Abstract
The cestode Hymenolepis nana is a common parasite of humans and mice. Fecal shedding in the absence of clinical disease has previously been reported in ring-tailed lemurs (Lemur catta). This report describes fatal, disseminated H. nana cestodiasis infection in an aged ring-tailed lemur in a zoological collection. The parasites were associated with severe multifocal to coalescing and regionally extensive pyogranulomatous hepatitis and moderate multifocal pneumonia. The morphology of the parasites was highly unusual. Profiles were variably sized, ellipsoid to irregularly serpiginous, lined by a thin tegument, and filled with lightly eosinophilic fibrillar stroma and numerous, round basophilic cells. Polymerase chain reaction targeting a portion of the 18S rRNA gene and DNA sequencing of the amplicon showed 100% homology with H. nana.
Prosimians are a group of small to medium-sized primates that includes strepsirhine lemurs, lorises, pottos and bushbabies, and haplorrhine tarsiers. Among them, a recent assessment identified lemurs as the most endangered primate group with 94% of lemur taxa listed as “critically endangered,” “endangered,” or “vulnerable” to extinction by the International Union for the Conservation of Nature (IUCN). 17 Main conservation threats include habitat destruction resulting from agriculture, logging, aquaculture, hunting, and climate-related change. 5 Ring-tailed lemurs (Lemur catta) are a common species exhibited in zoos around the world. They are currently listed as “endangered” by the IUCN. 5 In this report, we describe an unusual presentation of and death due to hymenolepiasis in an adult ring-tailed lemur.
A 13-year-old female spayed ring-tailed lemur that was housed at the Bronx Zoo in a multi-species, indoor exhibit with 13 other ring-tailed lemurs, collared lemurs (Eulemur collaris), multiple species of birds, and critically endangered Madagascar tortoises (Astrochelys yniphora) had a 1-day history of anorexia and self-separation from the troop. She was on intermittent cefpodoxime therapy for chronic urinary incontinence and lower urinary tract infections that developed secondary to pyometra and reconstructive genital surgery performed 3 years earlier. More recently, she was treated with diazepam to reduce anxiety related to new animal introductions, and for wounds from a conspecific that required surgical repair (4 and 2 months prior to presentation, respectively). Physical examination identified very thin body condition, severe tail base dermatitis, and a palpably firm liver. Pulmonary consolidation and pleural fluid were identified radiographically, and irregular echogenicity and possible hepatic cysts were identified ultrasonographically. Elevations in total bilirubin (104.3 μmol/L; reference interval [RI] = 3.4-17.1 μmol/L), γ-glutamyl transferase (175 U/L; RI = 6-57 U/L), and aspartate aminotransferase (217 U/L; RI = 8-81 U/L) were present. Alanine transaminase and alkaline phosphatase were 215 U/L (RI = 31-223 U/L) and 62 U/L (RI = 70-460 U/L), respectively. 18 Due to poor prognosis, euthanasia was elected. Clinical chemistry results at the time of wound repair 2 months earlier were largely unremarkable, with total bilirubin, alkaline phosphatase, and alanine transaminase values within reported reference intervals.
Grossly, the liver was diffusely dark brown, firm, and contained multifocal to coalescing, well-demarcated, unencapsulated, mottled dark red to tan-brown masses that ranged from pinpoint to 4 cm × 6 cm × 1 cm. When cut, many of the masses were mottled white, red and red-black, and had a large tan central area (Fig. 1). Numerous pinpoint to 0.1 cm diameter, well-demarcated, white nodules were present throughout the pulmonary parenchyma. Additionally, the lemur was in poor body condition with generalized skeletal muscle and adipose atrophy. Hepatic neoplasia with pulmonary metastasis was the primary differential diagnosis; inflammation or an infectious etiology were alternative differentials.
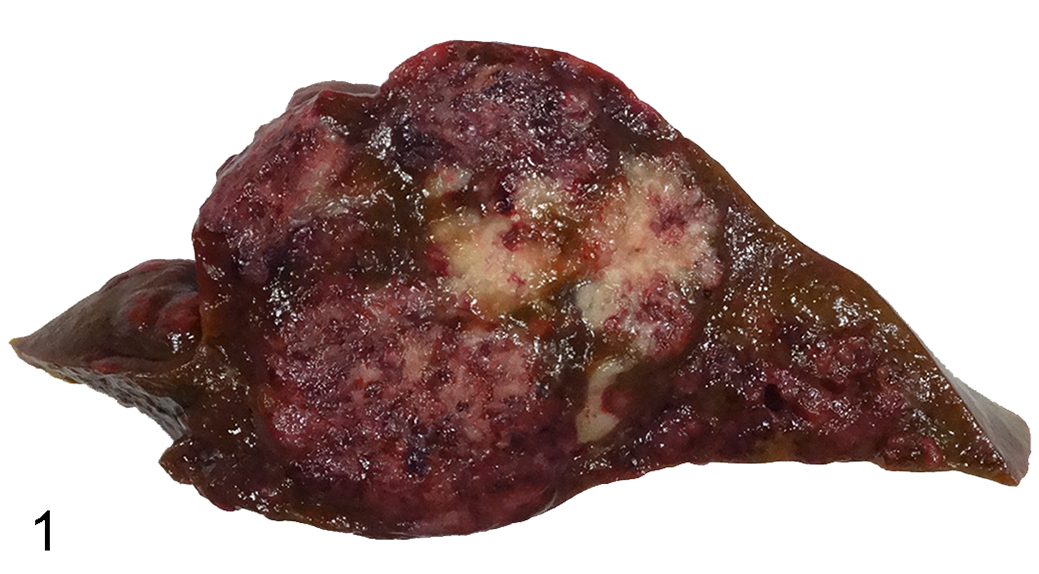
Cestodiasis, liver, ring-tailed lemur. The hepatic parenchyma is expanded by multifocal to coalescing, demarcated, unencapsulated, variably shaped and sized, mottled white, red, and red-black areas.
Histologically, approximately 75% of the hepatic parenchyma was effaced by variably sized, multifocal to coalescing and regionally extensive, roughly nodular areas of inflammation and necrosis that contained irregularly shaped, intralesional parasites (Fig. 2). Parasites were round to ellipsoid to irregularly serpiginous or folded, and up to 200 µm in width and 400 µm in length. They were lined by a thin, approximately 2-μm-thick, eosinophilic wall (tegument), and contained loose, lightly eosinophilic fibrillar stroma studded with numerous, round basophilic cells. Cells were generally 5 µm in diameter, had a high nuclear to cytoplasmic ratio, indistinct cell margins, and scant cytoplasm (Fig. 3), and were arranged individually, in dense rows lining the internal aspect of the tegument, in few to large aggregates (up to 30), or in sheets. Cell nuclei were generally round with coarse chromatin; many had a distinct central nucleolus. Some parasites had partial tegument loss that was associated with stromal vacuolation and degeneration. Intact and degenerate parasites were often embedded in loose collagenous matrix and admixed with variably degenerate neutrophils, fibrin, hemorrhage, congestion, parasite and host necrotic cellular and tissue debris, and occasional epithelioid macrophages and multinucleated giant cells. Moderate multifocal intracanalicular cholestasis, biliary hyperplasia, hemosiderin accumulation, and mild hepatocytic microvesicular lipid- and glycogen-type vacuolation were present throughout the remaining parenchyma. Similar parasites were also present multifocally in the lung and focally within the lumen of a mesenteric vein. Infection in these sites was mild and in the lung was associated with moderate, multifocal, pyogranulomatous pneumonia.
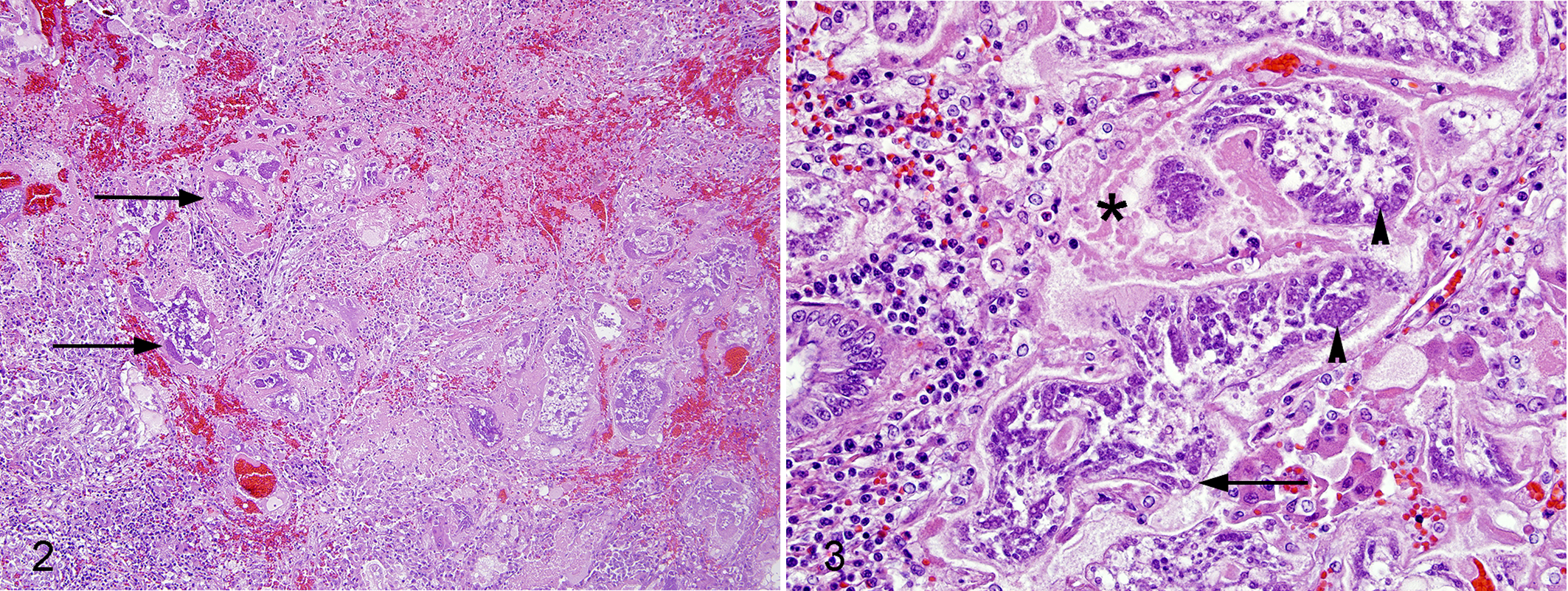
Cestodiasis, liver, ring-tailed lemur. Hematoxylin and eosin.
To further characterize and definitively identify the parasite, PCR (polymerase chain reaction) targeting a portion of the 18S rRNA gene was performed per previously published methods 19 (University of Florida, College of Veterinary Medicine, Zoological Medicine and Wildlife Disease Laboratory). DNA sequencing of the amplicon had 100% homology to sequences of the cestode Hymenolepis nana (the 516 nt product matched 100% with GenBank # MK874337).
Disseminated cestodiasis is generally uncommon, 1 and severe, hepatic cestodiasis with atypical parasite morphology was an unexpected finding in this lemur. The histologic appearance of the parasites was very different from intestinal adult cestodes and cysticercoids described in typical Hymenolepis infections of other species. Histologic identification of the parasites was challenging due to their unusual morphology. This was compounded by the absence of characteristic cestode morphologic features such as calcareous corpuscles, distinct proglottid segmentation, reproductive or digestive organs, or scolices. Also unusual in the current case was dissemination to the lungs and a mesenteric vein. 7
Of reported cestode-related diseases, Echinococcus granulosus occurs globally, is zoonotic, and causes cystic echinococcosis, also known as hydatid disease. Descriptions of abdominal, thoracic, and hepatic hydatid disease and subsequent death have been reported in a brown greater galago (Galago crassicaudatus), a ring-tailed lemur, and rhesus macaques (Macaca mulatta). Related complications have included ureteral compression and hydroureter, cardiac displacement, and hepatic necrosis with tissue replacement. 14 Abdominal and subcutaneous cysticercosis, due to either Taenia martis or Cysticercus longicollis (larval form of T. crassiceps), has been reported in a ring-tailed lemur, black lemur, and a red-ruffed lemur. Infections were associated with tissue swelling and/or replacement, organ displacement, abdominal distension, and one was associated with fibrinopurulent peritonitis. 3,8,9 Parasites in all of these cases had typical gross and/or histologic cestode features. A similar case to that of the current report is disseminated metacestode infection due to a novel species of Versteria in an orangutan (Pongo pygmaeus). 4 Histologically, cystic structures containing large numbers of round parasite cells were present in the liver and in the pulmonary and splenic vasculature. Death was attributed to acute respiratory distress secondary to disseminated infection. 4 Other reports of disseminated cestodiasis in the veterinary literature include disseminated Spirometra spp. infection in a cat and baboons. 2,12
The cestode Hymenolepis nana is a common parasite of humans and mice that has occasionally been described in nonhuman primates. 6,7,16 Infection in humans is typically intestinal and asymptomatic, although heavy infections can result in weakness, headaches, anorexia, abdominal pain, and diarrhea. 10 The parasite has either an indirect or a direct life cycle. 20 The former involves an arthropod intermediate host that ingests an embryonated egg from fecal material. Subsequent cysticercoid development occurs within the insect. The insect is then ingested by a definitive host and the cysticercoids develop into adult cestodes in the small intestine and produce eggs that pass into the feces. Alternatively, embryonated eggs may hatch in the human small intestine and release oncospheres that burrow into the mucosal villous lamina propria and develop into cysticercoids. This direct, auto-infective life cycle can potentiate superinfection. 7 Histologically, infection is typically characterized by intraluminal intestinal adults and intravillous lamina proprial cysticercoids with scolices. 7 Previous reports of H. nana infection in prosimians are few. Fecal shedding of H. nana in the absence of clinical signs has been reported in a zoological collection of ring-tailed lemurs. 6
Only a few cases of atypical cestode larval development with similar histologic features to the current case have been reported previously. 13,15 In humans, 2 similar cases, one in a patient with AIDS and another with Hodgkin’s lymphoma, have been described. Similar to the lemur, both human patients had disseminated infection and visceral masses composed of parasitic, cyst-like structures. Amid fibrosis, inflammation, and necrosis, the parasites were described as “numerous sacs containing peculiar small cells with prominent, large nucleoli, with average diameters of 85 µm and ∼100 μm.” 13,15 Cestodiasis was confirmed by PCR in both cases, and H. nana was identified in one. In naturally infected immunosuppressed athymic mice, infection was associated with near total effacement of the hepatic parenchyma by atypical cystic larvae (cysticercoids). 7 These were described as being variable in size and shape and up to 500 to 800 µm in length with empty centers or rare, eccentrically placed scolices. A tegument and a peripheral or central (in smaller cysticercoids) matrix of basophilic nuclei was also seen. Degeneration of the parasites was associated with parasite and host tissue necrosis and inflammation. Atypical cysticercoids were also seen in the ileum, mesenteric lymph nodes, pulmonary interstitium, and pulmonary vasculature. Morphologically, the parasites in the lemur were similar to those in both the immunosuppressed humans and mice.
The parasites in the lemur had morphologic features that were also somewhat similar to those in an immunosuppressed human with intestinal and extraintestinal H. nana infection in which the authors suggested the unusual parasite morphology was due to malignant transformation of the parasite. 11 H. nana was confirmed with cestode and hymenolepid-species-specific PCR and DNA sequencing. Comparative genome sequencing was performed and structural genomic variants compatible with oncogenic mutations were identified. 11 The authors suggested that parasite proliferation and subsequent somatic mutations in H. nana stem cells resulted in malignant transformation. 11 Whether a similar mechanism might be present in the current case was considered but not further pursued.
Multi-organ involvement in this case was consistent with aberrant larval migration (larval migrans). Possible routes for dissemination included parenchymatous migration in the liver and hematogenous spread based on pulmonary and intravascular parasites. Whether immunosuppression, which has been reported in human and mouse cases, was a predisposing factor was undetermined in the lemur. It was not thought to be present based on subjective assessment of lymphoid tissue histology. However, subclinical negative immunomodulation through a combination of chronic urogenital tract disease, social dynamics, conspecific trauma, and medications may have played a factor in susceptibility and/or parasite migration.
The duration and source of H. nana infection in this lemur was also undetermined. Elevated hepatic clinical chemistry results were present at the time of euthanasia but not in a clinical workup 2 months earlier. As to source, ingestion of contaminated food items or infected insects were considered possibilities. A retrospective review of our pathology database (1998–2021), which included necropsies of co-housed animals, did not identify cestodiasis or similar infections. However, as a result of this case, all co-housed lemurs were treated empirically with praziquantel (anthelmintic) subsequent to this animal’s diagnosis.
This report describes an unusual presentation of disseminated H. nana cestodiasis in a ring-tailed lemur. The most remarkable finding was the unusual morphology of the larval parasites and lack of characteristic features, which made identification of the parasites based on morphology alone a diagnostic challenge. Whether host immunosuppression might predispose to this form of abnormal parasite development is currently unknown.
Footnotes
Acknowledgements
The authors would like to extend their sincere gratitude to the Wildlife Conservation Society’s Zoological Health Program technicians, Elizabeth Connor, Alfred Ngbokoli, and Aleksandr Goldman, for their assistance with this case, and the University of Florida, especially Dr Robert Ossiboff, for performing parasite PCR. We would also like to thank the staff of the Zoological Health Program and Department of Mammalogy for the care of this animal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
