Abstract
We performed a retrospective examination of spontaneous hepatocellular carcinomas (HCCs) (primary and metastatic tumors) in 14 captive prosimians brought to the Veterinary Medical Diagnostic Laboratory in North Carolina State University over a period of 11 years (2003 to 2014) to characterize the tumors. These animals are endangered primates; a better understanding of the main fatal neoplasms is crucial. In addition to the histologic evaluation, an immunohistochemical study was also performed, using a hepatocyte marker (hepatocyte paraffin 1 [HepPar-1]) and 2 cholangiocyte markers (keratin 7 [K7] and keratin 19 [K19]), in an attempt to identify a specific profile for HCCs with metastatic behavior. Six of the 14 HCCs had pulmonary metastases. The most frequent histopathological findings were a trabecular pattern (14/14, 100%), presence of multinucleated cells (12/14, 85.7%), and foci of extramedullary hematopoiesis (9/14, 64.3%). The mitotic count was significantly higher in the metastatic HCCs (
Prosimians are the most primitive member of the Primate family, and the family includes lemurs and lorises. The most common spontaneous tumors in prosimians are hepatocellular neoplasia, followed by lymphoma/leukemia, biliary, and mammary neoplasms.
39
Hepatocellular carcinomas (HCCs) have been diagnosed at least 3 times more frequently than hepatocellular adenomas. The tumor pathogenesis remains unclear. Indeed, lemurs affected by HCC do not have excessive hepatic level of iron, copper, or molybdenum, and there is no evidence of hepadnavirus or hepatitis C virus infection.
57
It is also worth noting that spontaneous hepatic neoplasia has been rarely reported in anthropoids. HCCs have been described in 3 male cynomolgus monkeys (
Making a distinction between a primary
To the best of our knowledge, an immunohistochemical evaluation of HCCs in prosimians has never been reported in a series of tumors. The aims of this article are to review spontaneous HCCs (primary and metastatic neoplasms) diagnosed in North Carolina State University over a period of 11 years (2003 to 2014), to further characterize the lesions histologically and immunohistochemically using 3 markers (HepPar-1, K7, and K19), and to test the relevance of these markers in the distinction between HCCs with or without metastases in prosimians.
Materials and Methods
Case Material
The surgical and autopsy veterinary pathology database at North Carolina State University was searched for all cases of HCC in primates submitted from January 2003 through December 2014. Samples were collated and reviewed to determine their suitability for the investigation. One case (a 24-year-old female Collared brown lemur) with no tissue remaining was excluded, and a total of 14 cases were selected for the study (Table 1). Signalment and main gross pathologic findings were recorded.
Summary of hepatocellular carcinomas in captive prosimians.
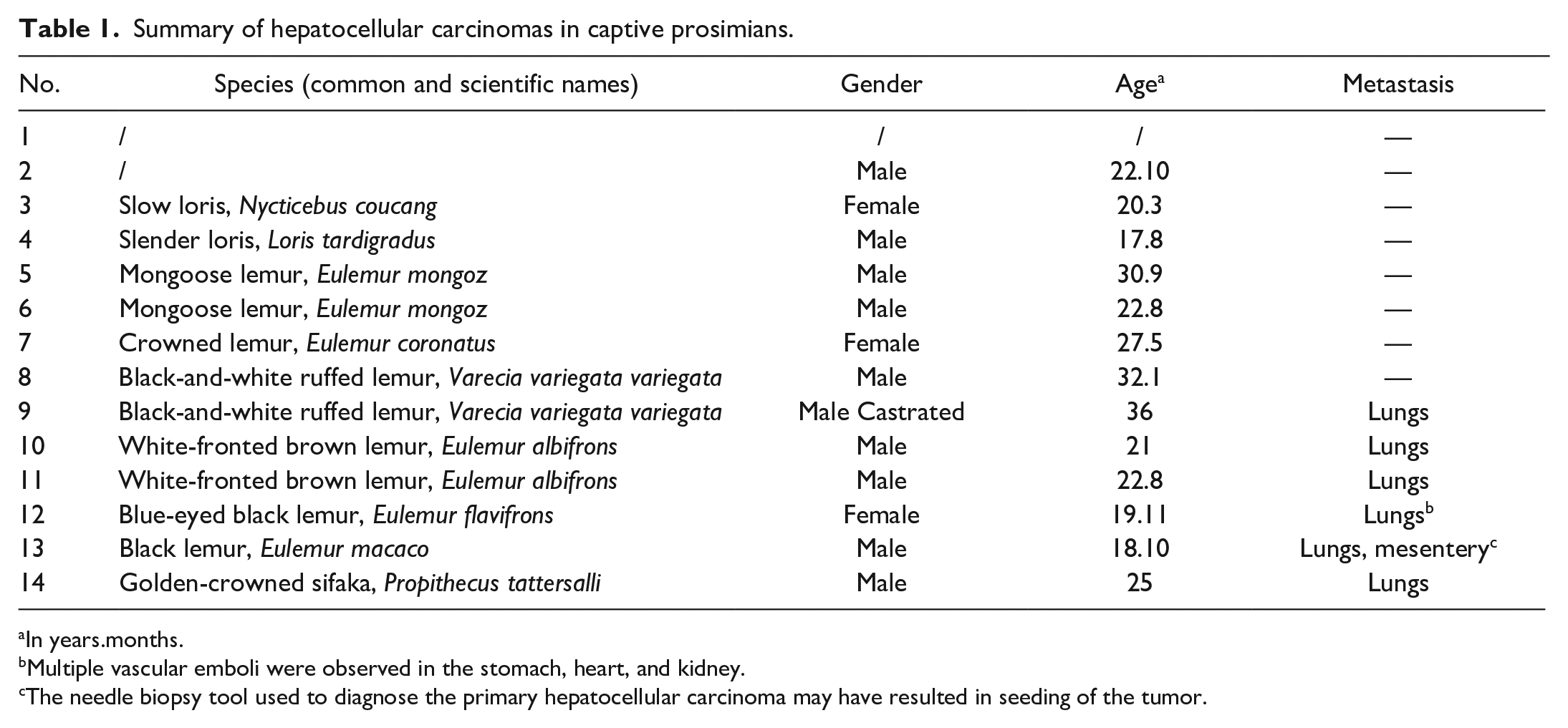
In years.months.
Multiple vascular emboli were observed in the stomach, heart, and kidney.
The needle biopsy tool used to diagnose the primary hepatocellular carcinoma may have resulted in seeding of the tumor.
Tissues were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin. The tumors were classified based on the nomenclature described for dogs and humans, that is, microscopic architectural patterns, degree of anaplasia, local invasion, mitotic count, and presence of metastasis.13,20,34 In 12 cases, at least 2 samples of each intrahepatic tumor were evaluated. Neoplastic cells were organized in short trabeculae of irregular thickness (trabecular pattern), acinus-like structures of variable shape and size (pseudoglandular pattern), 1 to 2 cell-layer-thick cords of neoplastic hepatocytes separated by large blood-filled spaces (pelioid pattern), and/or cords separated by abundant fibrous stroma (scirrhous pattern). Anaplasia was classified as mild, moderate, or marked based on the anisocytosis, anisokaryosis, and multinucleation. Variation at 2-fold or less in cell and nuclear sizes was graded as mild, 3-fold was moderate, and 4-fold or more was marked. Local invasion was characterized by focal infiltration of the surrounding parenchyma by the neoplastic population. The mitotic count was performed in 10 high-power fields, equivalent to 2.37 mm2 (diameter of the field of view = 0.55 mm; 40× objective and 10× ocular; Nikon Eclipse Ni microscope), starting from hotspots and avoiding areas with necrosis. Depending on the gross findings, several organs (including kidney, lung [except for the case 3], and frequently heart, spleen, and thyroid glands) were collected during the autopsy, and the presence of metastasis was based on the histologic evaluation.
Reagents and Immunohistochemistry Procedure
The details of immunohistochemical reagents and methods are given in Table 2. Immunohistochemistry was performed using an automatic stainer (Dako Autostainer, Carpinteria, CA). Deparaffinized sections were incubated with 3% H2O2 for 10 minutes to block endogenous peroxidase activity. The Envision-plus detection system, including an anti-mouse polymer, was used for secondary antibody labeling. The signal was developed in 3,3’-diaminobenzidine (DAB) solution for 5 minutes and finally counterstained with Mayer’s hematoxylin. Healthy hepatocytes and bile ducts served as an internal positive control for HepPar-1 and keratins 7 and 19, respectively. Internal positive controls for all antibodies used in the study demonstrated cross-reactivity with non-human primate tissues, and there was no background staining. Negative controls were performed by replacing the primary antibody with wash buffer. In addition, a negative labeling with HepPar-1 was observed in all non-hepatic tissues tested (kidney, lung, spleen, striated muscle, colon, parathyroid gland, uterus, testis, lymph node, adrenal gland), except the thyroid gland.
Primary antibodies and procedures used for the immunohistochemical examination.
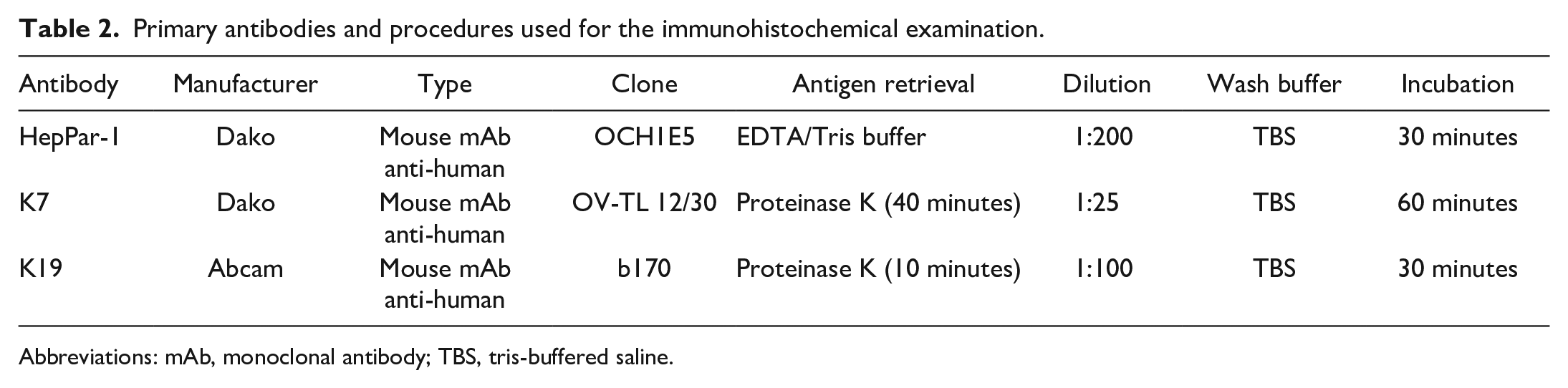
Abbreviations: mAb, monoclonal antibody; TBS, tris-buffered saline.
Scoring of HepPar-1, K7, and K19 Immunohistochemistry
Immunohistochemical evaluation was performed by a single board-certified pathologist (C.R.) in a blinded manner, using serial sections immunolabeled for HepPar-1, K7, and K19. For each sample, the overall immunoreactivity was semi-quantitatively evaluated with grading from 0 to 4 as follows: 0 = no staining; 1 = 5% to 15% of positive cells; 2 = 16% to 50%; 3 = 51% to 80%, and 4 = >80%. Cases with rare positive cells (<5%) were not observed. This scoring was based on a previously published study of Ramos-Vara et al 37 and applied to the whole tissue section at low magnification (2× objective) after verification of the specific staining at higher magnifications. When the score was different among the samples from the same animal, each score was mentioned. In addition, the percentages of K7-positive cells were evaluated manually by counting at least 1000 cells (except for case No. 14 because of a lack of adequate tumoral tissue) in the most representative areas, using a 40× objective.
Statistical Analysis
Data were compared using unpaired Student’s
Results
Fourteen HCCs in primates collected over a period of 11 years (2003 to 2014) were evaluated. Species identification was unavailable in 2 cases (Nos. 1 and 2); age and gender were unavailable in 1 case (No. 1). HCCs were identified in both lemurs and lorises with a sex ratio of 10 males (76.9%) to 3 females (23.1%). All animals were adults, ranging from 17 years and 8 months to 36 years, with an average of 24 years and 5 months. Metastases occurred in 6 animals (42.8%), all lemurs, and the lungs were always involved. One animal also had mesenteric metastases, which may have resulted from postoperative abdominal implantation with a needle biopsy tool.
Histopathological and immunohistochemical results are summarized in Table 3. Microscopically, 4 architectural patterns of HCC were identified, and at least 2 patterns were apparent in 11/14 cases (Figs. 1–4). The trabecular pattern was always present in both primary and metastatic tumors; it was the predominant pattern in the liver in 12/14 cases (85.7%). The pseudoglandular pattern and the pelioid pattern were identified in 6/14 cases (42.9%). A scirrhous pattern was focally identified in a single case (No. 6). The clear cell variant of HCC was observed in 2 cases (Nos. 5 and 9) (Supplemental Figs. S1–S2). Neoplastic cells maintained many characteristic hepatocellular features, including distinct cell borders, a cuboidal to polygonal shape, a central round nucleus, and an abundant eosinophilic cytoplasm. They were larger (2- to 5-fold) than nonneoplastic hepatocytes, and had an increased nuclear-cytoplasmic ratio. Nuclear pseudoinclusions, invaginations of cytoplasm into the nucleus, were frequently identified (Fig. 5). Moderate to marked anaplasia, including large multinucleated cells containing up to 30 nuclei, were observed in all HCCs except the clear cell variant (Fig. 6). Additional histopathological findings commonly found included foci of extramedullary hematopoiesis (9/14, 64.3%) (Fig. 7), peripheral ductular reaction defined by proliferation of irregular and tortuous ductules formed by cuboidal basophilic epithelium (6/14, 42.9%) (Figs. 9–14), and large necro-hemorrhagic areas. Less frequent findings were bile plugs (Nos. 7, 8, and 13), and the presence of intracytoplasmic eosinophilic inclusions that were variable in size and shape (Nos. 4, 10, and 12) (Fig. 8). The average mitotic count of the primary tumors for 2.37 mm2 was 4 in the nonmetastatic HCCs (Nos. 1-8) and 18 in the metastatic HCCs (cases 9-13), which was significantly different (
Summary of the histological and immunohistochemical results of hepatocellular carcinomas in captive prosimians.
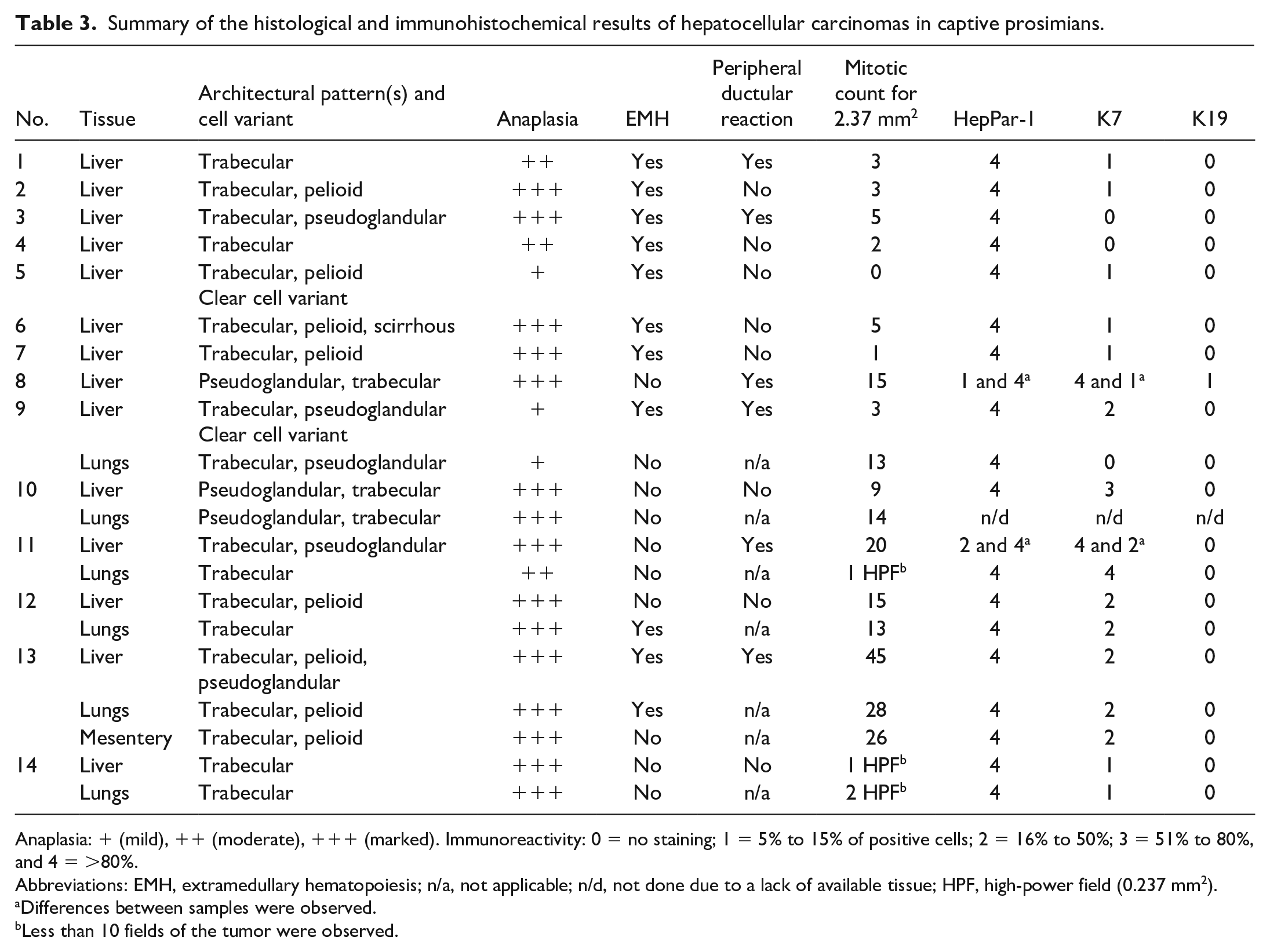
Anaplasia: + (mild), ++ (moderate), +++ (marked). Immunoreactivity: 0 = no staining; 1 = 5% to 15% of positive cells; 2 = 16% to 50%; 3 = 51% to 80%, and 4 = >80%.
Abbreviations: EMH, extramedullary hematopoiesis; n/a, not applicable; n/d, not done due to a lack of available tissue; HPF, high-power field (0.237 mm2).
Differences between samples were observed.
Less than 10 fields of the tumor were observed.
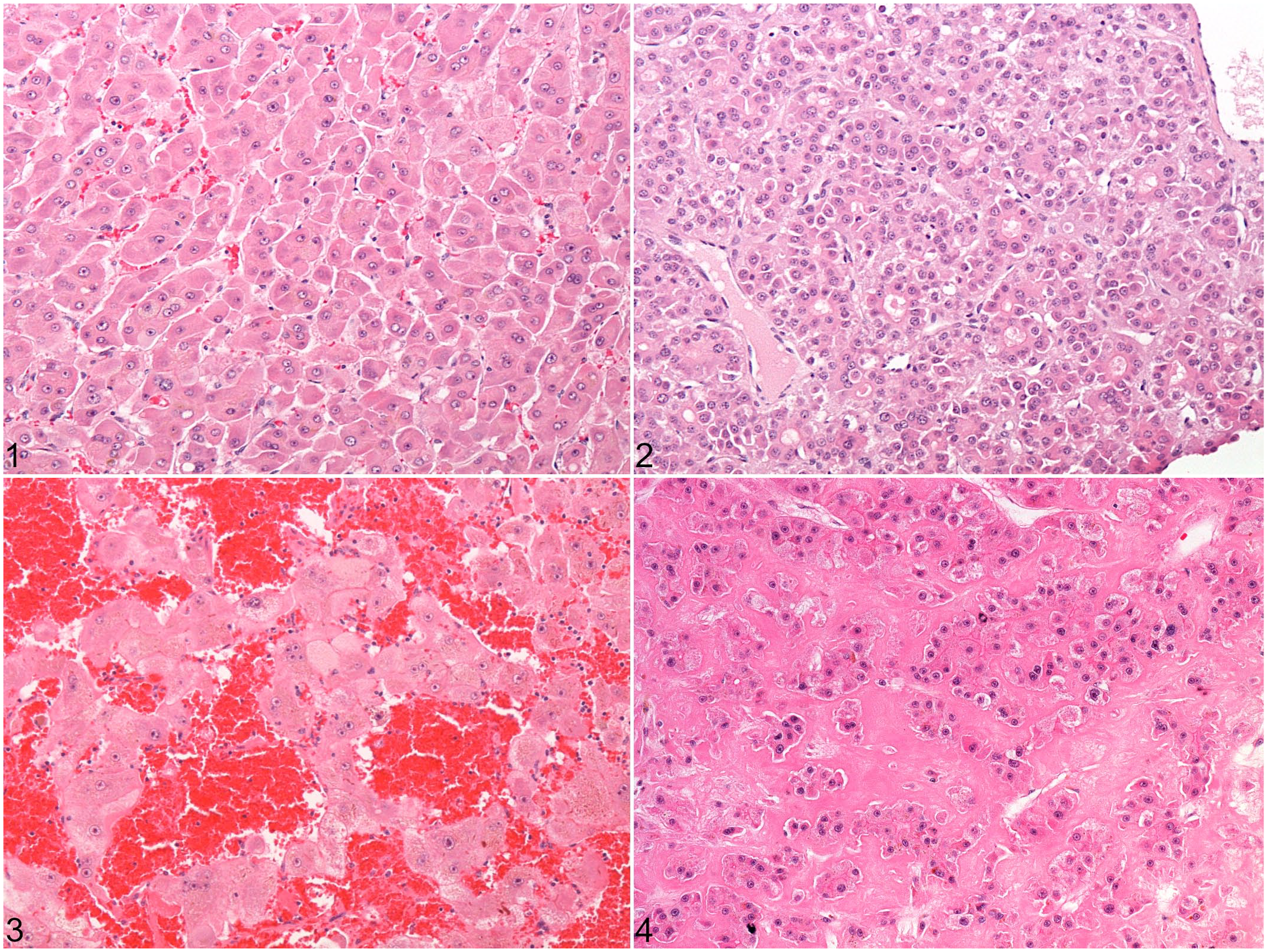
Architectural patterns of hepatocellular carcinoma in the liver of prosimians. Hematoxylin and eosin (HE).
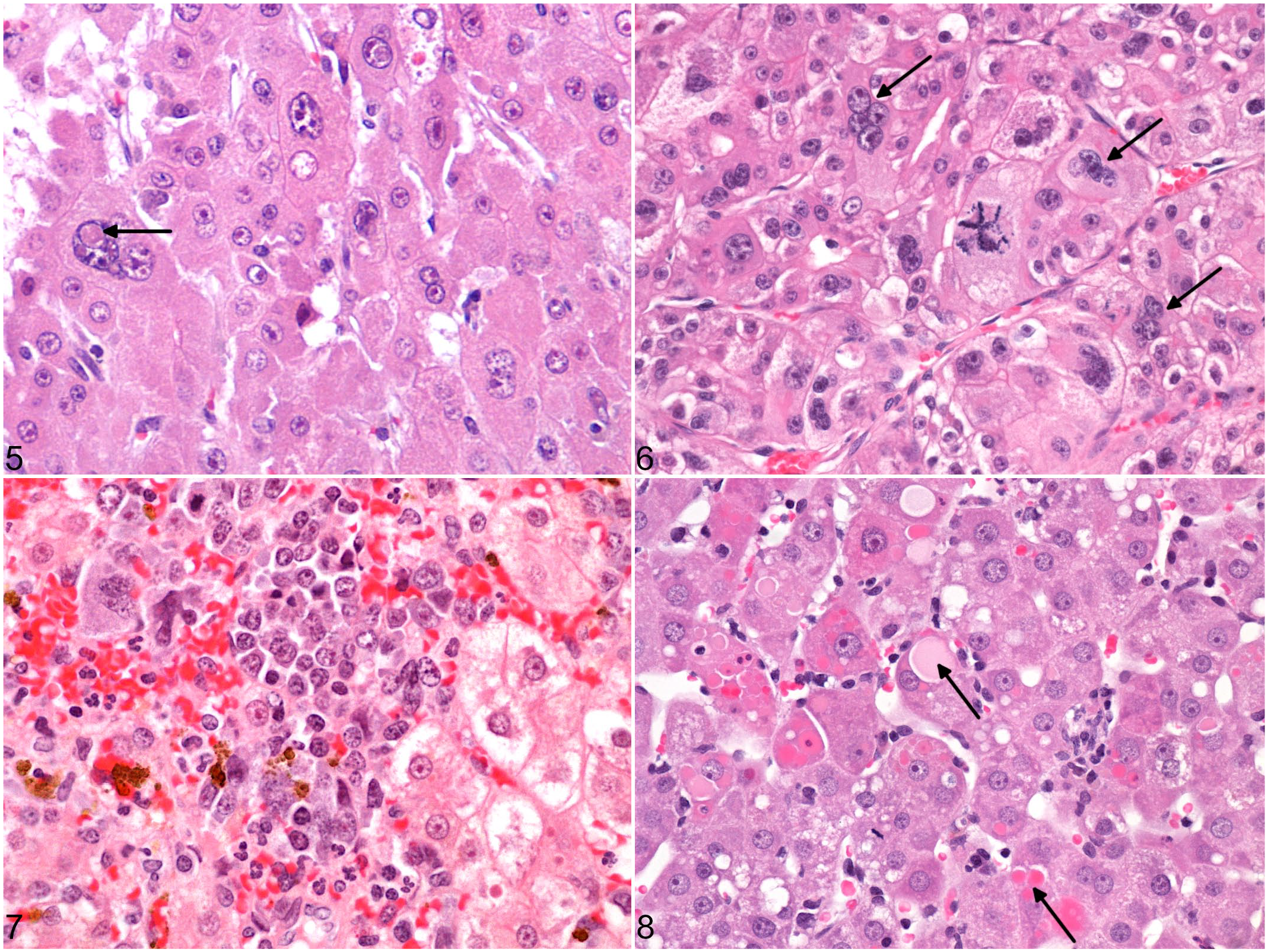
Histologic features of hepatocellular carcinoma in prosimians. HE.
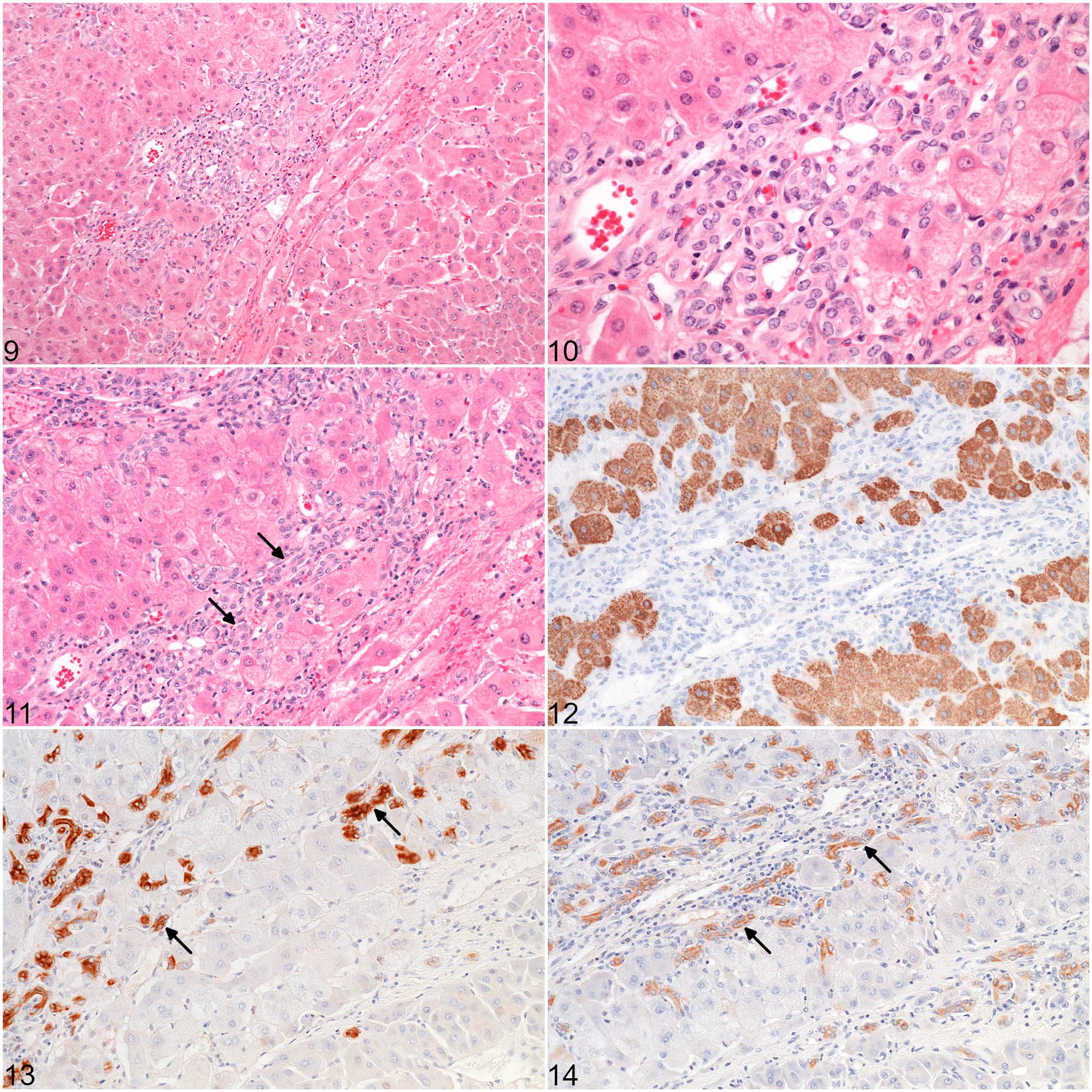
Peripheral ductular reaction in a hepatocellular carcinoma, case 1.
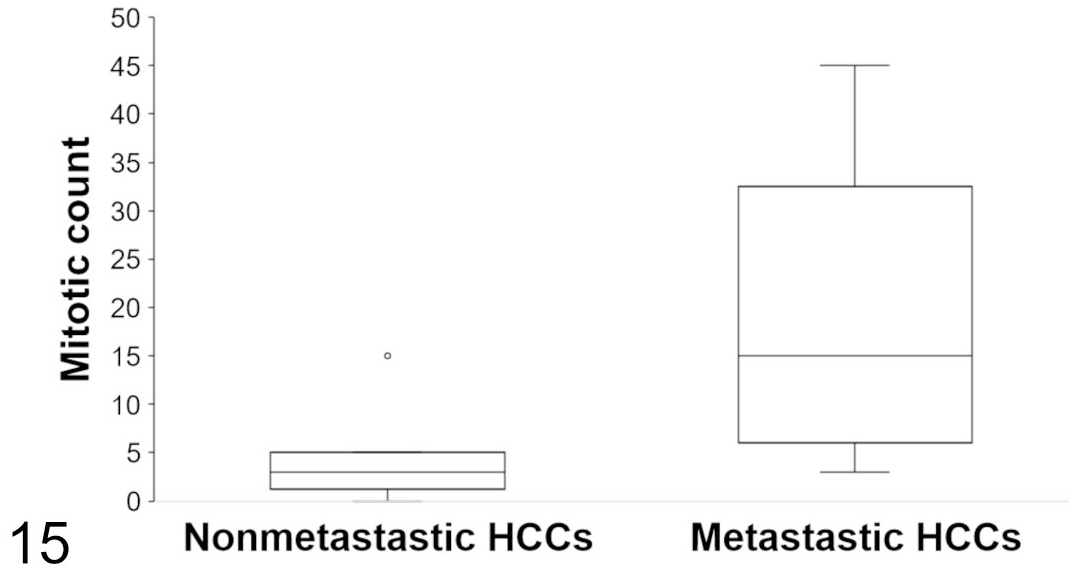
Mitotic count in primary hepatocellular carcinomas. Mitotic activity was evaluated in 10 high-power fields equivalent to 2.37 mm2. The average mitotic count is significantly higher in the metastatic HCCs (
The staining of normal hepatocytes with HepPar-1 was consistently cytoplasmic, granular, and strong. Differences in staining intensity among zones of the hepatic lobule were not apparent. Bile duct epithelium and connective tissue did not react with HepPar-1. HepPar-1 was detected in more than 80% of neoplastic cells in all HCCs, both primary and metastatic tumors, confirming the hepatocytic differentiation (Figs. 16–17). Neoplastic cells and normal hepatocytes had a similar staining with HepPar-1.
The labeling with K7 and K19 was cytoplasmic, diffuse, and with moderate to strong intensity in normal bile ducts of varying diameters. The ductular reaction surrounding HCCs had a similar staining pattern (Figs. 13–14). Normal hepatocytes and connective tissue did not react with K7 or K19. Positive immunohistochemical staining for K7 and K19 was evident in 12/14 HCCs (85.7%) and 1/14 HCCs (7.1%, case 8), respectively. Among all nonmetastatic HCCs except case 8, K7 staining was either negative (2/7; cases 3 and 4) or detected in 5% to 15% of neoplastic cells (5/7) (Fig. 18). For the case 8, 1 of the 2 samples revealed a pseudoglandular pattern with positive K7 labeling in more than 80% of tumor cells, and a positive HepPar-1 expression in 5% to 15% of cells. Staining for K19 was positive in 5% to 15% of tumor cells. Among the metastatic HCCs (cases 9-14), staining for K7 was detected in all primary tumors, and in all pulmonary metastases but one (the clear cell variant). More than 15% of neoplastic cells were positive in 5/6 primary tumors (Fig. 19) and 3/5 metastatic tumors. In both sites, the staining was cytoplasmic with increased perimembranous labeling, and was frequently patchy within the same tumor. The percentage of K7-positive neoplastic hepatocytes was significantly higher in metastatic HCCs compared to nonmetastatic HCCs (
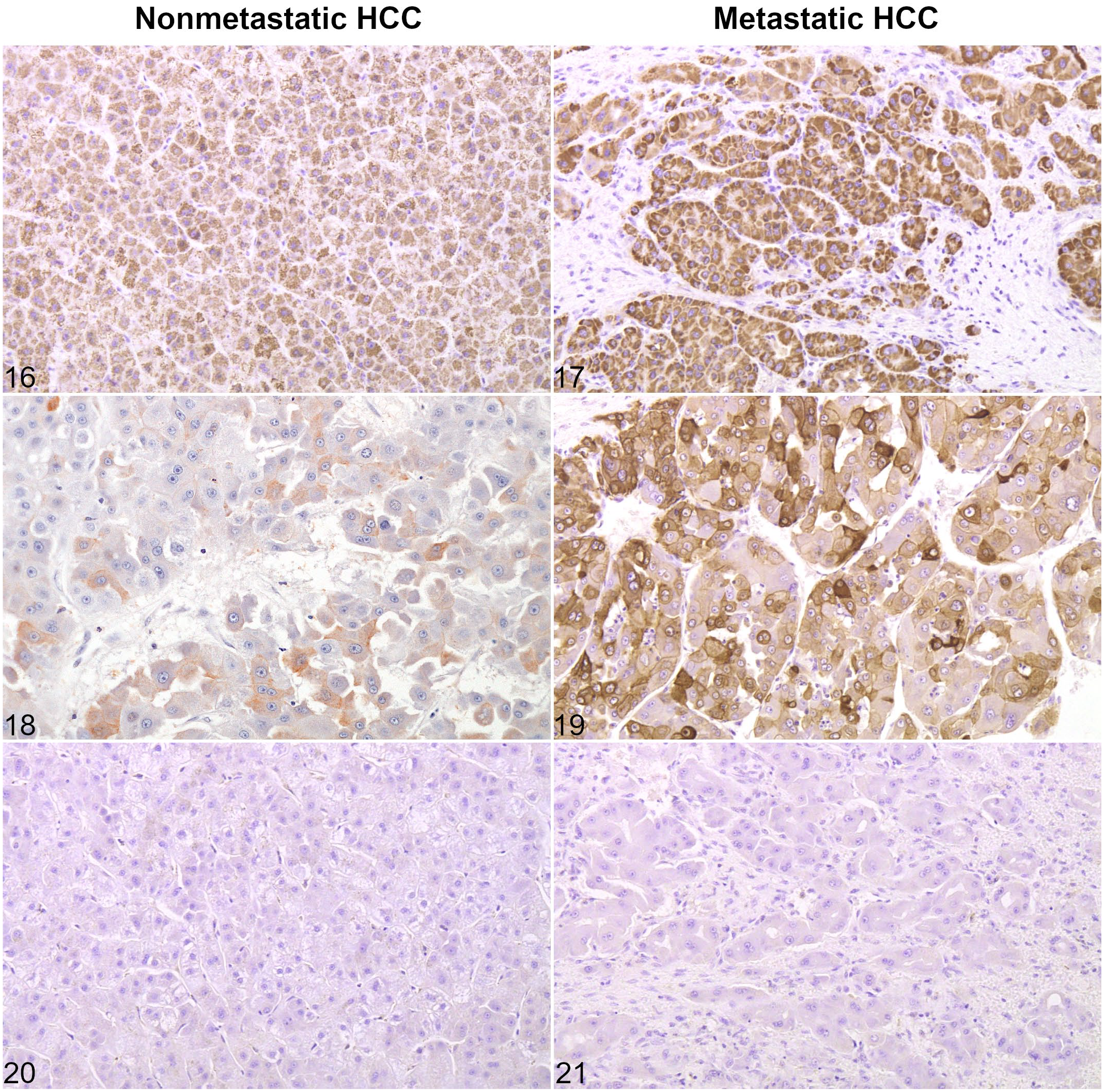
Expression of hepatocellular and cholangiocellular markers in hepatocellular carcinoma without and with metastases. Cases 6 (Figs. 16, 18, 20) and 10 (Figs. 17, 19, 21).
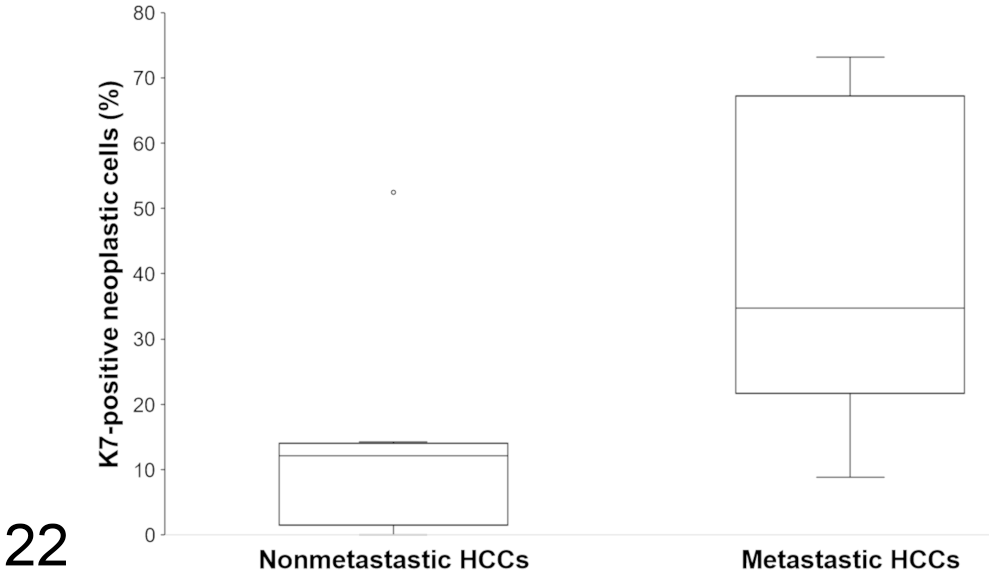
Percentage of K7-positive neoplastic cells in hepatocellular carcinomas with or without metastases. The difference was statistically different (
Discussion
To the best of our knowledge, this is the first study to characterize HCCs (both primary and metastatic tumors) in captive prosimians, histologically and immunohistochemically using HepPar-1, K7, and K19. Our initial search included all primates affected by HCC, but only lemurs and loris were recorded in the database at the North Carolina State University. These animals are endangered primates, so a better understanding of their most common fatal neoplasms is crucial. Hepatocellular neoplasia has been identified as the most common spontaneous neoplasm in prosimians. 39 In our study, all animals were older adults with HCCs occurring in the last one third of their life expectancy. Males were affected 3 times more than females. The same ratio is reported in humans, and the majority of patients are in their fifth to seventh decade. 4 Pulmonary metastases of HCC were found in 6/14 cases, showing a relatively aggressive behavior in prosimians. The lung is also the most common site of metastasis in man,6,25,56 captive prairie dogs,17,45 and woodchucks. 40 In dogs, metastases are less common and occur mostly in regional lymph nodes and lungs. 34
Microscopically, 4 architectural patterns (namely trabecular, pseudoglandular, pelioid, and scirrhous) of HCC were identified in lemurs and loris. No correlation was found between the pattern and the metastatic potential, as already described in humans. The trabecular pattern was present with the highest frequency; this has also been reported in dogs,
34
cats,
33
woodchuck,
40
captive prairie dogs,
17
Beechey’s and Richardson’s ground squirrels,29,44 and humans.
14
According to our study, the main histopathological features of HCCs in lemurs and loris were the presence of multinucleated cells and foci of extramedullary hematopoiesis. The latter finding has already been described in HCCs of several species, including woodchuck,
40
ground squirrel,
44
bovine,
52
and in porcine, and ovine hepatic tumors.5,8 The presence of intratumoral multinucleated cells has been identified more frequently in nonhuman primates compared to domestic animals,7,33,36,38 and has been a parameter of the grading system for human HCCs.
13
In our study, multinucleated cells were always visible, except in the 2 cases of the clear cell variant. Despite having mild atypia and low mitotic count, one clear cell HCC metastasized to the lungs. Interestingly, the 2 animals with clear cell HCC, that is, a mongoose lemur (
The etiologic factors responsible for the relatively high frequency of HCCs in prosimians remain uncertain. In humans, the development of the HCC is closely associated with chronic hepatitis or liver cirrhosis, caused by viral infection (e.g., hepatitis B virus or hepatitis C virus infections), alcohol consumption, aflatoxins in the diet, or hepatic metabolic disorders.9,16,54 Our study did not show that hemochromatosis, a well-known multifactorial disease in lemurs,
43
could be responsible for HCCs. Moreover, previous hepatic damage was absent in 10/14 animals. Mutations in
Immunohistochemistry is an important tool in the diagnosis of HCCs. HepPar-1 is an antibody to carbamoyl phosphate synthetase 1, a urea cycle enzyme in hepatocellular mitochondria. HepPar-1 was detected in all primary and metastatic HCCs, with a strong intensity of staining. Normal or hyperplastic bile ducts were not labeled. Consequently, HepPar-1 appears to be a highly sensitive marker for hepatocyte differentiation in lemurs and loris, as it has already been reported in canine, 37 feline, 49 and human studies.26,28,51,54
An aberrant cytokeratin pattern was observed in most cases (12/14). Expression of K7 and/or K19 has already been described in experimentally induced HCCs in rats and monkeys, 24 as well as spontaneous HCCs in 10% to 30% of humans.12,19,50 In our study, malignant hepatocytes expressed more frequently K7 than K19, as reported in humans. Interestingly, all nonmetastatic HCCs except 1 case revealed a positive K7 staining in 0% to 15% of neoplastic cells, whereas more than 15% of neoplastic cells were positive in 5/6 primary tumors for HCCs with metastases. Only one case (No. 8), a nonmetastatic HCC, expressed K19. In humans and dogs, positive K19 labeling is present in less than 15% of HCCs, and represents a prognostically relevant marker.12,18,27,48,53 The relevance of this marker might have been underestimated because of the small number of cases included in our study.
The histogenesis of HCC remains controversial. There are 2 possible explanations to account for the hepatocytic and biliary differentiation observed in HCCs. Either these HCCs may have arisen from progenitor cells with both features (maturation arrest theory), or alternatively neoplastic hepatocytes dedifferentiate and acquire progenitor cell characteristics (dedifferentiation theory). 41 For example, in this set of tumors, a combined hepatocholangiocarcinoma could not be ruled out in 1 case (No. 8), because 1 of the 2 samples revealed a pseudoglandular pattern with dual positivity for K7 and K19, and an HepPar-1 positivity only in 5% to 15% of cells. However, poorly differentiated HCC remained the principal diagnosis, considering the weak K19 immunoreactivity and the tendency of undifferentiated HCC to lose reactivity for HepPar-1 in humans.15,28
To conclude, 42.8% of lemurs had HCCs with pulmonary metastases, showing a relatively aggressive behavior. Metastatic HCCs had a significantly higher mitotic count, and K7 immunoreactivity with more frequency and in a significantly higher proportion of neoplastic hepatocytes compared to nonmetastatic HCCs. HepPar-1 appears to be a highly sensitive marker for hepatocyte differentiation in primary and metastatic tumors in captive prosimians.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221114471 – Supplemental material for Hepatocellular carcinomas in captive prosimians
Supplemental material, sj-pdf-1-vet-10.1177_03009858221114471 for Hepatocellular carcinomas in captive prosimians by Cynthia Robveille and John M. Cullen in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the histology laboratory staff, especially Ms Sandra Horton, at North Carolina State University College of Veterinary Medicine, for their technical assistance, and Drs. C. Williams and R. Schopler, at the Duke Primate Center, for providing the case material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
