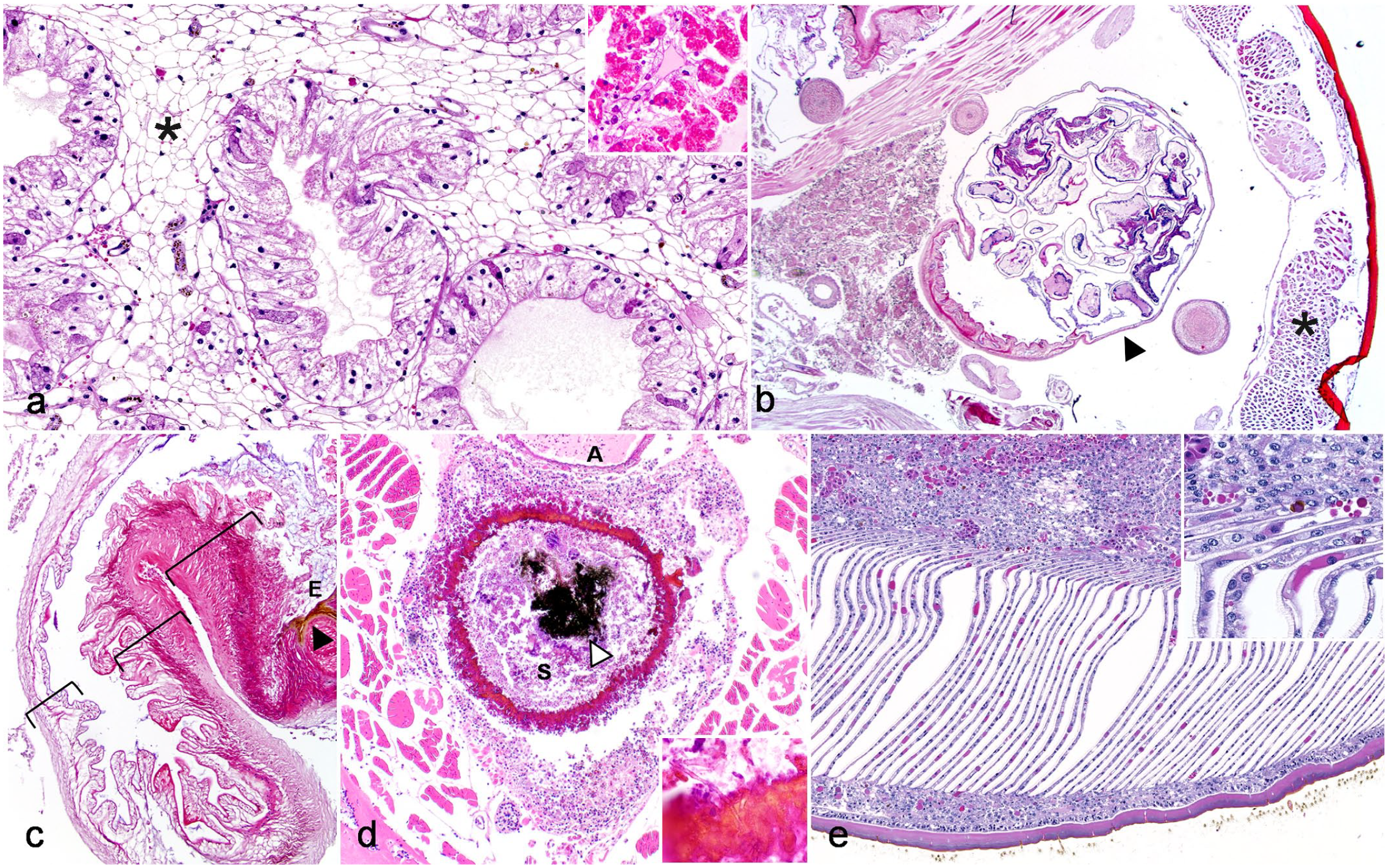
Histomorphology of gastrointestinal, respiratory, and reproductive tract lesions in captive arachnids. Hematoxylin and eosin. (a) Midgut diverticula atrophy, Costa Rican zebra tarantula (Aphonopelma seemanni, case 26). Midgut diverticula atrophy is characterized by loss of eosinophilic globules within interstitial cells (*) and tubular cells; inset shows normal midgut diverticula from a black widow (Latrodectus spp., case 5). (b–c) Gravid uterus with partial uterine intussusception, Asian forest scorpion (Heterometrus longimanus, case 13). (b) Uterus (arrowhead) contains cross sections of multiple developing nymphs. Skeletal myofiber atrophy is also apparent (asterisk). (c) Partial uterine intussusception. There is partial telescoping of uterus with corresponding mucosal erosion (E) and melanization (black arrowhead). The uterine wall is indicated in 3 separate locations by brackets. (d) Transmural and circumferential ulcerative hemocytic gastritis, Brazilian red and white tarantula (Nhandu chromatus, case 19). The wall of the sucking stomach is circumferentially perforated and replaced by hemocytic coagula and melanin with embedded fungal hyphae (inset). The aorta (A) is adjacent to the stomach (S). Coarse luminal granular black pigment (white arrowhead) is consistent with guanine crystals found in ingesta of arachnids. This material is birefringent and is a normal finding in the digestive tracts of arachnids. (e) Hemocytic pneumonia with melanization, book lungs, Arizona blond tarantula (Aphonopelma chalcodes, case 21). Book lung lamellae are expanded by hemocytes with spread of hemocytic inflammation into the adjacent opisthosomal coelom and melanin deposition (inset).
Hemocytic inflammation was less commonly identified in other alimentary segments (7/25 midgut tubes [28%], 2/9 sucking stomachs, 1/8 esophagi, and 1/20 stercoral pockets [5%]; Supplemental Table S1). Of these lesions, inflammation was associated with ulceration in 1 or more of these segments in 4/11 lesions (36%) and infectious agents in 6/11 lesions (54%), including bacteria (4/6 lesions), fungi (1/6 lesions), or bacteria and fungi (1/6 lesions; Supplemental Table S1). Ulcerative esophagitis, gastritis (Figs. 1b, d, 3d), and/or enteritis was associated with marked regional hemocytic inflammation of the prosoma, vasculitis, and presumed septicemia in 2 cases.
Respiratory System
Book lungs were present for histologic examination in 23 of the 26 arachnids (88%). Hemocytic pneumonia, the only lesion identified in book lungs, was identified in 13 animals (13/23 arachnids, 56%). This was characterized by increased numbers of granulated or nongranulated hemocytes expanding hemolymph channels in the lamellae. Most often, there was 1–2 cells thick expansion of hemolymph channels by hemocytes, but occasionally hemocytes formed more discrete aggregates at the base of lamellae within hemolymph channels that were associated with melanization (Fig. 3e). Infectious causes were associated with hemocytic pneumonia in 4/13 (42%) cases and included bacteria (3/4 cases) and parasitic (1/4 cases) etiologies. Bacterial agents were most often extracellular colonies of coccobacilli present within hemolymph channels. These were consistently seen with multi-systemic bacterial infection, suggesting hematogenous spread to the book lungs.
Body Wall and Musculoskeletal System
The body wall and the musculoskeletal system was histologically evaluated in all 26 arachnids. Skeletal muscle was more commonly affected (14/26 arachnids, 54%) than the body wall (cuticle and/or epidermis, 12/26 arachnids, 46%). Body wall lesions included lesions in the cuticle (9/26 arachnids; 35%) or epidermis (7/26 arachnids; 27%).
The most common lesion affecting the body wall was hemocytic dermatitis (8/12 arachnids, 67%). Dermatitis was defined as hemocytic inflammation within the cuticle and/or epidermis. Infectious agents were present in half of the cases of dermatitis (4/8 cases), and included either fungi (2/4 cases) or mixed fungi and bacteria (2/4 cases). Cases of dermatitis with visible infectious agents tended to be more severe (eg, hemocytic inflammation affecting all layers of the cuticle and epidermis, Fig. 4a), including a case of mixed bacterial and fungal dermatitis in a site of repeated hemolymph collection in a tarantula used for teaching purposes (case 1, Fig. 4b). Similarly, arachnids with infectious dermatitis often had concurrent multi-organ inflammation (3/4 arachnids). In contrast, cases that lacked histologically detectable etiologic agents tended to be milder (ie, only affecting partial layers of the cuticle) or affected only the epidermis (Fig. 4c; Supplemental Table S1). Dysecdysis (retained cuticle during molting) was also associated with mild dermatitis (see below).
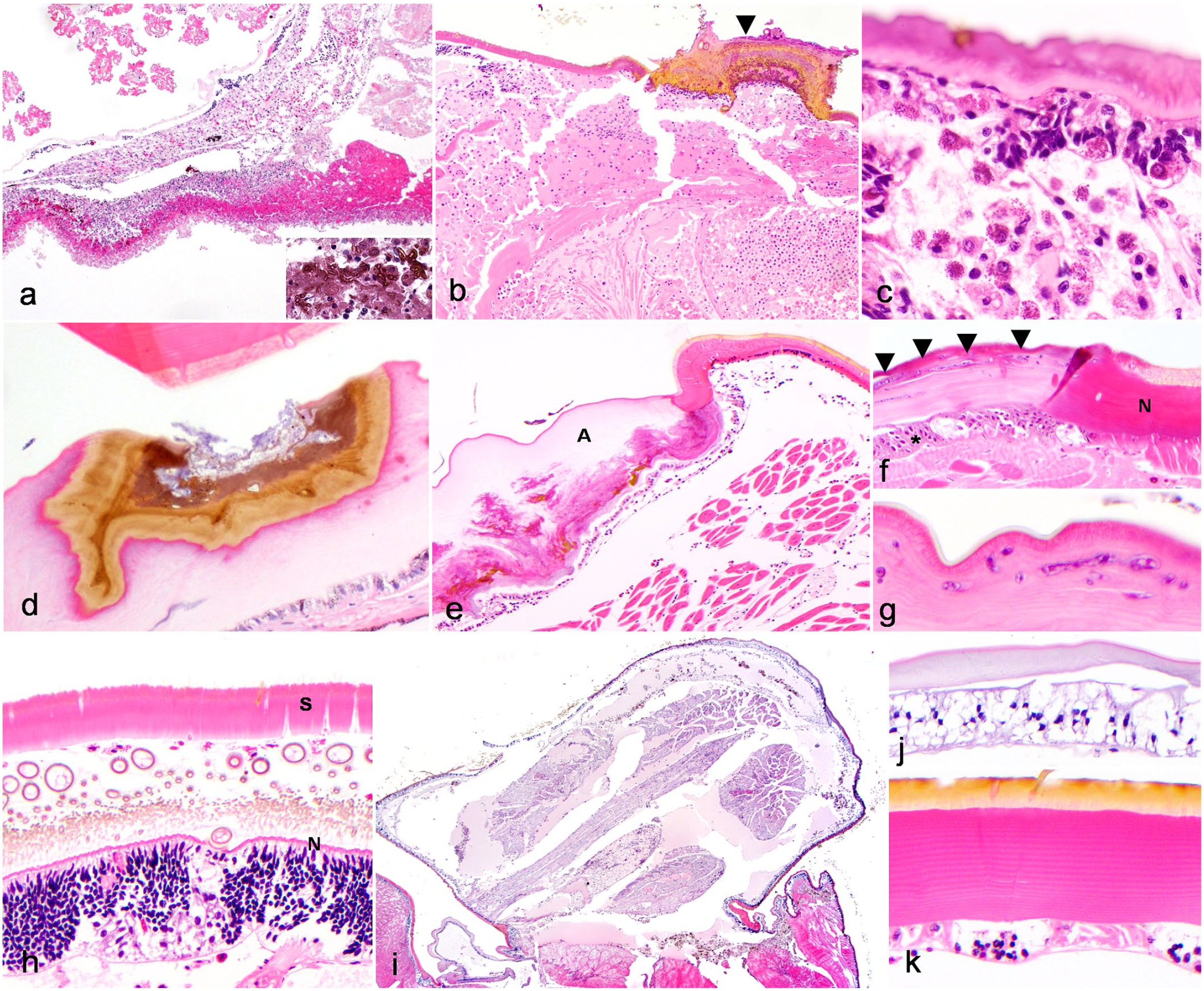
Histomorphology of body wall lesions in captive arachnids. Hematoxylin and eosin. (a) Full thickness cuticular ulceration, ventral opisthosoma, Chaco golden knee tarantula (Grammostola pulchripes, case 22). The cuticle and epidermis of the ventral opisthosoma are transmurally ulcerated and replaced by a thick crust of necrotic nuclear and cellular debris, hemocytic coagula, and foci of melanization. Inflammation and necrosis extend regionally into the hemocoel. Melanized fungal yeast, pseudohyphae, and hyphae are embedded in the surface crust (inset). Hyphae exhibit septation with nonparallel walls. (b) Intracardiac hemolymph sample site-associated dermatitis and myocarditis, dorsal opisthosoma, Arizona blond tarantula (Aphonopelma chalcodes, case 1). Focal dorsal ulcerative and crusting dermatitis with melanization (arrowhead) at a site of repeated hemolymph sampling. The underlying epidermis and myocardium are disrupted by regionally extensive hemocytic hemocoelomitis and myocarditis. Within the cuticular crust are abundant fungi and bacteria. (c) Epidermis-restricted hemocytic dermatitis, Chilean rose tarantula (Grammostola rosea, case 6). Hemocytes multifocally invade and disrupt the epidermis, with variable loss or piling (hyperplasia/dysplasia) of the epidermal cells. (d) Arthrodial membrane erosion, emperor scorpion (Pandinus imperator, case 24). Focal erosion of the cuticle of the arthrodial membrane, which is a flexible area of cuticle present at areas that require more movement (eg, appendage joints). Superficial melanization accompanies erosion; basophilic superficial material is consistent with bacteria on higher magnification (not shown). (e) Abnormal cuticular layering at arthrodial membranes, emperor scorpion (Pandinus imperator, case 20). The arthrodial membrane cuticle (A) is thickened and undulating, with abnormal layering in the mid to deep aspects and scattered foci of melanization. The epidermal cells are mildly hypertrophied with occasional dropout and scant hemocytes. (f–g) Abnormal cuticular layering associated with fungal invasion, Asian forest scorpion (Heterometrus longimanus, case 18). (f) Abrupt transition from normal cuticle (distinct epicuticle, exocuticle, and endocuticle; N) to a region lacking cuticular stratification. Fungal hyphae (arrowheads) are present in this area and the epidermis deep to this abnormal cuticle is moderately hyperplastic (asterisk) (g) Fungal hyphae have bulbous dilations, nonparallel walls, and irregular septation. (h) Dysecdysis with mild hemocytic dermatitis and epidermal hyperplasia, Brazilian black tarantula (Grammostola pulchra, case 4). Two separate cuticles are apparent. The superficial cuticle (S) represents the retained shed and has lost distinct stratification. Deep to this are scattered hemocytes and numerous cuticular hairs emerging from the newly forming cuticle. The new cuticle (N) is approximately 1/20 the thickness of the shed cuticle and lacks stratification. The subjacent epidermis is hyperplastic (approximately 10–15 cells thick), which is considered appropriate for new cuticle growth. (i–k) Limb regrowth post auto-amputation, Costa Rican zebra tarantula (Aphonopelma seemanni, case 7). (i) The limb is shortened and exhibits pallor and basophilia, wide spacing between myofiber bundles and peripheral nerves, and accumulation of abundant hemolymph. (j) The cuticle is thinned, basophilic, and lacks stratification that would be expected in normal cuticle (k).
Other cuticular lesions included erosion, ulceration, abnormal cuticle layering, and dysecdysis, while other epidermal lesions included ulceration. Cuticle erosions were defined as partial-thickness loss of the cuticle (ie, not extending to the epidermis) while cuticle ulceration was defined as full thickness loss of the cuticle extending to the level of the epidermis. Abnormal cuticle layering was defined as alterations in thickness, coloration, and organization of the epicuticle, exocuticle, and endocuticle. Both cases of erosion, which accounted for 2/9 cuticular lesions, occurred in the absence of dermatitis. In 1 case, erosion was associated with superficial bacterial colonization within the eroded cuticle of arthrodial membranes, which are flexible areas of cuticle present at anatomic sites requiring increased mobility (eg, appendage joints, Fig. 4d). Similarly, abnormal cuticle layering (occurring in 3/9 animals with cuticular lesions) was identified in the absence of concurrent dermatitis in all 3 cases (Fig. 4e). In 1 case, this lesion was associated with cuticular fungal colonization (Fig. 4f, g). Erosion and/or abnormal cuticle layering affected the arthrodial membranes in 3 animals and was associated with cuticle melanization (Fig. 4d, e). In contrast to these lesions, ulceration (3/12 body wall lesions, 25%) of the cuticle and/or the epidermis (3/12 body wall lesions, 25%) consistently occurred with hemocytic dermatitis. All cases of cuticle ulceration were associated with fungal infections, including some of the cases described above with subsequent systemic spread of infection (Fig. 4a). Ulceration of the epidermis was seen either in conjunction with cuticular ulceration (2/3, 67%) or with subjacent internal inflammation (ie, and “inside to out” type lesion, 1/3 cases, Fig. 4c).
Two tarantulas had histologic lesions related to dysecdysis. Dysecdysis was noted antemortem in both animals (Supplemental Table S1). In the first case (case 4), dysecdysis was characterized by a thicker, detached cuticle that lacked associated epidermis overlying a second, thinner cuticle that maintained typical histologic layering. (Fig. 4h). Subjacent to the deeper cuticle, the underlying epidermis was hyperplastic and infiltrated by hemocytic inflammation. Moderate numbers of granulated hemocytes, eosinophilic fluid, and trichobothria had accumulated between the cuticle layers. This animal also had systemic hemocytic inflammation, though an underlying etiologic agent was not identified. In a second case (case 7), there was a regionally extensive cuticle tear associated with a clinically reported previous dysecdysis. Hemolymph loss from this site prompted euthanasia in this animal (Supplemental Table S1). In addition, this animal had limb regrowth presumably secondary to autotomy related to dysecdysis (Fig. 4i). The new limb was severely shortened (approximately 2.5 mm long, compared with an estimated mature limb size of 4–5 cm long), and histologically the limb cuticle was markedly thinned (approximately 1/20 the thickness of normal mature cuticle), had abnormal layering, and stained with a diffuse basophilic pallor (Fig. 4j, k). The underlying muscle in the area of limb regrowth showed features similar to those seen in primitive developing vertebrate myoblasts/myotubes, with small diameter fibers with basophilic sarcoplasm, lack of sarcomeres, and occasionally enlarged central nuclei (Fig. 5a). This animal also had focal ulcerative dermatitis with fungi on the cuticle of the opisthosoma away from the limb regrowth site.
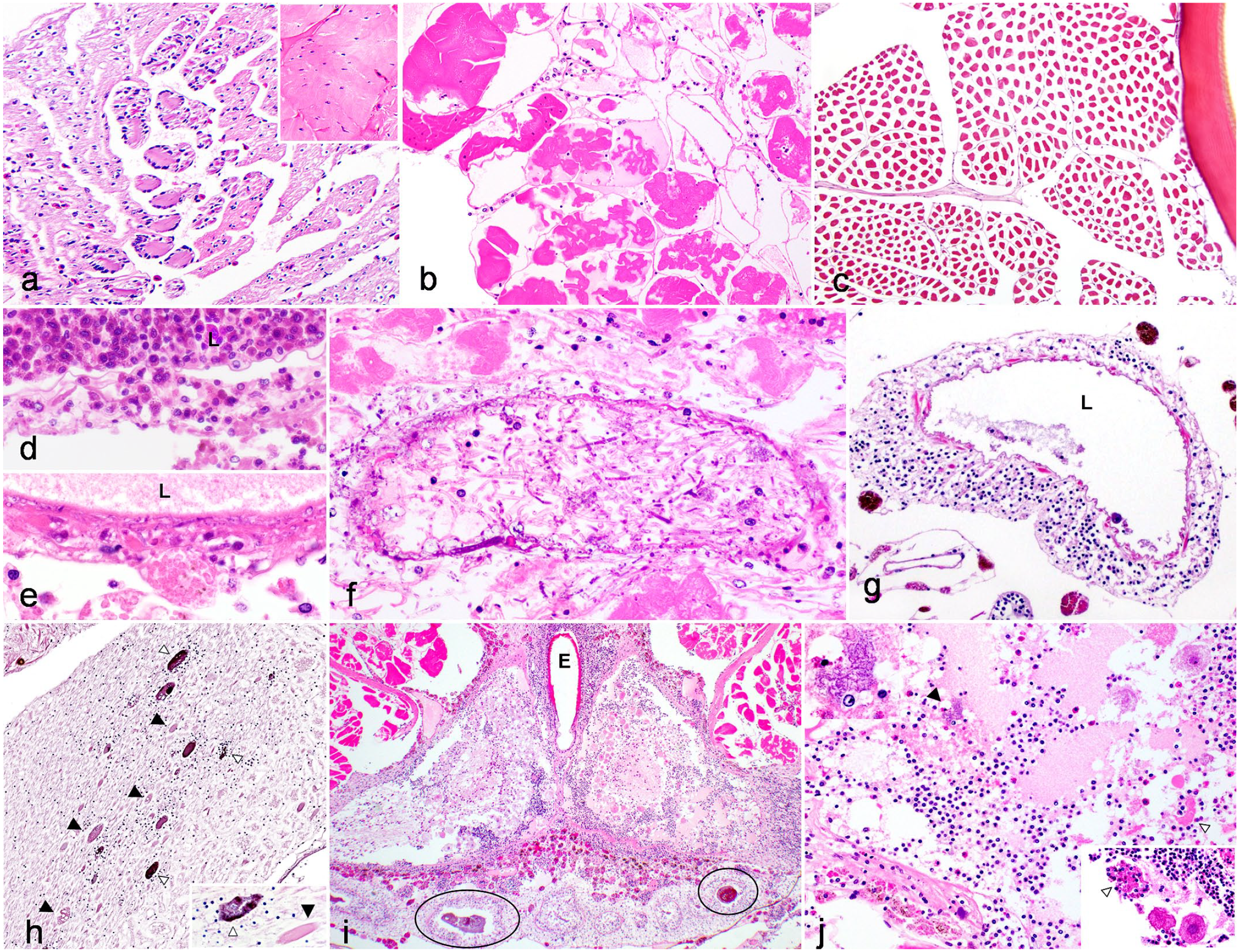
Histomorphology of skeletal muscle, vascular, and nervous system lesions in captive arachnids. Hematoxylin and eosin. (a) Myofiber regeneration in limb regrowth post-auto-amputation, Costa Rican zebra tarantula (Aphonopelma seemanni, Case 7). Myofibers are reduced in diameter with sarcoplasmic basophilia and hyperchromatic nuclei. Inset: normal skeletal muscle. (b) Skeletal myofiber degeneration and necrosis, prosoma, Brazilian red and white tarantula (Nhandu chromatus, Case 19). Rhabdomyolysis with sarcoplasmic hypereosinophilia, loss of cross striations, disruption and dissolution of the sarcoplasm, and accumulation of nuclear debris. (c) Skeletal myofiber atrophy, prosoma, Asian forest scorpion (Heterometrus longimanus, Case 13). Myofibers are reduced in diameter with detachment from the overlying endomysium. (d) Hemocytic aortic vasculitis, Chilean rose tarantula (Grammostola rosea, Case 6). Hemocytes infiltrate and expand the wall of the aorta, which is multifocally replaced by smudgy eosinophilic material (similar to fibrinoid vascular necrosis). The lumen (L) contains abundant hemocytes (hemocytosis). (e-f) Fungal vasculitis, Brazilian red and white tarantula (Nhandu chromatus, Case 19). The vessel lumen is indicated with an (L). (e) Invasion of the aorta by fungal hyphae intermixed with hemocytes and smudgy eosinophilic material. (f) Hyphae circumferentially infiltrate the vessel wall and extend into surrounding skeletal muscle undergoing rhabdomyolysis. Fungi have parallel 4 µm walls with septa and acute angle branching. (g) Prosoma perivascular hematopoietic tissue, Costa Rican zebra tarantula (Aphonopelma seemanni, Case 14). In contrast to cases of vasculitis, perivascular hematopoietic tissue cuffs but does not infiltrate or expand the vessel wall and is not associated with vascular degeneration. Lumen indicated with an (L). (h) Hemocytic neuritis, prosomal nerve, Brazilian salmon pink birdeater tarantula (Lasiodora parahybana, Case 8). Few non-granulated hemocytes infiltrate the nerve with multifocal axonal swelling and degeneration (black arrowhead, full size image and inset) and melanization (white arrowhead, full size image and inset). Inset: bacterial colonies are embedded within melanization (white arrowhead), and swollen axons resemble spheroid-like lesions (black arrowhead). (i-j) Hemocytic encephalitis with liquefactive necrosis, Brazilian red and white tarantula (Nhandu chromatus, Case 19). (i) There is widespread liquefactive necrosis and marked hemocytic encephalitis present in association with erosive midgut diverticulitis with bacteria and melanization (circles). The esophagus (E) traverses through the brain. (j) Within areas of necrosis, there are numerous hypereosinophilic neurons (neuronal necrosis, lower right inset) with neuronophagia (white arrowheads). Within areas of lytic necrosis, rare bacterial colonies (black arrowhead, upper left inset) were present.
The most common lesions affecting skeletal muscle were myositis (7/13 cases, 58%) and atrophy (6/13 cases, 50%). Myositis was characterized by infiltration, expansion, separation, and replacement of myofibers by granulated and nongranulated hemocytes, and in severe cases was defined by associated myofiber degeneration, fragmentation, and necrosis (Fig. 5b). Myositis was most commonly seen with systemic inflammation and hemocytosis (7/7 cases), with hemocytes infiltrating myofibers directly from the hemolymph of the open circulatory system. Myositis was also common as an extension from dermatitis (4/7) or ulcerative gastritis/midgut diverticulitis (2/7, 29%). Myocyte atrophy was characterized by focal, multifocal, or diffuse shrinking of myocytes away from the endomysium (Fig. 5c). This lesion was seen in a variety of scenarios, including the only 2 male animals examined and the case of uterine intussusception.
Circulatory System
Components of the circulatory system, including the heart (22/26 arachnids, 85%), hemolymph vessels (25/26 arachnids, 96%), and hemolymph sinuses (25/26 arachnids, 96%) were present for examination in the majority of cases. Hemocytic myocarditis was present in 8/26 arachnids (31%), and was characterized by infiltration, expansion, and disruption of the myocardium by hemocytes (Fig. 1e, 2a). Accompanying lesions included myofiber necrosis, melanization, and/or association with infectious agents, including bacteria (3/8 hearts) or a combination of fungi and bacteria (1/8 hearts). Myocarditis was consistently associated with multi-organ inflammation (8/8 hearts) and systemic hemocytosis (7/8 hearts). Vasculitis, characterized by disruption of aortic walls by hemocytes and fibrinoid vascular necrosis-like lesions, was uncommonly identified (2/25 arachnids, 8%, Fig. 5d, e). Both cases of vasculitis showed adjacent segmental ulcerative esophagitis and/or gastroenteritis within the prosoma. One case was associated with intralesional bacterial colonies (Fig. 5d, see also Fig. 1b, d) while the second case had a fungal and bacterial coinfection, with extensive fungal invasion of the aorta (Fig. 5e, f). Care was taken to distinguish true lesions of vasculitis from the presence of perivascular hemocytes within the prosoma, which we believe represents hematopoietic tissue (Fig. 5g).
Nervous System
The brain was identified in 16/26 animals (61%) while peripheral nerves were identified in 24/26 animals (92%). Hemocytic inflammation within the nervous system affected both the brain (4/16 animals, 25%) and peripheral nerves (7/24 animals, 29%). Histologic features of nervous system inflammation were similar to those seen in the nervous system of vertebrates, with axonal swelling/spheroid formation (Wallerian-type degeneration; Fig. 5h), neuritis, lytic necrosis, and encephalitis (Fig. 5i, j). Encephalitis and neuritis often represented direct extension of inflammation from adjacent prosomal organs. In 1 tarantula (case 19), severe necrotizing encephalitis (Fig. 5i, j) was a direct extension of adjacent ulcerative fungal and bacterial esophagitis, gastritis, and aortic vasculitis (Figs. 3d, 5e, f). Encephalitis and neuritis were frequently associated with systemic hemocytosis (3/4 arachnids; and 6/7 arachnids, respectively) and multi-organ inflammation (4/4 arachnids; and 7/7 arachnids, respectively). Infectious agents were uncommonly associated with encephalitis (mixed fungal and bacterial infection in 1 animal) and neuritis (bacterial infections in 2/7 animals; and fungal infection in 1 animal).
Discussion
In this study, we characterized the histomorphology of lesions in captive arachnids (Scorpiones and Araneae), animals that are commonly encountered in zoologic collections and the exotic pet trade. Inflammation characterized by hemocytes, melanization, and nodulation was the most common histologic abnormality. This histologic pattern is consistent with prior descriptions of inflammation in other invertebrate species.2–4,12,14,16,18,27,29 Inflammation was frequently associated with bacterial and fungal infections, suggesting that these infections may significantly contribute to captive arachnid morbidity and mortality. Infectious agent identification in this study was limited to histologic descriptions. Historically, further characterization of microbial infections in invertebrate species via traditional methods, such as bacterial and fungal culture, has been unrewarding due to postmortem bacterial/fungal overgrowth and the difficulty growing poikilotherm pathogens using traditional culture techniques. 26 Opportunistic invasion of the opisthosomal hemolymph and tissues by enteric bacteria may occur rapidly postmortem, and even occasionally antemortem in debilitated animals. 26 Similarly, postmortem overgrowth of saprophytic cuticular fungal infection may be difficult to differentiate from a true systemic entomopathogenic fungal infections on culture results alone. 26 Given these limitations, histologic identification of organisms within hemocytic inflammation may be a more sensitive and specific means for detecting true infections.
Advanced diagnostic techniques, including histochemical stains, polymerase chain reaction (PCR), and transmission electron microscopy are important tools that can aid in further characterization and identification of infectious agents in invertebrate species.2,12,13,17 Laser capture microdissection has been used in conjunction with PCR to identify pathogens within histologic lesions in veterinary species. 15 In this study, histochemical stains were sporadically utilized in cases to either characterize identifiable organisms or interrogate for the presence of potential infectious agents. The lack of application of a uniform panel of histochemical stains to cases with confirmed or suspect infectious agents, and lack of utilization of either PCR or electron microscopy, are inherent limitations to this study. Ultimately, as with any species, the ideal evaluation for presence of infectious agents would include a combination of histology, microbial culture, PCR, and electron microscopy, as applicable.
Previous descriptions of bacterial, fungal, and parasitic infections in captive arachnids are predominantly limited to clinical descriptions of suspected infections by organisms cultured either ante- or postmortem that lack documented histologic confirmation. Bacillus and Proteus spp. have been indicated as opportunistic infections in captive tarantulas, with the former isolated in low-humidity induced mortality events and the latter cultured in conjunction with Panagrolaimidae nematode infections. 26 Several organisms have been isolated from captive scorpions, including Bacillus megatherium, Pseudomonas diminuta, Staphylococcus aureus, Acinetobacter calcoaceticus, Moraxella sp., Serratia marcescens, Bacillus sp., E. coli, Proteus vulgaris, Lactobacillus, and Rickettsiella.11,12 However, aside from Rickettsiella spp., clinical signs and lesions associated with these isolated organisms have not been reported (ie, it is unclear if these organisms were identified in diseased or healthy scorpions). Bacteria isolated postmortem from captive nonarachnid invertebrates include Serratia marcescens and Pseudomonas aeruginosa in Lord Howe Island stick insects 2 and Pseudomonas putrefaciens, Aeromonas hydrophila, and Micrococcus sp. in horseshoe crabs. 18 Notably, in the study documenting mortality of Lord Howe Island stick insects, postmortem bacterial culture frequently isolated mixed infections and this precluded definitive identification of pathogens. 2 While this study is limited by the lack of bacterial culture, histologically bacterial agents were cocci or short coccobacilli that formed large extracellular colonies. In the 3 cases where Gram stain was applied, organisms were gram positive. Example genera of bacteria considered for these findings are Staphylococcus, Streptococcus, or Corynebacterium spp., amongst others. Whether invertebrates are susceptible to infection by the latter 2 genera is unknown. Given the tenuous association of previously reported invertebrate bacterial agents with disease, as well as the overall low number of animals examined in this study, meaningful comparison is limited.
Rickettsiella spp. have been reported as pathogens in emperor scorpions as well as other arthropods.11,12 In this study, lesions described with Rickettsiella infection were not identified in any animal, but only 2 emperor scorpions were examined. Notably, Rickettsiella spp. may be more challenging to detect histologically, and in previous studies were visualized more apparently with application of a variety of histochemical stains, including GMS, Fite’s acid fast, and Machiavello stains. 12 In the 6 scorpions examined in this study, there was only 1 bacterial infection, which was restricted to cuticular colonization of an arthrodial membrane erosion (case 13, Heterometrus longimanus).
Fungal infections in invertebrates have been previously classified as opportunistic cuticular infections by saprophytic fungi or systemic infections by entomopathogenic fungi. 26 However, fungal pathogens of tarantulas and scorpions have not been characterized to the level of genus and species. Fungal infections were found to be common in captive horseshoe crabs in a retrospective histological study, with Fusarium spp. isolated in some cases. Infections were associated with external lesions, such as cutaneous ulceration, branchitis, and ophthalmitis. 18 In this study, entomopathogenic infections and cuticular infections were equally represented. Of these, only 1 animal had a definitive cuticular and systemic fungal infection, suggesting that systemic spread of cuticular fungi is uncommon. Based on histologic morphology alone, Fusarium infection could be possible in cases in this study. However, similar to the limitations of histologic bacterial identification, identification of fungi is speculative at best given the lack of fungal culture and/or molecular characterization. Microsporidial fungi (Nosema spp.) are considered major pathogens of honey bees. 13 Organisms consistent with microsporidia were not identified in any of the currently reviewed arachnid cases.
Endo- and ectoparasitic infections of arachnids include nematodes as well as wasps, flies, and mites. 26 Oral Panagrolaimidae nematodes have been reported in tarantulas, 26 while systemic nematodiasis has been sporadically reported in both tarantulas and scorpions.11,26 The nematode larvae seen in a single gravid female scorpion in this study was not morphologically consistent with any previously described nematods in arachnids. This organism is suspected to represent an opportunistic infection, perhaps secondary to immunosuppression due to pregnancy, although this is speculative in invertebrate species. In 1 case, the extensive arthropod larval infection may reflect parasitism by either pompilid or sphecidid wasps, with fly larval infestation considered less likely. 26 The presence of this lesion in an animal from a zoologic collection underscores the importance of husbandry and biosecurity in caring for invertebrates, including the importance of keeping native insects from entering enclosures. In both of these cases, there were limited inflammatory responses to the parasites; however, the low number of cases examined in this study precludes meaningful interpretation of this finding.
Viral infections have been reported in spiders and scorpions,11,26 including a single case report of a baculovirus in a nursery web spider, but gross and histologic lesions associated with viral infection have not been characterized. In this study, there was no histologically detectable features of viral infection, such as inclusion bodies, which can be seen in other invertebrates; 25 however, it is possible that unidentified viruses may have contributed to disease. Future similar studies should continue to be open to this possibility.
Frequent sites of infection and inflammation identified in this study included the digestive tract, particularly the midgut diverticula, and to a lesser extent, the book lungs and cuticle/body wall. This tissue infection pattern is similar to findings previously described in horseshoe crabs. 18 Infection at these 3 sites is likely opportunistic with a multi-factorial pathogenesis. First, we hypothesize that consumption of abrasive substrate and insects may cause micro-trauma of the noncuticle lined segments of the digestive tract (ie midgut tube, stercoral sac), leading to colonization and infection by intestinal yeast and bacteria. The labyrinthine anatomy of the midgut diverticula provides ample opportunity to trap ingesta with subsequent epithelial injury. Midgut diverticula interstitial cells, which store lipids, proteins, and glycogen18,19 are likely to provide abundant nutrients to organisms to serve as an optimal “growth media.” Second, the ventrally oriented book lungs are in direct opposition to substrate and may be prone to traumatic injury and accumulation of environmental organisms. Third, while the cuticle is a formidable barrier to infection, some anatomic areas, such as arthrodial membranes, which lack an exocuticle, 26 or the thinner cuticle overlying the ventral opisthosoma may be more prone to traumatic erosion and infection. This theory is supported by the propensity for cuticle lesions to affect arthrodial membranes and ventral opisthosoma in this study. Furthermore, we speculate that the process of dysecdysis may leave an arachnid susceptible to disease due to cuticular barrier compromise 26 or systemic immunosuppression secondary to increased metabolic demand.
Systemic and/or multi-organ spread of bacteria and fungi was common in our study and was likely enabled by multiple factors. First, systemic spread appeared facilitated by the vascular system as evidenced by the high incidence of myocarditis, vasculitis, or intravascular/intra-hemolymph organisms. The open circulatory system of invertebrates, which lacks capillaries and relies on pressure gradients for return of venous blood to the heart, may predispose these animals to widespread multi-organ infection and rapid debilitation once the animal is septicemic. Second, arachnid anatomy apparently predisposed few animals to a unique combination of lesions. In the prosoma, the digestive tract, brain, and aorta are in intimate contact with one another, with the esophagus passing directly through the brain. In 2 cases in this study, ulcerative gastritis was associated with infection of other prosomal organs from enteric flora, resulting in a florid encephalitis and vasculitis in 1 case. In the opisthosoma, the heart runs along the dorsal midline ventral to the cuticle and immediately adjacent to the midgut diverticula. In 1 case in a teaching animal, repeated cardiac puncture resulted in a fungal and deep bacterial deep dermatitis, myocarditis, and regional hemocoelomitis. These findings reinforce that consideration of the unique anatomy of arachnids is critical in medical management and in the accurate trimming and histologic evaluation of tissues.
In addition to inflammation, atrophy of the midgut diverticula and/or skeletal muscle was common. Atrophic lesions were interpreted as indicators of decreased nutritional status18,21 and have been documented in other arachnids in times of starvation. 19 During starvation, electron microscopy of midgut diverticula cells has demonstrated increased autophagy and reduced intracellular lipid, glycogen, and protein, indicative of depletion of energy stores. 19 Atrophic lesions were commonly identified in animals with infections, and we suspect this reflects a combination of increased metabolic demand and general poor-doing secondary to systemic disease. These lesions were present in gravid females and all males in the study. We suspect that these individuals are under higher metabolic demands and may have increased and/or more specialized nutritional requirements that lead to depletion of energy stores in the midgut diverticula. Appropriate nutrition has a direct impact on reproductive performance in female spiders.22,28,30 Furthermore, male tarantulas have a much shorter life span (1–2 years postreproductive maturation) than females (up to 20 years postreproductive maturation), which has been attributed to higher metabolic rate required to actively search for mates with subsequent cellular oxidative damage.7,26 In general, the nutritional requirements of arachnids are poorly understood, and diverse prey diets have been shown to be required in some spiders. 26 Nutritional needs should be considered in the management of arachnids which are breeding or showing signs of disease, and invertebrate husbandry would likely benefit from future nutritional studies of arachnids in different life stages.
Interestingly, neoplasia was not identified in any arachnid in this study. Neoplasia has been documented across several taxa of invertebrates, 24 including a single report in an orb-weaver spider, 20 but is likely under-recognized in invertebrates due to overall lack of histologic evaluation and pathologist unfamiliarity with arachnid tissue histomorphology. We suspect that as invertebrate pathology becomes more widespread amongst veterinary pathologists that more neoplastic diseases will be identified.
Correlation of histologic lesions to antemortem clinical data is challenging in this study due to limited reporting and/or observation of morbidity in these animals. A few connections can be made despite this limitation. All animals with reported molting issues and/or dysecdysis had histologically confirmed cuticular abnormalities, including confirmed dysecdysis and/or cuticular regrowth as well as fungal and/or mixed fungal and bacterial dermatitis. Two of these animals also showed systemic inflammation. Collectively, these observations suggest that animals with clinically identified dysecdysis may be at risk for either further cuticular injury and/or opportunistic infection, and should be monitored and/or treated appropriately. None of the 3 animals with reports of suspected trauma had histologic features to support traumatic injury. All reported traumatic incidents were considered mild (eg, dropped by owner, suspected conspecific fighting) with either no overt gross lesions or clinical resolution of gross lesions by the time of death, and so that, we suspect that reports of “trauma” were unrelated to death. Finally, all 3 animals for which lethargy was observed had multi-organ inflammation and infection of at least 1 visceral tissue by either fungi or bacteria. Combined with the high prevalence of infectious disease identified in this study, we recommend that infectious agents are prioritized as differential diagnoses for spiders and scorpions exhibiting nonspecific signs of ill thrift, such as lethargy.
Several limitations to this study preclude extensive conclusions. First, the number of animals examined is relatively low, and the animals were all from a small geographic region with the majority being housed at the same institution with similar climatic conditions, husbandry practices, and degree of handling. This introduces sampling bias and it is unclear whether the frequency and types of lesions identified in this study can be broadly extrapolated to other populations of captive arachnids. Future studies would benefit from sourcing arachnids from several institutes that both span a larger geographic area as well as represent a diversity of husbandry and handling scenarios. Second, lack of bacterial and fungal culture, viral isolation, molecular diagnostics, or transmission electron microscopy limits the identification of infectious agents. We recommend a multimodal approach to postmortem evaluation of arachnids that includes prospective collection and appropriate storage of fresh tissues commonly affected by infectious agents (midgut diverticula, book lung, and cuticle) for culture, molecular diagnostics, and electron microscopy.2,12 Third, postmortem autolysis limited histologic tissue evaluation. Postmortem overgrowth of bacteria and fungi made interpretation of some histology challenging and 16% of archived specimens were excluded due to autolysis. Future studies of arachnids should optimize tissue fixation to maximize the utility of histologic evaluation.
To this point, given that many pathologists are unfamiliar with processing arachnids, we would be neglect to not briefly discuss important steps that aided in histologic evaluation in this study. First, appropriate and rapid fixation of tissues, preferably of the whole animal, is critical to maintain tissue integrity, prevent autolysis, and minimize postmortem overgrowth of bacteria and fungi. We suggest removal of all limbs and parasagittal/lateral incision of the opisthosoma to allow for penetration of fixative. Second, optimal tissue processing prior to and after trimming requires incubation in a number of different solutions, including formalin, formic acid, ethanol, and Bouin’s or Davidson’s solutions. The methodology described in this study produces excellent slides. Improvements to this methodology since completion of this study include the use of Davidson’s solution in place of Bouin’s solution, in which formic acid is often not required. Third, we recommend examining the animal in several serial transverse sections that captures the entire prosoma and opisthosoma. This will vary by animal size, but in our experience in average adult tarantulas and scorpions includes 2–4 sections of the prosoma and 3–5 sections of the opisthosoma. Legs can be examined if clinically relevant (ie, in cases of known autotomy or limb trauma), but in our experience few lesions of overall significance (ie, contributors to death) are identified in the limbs. Last, given this method of whole-body evaluation, deeper sections through tissue blocks may be required for thorough evaluation of histologic lesions. In this study, evaluation of several serial sections of tissue was required to identify lesions in multiple arachnids.
Our study identifies and describes histologic lesions in captive arachnids submitted for histologic evaluation in North Carolina. Our findings are a resource for veterinary pathologists who encounter arachnids in their diagnostic or research case load. Further studies are needed to add to the arachnid pathology knowledge base, particularly to explore the mechanisms underlying bacterial and fungal infection in captive arachnids.
Supplemental Material
sj-pdf-2-vet-10.1177_03009858231162948 – Supplemental material for Retrospective analysis of histologic lesions in captive arachnids
Supplemental material, sj-pdf-2-vet-10.1177_03009858231162948 for Retrospective analysis of histologic lesions in captive arachnids by Chris Gaudette, Elise E. B. LaDouceur, Brigid V. Troan, Nathan Whitehurst, Daniel S. Dombrowski, Gregory A. Lewbart, Keith E. Linder, Kent Passingham, Larry S. Christian and Megan E. Schreeg in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858231162948 – Supplemental material for Retrospective analysis of histologic lesions in captive arachnids
Supplemental material, sj-xlsx-1-vet-10.1177_03009858231162948 for Retrospective analysis of histologic lesions in captive arachnids by Chris Gaudette, Elise E. B. LaDouceur, Brigid V. Troan, Nathan Whitehurst, Daniel S. Dombrowski, Gregory A. Lewbart, Keith E. Linder, Kent Passingham, Larry S. Christian and Megan E. Schreeg in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank the NC State University College of Veterinary Medicine Histology Lab for their contribution. They would also like to thank the staff at the North Carolina Museum of Natural Sciences and Dr. Kelsie Dougherty for their assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
