Abstract
Despite increasing concern for coral reef ecosystem health within the last decade, there is scant literature concerning the histopathology of diseases affecting the major constituents of coral reef ecosystems, particularly marine invertebrates. This study describes histologic findings in 6 species of marine invertebrates (California sea hare [Aplysia californica], purple sea urchin [Strongylocentrotus purpuratus], sunburst anemone [Anthopleura sola], knobby star [Pisaster giganteus], bat star [Asterina miniata], and brittle star [Ophiopteris papillosa]) with spontaneous copper toxicosis, 4 purple sea urchins with experimentally induced copper toxicosis, and 1 unexposed control of each species listed. The primary lesions in the California sea hare with copper toxicosis were branchial and nephridial necrosis. Affected echinoderms shared several histologic lesions, including epidermal necrosis and ulceration and increased numbers of coelomocytes within the water-vascular system. The sunburst anemone with copper toxicosis had necrosis of both epidermis and gastrodermis, as well as expulsion of zooxanthellae from the gastrodermis. In addition to the lesions attributed to copper toxicosis, our results describe normal microscopic features of these animals that may be useful for histopathologic assessment of marine invertebrates.
There is increasing concern about the degradation of the world’s coral reef ecosystems. 3,9,37 Due to the interdependence of animals and plants in reef communities, shifts in abundance and diversity strongly affect the various trophic levels in these complex ecosystems. 3,19,39 Despite increased attention to reef ecosystem health within the last decade, there remains relatively scant literature concerning the histopathology of diseases affecting the major constituents of coral reef ecosystems, particularly marine invertebrates. 16,40
To address the need for better information relevant to coral conservation, the National Oceanic and Atmospheric Administration sponsored a workshop to develop a consensus on the histopathology and terminology of several common coral diseases. 12,29 No such consensus has been reached for the majority of marine invertebrates, and gaps as well as inconsistencies exist in the terminology and reports of pathophysiology, microanatomy, and embryology for these groups. Research in corals has shown that some cnidarian lesions that appear similar grossly are widely divergent microscopically. 35 Systematic gross and microscopic descriptions, as well as attempted identification of etiologic agents, are important steps to developing an understanding of the pathogenesis of disease in these animals. 41
Copper is a common pollutant of marine ecosystems that emanates from industrial effluent, urban/agricultural runoff, sewage treatment discharge, heavy metal mining areas, and antifouling paints and refineries. 24,27 Marine organisms can concentrate heavy metals from 10 to >20 000 times the environmental level, causing extremely high tissue loads. 10 The toxic effects of heavy metals are well documented in a variety of marine organisms, but there is limited research on the histologic lesions of heavy metal toxicosis in marine invertebrates other than molluscs. 13 Copper exposure occurs in wild populations due to a variety of anthropogenic activities and in captive populations secondary to accidental exposure (eg, water contamination from copper pipes, brass bushings, and substrates), algae eradication, or protozoacide treatment in mixed-species exhibits. 14,16,21,26
This study details the histologic lesions and normal histology of target tissues in marine invertebrates with and without copper toxicosis, respectively. Microscopic features are described in 6 species: California sea hare (Mollusca: Gastropoda: Aplysia californica), purple sea urchin (Echinodermata: Echinoidea: Strongylocentrotus purpuratus), sunburst anemone (Cnidaria: Anthozoa: Anthopleura sola), knobby star (Echinodermata: Asteroidea: Pisaster giganteus), bat star (Echinodermata: Asteroidea: Asterina miniata), and brittle star (Echinodermata: Asteroidea: Ophiopteris papillosa).
Materials and Methods
Three groups were examined histologically: Group 1: 6 animals with spontaneous copper toxicosis—a California sea hare, purple sea urchin, sunburst anemone, knobby star, bat star, and brittle star Group 2: 4 purple sea urchins with experimentally induced copper toxicosis Group 3: 6 wild-caught animals representing all aforementioned species, used as histologic controls
Group 1 was submitted in 10% neutral buffered formalin by an aquarium in Southern California that had a mass mortality event due to copper toxicosis; 333 of 441 wild-caught marine invertebrates died due to a water copper concentration of 33 ppb. 42 The clinical investigation and diagnostic methods used to determine that copper was the etiologic agent are available. 42 Six animals were euthanized with 30% magnesium sulfate (Safeway Inc, Pleasanton, CA) and submitted for histologic examination. Following fixation in 10% neutral buffered formalin, entire specimens or calcified tissues from purple sea urchins and sea stars were decalcified using 0.5 M ethylenediamine tetra-acetic acid, pH 8.0 (Fisher Scientific, Pittsburg, PA) for 24 to 72 hours prior to trimming. Serial transverse sections of entire specimens were embedded in paraffin, sectioned at a thickness of 5 µm, and stained with hematoxylin and eosin.
Group 2 was submitted in formalin by the same aquarium and consisted of 4 purple sea urchins that were experimentally exposed to 40-ppb copper in the water. 42 For this experiment, 4 newly caught purple sea urchins from the same wild population as group 1 were placed in a test tank with 2-ppb copper for 1 week. 42 At the end of 1 week, copper was added to the test system until the water concentration equilibrated to 40 ppb. 42 When purple sea urchins became moribund, they were euthanized and examined histologically via the aforementioned protocols.
Group 3 consisted of 6 apparently healthy marine invertebrates representing the aforementioned species submitted in formalin by the same aquarium. These animals were caught from the same wild population as previous groups and were used as histologic controls. These newly caught animals were euthanized and examined histologically through the aforementioned protocols.
A checklist of organs/components to be evaluated histologically was developed for all 4 orders represented (Gastropoda, Anthozoa, Asteroidea, Echinoidea). This checklist was designed to focus on primary organs only. An exhaustive study of all organs in 6 species of invertebrates was considered beyond the scope of this study. Classic texts were used as guidelines for the nomenclature 6,8,11,23,31 ; these references were supplemented as needed with primary literature.
Results
Tissues that were evaluated histologically are listed in Table 1. Histologic findings were similar across some species or groups (ie, purple sea urchins from groups 1 and 2; all echinoderms from group 1; and all echinoderms from group 3); most of the findings in these animals were described collectively. Normal histology and histopathologic findings were organized according to organ systems. Histologic lesions are summarized in Table 2.
Anatomic Features for Evaluation by Light Microscopy in Sea Hare, Sea Star, Sea Urchin, and Anemone.
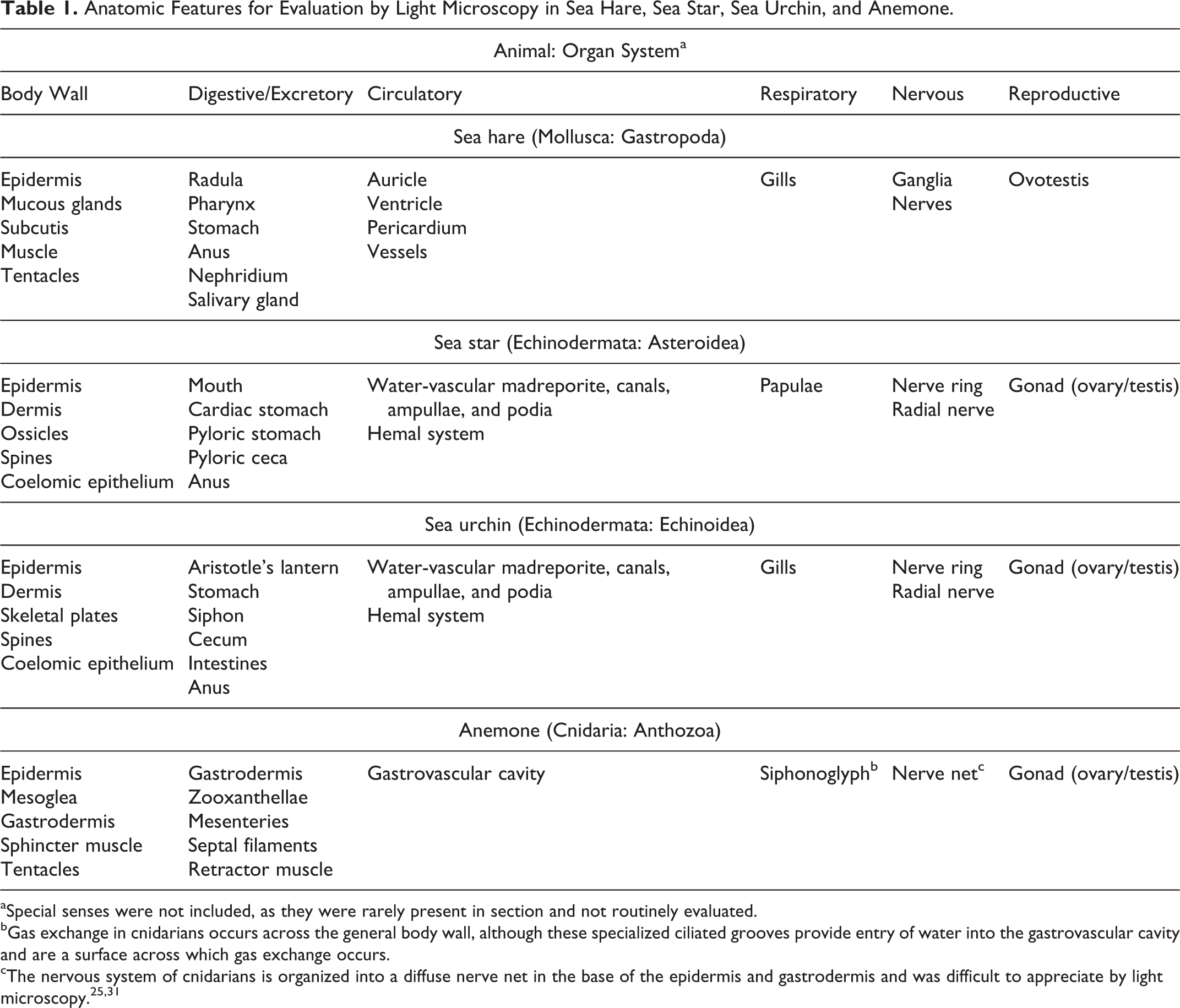
aSpecial senses were not included, as they were rarely present in section and not routinely evaluated.
bGas exchange in cnidarians occurs across the general body wall, although these specialized ciliated grooves provide entry of water into the gastrovascular cavity and are a surface across which gas exchange occurs.
Histologic Lesions Attributed to Copper Toxicosis in a California Sea Hare, 3 Sea Stars (Knobby, Bat, Brittle), Purple Sea Urchin, and Sunburst Anemone.
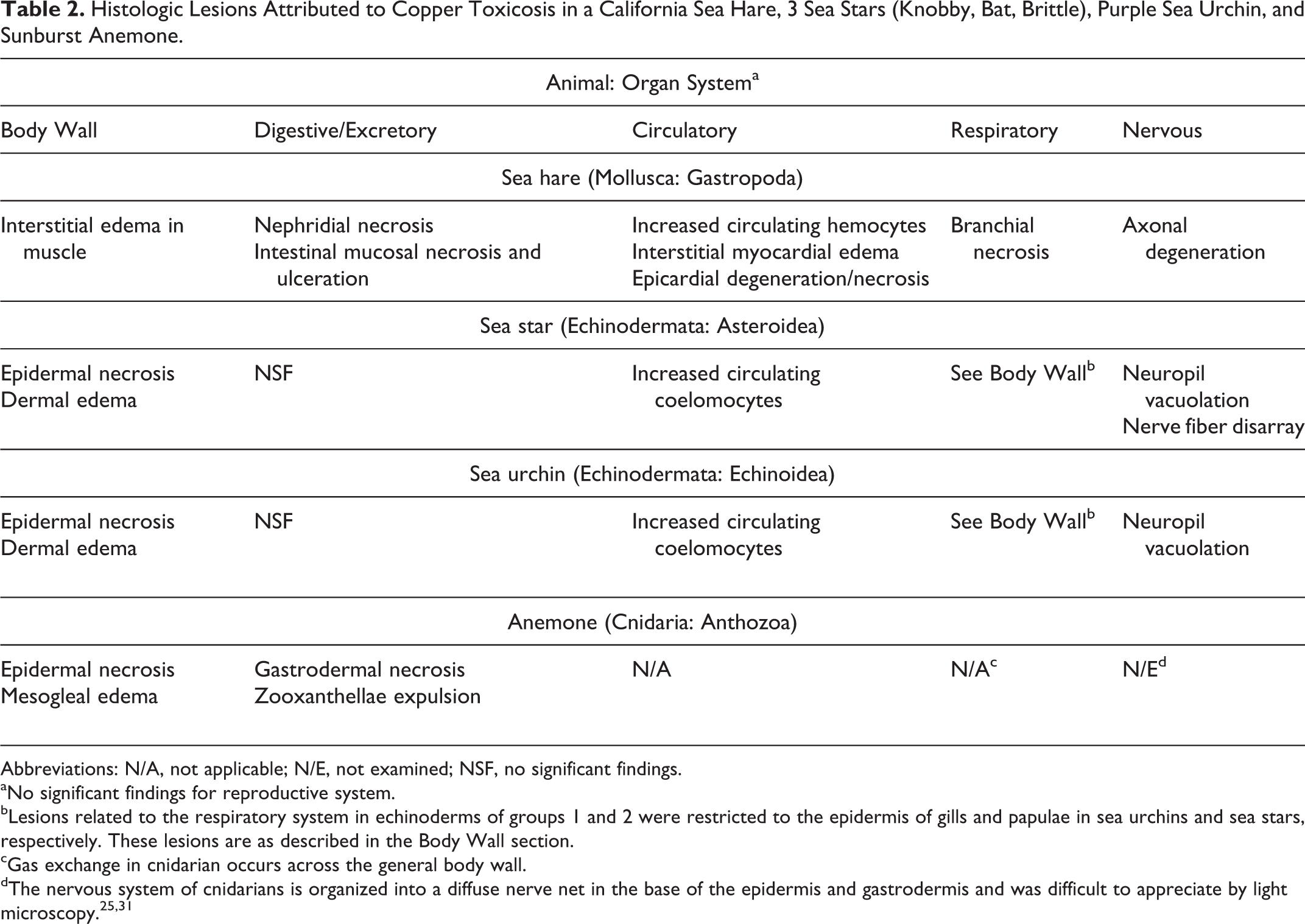
Abbreviations: N/A, not applicable; N/E, not examined; NSF, no significant findings.
aNo significant findings for reproductive system.
bLesions related to the respiratory system in echinoderms of groups 1 and 2 were restricted to the epidermis of gills and papulae in sea urchins and sea stars, respectively. These lesions are as described in the Body Wall section.
cGas exchange in cnidarian occurs across the general body wall.
Body Wall
The California sea hare epidermis is composed of a variably pigmented, simple, cuboidal to columnar, variably ciliated and microvillus epithelium. The epidermis is covered by a fine mucous layer and subtended by subepidermal mucous cells. 23,33 Within the dermis, myocytes and collagen bundles are arranged in orthogonal to random orientation, and vascular channels are scattered. 23,33 In comparison with the pharyngeal muscle of the California sea hare in group 3, myocytes of the pharyngeal muscle in group 1 were separated and replaced by increased numbers of variably sized, clear vacuoles and increased basophilic, mucinous matrix (Figs. 1, 2). The remainder of the body wall was unremarkable in California sea hare in groups 1 and 3.
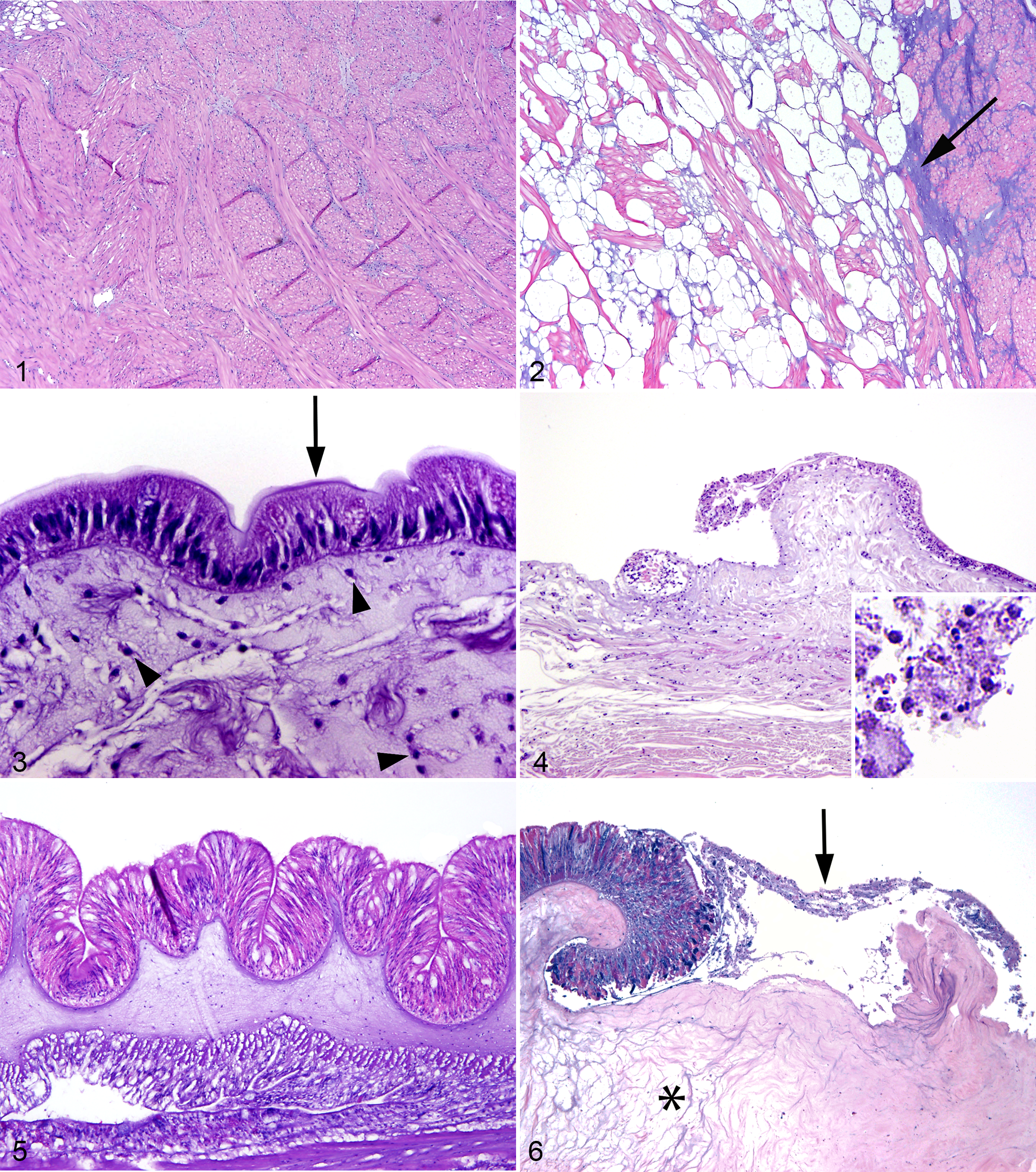
The body wall of echinoderms is composed of a fine outer cuticle, epidermis, dermis, and coelomic epithelium. 31 The simple columnar epidermis has variably ciliated, microvillus epithelial cells and mucus-secreting cells (Fig. 3). 31 The dermis is generally composed of specialized collagen called mutable collagenous tissue and an endoskeleton composed of spines and either ossicles or sutured skeletal plates in sea stars and sea urchins, respectively. 6,8,31 Coelomic epithelium covers the interior of the body wall, the outer surface of the digestive and reproductive systems, and the outer and inner surfaces of some segments of the water-vascular system (see Circulatory System section). 8 This simple coelomic epithelium comprises myoepithelial, supporting, and secretory cells of varying shape (commonly low cuboidal), with microvilli and occasionally cilia. 8 In echinoderms from group 1, epidermal cells were variably swollen and pale with fragmented cytoplasm and karyorrhectic, pyknotic, or absent nuclei (necrosis). Multifocally, the epidermis had small ulcers subtended by dermal edema (Fig. 4). Ossicles and skeletal plates of echinoderms in groups 1 and 3 frequently had loss of the center of their lattice; the border of these central defects had spicules of compressed lattice admixed with increased density of intraossicle cells (presumed sclerocytes and coelomocytes). The cuticle was frequently absent in echinoderms in group 1 and occasionally absent in group 3.
The sunburst anemone body wall is composed of 3 layers: epidermis, mesoglea, and gastrodermis (Fig. 5). 11 The tentacles are composed of evaginations of all 3 layers extending from the body wall around the oral disc. 11 The most abundant cells in the epidermis are simple, elongated, columnar, myoepithelial cells; less common are interstitial cells, mucus-secreting cells, receptor/nerve cells, and cnidocytes, which contain cnidae, an eversible cnidarian organelle that commonly functions in stinging. 31 In the sunburst anemone in group 1, epidermal cells were variably swollen and pale with fragmented cytoplasm and karyorrhectic, pyknotic, or absent nuclei (necrosis); the epidermis was multifocally ulcerated; and the mesoglea subtending ulcers was edematous (Fig. 6).
Digestive/Excretory System
The California sea hare was the only examined species that is considered to have a true excretory system. In the California sea hare in group 3, nephridial lobules were well defined and contained nephrocytes that variably had yellow-brown cytoplasmic concretions (excretory concretions; Fig. 7). In group 1, nephridial lobular outlines were evident, but individual nephrocytes could not be discerned, as the lobule was filled with an amalgamation of heterogeneous eosinophilic to basophilic karyorrhectic debris; the structure of excretory concretions was maintained (Fig. 8).
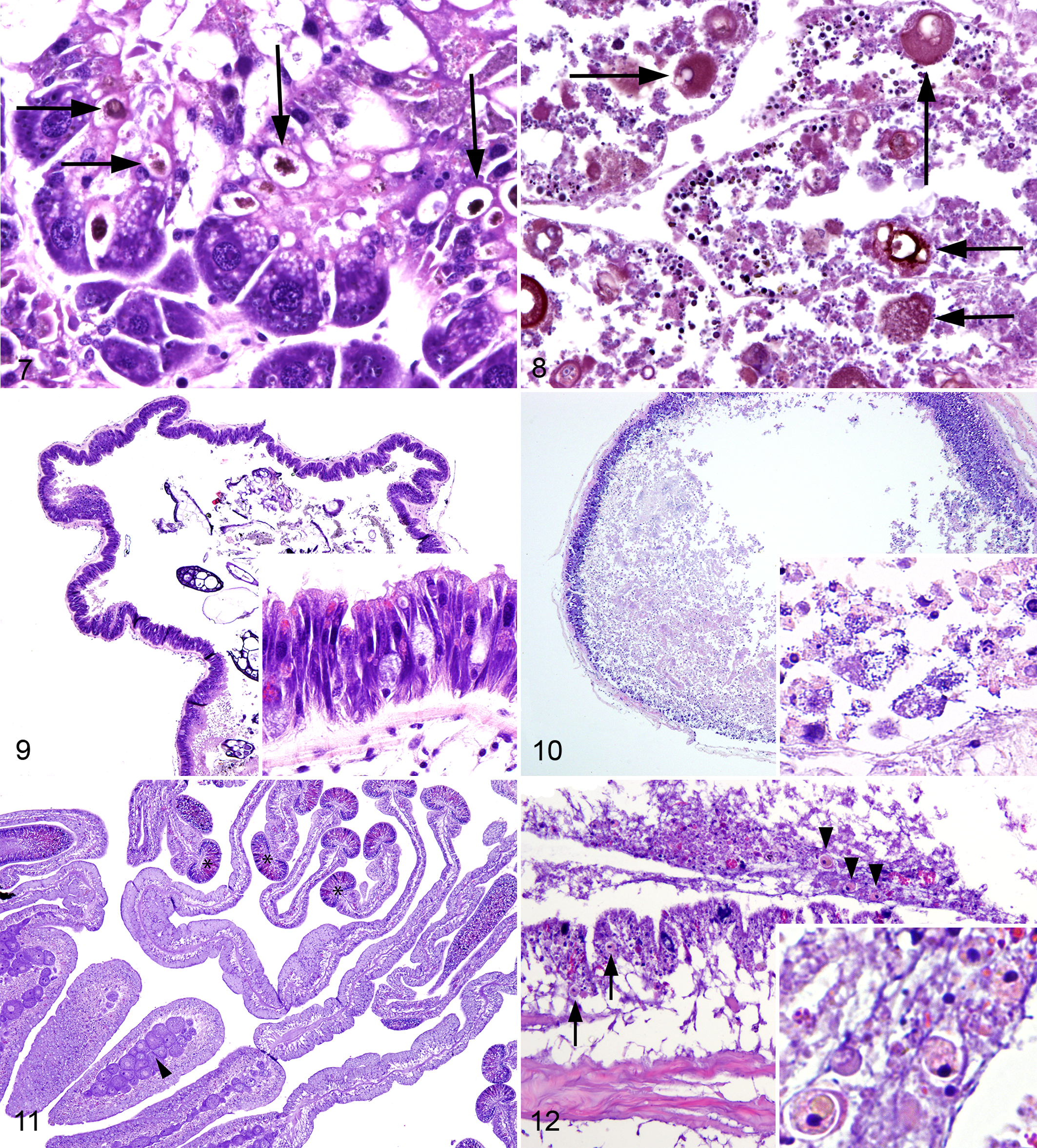
The digestive system of the California sea hare starts at the radula, which has chitinous teeth, and continues to the esophagus, stomach (also called gizzard due to its chitinous inner membrane), intestines, and anus. 31 In the California sea hare in group 3, ciliated and mucous cells were present throughout the tract and ranged from simple columnar to pseudostratified (Fig. 9). In the California sea hare in group 1, epithelial and glandular cells were variably swollen and pale with fragmented cytoplasm and karyorrhectic, pyknotic, or absent nuclei (necrosis); necrotic cells were frequently detached from the basement membrane and free in the digestive tract lumen where they were admixed with small, granular basophilic material (presumed gland cell secretions and bacteria; Fig. 10).
The digestive system of sea stars is broadly arranged into a mouth, 2 stomachs, and pyloric ceca. In purple sea urchins, the digestive tract is divided into the mouth, stomach (which has a parallel canal called a siphon), cecum, intestines, and anus. Additionally, purple sea urchins have a specialized masticatory apparatus called Aristotle’s lantern. The histologic organization of the echinoderm gastrointestinal tract is similar throughout the tract and is arranged into a gastrodermis, connective tissue layer, and coelomic lining. 6,8 The gastrodermis of echinoderms in groups 1–3 was generally pseudostratified, microvillus, variably ciliated, and columnar with large numbers of cells containing mucus or other secretory products; no lesions were observed.
The sunburst anemone gastrovascular cavity is composed of mesenteries with trilobate or unilobate filaments (Fig. 11), all of which are overlain by a simple gastrodermis composed of myoepithelial cells, mucus-secreting cells, glandular cells, and cnidocytes. 11,31 Retractor muscles occur on one face of the mesenteries and are arranged in longitudinal, pleated sheets composed of 2 monolayers of contractile cells within a central band of mesoglea, to which they are anchored. 11 In the sunburst anemone in group 1, gastrodermal cells were multifocally swollen and pale with fragmented cytoplasm and karyorrhectic to pyknotic nuclei (necrosis); the gastrodermis was variably ulcerated. In the sunburst anemone in group 1, zooxanthellae were present in the gastrodermis or were occasionally free in the gastrovascular cavity (expulsion) admixed with karyorrhectic debris and necrotic gastrodermal cells (Fig. 12). Zooxanthellae were not identified in group 3. Retractor muscles were within normal limits in groups 1 and 3.
Circulatory System
The circulatory system of California sea hare is open and includes a heart that lies in a pericardial sac. The outer surface of the heart is overlain by a simple epicardium, the cells of which are columnar when the heart is contracted and flattened when the heart is distended. The heart is composed of a trabecular arrangement of striated muscle fibers that are not lined by an endocardium (Fig. 13). 23 In the California sea hare in group 3, cardiac chambers contained clear space, and vessels contained small numbers of hemocytes (formerly called amebocytes). 22 In the California sea hare in group 1, the cardiac chambers contained mildly increased numbers of hemocytes, and the myocardium was expanded by interstitial edema (Fig. 14). In the same animal, the epicardium was frequently absent; in areas of remnant epicardium, the cells were piled and had fragmented cytoplasm.
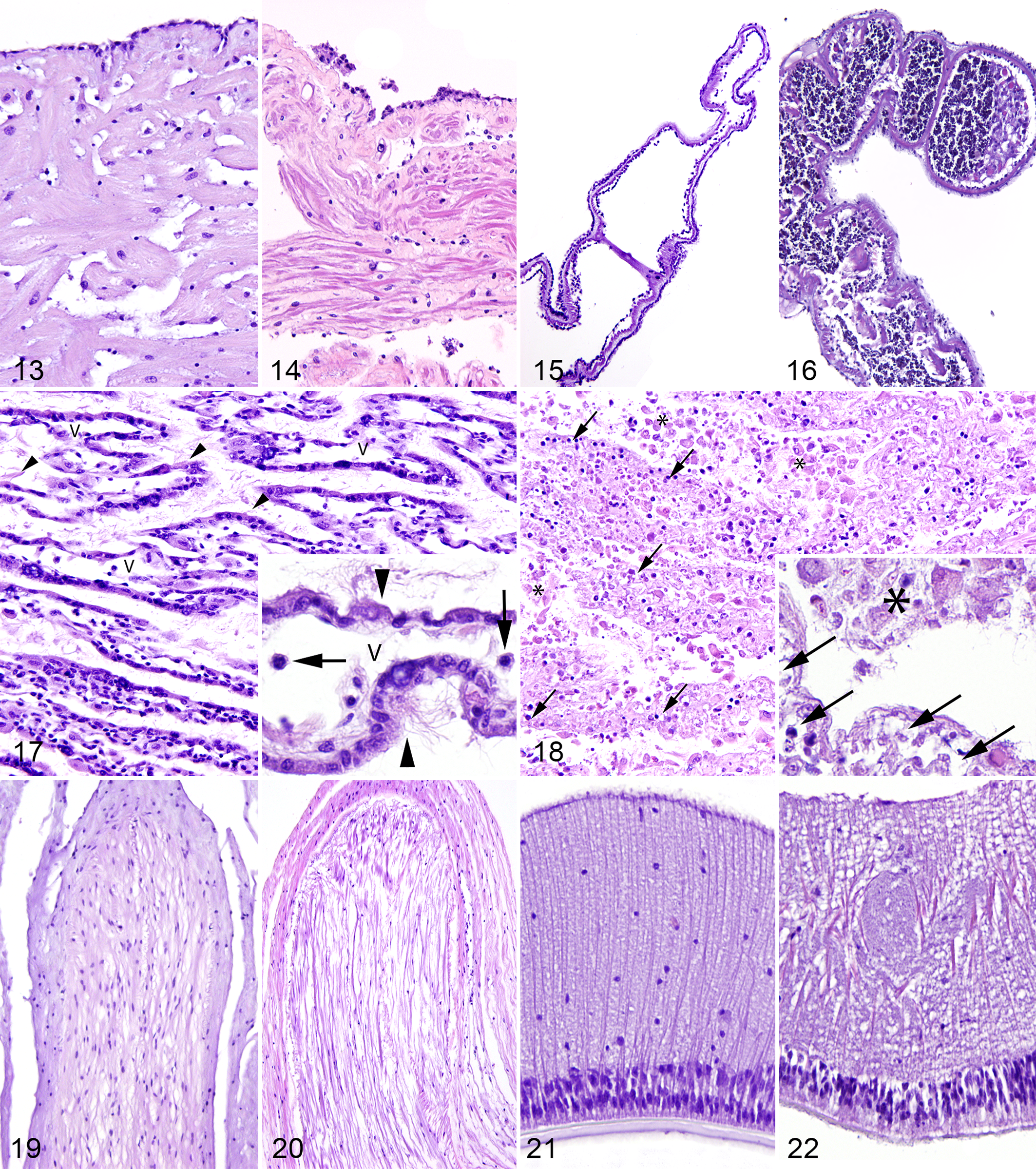
The circulatory systems of echinoderms include a water-vascular system used for locomotion and filtering of external water, as well as a hemal system that produces an internal current of coelomic fluid. 31 The hemal system is rudimentary, and circulation is largely facilitated by simple, ciliated coelomic epithelium. The water-vascular system consists of canals and appendages of the body wall that originate at the madreporite on the aboral surface, lead to a series of canals that open into small muscular sacs called ampullae, and terminate in tube feet, or podia, on the oral surface. 31 The inner lining of the madreporite and stone canal in echinoderms in groups 1–3 was pseudostratified, microvillus, and ciliated. The remaining canals were lined internally by coelomic epithelium (see Body Wall) subtended by connective tissue. 8 Podia in echinoderms in groups 1–3 had similar layers to the body wall (ie, outer epidermis, midlayer of connective tissue, and inner coelomic lining), although musculature of the myoepithelium was more extensive. The water-vascular system of echinoderms in group 3 contained scant, coelomic fluid and rare, small aggregates of coelomocytes (Fig. 15). In echinoderms in groups 1 and 2, the water-vascular system contained a small amount of coelomic fluid and moderately sized to large aggregates of coelomocytes (Fig. 16).
Cnidarians do not have a true circulatory system, as the “gastrovascular cavity” functions in digestion and circulation (see Digestive/Excretory System). 31
Respiratory System
The gills of the California sea hare in group 3 formed lamellar structures composed of vascular spaces that contained clear space and rare hemocytes (Fig. 17). 2,33 Gills from the California sea hare in group 3 were lined by highly ciliated epithelium interspersed with small numbers of secretory cells supported by variable amounts of delicate muscular trabeculae. In the California sea hare in group 1, muscle trabeculae were no longer lined by ciliated epithelium, and epithelial cells were sloughed into the mantle cavity; sloughed cells were swollen and pale with fragmented, vacuolated cytoplasm and karyorrhectic, pyknotic, or absent nuclei (necrosis; Fig. 18). In this case, both vascular spaces and the mantle cavity were filled with granular, pale eosinophilic, necrotic cellular debris admixed with discernible necrotic cells and, within the vascular spaces, mildly increased numbers of hemocytes. The base of lamellae in California sea hare from groups 1 and 3 opened into gill ducts lined by 1 to several layers of poorly cohesive polygonal cells with abundant basophilic cytoplasm and poorly defined perinuclear clearing.
Podia provide principal gas exchange surfaces for echinoderms and are described in the Circulatory System section. Additionally, echinoderms have hollow outpocketings of the coelom through the body wall that aid in respiration; these are called gills and papulae in sea urchins and sea stars, respectively. 6,8,31 Gills and papulae both have an outer epidermis, thin middle connective tissue layer, and inner coelomic lining. Lesions related to the respiratory system in echinoderms in groups 1 and 2 were restricted to the epidermis associated with gills and papulae (see Body Wall). The gills and papulae in echinoderms in group 3 were unremarkable.
Gas exchange in cnidarians occurs across the general body wall, although specialized ciliated grooves called siphonoglymphs provide entry of water into the gastrovascular cavity and are another surface across which gas exchange occurs. 11,31 This area was not definitively discerned from other regions of gastrodermis in sunburst anemone from groups 1 and 3.
Nervous System
The California sea hare in group 3 had ganglia with very large neurons that were distributed throughout the body and connected by nerves; ganglia and nerves were both surrounded by a fibrous sheath (Fig. 19). In the California sea hare in group 1, axonal filaments were partially replaced by increased interstitial clear space (edema); axonal supporting cells were small with condensed nuclei and were less numerous than those seen in group 3 (Fig. 20). Ganglia were not observed in sections of the California sea hare in group 1.
The nervous system of echinoderms comprises a basiepithelial nerve plexus, which condenses to form the circumoral nerve ring, from which project radial nerves. Radial nerves form V-shaped ridges that extend down each arm or each ambulacral groove in sea stars and sea urchins, respectively. 31 The radial nerve comprises epidermal cell bodies resting on a layer of nerve cell bodies enmeshed in a thick layer of concentrated ectoneural axons (Fig. 21). 8 The radial nerve of echinoderms in groups 1 and 2 had abundant, fine neuropil vacuolation (Fig. 22) that was not observed in group 3. In sea stars in group 3, the parallel orientation of nerve fibers was evident histologically; in group 1, fibers lost normal orientation and were slightly curved and intersecting.
The nervous system of cnidarians is organized into a diffuse nerve net in the base of the epidermis and gastrodermis and was difficult to see by light microscopy in sunburst anemone in groups 1 and 3. 25,31
Reproductive System
Ovotestis of the California sea hare in group 1 was arranged in lobules with an outer rim of developing oocytes and central region of spermatogenesis. Ovotestis was not observed in the California sea hare in group 3.
Echinoderms are dioecious with ovaries or testes suspended in the body cavity. Gametogenesis was present at approximately equivalent levels in sea stars in groups 1 and 3; no gonadal lesions were appreciated in these groups. Female purple sea urchins in groups 1–3 had similar-appearing ova with equivalent levels of folliculogenesis. Male purple sea urchins in group 2 had abundant testicular lumina, approximately 20% of which contained spermatozoa. None of the purple sea urchins in group 1 or 3 were male.
Anemones do not have true “gonads” but rather gametogenic regions in the mesenteries (Fig. 11). 11 The anemone in group 1 was male and in group 3 was female; both anemones had unremarkable gametogenesis.
Discussion
Copper is an essential nutrient required for all organisms but at toxic levels can be harmful to aquatic organisms, including marine invertebrates. 42 Many mechanisms have been proposed for the pathogenesis of copper toxicosis in these species, including oxidative stress, DNA damage, impaired ion regulation, and metabolic derangements due to impaired oxygen uptake. 20 A study on copper toxicosis in marine invertebrates with differing osmoregulatory strategies revealed that osmoregulatory capacity influenced susceptibility to copper toxicosis. 20 The study suggested that the impacts of copper are related to physiology of the animal. Hence, it is not surprising that histologic lesions of copper toxicosis presented here varied widely among marine invertebrate species in the 3 phyla represented in this study (Mollusca, Echinodermata, and Cnidaria). While species from different phyla had different lesions, animals from different species within the same class (ie, bat star, brittle star, and knobby star in class Asteroidea) and different classes within the same phylum (ie, sea stars and sea urchins in phylum Echinodermata) showed almost identical histologic lesions.
The immune system of echinoderms is mediated by coelomocytes, which were increased in number in all echinoderms with copper toxicosis. Coelomocytes are sensitive, stress-activated effectors of the echinoderm immune response and have a wide range of functions. 34 Increased numbers of coelomocytes have been observed in stressed echinoderms and those from polluted environments; coelomocytosis in the cases presented here is considered a generalized and nonspecific immune response to copper toxicosis. 34 Echinoderms had frequent epidermal lesions, including necrosis and ulceration. Copper toxicosis has been shown to cause irregular epidermal contour and ulceration in marine invertebrates. 10,32 This effect is attributable to copper-induced membrane lipid peroxidation resulting in impairment of structure and function of epidermal cellular membranes. 10
Cnidarians are diploblastic, and the ectoderm (ie, epidermis) and endoderm (ie, gastrodermis) function as the primary immune and metabolizing organs. 1 Cyanide toxicosis in cnidarians, specifically anemone and corals, has been associated with epidermal disruption, loss of gastrodermal integrity, and mesogleal edema. 7 These findings are morphologically similar to lesions observed in the sunburst anemone presented here. Disruptions in cellular metabolism caused by copper, similar to those seen in cyanide toxicosis, may be the cause for cellular degeneration and necrosis in the epidermis and gastrodermis of the sunburst anemone. That being said, degeneration and necrosis are likely nonspecific lesions in an animal that has a limited array of responses to injury. Mesogleal edema likely follows epidermal ulceration due to loss of an osmotic barrier.
Copper exposure in anemone has been reported to cause expulsion of zooxanthellae, as seen in the sunburst anemone presented here. 17 Studies have demonstrated that zooxanthellae uptake excessive environmental copper, thereby buffering host anemone from the effects of copper accumulation. 17,26 Expulsion of zooxanthellae may be a regulatory response to increased copper storage. 17 This theory is supported by the fact that aposymbiotic, or azooxathellate, anemone accumulate copper at a faster rate as compared with symbiotic anemone. 17 Alternatively, zooxanthellae expulsion in the sunburst anemone presented here may be a nonspecific response due in part to gastrodermal necrosis/ulceration. Cyanide exposure in cnidaria reportedly causes several morphologic changes to zooxanthellae, including swelling, increased mitotic rate, abnormal expulsion into the gastrovascular cavity, and partial bleaching. 7 Definitively identified changes to zooxanthellae in the sunburst anemone presented here, however, were limited to expulsion into the gastrovascular cavity. Morphologic comparisons were not possible, because the control sunburst anemone in this study was azooxanthellate. A recent study revealed that a species of coral that is zooxanthellate at 20- to 30-m depth may be azooxanthellate at 60-m depth, suggesting that some cnidarian species may have facultative symbioses depending on their environment/depth. 15
In the California sea hare with copper toxicosis, the most severely affected organs were the gills and nephridia. Gills are reportedly the primary site of entrance for dissolved copper in gastropods. 4 Gills are a commonly injured organ in gastropods with copper toxicosis, and branchial epithelial necrosis is among the most commonly described lesion of this disease; the specificity of these lesions, however, has not been investigated in the referenced studies. 4,13,18,22,32 Injury to the branchial system presumably results in a reduction in oxygen consumption and osmoregulatory capacity. 18,32 Another lesion observed in the California sea hare presented here was axonal degeneration, attributed to a combination of elevated pressure due to osmoregulation dysfunction and direct necrotizing affect to neural cells, the latter of which may be due to cellular metabolic derangements induced by copper toxicosis. 5 Defects of osmoregulation may have contributed to interstitial edema within the heart and pharyngeal muscle in this case, although a contribution from an inflammatory response, evidenced by increased numbers of circulating hemocytes, was also considered possible. One of the most striking lesions in the California sea hare with copper toxicosis was necrosis within nephridia, the functions of which include filtration, excretion, and water balance. 23,31 As nephridia are among the most heavily perfused organ with high metabolic activity, it follows that the toxicant effect would be relatively high in this organ. Mechanisms of nephridial necrosis outside of direct toxicity are also considered possible.
The histologic findings presented here can be extrapolated to future mortality investigations of captive and free-ranging marine invertebrates. The specificity of the lesions associated with copper toxicosis in these animals is unknown. As similar lesions have been reported in marine invertebrates exposed to other pollutants/heavy metals, toxicosis may be considered a differential in cases where lesions similar to those presented here are identified along with history suggestive of toxicosis. 7,34 There is considerable variation in the normal anatomic structures within a single invertebrate class. Hence, the list of histologic components in Table 1 should be used as a guideline to be further tailored for various species. Additionally, the histologic review of marine invertebrates in these cases is not nearly inclusive of the complete histology for all 6 species represented.
Every year, millions of marine invertebrates are removed from the wild as part of an expanding, multibillion-dollar trade to supply food markets, curio trade, pharmaceutical and research industries, and aquaria. 28,30,36,39 A primary detriment to the trade’s sustainability is the high mortality rates that occur during holding and transport of collected animals. 36 Copper is a common pollutant in free-ranging populations, and the US Environmental Protection Agency has stressed the need for histopathologic investigations to evaluate toxicant effects on aquatic life. 38 As with vertebrate species, diagnostic reference materials likely will help to curb preventable mortality in wild-caught captive animals and understand disease emergence in free-ranging populations.
Footnotes
Acknowledgements
We thank Christy Buie and Liz Post of Northwest ZooPath; Kurt Takahashi, Barry Puget, Felicia DonRussello, and Christopher Kwong of the Veterinary Medical Teaching Hospital histology laboratory at the University of California-Davis; Roy Brown of Histology Consulting Service; and E. J. Hurley of California Animal Health and Food Safety Laboratory–San Bernadino histology laboratory for specimen preparation. Additionally, we thank Scott Fish of California Animal Health and Food Safety Laboratory–Davis for preliminary electron microscopy work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Phil and Karen Drayer Wildlife Health Center Fellowship Award from the Wildlife Health Center at the University of California–Davis.
