Abstract
Rickettsiella infection was diagnosed in 4 adult emperor scorpions (Pandinus imperator) from 2 different collections over a 3-year period. One case had a 2-day history of weakness, failure to lift the tail, or respond to stimulation, with rapid progression to death. The other 3 cases were found dead. There were no gross lesions, but histologically the hemolymphatic vasculature and sinuses, presumed hematopoietic organ, heart, midgut and midgut diverticula, nerves, and skeletal muscle were infiltrated with phagocytic and granular hemocytes with necrosis. Phagocytic hemocytes contained abundant intracellular microorganisms that were Fite’s acid-fast-positive, Macchiavello-positive, variably gram-positive or gram-negative, and Grocott’s methenamine silver-negative. By transmission electron microscopy, hemocytes contained numerous phagocytic vacuoles with small dense bacterial forms (mean 0.603 × 0.163 μm) interspersed with large bacterial forms (mean 1.265 × 0.505 μm) and few intermediary forms with electron-dense nucleoids and membrane-bound crystalline arrays (average 4.72 μm). Transmission electron microscopy findings were consistent with bacteria of the family Coxiellaceae. Based on sequencing the 16S ribosomal RNA gene, the identity was confirmed as Rickettsiella, and phylogenetic analysis of protein-coding genes gidA, rspA, and sucB genes suggested the emperor scorpion pathogen as a new species. This study identifies a novel Rickettsiella causing infection in emperor scorpions and characterizes the unique pathological findings of this disease. We suggest this organism be provisionally named Rickettsiella scorpionisepticum.
Scorpions are invertebrates of the phylum Arthropoda and class Arachnida, along with spiders, ticks, and mites. Scorpions exist on all continents, with the exception of Antarctica, and include approximately 13 families and 1750 different species inhabiting a wide variety of environments. 6,16 Scorpions are commonly kept as pets and maintained in zoological collections, with emperor scorpions (Pandinus imperator) considered one of the most popular captive species due to its large size and general docile temperament. 47
Published reports of diseases and the medical management of captive scorpions and other arachnids is growing, with recorded diseases including traumatic injuries of the limbs and exoskeletal tergum, endo- and ectoparasites, metabolic derangements, and infectious diseases. 19,21,39,59 Among these reports is an article identifying a Rickettsia-like infection in wild-caught common yellow scorpions (Buthus occitanus). 37 This bacterium was originally classified as Porochlamydia buthi belonging to either the genus Rickettsiella or Chlamydia. In this article, the author orally infected common yellow scorpions with tissue homogenates derived from dead common yellow scorpions, noting death in up to 100% of common yellow scorpions over an 8-month period. Based on DNA sequence evidence, this bacterium has since been reclassified as Rickettsiella chironami. 59
Rickettsiella spp. are well recognized as common pathogens and occasionally symbionts of a wide variety of arthropods. 2,12,31,45,55 Rickettsiella were previously considered to be a subspecies of the genus Rickettsia, but recent reclassification based on 16S ribosomal RNA (rRNA) gene sequences 45 has placed Rickettsiella in a separate genus in the family Coxiellaceae, order Legionellales, class Gammaproteobacteria. 22 There are currently 4 recognized species of Rickettsiella: R. grylli, R. chironami, R. popilliae, and R. stethorae with several pathotypes identified based on clinical presentation, although no type-strain isolates of these pathotypes have been successfully cultured. 20,24 Rickettsiella is an important and often fatal pathogen in many arthropods including beetles, 1,30 wireworms (Argiotes spp.), 49 cockroaches (Blatta orientalis), 13,40 and grasshoppers (Zonocerus variegatus), 25 but also affecting crustaceans 51 such as the red claw crayfish (Cherax quadricarinatus) 43 and isopod crustaceans (lice), 12 with reports in arachnids including poultry red mites (Dermanyssus gallinae), 14 spiders (Pisaura mirabilis), 38 and common yellow scorpions (Buthus occitanus) 37 as examples. Rickettsiella infection in most arthropods produces severe infection and death, such that investigations into using the bacterium as a biocide for control of pest arthropods are currently of interest. 14,48
This report details the clinical presentation and pathological findings in Rickettsiella infection in captive emperor scorpions.
Materials and Methods
Case Material
Captive adult male and female emperor scorpions were derived from a zoo in Denver, Colorado, United States (cases 1-3), and from a private collection in Saskatoon, Saskatchewan, Canada (case 4). Cases 1 to 3 were purchased as captive reared adults from commercial vendors (case 1: Glades Herp Farm, Inc, Bushnell, FL; cases 2 and 3: LLL Reptile and Supply Co, Oceanside, CA). Cases 1 to 3 were housed separately in acrylic containers with optimal temperature of 36 °C, humidity of 80%, and substrate composed of fresh coconut bark and dirt (Zoo Med Laboratories Inc, San Luis Obispo, CA). Cases 1 to 3 were held for 1 month in quarantine prior to display and were never physically handled or co-housed. They were fed a diet of live adult crickets (Gryllus sp.) dusted with a commercial multivitamin supplement (Reptivite with D3, Zoo Med Laboratories Inc). Crickets were purchased live from various vendors. The source, husbandry, and diet of case 4 was not known, and the case was presented after death as a privately owned pet. Histological sections from a healthy adult emperor scorpion and 2 healthy younger emperor scorpions, archived within the Wildlife Conservation Society, were used as a normal reference.
Histopathology
Dead emperor scorpions were fixed whole by incising the chitin along the ventrum and immersing the bodies in 10% neutral buffered formalin for 48 to 72 hours. Cases 2 and 3 were grossly dissected by removing the visceral mass in toto from the prosoma and mesosoma, and tissues were then serially sectioned in the transverse plane in situ into individual cassettes representing the prosoma and mesosoma. Metasoma and limbs were processed whole by immersion in a solution of 17% potassium hydroxide and 10% formalin (StatLab, McKinney, TX) until the chitin was pliable and easily sectioned with a scalpel. Metasoma and limbs were embedded and sectioned in the sagittal plane. Cases 1 and 4 were similarly formalin fixed whole and the entire body was softened in potassium hydroxide solution until the chitin was adequately softened for sectioning (approximately 3 hours). The entire emperor scorpion was serially sectioned in the transverse plane and placed in cassettes for in situ histopathology of the prosoma and mesosoma; the metasoma and limbs were processed as described above. This latter method was found to be preferable with the highest quality of histologic sections. Using standard methods, cassettes were processed, embedded in paraffin, and 5 µm thick sections were mounted on glass slides and stained with hematoxylin and eosin. The following structures were examined by light microscopy: exoskeletal tergum, skeletal muscle, hemolymphatic system (hemolymphatic vasculature, sinuses, presumed hematopoietic organ [note the hematopoietic organ previously described in scorpions and noted in this article may conflict somewhat with literature describing the hematopoietic source in other arachnids, particularly the spider]), 17,19 circulatory system (heart, arteries), central nervous system (brain, nerves), eyes, digestive system (pharynx, midgut, midgut diverticula), excretory system (coxal glands, Malpighian tubules), respiratory system (book lungs, tracheal tubes), and reproductive system (ovary, testis). Tissue sections with inflammation were stained with Fite’s acid-fast, 50 Grocott’s methenamine silver (GMS), 23 Brown and Brenn Gram, 36 and Macchiavello stains, 8 and examined by light microscopy.
Transmission Electron Microscopy
Tissues from cases 1 and 3 were best preserved and included the most representative lesions. Mesosoma with histologically confirmed lesions containing intracellular acid-fast-positive bacteria were selected for transmission electron microscopy (TEM), including nerve, midgut, midgut diverticula, skeletal muscle, and associated connective tissue with hemolymphatic sinuses. Tissues were stored in 10% neutral buffered formalin for approximately 3 weeks before processing, then sectioned into 1.0 mm3 tissue blocks and transferred to 3% glutaraldehyde solution with 0.166 M cacodylate buffer (Electron Microscopy Sciences) until processing. Similar 1.0 mm3 tissue blocks from the prosoma underwent a second fixation in 1% osmium tetroxide. Tissue fragments were dehydrated using a graded series of ethyl alcohol and embedded in Embed 812 resin (Electron Microscopy Sciences). Embedded samples were sectioned on a Leica UC6 Ultramicrotome (Leica Microsystems) to 1 µm thickness and stained with toluidine blue to visualize areas of interest. Selected sites were then processed into ultrathin sections (60–90 nm) and collected on 100-mesh copper grids (Electron Microscopy Sciences), and stained with 5% uranyl acetate for 20 minutes and Satos lead citrate for 6 minutes. The sections were examined under a JEOL transmission electron microscope (JEOL Ltd). The microorganism structures were measured and analyzed using iTEM software (Olympus SIS).
Molecular Identification
All polymerase chain reaction (PCR) primers used in this study are shown in Table 1.
Polymerase Chain Reaction Primers Used for Identifying a Novel Rickettsiella Species in Emperor Scorpions (Pandinus imperator).

Five 7-μm-thick scrolls of paraffin-embedded tissues from case 1, which included histologically identified intracellular microorganisms, were processed for PCR at the Washington Animal Disease Diagnostic Laboratory. DNA was extracted from the sample using a commercial kit (Qiamp DNA Minikit; Qiagen) according to the manufacturer’s instructions with variation as described. 60 Two Mycobacterium genus-specific PCR assays were performed as described previously. 27,44 PCR amplification of a portion of the 16S rRNA gene was performed using universal Rickettsia primers, 60 and the resulting product was sequenced and compared to the National Center for Biotechnology Information GenBank database.
Seven-micrometer-thick paraffin scrolls were prepared from paraffin-embedded lesions from all cases, and PCR was performed in the Hill Laboratory, Department of Veterinary Microbiology, University of Saskatchewan. Total DNA was extracted from the embedded tissues using the QiaAmp DNA FFPE tissue kit (Qiagen). The presence of amplifiable DNA was confirmed with PCR using primers targeting a 229 bp region of the cytochrome oxidase subunit 1 (cox1) gene in genera Pandinus (Table 1). PCR targeting the V1-V3 region of the 16S rRNA gene of bacteria was performed using “universal” bacterial primers F1 and R2, and the resulting product was sequenced using the amplification primers. For further identification and comparison to other species, 3 protein-coding genes previously demonstrated to provide robust resolution within entomopathogenic Rickettsiella were targeted for PCR and sequencing. Genes encoding rpsA (30S ribosomal protein A), gidA (glucose inhibited cell division protein A), and sucB (dihydrolipoamide succinyl-transferase component E2) were amplified using primers and conditions described previously. 2,34 PCR amplicons were purified and sequenced using the amplification primers.
For phylogenetic analysis of protein-coding genes, nucleotide sequences for all 3 genes were concatenated, and aligned with ClustalW. 54 Distance matrices were calculated using dnadist or protdist within the PHYLIP package 18 using the F84 and PAM scoring methods, respectively. Trees were produced by neighbor-joining with neighbor, consensus trees generated with consense, and branch lengths added in fitch. Trees were viewed and edited using FigTree (v1.4.2; http://tree.bio.ed.ac.uk).
Evaluation of Crickets
Four adult feeder crickets from the emperor scorpion facility of Denver, Colorado, were randomly selected live and immersed in 70% ethanol for euthanasia and preservation. 11 Crickets were bisected whole and processed in cassettes, embedded in paraffin and sectioned on a microtome as described for emperor scorpion tissues. Slides were stained with hematoxylin and eosin, Fite’s acid-fast, 50 Grocott’s methenamine silver (GMS), 23 Brown and Brenn Gram stains, 36 and examined by light microscopy. PCR and TEM were not performed.
Results
Clinical and Pathologic Findings
The 4 adult, captive-reared emperor scorpions were from 2 separate collections in the United States and Canada and had no previous clinical conditions or treatments. Case 1 had been in the facility for 5 months and presented with a 2-day history of weakness and inability to lift the tail, after which time it was found dead in the enclosure. Cases 2 to 4 were found dead in the enclosure with no prior signs. These cases had been in the facility for 1 year, 1.2 years, and an unknown number of months, respectively. None of the cases had gross lesions or an obvious cause of death.
Histopathologic findings were compared to archived tissues from normal emperor scorpions (Figs. 1, 5, 8). In all 4 cases, the hemolymphatic system (composed of the hemolymphatic vasculature, sinuses, and the presumed hematopoietic organ) was infiltrated with numerous large granular hemocytes that were individualized or formed coalescing aggregates (nodulation). Hemocytes contained abundant finely granular and basophilic stippled material consistent with phagocytized microorganisms (Figs. 2, 3), which were often strongly positive with Fite’s acid-fast stain suggestive of either Mycobacteria or Coxiella-like bacteria (Fig. 4). In the heart, hemocytic inflammation infiltrated the superficial epicardium in cases 1 and 4, and additionally effaced the myocardium with nodulation filling the lumen in cases 2 and 3. Within the nervous system, 3 of 4 scorpions (cases 1–3) had severe infiltration of nerves with hemocytic inflammation that underwent nodulation, with associated axonal swelling and degeneration (Fig. 2). The brain and globes were unaffected. In all cases, striated skeletal muscle was extensively infiltrated by individualized and nodular aggregates of hemocytes admixed with fewer brightly granular hemocytes (Fig. 6). Most hemocytes contained intracellular microorganisms. Multiple myocytes were infiltrated by hemocytes containing intracellular microorganisms. Additionally, myocytes contained similar microorganisms that did not appear to be within hemocytes and formed dense aggregates free within the sarcoplasm (Fig. 6). The microorganisms within hemocytes and free within the myocyte sarcoplasm (not within hemocytes) were strongly Macchiavello-positive, suggestive of Rickettsia-like bacteria (Fig. 7). In the respiratory system (which included the book lungs and tracheal tubes), cases 1 and 4 had hemocytic inflammation infiltrating the hemolymphatic vasculature of the book lung lamellae. Hemocytes contained Macchiavello-positive phagocytized bacteria (Figs. 9, 10). The lamellae of the book lung were not inflamed or degenerated and the tracheal tubes were unaffected. Within the digestive system (which included the pharynx, midgut, and midgut diverticula), all cases had severe effacement of the midgut diverticula with hemocytic inflammation and nodulation, confluent with inflammation throughout the surrounding hemolymphatic sinuses. There was moderate serosal and mural hemocytic inflammation of the midgut in case 1. The pharynx was unaffected. There were no lesions or microorganisms within the exoskeletal tergum, the excretory system (which included the coxal glands and Malpighian tubules) or the reproductive system (which included ovaries and testes).
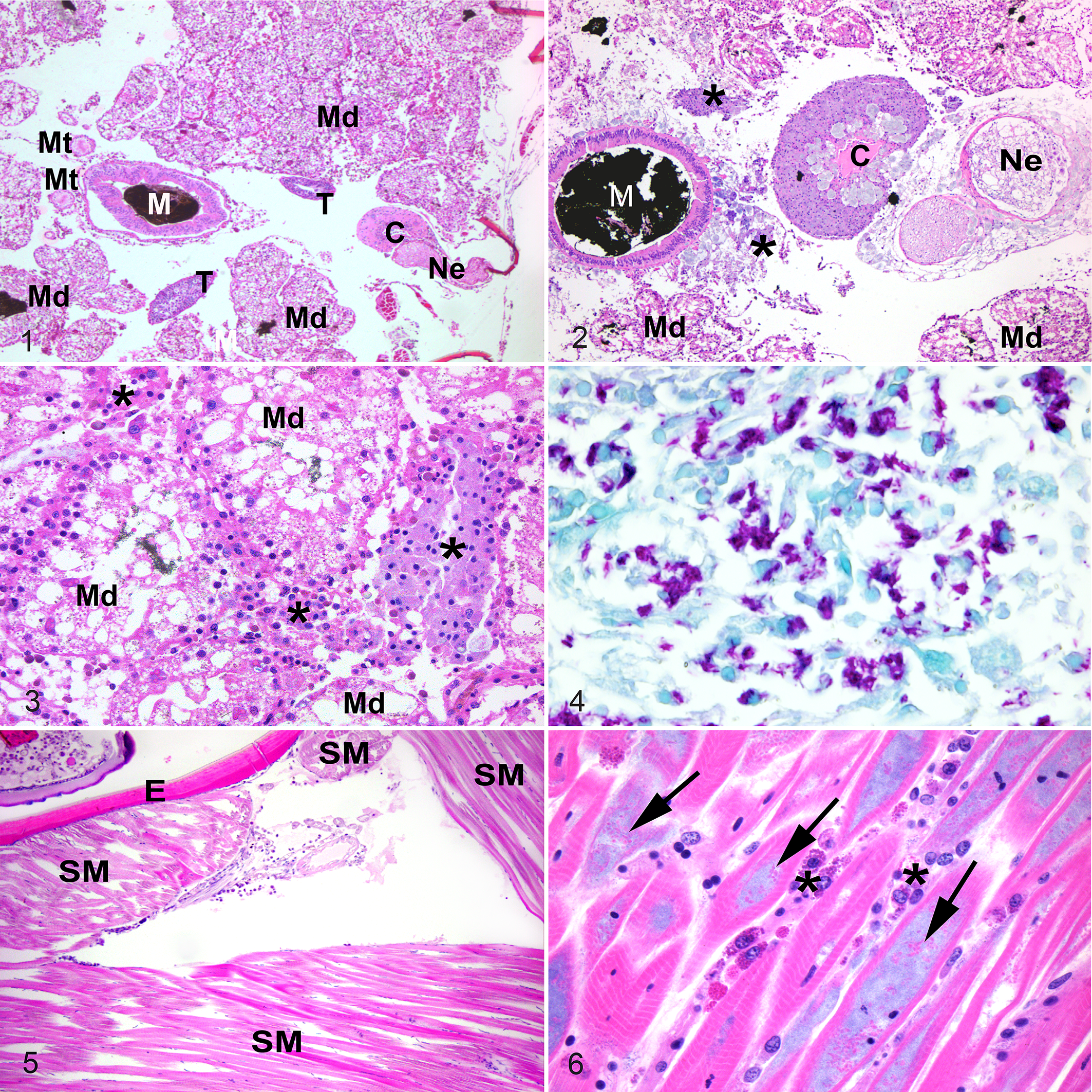
Emperor scorpions.
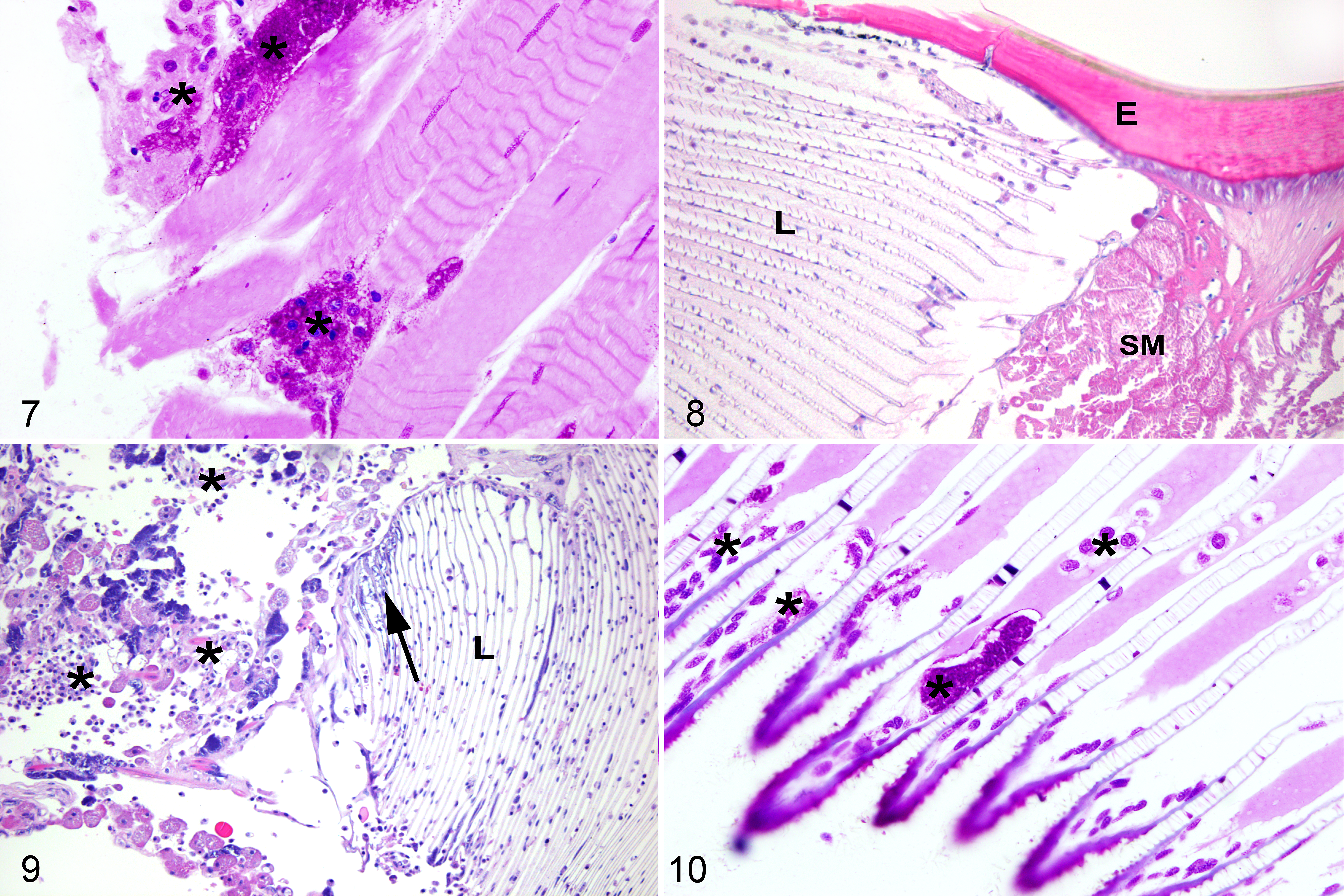
Emperor scorpions.
Tissues from each case with lesions consisting of hemocytic inflammation were stained with a panel of histochemical stains. The intracellular microorganisms were negative with Grocott’s methenamine silver (4/4 cases) and positive with Fite’s acid fast (3/4 cases). The Gram staining was variable: microorganisms were gram-positive in 2/4 cases, gram-negative in 1/4 cases, and unstained in 1/4 cases. Tissues from case 4 were additionally stained with Macchiavello, and the intracellular microorganisms were strongly Macchiavello-positive (Figs. 7, 10). A diagnosis of systemic hemocytic inflammation and nodulation with phagocytized Rickettsia-like or Coxiella-like bacteria was made.
Feeder crickets (Gryllidae sp.) from one institution were examined histologically and there was no detectable hemocytic inflammation or pathogenic or non-pathogenic intracellular microorganisms.
Transmission Electron Microscopy
Mesosomal tissues were examined ultrastructurally from cases 1 and 3, which had the best preserved and most characteristic lesions. The tissues included nerve, midgut, midgut diverticula, skeletal muscle, and associated connective tissue with hemolymphatic sinuses. Lesions in both cases were heavily infiltrated with hemocytes (Figs. 11–13) that were swollen with numerous intracellular phagocytic vacuoles of variable dimensions containing myriad bacteria (Figs. 13, 14). The bacteria had 2 distinct phenotypes: a small electron-dense bacterial form and a large bacterial form (Figs. 14–17), and these findings were most consistent with the family Coxiellaceae. The small dense bacterial form was most common with elongated bacilli measuring 0.603 µm (mean ± SD: 0.057 μm) by 0.163 ± 0.014 μm, with a fibrillary electron-dense nucleoid. The bacterial wall was composed of a 4-layered membrane that was 15.25 ± 2.51 nm thick (Figs. 15, 16). The large bacterial forms were far less numerous and were elongated 1.265 ± 1.149 µm by 0.505 ± 0.058 μm, and had a dispersed nucleoid surrounded by cytosol with increased electron density. The bacterial wall was composed of a 2-layered membrane, 24.66 ± 4.88 nm thick (Fig. 17, inset). Large bacterial forms were occasionally in an intermediary stage characterized by an electron-dense nucleoid (Figs. 14 and 20). The small dense bacterial form, intermediary and large bacterial forms were found in large phagocytic vacuoles (Figs. 14 and 21). Additionally, large vacuoles contained several membrane-bound crystalline arrays (Fig. 20) that varied from spherical to elliptical, with a diameter of 4.72 ± 0.845 μm, and an electron-dense core with a single-layer membrane. Occasionally, the large bacterial forms were extra-hemocytic and were free within the sarcoplasm of myocytes, and in this form they were surrounded by a thick envelope likely comprised of host cell intermediary filaments and microtubules (Fig. 17, inset). The large bacterial forms within phagocytic vacuoles were devoid of envelopes (Figs. 17–19).
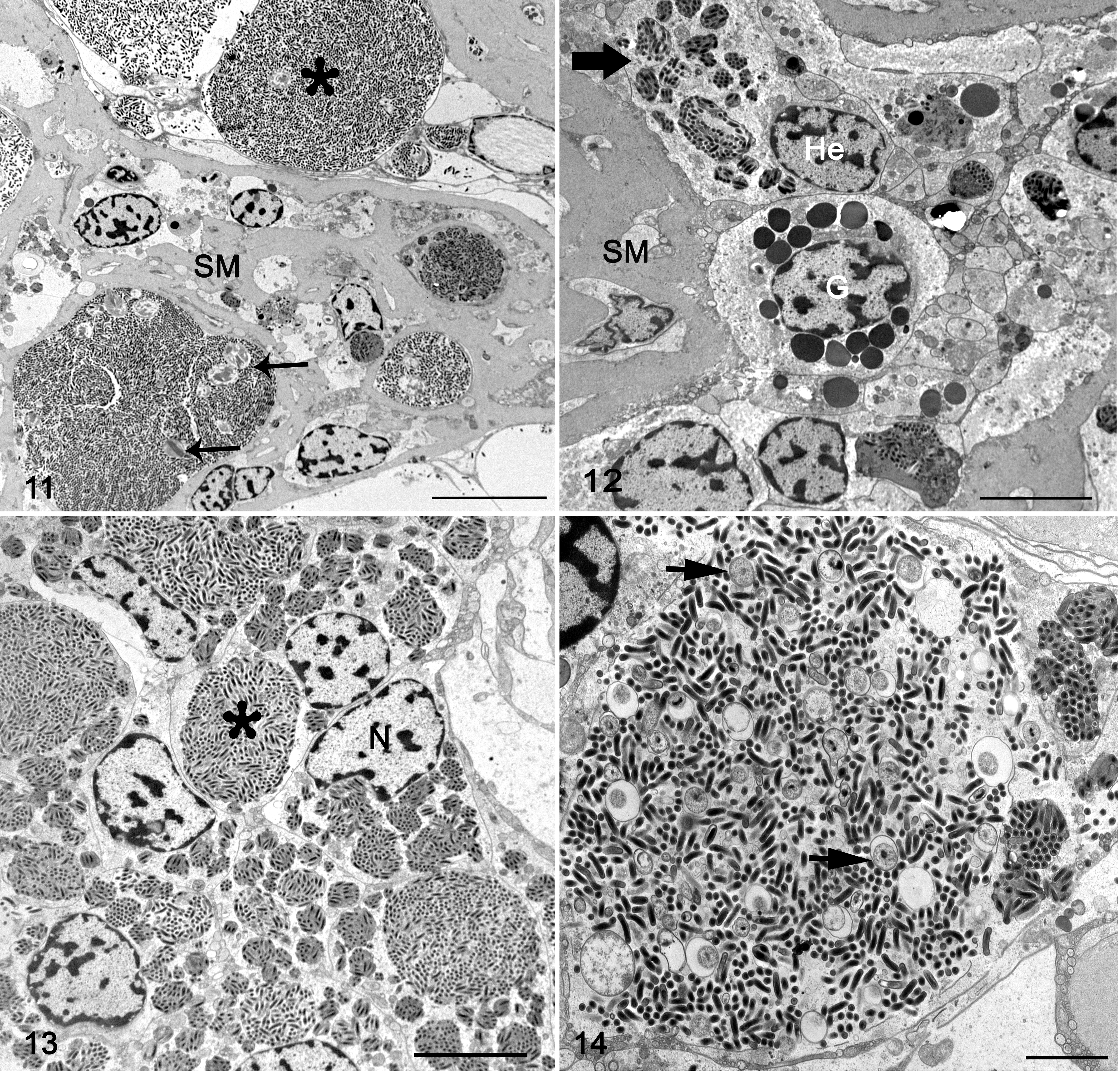
Rickettsiella scorpionisepticum infection, emperor scorpion, case 1. Transmission electron microscopy.
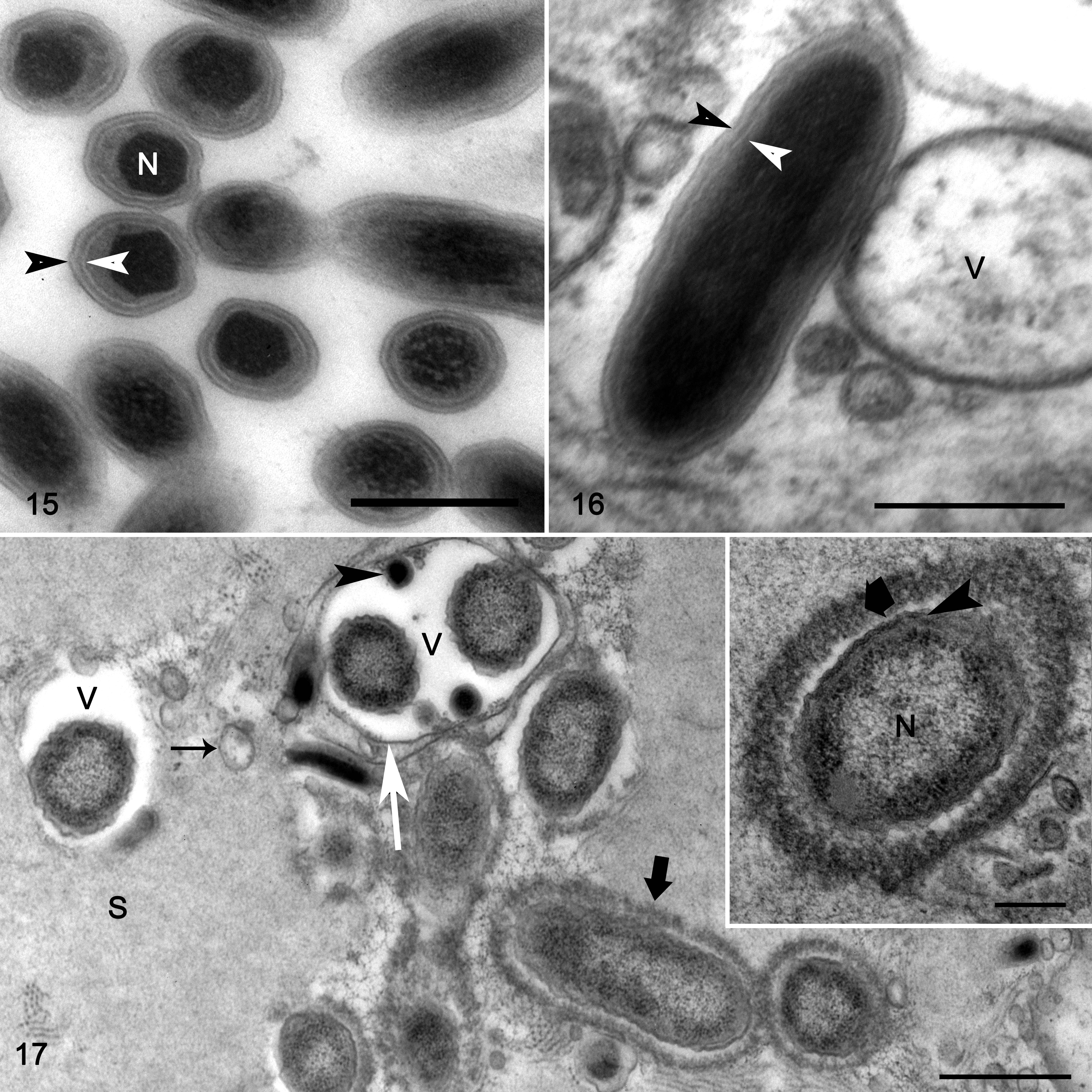
Rickettsiella scorpionisepticum infection, emperor scorpion, case 1. Transmission electron microscopy.
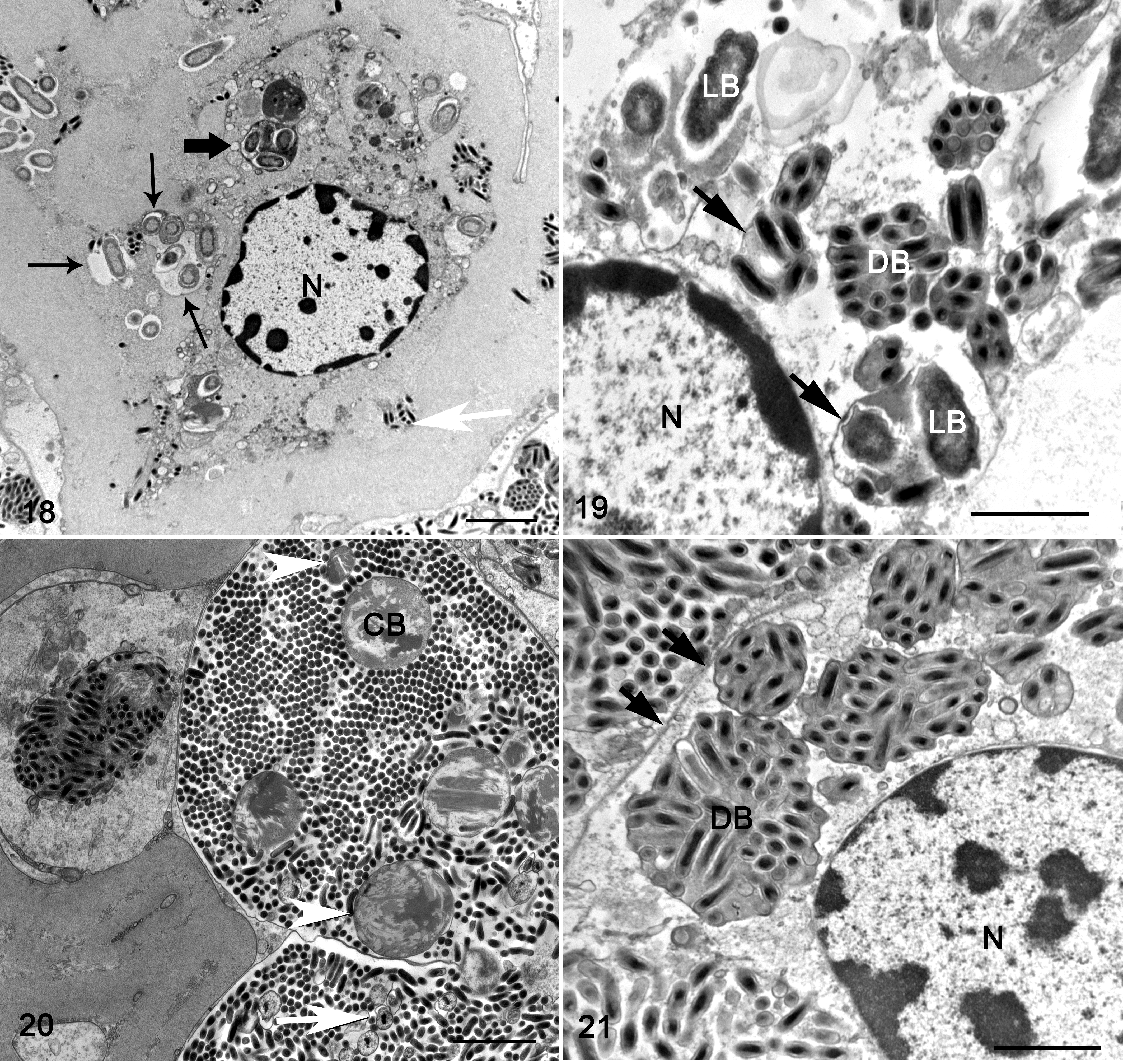
Rickettsiella scorpionisepticum infection, emperor scorpion, case 1. Transmission electron microscopy.
Molecular Identification
In case 1, Rickettsia-specific primers targeting the V3-V6 regions of 16S rRNA gene produced a product of the expected size and the sequence of the amplicon was 98% identical (704/719 bp) to a portion of the 16S rRNA gene from Rickettsiella agriotidis (GenBank Accession HQ640943) and to sequences from Rickettsiella derived from cockroaches and beetles (GenBank Accessions JX457300, JX457245, and KX363645). Independently, during the initial investigation of case 4, universal bacterial 16S rRNA PCR targeting the V1-V3 regions was performed. The sequence clusters had good bootstrap support within Rickettsiella but was no more than 97% identical to any of the other taxa over 504 bp of the V1-V3 region. The 16S rRNA gene sequence from case 4 was deposited in Genbank (Accession KU597420).
DNA extracts from paraffin-embedded tissues of all 4 cases were positive for cox1 PCR, indicating the recovery of amplifiable DNA. Specific PCR targeting 3 protein-coding genes (rpsA, gidA, and sucB) 2 of entomopathogenic Rickettsiella was then performed. DNA extracts from cases 1, 3, and 4 yielded PCR-amplicons for all 3 targets, and a weak amplification of gidA was detected from case 2. High-quality sequence data were generated for all 3 gene targets for cases 3 and 4, and for sucB of case 1. Sequences of rpsA and gidA were identical between cases 3 and 4, and sucB sequences were >99% identical (733/734 bp). The sucB sequences of cases 1 and 3 were also identical. Sequences from case 4 were deposited to Genbank (Accessions KU597421-KU597423). The relationships between the nucleotide and translated protein sequences of the emperor scorpion Rickettsiella and other related species are shown in Figure 22. The maximum nucleotide sequence identity between the emperor scorpion Rickettsiella and the published sequences of other related species were 91%, 82%, and 83% for rpsA, gidA, and sucB, respectively.
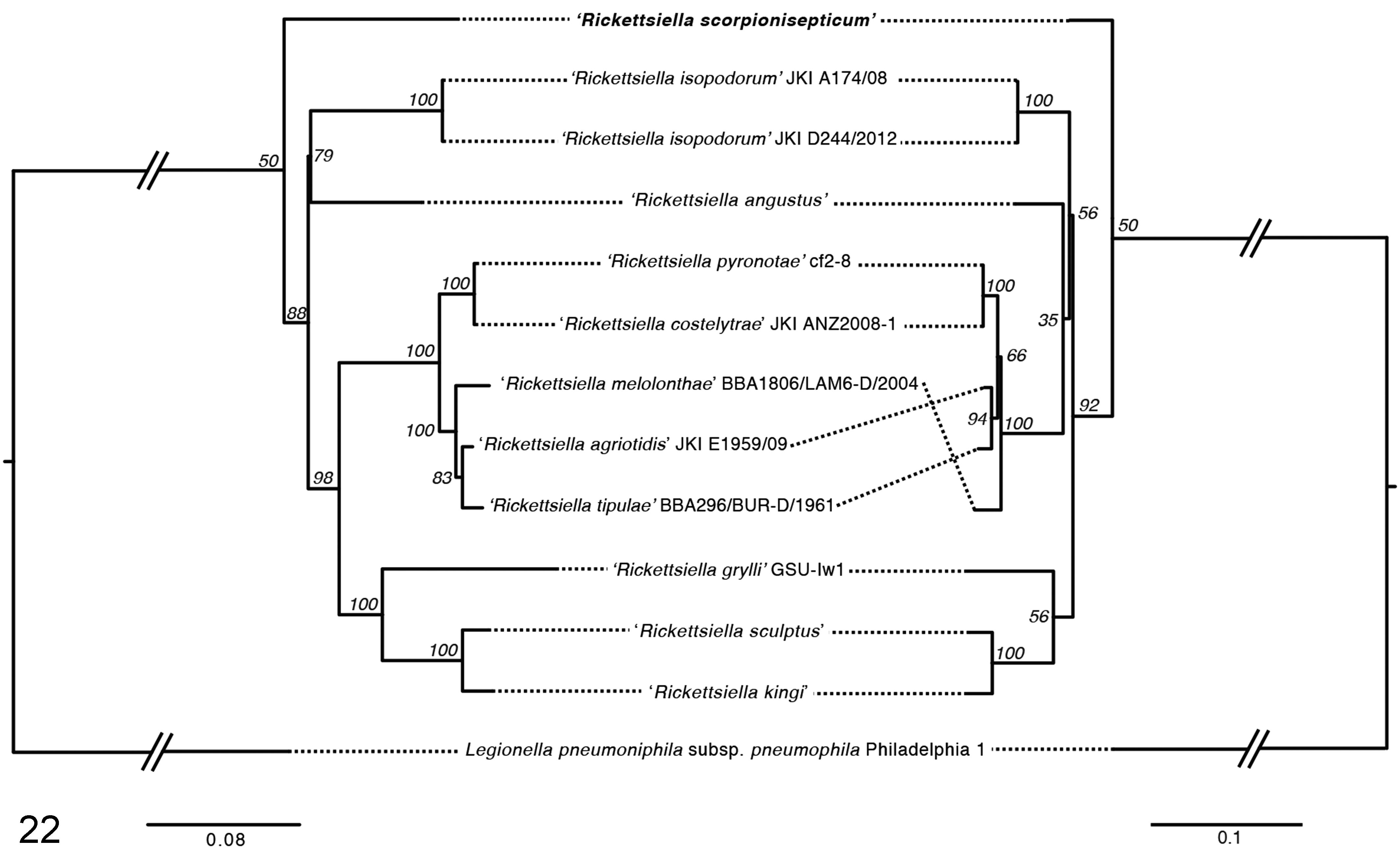
Phylogenetic relationships of Rickettsiella spp. based on concatenated nucleotide (left) and protein (right) sequences of gidA, rpsA, and sucB (total alignment length, 2512 bp). The trees are the neighbor-joined consensus of 100 bootstrap iterations of the alignments. Sequences included are from Rickettsiella derived from case 4 (KU597421), Rickettsiella melolonthae BBA1806/LAM6-D/2004 (EF694040), Rickettsiella angustus (HG792871), Rickettsiella kingi (HG792872), Rickettsiella sculptus (HG792873), Rickettsiella tipulae (JF288927), Rickettsiella agrioditis JKI E1959/09 (JN565687), Rickettsiella costelytrae JKI ANZ2008 (JN565689), Rickettsiella pyronotae cf2-8 (JN565691), Rickettsiella grylli GSU-Iw1 (JQ070345), Rickettsiella isopodorum JKI A174/08, and Rickettsiella isopodorum JKI D244/2012 (JX406182). Bootstrap values are indicated at the nodes. Legionella pneumoniphila subsp. pneumophila (NC_002942) is included as an outgroup. Scale bars indicate substitutions per site.
Because acid-fast-positive intracellular bacteria were detected, DNA extracted from paraffin-embedded lesions of case 1 were tested for mycobacterial infection using 2 different Mycobacteria genus-specific assays. Both PCR tests for Mycobacteria sp. were negative.
Discussion
This article describes a novel species of Rickettsiella associated with fatal infection in 4 adult emperor scorpions that presented with a brief history of generalized weakness or sudden death with no gross lesions. Histologically, scorpions had severe hemocytic inflammation involving the hemolymphatic, circulatory, nervous, digestive, and musculoskeletal systems. No lesions were detected in the brain, reproductive tract, or excretory system. Hemocytes within the lesions contained myriad intracellular bacteria with histochemical staining characteristics and ultrastructurally distinct phenotypic forms consistent with bacteria of the family Coxiellaceae as described in arthropods and other hosts. 9,29,30,40,43 In Vero cell cultures, the small dense bacterial forms have been shown to represent a stationary phase with no bacterial growth, the intermediary form is considered a transitional phase to active growth, and the large bacterial forms represent an exponentially replicative form, where increasing expression of specific proteins were used as markers for cell replication. 9,10 Bacterial forms described in these cases of emperor scorpion Rickettsiella are unique from many varieties of Rickettsiella sp. detailed ultrastructurally in other arthropods. Other Rickettsiella sp. exhibit distinct elementary body and initial body forms with common binary fission, 1,13,37,40,43 features which were not detected in the emperor scorpion Rickettsiella. Additionally, phagocytic vacuoles in these cases contained several large membrane-bound crystalline arrays, which are recognized in R. popilliae and R. popilliae pathotype pyronatae, 29,30 but distinct from single, intra-bacterial crystals that are commonly described in other species of arthropod Rickettsiella sp., 1,40,43 thus indicating the marked phenotypic diversity of this bacterium in arthropods in general. Notably, ultrastructural characteristics of Rickettsiella sp. in these affected emperor scorpions were distinct from pathogenic intracellular bacteria previously identified as Porochlamydia buthi in common yellow scorpions. 37 This bacterium has been since reclassified based on 16S rRNA gene sequence as Rickettsiella chironami. 59
DNA sequencing provided further evidence of the novelty of the Rickettsiella detected in these cases. An initial identification as Rickettsiella was based on partial sequencing of the 16S rRNA gene. Previous investigations, however, have demonstrated that phylogenetic relationships between Rickettsiella are better resolved using protein-coding gene sequences, with rpsA, gidA, and sucB identified as particularly informative. 2,32,34 Results of this analysis clearly illustrate the discrimination of the emperor scorpion Rickettsiella detailed in this article from other hosts, and also provide evidence of the utility of multi-locus sequence typing primers for the detection and characterization of these organisms. 32 One limitation of our study was that protein-coding gene sequence data were not obtained from case 2, perhaps due to poor-quality DNA template derived from the fixed and embedded tissue, and though partial sequence of the 16S rRNA gene was positive for Rickettsiella, this case cannot be conclusively connected to the other cases based on sequence data. Importantly, case 2 had histopathologic presentation of severe hemocytic inflammation, with similar histochemical staining qualities of the intracellular microorganisms, suggesting that this was the same condition.
Rickettsiella is a pathogenic bacteria of insects, crustaceans, and arachnids with a worldwide distribution. 1,2,4,14,29,30,43 Rickettsiella infection in arthropods is associated with weakness, emaciation, and death following what is thought to be a chronic infection developing slowly in both larvae and adults. 5,13,29,30,37,40,43,49,58 Rickettsiella is an obligate intracellular bacteria in the order Legionellales of the family Coxiellaceae class Gammaproteobacteria. 22,33 There are currently 4 recognized species of Rickettsiella: R. grylli, R. chironami, R. popilliae, and R. stethorae, with several pathotypes identified and characterized by the species of host infected and the infection produced. 20,24,32 In contrast, Rickettsiella has been shown to be a mutualistic endosymbiont in few insects and crustaceans, specifically the pea aphid, 55,56 leaf hopper, 26 and woodlouse, 30 and may have a symbiotic role in some arachnids, specifically the poultry red mite 14 and ixodid ticks. 2 Symbiotic Rickettsiella does not cause obvious disease in the host and is generally restricted to specific organs and cells where it has no pathological consequences, 2,14,26,29,55 though in mutualistic cases, it may additionally provide an advantage. In the pea aphid, Rickettsiella sp. is maintained in secondary bacteriocytes throughout the body, and in combination with other symbionts has been shown to alter the body color of aphids from red to green, providing an important advantage of camouflage and improving survivability of aphids. 55,56 Symbiotic Rickettsiella is also associated with the ovary and accessory glands of female insects, allowing for vertical transmission of bacteria to offspring. 26,55 Conversely, as a pathogenic bacteria, Rickettsiella is nonrestricted and infects most major vital organs distributing via hemocytes throughout the hemolymph in many arthropods. Rickettsiella is generally found throughout host cells in microcolonies sequestered within intracellular vacuoles. It is associated with cell destruction and inflammation, with eventual death of the host, as was recognized in these emperor scorpions. 1,13,30,37,40,49
Natural transmission of pathogenic Rickettsiella is suspected to be fecal-oral with release of Rickettsiella from the infected midgut and midgut diverticula and ultimately the feces of the host. 37,40,53 The route of infection in these emperor scorpion cases is suspected to be oral as significant infection of the midgut and midgut diverticula was noted. Rickettsiella-like infection was previously examined by feeding homogenized midgut diverticula from infected common yellow scorpions to healthy common yellow scorpions, and this resulted in disease in 100% of inoculated individuals. 37 In that study, infected common yellow scorpions generally died between 1.5 and 8 months post-exposure. High dosages of bacteria caused death between 40-60 days post-infection with no clinical signs, and lower doses caused death between 3 and 8 months post-infection, with approximately 45 days of clinical weakness and inappetence prior to death. 37 Rickettsiella is generally persistent within the environment, 53,58 and healthy common yellow scorpions succumbed to disease months after introduction to aquaria previously inhabited by infected common yellow scorpions. 37 Environmental contamination and chronic exposure, likely orally, may be an important source of Rickettsiella infection in the emperor scorpions in this study. As Rickettsiella can be both endosymbiotic and pathogenic in such a wide variety of arthropods, it is useful to consider that the cases in this study might have been infected by ingestion of commercially purchased adult crickets (Gryllidae sp.). Crickets have been documented to carry and succumb to pathogenic Rickettsiella grylli infection. 57 However, gross or histologic lesions were not detected in feeder crickets examined from one facility in the current study, although PCR for Rickettsiella was not performed.
The inflammatory response to Rickettsiella detailed in this article is typical of the response to intracellular bacterial infection in invertebrates, and specifically arachnids, which is considered to be an innate immune response triggered by pathogen-associated molecular patterns such as lipopolysaccharide, (1,3)-β-glucans, and a wide range of peptidoglycans. 3,7,28,35,44,52 The cell membrane of Rickettsiella is rich in peptidoglycans and lipopolysaccharides, thought to be an important trigger for inflammation while providing a means for obligate intracellular existence of this microorganism. 1,30,33,34,49 The inflammatory cascade in arachnids includes recruitment of hemocytes and activation of a compliment-like clotting cascade, intended to sequester hemolymph and prevent loss of hemolymph from internal or external injuries. 3,7 Hemocytopoiesis in arachnids is described as arising from the internal wall of the myocardium 19 while conflicting literature in scorpions describes a presumed hematopoietic organ arising from the supraneural artery. 17 Hemocytes in each system then enter the hemolymph and are circulated throughout vessels and hemal sinuses. 17,19 Hemocytes are classified into 3 basic categories consistent with light microscopic features of cytoplasmic granularity: hyaline hemocytes have small spherical nuclei and no cytoplasmic granularity with a robust phagocytic capacity; semi-granular hemocytes have moderate cytoplasmic granularity; and granular hemocytes have coarse eosinophilic granular cytoplasm. Cytoplasmic granules of semi-granular and granular hemocytes contain prophenoloxidase as well as defensins, glycine-rich peptides, small cyclic-peptides, and hemocyanin, all important components of a prophenoloxidative reaction that follows degranulation in activated hemocytes. 3,7,28,41,52 Hemocytes respond to innate stimuli by phagocytosis and degranulation, as was noted by numerous swollen hemocytes in Rickettsiella-infected emperor scorpions. Phagocytosis and degranulation are followed by nodulation, which serves to entrap and sequester phagocytized microorganisms and closely resembles granulomas in vertebrate species. 28,46,52 Chronic nodulation can progress to encapsulation, where aggregated hemocytes form a distinct capsule encircling the nodule, often associated with intense central degranulation and necrosis, similar to chronic caseating granulomas in vertebrate species. 28,42,46,51 Nodulation was common in infected scorpions with no encapsulation detected; however, nodulation did include chronic melanization, a process that follows prophenoloxidase activation as part of the inflammatory cascade. 28
The findings of this study suggest that these emperor scorpions were infected by a novel Rickettsiella that we propose be provisionally named Rickettsiella scorpionisepticum. In the current study, we show this infection was associated with morbidity and mortality in captive emperor scorpions.
Footnotes
Acknowledgements
The authors are grateful to Dr Neil Chilton, Department of Biology, University of Saskatchewan, for generous provision of Rickettsiella PCR primers. We thank the histotechnologists at the Diagnostic Medicine Center at Colorado State University and Prairie Diagnostic Services, Inc. The authors also thank Dean Muldoon at the University of Minnesota Veterinary Diagnostic Laboratory for the electron microscopy preparations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
