Abstract
Spontaneous hepatocellular carcinoma has been reported as a relatively common neoplasm in prosimians; however, the cause is unknown. To investigate possible pathogenic mechanisms, the authors performed a review of all adult animals from a captive prosimian population that had postmortem examinations over the past 10 years. They performed a detailed histologic evaluation of all suspected proliferative liver lesions and diagnosed hepatocellular carcinoma in 14 of 145 lemurs (9.7%). Affected animals ranged between the ages of 6 and 40 years old. The tumors had an unusually aggressive growth pattern for animal species; metastasis to the lungs or mediastinum was evident in 7 of 14 animals. Thirty-one animals—9 with hepatocellular carcinomas and 22 age-matched controls without hepatic neoplasia—were tested to evaluate the relationship between hepatic iron stores (as well as other trace metals) and the presence of hepatocellular carcinoma. There was no difference between the hepatic iron, copper, or molybdenum in lemurs with hepatocellular carcinoma and those without, suggesting that iron is not a key element in the pathogenesis of liver tumor formation. Analysis of 22 serum samples from animals with and without liver tumors indicated no evidence of active infection with a hepadnavirus, the virus family that includes hepatitis B virus. Hepatitis C virus and aflatoxin B1 were considered as potential causes and ruled out owing to lack of associated histopathologic lesions. In conclusion, hepatocellular neoplasia is relatively common in captive prosimians, although previously suspected etiologies seem unlikely.
Lemurs and lorises are members of the suborder Prosimii, the most primitive members of the Primate family. 15 Lemurs are native to the island of Madagascar, whereas lorises are found in southern Asia. 15 Most of these species are endangered in their natural habitat, and small breeding populations are maintained at various sites around the globe. Our understanding of the general background of neoplastic disease in prosimians is limited to a few publications. 17 In a recent publication, hepatocellular neoplasia was identified as the most common spontaneous neoplasm in prosimians. 17 Before this review article, spontaneous hepatic neoplasms were sporadically reported in the literature with only occasional mention of metastasis.
Liver tumors in domestic animals are uncommon, representing approximately 1% of overall tumor incidence. 5 Similarly, malignant hepatic neoplasia in anthropoid primates is uncommon. Review of the literature reveals descriptions of fewer than 16 hepatocellular carcinomas (HCCs) and 5 cholangiocarcinomas in nonhuman primates. 16 Only 5 chimpanzees with hepatocellular neoplasia have been reported. Three of the tumors were carcinomas: One occurred in a chimpanzee with chronic hepatitis B virus (HBV), a member of the hepadnaviral family, with concurrent hepatitis D virus, a satellite virus of HBV. 20 A second HCC developed in a chimpanzee infected with hepatitis B and C viruses, and a third animal had no evident predisposing conditions. 11,16 Known risk factors for HCC in humans include chronic viral infections with HBV or hepatitis C virus (HCV), 6,24 excessive hepatic iron levels, 1,2,12 excessive alcohol consumption, and aflatoxin B1 contamination of the diet. 24
The purpose of this article is to review a series of spontaneous proliferative hepatic lesions in prosimians from the Duke Lemur Center (Durham, NC) from the past 10 years to further characterize the lesions histologically. In addition, we evaluated select cases for two recognized risk factors for HCC in humans and great apes: chronic hepadnavirus infection and hepatic iron levels.
Materials and Methods
Case Material
We performed a retrospective review of pathology reports of all prosimians from the Duke Lemur Center between 1999 to 2009. Ages at death were broken down into 5-year increments ranging from 0 to 40 years old (184 total deaths). Given that the majority of prosimians aged < 6 years old died at less than 1 week of age and that the first reported hepatic neoplasm occurred in a 10-year-old loris, we calculated the incidence of hepatic neoplasms in prosimians ranging from 6 to 40 years old. All animals had been maintained in accordance with the existing standards of the Duke University Institutional Animal Care and Use Committee guidelines. We reviewed histopathology slides from necropsy and biopsy samples of all animals with reports of proliferative lesions in the liver. All samples had been fixed in 10% neutral buffered formalin and then routinely processed into paraffin, sectioned at 5-μm thickness, and stained with hematoxylin and eosin for routine histology. Liver tumors were classified using standards developed for canine and feline liver. 18 All available tissues were examined for evidence of metastasis. Stains for copper (rhodanine), iron (Perl’s iron stain), and lipofuscin (Schmorl’s stain) were used on selected liver samples.
Polymerase Assay
Serum was collected from 23 prosimian primates representing 8 species: white-fronted brown lemur (
Trace Metal Assay
Samples of frozen liver (1 to 2 g) were collected from 31 prosimians that died of HCC or nonrelated natural causes. Livers from 5 lemur and 1 loris species were represented: fat-tailed dwarf lemur (
Statistical Analysis
Statistical analysis was performed using JMP software (SAS Institute Inc, Cary, NC). The liver trace mineral concentrations were logarithmically distributed; therefore, the values of these measurements were natural log transformed before statistical evaluation. Comparison of means for liver trace mineral levels between animals with and without liver tumors was performed using the Student’s
Results
According to the 145 necropsy reports reviewed from the animals in this range, 14 lemurs (9.7%) had HCC (case Nos. 1–14), 6 had hepatocellular adenoma (case Nos. 15–20), and 1 had a preneoplastic hepatocellular lesion (case No. 21). Affected animals represented 14 different prosimian species (Table 1 ). The age of death of the animals with HCC ranged from 10 to 36 years, with an average of 22.3. The sex ratio of affected animals was 9 males (64.3%) to 5 females (35.7%).
Summary of Spontaneous Hepatic Neoplasms in Captive Prosimians a
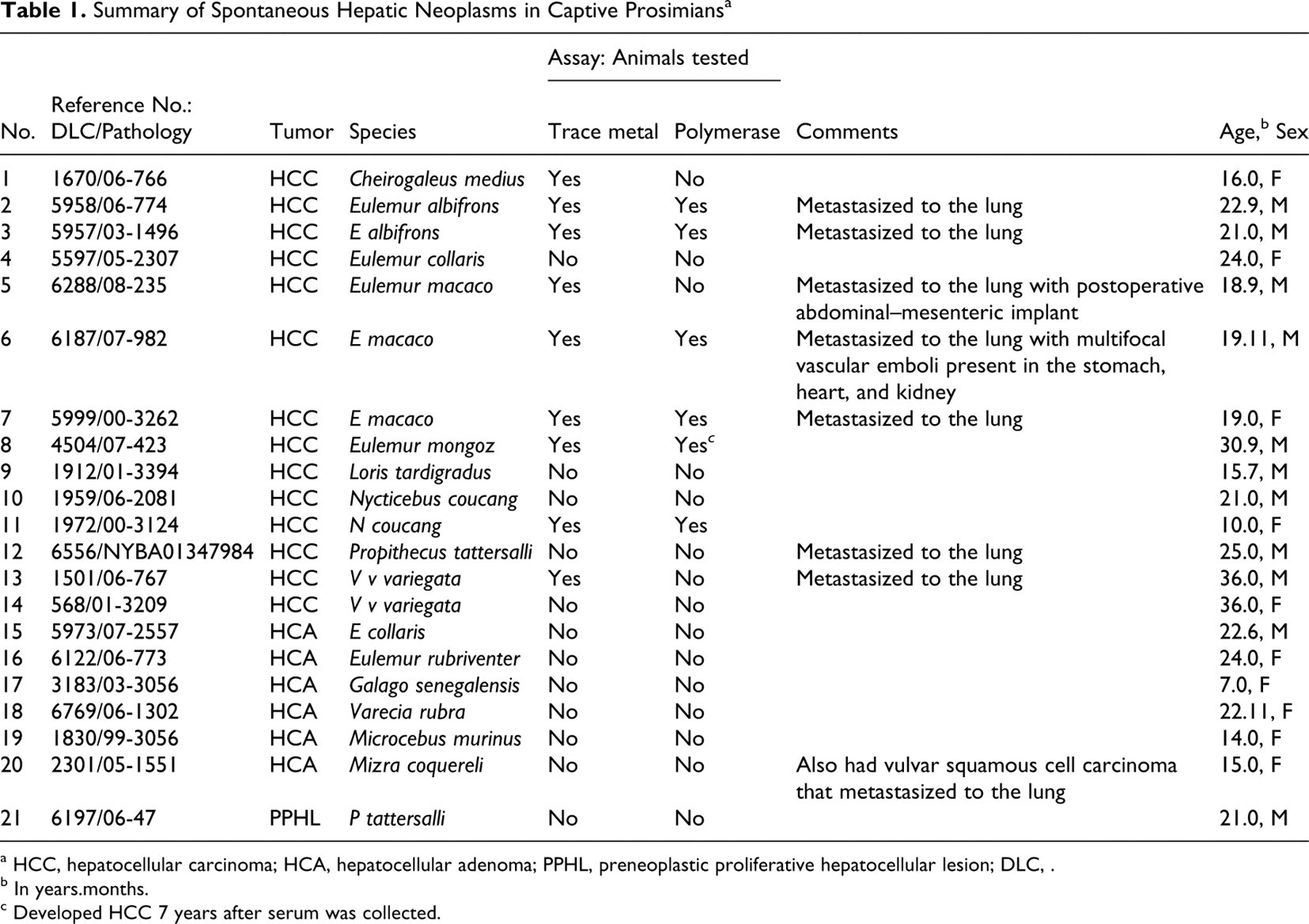
a HCC, hepatocellular carcinoma; HCA, hepatocellular adenoma; PPHL, preneoplastic proliferative hepatocellular lesion; DLC, .
b In years.months.
c Developed HCC 7 years after serum was collected.
Grossly, the hepatocellular neoplasms varied from relatively small, well-demarcated 1-cm masses involving a single lobe to expansive and invasive masses that essentially involved the entire liver (Fig. 1 ). The masses varied from pale brown to dark red brown. The cut surfaces often contained areas of necrosis and hemorrhage.
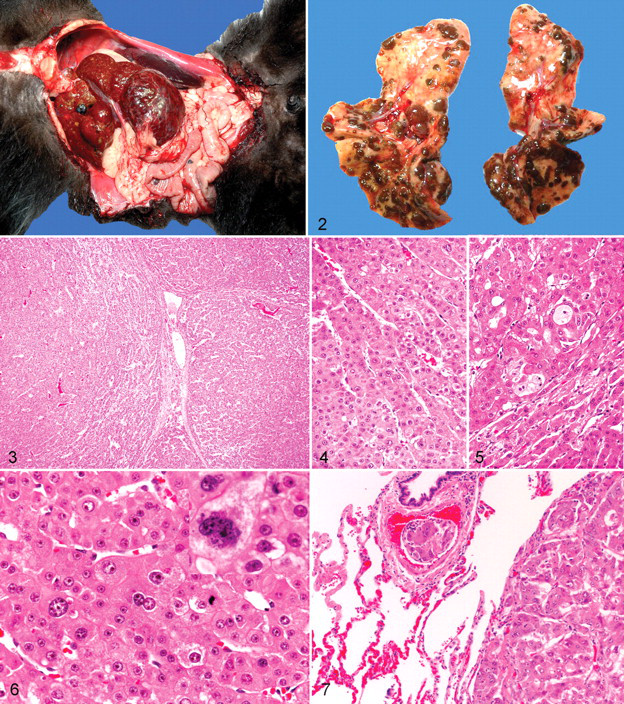
Histologically, hepatocellular adenomas were rounded expansive masses characterized by sharply demarcated borders with adjacent hepatic parenchyma. Within the adenomas, areas of cystic degeneration were filled with blood and, in some areas, proteinaceous fluid. The masses lacked normal portal tracts and instead contained individual veins and arteries. Hepatocytes in the adenomas formed regular trabeculae that were typically 2 to 3 cells thick, were well differentiated, had focal areas of finely vacuolated cytoplasm, and had round nuclei with single nucleoli.
More often, HCC had irregular borders and tended to invade adjacent hepatic parenchyma (Figs. 3, 5). Irregular, often extensive, areas of necrosis were present. Necrotic areas were often filled with blood or proteinaceous fluid. Hepatocytes were variably but often dramatically pleomorphic, with up to fourfold variation in size, including that of the nuclei (Fig. 6). Mitotic figures were generally uncommon but, when present, were frequently bizarre.
Metastasis was identified in 7 of 14 cases (case Nos. 2, 3, 5–7, 12, 13). Most metastatic lesions were found in the lungs, although 1 animal had multiple sites of metastasis (or intravascular spread that included the stomach, heart, and kidney) and 1 animal had metastasis to the mediastinum. Multiple masses of metastatic tumor were typically found in the lungs (Figs. 2, 7). The metastases often exceeded 1 cm in diameter, replacing up to 70% of the lung volume. The tumor cells were pleomorphic with a higher number of mitotic figures than that found in the primary neoplasm. Necrotic foci were less common in metastatic lesions than in the primary mass. One lemur had intra-abdominal metastasis, but the primary HCC had been biopsied with a needle biopsy tool, which may have resulted in seeding of the tumor.
Nonneoplastic sections of liver from prosimians with and without hepatocellular neoplasms typically lacked significant inflammation, fibrosis, and steatosis. Abundant hemosiderin was common within the cytoplasm of hepatocytes and Kupffer cells. Inflammation was not evident in any of the liver tumors, nor was there evidence of inflammation or fibrosis in animals with markedly increased amounts of stainable iron within their livers.
Polymerase Assay
There was no evidence of endogenous polymerase activity characteristic of hepadnaviruses in any of the 23 samples examined (Table 1). This finding suggests that there was no hepadnavirus viremia present in tissues at the time of testing, but it does not rule out the possibility of prior infection in some animals.
Trace Metal Assay
There was no correlation between liver iron concentrations and the presence or absence of hepatocellular tumors. Mean liver iron concentrations for animals without hepatic tumors were slightly lower than for those with hepatic tumors (3588 parts per million [ppm] versus 4138 ppm, respectively); however, the difference was not statistically significant (
Discussion
Given the small number of animals available for this study, we pooled several species of lemurs and included one member of a related species, the loris, a prosimian but not in the lemur family. Because the total number of animals in the colony, as well as the number of animals representing a specific species, fluctuated over the length of time covered by this study period, it was not possible to evaluate the incidence of hepatic neoplasia in either the colony as a whole or the different species. However, a general pattern can be discerned. In this population, the number of animals with HCC appears to be higher than that of many other mammalian species reported in the literature. In addition, the likelihood that HCCs will metastasize to the lungs in prosimian primates appears to be dramatically higher than that of spontaneously occurring HCC in other veterinary species. Phenotypically, malignant hepatocytes from HCC in lemurs have a greater tendency to be highly pleomorphic than do those from domestic species. 18
The natural life span of prosimians varies by species: thus, calculating a mean age of death for the 14 animals with HCC is somewhat misleading. However, with the exception of 1 animal, a slow loris (
The pathogenesis of HCC in this population of prosimians could not be determined. However, two known risk factors for HCC in humans were evaluated and eliminated with a reasonable degree of certainty. The lack of any evidence of hepadnavirus viremia in any of the serum samples evaluated minimizes the possibility of a HBV-related etiology for tumors in this population of animals, although it is possible that all animals had recovered from a previous infection and cleared all virus particles from their circulation. A HBV-like virus has been described in a black-and-white ruffed lemur (
Increased tissue iron concentrations have been shown to increase the risk of developing liver cancer in humans and rats. 1,7,12 Despite previous suggestions that lemurs with increased tissue iron deposition (hemosiderosis) may be at risk for developing liver cancer, 3,19 there was no correlation in this study between liver iron levels and the presence or absence of hepatic neoplasia. The fact that inflammation and fibrosis was not seen in animals with higher liver iron levels also suggests that iron is not a causative factor leading to tumor production in the animals in the population studied. These findings are consistent with those of Williams et al 22 in which typical cellular pathology associated with iron overload in the liver was not seen in lemurs that had hepatic iron concentrations up to 12,800 ppm dry weight (the lemurs were from the same colony studied in this work). In humans with genetic hemochromatosis (an inherited disorder of iron metabolism), the threshold level above which fibrosis occurs is approximately 22,000 ppm. 2,9 No animals in this study had levels above 22,000 ppm, although 1 animal, a ruffed lemur, had a value of 19,877 ppm. A trend for reduced cobalt levels in the animals with spontaneous HCC is a novel finding. This is in accord with a report in which diethylnitrosamine-treated mice were protected from liver tumor development by cobalt supplementation, suggesting that cobalt deficiency could increase liver tumor risk. 13 Another potential cause of hepatic neoplasia, aflatoxin B1, could not be assessed, because the diet was not analyzed for this mycotoxin. However, chronic aflatoxicosis typically produces biliary hyperplasia, fibrosis, and hepatocellular steatosis, lesions not seen in these animals. 14
In summary, the incidence of spontaneously occurring HCC in a group of captive prosimians has been described. Thorough histologic evaluation of the tissues supplied revealed a relatively high rate of metastasis in HCC-bearing prosimians, similar to the 40 to 60% reported for humans. 8,25 In addition, the lung was the most common site of hematogenous metastasis in both species.8 Lymphatic metastasis could not readily be assessed, because most cases did not include hepatic lymph nodes. Although the cause of HCC in prosimians remains unclear, we have eliminated two well-known risk factors reported in humans and great apes: hepadnavirus and excessive iron accumulation. Additional causative agents, aflatoxin B1 and chronic inflammation, are not likely associated with the development of HCC; that is, there were minimal to no supporting histologic changes. Given the endangered nature of these animals, further research may be warranted to determine the molecular pathogenesis of HCC in an effort to better preserve this primitive species.
Footnotes
Acknowledgements
We would like to thank the staff at the Duke Lemur Center, Durham, North Carolina, for their dedication to the conservation of prosimian species. We also express our gratitude to Dr William Mason, Fox Chase Cancer Center, Philadelphia, Pennsylvania, for polymerase assays; Carie Kimbrough, GlaxoSmithKline, Research Triangle Park, North Carolina, for review of statistical analysis; the histology laboratory staff at North Carolina State University College of Veterinary Medicine, Raleigh, North Carolina, for their technical assistance; and Dr John Barnes for his assistance with the images.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
