Abstract
Canine cutaneous epitheliotropic T-cell lymphoma is a neoplasm with heterogeneous clinical and histopathological presentations. Survival times and responses to therapy are variable, and indicators to predict outcomes are lacking. Clinical and histopathological parameters from 176 archival cases from the University of Pennsylvania and University of Bern (2012–2018) were investigated for associations with clinical outcomes. Histopathological evaluation used digitized whole slide images and QuPath software. Cases included 107 female and 69 male dogs from 48 breeds, with a mean age of 10.4 years. Most common clinical signs were erythema (n = 131), crusting (n = 108), and scaling (n = 102). Affected sites were haired skin (n = 159), lip (n = 74), nasal planum (n = 49), and paw pads (n = 48). The median survival time (MST) was 95 days (1–850). Dogs had 4.26-fold and 2.82-fold longer MST when treated with chemotherapy and prednisone, respectively, than when receiving supportive care. Haired skin involvement (hazard ratio [HR]: 2.039, 95% confidence interval [CI]: 1.180–3.523), erosions/ulcers (HR: 1.871, 95% CI: 1.373–2.548), nodules (HR: 1.496, 95% CI: 1.056–2.118), and crusting (HR: 1.454, 95% CI: 1.061–1.994) were clinical parameters predicting poor outcomes, whereas complete posttherapeutic clinical remission (HR: 0.469, 95% CI: 0.324–0.680) and a stable disease (HR: 0.323, 95% CI: 0.229–0.456) were associated with longer survival. Histopathological features associated with the increased risk of death were extensive infiltration of the panniculus (HR: 2.865, 95% CI: 1.565-4.809), mitotic count ≥7/high-power field (HR: 3.027, 95% CI: 2.065–4.439), cell diameter ≥10.0 µm (HR: 2.078, 95% CI: 1.281–3.372), and nuclear diameter ≥8.3 µm (HR: 3.787, 95% CI: 1.647–8.707).
Canine cutaneous epitheliotropic T-cell lymphoma (CETCL) is a spontaneous neoplastic skin disease with a variety of clinical presentations and morphological features. Dogs diagnosed with CETCL may present with erythema, scaling, depigmentation, crusts, erosions, and ulcers affecting the haired skin, mucocutaneous junctions, and mucous membranes.4,17 In later stages, there is formation of plaques and nodules that spread to lymph nodes and visceral organs. Response to chemotherapy is often poor, and the disease progresses despite treatment.3,9 The rapidity of progression, however, is variable, with survival times ranging from a few days or weeks to up to 5 years after diagnosis.3,4,10,11 One study found the occurrence of solitary lesions and the restriction of lesions to mucous membranes and mucocutaneous junctions to be associated with longer survival, albeit with a highly variable survival time in both groups. 3
The diagnosis of CETCL is routinely made by histopathology and immunohistochemistry (IHC). Canine CETCL belongs to the group of peripheral (mature) T-cell lymphomas and is subclassified according the updated Revised European-American Lymphoma/World Health Organization (REAL/WHO) classification system of lymphoid tumors as pagetoid reticulosis (PR; neoplastic lymphocytes confined to epidermis and adnexa), mycosis fungoides (MF, neoplastic lymphocytes infiltrating epidermis, adnexa, and dermis), and Sézary syndrome (MF in combination with leukemia). 15 In addition to the 3 major canine CETCL subtypes, a few reported cases resemble the human folliculotropic subtype of MF.2,4,18 In this subtype, the neoplastic infiltrate is centered on hair follicles, while the epidermis is spared. In humans, this subtype is frequently associated with follicular mucinosis. The classification of canine CETCL was adapted from the human European Organization for Research and Treatment of Cancer (WHO-EORTC) classification, although canine CETCL is mainly a neoplasm of CD8+ lymphocytes, whereas most human CETCL forms are derived from CD4+ lymphocytes.2,11,18
In humans, different CETCL subtypes, characterized by subtype-specific clinical signs, histomorphologic features, and immunohistochemical and molecular properties, are associated with different prognoses. Corresponding data from canine CETCL are lacking.2,18 Currently, there are no clinical, histomorphological, or molecular parameters available to reliably predict the survival and the response to chemotherapy for dogs with CETCL. The goal of this study is to evaluate multiple parameters for associations with response to treatment and survival outcomes through systematic histomorphological evaluation on a large cohort of canine biopsies with CETCL.
Materials and Methods
Case Collection
Electronic medical records and surgical biopsy archives from the University of Pennsylvania School of Veterinary Medicine (Penn Vet) and the University of Bern Institute of Animal Pathology were searched from 2012 to 2018 for all canine cases diagnosed with the following terms: cutaneous epitheliotropic lymphoma, and cutaneous lymphoma. Cases with the cutaneous lymphocytosis diagnosis were not included. Clinical data were obtained via an emailed questionnaire or phone interview with referring veterinarians and included the following information: clinical signs, date of onset, anatomic locations and progression, treatment type, occurrence and duration of clinical remission, and the date and reason for euthanasia/death. An inclusion criterion was a completed questionnaire that indicated either euthanasia or death was related to CETCL or the animal was still alive at the time of the survey.
Histopathology and IHC
The original hematoxylin and eosin (HE) slides, and IHC slides where available, of the included cases were retrieved from the archives. For each case, the biopsy with the largest tumor burden was selected, and new HE and CD3 IHC slides were created according to routine protocols of the Penn Vet Diagnostic Laboratory. For cases with an equivocal or no CD3 labeling, a subsequent IHC for granzyme B was performed. All tissues were fixed in buffered 10% formalin, embedded in paraffin, sectioned at 5 µm, and stained with HE. For IHC on a formalin-fixed paraffin-embedded material, 5-µm-thick paraffin sections were mounted on ProbeOn slides (Thermo Fisher Scientific, Waltham, MA) and immunohistochemically labeled for CD3 (1/600 dilution, Bio-Rad, MCA1477T, Rat mAb, Hercules, CA) or granzyme B (1/200 dilution, Abcam, ab4059, Rb pAb, Cambridge, UK). The immunolabeling procedure was performed using a Leica BOND RXm automated platform combined with a Bond Polymer Refine Detection kit (Leica DS9800), and immunoreactivity was revealed with the diaminobenzidine chromogen reaction. Slides were counterstained in hematoxylin, dehydrated in an ethanol series, cleared in xylene, and permanently mounted with a resinous mounting medium (Thermo Fisher Scientific ClearVueTM coverslipper, Waltham, MA). Control tissues were used and labeled as expected. Positive controls were lymph node and canine regressing histiocytoma for CD3 and granzyme B, respectively. Negative controls were an irrelevant isotype-matched rat monoclonal antibody and an irrelevant rabbit polyclonal antibody for CD3 and granzyme B, respectively. IHC specimens were evaluated by 2 authors (M.D. and A.C.D.).
Digital Pathology
The generated HE slides were scanned at 40× magnification using either the Aperio AT2 or Aperio Versa scanner (Leica Biosystems, Inc., Buffalo Grove, IL), and whole slide images (WSIs) were saved in the svs format. The WSIs were analyzed using the open-source digital pathology software QuPath. 1 On each WSI, the most severely affected specimen was selected (Fig. S1a), within which a representative area of 0.237 mm2 (corresponding to a single high-power field [HPF]), comprising epidermis and/or adnexa, was highlighted using the rectangle tool (Fig. S1b). Within this rectangle, the number of mitotic figures was counted using the points tool (Fig. S1c). To measure the diameter of the neoplastic cells and their nuclei, the line tool was applied on 100 cells within the rectangle measuring across the widest points of the cell and nucleus (Fig. S1d). In addition, the diameter of at least 20 red blood cells (RBCs) per WSI was measured similarly (Fig. S1e). The metadata of these line annotations were exported into an Excel sheet, and the mean, median, and smallest and largest cell and nuclear diameters were calculated. For cases with less than 100 cells within 1 rectangle, additional rectangles were drawn and annotations were applied similarly until 100 cells were measured. Neoplastic cell size category was determined as a ratio of the mean nuclear diameter/RBC diameter as shown in Table S3. 16 Image acquisition for HE and IHC photomicrographs was performed with QuPath and ImageScope (Leica Biosystems, Inc., Buffalo Grove, IL), respectively.
In addition to the measurements, the WSIs were evaluated for the degree of infiltration by neoplastic cells, cellular morphology, and additional lesions related to the neoplastic infiltration. The degree of infiltration was scored as absent, mild, moderate, or severe for the epidermis, hair follicles, sebaceous glands, epitrichial apocrine glands, dermis, and panniculus. The degree “mild” was assigned when the neoplastic cells occupied less than 30% of the infiltrated structures. The degree “moderate” corresponded to an infiltrate occupying between 30% and 60% of the infiltrated structures. The infiltrate was graded “severe” when neoplastic cells occupied more than 60% of the respective structures. Individual neoplastic cells were evaluated for the following features: amount of cytoplasm (scant vs moderate/abundant), color of the cytoplasm (eosinophilic, basophilic, amphophilic), character of the cytoplasm (finely granular vs feathery/vacuolated), eosinophilic cytoplasmic granules (present vs absent), nuclear shape (round/oval vs pleomorphic/cleaved), nuclear color and chromatin quality (dark nuclei with clumped chromatin vs clear nuclei with clumped chromatin vs dark nuclei with dispersed chromatin vs clear nuclei with dispersed chromatin), chromatin margination (yes vs no), and number of nucleoli (1 vs multiple vs poorly visible vs variable). The presence or absence of the following additional features was assessed: Pautrier’s microabscesses (intraepithelial aggregates of at least 5 neoplastic lymphocytes), follicular changes (eg, external root sheath destruction with secondary granulomatous inflammation, mucin deposition), apoptotic keratinocytes, epidermal parakeratosis, subepidermal clefting and/or ulceration, neoplastic lymphocytes in dermal lymphatic vessels, eosinophils, and reactive small lymphocytes.
Statistical Methods
All statistical analyses were conducted by a statistician (D.S.) with Stata 17MP (StataCorp, College Station, TX) with 2-sided tests of hypotheses and a P < .05 as the criterion for statistical significance. To obtain the prevalence ratio for dog breeds, the ratio between dogs of a particular breed in the CETCL cohort and in the biopsy database 2012–2018 was calculated. Dogs of breeds with n < 3 in the cohort were grouped as “other purebred dogs,” and the prevalence rate was calculated with the number of dogs of the respective breed. For the calculation of cutoff values for the mitotic count, mean cell diameter, and mean nuclear diameter, finite mixture modeling was used assuming 2 subcohorts. The cutoffs were specified at the lower 95% confidence limit of the second subcohort.
Descriptive analyses included computation of medians and ranges of continuous variables and tabulation of categorical variables. Frequency counts and percentages were used for categorical variables, such as sex and breed. An inferential statistical analysis was conducted in 2 steps. To investigate a potential clustering of clinical and/or histopathological features in terms of different CETCL subtypes, a principal component analysis (PCA) was performed as the initial exploratory step. Furthermore, a univariate Cox regression analysis as an additional exploratory analysis was used to evaluate the association between the clinical, histopathological, and immunohistochemical parameters and the risk of instantaneous death. Survival probabilities and the median survival time were estimated nonparametrically with the Kaplan-Meier method. Treatment modalities were stratified as surgery/supportive therapy, prednisone and supportive therapy, and chemotherapy including retinoids (with or without prednisone). For the survival analysis, the interval between the day of the biopsy report and the day of death due to CETCL or a related disease was considered. All factors with a P value ≤.1 in the univariate analyses that were considered easily identifiable features on routine biopsy evaluation were considered for inclusion in multivariable Cox regression. In all, 3 multivariable Cox regression models were explored: a clinical model based on clinical independent variables, a histopathological model based on histological independent variables, and a combined model. Purposeful backward and forward search algorithm was used to identify the subset of predictors for each of the 3 models mentioned above. For the 3 models identified, in silico validation based on a bootstrap methodology with 1000 repetitions was used to establish the independence of the model from the observed data. The proportional hazard assumption was checked by means of the test based on the Schoenfeld residuals. To assess the classification power of the model to predict which animals have the highest likelihood of dying, a nonparametric receiver operating characteristic (ROC) analysis was used. The classification power was graded as acceptable, excellent, or outstanding according to Hosmer-Lemeshow proposed scale. 7
Results
Study Cohort
Surgical biopsy archives comprised 491 cases of canine CETCL, and seeking information from the referring veterinarians resulted in 233 completed questionnaires. Inclusion criteria were met by 176 cases; 173 dogs were euthanized or died within the study period due to CETCL or related diseases. Three dogs were still alive at the end of the study. Ninety-nine dogs (56.25%) were neutered females, 56 dogs (31.82%) were castrated males, 13 dogs (7.39%) were intact males, and 8 dogs (4.55%) were intact females. The mean age at the time of diagnosis was 10.4 years (range: 2.9–17.3 years, median: 10.7 years), with 1 dog under 3 years, 1 dog between 3 and 4 years, and 7 dogs between 4 and 5 years of age. The most frequently represented breeds were Golden Retriever (n = 28) and Labrador Retriever (n = 13), while 33 dogs were mixed breed dogs. When compared to the number of dogs per breed in the biopsy database, the Gordon Setter had the highest prevalence rate (56.60 per 1,000 dogs). All breeds of the cohort and their prevalence rates are listed in Supplemental Table S1.
Clinical Findings
Clinical signs were noted on average 169.33 days (range: 9–1189 days) before diagnoses were confirmed by biopsy. Most commonly reported signs were erythema (131/176, 74.43%), crusting (108/176, 61.36%), and scaling (102/176, 57.95%). Most affected anatomic locations were haired skin (159/176, 90.34%), lip (74/176, 42.05%), nasal planum (49/176, 27.84%), and paw pads (48/176, 27.27%). Twenty-two of 177 (12.50%) dogs had regional lymph node involvement. A complete list of clinical signs and anatomic locations is presented in Table 1.
Clinical parameters of 176 dogs with cutaneous epitheliotropic T-cell lymphoma.
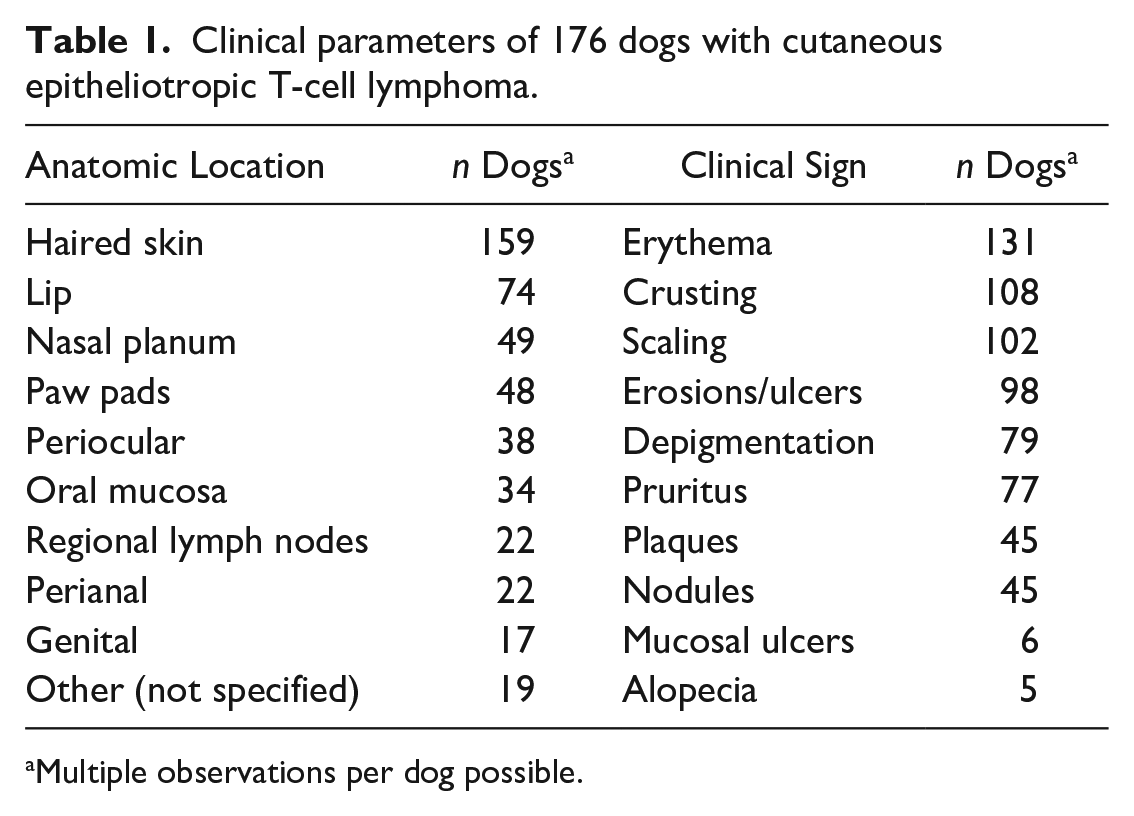
Multiple observations per dog possible.
One hundred seventeen dogs received pharmaceutical therapy upon diagnosis, while in 57 dogs, surgical excision was the only treatment. Treatment data were not available for 2 dogs. Among the dogs receiving pharmaceutical therapy, 37 dogs were treated with prednisone, 77 dogs received chemotherapy with or without prednisone, and 3 dogs did not have treatment modalities indicated. Chemotherapy treatment among dogs was variable and included single agents or combinations of the following drugs: lomustine (n = 69), L-asparaginase (n = 27), cyclophosphamide (n = 10), isoretinoin (n = 8), mechlorethamine (n = 6), vincristine (n = 5), vinblastine (n = 4), rabacfosadine (n = 4), adriamycin (n = 3), procarbazine (n = 3), chlorambucil (n = 2), doxorubicin (n = 2), masitinib (n = 2), and cytarabine (n = 1). Additional prednisone was applied in 56 dogs receiving chemotherapy. Two dogs underwent radiation therapy in addition to chemotherapy.
Clinical remission after surgery and/or medical treatment was reported as complete in 47 dogs and partial in 22 dogs, while 76 dogs experienced no remission at all (data not available for 31 dogs). In 67 cases, a phase of disease stability of at least 1 month was recorded. This phase of nonprogression lasted on average 4.8 months (range: 1–30). The survival times are listed in Table 2. Dogs treated with chemotherapy had 4.26-fold and dogs treated with prednisone had 2.82-fold longer median survival times than dogs treated with supportive care only, but survival times were highly variable in all 3 groups (Fig. 3). When including the period since the first occurrence of clinical signs, survival times were less different among groups (Table 2).
Survival times of 176 dogs with cutaneous epitheliotropic T-cell lymphoma.

Histopathological Findings
The results of the histopathological evaluations are shown in Supplemental Tables S2 and S3. A vast majority of cases had not only epithelial (Fig. 1a) but also dermal infiltration by neoplastic lymphocytes (n = 167). Infiltration of the panniculus was present in 62 of 140 cases with panniculus available in the specimen (Fig. 1b). Epitrichial apocrine glands and sebaceous glands were also infiltrated in a majority of cases; however, the structures were frequently not visible due to the severity of the neoplastic infiltrate (apocrine glands: n = 25; sebaceous glands: n = 77). Common concurrent epithelial changes were apoptotic keratinocytes (n = 58), parakeratosis (n = 90), and subepidermal clefting and/or ulceration (n = 74). Follicular mucinosis confirmed by Alcian blue staining was present in 40 cases. Mitotic counts and cellular and nuclear size assessments are presented in Table 3. In 95 cases, the mean nuclear diameter of the neoplastic lymphocytes was smaller than 1.5 RBC diameters (mean RBC diameter of all cases: 5.70 µm), compatible with small-cell lymphoma (Fig. 1c). In 79 cases, the cells were of intermediate size (nuclear diameter between 1.5 and 2 RBC diameters), and 2 cases were large cell lymphomas (nuclear diameter larger than 2 RBC diameters) (Fig. 1d).
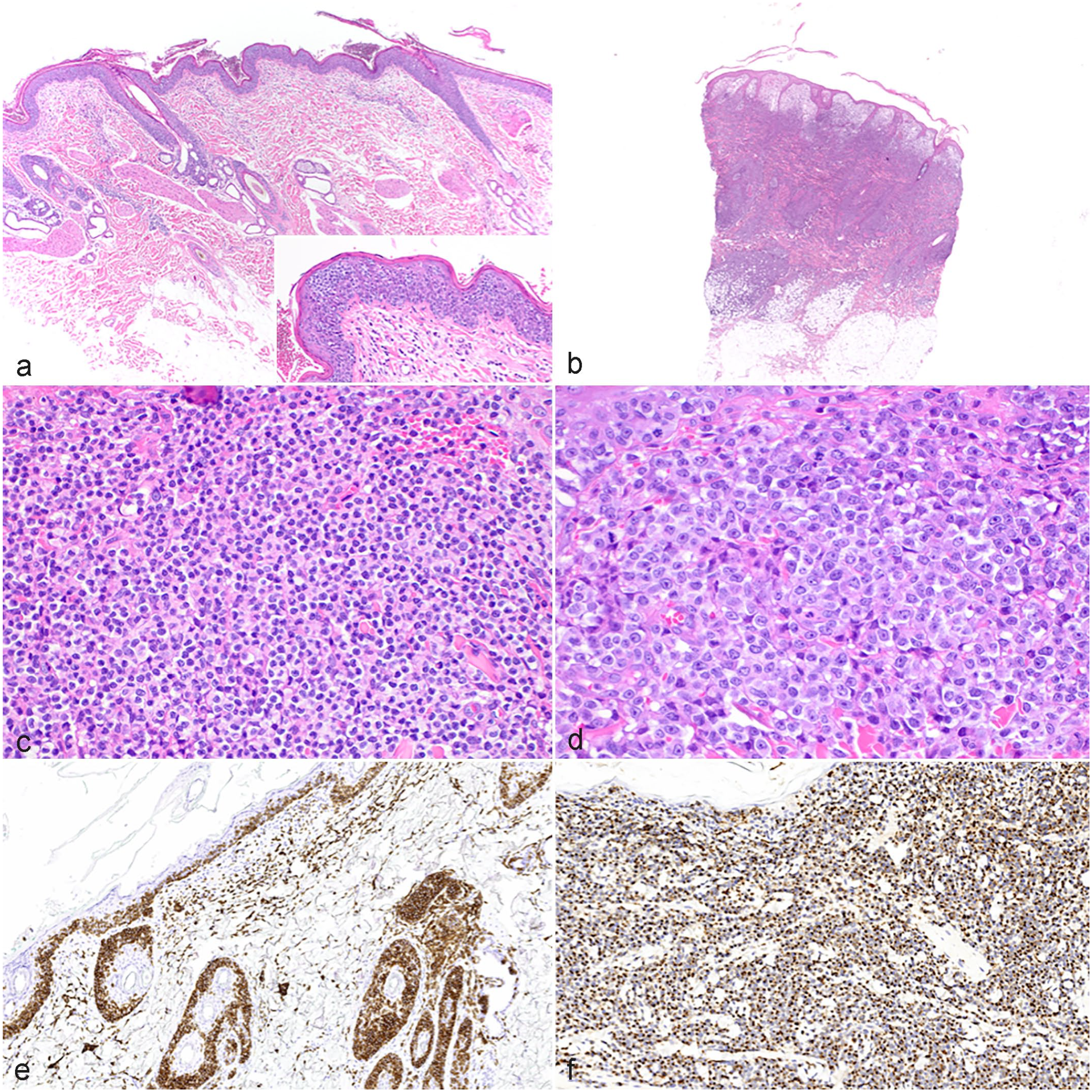
Canine cutaneous epitheliotropic T-cell lymphoma (CETCL). (a) Lumbar skin, 13-year-old mixed breed. Neoplastic lymphocytes are confined to the epidermis and adnexal epithelium (pagetoid reticulosis). The dermis and subcutis are not involved. Inset shows Pautrier’s aggregates in the epidermis. Case 84. HE, hematoxylin and eosin. (b) Haired skin, 14-year-old American Pit Bull Terrier. Marked infiltration of the dermis, adnexal units, and superficial panniculus by neoplastic lymphocytes. Case 21. HE. (c) Haired skin, 9-year-old mixed breed dog. The cells have small nuclei (mean diameter: 3.943 µm) with clumped, dark chromatin. Mitoses are not evident. Case 76. HE. (d) Haired skin, 10-year-old English Cocker Spaniel. The cells have large nuclei (mean diameter: 8.603 µm) with marginalized chromatin and a single nucleolus. Several mitotic figures are evident. Case 174. HE. (e) Haired skin, 8-year-old Golden Retriever. Neoplastic cells are CD3 positive. Case 25. Immunohistochemistry (IHC) for CD3. (f) Haired skin, 11-year-old German Shepherd Dog. Neoplastic cells are granzyme B positive. Case 70. IHC for granzyme B.
Quantifiable cellular features of 176 dogs with cutaneous epitheliotropic T-cell lymphoma and their association with the risk of instantaneous death.
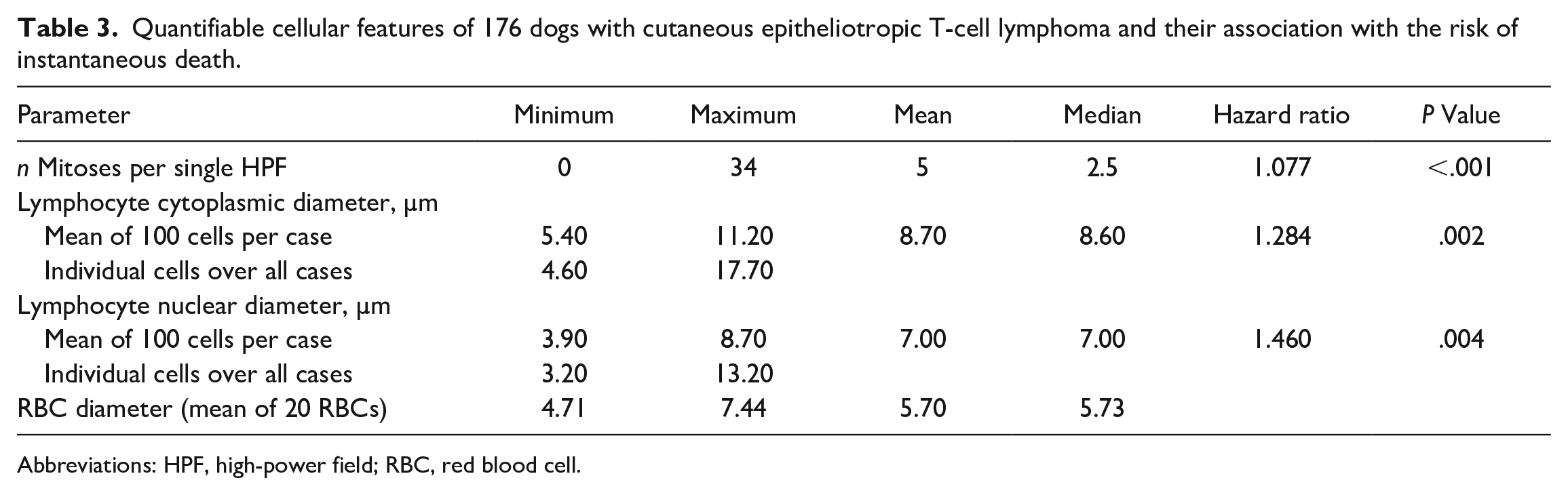
Abbreviations: HPF, high-power field; RBC, red blood cell.
Most of the cases (170/176) showed a moderate to strong, diffuse immunohistochemical signal for CD3 (Fig. 1e). In 3 cases, the signal was weak in the majority of cells or had a patchy distribution among neoplastic cells, whereas 3 cases were completely negative for CD3; all 6 cases displayed the typical epidermotropism and a uniform expression of granzyme B (Fig. 1f), compatible with CETCL.
Statistical Results
The PCA did not yield any clustering of cases in terms of subtypes. The results of the univariable Cox regression analysis for all parameters assessed are listed in Table 4. A mitotic count of ≥7 per single 0.237 mm2 HPF (hazard ratio [HR]: 2.891, P < .001), a mean cytoplasmic diameter of ≥10 µm (HR: 1.994, P = .005), and a mean nuclear diameter of ≥8.3 µm (HR: 3.701, P = .002) were identified as cutoff values to indicate a higher risk of instantaneous death (Fig. 2).
Clinical and histopathological parameters significantly associated with a risk of instantaneous death in univariable Cox regression analysis.
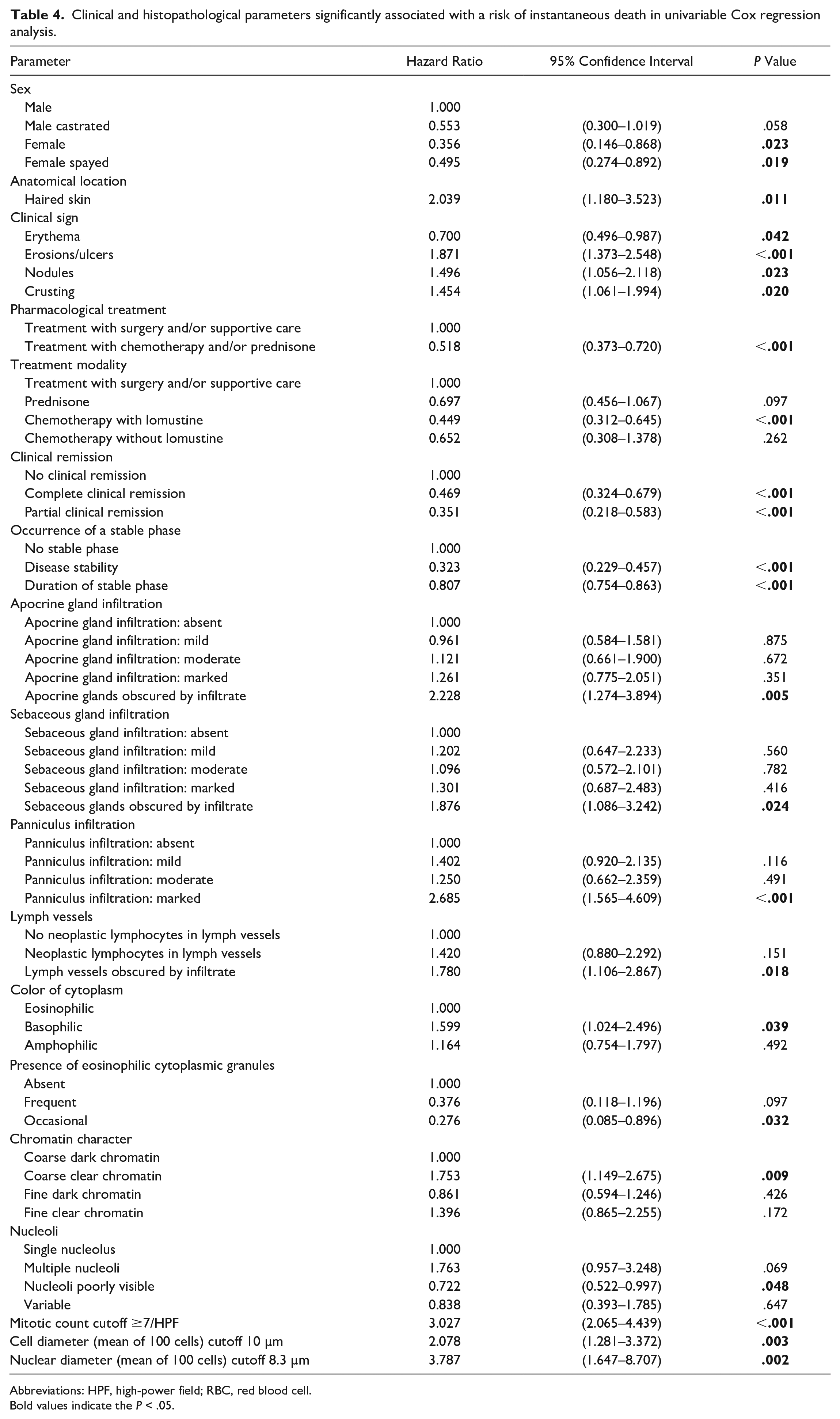
Abbreviations: HPF, high-power field; RBC, red blood cell.Bold values indicate the P < .05.
Based on the origin of the independent variables, histopathological, clinical, and combined multivariable models were constructed (Table 5). Using the methodology outlined earlier, the histopathological model included the nuclear diameter and mitotic count cutoff as categorical variables. While the model showed good robustness independent from the data and that the proportional-hazard assumption holds (P = .605; Supplemental Table S4), it showed poor predictive power (ROC = 0.65, 95% CI: 0.29–1.00). The clinical model included erosions/ulcerations and nodules as categorical variables. It showed good robustness independent from the data and a valid proportional-hazard assumption (P = .121; Supplemental Table S5). The model had excellent predictive power (ROC = 0.83, 95% CI: 0.79–0.88). Finally, the combined model including the variables erosions/ulcerations, nuclear diameter, and mitotic count cutoff had excellent robustness, and the proportional-hazard assumption held (P = .541; Supplemental Table S6). The combined model also had an excellent predictive power (ROC = 0.83, 95% CI: 0.64–1.00).
Multivariable Cox regression models calculating the risk of instantaneous death for clinical and histopathological parameters.
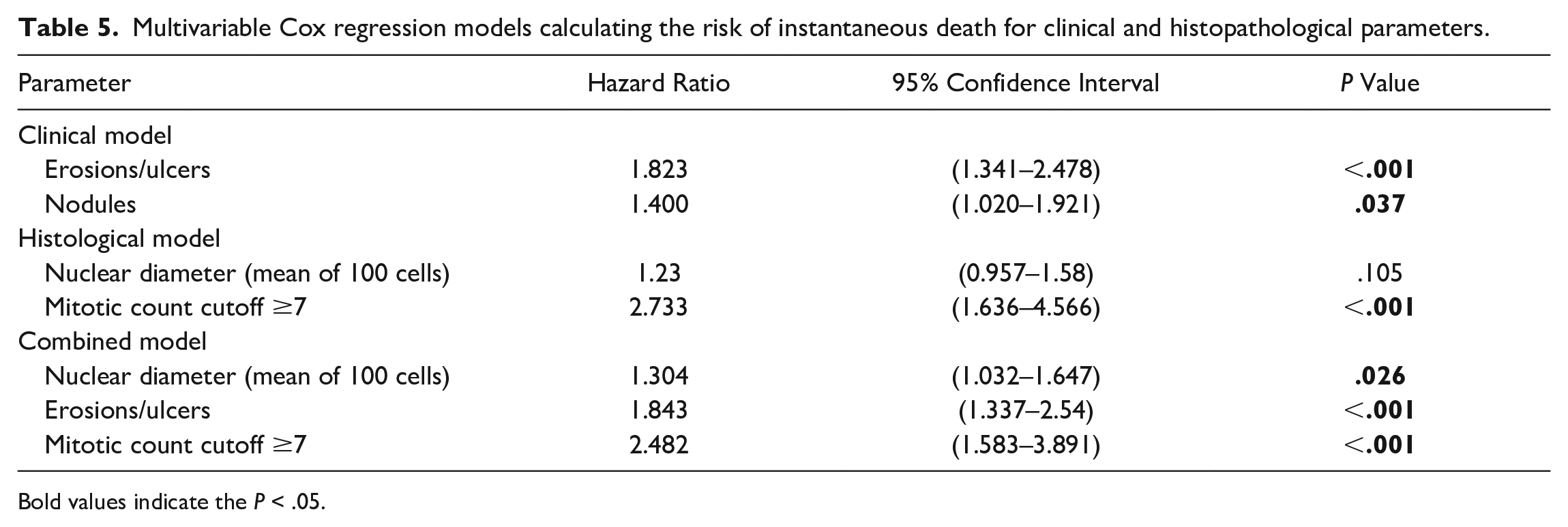
Bold values indicate the P < .05.
Discussion
This comprehensive investigation of canine CETCL reveals histopathological and clinical features that impact survival time. The features enable a pathologist to directly provide data to aid in case management and outcome prediction. Prior studies on canine CETCL have focused on case description and clinical outcomes, but not on features seen with light microscopy via evaluation of routine punch biopsies.3,4,11,17
Histologic features that predict clinical outcome include deep tissue infiltration, cell/nuclear size, character of chromatin, and mitotic activity. Extensive infiltration of the pannicular adipose tissue by neoplastic cells is associated with an increased risk of instantaneous death and likely represents a more advanced disease seen clinically as nodules or plaques. The presence of clear nuclei with clumped chromatin and a basophilic color of the cytoplasm is also an increased risk factor of instantaneous death. However, chromatin character and the color of cytoplasm may be strongly associated with the degree of formalin fixation and staining protocols, respectively. Thus, these features must be interpreted with caution due to the possibility of inadequately fixed samples and staining variations in different histopathology laboratories, respectively. Additionally, this study provides justification for including the cell size and mitotic count in histologic interpretations of CETCL. An increased cell size (mean cell diameter ≥ 10.0 µm, mean nuclear diameter ≥ 8.3 µm) and a mitotic count of ≥7 per single 0.237 mm2 HPF clearly divide cases with poor outcome from those with significantly longer survival times (Fig. 2). As an increasing number of diagnostic laboratories work with digitized specimens and WSI viewing platforms enabling distance measurements and counts, the calculation of these cutoff values may be more readily obtained.
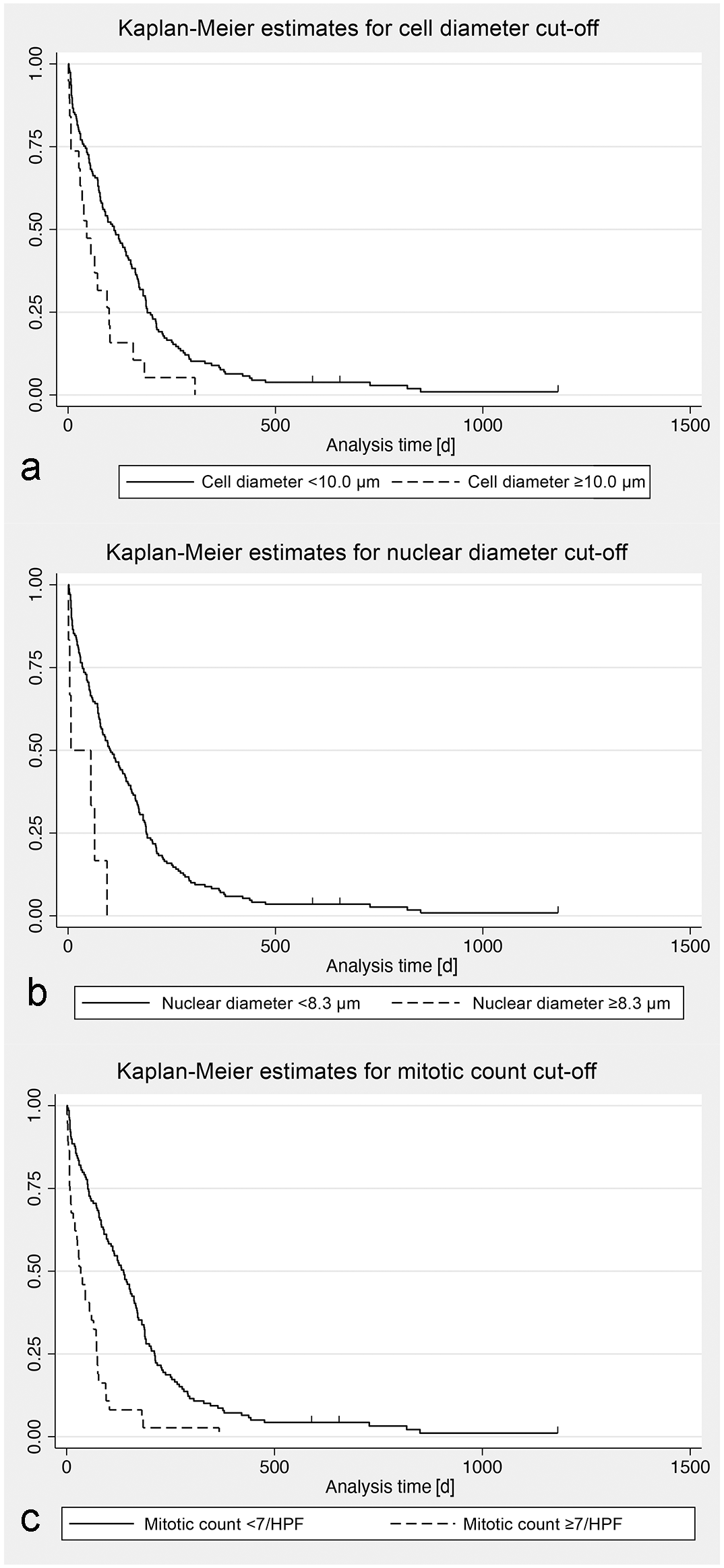
Kaplan-Meier survival curves for mortality in 173 dogs with cutaneous epitheliotropic T-cell lymphoma (CETCL), stratified according to specified histopathological parameter cutoffs. (a) Kaplan-Meier estimates for a cell diameter cutoff of 10.0 µm. Censored individuals are indicated with a vertical dash. [d] = days. (b) Kaplan-Meier estimates for a nuclear diameter cutoff of 8.3 µm. Censored individuals are indicated with a vertical dash. [d] = days. (c) Kaplan-Meier estimates for a mitotic count cutoff of 7/HPF (2.37 mm2). Censored individuals are indicated with a vertical dash. [d] = days.
Among clinical parameters, the involvement of haired skin irrespective of the involvement of other anatomic locations was an indicator of a poor outcome, which has been described previously. 3 However, in contrast to the previous study, which compared the outcome of dogs with cutaneous versus mucocutaneous/oral CETCL, dogs with lesions restricted to the oral cavity were not included in the present study. Clinical remission and a stable disease phase had a significant positive effect on outcomes. Correlating with the results of the survival analysis (Fig. 3), dogs receiving chemotherapy with lomustine had a significantly decreased risk of instantaneous death compared to dogs receiving only surgery and supportive care. The risk of instantaneous death was also lower with other chemotherapy protocols and prednisone, but differences were not significant. Chan et al found dogs treated with chemotherapy survived significantly longer than dogs not having received chemotherapy. 3 However, the authors of that study did not further stratify the different chemotherapy protocols and the nonchemotherapy group into dogs that did and did not receive prednisone. The data of the present study suggest that prednisone treatment alone also prolongs survival but is less effective than chemotherapy with lomustine.
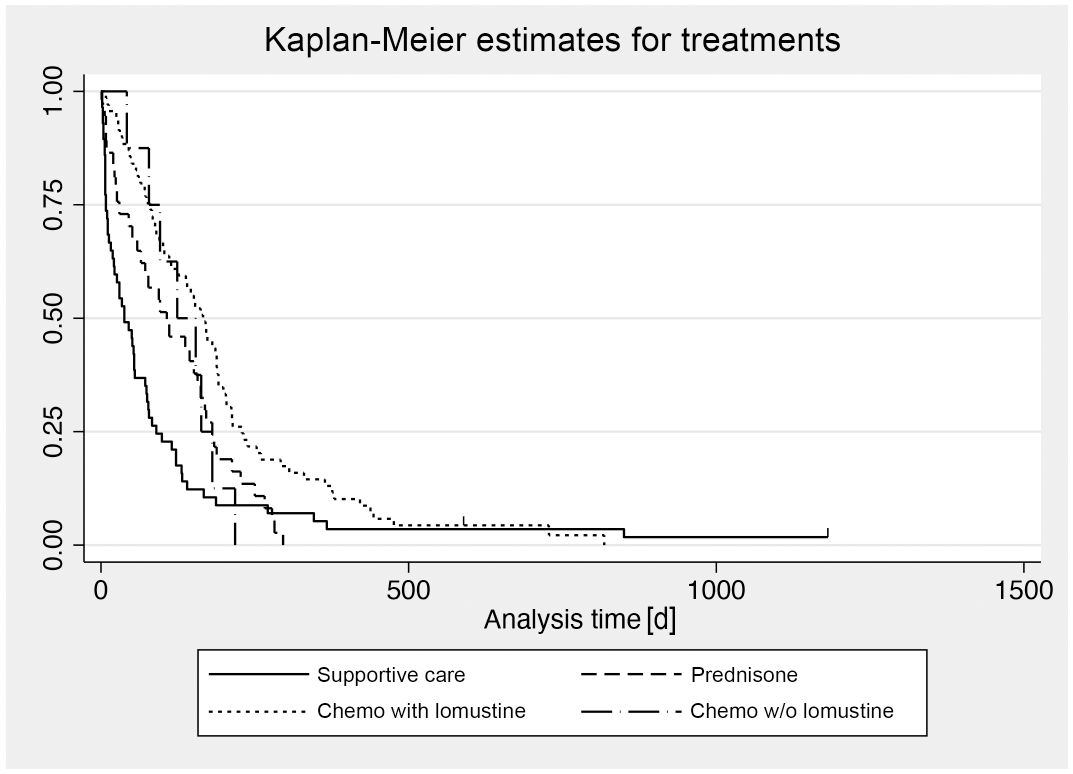
Kaplan-Meier survival curves for mortality in 176 dogs with cutaneous epitheliotropic T-cell lymphoma (CETCL), stratified according to treatment modality. Censored individuals are indicated with a vertical dash.
In concordance with previous reports, the study cohort presented a variety of breeds, clinical signs, and survival times, as well as a predilection for older dogs.3,4,17 Similar to the study of Chan et al, female dogs were overrepresented with 60.8% of the cases. 3 In humans, a male-to-female incidence ratio of 1.57 has been reported for MF/Sézary syndrome cases in the United States. 8 According to the results of Chan et al, mixed breed dogs and Golden Retrievers were most commonly represented. 3 However, the prevalence rates in our study, calculated on the basis of the biopsy database for both Golden Retriever and mixed breed dogs, were only slightly above the mean prevalence rate of the cohort (3.27 per 1,000 dogs) (Supplemental Table S1). Breeds with the highest prevalence rates in this study were Gordon Setter, Shar Pei, and Havanese. Also histopathological findings were largely compatible with previous studies.4,11 In contrast to older literature, lesions in this study were not classified as PR and MF, due to the uncertain impact on prognosis in canine CETCL. 10 Moreover, in study cases with more than 1 biopsy available, both histological forms were present in some cases, corroborating the theory that PR is an earlier form of canine CETCL. 10 According to the human histological subtype classification, 18 cases resembled folliculotropic MF, as reported previously for canine CETCL.2,4 As in the human entity, adnexal neoplastic infiltration was frequently associated with mucin deposition and mucinous degeneration. 2 While this subtype has a slightly reduced survival time in human medicine, a significant difference in survival was not found for this histopathological subtype in the present study. Owing to the study design, it was not possible to identify cases with Sézary syndrome because hematology data were not requested in the questionnaire. According to the literature, this is a very rare entity in dogs, with only a handful of published cases.5,13
To the authors’ knowledge, this is the first digital morphometric analysis performed with canine lymphoma histopathology slides. Compared with the descriptions of Fontaine et al and the commonly accepted WHO classification for canine lymphoma, the absolute individual cell and nuclear diameters found in the present study were considerably smaller (Table 3).4,16 With respect to the mean RBC diameter per case, most cases fell into the small-cell and intermediate-cell lymphoma category (Supplemental Table S3). This result was unexpected because based on the initial visual evaluation, more cases were thought to be in the large-cell category, emphasizing the importance of accurate measurement. It remains unknown if a similar result would also occur when performing the same analysis with canine nodal lymphoma WSIs. The mitotic count per single 0.237 mm2 HPF was generally low, albeit highly variable (Table 3). According to the WHO classification system for canine nodal lymphoma, 73% of our cases were compatible with a low-grade lymphoma (mitotic count ≤5; data not shown). 16 Low mitotic counts and proliferative activity have been reported for CETCL previously but do not obviously reflect the grade of biological behavior seen in other lymphoma types.4,6,16
In addition to the identification of prognostic parameters, the authors expected to be able to group histological subtypes based on their morphology and outcome. With the used approach of a PCA including all assessed parameters, no case clustering providing evidence of canine CETCL subtypes, as described in the human WHO-EORTC classification, was found.2,18 However, the human classification is not only based on clinical and histomorphological criteria but also largely relies on the expression pattern of a plethora of immunohistochemical markers, as well as on specific genetic alterations. An immunohistochemical analysis was beyond the scope of this study. Additionally, a large number of human IHC markers do not work on canine formalin fixed paraffin embedded tissues or protocols for them have not been established yet. Recent advances in canine lymphoma research have made available immunohistochemical markers for CD5, CD30, CD56, Bcl-2, Bcl-6, and others, which could be used to further characterize the neoplastic cell types as in human medicine.12,14 Such additional work would probably enable a more detailed classification of canine CETCL.
In conclusion, this study identified several novel, independent clinical and histopathological prognostic factors for canine CETCL. As clinical parameters, haired skin involvement, erosions/ulcers, nodules and crusting, and the histopathological parameters of extensive infiltration of panniculus, a mitotic count ≥7 per single 0.237 mm2 HPF, a mean cell diameter ≥10.0 µm, and a mean nuclear diameter ≥8.3 µm were independent predictors of an increased risk of instantaneous death. Complete posttherapeutic clinical remission and stable disease were associated with a decreased risk of death. In particular, infiltration of the panniculus and mitotic count can be easily assessed in routine diagnostic procedures and are thus very helpful prognostic histologic markers. As expected, a wide variety of clinical presentation, outcomes, and histomorphological features were recorded. Further characterization of potential canine CETCL subtypes will require additional analyses, such as IHC or mutational profiling.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221140818 – Supplemental material for Prognostic clinical and histopathological features of canine cutaneous epitheliotropic T-cell lymphoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858221140818 for Prognostic clinical and histopathological features of canine cutaneous epitheliotropic T-cell lymphoma by Martina Dettwiler, Elizabeth A. Mauldin, Sara Jastrebski, Deborah Gillette, Darko Stefanovski and Amy C. Durham in Veterinary Pathology
Footnotes
Acknowledgements
We are thankful to the referring veterinarians in the United States and Europe for the provision of clinical data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded with institutional resources of the Department of Pathobiology, School of Veterinary Medicine, University of Pennsylvania, and the Institute of Animal Pathology, Vetsuisse Faculty, University of Bern. Immunohistochemical stains were provided by the Penn Vet Comparative Pathology Core, a shared resource supported by the Abramson Cancer Center Support Grant (P30 CA016520). The Aperio Versa 200 scanner used for whole slide imaging and the image analysis software was supported by a NIH Shared Instrumentation Grant (S10 OD023465-01A1).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
