Abstract
Keratinic primary localized cutaneous amyloidosis is a disease in humans; however, no similar condition has been reported in animals. This study aimed to investigate cutaneous keratinic amyloid deposition in dogs and elucidate its etiology. Canine hair follicle tumor tissues were histopathologically analyzed. Immunohistochemistry and mass spectrometry–based proteomic analyses were performed to identify precursor protein candidates. Structural prediction and in vitro fibrillization analyses were conducted to determine the amyloidogenic region and gene sequencing analysis was performed to assess mutations. Of the 266 samples, 16 had amyloid deposition. Amyloid deposits were found in the stroma of tumors and in the margins of keratin debris and around normal hair follicles. Cytokeratin 5 (CK5) was identified as a precursor protein candidate. C-terminal truncation of CK5 was observed in amyloid deposits, and the truncation sites varied depending on the deposition pattern. There was a significantly higher incidence of amyloid deposition in Shiba dogs, and CK5 amino acid polymorphisms were identified in these dogs. A part of the C-terminal region of both canine and human CK5 exhibited highly amyloidogenic properties in vitro. This study revealed the existence of cutaneous keratinic amyloid deposition in animals and identified CK5 as an amyloid precursor protein, providing novel insights into understanding the etiology of cutaneous amyloidosis.
Primary localized cutaneous amyloidosis (PLCA) is a disease group in which amyloid deposits are limited to the skin without systemic involvement. 22 PLCA is further divided into keratinic PLCA (kPLCA) and nodular PLCA based on pathological features. 22 Lichen amyloidosis and macular amyloidosis are kPLCAs commonly detected in Asians and South Americans.21,24 In this disease, amyloid deposits accumulate under the basal layer of the epidermis, resulting in the deterioration of the patient’s quality of life owing to severe pruritus, pigmentation, and lichenification.9,21,24 The kPLCAs occur independently or secondary to underlying diseases, such as basal cell tumors, nevus, seborrheic keratosis, and actinic keratosis. 22 In basal cell carcinomas, which is one of the most common cancers in humans, 3 amyloid deposits are observed at an incidence ranging from 11% to 75%. 7 Cytokeratins (CKs) are suspected as possible amyloid precursor proteins,1,13 and especially CK5 has been detected in amyloid deposits of the skin.4,24
In animals, cutaneous amyloidosis is reported as a part of systemic amyloid A amyloidosis 25 or plasmacytoma-associated localized immunoglobulin light-chain amyloidosis, which corresponds to human nodular PLCA. 28 However, pathological conditions corresponding to human kPLCA have not been reported in animals. In this study, we searched for amyloid deposition in canine hair follicle (HF) tumors. Furthermore, we attempted to identify the main amyloidogenic protein within the amyloid deposits in canine HF tumors and elucidate the mechanism of amyloidogenesis using proteomic and genetic analyses.
Materials and Methods
Case Information
Formalin-fixed paraffin-embedded (FFPE) tissues from 266 HF tumors were used for subsequent analyses. There were 69 trichoblastomas, 36 pilomatricomas, 101 trichoepitheliomas, and 60 infundibular keratinizing acanthomas. Details are presented in Supplemental Table S1. All samples were biopsied from pet dogs from 2016 to 2019, submitted to the Laboratory of Veterinary Pathology at Gifu University for pathological diagnosis, and histopathologically diagnosed by at least 2 veterinary pathologists.
Histological and Immunohistochemical Analyses
To histologically examine amyloid deposition, FFPE tissues were sectioned at 3 µm, stained with Congo red dye, and examined using a polarizing microscope (BX43; Olympus, Tokyo, Japan). Amyloid deposits were confirmed as yellow to green birefringence under polarized light. Amyloid-positive cases were further analyzed using immunohistochemistry (IHC). Primary antibodies and process information are listed in Supplemental Table S2. Followed by antigen retrieval, sections were treated with 3% hydrogen peroxide in methanol, blocked with 1% bovine serum albumin in phosphate-buffered saline, and then incubated with the primary antibodies. Horseradish peroxidase–labeled polymer anti-rabbit or anti-mouse antibodies (Envision+; Dako, Carpinteria, California) were used as secondary antibodies. 3,3′-diaminobenzidine-4HCl and hematoxylin were used for color development and counterstaining, respectively. For negative controls, the primary antibody was omitted.
Laser Microdissection-Liquid Chromatography-Tandem Mass Spectrometry
Protein extraction using laser microdissection was performed on FFPE specimens from cases 1, 11, and 15. Each FFPE specimen was sectioned at 5 µm, applied on polyethylene naphthalate membrane glass slide (Leica, Wetzlar, Germany), and stained with Congo red. Congo red–positive amyloid areas (500,000 µm2) were collected by laser microdissection (PALM MicroBeam; Carl Zeiss AG, Jane, Germany). Tryptic digestion of samples followed by liquid chromatography-tandem mass spectrometry (LC/MS/MS) was performed using QExactive HF (Thermo Fisher Scientific, Tokyo, Japan) as described previously. 14 The resulting MS/MS data were correlated to the theoretical fragmentation patterns of tryptic peptide sequences from the NCBInr database using Mascot Server (Matrix Science Inc., Boston, Massachusetts). Trypsin was used as the in silico proteolytic enzyme and semi-trypsin was used to identify additional nontryptic peptides. Statistically significant proteins/peptides (P < .05) were extracted using Mascot’s probability-based scoring algorithm.
Prediction of Structure
To predict amyloidogenic properties and disorderness, the amino acid sequence of canine CK5 (NCBI accession, NP_001332964.1) and human CK5 (UniProt accession, P13647) were analyzed using PASTA 2.0. 27 To investigate the effect of amino acid substitutions on amyloidogenic potential, the sequences of canine CK5 with Met499 replaced by valine and human CK5 with Val498 replaced by methionine were also subjected to analysis.
In Vitro Fibrillization of Synthetic Partial Peptides
Synthetic partial peptides (>98.0% purity) of canine CK5 (NCBI accession, NP_001332964.1; 490–504 aa: VNISVITNSMSSAY) and human CK5 (UniProt accession, P13647; 489–503 aa: VNISVVTSSVSSGY) were obtained from GL Biochem (Shanghai, China). Each synthetic peptide was suspended at 100 µM in buffer (pH 6.8; 50 mM citrate, 0.1 M NaCl, 5% DMSO [dimethylsulfoxide], and 0.05% sodium azide). Mixtures without synthetic peptides were used as negative controls. The mixtures were incubated for 3 days at 37°C with agitation at 300 rpm and were used for further assays.
After incubation, 10 µL of each reaction mixture was placed onto a glass slide and evaporated. The dried smears were stained with Congo red and examined under polarized light. To determine the ultrastructure of peptide-derived fibrils, the mixtures were subjected to transmission electron microscopy. After incubation, each sample was prepared by applying tiny drops of the mixture on 200-mesh copper grids supported by Formvar-carbon film, which were then blotted to remove excess water and air-dried. The grids were stained with EM Stainer (Nisshin EM, Tokyo, Japan) and examined under a transmission electron microscope (JEM-1400; JEOL, Tokyo, Japan) at 80 kV.
Real-Time Reverse-Transcription Polymerase Chain Reaction
To compare the expression levels of the CK5 gene (KRT5) in amyloid-positive and amyloid-negative cases (n = 10 each), a real-time reverse-transcription quantitative polymerase chain reaction (RT-qPCR) was performed (Supplemental Table S3). Total RNA was extracted from each FFPE tissue section using an innuPREP FFPE total RNA Kit (Analytik Jena, Überlingen, Germany) following the manufacturer’s instructions. Complementary DNA samples were prepared from 500 ng of total RNA using PrimeScript RT Master Mix (Takara Bio Inc.,, Shiga, Japan) in a MiniAmp Thermal Cycler (Applied Biosystems, Waltham, Massachusetts) (37°C for 15 minutes and 85°C for 5 seconds). Real-time PCR was performed as previously described. 12 Primer sequences were designed using Primer Blast in NCBI based on the mRNA sequences of Canis lupus familiaris KRT5 from the GenBank database (accession number: NM_001346035.1) and are listed in Supplemental Table S4. Results were normalized to the expression of endogenous control (β-2-microglobulin; B2M); KRT5 expression of amyloid-positive samplesrelative to that of amyloid-negative samples was calculated using the ΔΔCt method. The melting curve of amplification products was analyzed at the end of each PCR reaction to verify specific amplification.
Gene Sequencing Analysis
Gene sequencing analysis was performed to evaluate the mutations in the amyloidogenic region of CK5 as predicted by PASTA 2.0. The sample list (7 amyloid-positive cases and 5 amyloid-negative cases) for gene sequencing is shown in Supplemental Table S3. Total RNA extraction from each FFPE tissue section was performed as described above. The primer set was designed to cover codons corresponding to the predicted amyloidogenic region (Supplemental Table S4). One-step RT-PCR using a PrimeScript One-Step RT-PCR Kit (Takara Bio) was performed as follows: reverse transcription at 50°C for 30 minutes, 30–40 cycles each of 94°C for 30 seconds, 60°C for 30 seconds, and 72°C for 1 minute, followed by a final extension at 72°C for 3 minutes. The product size was analyzed by agarose gel electrophoresis, and target cDNA fragments (330 bp) were purified from the gel. Nucleotide sequences were determined using the BigDye Terminator v3.1 Cycle Sequencing Kit with an ABI 3730xl DNA Analyzer (Applied Biosystems, Foster City, California). Each amino acid sequence was translated from the sequenced codons using EMBOSS Transeq. 18 Multiple sequence alignment of the determined amino acid sequences with canine CK5 in the database (NCBI accession: NP_001332964.1) was analyzed using EMBOSS Omega. 18
Statistical Analysis
Data were analyzed using Fisher’s exact test or Mann-Whitney U test, depending on the purpose of analysis. P value of <.05 was considered statistically significant. All statistical tests were performed using GraphPad Prism version 8.0 software (GraphPad Software, San Diego, California).
Results
Pathological Findings
Histologically, amyloid deposition was observed in 16 of 266 cases comprising 4/88 trichoblastoma cases (5%), 10/103 trichoepithelioma cases (10%), 2/36 pilomatricoma cases (6%), and 0/101 in infundibular keratinizing acanthoma cases (0%). The case information is shown in Supplemental Tables S1 and S5. Amyloid deposits were observed as eosinophilic amorphous materials by H&E (Fig. 1a–c), stained pale red by Congo red, and exhibited yellow to green birefringence under polarized light (Fig. 1d–f).
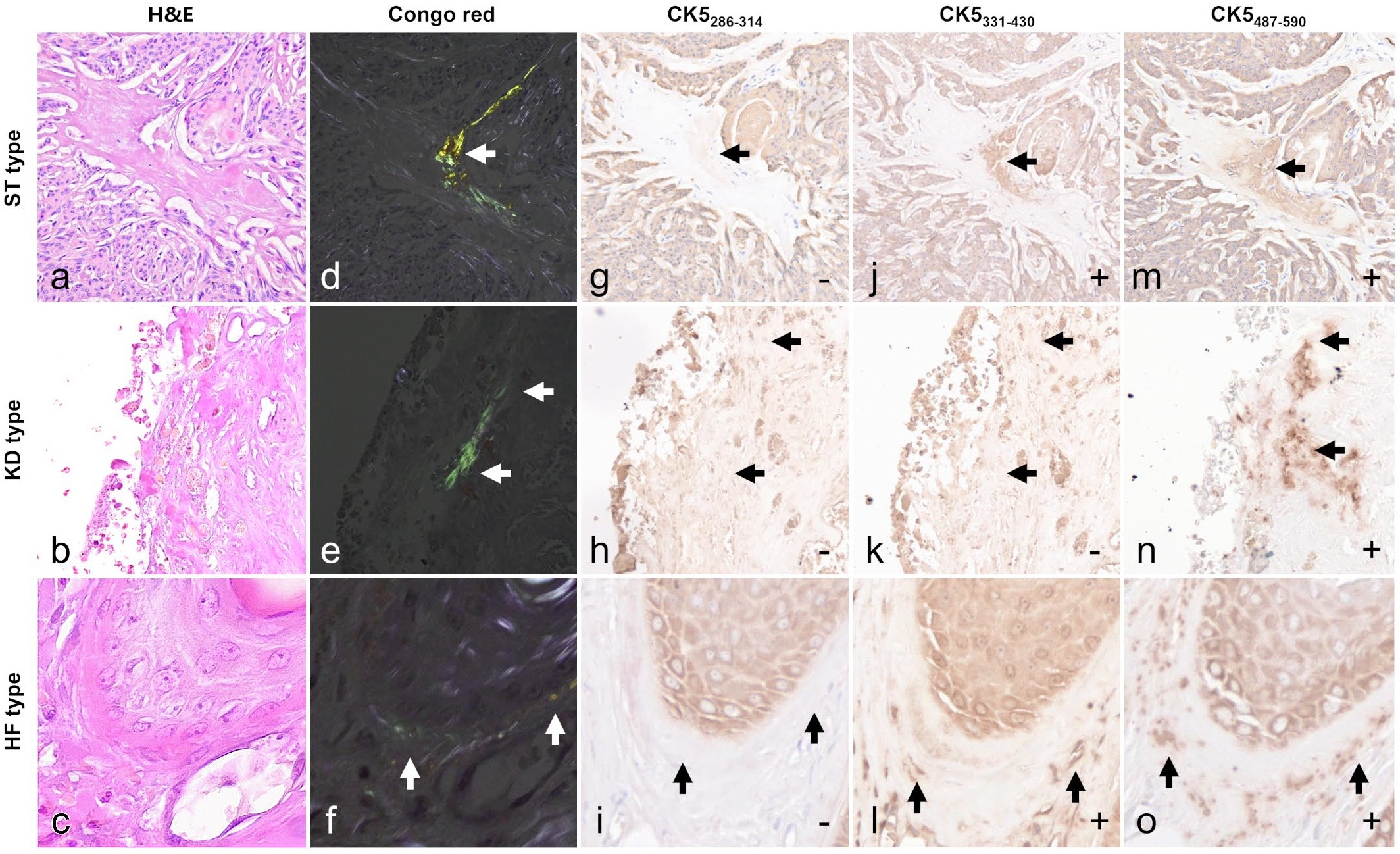
Amyloidosis, hair tumors and hair follicle, dog. Arrows indicate the amyloid deposits. (a–c) Hematoxylin and eosin (H&E) stain. (d–f) Congo red stain. Amyloid deposits show green to yellow birefringence under polarized light. (g–o) Immunohistochemical images. The symbols at the bottom right of the figures indicate immunoreactivity. Amyloid deposits in the stroma of a trichoblastoma (ST; case 1) and around the normal hair follicles adjacent to a trichoepithelioma (HF; case 16) were positive for both cytokeratin 5 (CK5) 331–430 and CK5487–590, whereas amyloid deposits in keratin debris of a trichoepithelioma (KD; case 11) were positive for CK5487–590 only.
Based on amyloid deposition pattern, 3 types were identified as follows: intratumoral stroma (ST) type (n = 10), keratin debris (KD) type (n = 4), and HF type (n = 2). ST types had amyloid deposits scattered in the intratumoral stroma. KD types showed amyloid deposits attached to the margins of keratin accumulated in neoplastic follicles. In the HF type, amyloid deposits were observed in strips to dots around normal HFs outside the tumor. All cases had small amyloid deposit lesions and no severe deposits that would compress or replace existing tissue.
IHC analysis showed that the amyloid deposits in all amyloid-positive cases were negative for serum amyloid A, immunoglobulin κ and λ light chains, and transthyretin (Supplemental Table S6).
Statistical Analysis
The statistical results are listed in Table 1. The incidence of amyloid deposition in Shiba dogs was significantly higher than that in other breeds (P = .0281, Fisher’s exact test). The mean age of amyloid-positive cases was significantly higher than that of amyloid-negative cases (P = .0016, Mann-Whitney U test). The incidence of amyloid deposition was higher in female dogs (7.9%) than in male dogs (4.2%), although the difference was not statistically significant (P = .3022, Fisher’s exact test).
Statistical analysis of canine hair follicle tumor samples.
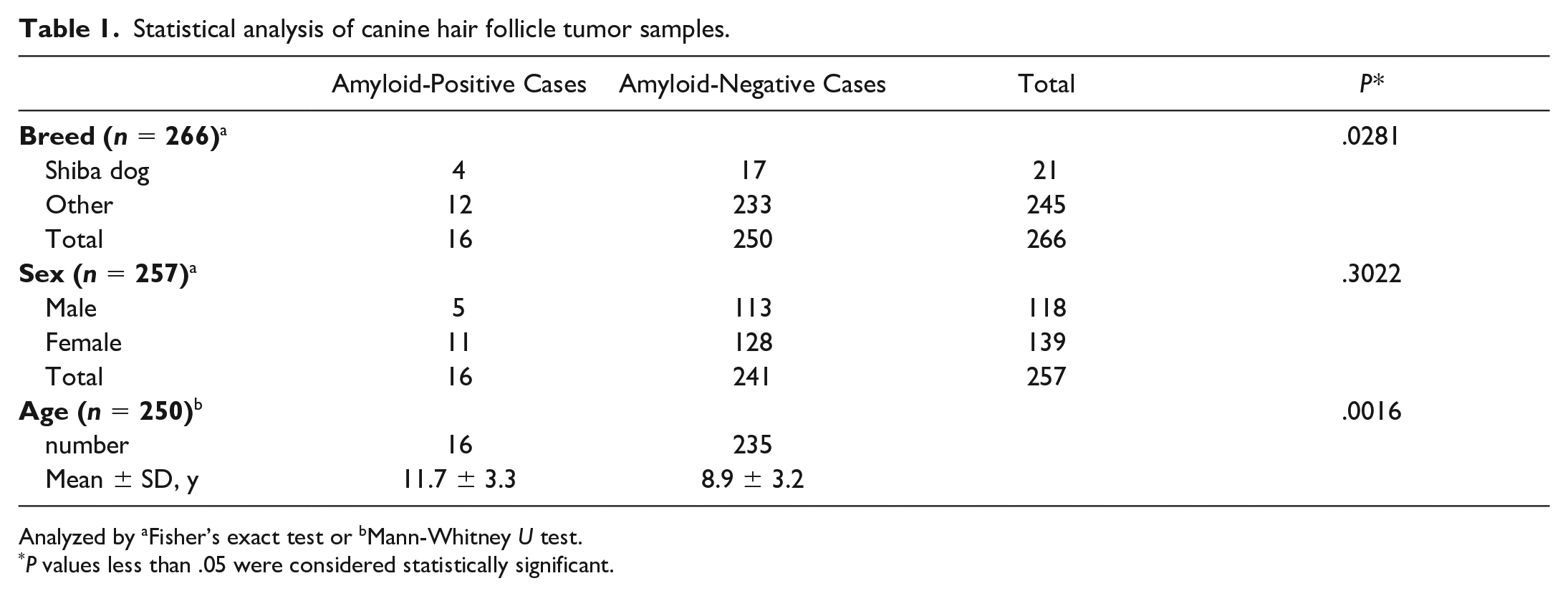
Analyzed by aFisher’s exact test or bMann-Whitney U test.
P values less than .05 were considered statistically significant.
Proteome Analyses
CK5 and CK14 were consistently among the top 6 detected proteins in all cases (Table 2). CK10 was the second most prevalent protein in case 15. Collagen was detected at high levels in all cases; however, its exponentially modified protein abundance index (emPAI) value, a quantitative index of MASCOT analysis, was lower than those of CKs. Selective collection of amyloid deposits by laser microdissection was ensured by detecting apolipoprotein A-I, apolipoprotein A-IV, and apolipoprotein E as amyloid signature proteins. 23 Although the lambda light chain was detected in case 11 as an amyloid-formable protein, its score and emPAI value were lower than those of CKs.
List of proteins identified by mass spectrometry.
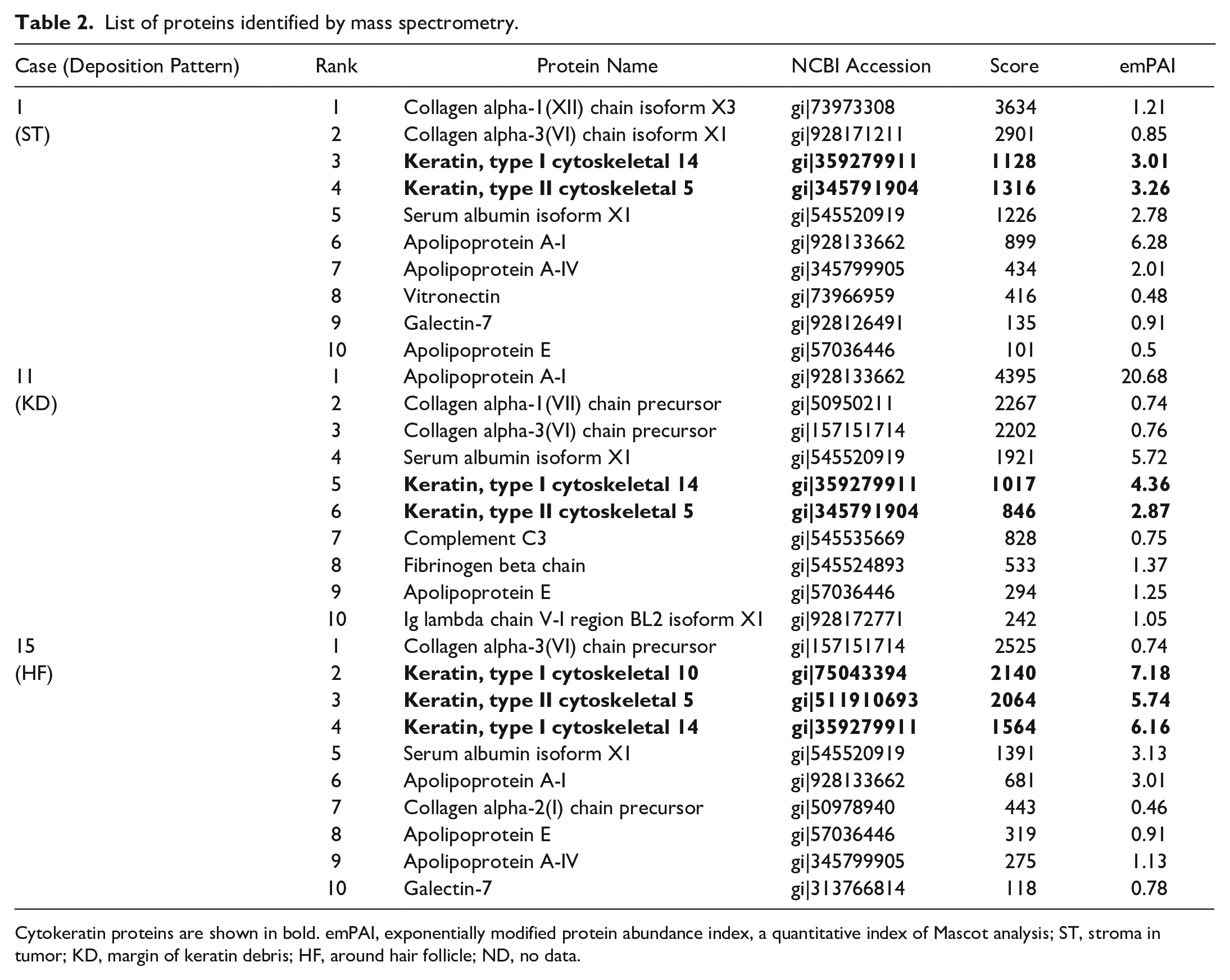
Cytokeratin proteins are shown in bold. emPAI, exponentially modified protein abundance index, a quantitative index of Mascot analysis; ST, stroma in tumor; KD, margin of keratin debris; HF, around hair follicle; ND, no data.
Prediction of Fragmentation Site
Several fragmentation sites of the CK5 peptide were detected at 333–337 aa in cases 1 and 15, which were ST and HF types, respectively. In case 11 (KD type), several fragmentation sites were detected at 397–423 aa (Fig. 2, Supplemental Fig. S1). Fragmentation sites around 550 aa were also detected in cases 1, 11, and 15 (Fig. 2).
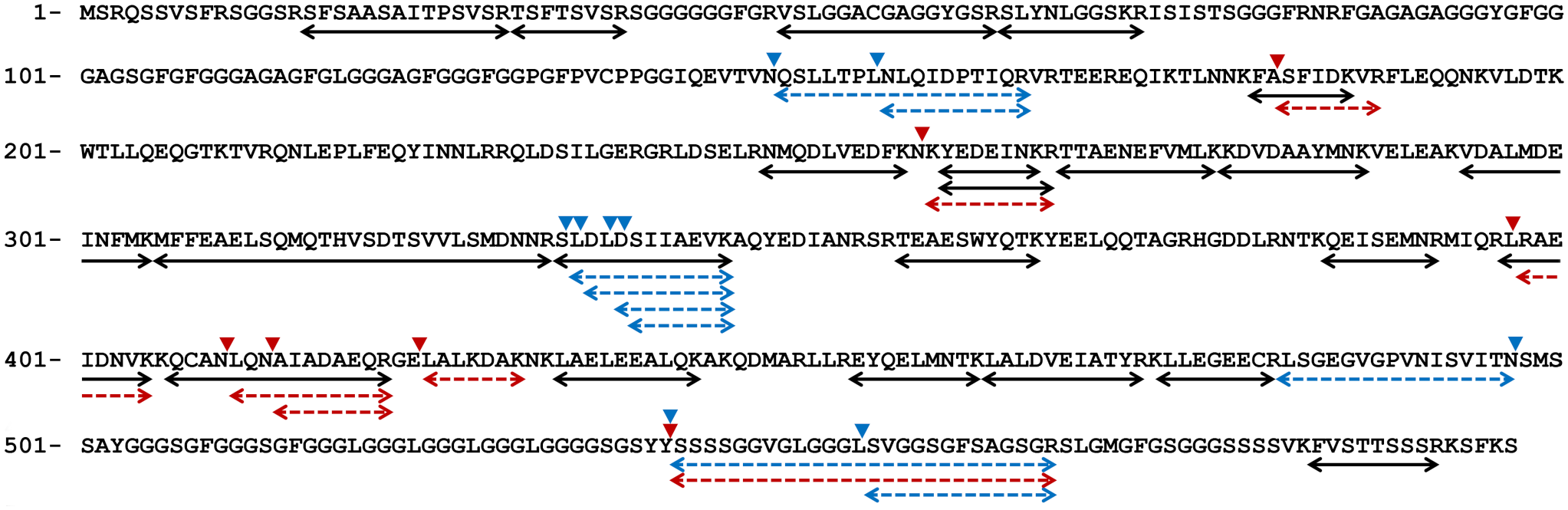
Peptide fragments detected by mass spectrometry. Double-headed arrows indicate peptides detected from all cases (black lines), cases 1 and 15 (stroma in tumor type and hair follicle type, blue dashed lines), and case 11 (keratin debris type, red dashed lines). Arrowheads indicate the truncation site, ie, the site indigestible by trypsin.
IHC Results
Amyloid deposits in all samples did not label for CK10poly, CK10DE-K10, CK14poly, CK14LL022, and CK5286–314 (Fig. 1g–o, Supplemental Fig. S2). The ST and HF types were positive for CK5331–430 and CK5487–590, whereas the KD types were positive for CK5487–590 but negative for CK5331–430 (Supplemental Table S6). Cases lacking amyloid deposits did not have any labeling other than follicular labeling, and no labeling was observed in primary antibody-omitted negative controls.
In infundibular keratinizing acanthoma, CK5 was positive in the spinous layer cells, CK14 was positive across all layers, and CK10 was positive in the spinous layer, granular layer, and a part of the stratum corneum (Supplemental Fig. S3). In trichoblastomas and trichoepitheliomas, CK5 and CK14 were positive in almost all tumor cells, whereas CK10 was sparsely positive. In pilomatricomas, CK5 and CK14 were positive in the basal layer, whereas CK10 was positive in spinous cells.
Prediction of Structure
PASTA 2.0 analysis predicted that the C-terminal regions at 490–504 aa of canine CK5 and at 489–503 aa of human CK5 have high amyloid propensity (Fig. 3). Intrinsically disordered regions of approximately 100 aa in length at the N-terminal and C-terminal of both canine CK5 and human CK5 proteins were identified.
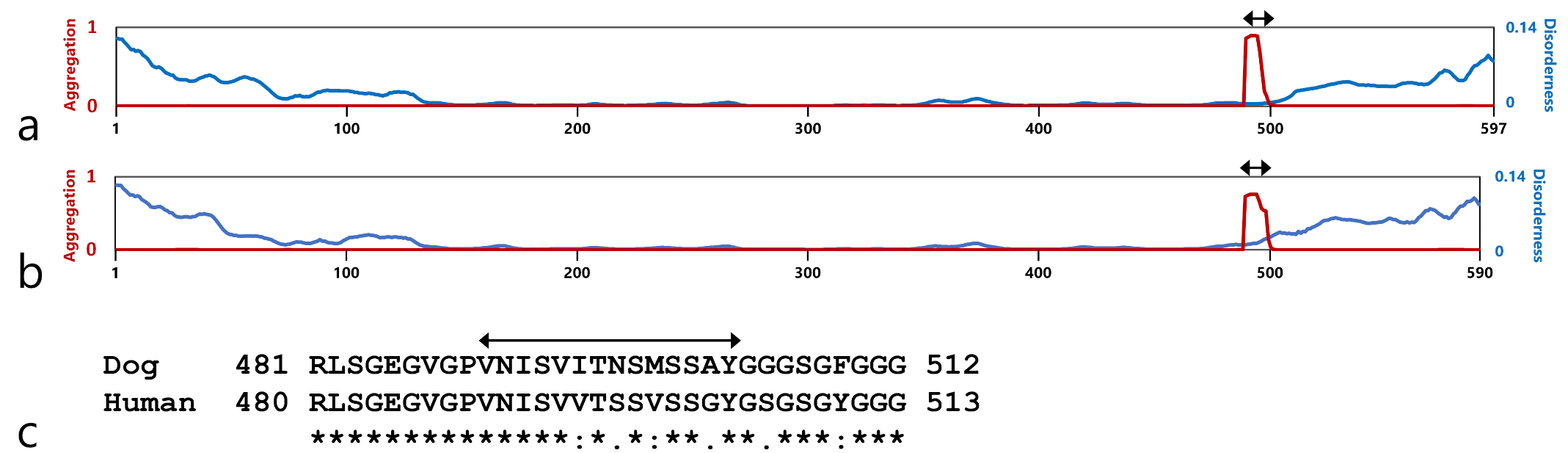
Structural analysis by PASTA2.0 of canine CK5 (a) and human CK5 (b). Red and blue lines indicate the amyloid propensity and disorderness of CK5, respectively. The red lines indicate a sharp increase in the aggregation potential of amino acids 489–503. The blue lines indicate increased disorderness of the N- and C-terminal domains. (c) Pairwise alignment of highly amyloidogenic sequences of canine and human CK5. Asterisks (*) indicate fully conserved residues. Colons (:) and periods (.) indicate conserved residues between groups with strongly or weakly similar properties, respectively. Double-head arrows indicate the reference site of the synthetic peptide used for in vitro fibrillization (Dog, 490–504 aa, VNISVITNSMSSAY; Human, 489–503 aa, VNISVVTSSVSSGY).
In Vitro Fibrillization
Both dog and human CK5 peptides showed Congo red birefringence under polarized light (Fig. 4a, b) and formed fibrils with a diameter of 15 nm (Fig. 4c, d).
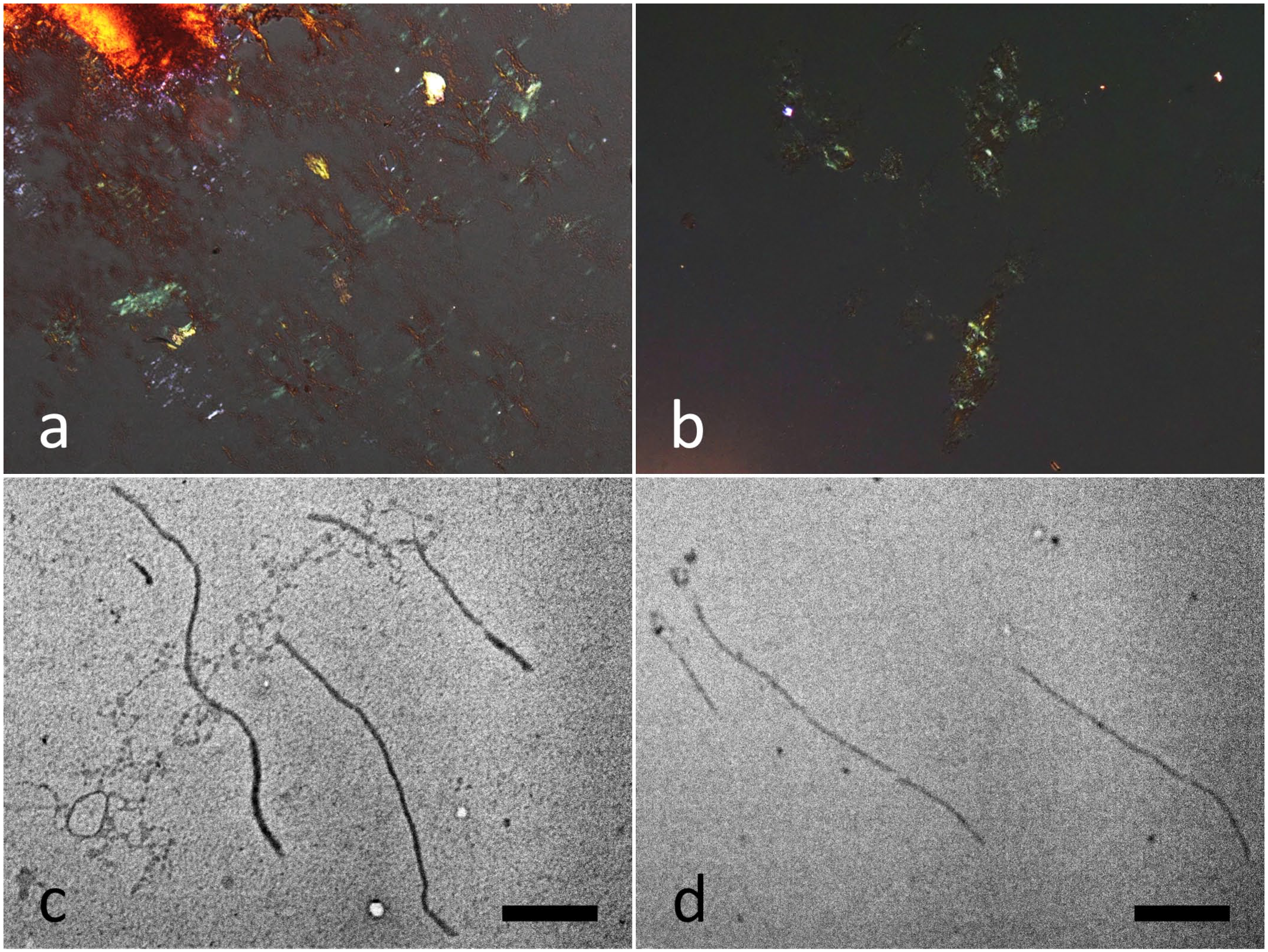
In vitro fibrillization of peptides derived from canine or human CK5. Congo red polarization of canine CK5 peptide-derived amyloid (a) and human CK5 peptide-derived amyloid (b). Transmission electron microscopy image of canine CK5 peptide-derived fibrils (c) and human CK5 peptide-derived fibrils (d). Bars = 1 µm.
RT-qPCR Analysis
There was no significant difference in the expression level of KRT5 between amyloid-positive and amyloid-negative cases (Supplemental Fig. S4).
Gene Sequencing Analysis
An amino acid polymorphism (p.Met499Val) due to a single-base substitution (c.1495A>G) was detected in cases 3, 8, 10, and 11 and in amyloid-negative cases (Fig. 5, Supplemental Fig. S5). This polymorphism was heterozygous (G/A) in case 3 and amyloid-negative cases, and homozygous (G/G) in cases 8, 10, and 11 (Shiba dogs, Supplemental Fig. S5). PASTA 2.0 analysis suggested the extension of the high amyloidogenic region in CK5 caused by the Met499Val polymorphism (Supplemental Fig. S6). Insertion of 4 amino acids (p.Lue527_Gly528insGlyGlyGlyLeu) due to a 12-base insertion (c.1581_1582insGGCGGTGGTCTT) was detected in cases 8, 10, and 11. An amino acid polymorphism (p.Val547Ile) due to a single-base substitution (c.1639G>A) was also found in cases 8 and 10. Single-base substitutions (c.1573G>A and c.1641C>T) were similarly observed in case 3 and amyloid-negative cases, resulting in amino acid replacement (p.Gly525Ser). Amino acid insertions and polymorphisms other than Met499Val did not affect the prediction results for the amyloidogenic region.

Amino acid sequences of CK5 translated from sequenced codons. The top row shows the amino acid sequence obtained from the database (NCBI accession: NP_001332964.1). Black shadings indicate the homozygous polymorphism site determined by gene sequencing. Gray shadings indicate the heterozygous polymorphism site. Amyloid (−): amyloid-negative case. Refer Supplemental Table S5 for each case information.
Discussion
In this study, we screened canine HF tumors and found amyloid deposits. Laser capture microdissection followed by LC/MS/MS analysis revealed CK5, CK10, and CK14 as the major components of amyloid deposits in canine kPLCA. IHC analysis showed that amyloid deposits were positive for CK5 and negative for CK10 and CK14, suggesting that CK5 may be an amyloid precursor protein. The reason why CK10 and CK14 were detected by mass spectrometry is thought to be contamination from the tissue surrounding the amyloid during laser microdissection. Fragmentation of amyloid proteins has been reported. 20 We identified several fragmentation sites of CK5 in the amyloid deposits.
Interestingly, amyloid deposits were found not only in the stroma of tumors (ST type), but also in the margin of keratin debris (KD type) and around normal HFs (HF type). Structural prediction and in vitro fibrillization test results showed that peptides at 490–503 aa might be involved in canine CK5 amyloid formation. Notably, the fragmentation sites of CK5 varied across each amyloid deposition pattern. The mass spectrometry and IHC results indicated that fragmented CK5 polypeptides after 333–337 aa in the ST and HF types and those after 397–423 aa in the KD type are major amyloid components, respectively. Thus, we attribute the difference in fragmentation sites to the difference in the amyloid formation process. For the ST and HF types, amyloid deposits are speculated to originate from basolateral-released CK5. In human kPLCA, according to the filamentous degeneration theory, filamentous degenerated keratinocytes are released into the fibrous tissue of stroma around HFs and form amyloid via some interaction.8,15,17 In this study, we suspected that CK5, which is released into the interstitium, was fragmented to form amyloid. For the KD type, CK5 released from keratinocytes during keratinization is hypothesized to be fragmented to form amyloid. However, to confirm these amyloidogenic mechanisms, further studies will be necessary on degenerated keratinocytes and intracellular changes.
Amyloid deposition was not observed in infundibular keratinizing acanthoma. Canine infundibular keratinizing acanthoma consists of cells that differentiate into the squamous epithelium, whereas trichoblastoma, pilomatricoma, and trichoepithelioma have abundant basaloid cells,11,19 indicating that basal cells might be more involved in the pathogenesis of kPLCA than spinous cells. These results also support that CK5, an intermediate filament of basal cells, and not CK10 which is expressed in spinous cells, is the precursor protein of amyloid fibrils. 16
Increased protein expression destabilizes proteins and promotes amyloid formation. 26 However, we found no significant difference in KRT5 expression levels between amyloid-positive and amyloid-negative cases. In addition, amyloid deposits were found not only in the tumor, but also around the normal HFs. These results indicate that elevated protein expression associated with tumorigenesis may not necessarily be required for amyloidogenesis of canine CK5.
The incidence of kPLCA was significantly higher in Shiba dogs than in other breeds, suggesting that genetic factors are involved in the pathogenesis. Several amino acid polymorphisms in CK5 were detected in Shiba dogs (p.Met499Val, p.Lue527_Gly528insGlyGlyGlyLeu, and p.Val547Ile); the Met499Val polymorphism predicted increased amyloidogenicity of canine CK5. Interestingly, the homozygous Met499Val polymorphism of CK5 in Shiba dogs is consistent with the Val498 of human CK5. When Val498 was replaced with methionine in human CK5, PASTA analysis showed that the predicted region of high amyloidogenicity was shortened by 4 amino acids (Supplemental Fig. S6). In various types of amyloidoses, a single amino acid substitution, such as Val30Met mutation in human ATTR amyloidosis, 10 is known to have a significant effect on the amyloidogenicity of precursor proteins. The milder disease state in dogs compared with humans and the significantly higher incidence in Shiba dogs are both likely attributed to the unique amino acid sequence of their CK5. Genetic examinations of the total length of KRT5 using a larger number of samples are necessary to clarify genetic involvement in the amyloid deposition of canine HF tumors.
Human kPLCA has a higher incidence in women than in men,5,21 and basal-cell carcinoma-associated human kPLCA is more common in elderly patients. 7 Similarly, we found that the incidence of amyloid deposition in female dogs was 1.9 times higher than that in male dogs (albeit without statistical significance) and identified aging as a risk factor for amyloid deposition. Therefore, sex and aging are associated with the pathogenesis of cutaneous keratinic amyloid deposition in both humans and dogs.
A partial peptide of the C-terminal region in both human and canine CK5 formed amyloid in vitro, suggesting that human CK5 may form amyloid as well as canine CK5. Previous studies on human tumor–associated kPLCA mostly used IHC to analyze the amyloid protein.1,8 There are 2 reports that performed both mass spectrometry and IHC; pan-CK primary antibodies were used for IHC in these studies.2,6 As CK proteins have many common sequences, it is essential to analyze them using a combination of antibodies specific to each CK protein. The fragment-based IHC used in this study may also be useful in identifying an amyloid precursor protein in human kPLCA.
In conclusion, we discovered cutaneous keratinic amyloid deposition in canines and revealed CK5 as an amyloid precursor protein. CK5 fragmentation may be involved in its amyloidogenesis, and fragmentation sites varied according to the histological deposition pattern. Canine cutaneous keratinic amyloid deposition shares similar underlying diseases and risk factors with human kPLCA. Furthermore, our results suggest that the KRT5 genetic variation plays a role in canine amyloid pathology. Although further investigation is required to determine whether CK5 is a solo amyloidogenic protein in kPLCA, in vitro fibrillization test suggested the possible amyloidogenic properties of human CK5. This study provides valuable insights into veterinary medicine and the etiology of human kPLCA and may contribute to an integrated understanding of the pathophysiology of amyloidosis.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221128924 – Supplemental material for Keratinic amyloid deposition in canine hair follicle tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858221128924 for Keratinic amyloid deposition in canine hair follicle tumors by Kyoko Kobayashi, Susumu Iwaide, Hiroki Sakai, Fuyuki Kametani and Tomoaki Murakami in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by the JSPS KAKENHI (grant no. 20K15660) and the Program on Open Innovation Platform with Enterprises, Research Institute and Academia from the Japan Science and Technology Agency.
Supplemental Material for this article is available online.
