Abstract
Lymphoma is a common neoplasm in cats, in which alimentary lymphoma is a common subtype, and it is usually diagnosed in elderly, feline leukemia virus (FeLV)-negative cats. This study aimed to describe the pathological features of lymphoma with involvement of the alimentary tract in FeLV-antigen-positive cats. In a 12-year retrospective study, 32 necropsied and FeLV-infected cats with lymphoma affecting the alimentary tract were identified. Twenty-one cases were multicentric lymphomas with secondary involvement of the alimentary tract, and the remaining 11 cats were considered to have primary alimentary lymphoma. The small intestine was the most common anatomic location (23/32; 72%), followed by the large intestine (19/32; 59%) and stomach (18/32; 56%). In 22/32 cases (69%), multiple organs within the alimentary tract were concomitantly affected. Thickening of the intestinal and gastric walls was the most common gross lesion (23/32; 72%), while mural nodules were observed in 16/32 cats (50%). The mesenteric lymph nodes were frequently affected (22/32; 69%). Most lymphomas were composed of large (17/32; 53%) and intermediate cells (14/32; 44%). B-cell lymphomas were more frequent (24/32; 75%), and diffuse large B-cell lymphoma was the most common diagnosis (15/32; 47%). In 31/32 (97%) cases, FeLV gp70 antigen was detected in neoplastic lymphocytes by immunohistochemistry. Lymphomas affecting the alimentary tract may be observed in FeLV-infected, young adult cats, in which large to intermediate cell and B-cell lymphomas are more frequently observed, and small cell T-cell intestinal lymphoma is unlikely to be diagnosed.
Keywords
Lymphoma is the most commonly diagnosed malignant neoplasm in cats, and feline leukemia virus (FeLV) is a known causative agent. FeLV-infected cats are 3.9 times more likely to develop lymphoma when compared with non-infected cats. 8 In many countries, successful control programs and vaccination have been effective in reducing the number of FeLV infections and FeLV-associated neoplasms.2,20,22 Still, lymphoma is one of the main causes of death in cats progressively infected with FeLV, accounting for 24%–58% of the deaths.8,36 In Brazil, FeLV is highly prevalent and association between lymphoma and FeLV infection is still described;3,7,8,19,33 approximately 55% of cats with lymphoma are progressively infected with FeLV.8,19
Lymphomas may be classified according to anatomic location, histologic distribution within organs, cell size, immunophenotype, and when known, biologic behavior and molecular features. 39 Regarding the anatomic distribution, alimentary lymphoma comprises those neoplasms affecting the alimentary tract with or without involvement of regional lymph nodes, and it may also occasionally affect the pancreas, liver, and spleen.1,13,25 In cats, it is the most frequent gastrointestinal tumor18,30 and the most common form of lymphoma, accounting for approximately half of the lymphoma cases in this species.6,20,22,32,41 In addition, it is more common in older and FeLV-negative cats.1,13,35,38 In Brazil, alimentary lymphoma is the most common type of lymphoma;19,33 however, mediastinal lymphoma is still common, being the second most frequent diagnosis.7,19 This may be explained by the high prevalence of FeLV infection in Brazil, as this retrovirus is frequently detected in cases of mediastinal lymphoma, whereas in alimentary lymphoma, only up to 30% of affected cats are FeLV-positive.11,17,19
Studies of lymphomas affecting the alimentary tract usually include aged and FeLV-negative cats. In most of these cases, lymphomas affect the small intestine and are histologically classified as small cell lymphoma of T-cell origin. This tumor has an indolent behavior and distinction from lymphoplasmacytic enteritis is challenging. Thus, many of these studies focus on making this distinction, as well as determining the etiology and possible association with human intestinal lymphoma.9,10,23,26,31,42 On the contrary, studies describing lymphoma in FeLV-infected cats include all types of lymphoma, and the types more frequently associated with FeLV infection, such as mediastinal and multicentric, are overrepresented.7,11,19,33,35 In general, FeLV-associated lymphomas are aggressive tumors and have a distinct biologic behavior from that of small cell, non-FeLV-associated intestinal lymphoma.38,40 No investigation to date has focused only on lymphomas affecting the alimentary tract in FeLV-infected cats, and few studies classify the lymphomas of the alimentary tract according to the Revised European and American Lymphoma/World Health Organization (REAL/WHO) classification system.23,28,41,42,45 Thus, this study aimed to evaluate the gross, histologic, and immunohistochemical features of lymphomas affecting the alimentary tract in FeLV-infected cats, and to determine whether these differ in type and pattern from lymphomas previously described in FeLV-infected cats and from those observed in FeLV-negative cats.
Materials and Methods
Case Selection
A retrospective study of the necropsy database (book records and digital search) from the Veterinary Pathology Laboratory was conducted to search for cases of lymphoma with involvement of the alimentary tract in naturally FeLV-infected cats from January 2010 to May 2022. Combinations of keywords “feline,” “lymphoma,” “alimentary,” “alimentary lymphoma,” “gastrointestinal,” “intestinal,” and “FeLV” were used for the digital database search. The search in book records was performed by reviewing the histologic description and diagnosis of necropsied cats. The initial selection included cases that had lymphoma in the final diagnosis and description of neoplastic lymphocytes in organs of the alimentary tract. Information on age, breed, sex, and clinical history was retrieved from the necropsy reports. Clinical signs and time from diagnosis to death, when available, were retrieved from the clinical records. FeLV-antigen-positive cats were selected based on either positive rapid immunochromatographic tests results reported in their medical history or by postmortem detection of infection through immunohistochemistry (IHC) of bone marrow and lymph node, when clinical history did not provide retrovirus status. Feline immunodeficiency virus (FIV) status was retrieved from clinical records or from necropsy reports in which anti-FIV IHC was performed on bone marrow samples.
Involvement of the alimentary tract in this study refers to the presence of neoplastic lymphocytes in the upper and/or lower alimentary tract and did not necessarily originate from these sites. Thus, primary alimentary lymphomas and multicentric lymphomas with secondary involvement of the alimentary tract were included in the present study. If the cat had a previous diagnosis of alimentary lymphoma via biopsy or presented with gastrointestinal signs as the main clinical issue, these cases were considered primary lymphomas. This assessment was further analyzed in conjunction with the gross, histologic, and immunohistochemical results. If only the stomach and small and large intestines were affected, or if the mesenteric lymph nodes, pancreas, and/or liver were concomitantly affected, but without involvement of other organs, the cases were considered primary. Multicentric lymphomas comprised the cases with masses in the mediastinum (mediastinal lymphomas) or with multiple affected organs in both abdominal and thoracic cavities, in which there was no clinical history of gastrointestinal disease or diagnosis of alimentary lymphoma, or there were clinical signs and findings compatible with lymphoma in a different anatomic location.
Gross and Histologic Evaluation
The photographs taken at necropsy and the necropsy reports were analyzed to determine the organs with macroscopic lesions and the main gross aspects of each site. Standard necropsies were performed in all cases with collection of samples of multiple organs, with and without lymphoma, including the spleen, liver, lymph nodes, kidneys, bone marrow, lungs, heart, adrenal glands, thyroid, tongue, esophagus, stomach, small and large intestines, pancreas, and urinary bladder. The cerebrum, cerebellum, and brainstem were routinely collected, while the spinal cord was only assessed when the clinical history and gross findings indicated possible lesions.
The mesenteric lymph nodes were grossly assessed for enlargement (mild, moderate, or marked) and loss of corticomedullary distinction. The presence and degree of lymph node enlargement were identified and assessed by lymph node measurements, when provided in the necropsy report, and subjectively based on photographs and description of enlargement in necropsy reports. To the best of our knowledge, there are no studies reporting normal mesenteric lymph node sizes in cats based on necropsy or biopsy databases. Therefore, we relied on a study using computed tomography in healthy cats. 27 In that study, jejunal lymph nodes had a mean length of 1.7 cm. Although other lymph nodes within the cranial mesenteric lymph center were smaller, we chose to establish 1.7 cm as the cut-off value to avoid including borderline cases that could potentially represent normal variation and ensured that only truly enlarged lymph nodes were categorized as such. Mild enlargement was defined as lymph nodes twice their normal size (~ 3.5 cm in length), moderate as 3–4 times their normal size, and marked as 5 times their normal size. The mesenteric lymph nodes were considered involved when the cranial mesenteric lymph center, comprised cranial mesenteric, jejunal, cecal, and colic lymph nodes, was affected. Involvement of other regional lymph nodes associated with the alimentary tract, such as gastric lymph node, pancreaticoduodenal, and caudal mesenteric lymph nodes, was also evaluated.
All tissues were fixed in 10% neutral-buffered formalin, routinely processed for histology, and stained with hematoxylin and eosin. Only FeLV-infected cats with a definitive diagnosis of lymphoma involving the alimentary tract were included in the study, and cases in which formalin-fixed, paraffin-embedded tissue blocks were not found or that had advanced autolysis were excluded. The histologic slides were reviewed by 3 of the authors (PRR, EMSS, and SPP) to confirm the diagnosis and determine the affected sites. The anatomic locations of lymphoma were initially assessed by evaluation of macroscopic photographs and information present on necropsy reports and were histologically confirmed by the presence of neoplastic lymphocytes within these sites. The histologic distribution of the neoplastic cells within the stomach and small and large intestines was assessed and classified as affecting only the mucosa, the mucosa and the submucosa, or all layers (transmural invasion).
Immunohistochemistry and Classification of Lymphomas
IHC was performed in at least 3 sections of selected tissues containing the tumor, which included the tongue, stomach, and/or small and large intestines, in addition to a section of lymph node, kidney, or liver with lymphoma to properly immunophenotype the neoplastic lymphocytes. Three micrometer sections were cut from selected paraffin blocks and IHC was performed following previously described protocols. 29 Polyclonal rabbit anti-CD3 (ready-to-use; code A0452; Dako) and polyclonal rabbit anti-CD20 antibodies (1:400; code PA516701; Thermo Fisher Scientific) were employed in tumor sections to determine the cell of origin. Expression of FeLV was assessed in sections of bone marrow, lymph node, and lymphoma using monoclonal mouse anti-FeLV gp70 antigen antibody (1:500; clone C11D8; Bio-Rad). The amplification signal was obtained with NovoLink Max Polymer Detection System (Leica Biosystems), and the reactions were revealed with 3-amino-9-ethylcarbazole chromogen kit (AEC, Biocare Medical). All slides were counterstained with hematoxylin. A lymph node from a cat with confirmed FeLV infection was used as positive control, and for negative control, the primary antibodies were replaced by Universal Negative Control Serum (Biocare Medical, Pacheco, California).
Immunoreactivity for T- and B-cell markers was evaluated by 2 of the authors (PRR and SPP). Areas with the highest density of positive cells were assessed and areas of necrosis were avoided. Tumors were then classified according to the REAL/WHO classification of lymphoid neoplasms, as recommended for use in animals. 39 Semiquantitative analysis of FeLV-specific IHC on tumoral sections was performed and cases were classified as having mild (20%), moderate (21%–50%), and marked (> 51%) immunolabeling based on the percentage of positive neoplastic lymphocytes.
Statement of Ethics
This study was conducted according to relevant institutional guidelines for retrospective studies involving necropsy data and embedded paraffin tissue samples, and it was approved by the Research Committee under the number 42120.
The data analyzed in this study are available upon reasonable request to the author.
Results
Epidemiologic and Clinical Information
In the 12-year period, 1913 cats were necropsied, and FeLV status was available for 1664 cats, with a 37% prevalence of FeLV infection (616/1664). Of the 616 FeLV-infected cats, 155 (25%) had lymphoma, of which 32 cases, representing 21% of all lymphomas diagnosed in FeLV-infected cats, met the inclusion criteria and were selected for the study. Regarding the affected breeds, 31/32 cases (97%) were mixed-breed cats, with only 1 Siamese cat. Male and female cats were equally affected, with 16 cases each. The age of the cats ranged from 1 to 14 years, with a median age of 4 years and mean age of 62 months (5 years and 2 months old). The FIV status was available in 26 cats, of which 7 were infected, with a prevalence of 27%.
In the same period, lymphoma, regardless of anatomic location and retrovirus status, was diagnosed in 299 cats (299/1913), comprising 17% of all diagnoses in necropsied cats. The alimentary tract was involved (either primarily or secondarily) in 114 of the 299 cases (38%). Retrovirus status was available in 102 of these cases (102/114; 89%), with lymphoma being more commonly diagnosed in FeLV-negative cats (70/102; 69%). Of the 70 FeLV-negative cats with lymphoma involving the alimentary tract, the median and mean age of death of the cats were 13 and 12 years old, respectively.
Lymphoma Subtype and Gross Evaluation
Lymphoma was more commonly classified as multicentric with secondary involvement of the alimentary tract (21/32; 66%), in which the alimentary lymphoma was considered part of a disseminated disease, and the cat initially did not have gastrointestinal signs (Table 1). In 11/32 cases (34%), lymphoma was classified as primary, and involvement of organs outside the alimentary tract was considered secondary (Table 2). The small intestine was the most common anatomic location affected (23 cases), followed by the large intestine (19 cases) and stomach (18 cases). Thickening of the intestinal and gastric walls was the most common gross lesion (23 cases; 16 in the small intestine, 6 in the large intestine, and 1 in the stomach), while nodules were observed in 16 cats (10 in the small intestine, 6 in the large intestine, and 4 in the stomach), of which 5 affected the ileocecocolic junction. In most cases (22/32; 69%), multiple organs within the alimentary tract were concomitantly affected.
Epidemiologic and pathological characterization of 21 cases of multicentric lymphomas with secondary involvement of the alimentary tract in cats infected with feline leukemia virus.
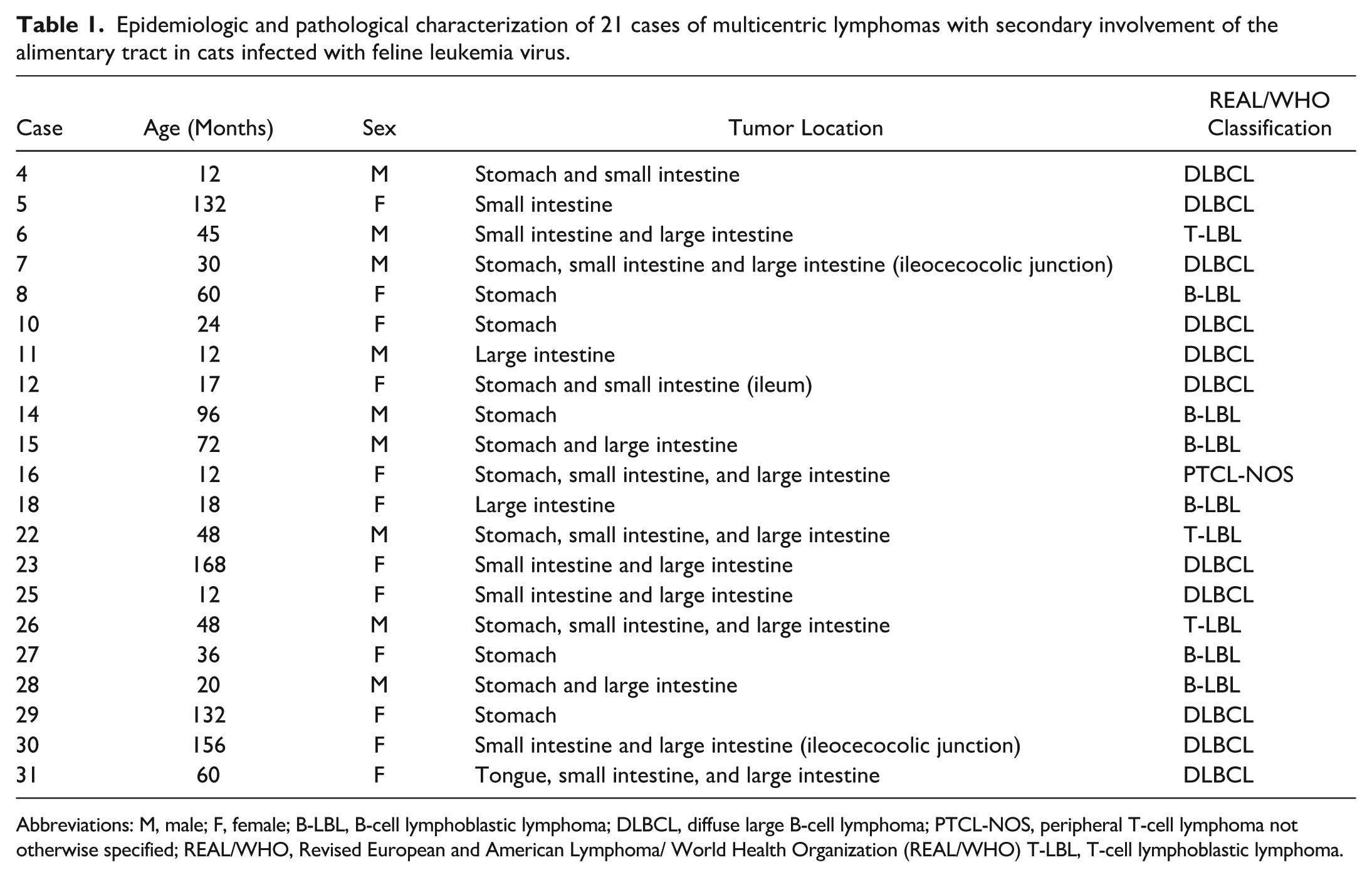
Abbreviations: M, male; F, female; B-LBL, B-cell lymphoblastic lymphoma; DLBCL, diffuse large B-cell lymphoma; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; REAL/WHO, Revised European and American Lymphoma/ World Health Organization (REAL/WHO) T-LBL, T-cell lymphoblastic lymphoma.
Epidemiologic and pathological characterization of 11 cases of primary alimentary lymphoma in cats infected with feline leukemia virus.

Abbreviations: M, male; F, female; B-LBL, B-cell lymphoblastic lymphoma; DLBCL, diffuse large B-cell lymphoma; EATL, enteropathy-associated T-cell lymphoma; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; REAL/WHO, Revised European and American Lymphoma/ World Health Organization.
The stomach was involved in 18/32 cats (56%), and it was the only affected organ of the alimentary tract in 6 of these cases. In most cases (12/18; 67%), the stomach was unremarkable at necropsy. Macroscopic lesions were observed in 6/18 cases (33%), which included 2 cases of ulcerated nodules (Fig. 1a), 2 cases of nonulcerated nodules (Fig. 1b), 1 case with gastric wall thickening, and 1 case with multiple ulcers.
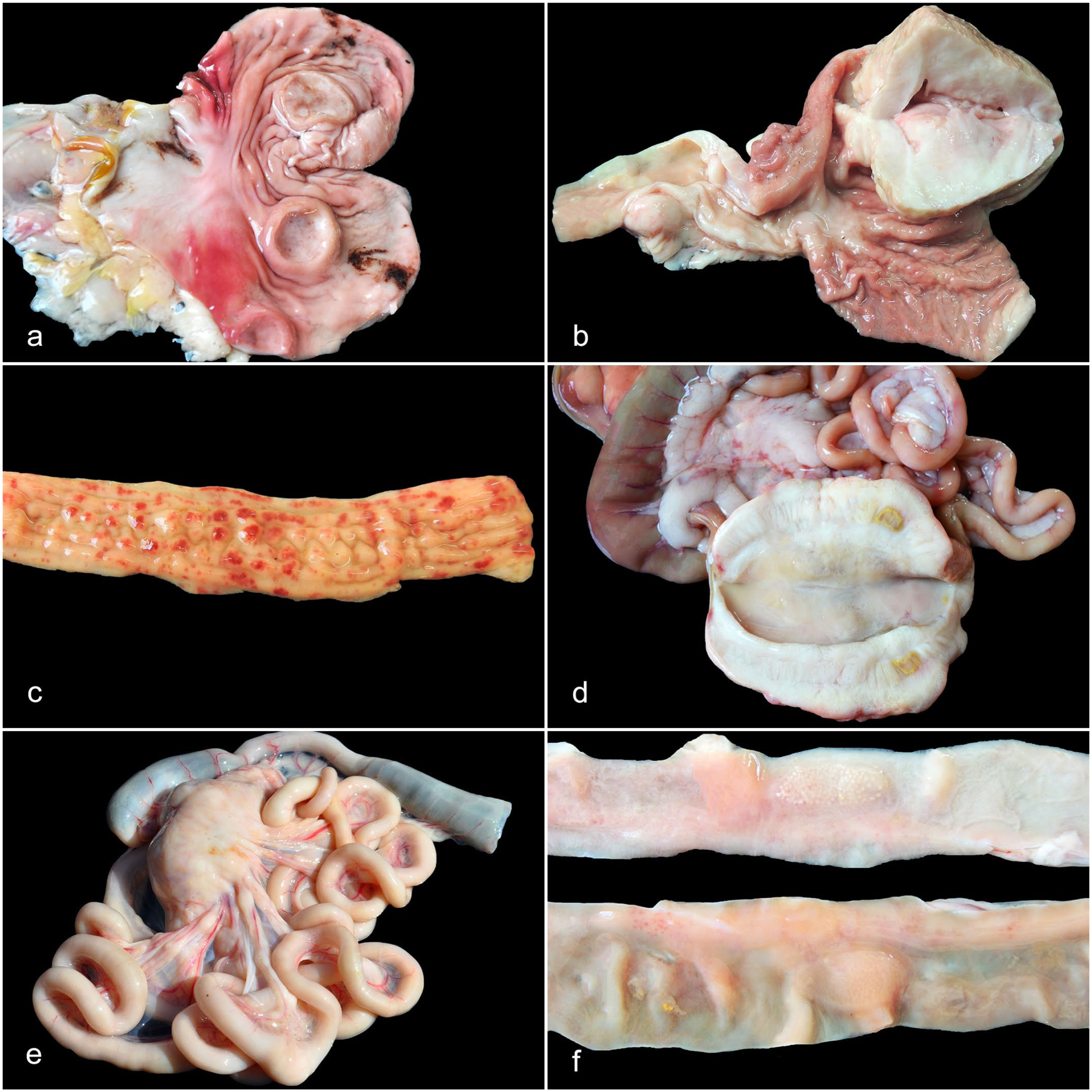
Gross aspects of lymphoma with involvement of the alimentary tract in cats infected with feline leukemia virus. (a) B-cell lymphoblastic lymphoma, stomach, cat. There are ulcerated nodules up to 2.0 cm in diameter protruding from the gastric mucosa, which has brown ulcerated areas. Case 14. (b) Diffuse large B-cell lymphoma (DLBCL), stomach, cat. A 7 × 6 × 7.5 cm, white mass partially obstructs the gastric lumen. Case 29. (c) Intestinal T-cell lymphoma (compatible with enteropathy-associated T-cell lymphoma [EATL] type I), small intestine, cat. There is diffuse and severe thickening of the intestinal wall, with areas of hyperemia. Case 21. (d) Intestinal T-cell lymphoma (compatible with EATL type I), small intestine, cat. The ileum is surrounded by an 8 × 4 × 3 cm, white mass, which diffusely thickens the intestinal wall and narrows the lumen. Case 17. (e) DLBCL, small and large intestines, and mesenteric lymph nodes, cat. The small intestine is diffusely thickened and has nodules expanding the intestinal wall. The mesenteric lymph nodes are markedly enlarged. Case 19. (f) DLBCL, small intestine, cat. Peyer’s patches are enlarged and prominent, and there is mild thickening of the intestinal wall. Case 25.
The small intestine was affected in 23/32 cases (72%), and it was the most commonly affected site of the alimentary tract in the present study. Of these, only 4/23 were confined to the small intestine. Most cases had gross lesions (20/23; 87%), and segmental thickening of the intestinal wall was frequently observed (16/23 cases; 70%) (Fig. 1c), followed by nodules/masses in 10/23 cats (43%) (Fig. 1d). Six cats had both wall thickening and nodules/masses (Fig. 1e). In 1 case, besides thickening of the wall, prominent and enlarged Peyer’s patches were also observed (Fig. 1f).
The large intestine was affected in 19/32 cases (59%), and in 1 case, it was the only site of the alimentary tract involved. On gross evaluation, 9/19 cats (48%) did not have any lesions in the large intestine, whereas 6/19 (32%) had thickening of the large intestinal wall, one of which also had prominent and enlarged lymphoid follicles. In 5/19 cats (26%), the ileocecocolic junction was affected, involving both small and large intestines, with enlargement and mass formation in the region. It was associated with wall thickening in 2 cases, and perforation of the cecum in 1 cat (Fig. 2a). The remaining cat (5%) only had a nodule.
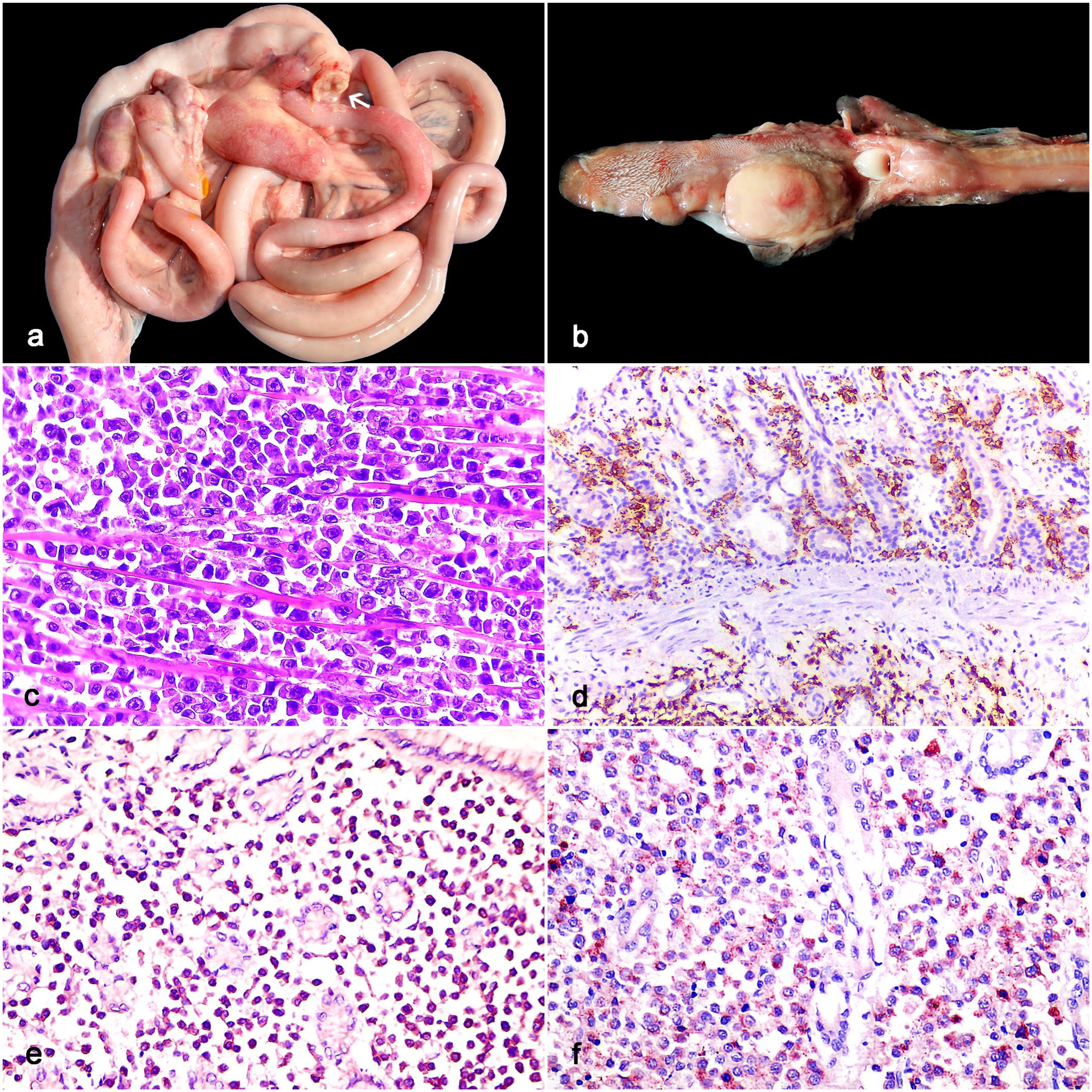
Gross, histologic, and immunohistochemical features of lymphoma with involvement of the alimentary tract in cats infected with feline leukemia virus (FeLV). (a) Diffuse large B-cell lymphoma (DLBCL), small and large intestine and mesenteric lymph node, cat. The intestinal wall is thickened and expanded by a white to pink mass in the ileocecocolic junction, which is associated with perforation of the cecum (arrow). Case 1. (b) DLBCL, tongue, cat. There are 2 white masses in the caudal portion of the dorsal surface of the tongue. Case 31. (c) Intestinal T-cell lymphoma (compatible with enteropathy-associated T-cell lymphoma type I), small intestine, cat. A sheet of large (nuclei > 2 red cell in diameter) neoplastic lymphocytes effaces the muscular layer. Neoplastic lymphocytes have stippled chromatin and single prominent nucleoli. Case 20. Hematoxylin and eosin (HE). (d) DLBCL, small intestine, cat. The neoplastic lymphocytes within the intestinal mucosa and submucosa are immunolabeled for the B-cell marker CD20. Case 7. CD20 immunohistochemistry (IHC). (e) Peripheral T-cell lymphoma not otherwise specified, stomach, cat. The neoplastic lymphocytes infiltrating the gastric mucosa are diffusely and moderately immunolabeled for the T-cell marker CD3. Case 16. CD3 IHC. (f) DLBCL, small intestine, cat. The neoplastic lymphocytes within the intestinal mucosa are large (nuclei > 2 red cell in diameter) and exhibit granular and cytoplasmic immunolabeling for FeLV gp70 antigen. Case 7. FeLV gp70 IHC.
The tongue was affected in one case, which was characterized by 2 white, soft nodules, measuring 5 × 3 × 3 cm and 1 × 1 × 0.5 cm, in the caudal portion of the dorsal surface of the tongue (Fig. 2b). It was part of disseminated disease, in which neoplastic lymphocytes were also observed in the small and large intestines, lymph nodes, tonsils, liver, kidneys, pancreas, and urinary bladder.
The mesenteric lymph nodes were affected in 22/32 cases (69%), which had different degrees of involvement, ranging from mild (2/22) to moderate (6/22) and marked (14/22) enlargement with loss of corticomedullary distinction and replacement by a white, soft mass (Supplemental Figure S1). Other lymph nodes associated with the alimentary tract were affected in 6 cats, but the mesenteric lymph node was involved in all of these cases. These included the pancreaticoduodenal lymph node in 4 cats, and the caudal mesenteric, hepatic, and gastric lymph nodes in 1 cat each.
In most cases (30/32; 94%), neoplastic lymphocytes were observed in more than 2 organs unrelated to the alimentary tract. Distant lymph nodes were affected in 18/32 cats (56%), and no neoplasm was found in any lymph nodes in 4/32 cases (13%). A large mass in the mediastinal region, which occupied most of the thoracic cavity and compressed the lungs and heart, was noted in 7/32 cases (22%). The liver and kidneys were commonly involved, and included 25/32 (78%) and 24/32 (75%) cases, respectively. The lungs were affected in 13/32 cats (41%), the spleen in 11/32 cases (34%), and the pancreas in 10/32 cases (31%). The bone marrow, the adrenal glands, and the urinary bladder were affected in 7/32 cases (22%) each. Less commonly affected sites included the heart, thyroid gland, and brain, which were affected in 5, 4, and 3 cases, respectively, and bone (vertebra), skeletal muscle, tonsils, and diaphragm, which were affected in 1 case each.
Histological and Immunohistochemical Features and Revised European and American Lymphoma/World Health Organization Classification
Histologically, 17/32 cases (53%) were composed of large cells (Fig. 2c), followed by 14/32 (44%) cases of intermediate-sized lymphocytes, and only 1/32 case (3%) of small cell lymphoma. On immunohistochemical analysis, in 24/32 cases (75%), the neoplastic cells had multifocal to diffuse, moderate to marked, membranous labeling for CD20 and were classified as B-cell lymphomas (Fig. 2d). The remaining 8/32 cases (25%) had a T-cell origin and were characterized by multifocal to diffuse, moderate to marked, cytoplasmic and membranous CD3 immunolabeling (Fig. 2e). Regardless of the anatomic location, B-cell lymphomas predominated, comprising 14/18 cases (78%) in the stomach, 15/23 cases (65%) in the small intestine, and 12/19 cases (63%) in the large intestine. The only case affecting the tongue was of B-cell origin and was diagnosed as diffuse large B-cell lymphoma (DLBCL). Regardless of immunophenotype, neoplastic lymphocytes immunolabeled for FeLV antigen in most cases (31/32; Fig. 2f), with the percentage of positive neoplastic lymphocytes varying from mild (8/31) to moderate (11/31) to marked (12/31).
The extent of the neoplasm in the stomach and intestines varied among cases and, within the same cat, it also varied according to the affected anatomic location. In the stomach, the neoplastic lymphocytes were mostly observed multifocally in the mucosa in 9/18 cases (50%) and extended into the submucosa in 5/18 cases (28%). Transmural infiltration was less frequently observed (4/18; 22%), which grossly corresponded to nodules in three cats. In the small and large intestines, transmural invasion was the most frequent pattern, accounting for 15/23 cases (65%) and 12/19 cases (63%), respectively, followed by neoplastic cells mostly involving the mucosa and multifocally extending into the submucosa in 7/23 cases (30%) in the small intestine and in 4/19 cases (21%) in the large intestine. Rarely, the neoplastic cells expanded the lymphoid follicles of the submucosa, occasionally extending into the mucosa (1 case and 3 cases in the small and large intestine, respectively). This last pattern was grossly characterized by enlarged and prominent Peyer’s patches in 1 case.
According to the REAL/WHO classification system as recommended for use in animals, the most common diagnosis was DLBCL, which accounted for 15/32 cases (47%), followed by B-cell lymphoblastic lymphoma (8/32; 25%). Less commonly, enteropathy-associated T-cell lymphoma type I was diagnosed in 4/32 cats (13%), followed by T-cell lymphoblastic lymphoma in 3/32 cases (9%), and peripheral T-cell lymphoma not otherwise specified and B-cell lymphocytic lymphoma in 1 case each. The distribution of cases according to the REAL/WHO classification system with respect to organ involvement is provided in Table 3.
Distribution of lymphomas with involvement of the alimentary tract in cats infected with feline leukemia virus according to REAL/WHO classification system as applied for use in animals.
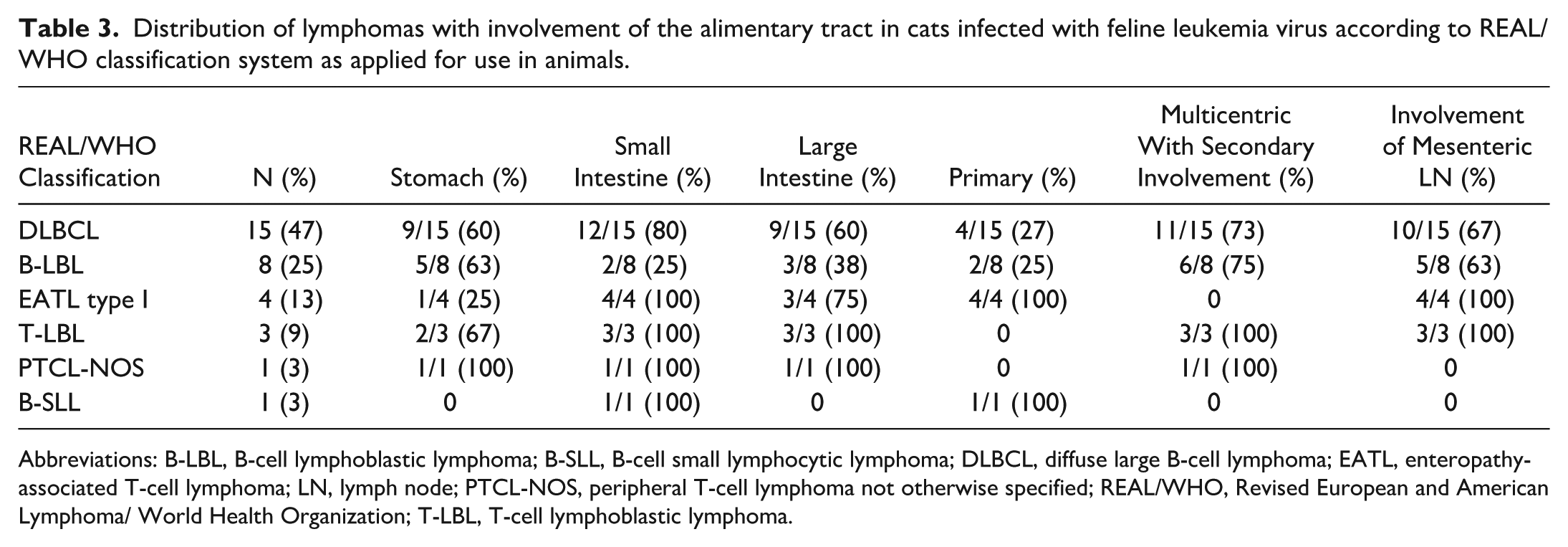
Abbreviations: B-LBL, B-cell lymphoblastic lymphoma; B-SLL, B-cell small lymphocytic lymphoma; DLBCL, diffuse large B-cell lymphoma; EATL, enteropathy-associated T-cell lymphoma; LN, lymph node; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; REAL/WHO, Revised European and American Lymphoma/ World Health Organization; T-LBL, T-cell lymphoblastic lymphoma.
Discussion
This study described 32 cases of lymphoma affecting the alimentary tract in FeLV-infected cats, with a median age of 4 years and mean age of 5.2 years, which is different from that described for feline alimentary lymphomas in general, wherein the mean age varies from 10 to 12 years.18,23,28,37 However, in other studies, the cats are frequently FeLV-negative, as commonly observed in intestinal lymphomas, which explains this difference of age in the affected cats. FeLV-infected cats have a significantly lower median age of death than non-infected cats, 8 and in our study, the age of death for FeLV-infected cats that died with alimentary lymphoma was similar to that described for FeLV-infected cats that died of multiple causes. 4 This suggests that FeLV may have played a more important role in reducing life expectancy than the anatomic form or distribution of lymphomas in this study. Our findings reinforce this reduction in life expectancy in FeLV-infected cats, including in cats with lymphoma affecting the alimentary tract. Most cats were mixed-breed cats, which is representative of the cat population submitted to necropsy. 8 Male and female cats were equally affected, and this was expected, as no sex predisposition has been observed for FeLV infection, which is different from that described for FIV. 8
Alimentary lymphoma is the most commonly diagnosed type of lymphoma in cats, both in countries where vaccination and control programs for FeLV have been successfully implemented,6,20,32,41 and in Brazil, a country with high prevalence of FeLV infection.19,33 However, a difference is noted among these countries, in which mediastinal lymphoma still represents the second most common lymphoma in Brazil, with prevalence ranging from 28% to 34%,7,19 while it is infrequently diagnosed in Europe and the United States (1%),20,41 and in Japan (11%).6,32 This highlights the difference in the studied population of these investigations, and may represent a bias in our study, in which 22% of the cases also had mediastinal masses.
Regarding the anatomic location, the small intestine was the most affected site, as reported in previous studies.18,23,28,30,45 The small intestine has been described to be exclusively affected in 80% of the cases,28,45 which is different from our study, with the small intestine being solely involved in only 17% of cases. This may be explained by the fact that most of our cases represented advanced forms of the disease, in which the tumor may have spread and affected different portions of the alimentary tract, as well as parenchymal organs. Lymphomas affecting the stomach and oral cavity are uncommonly confined to these locations, 24 as observed in the present study. As this was a retrospective study of necropsied cats, there was difficulty in categorizing the lymphomas, as previously described, 12 and many cases may fit into more than one anatomic location with unusual associations of affected organs. Thus, prospective and clinically controlled studies should be performed to better determine and characterize these anatomic forms.
Grossly, for the small intestine, the most common lesion was segmental thickening of the intestinal wall, in accordance with that described for intestinal lymphomas, 23 whereas the stomach did not have gross lesions in most cases in this study. This may be explained by the fact that in these cases the neoplastic cells were restricted to the gastric mucosa and submucosa rather than transmural, and the latter is more common in more advanced stages of the disease and with nodular formation. 23 Our findings corroborate that it may be common for lymphomas in the stomach to be grossly normal at necropsy or have mild or nonspecific lesions, such as thickening of the wall, and the absence of macroscopic lesion or nodules does not rule out the possibility of lymphoma.
Studies of gastrointestinal lymphomas are frequently performed using clinical biopsy samples,16,21,31 unlike our study on necropsied animals. Comparisons of gross lesions between such investigations should be done with caution, due to autolysis with destruction of soft tissue and postmortem changes, such as hemoglobin imbibition, and distension by gas. 5 Also, in our cases, the time between death and necropsy could not be determined for most cases and it varied among cats, which hinders further assessment. On the contrary, endoscopy requires a systematic, rigorous, and complete examination of the gastrointestinal tract, and endoscopic reports should provide detailed gross findings to be correlated with the histologic findings. 43 Subtle lesions may be more frequently noted endoscopically, and these could be obscured by postmortem changes during necropsy. Still, necropsy specimens allow evaluation of the entire gastrointestinal tract and collection of larger, full-thickness specimens of various sites and organs. Endoscopic, full-thickness biopsy, and necropsy samples can all contribute to identifying the gross, histologic, and immunohistochemical features of gastrointestinal lymphomas. 23
Histologically, 97% of the lymphomas were composed of large and intermediate lymphocytes, which is different from that described for gastrointestinal lymphomas as a whole, as small cells are more frequently observed in enteric lymphomas in cats.16,17,23,38 This may be explained by the fact that this study included multicentric disease with involvement of the alimentary tract and that all cats were FeLV-positive. FeLV infection is often associated with aggressive lymphomas that have high mitotic count and are frequently classified as high grade by the National Cancer Institute Working Formulation, 38 but it is rarely detected in low-grade, small cell intestinal lymphomas.1,17 In contrast to small cell T-cell lymphomas, which is frequently diagnosed in aged and FeLV-negative cats,23,40 our study did not find this type of lymphoma in young, FeLV-positive cat, with large and intermediate-cell lymphomas being more common. In the 12-year search we performed in the necropsy database of the pathology laboratory, small cell T-cell lymphoma was not observed in any of FeLV-infected cats with lymphoma of the alimentary tract. Thus, when an FeLV-infected cat is presented for clinical evaluation and an intestinal lymphoma is diagnosed, it is likely to be a more aggressive tumor, as has been described for all types of lymphomas in FeLV-positive cats. 40
Even though immunophenotype has been associated with anatomic location, in which B-cell lymphomas predominated in the stomach and large intestine,18,28 and T-cell lymphomas predominated in small intestine, in our study of FeLV-infected cats, B-cell lymphomas predominated in all anatomic sites within the alimentary tract. When National Cancer Institute Working Formulation and REAL/WHO schemes are used in conjunction, B-cell and T-cell lymphomas are variably diagnosed in intermediate to high-grade alimentary lymphomas, while low-grade alimentary lymphomas are mostly of T-cells, 1 which could explain the difference in our findings from the other studies.
The most common type of B-cell lymphoma according to the REAL/WHO classification system was DLBCL, accounting for 63% of these cases, which is in accordance with previous studies.23,25,28,41,45 Still, this could also be influenced in the present study by the large proportion of B-cell lymphoma that have been diagnosed in similar populations of cats in Brazil.19,33 In addition to that, large-cell lymphomas are mostly of B-cell origin (68%), 18 and in our study, many lymphomas (53%) were of large lymphocytes. Following DLBCL, B-cell lymphoblastic lymphoma was the second most common type of lymphoma in this study, and lymphoblastic lymphomas are known to be aggressive tumors and of T- or B-cell origin. Categorizing these tumors as lymphoblastic relies mostly on cell features, such as nuclei of intermediate size (1.5–2 red cell in diameter), with dispersed nuclear chromatin and inconspicuous nucleoli. 40 B-cell lymphoblastic lymphoma has been rarely described in investigations of alimentary lymphoma, 41 but it accounted for half of the cases in a study that included multicentric diseases with gastrointestinal involvement. 42 We believe that our findings differed from the study conducted by Vezzali et al 41 because multiple cases herein comprised disseminated disease, in which lymphoblastic lymphoma/leukemia is more frequently observed. 40 The large difference in the composition of the studied population among investigations may also explain these discrepancies, in which the cats of our study were young and FeLV-infected, compared with aged FeLV-negative cats from other studies. Moreover, when primary alimentary lymphomas and multicentric lymphomas with gastrointestinal tract involvement were evaluated, B-cell tumors predominated, 42 which may indicate that inclusion of multicentric disease increases the diagnosis of B-cell lymphomas. Finally, T-cell lymphoblastic lymphoma is the most common type of thymic lymphoma in FeLV-infected cats; 39 however, in our study, mediastinal masses comprised only 7 cats of 32 cases. This may explain the high ratio of B-cell lymphoblastic lymphoma and T-cell lymphoblastic lymphoma in our study and demonstrate that the prevalence of this type of lymphoma may vary according to the anatomic distribution.
The FeLV antigen is detected in only up to 30% of feline alimentary lymphomas,11,17,19 similar to that observed in our entire population of necropsied cats in a 12-year period. However, previous studies have shown that FeLV may be more frequently detected in lymphomas, including alimentary lymphomas, when PCR is used to detect provirus.11,44 PCR was not conducted in the current work, and could have increased the number of cats included in the study; however, it is unlikely that FeLV-regressive infection is responsible for most lymphomas in cats. 35 More studies should be conducted with intestinal lymphomas of cats to determine the proportion of antigen-negative but provirus-positive tumors, 34 and it would be ideal to concomitantly perform IHC and PCR in sections of lymphomas to identify both progressively and regressively infected cats.11,33,44 Even though FIV has been historically associated with lymphoma 14 a recent study with a similar population of necropsied cats in Brazil showed that FIV-infected cats were not more likely to die from lymphoma when compared with non-infected cats. 8 FIV infection was detected in 27% of cats of the current work, which is considered higher than other studies in Brazil, 4 but similar to that observed in the same population of cats that died of multiple causes (23%). 8 Thus, considering the overall population and FIV prevalence, this virus did not appear to influence the pathological presentation of lymphomas.
Alimentary lymphomas are not exclusively of FeLV-negative and old cats and can occur in young FeLV-infected cats, as seen in our study. Causes other than FeLV have been suggested to play a role in lymphomagenesis, as cases of lymphoma, especially gastrointestinal lymphoma, have increased concomitantly with a decrease in prevalence of FeLV infection.20,22,35 However, these studies were conducted in countries with a low prevalence of FeLV, unlike that observed in our population, in which FeLV-associated lymphomas are still frequent, including the ones affecting the alimentary tract. Small cell mucosal T-cell lymphoma was not diagnosed in any of our cases, reinforcing a significant difference in immunophenotype of alimentary lymphomas when FeLV-infected cats are evaluated, and consequently, possibly a different oncogenic mechanism. The lymphomas affecting the alimentary tract of FeLV-infected cats were more frequently of B-cell phenotype, independent of the gastrointestinal location, and of intermediate- or large-cell sizes. Even though FeLV has been historically associated with T-cell lymphomas,14,15 we have demonstrated that in cases of lymphoma affecting the alimentary tract in FeLV-infected cats, either as a primary or multicentric lymphoma, it is mostly of B-cell origin. FeLV antigen was found within the neoplastic lymphocytes in most cases, reinforcing the presence of the virus in the tumoral environment. Our cases were commonly associated with involvement of other organs, either as part of multicentric lymphoma or as secondary involvement in a primary alimentary lymphoma, and with multiple organs within the alimentary tract being concomitantly involved, suggesting a more aggressive behavior. The aggressive behavior is reinforced by the fact that the most common types of lymphomas diagnosed according to the REAL/WHO classification system were DLBCL and B-cell lymphoblastic lymphoma. Lymphomas affecting the alimentary tract of FeLV-infected cats have markedly distinct clinicopathological features from those of FeLV-negative cats, demonstrating an aggressive behavior with multiple organ involvement and evidence for the participation of the retrovirus in its oncogenesis.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251400689 – Supplemental material for Pathological characterization of lymphomas with involvement of the alimentary tract in feline leukemia virus-infected cats
Supplemental material, sj-pdf-1-vet-10.1177_03009858251400689 for Pathological characterization of lymphomas with involvement of the alimentary tract in feline leukemia virus-infected cats by Paula Reis Ribeiro, Lauren Santos de Mello, Emanoelly Machado Sousa da Silva, Kalvin Ferreira Siqueira, Paula Reis Pereira, Marcele Bettim Bandinelli, Welden Panziera, David Driemeier, Luciana Sonne and Saulo Petinatti Pavarini in Veterinary Pathology
Footnotes
Acknowledgements
We are thankful to the technician Bárbara Krebs for her technical support in the present study.
Supplemental material for this article is available online.
Author Contributions
PRR, LSM, EMSS, and MBB contributed to conceptualization, investigation, validation, methodology, and writing original draft. KFS, PRP, WP, DD, and LS contributed to investigation, writing—review, and editing. SPP contributed to conceptualization, investigation, validation, methodology, resources, supervision, project administration, funding acquisition, writing—review, and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grant number 305460/2021-8), and it was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). Brasil—Finance code 001.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
