Abstract
Hamster polyomavirus (HaPyV) infection has been associated with lymphomas in Syrian hamsters. In the present study, 14 cases of lymphoma in pet Syrian hamsters were pathologically examined and the involvement of HaPyV was investigated. Among 14 cases, 11 were abdominal and 3 were cutaneous lymphomas. The average ages of hamsters with abdominal lymphoma and cutaneous lymphoma were 7 months (range: 4-12 months) and 14 months (range: 6-23 months), respectively. Histologically, abdominal lymphomas were characterized by the diffuse growth of tumor cells with intermediate or large nuclei, low mitotic rates, the presence of tingible body macrophages, and the T-cell immunophenotype. Furthermore, 4/11 abdominal lymphomas were immunopositive for T-cell intracellular antigen-1, suggesting cytotoxic T-cell lymphomas. Cutaneous lymphomas were diagnosed as nonepitheliotropic T-cell lymphoma. Polymerase chain reaction (PCR) detected HaPyV DNA in 12/14 samples, and a sequence analysis of PCR amplicons confirmed >99% nucleotide identity to the published HaPyV sequences. In situ hybridization (ISH) for HaPyV DNA resulted in diffuse nuclear signals within tumor cells in 10/14 cases. Consistent with previous findings, all HaPyV-associated lymphomas were observed in the abdominal cavity of young hamsters. Polymerase chain reaction and ISH were useful for identifying the involvement of HaPyV in lymphomas, and ISH results indicated the presence of episomal HaPyV in neoplastic lymphocytes. The present study suggests that HaPyV infection is highly involved in abdominal lymphomas in young pet Syrian hamsters in Japan and provides diagnostic information on HaPyV-associated lymphoma.
Tumors of the hematopoietic system are the second most common tumors (17%), after integumental tumors (62%), in pet hamsters. 23 Hematopoietic tumors commonly occur in Syrian (golden) hamsters (Mesocricetus auratus) (87%) and rarely in other hamsters (13%), and most of them are lymphomas. 23 One type of lymphoma in Syrian hamsters is an epizootic disease caused by Hamster polyomavirus (M. auratus polyomavirus 1, HaPyV) infection.15,26 HaPyV is a non-enveloped circular double-stranded DNA virus that is transmitted horizontally via urine or by biting and grooming behaviors. Cases of HaPyV infection have been reported in the United States and Europe.5,11,20,27 There are 3 main outcomes of HaPyV infection: subclinical effects, trichoepithelioma, and lymphoma.4,15,26 HaPyV infection in older hamsters is typically subclinical and the virus persists in the renal tubular epithelium, resulting in continued viruria. In enzootically infected Syrian hamsters, HaPyV infects hair follicle keratinocytes and induces multicentric trichoepithelioma containing abundant HaPyV particles in the stratum corneum.4,11,15 In naive young hamsters, HaPyV infection causes lymphoma.3,4,15 Mortality rates among hamsters with lymphoma may reach 80% within 4 to 30 weeks of infection. 4 Once enzootic in laboratories, the virus cannot be effectively eliminated without euthanizing the entire population and thoroughly decontaminating the premises. In 2013, a pandemic of HaPyV infection occurred in a colony of GASH:Sal Syrian hamsters in the University of Salamanca, Spain. 20 Among 975 animals, 150 developed lymphoma over a 5-year period and exhibited rapid disease progression with mortality within 1 to 2 weeks.
Although the life cycle and pathogenicity of HaPyV have been extensively examined in experimental infections, few clinical studies have investigated HaPyV-associated lymphoma in Syrian hamsters outside of the laboratory, and most are individual case reports.5,27 Previous epidemiological studies on spontaneous tumors in pet hamsters did not examine the involvement of HaPyV in lymphoma cases.16,23 Therefore, the aims of the present study were to characterize the pathological findings of lymphomas in pet Syrian hamsters and investigate the prevalence of HaPyV in these cases.
Materials and Methods
Samples
Biopsy samples from 14 Syrian hamsters that were histopathologically diagnosed as lymphoma were examined. Samples were from the archives of the Laboratory of Veterinary Pathology, the University of Tokyo, between 2009 and 2022. Histological slides were reviewed by 2 veterinary pathologists approved by the Japanese College of Veterinary Pathologists (J.K.C. and K.U.).
Histopathology and Immunohistochemistry (IHC)
Specimens were routinely fixed in 10% phosphate-buffered formalin, embedded in paraffin, sectioned at a thickness of 4 μm, and stained with hematoxylin and eosin. According to the classification criteria of canine malignant lymphomas, 29 the following features of tumors were examined sequentially for each case: growth pattern (nodular or diffuse), nuclear size (small, intermediate, or large), cytoplasmic volume (scant, moderate, or abundant), mitotic rate (grades 1, 2, or 3), the presence of necrosis, and the infiltration of tingible body macrophages, neutrophils, and eosinophils. Nuclear sizes were classified as small (<1.5× the size of a red blood cell), intermediate (1.5-2× the size of a red blood cell), or large (>2× the size of a red blood cell). Cytoplasmic volumes were classified based on the maximum width between the plasma membrane and nuclear membrane as follows: scant (<0.5× the diameter of a red blood cell), moderate (0.5-1× the diameter of a red blood cell), and abundant (>1× the diameter of a red blood cell). The mitotic rate was measured as the average number of mitotic figures per 10 consecutive high-power fields (HPF; diameter of the field of view = 0.55 mm; HPF = 0.237 mm2; 400× magnification; Olympus CX31 microscope) starting from the most mitotically active areas. 18 Only fields with representative tumor cells were assessed. After counting mitoses in the first HPF, if no mitoses were found in the next 3 consecutive HPF, the process was repeated by moving to another mitotically active area. Lymphomas with 0 to 5 mitoses/HPF were classified as grade 1, those with 6-10 mitoses/HPF as grade 2, and those with more than 10 mitoses/HPF as grade 3. Regarding cutaneous lymphomas, the histological location of tumor tissues and the epitheliotropism of tumor cells were also assessed.
The immunophenotypic characterization of tumor cells was performed using immunohistochemistry (IHC). A primary antibody for CD3 (ready to use; rabbit polyclonal antibody; Dako) was used as a T-cell marker. Primary antibodies for CD10 (ready to use; mouse monoclonal antibody; clone 56C6; BioGenex) and CD20cy (1:500; mouse monoclonal antibody; clone L26; Dako) were used as B-cell markers. The cross-reactivity of these antibodies with Syrian hamsters was confirmed in a previous study. 20 Furthermore, a primary antibody for T-cell intracellular antigen-1 (TIA-1; 1:50; mouse monoclonal antibody; clone 2G9A10F5; Beckman Coulter), which was not validated for Syrian hamster, was used as a marker of cytotoxic granules. Deparaffinized tissue sections were immersed in 10% hydrogen peroxide (H2O2) in methanol at room temperature for 5 minutes. Antigen retrieval was performed by an autoclave pretreatment at 121°C for 10 minutes in 10 mM citrate buffer, pH 6.0 for CD3, CD20cy, and CD10, or in Target Retrieval Solution, pH 9.0 (Dako) for TIA-1. Sections were incubated in 8% skim milk in Tris-buffered saline at 37°C for 40 minutes to avoid nonspecific reactions. Each tissue section was then treated with a primary antibody at 4°C overnight. An anti-mouse IgG or anti-rabbit IgG polymer labeled with horseradish peroxidase (Envision, Agilent Technology) was applied at 37°C for 40 minutes and sections were then rinsed with Tris-buffered saline. Reactions were visualized with 0.05% 3-3’-diaminobenzidine (Dojindo) containing 0.03% H2O2 in Tris-hydrochloric acid buffer, followed by a counterstain with Mayer’s hematoxylin (Muto Pure Chemicals). A normal lymph node tissue collected from a Syrian hamster was applied both as a positive and negative control for the IHC.
Polymerase Chain Reaction (PCR) and Sequence Analysis
Genomic DNA was extracted from all formalin-fixed paraffin-embedded (FFPE) tissue samples using the QIAamp DNA FFPE Tissue Kit (QIAGEN). HaPyV DNA was amplified by PCR targeting the VP1 gene (nucleotides 3,277-3,737) using the following primers: HaPyV-F (5’-GCCCAACCCCATTT TCATCC-3’) and HaPyV-R (5’-ACCCTTCAAATGTGGGA GGC-3’). 27 PCR was performed using KOD FX Neo DNA polymerase (TOYOBO) by preparing the following reaction mixture: 12.5 µL 2×PCR Buffer for KOD FX Neo, 5 µL dNTPs (2 mM), 0.75 µL forward primer (10 µM), 0.75 µL reverse primer (10 µM), and 0.5 µL KOD FX Neo (1 U/µL). Template DNA was added at between 30 and 40 ng per PCR reaction followed by distilled water to a total volume of 25 µL. Distilled water instead of template DNA was applied for negative controls. The PCR cycle was performed using a 3-step cycling condition according to the manufacturer’s instructions as follows: pre-denaturation at 94°C for 2 minutes followed by 45 cycles of denaturation at 98°C for 10 seconds, annealing at 58°C for 30 seconds, and extension at 68°C for 30 seconds. Polymerase chain reaction–amplified products were electrophoresed on 2% agarose gels and stained with ethidium bromide solution (Invitrogen). Bands were visualized by UV exposure using the ChemiDoc XRS+ System (Bio-Rad Laboratories). The PCR-amplified products of cases 1, 2, 3, 4, 9, and 10 were purified using the High Pure PCR product Purification Kit (Roche) and subjected to a sequence analysis (FASMAC). The identity of the products was established by sequencing the 364-bp PCR amplicon. Sequences were compared with reference HaPyV sequences (GenBank accession number: JX416853.1, JX036360.1, AF073287.1, and MT925638.1) using the Unipro UGENE software and Basic Local Alignment Search Tool from the National Center for Biotechnology Information (https://blast.ncbi.nlm.nih.gov/Blast.cgi).1,8,21
In Situ Hybridization (ISH)
Using the extracted DNA of case 10 as a template, a digoxigenin (DIG)-labeled DNA probe for the HaPyV VP1 gene was obtained by PCR amplification using the PCR DIG Probe Synthesis Kit (Roche Diagnostics) according to the manufacturer’s instructions. After amplification, the product was confirmed by gel electrophoresis as described above. Chromogenic ISH was performed for all cases. Deparaffinized tissue sections were immersed in 10% H2O2 in methanol at room temperature for 5 minutes, followed by enzyme digestion with the Carezyme III: Pronase Kit (Biocare Medical LLC) at room temperature for 7 minutes. After an autoclave pretreatment at 105°C in 10 mM citrate buffer, pH 6.0 for 15 minutes, DNA was denatured by heating at 95°C for 5 minutes. Sections were hybridized with 1 ng/μL of a DIG-labeled DNA probe at 40°C for 16 hours. After washing, sections were incubated in 1.5% Blocking Reagent (Roche Diagnostics) in Tris-buffered saline at room temperature for 30 minutes, and then incubated with a mouse anti-DIG antibody (1:500, Sigma-Aldrich) at 37°C for 1 hour. An anti-mouse IgG polymer labeled with horseradish peroxidase (Envision, Agilent Technology) was applied at 37°C for 40 minutes and sections were then rinsed with Tris-buffered saline. Reactions were visualized with 0.05% 3-3’-diaminobenzidine (Dojindo) containing 0.03% H2O2 in Tris-hydrochloric acid buffer, followed by a counterstain with Mayer’s hematoxylin (Muto Pure Chemicals). Non-neoplastic cells within the tissue sections and HaPyV PCR-negative lymphoma tissues were used as negative controls.
Results
Clinical Features
Clinical information on the 14 hamsters examined is summarized in Table 1. The average age of hamsters was 8.6 months (range: 5-23 months). There were 6 females and 7 males; the sex of 1 hamster (case 11) was unknown. Lymphomas were located in the abdominal cavity in 11 cases and skin in 3 cases. The average ages of hamsters with abdominal lymphoma and cutaneous lymphoma were 7 months (range: 4-12 months) and 14 months (range: 6-23 months), respectively. Hamsters with abdominal lymphoma showed reduced activity in 4 cases, abdominal distention in 4 cases, anorexia in 2 cases, and constipation in 2 cases. Macroscopically, abdominal lymphoma presented as a whitish, solid mass occupying the abdominal cavity (Fig. 1a). In 3 cases, tumors invaded the intestinal wall (case 6), intestinal wall and pancreas (case 1), or the abdominal wall (case 11). Cutaneous lymphomas presented as solitary masses in 2 cases (cases 12 and 14) or generalized alopecia with erythema and crusts in 1 case (case 13).
Clinical information, histological features, and results of immunohistochemistry, PCR, and in situ hybridization on 14 cases of lymphoma in pet Syrian hamsters.
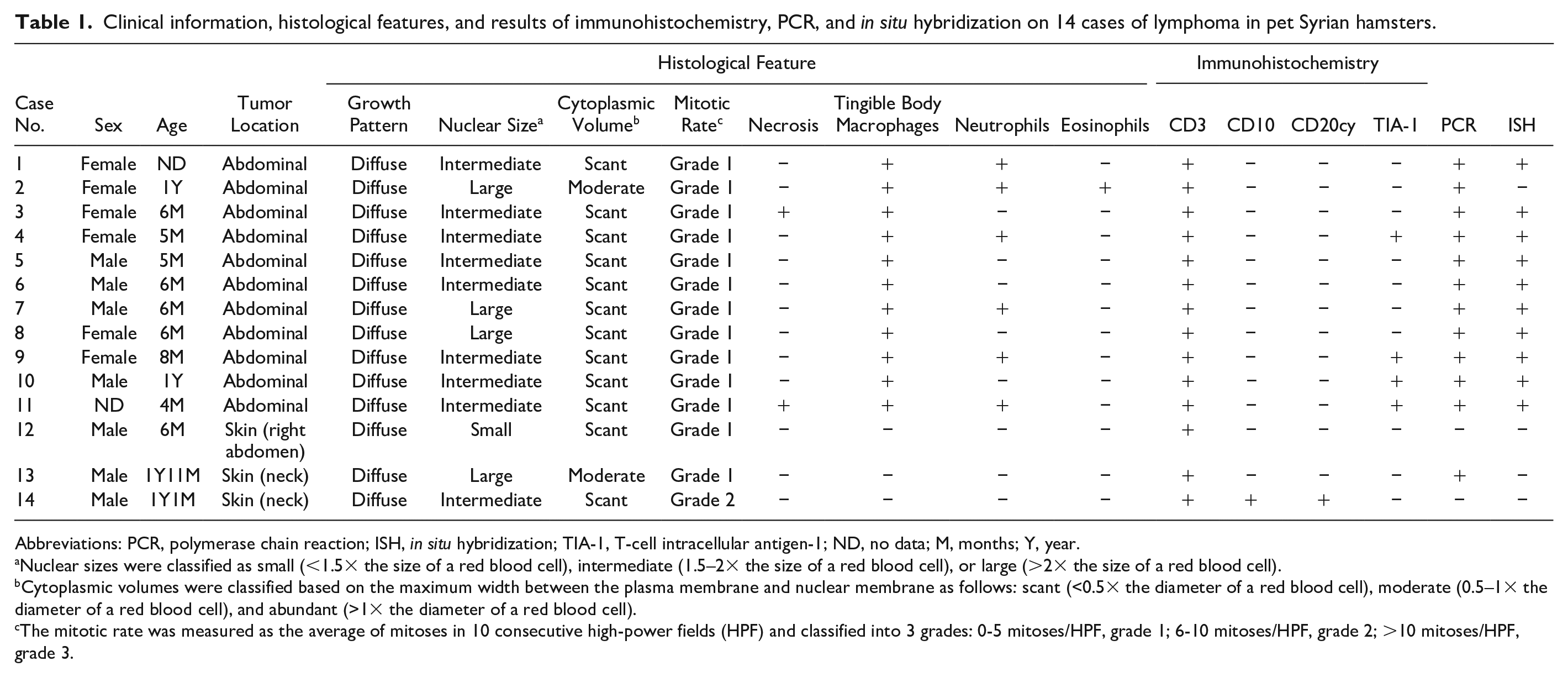
Abbreviations: PCR, polymerase chain reaction; ISH, in situ hybridization; TIA-1, T-cell intracellular antigen-1; ND, no data; M, months; Y, year.
Nuclear sizes were classified as small (<1.5× the size of a red blood cell), intermediate (1.5–2× the size of a red blood cell), or large (>2× the size of a red blood cell).
Cytoplasmic volumes were classified based on the maximum width between the plasma membrane and nuclear membrane as follows: scant (<0.5× the diameter of a red blood cell), moderate (0.5–1× the diameter of a red blood cell), and abundant (>1× the diameter of a red blood cell).
The mitotic rate was measured as the average of mitoses in 10 consecutive high-power fields (HPF) and classified into 3 grades: 0-5 mitoses/HPF, grade 1; 6-10 mitoses/HPF, grade 2; >10 mitoses/HPF, grade 3.
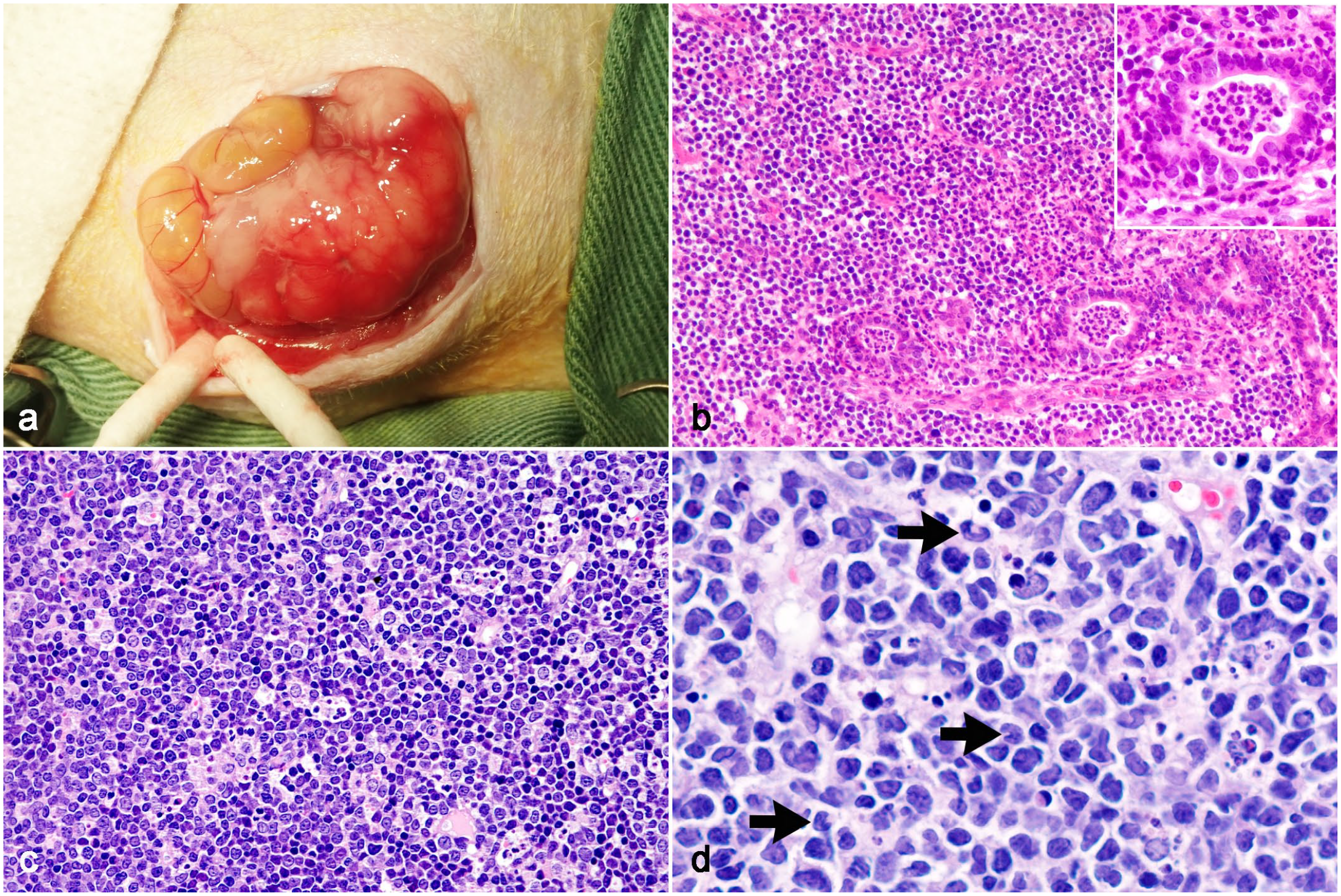
Lymphoma, abdominal cavity, Syrian hamster. (a) Gross image during surgery. A large whitish mass adjacent to the intestine. Case 10. (b) Invasion of tumor cells into the pancreas. Neutrophil infiltration is observed in the remaining ducts of exocrine glands (inset, higher magnification). Case 1. Hematoxylin and eosin (HE). (c) Sheets of tumor cells accompanied by tingible body macrophages. Case 10: HE. (d) Tumor cells with large (>2× the size of a red blood cell) oval or cleaved nuclei (arrows) and scant amphophilic cytoplasm. Case 8: HE.
Histopathology and IHC
The histological features of lymphoma in 14 cases are summarized in Table 1. All lymphomas were characterized by the diffuse growth of neoplastic lymphocytes. Abdominal lymphomas (cases 1-11) were unencapsulated and composed of sheets of tumor cells with scant fibrovascular stroma (Fig. 1b–d). Tumor cells often infiltrated peritoneal adipose tissue. In case 1, the structures of the intestine and pancreas were lost due to tumor invasion, and few mucosal glands and ducts of the exocrine glands remained in the tumor tissue (Fig. 1b). Cutaneous lymphomas (cases 12-14) were located in the dermis and obscured the adnexa (Fig. 2a). Tumor cells infiltrated the subcutaneous tissue in case 12 and skeletal muscle in case 14. Epitheliotropism was not observed in any of the cases. The nuclei of tumor cells were large in 4 cases (3 abdominal and 1 cutaneous), intermediate in 9 cases (8 abdominal and 1 cutaneous), and small in 1 case (cutaneous). Multinucleated giant tumor cells resembling Reed-Sternberg cells were often observed in 1 cutaneous lymphoma (case 14, Fig. 2b). Mitotic rates were low (grade 1) in all cases, except for 1 cutaneous lymphoma (case 14, grade 2). Intratumoral necrosis was present in 2 abdominal lymphomas. In all abdominal lymphomas, tingible body macrophages containing phagocytized apoptotic bodies were diffusely present in the background, showing a “starry sky” appearance (Fig. 1c). Neutrophil infiltration was observed in 6 cases, and eosinophil infiltration was observed in 1 case.
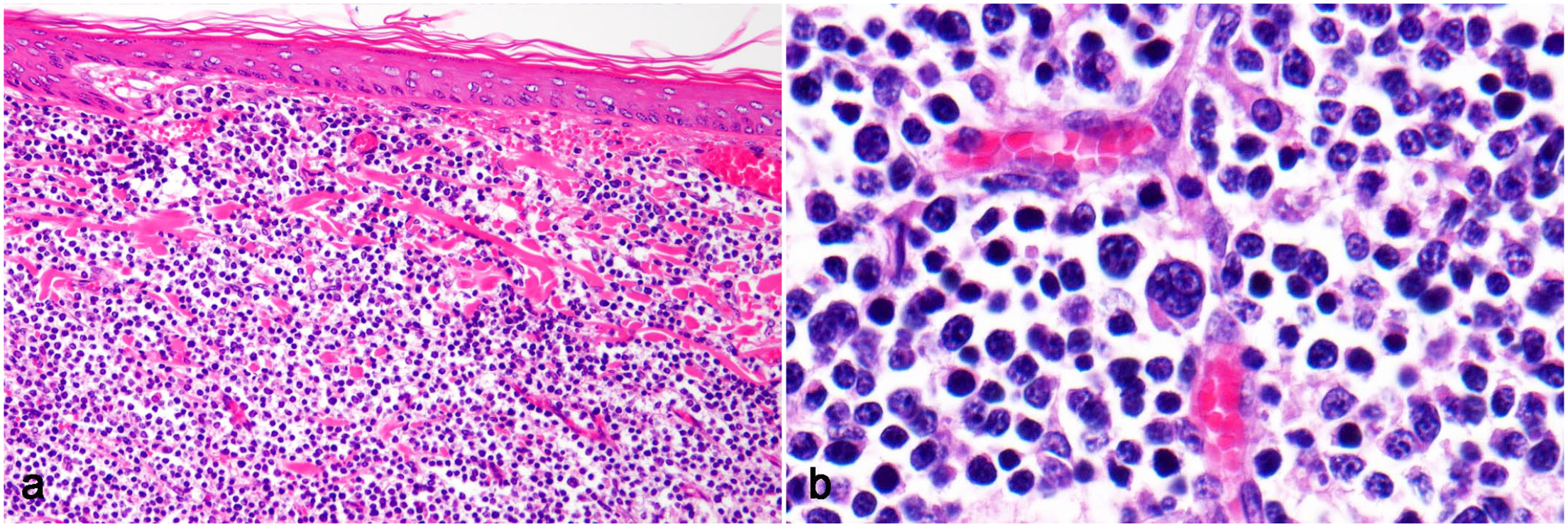
Lymphoma, skin, Syrian hamster. (a) Tumor cells diffusely infiltrate the dermis without epitheliotropism. Adnexal structures are replaced by tumor cells. Case 12. Hematoxylin and eosin (HE). (b) Tumor cells with round, intermediate-sized nuclei (1.5-2× the size of a red blood cell) and a scant eosinophilic cytoplasm. Multinucleated giant tumor cells are observed. Case 14: HE.
IHC results are summarized in Table 1. Tumor cells were positive for CD3 and negative for CD10 and CD20cy in all abdominal lymphomas (Fig. 3a, b). The CD10- and CD20cy-positive lymphocytes were scattered throughout tumor tissues, and their nuclei were dense and smaller than those of neoplastic lymphocytes (Fig. 3b). Cutaneous lymphoma tumor cells were positive for CD3 in 3/3 cases and negative for CD10 and CD20cy in 2/3 cases. In 1 case of cutaneous lymphoma (case 14), a small number of tumor cells were positive for CD10 and CD20cy as well as CD3 (Fig. 3c, d). T-cell intracellular antigen-1 immunolabeling in tumor cells was occasionally observed in 4/11 abdominal lymphomas and characterized by a granular cytoplasmic pattern (Fig. 3e).
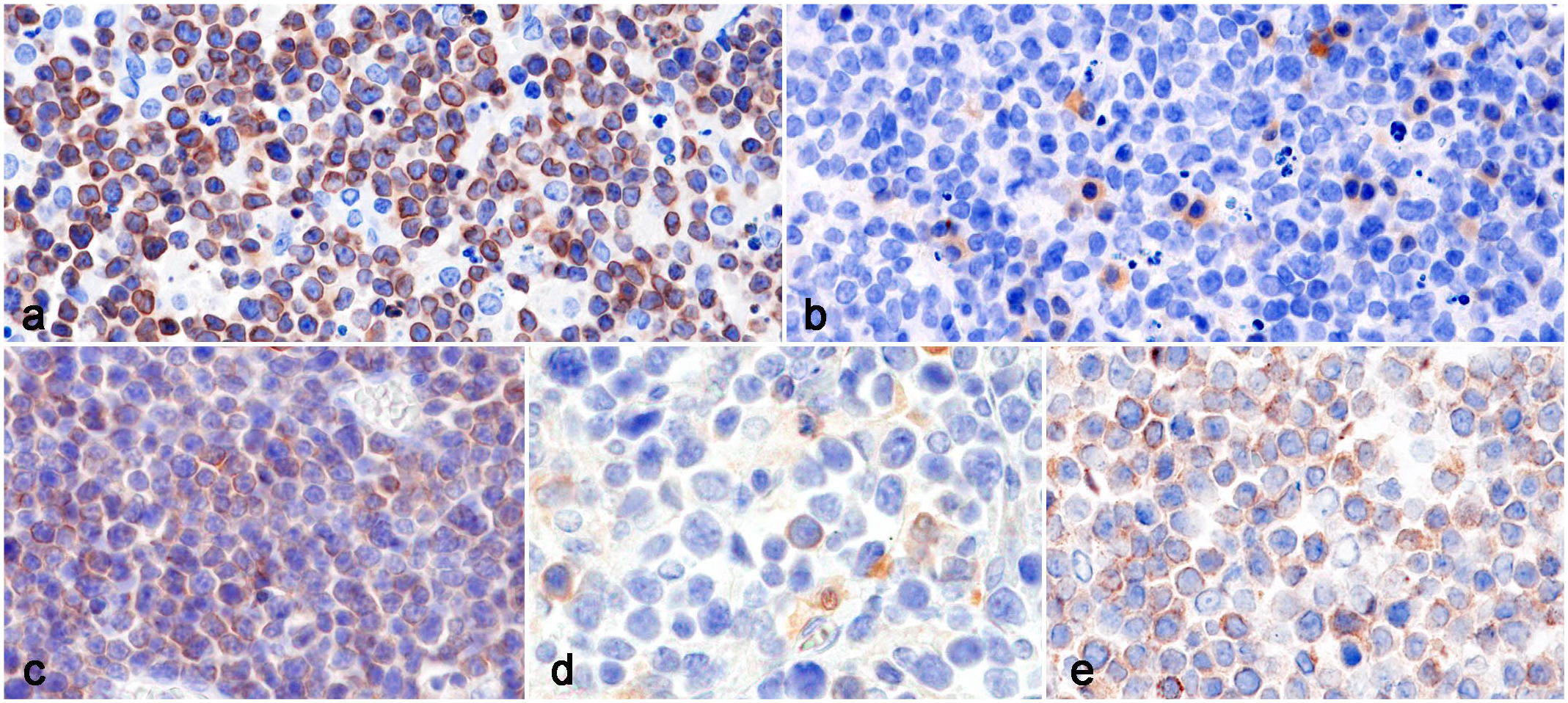
Lymphoma, Syrian hamster. Immunohistochemistry. (a) Abdominal lymphoma. Tumor cells are immunolabeled for CD3. Case 5. (b) Abdominal lymphoma. Tumor cells are negative for CD20cy, and mature lymphocytes with small densely stained nuclei are immunolabeled for CD20cy. Case 7. (c) Cutaneous lymphoma. Tumor cells are immunolabeled for CD3. Case 14. (d) Cutaneous lymphoma. A small number of tumor cells are immunolabeled for CD20cy. Case 14. (e) Abdominal lymphoma. Tumor cells are immunolabeled for T-cell intracellular antigen-1 in a granular cytoplasmic pattern. Case 11.
PCR and Sequence Analyses
HaPyV DNA was detected in 12/14 samples by PCR (Table 1, Fig. 4). All abdominal lymphomas were PCR-positive, while only 1 cutaneous lymphoma (case 13) was PCR-positive. Of the 5 cases that were sequenced, the nucleotide sequence from 4 cases had 100% identity to each other while the sequence from the remaining case had a single nucleotide substitution (c. 3663C>T). In comparisons with 4 reference sequences of the HaPyV VP1 region, the 4 samples with the same sequence showed 100% identity to the reference sequence identified in Spain (JX416853.1), 96.43% identity (351/364) to those identified in United States (AF073287.1) and United Kingdom (MT925638.1), and 96.15% identity (350/364) to that identified in Germany (JX036360.1). The single nucleotide substitution detected in 1 sample was not found in any of the reference sequences.
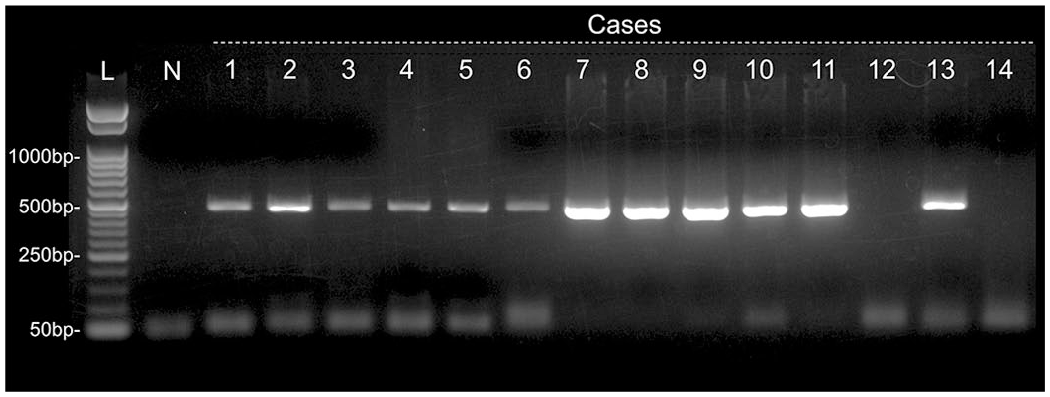
Gel electrophoresis of PCR amplicons for Hamster polyomavirus VP1. PCR-positive bands in cases 1-11 and 13, but not in case 12 or 14. Abbreviations: bp, base pairs; L, 50-bp ladder; N, negative control.
ISH
The cellular localization of HaPyV DNA was examined by chromogenic ISH and the results obtained are summarized in Table 1. Positive hybridization signals were detected in 10/14 cases (cases 1 and 3-11). All cases with positive hybridization signals were also PCR-positive for HaPyV: 10/11 abdominal lymphoma cases were ISH-positive, while all 3 of the cutaneous lymphoma cases were ISH-negative. Hybridization signals were characterized by diffuse nuclear signals within neoplastic lymphocytes (Fig. 5a). No hybridization signals were observed in 2 PCR-positive and 2 PCR-negative cases (Fig. 5b), as well as non-tumor tissues.
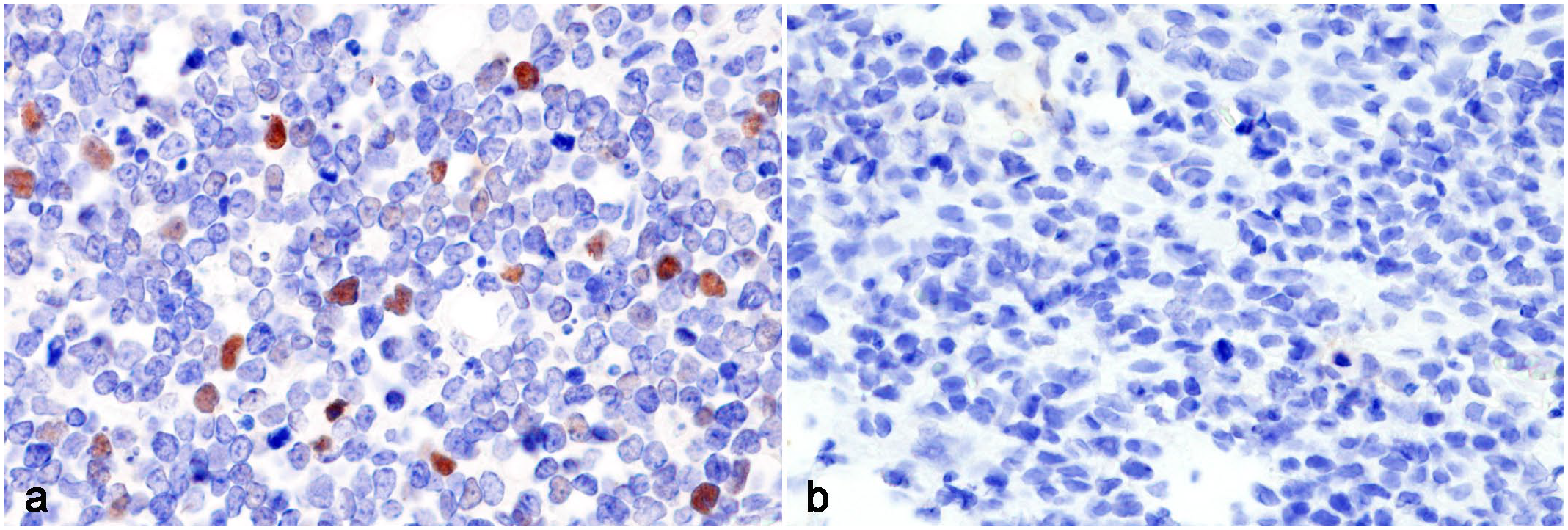
Lymphoma, Syrian hamster. In situ hybridization for Hamster polyomavirus VP1 DNA. (a) Abdominal lymphoma. Strong diffuse hybridization signals within the nuclei of tumor cells. Case 5. (b) Cutaneous lymphoma. No hybridization signals are observed. Case 13.
Discussion
In the present study, 10/14 cases (cases 1 and 3-11) were confirmed to be HaPyV-associated lymphoma by PCR and ISH. This is the first study on HaPyV infection in pet Syrian hamsters in Asia. HaPyV-associated lymphomas examined in the present study had common clinical and pathological features. All tumors occurred in the abdominal cavity of young Syrian hamsters (1 year old or less) and were histologically characterized by the diffuse growth of neoplastic lymphocytes with intermediate or large nuclei, low mitotic rates, the presence of tingible body macrophages, and the T-cell immunophenotype. These results are consistent with previous findings and are characteristic features that suggest HaPyV involvement.4,15,26 HaPyV generally causes lymphoma in the mesenteric lymph nodes and gut-associated lymphoid tissues without the involvement of the spleen, forming a palpable abdominal mass.4,15,20 In this study, however, HaPyV-associated lymphomas contained only a small amount of normal tissue, such as peritoneal adipose tissue, making it difficult to identify where the tumors originated. Since HaPyV-associated lymphoma is a lethal enzootic disease, 4 veterinary clinicians and pathologists need to be aware of the above-mentioned findings and consider the possibility of a HaPyV pandemic in the rearing environment of animals, especially in breeding facilities.
HaPyV may induce T-cell and B-cell lymphomas.4,5,15 A previous study on the HaPyV pandemic in a colony of GASH: Sal Syrian hamsters reported a high incidence of non-Hodgkin, Burkitt-type B-cell lymphoma in affected hamsters. 20 HaPyV-associated lymphomas in the abdominal cavity have been suggested to possess the B-cell immunophenotype, while those in the thymus possess the T-cell immunophenotype. 4 However, all HaPyV-associated lymphomas examined in the present study occurred in the abdominal cavity and had the T-cell immunophenotype. In addition to the primary tumor locations, other factors, such as viral strains and genetic differences among hamsters, may be related to tumor immunophenotypes. Furthermore, 4/11 (36%) HaPyV-associated lymphomas were immunohistochemically positive for TIA-1. T-cell intracellular antigen-1 is generally expressed in lymphocytes with cytolytic potential, including natural killer (NK) cells and cytotoxic T-cells.2,28 NK cells and cytotoxic T-cells are characterized by the presence of cytoplasmic granules containing cytotoxic molecules, such as TIA-1, granzyme B, and perforin. T-cell intracellular antigen-1 is a cytotoxic granule-associated RNA-binding protein that characteristically shows granular cytoplasmic immunolabeling in IHC. The present results suggest that HaPyV infection is associated with the development of cytotoxic T-cell lymphomas. Among polyomaviruses, HaPyV is unique to developing hematopoietic tumors. 24 The HaPyV genome encodes 3 tumor antigens (large-, middle-, and small-T), whereas other members of the family, except for murine polyomavirus, encode only large- and small-T. 10 The binding of HaPyV middle-T to Fyn, which is a proto-oncogene tyrosine-protein kinase associated with T-cell receptor signaling, 22 has been proposed to play a role in the development of lymphoma.7,26 Although trichoepithelioma is also caused by HaPyV and has only been described in association with HaPyV infection, 15 the concurrent formation of trichoepithelioma and lymphoma in individual hamsters is a rare event.5,15 In general, HaPyV induces trichoepithelioma in older hamsters and lymphoma in young hamsters. Consistent with previous reports, all hamsters that developed HaPyV-associated lymphoma in the present study were young and had no neoplastic skin lesions.
Two PCR-positive cases (cases 2 and 13) were ISH-negative in the present study. Case 2 had similar clinical and histological features to other PCR- and ISH-positive cases, suggesting a false-negative result for ISH. One possible explanation for the loss of hybridization signals is the degradation of DNA in FFPE tissue. 19 One cutaneous lymphoma (case 13) was positive for HaPyV by PCR. However, the viral gene was not detected in the tumor cells by ISH, and the clinical and pathological features, such as age, tumor site, and presence of tingible body macrophages, were different from those in other cases of HaPyV-associated lymphoma. Therefore, the development of lymphoma was assumed to be unassociated with HaPyV in this case. In general, the occurrence of HaPyV-associated lymphoma is uncommon in the skin of older hamsters.15,26 HaPyV infection in older hamsters results in (1) subclinical effects with viral persistence in the renal tubular epithelium or (2) induction of trichoepitheliomas.15,26 In case 13, a low copy number of viral DNA, which was undetectable by ISH, may have been present in hair follicle keratinocyte and amplified by PCR. Polymerase chain reaction was useful for detecting HaPyV DNA from FFPE samples in the present study; however, the potential risk of false-positive PCR results in diagnostic testing for HaPyV-associated lymphoma needs to be considered. Our results show that both PCR and ISH modalities are effective to prove causal relationship between HaPyV infection and lymphoma.
ISH is useful for examining not only the localization of viral DNA, but also the status of the virus (episomal or integrated) in infected cells. Diffuse nuclear hybridization signals represent episomal forms, while punctate nuclear hybridization signals represent integrated forms.6,12 In the present study, ISH revealed diffuse nuclear signals in HaPyV-associated lymphomas, suggesting the presence of episomal HaPyV in tumor cells. Tumorigenesis associated with polyomaviruses is commonly triggered by the integration of their viral DNA into the host genome.9,30 Conversely, the integration of HaPyV DNA has only been found in 9% of HaPyV-associated lymphomas, while the remaining lymphomas contained HaPyV DNA as extrachromosomal episomal copies.17,25 However, in a previous study, virus particles were not present in the tumor cells of HaPyV-associated lymphomas. 15 The formation of lymphomas is a side effect of HaPyV infection and not critical to the virus life cycle. 4
Cutaneous lymphoma generally presents in adult hamsters and causes clinical signs, including anorexia, weight loss, alopecia, crusts, and exfoliative erythroderma. 14 A previous study reported epitheliotropic cutaneous T-cell lymphomas resembling mycosis fungoides in 6 Syrian hamsters. 13 In the present study, 3 cases were considered to be spontaneous cutaneous lymphomas that were not associated with HaPyV. Although all 3 cases were diagnosed as non-epitheliotropic T-cell lymphoma, and the histological features, such as the nuclear morphology, mitotic rates, and the immunophenotypes of tumor cells, varied among the cases. Two out of the 3 cases presented with a localized mass, whereas 1 hamster (case 13) developed widespread skin lesions without mass lesions. Therefore, there appears to be several types of cutaneous T-cell lymphoma in Syrian hamsters.
The present study revealed that HaPyV is often involved in the development of lymphomas in pet Syrian hamsters. HaPyV-associated lymphomas had common features, which may have diagnostic value for distinguishing between viral and non-viral lymphoma. In particular, our results indicated that abdominal, but not cutaneous, lymphomas are frequently associated with HaPyV infection. However, to discuss the overall prevalence of HaPyV infection and its association with neoplastic diseases in pet Syrian hamsters, investigation on a larger sample set including other types of tumors is needed.
Conclusions
In the present study, 10/14 (71%) lymphomas in pet Syrian hamsters developed in the abdominal cavity of young individuals in association with HaPyV infection. They were histologically characterized by the diffuse growth of tumor cells with intermediate or large nuclei, low mitotic rates, the presence of tingible body macrophages, and a T-cell immunophenotype. Polymerase chain reaction and ISH were useful for identifying the involvement of HaPyV in lymphomas. The present results suggest the high prevalence of HaPyV in the lymphomas of pet Syrian hamsters and provide useful information for the diagnosis of HaPyV-associated lymphoma.
Footnotes
Acknowledgements
The authors thank Professor Haga and Ms. Dung of the Division of Infection Control and Disease Prevention, Graduate School of Agricultural and Life Sciences, the University of Tokyo, for the precious discussion about virus detection and for the excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
