Abstract
Malignant pilomatricoma, also known as pilomatrix carcinoma and calcifying epitheliocarcinoma (in the human literature), has been considered a rare neoplasm of dogs. The authors present 3 canine cases of malignant pilomatricoma (2 with distant metastasis) and compare its behavior with reported behavior. Cases include an 8-year-old spayed female Airedale Terrier, a 7-year-old male Bassett Hound, and a 12-year-old intact male Giant Schnauzer. In all cases, the histologic features included trabeculae of basaloid cells, abrupt keratinization, “ghost” or “shadow” cells, and various features of malignancy consistent with a diagnosis of malignant pilomatricoma. Metastasis, including that to bone, was confirmed in 2 cases. Four cases of the 13 canine pilomatricomas diagnosed within a 24-month period (2006–2008) at the Ohio State University (2 of which are discussed in this report) were classified as malignant, which suggests that malignant pilomatricoma is more common than previously reported.
Pilomatricoma is a benign tumor that arises from the germinative cells of the follicular matrix, or hair bulb, and is reported mainly in dogs 9 and humans; in the latter species, it is known as necrotizing and calcifying epithelioma of Malherbe or pilomatrixoma. 8 The tumor is rare in aged Sprague-Dawley rats 34 and unreported in cats. 9
Pilomatricoma accounts for 1 to 3% of canine skin tumors 9,26 and is most frequently diagnosed in 4- to 8-year-old dogs, 6 without apparent sex predilection. 8 Kerry Blue Terriers, Soft Coated Wheaten Terriers, Standard Poodles, and other breeds with continuously growing coats and predominantly anagen hair follicles are overrepresented, perhaps because the relative increase in anagen follicles provides a larger pool of replicating cells in which neoplastic transformation can occur. 9
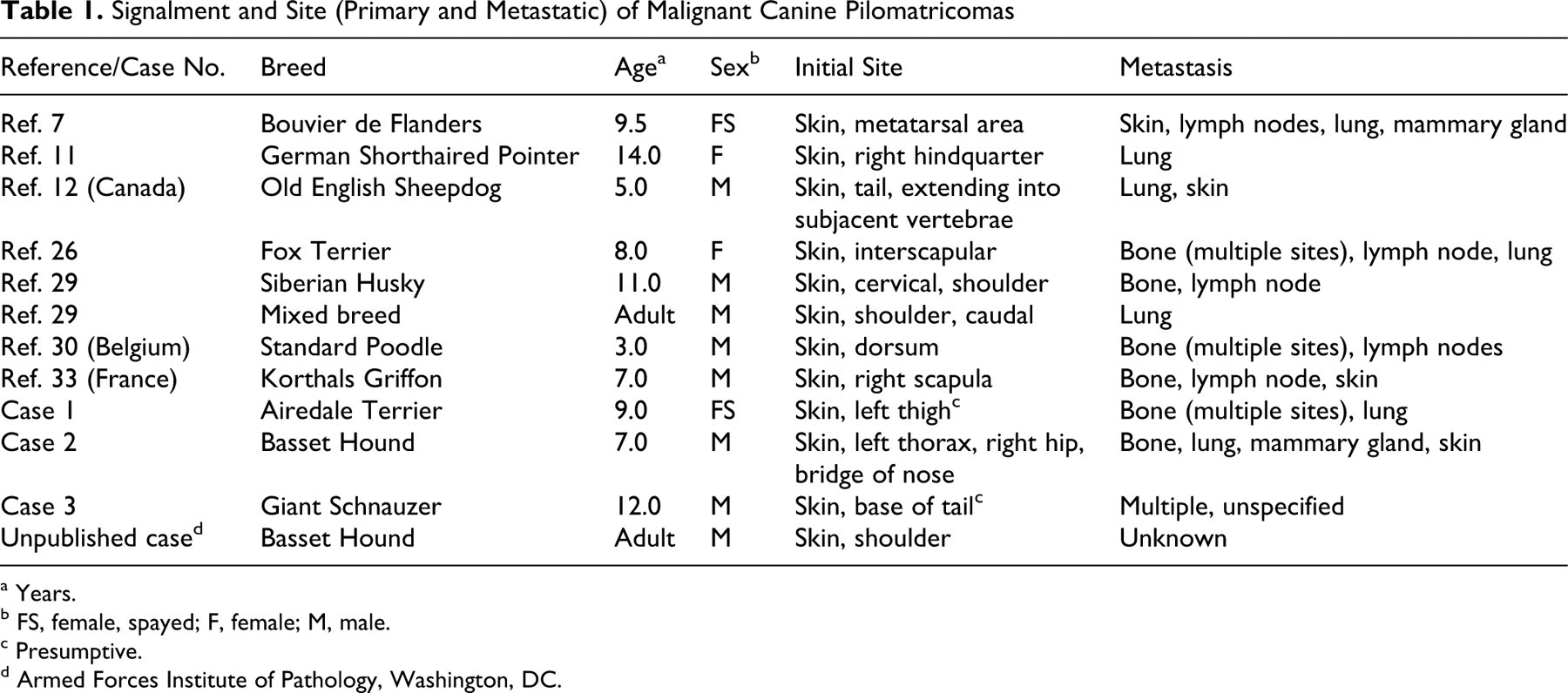
Malignant pilomatricoma has been considered rare in dogs, with only 5 published cases in the United States (Table 1 ). 7,11,26,29 Additional single cases have been reported from Canada, 12 Belgium, 30 and France. 33 Reported cases of malignant pilomatricoma were sufficiently differentiated for histologic recognition but were locally aggressive, with invasion of bone 9,12 and metastasis to bone, 4,26,29,30,33 lymph nodes, 4,7,26,29,30,33 and lung. 7,11,12,26,29 Dogs with malignant pilomatricoma have been presented with lameness, as reported here, or neurological signs. 26,30 The reason for metastasis of a hair follicle tumor to bone is unknown but not surprising, considering that bone is exceeded only by lung and liver as the most common site for metastasis of certain human carcinomas (breast, prostate, lung, and others). 21 Additionally, osseous metaplasia is common in benign and malignant pilomatricoma. 9 We present 3 cases of malignant pilomatricoma, 2 of which had distant metastasis, and discuss the behavior of this neoplasm.
Signalment and Site (Primary and Metastatic) of Malignant Canine Pilomatricomas
a Years.
b FS, female, spayed; F, female; M, male.
c Presumptive.
d Armed Forces Institute of Pathology, Washington, DC.
Materials and Methods
Cases were selected from the archives of the Ohio State University College of Veterinary Medicine and the Armed Forces Institute of Pathology (AFIP). Four cases met the selection criteria of a histologic diagnosis of malignant pilomatricoma: nuclear or cellular atypia, erratic or rapid growth rate, increased number (over normal tissue) of mitotic figures, infiltrative growth, and absence of a capsule. One of the 4 cases was excluded from the study because of incomplete information on the extent of the tumor and outcome of the case. Tissues were fixed in 10% neutral buffered formalin, routinely processed, sectioned to a thickness of 5 μm, and stained with hematoxylin and eosin (HE). Bony tissue from case No. 1 was decalcified in Rapid Bone Decalcification solution (Master Tech Scientific, Inc, Lodi, CA) for 24 hours, sectioned, and stained with HE or evaluated immunohistochemically at AFIP with a Kermix cocktail made from a solution of monoclonal mouse anti-human cytokeratin antibodies LP34, 34BE12, and 35BH11 (1:400, code No. N1589, Dako, Carpinteria, CA) and a prediluted mouse anti-human cytokeratin epithelial monoclonal antibody blend of AE1/AE3 (1:400, catalog No. IHC 2025-6, Chemicon International, Temecula, CA). An avidin–biotin complex method was used at the Ohio State University (see http://vet.osu.edu/905.htm) for cytokeratin and vimentin immunohistochemistry.
Case Histories
Table 1 summarizes signalment, site of the primary tumor, and metastatic sites.
Case 1
An 8-year-old spayed female Airedale Terrier was presented with lameness of the right rear limb that had progressed to complete nonweightbearing within a month. A 4-cm-diameter mass was in the skin of the left thigh. An oval lytic lesion was radiographically evident in the distal metaphysis of the right femur with thinning of the overlying cortex and irregular periosteal new bone formation. The limb was amputated at the hip. In radiographs on the day of surgery, no pulmonary masses were detected, but the distal aspects of the right tibia and femur had radiating “starburst-like” periosteal bony proliferation with a possible pathologic fracture of the distal tibial diaphysis (Fig. 1). Neoplastic tissue from the amputated limb was submitted to Antech Diagnostics (Baltimore, MD), where it was evaluated and then forwarded to the Division of Veterinary Pathology, AFIP, for consultation. Three weeks postsurgery, 3 new masses appeared on the dog’s head and lateral aspect of the left thigh. Five weeks postamputation, the dog was returned to the clinic in pain, with lethargy, and with additional masses in a left mammary gland and in the skin on the right side of the thorax. The dog was euthanized, and selected tissues were submitted to Antech and forwarded to the AFIP.
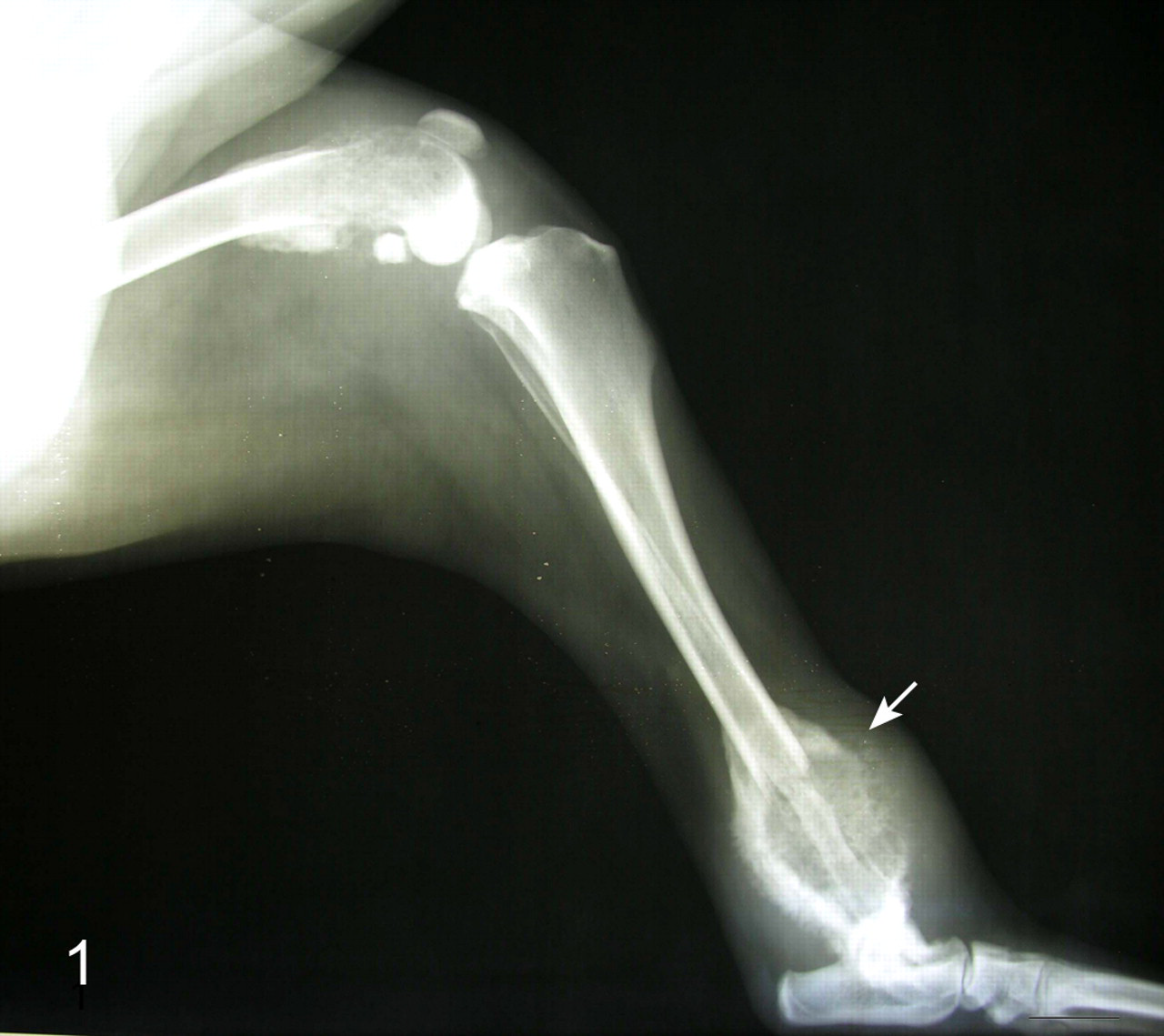
Radiograph of right hind limb; dog No. 1. The distal aspects of femur and tibia have areas of bony lysis with radiating periosteal new bone formation. The tibia is fractured (arrow).
Case 2
A 7-year-old intact male Basset Hound was presented for necropsy to the Ohio State University College of Veterinary Medicine. The dog had been euthanized by a local humane society for neglect. The dog had reportedly had a cutaneous mass and paraphimosis for an “extended period of time.” Cutaneous masses on the left thorax, right hip, and bridge of the nose had been removed approximately 18 months earlier without histologic evaluation. The submitting veterinarian described those masses as being 3 to 7 cm in diameter, erosive, necrotic, and infiltrative. At necropsy, a moderately firm subcutaneous mass (diameter, 1.5 cm) on the left shoulder was, on cross section, tan to dark brown with gray layers of soft material. Within the abdominal cavity, a lobulated, rubbery to hard, spherical to irregular, white gritty mass (diameter, 12 cm) involved the pelvis and each vertebra from the seventh lumbar to the first coccygeal, with sparing of intervertebral discs. A mass (diameter, 6 cm) was in the right perianal tissue, and a mediastinal mass (diameter, 5 cm) effaced a presumptive mediastinal lymph node. Approximately 25% of the lung was replaced by firm-to-hard white gritty nodules (3–5 cm). The same white gritty material was in the pelvic mass, the mediastinal lymph node, and the pulmonary nodules; therefore, the nodal mass and pulmonary nodules were believed to represent metastases from the same primary neoplasm, the site of which was undetermined.
Case 3
A 12-year-old intact male Giant Schnauzer was presented to the Ohio State University College of Veterinary Medicine for evaluation of left tibial and fibular fractures after being struck by a car. During physical examination, multiple masses were noted at various unspecified locations, including a firm mass (diameter, 10 cm) just cranial to the tail base. Multiple wedge biopsy specimens were submitted for histologic evaluation.
Histologic Findings
In case No. 1, the distal aspect of the femur was infiltrated by islands and trabeculae of neoplastic cells that surrounded lakes of eosinophilic material to form cystlike structures. The neoplastic cells were polygonal with scant cytoplasm, hyperchromatic oval nuclei, moderate anisocytosis, mild anisokaryosis, and 4 mitotic figures per 10 fields (400×). Scattered cells had diffuse eosinophilic intracytoplasmic material that was interpreted as keratin. The eosinophilic extracellular material contained myriad oval pale foci (4–6 μm). The proximity of this material to basaloid cells was consistent with the formation of “ghost” cells resulting from abrupt keratinization. Neoplastic cells, mixed with numerous lymphocytes, infiltrated the marrow and trabecular bone.
A rib had focal cortical effacement and replacement by a poorly circumscribed, unencapsulated nodule that consisted of islands and trabeculae of moderate cellular density with similar foci of pale eosinophilic material. The periosteum was markedly elevated by narrow trabeculae of woven bone oriented perpendicularly to the cortex; this was interpreted as subperiosteal reactive bone. The nodule was interpreted as a metastasis of the pilomatricoma.
Similar neoplastic basaloid cells, arranged around cystlike structures filled by the eosinophilic material, were in skin adjacent to mammary glands, in the left thigh (Figs. 2, 3) and in soft tissue at the hind limb amputation site. The mammary metastasis was associated with desmoplastic proliferation of spindle-shaped cells in amphophilic fibrillar matrix. Areas of abrupt keratinization were often covered by intensely basophilic homogeneous material interpreted as dystrophic mineralization. Metastatic neoplastic cells also distended and surrounded a few vessels in pulmonary interalveolar septa.
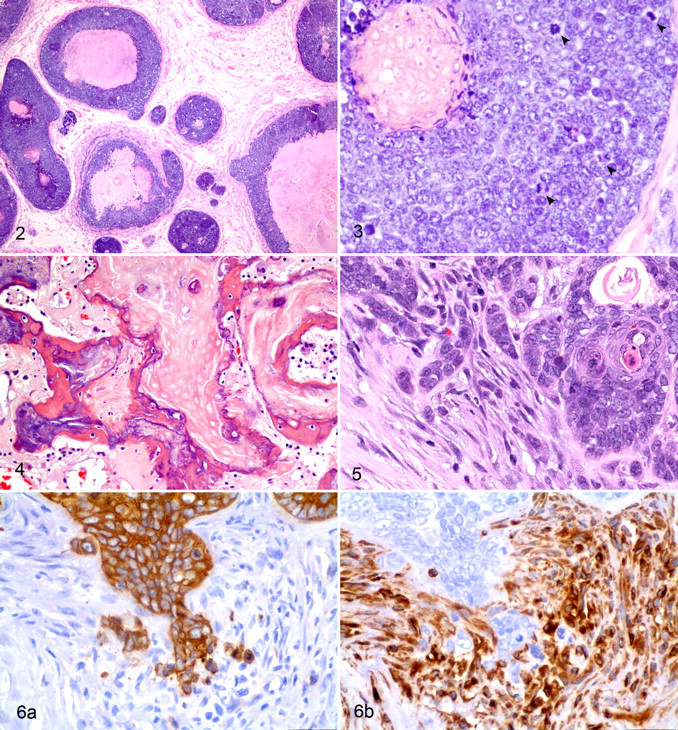
Immunohistochemically, 5 to 8% of the neoplastic polygonal cells had moderate to strong expression of cytokeratin. Roughly 10% of the basaloid neoplastic cells had faint perinuclear immunoreactivity for cytokeratin. The ghost cells were generally negative to faintly immunoreactive. Most infiltrating lymphocytes were CD3-positive T cells, with rare CD79a-positive B cells. Many round cells were not immunoreactive to the applied antibodies, presumably owing to loss of antigenic determinants during decalcification.
In case No. 2, the histologic findings were generally similar to those of case No. 1. The subcutaneous shoulder mass was diagnosed as a malignant pilomatricoma and was presumed to be the primary tumor. In a section including 2 vertebrae, most of the vertebral body on either side of the intervertebral disc was replaced by a mass of anastomosing trabeculae of stratified squamous epithelial cells with coalescing areas of necrotic (coagulation) epithelial cells (ghost cells) and extensive woven bone formation. Nuclear and mitotic atypia was minimal to mild. Preexisting bone had undergone marked osteolysis at the interface with neoplastic tissue. Case No. 2 also had abundant osseous metaplasia in areas of ghost cells (Fig. 4). The vertebral neoplasm was believed to be a metastasis from the subcutaneous pilomatricoma of the shoulder.
In case No. 3, the dermal mass at the base of the tail consisted predominantly of thin cords and nests of multilayered basaloid to polygonal epithelial cells with scattered foci of squamous differentiation. These cells surrounded nodular aggregates of keratin with pale ghost cells reminiscent of hair matrix. Case No. 3 differed from case Nos. 1 and 2 by the presence of marked anisocytosis and anisokaryosis, abundant desmoplasia, and lack of observed vascular invasion. However, neoplastic cords and islands focally invaded tissue at the deep margin of the specimen. Additionally, proliferative spindle cells were mixed with neoplastic epithelial cells in what resembled epithelial-to-mesenchymal transition (Fig. 5). The polygonal neoplastic cells were cytokeratin positive, and the spindle-shaped cells were vimentin positive, with minimal gradation of immunoreactivity between the 2 morphological cell types (Fig. 6).
Discussion
Most canine pilomatricomas are benign, 4,31 so recognition of a malignant pilomatricoma is paramount. Malignancy is generally distinguished histologically from benign processes by predominance of the following features: lack of differentiation, atypical histologic structure, anaplasia, erratic or rapid growth, increased mitotic index, infiltrative growth, and—the definitive criterion—metastasis. 18 The diagnosis of malignant pilomatricoma “is best made when lymphatic invasion can be identified, usually at the periphery of the tumor.” 5
In this study, the behavior of all 3 cases was clearly malignant. In contrast, the histologic features of the primary tumors varied from case to case and met only some criteria of malignancy. The presumed primary tumor of case No. 1 was well differentiated, but it metastasized early. Case No. 2 was less well differentiated, but the presumed primary tumor in case No. 3 had the most obvious histologic features of malignancy. Thus, histologic appearance in malignant pilomatricoma may not correlate with behavior.
Dystrophic calcification and osseous metaplasia, including lamellar bone formation, often occur in aggregates of ghost cells in pilomatricoma. 6,10,24 Dystrophic calcification can occur wherever there is necrosis. It arises intracellularly and extracellularly and has two phases. 17 Initiation of intracellular calcification occurs in the mitochondria of dead or dying cells. Extracellular dystrophic calcification begins with membrane phospholipids, which form vesicles upon being released from degenerating or aging cells. Following initiation, propagation of calcium phosphate occurs, similar to the formation of hydroxyapatite of bone. 27 Organic matrix attracts inorganic minerals that lead to its solidification.
Osseous metaplasia, however, is less well understood but regularly reported in canine pilomatricoma, particularly in the later stages of tumor development. 4,8,9 Osseous metaplasia has been reported in various neoplasms in dogs and cats. 3,4,8,28 It has also been reported in mice with metal ear tags, 15 in the retinal pigmented epithelium of phthisical eyes, 17 and in an otherwise normal canine eye. 20 Metaplasia results not from a change in phenotype of differentiated cells but from reprogramming of either stem cells in normal tissues or undifferentiated mesenchymal cells in connective tissue. 17 The differentiation of stem cells to the new lineage is induced by cytokines, growth factors, and signals from extracellular matrix components in the cell’s environment. 17,25
In a human rectal adenocarcinoma with osseous metaplasia, Kypson et al 19 found overexpression of bone morphogenetic protein 2, a known inducer of osteoblastic differentiation, in neoplastic cells. They hypothesized that stimuli such as bone morphogenetic protein 2, as generated by malignant epithelial cells, induce pluripotent mesenchymal cells to become osteoblasts and produce bone. 19 A similar mechanism may occur in malignant pilomatricoma.
Malignant pilomatricoma seems predisposed to metastasize to bone. Of the 13 reported cases (including the 3 reported here), 7 metastasized to or extended directly into bone, including vertebrae, ribs, mandible, maxilla, and femur. 26,29,30,33 Four of the 6 cases without reported bone metastasis were not examined by necropsy; thus, bony lesions may have been missed.
Metastasis to bone by human tumors (myeloma and solid tumors) is most common in the best-vascularized parts of the skeleton (eg, the axial skeleton, proximal aspect of long bones, and ribs). 21 Early investigators injected dyes to demonstrate venous return from the prostate gland—tumors of which metastasize to bone—to the vertebral column via the vertebral venous plexus. 1,21 Properties of the “landing” site that determine whether metastasis is possible 13,21 include a micromilieu that permits metastatic cells to adhere and proliferate. 13 Paget described his “seed and soil” hypothesis of tumor metastasis more than 100 years ago. 24 The bone microenvironment is “fertile soil” for numerous solid carcinomas. 21 For example, human breast cancer metastasizes to bone, where it produces colony stimulating factor 1, parathyroid hormone–related protein, and tumor necrosis factor–alpha, which activate the receptor activator of nuclear factor–kappa B ligand, which stimulates the formation and activity of osteoclasts. 14 Osteoclasts degrade bone matrix, which in turn releases insulin-like growth factor 1, transforming growth factor–beta, and bone morphogenetic proteins, thereby promoting survival of metastatic cells and stimulating parathyroid hormone–related protein synthesis. 22,23 The result is a positive feedback loop of osteolysis and metastatic growth. 22,23 Additional factors that promote metastasis to bone include the production of chemokines in bone that attract circulating receptor-bearing tumor cells, 13 increased expression of cytokines that bind tumor cells bearing cognate adhesion molecules (eg, E-cadherin and laminin), 2,21 and the presence of tumor-associated macrophages. 10
The results of immunohistochemistry of pilomatricoma are equivocal. Walter 32 tested 205 canine epithelial skin tumors of epidermal, hair follicle, sebaceous gland, apocrine gland, and perianal (hepatoid) gland origin. Cytokeratin 6 was present in all tumor types except pilomatricoma, which did not react with any of the 13 antibodies tested, typical of hair follicle matrix cells in healthy canine skin. In contrast, trichoepithelioma was labeled with antibody to cytokeratins 1, 5, 6, 8, 10, 14, 16, 18, and 19. Basal cells of intracutaneous cornifying epithelioma (infundibiular keratinizing acanthoma) were labeled with antibodies to cytokeratins 6, 14, and 18. Basal cell carcinomas were labeled by cytokeratins 5, 6, 8, 14, and 18. 32 In other studies, 16,30 basaloid and maturing cells (but not ghost cells) of malignant pilomatricoma variably reacted with antibodies to cytokeratin 8. Cytokeratin 5 antibodies variably labeled malignant pilomatricoma basaloid cells, ghost cells, and squamoid areas, with numerous nonreactive areas. 30 Ghost cells of the same tumor were both vimentin and cytokeratin positive. 30
Pilomatricoma is a skin tumor. Malignant pilomatricoma usually has the same clinical presentation, 7,11,12,29,33 but in some cases, the primary clinical complaint is dysfunction at a metastatic site, such as lameness (case No. 1) or neurological disease. 26,30 Thus, metastatic carcinoma, including malignant pilomatricoma, should be in the differential diagnosis for lameness, paraplegia, and other neurologic diseases.
Histologic examination may distinguish malignant pilomatricoma from other follicular tumors, such as benign pilomatricoma and malignant trichoepithelioma. Malignant pilomatricoma generally has more basaloid cells relative to the keratinized ghost cells, poor circumscription with infiltration, more mitotic activity, and nuclear and mitotic atypia of the basaloid cells. 9 Malignant pilomatricomas can be difficult to distinguish from malignant trichoepitheliomas, but the former “usually have larger epithelial aggregates, a marked predominance of atypical matrical cells, and larger zones of ghost cells.” 9 Malignant trichoepitheliomas have trichohyalin granules and prominent focal inner and outer root sheath differentiation, usually without calcification or ossification. 9 Keratinizing basal cell carcinoma differs from malignant pilomatricoma by its superficial plaque-like architecture and contiguity with the epidermis. 9
Malignant pilomatricoma may be more common, aggressive, and life-threatening than previously thought. Although reportedly rare, these 3 cases of malignant pilomatricoma—plus 3 additional, unpublished cases (1 at AFIP, 2 at Ohio State University)—were submitted within a 24-month period (January 2006–January 2008). During the same period, 9 benign canine pilomatricomas were submitted to the Ohio State University. Dogs with malignant pilomatricomas were presented late in the course of disease, and the tumor led to the demise of the patient in at least 3 cases. Awareness of the behavior of malignant pilomatricoma may lead to accurate earlier diagnosis and treatment and potentially longer lives in affected dogs.
Footnotes
Acknowledgements
Our thanks to Mr David Miles, Walter Reed Army Institute of Research, for imaging technical expertise and to Dr Kristine Wilson, Prospect Veterinary Clinic, Frederick, Maryland, for detailed case information.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
