Abstract
A 14-year-old female spayed Dachshund was presented with generalized scaling, erythema, pruritus, poor quality of hair coat, and progressive weight loss. Cutaneous epitheliotropic T-cell lymphoma (CETCL) was suspected. Skin biopsies were suggestive of CETCL. However, immunohistochemistry revealed the presence of numerous CD20+ and CD3+ cells. Clonality assay demonstrated a clonal T-cell receptor gamma rearrangement and a polyclonal IgH gene rearrangement. Double-label immunofluorescence confirmed coexpression of CD3 and CD20 by neoplastic cells. By double immunohistochemistry, neoplastic cells were CD3+ and PAX5–. The results are compatible with a CD3+, CD20+ CETCL. Coexpression of CD20 and CD3 has been recognized in peripheral T-cell lymphomas. Although documented in human CETCL, it has not been reported in canine CETCL. The pathogenetic basis of CD20 expression in mycosis fungoides is explored.
Keywords
Cutaneous epitheliotropic T-cell lymphoma (CETCL) presents in different forms, including classic mycosis fungoides (MF; patch, plaque, and tumor form), pagetoid reticulosis, and Sézary syndrome. 2 Typically, neoplastic T cells are seen within the dermis as well as in epithelial structures, including epidermis and adnexa, and can extend into the subcutis. 2 Occasionally, the neoplastic infiltrate is limited to the epithelia (pagetoid reticulosis). The disease occurs uncommonly in dogs and is rare in cats. It follows a progressive course and usually affects older animals. Clinical features range from exfoliative erythroderma to patch, plaques, or nodules (tumor stage) with final dissemination to peripheral lymph nodes, ulcerative disease of the oral mucosa, and a mucocutaneous form. Patches and plaques often coexist, and progression to tumor stage is generally faster in dogs than in humans. According to the clinical picture, differential diagnoses range from allergic diseases to immune-mediated/autoimmune conditions, and a careful histopathologic examination is often fundamental for a definitive diagnosis.
Canine CETCL is composed of CD3+, CD8+ T lymphocytes. Classical MF is either composed of TCRαβ+ T cells or TCRγδ+ T cells, whereas pagetoid reticulosis is generally TCRγδ+ T cells. 9 This is in contrast to CETCL in humans, which is mostly composed of CD4+ TCRαβ+ T cells. 6 Loss of T-cell surface antigens is a known aberration associated with disease progression in humans and dogs. Coexpression of CD20+ was recently documented in MF and peripheral T-cell lymphomas with skin involvement in humans, and the potential pathogenetic or prognostic significance of this marker in disease progression has been investigated. 7 To our knowledge, this represents the first report of CD3+, CD20+ CETCL in dogs.
A 14-year-old female spayed Dachshund was presented with generalized scaling and erythema accompanied by pruritus. The dog had been suffering from recurrent otitis and pruritic dermatitis for >6 years. In the last 8 months, she was losing weight and had a poor hair coat quality. At referral, a generalized exfoliative erythroderma was noted (Fig. 1), which was incompletely blanching at diascopic examination. Footpads were hyperkeratotic, and there was focal pad ulceration. An eroded plaque on the ventral neck and a generalized lymphadenomegaly were noted. Differential diagnoses included CETCL, erythema multiforme, paraneoplastic syndrome, demodicosis, and dermatophytosis. Skin scrapings and microscopic examination of plucked hairs were negative. Hairs were sampled for fungal culture, and while waiting for results, the dog was bathed with 2% chlorhexidine shampoo twice a week. Microsporum canis was identified; however, a second culture, performed after 1 month, was negative.
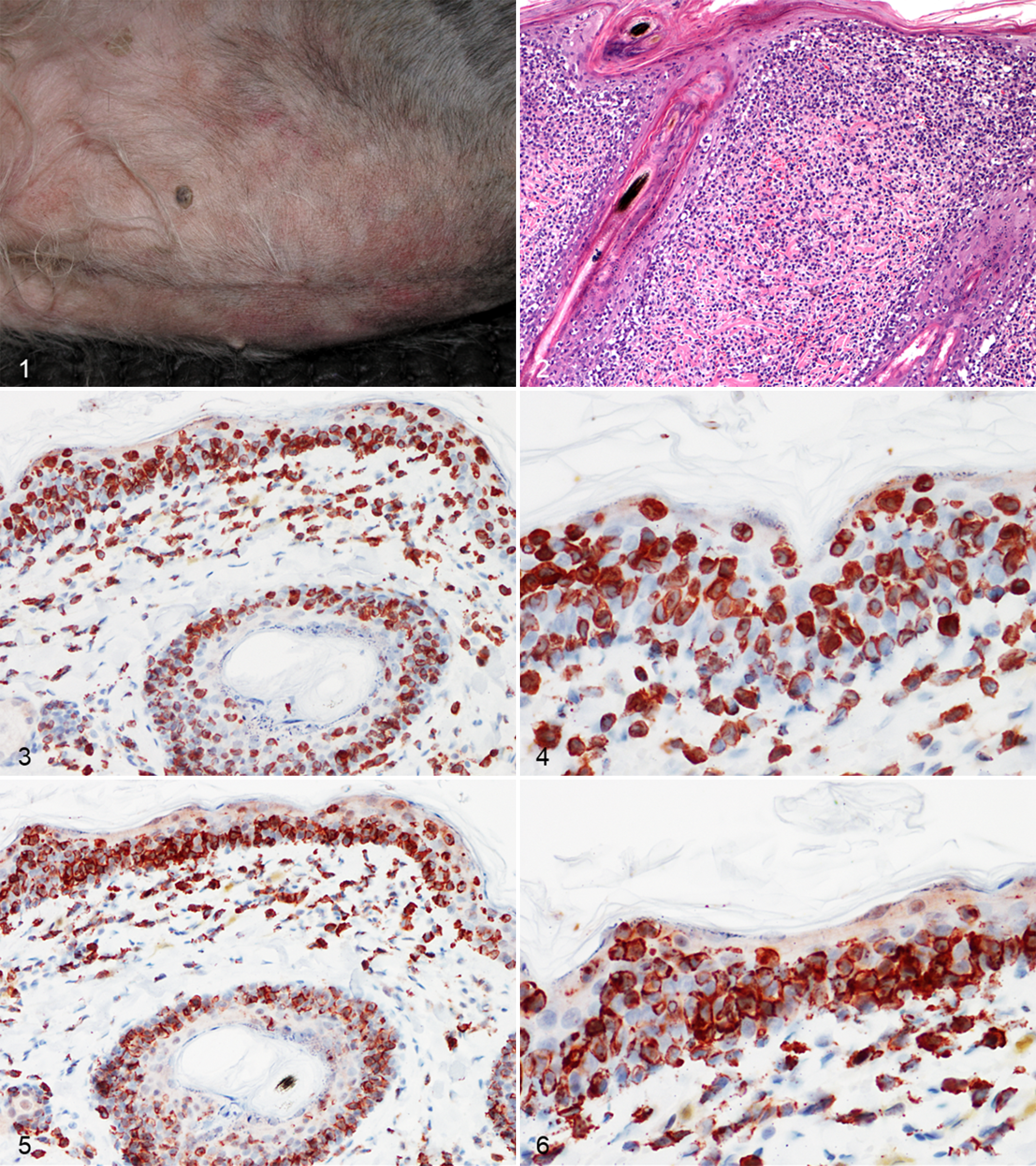
Cutaneous epitheliotropic T-cell lymphoma, skin, Dachshund.
Punch biopsies taken at the first consultation were characterized by a monomorphous round cell infiltration in the superficial dermis, multifocally obscuring the dermoepidermal junction of the epidermis and hair follicles (Fig. 2). Infiltrating cells had sharply demarcated cell borders, scant to moderate amount of pale eosinophilic cytoplasm, and central round to indented nuclei with a diameter of about 2 red blood cells. Admixed with these cells there were fewer smaller lymphocytes. Small intraepithelial aggregates of round cells (Pautrier microabscesses) as well as scattered single-keratinocyte necrosis/apoptosis and rare subepidermal vesicles were noted. In 1 biopsy, round cells extended to the mid dermis, forming a bandlike infiltrate, where numerous eosinophils and fewer mast cells and neutrophils were admixed. Periodic acid–Schiff stain on serial sections ruled out the presence of dermatophytes. Histopathologic findings were consistent with a diagnosis of CETCL.
Immunohistochemistry was performed as previously described, 1 following a modified protocol. Briefly, a commercially available rabbit antibody against CD20 (1:600, RB-9013; Thermo Scientific, Fremont, CA, USA) and a rabbit polyclonal antibody against CD3 (1:300; A 0452; DakoCytomation, Glostrup, Denmark) were used. Antigen retrieval was achieved in a preheated Tris-EDTA buffer solution, pH 9.0, for the CD3 antibody. No antigen retrieval was performed for the CD20 antibody. Negative controls were treated, omitting the primary antibody and incubating tissue sections with Tris-buffered saline buffer. Normal canine lymph node was used as positive control. Intraepithelial and dermal round cells consistently and intensely expressed CD3 (Figs. 3, 4). However, numerous CD20+ cells were also seen, sometimes equating the number and the distribution pattern of CD3+ T cells (Figs. 5, 6). The dog was treated with prednisolone (1 mg/kg orally, once daily), without satisfactory response. A polymerase chain reaction for antigen receptor rearrangement was performed, as previously described. 8,13 Clonality testing revealed clonal T-cell receptor γ locus (TRG) rearrangement for both primer sets. A polyclonal and pseudoclonal rearrangement for immunoglobulin heavy chain (IgH) gene and kappa deleting element (KDE) was also found, respectively. Duplicate samples were tested to validate the results. A double immunofluorescence sequential protocol was used to investigate the colocalization of CD3 and CD20 on neoplastic cells, following a previously reported modified protocol. 11 Briefly, CD3 (1:100) was applied overnight at 4°C. Remaining steps and washes between steps were performed at room temperature. Biotinylated goat anti-rabbit antibody (1:200; Vector Laboratories, Burlingame, CA, USA) was applied for 30 minutes, and slides were then incubated with fluorescein-conjugated avidin (1:100; Vector Laboratories) for 10 minutes. For avidin/biotin blocking step, slides were incubated for 15 minutes with avidin and then with biotin (Avidin/Biotin Blocking Kit, code SP-2001; Vector Laboratories). CD20 (1:200) was applied for 1 hour, followed by a second step with the secondary antibody. The slides were then incubated with Texas Red–conjugated avidin (1:100; Vector Laboratories) for 10 minutes. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (Vector Laboratories). CD3-CD20 colocalization was evident in the majority of neoplastic cells infiltrating both the epidermis and the dermis (Fig. 7). Double immunohistochemistry with another B-cell marker was performed, with the CD3 antibody and an anti-human monoclonal mouse PAX-5 antibody (1:20, clone 24/Pax-5; BD Bioscience, Franklin Lakes, NJ, USA). The first primary antibody (PAX5) was applied to the tissue section, followed by a streptavidin–biotin–horseradish peroxidase–labeled detection system with a DAB chromogen, whereas the second primary antibody (CD3) was applied after the excess DAB was rinsed off, followed by labeling with a streptavidin–biotin–alkaline phosphatase detection system and a fast red chromogen (EnVision Doublestain System, code K1395; DakoCytomation). Neoplastic cells did not express PAX5 (data not shown). The final diagnosis was CD3+, CD20+ CETCL. The dog was treated with the same daily dosage of prednisolone for approximately 1 month; then, the frequency was reduced to every 48 hours due to polyuria, polydipsia, and polyphagia. Due to deterioration of its general condition, the dog was euthanized after 6 months. The owner declined necropsy.
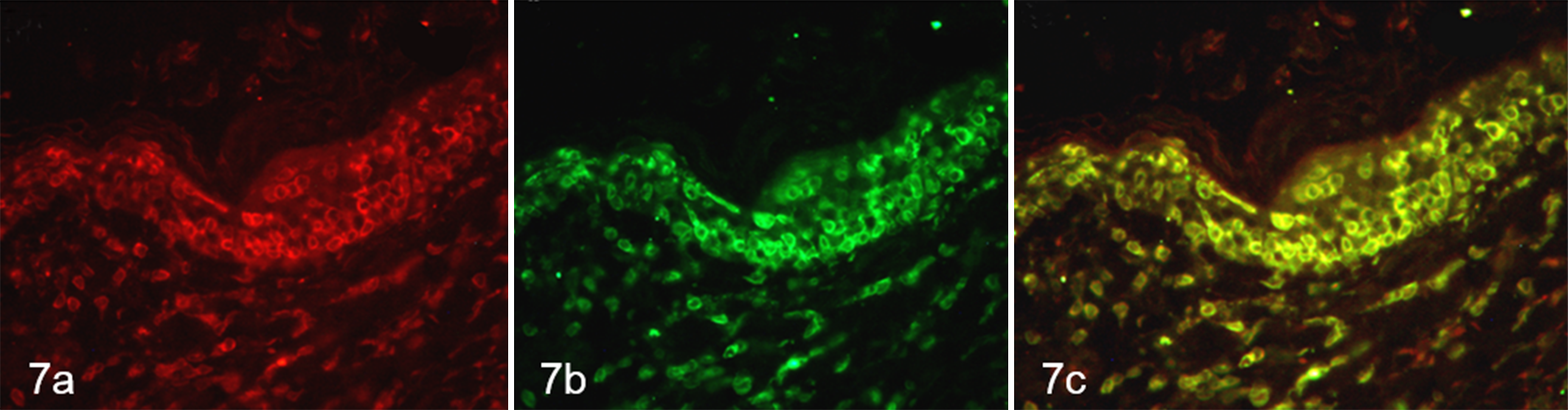
Cutaneous epitheliotropic T-cell lymphoma, skin, Dachshund. Labeling of cells with anti-CD20 (red; a) and anti-CD3 (green; b) antibodies shows colocalization of staining (merge; c) in neoplastic lymphocytes in epidermis and dermis. Double sequential immunofluorescence.
Canine CETCL can be limited to epidermis and adnexa (pagetoid reticulosis) or include dermal infiltrates forming patches, plaques, and tumors (MF). The latter is the most common form. Canine CETCL is characterized by a proliferation of CD3+, CD8+ T cells. 9 This is in contrast to CETCL in humans, which is mostly of CD4 T-cell origin. 6
In view of the heterogenic infiltrate of CD3+, CD20+ cells within the current case, a dermatophytosis, a lymphoid drug reaction, and an unusual T cell–rich B-cell lymphoma were considered as differential diagnoses. Presence of prominent epitheliotropic aggregates of CD20+ is not a primary feature of either lymphoid drug reaction or T cell–rich B-cell lymphoma. The periodic acid–Schiff staining of biopsies and the second skin culture were negative, making a dermatophytosis unlikely. The positive result of the first fungal culture may therefore reflect a contamination or growth of dermatophytes passively carried on the hair coat. Alternatively, an initial dermatophytosis was successfully treated with topical therapy.
Clonality testing supported the presence of a CD3+, CD20+ T-cell lymphoma, as duplicate samples confirmed a clonal TCRγ rearrangement of the infiltrating lymphocytes. Expression of CD20 is most unusual by neoplastic T cells. As a transmembrane phosphoprotein, CD20 is thought to regulate cellular calcium transport and cell activation of B cells. It is lost during differentiation to plasma cells. Although considered a marker for B cells, CD20 in humans is expressed on a small subset of T cells in peripheral blood and bone marrow of healthy individuals. 4 These CD20+ cells are more likely to be γδ–T cell antigen receptor positive and CD8+ as compared with CD20– T cells. 5 Recent reports described CD20 expression in rare cases of T-cell lymphomas and less commonly in cutaneous T-cell lymphomas in humans. 12
Cutaneous epitheliotropic CD3+, CD20+ T-cell lymphomas could derive from the CD20+ subset of normal peripheral T cells. Canine CETCL can be of CD8 αβ–T cell or CD8 γδ–T cell origin, 9 and the current case may be of CD8 γδ–T cell origin with coexpression of CD20. Unfortunately, the lack of snap-frozen tissue precluded the evaluation of CD4 versus CD8 and αβ-TCR versus γδ-TCR in our case.
Double immunohistochemical method for the detection of a coexpression of CD3 and PAX5 by neoplastic cells was negative. The lack of expression of other B-cell markers (CD79, CD19, PAX5) has already been reported in cases of CD20+ MF in humans. 3,4
Due to the low number of reported cases in human medicine, the prognostic significance of CD20 expression in CETCL is unclear. CD20 could be regarded as an activation marker; its expression has been observed in human patients with acute lymphoblastic B-cell leukemia following induction treatment with glucocorticoids. In some cases, CD20+ T-cell lymphomas were characterized by an aggressive course, 10 whereas in other cases CD20 expression appeared over time and correlated with disease progression. 4 In our case, the dog was euthanized 6 months after the initial diagnosis, and no conclusion can be drawn from this single report.
In summary, we present a case of CD3+, CD20+ epitheliotropic lymphoma, which is considered a CETCL based on clonal TCRγ rearrangement. More cases with careful follow-up need to be identified to allow any possible prognostic value of CD20 expression in CETCL. Importantly, conflicting results of immunohistochemistry, such as coexpression of CD3 and CD20, need to be followed up by additional tests (eg, clonality testing) to achieve the final diagnosis of CETCL.
Footnotes
Acknowledgements
We thank Luca Stefanelli and Dr Emilia Del Rossi for their technical assistance with histologic and immunohistochemical processing. We are also grateful to Kristy Harmon for her help in running the clonality test.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
