Abstract
Carcinoma in situ of the breast is a well-known entity in humans. In veterinary medicine, particularly in canine and feline mammary literature, there is no agreement whether the term in situ should be used to indicate a specific carcinoma histotype or the noninvasive status of a carcinoma of any histotype. Moreover, in the most recent histologic classification of mammary tumors published by the Davis-Thompson Foundation, it is suggested to abandon the term carcinoma in situ given the lack of standardized criteria defining this entity, replacing it with epitheliosis or ductal/lobular hyperplasia with severe atypia. This publication presents a critical review of the term in situ in human and veterinary medicine considering the evolution of the term over the years and its heterogeneous use by different authors, including variations in immunohistochemical markers for classification. This review aims to point out the lack of uniformity in the nomenclature and classification issues in veterinary medicine regarding the use of the term in situ, laying the ground for a process of standardization in future publications.
Keywords
In the intact adult female dog, spontaneous mammary tumors are the most common neoplasm, with malignant tumors accounting for up to 50% of cases. 10 Mammary tumors constitute approximately 17% of all feline neoplasms, with malignancies comprising up to 80% to 90%. 21 Thus, canine mammary tumors (CMTs) and feline mammary tumors (FMTs) have been the focus of intense research by veterinary oncologists and pathologists for decades. Both dogs and cats have been proposed as potential spontaneous animal models for human breast cancer (HBC) based on some clinical, pathological, and molecular similarities between CMTs, FMTs, and HBC. 2 Despite these similarities, there are some unique histologic features of canine and FMTs, and therefore, mammary tumors in these species are classified using species-specific histologic classification systems.
There is no agreement in the veterinary literature as to whether the histologic term in situ should be used. In the 1974 International Histological Classification of Tumours of Domestic Animals, 28 the authors have not “felt sure enough to make the diagnosis of mammary carcinoma in situ, in either its ductal or its ductulolobular forms, in bitches and cats” and did not include the histotype in the classification system. Moreover, in the 1999 36 and 2011 26 histologic classifications for mammary tumors, the term indicates a histopathological diagnosis of a specific entity (ie, an epithelial neoplasm—carcinoma in situ, CIS—with histological features of malignancy but without invasion of the basement membrane [BM] of preexisting structures), while some other authors used the term to indicate a type of growth of any carcinoma (see below).1,16,17,41 More recently, the authors of the histologic classification system published by the Davis-Thompson Foundation (2019) 72 include the term in situ, but suggest no longer using the histotype CIS given the lack of standardized histologic diagnostic criteria.
In addition, in any case, there is no standardization regarding how to assess the in situ status.
Historically, CIS, as a distinct classification entity, was described in the veterinary literature in the second edition of the World Health Organization (WHO) Histologic Classification for canine and FMTs, published in 1999 36 and replacing the first edition of 1974. 28 CIS was also maintained as a specific histotype in the 2011 26 proposal for the updated histologic classification of CMTs. In the 1999 36 and 2011 26 classifications, the CIS histotype (ie, noninfiltrating) was used to designate a neoplastic lesion that was often multicentric, arise in preexisting ducts or lobules, and had morphologic features of malignancy, such as loss of cell polarity and loss of normal architecture, but had not invaded the preexisting BM. The authors of the 1999 and 2011 classifications emphasized the difficulty in differentiating between CIS, lobular hyperplasia with atypia, and atypical epitheliosis. In the most recent updated histologic classification system (2019) 72 published by the Davis-Thompson Foundation, the authors suggested no longer using the histotype CIS given the lack of standardized histologic diagnostic criteria. The authors preferred terms such as epitheliosis or ductal/lobular hyperplasia with severe atypia to refer to those intraductal or intralobular lesions that do not form a nodule, do not exhibit sufficient malignant features to be classified as a malignancy, and have no apparent extension through the preexisting BM and no interruption of the peripheral layer of preexisting myoepithelial cells. 72 Furthermore, the authors mentioned that immunohistochemistry (IHC) applied to identify a continuous layer of myoepithelial cells in an in situ type of growth can be misleading in veterinary species, as these cells, especially in complex canine tumors, are not the original preexisting myoepithelial cells but a proliferation beneath the luminal epithelial cells, creating a new peripheral barrier. 72
More recently, the term in situ has been used to describe a type of growth of any mammary carcinoma in dogs and cats, regardless of the histotype, especially by some authors focusing on comparative aspects between mammary tumors of humans, cats, and dogs.1,16,17,41 In particular, a research group, attracted by the potential prognostic impact of recognizing lesions restricted to their preexisting mammary anatomic limits, has performed studies to investigate the usefulness of an in situ, noninvasive compared with an invasive status, applied to any histotype, with the former status based on a continuous immunohistochemical layer of myoepithelial cells.1,16,17,41
To lay the groundwork for reaching a standardization and a consensus in the future regarding the use of the term in situ, the present publication aimed to review the use of the term “in situ” when referring to CMTs and FMTs in the veterinary literature and compare this with the HBC classification, mainly referring to the latest WHO classification of breast tumors (herein WHO Breast 2019). 67
In Situ Human Breast Cancer
The term “in situ” is a Latin phrase that translates to “on-site” or “in its place.” In human breast pathology, this adjective, as described by Broders in 1932,
8
refers to a condition in which malignant epithelial cells and their progeny are found in or near positions occupied by their ancestors before the ancestors underwent malignant transformation. At least they have not migrated beyond the juncture of the epithelium and connective tissue or the so-called basement membrane. Such migration would be manifested by the cells entering the connective tissue.
In the modern era, and according to the WHO Breast 2019, this term is used as an antonym for an invasive epithelial neoplasm. 67 The qualifier in situ has been assigned to some specific noninvasive histotypes of HBC: lobular carcinoma in situ (LCIS), ductal carcinoma in situ (DCIS) including the papillary subtype, and solid papillary CIS (Table 1). 67
Histopathological characteristics of in situ human breast neoplasms. a
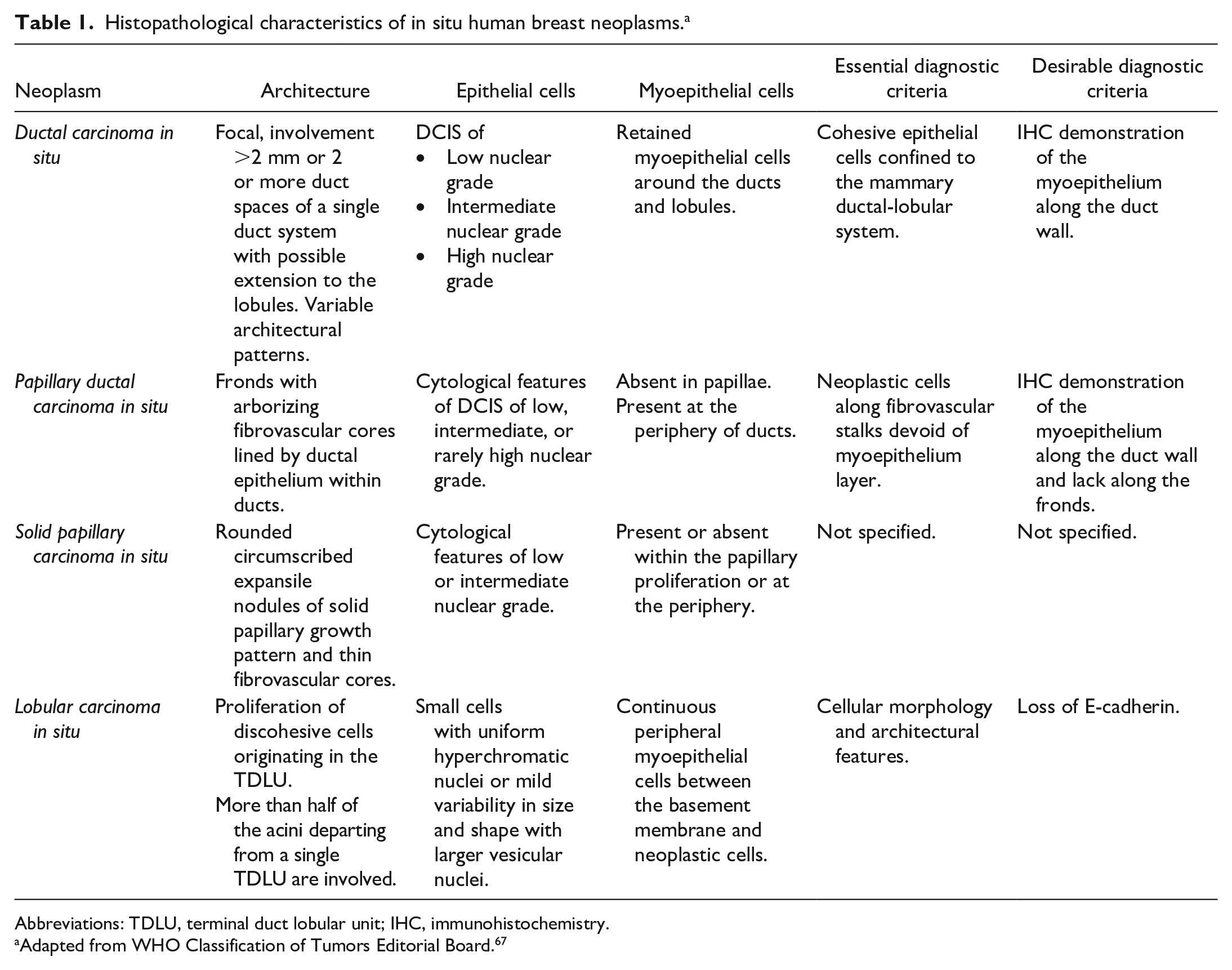
Abbreviations: TDLU, terminal duct lobular unit; IHC, immunohistochemistry.
Adapted from WHO Classification of Tumors Editorial Board. 67
Generally speaking, and as reported by the WHO Breast 2019, invasive carcinoma permeates the surrounding stroma and consequently disrupts the integrity of myoepithelial layers at their periphery; therefore, the term in situ should be accompanied by the lack of invasion of the surrounding stroma suggested by regular contours and by the evidence of a peripheral, intact myoepithelial layer. 67 However, the absence of a continuous peripheral myoepithelial layer does not unequivocally rule out an in situ carcinoma. Indeed, some forms of noninvasive lesions such as solid papillary carcinoma might not retain a myoepithelial lining.32,67 Solid papillary carcinoma is a biologically indolent tumor diagnosed when the nodules have rounded, well-circumscribed contours and a distribution pattern consistent with an in situ process. 32 In this specific entity, the absence of detectable myoepithelial cells at the tumor-stromal interface does not necessarily indicate stromal invasion, and therefore, investigation of myoepithelial cells is not considered mandatory.32,42,55
IHC for myoepithelial cell markers, such as calponin, smooth muscle actin (SMA), smooth muscle myosin heavy chain (SMMHC), p63, and basal cytokeratins (CK), can be helpful in demonstrating an intact myoepithelial layer around the neoplasm.70,71 Unfortunately, these markers have variable sensitivities and specificities, and the correct interpretation of the immunolabeling is not always clear cut, especially in differentiating stromal and vascular elements from myoepithelial cells.29,70 Cells that are most commonly misinterpreted as myoepithelial cells include reactive, calponin/SMA-positive myofibroblasts,28,69 calponin/actin/SMMHC-positive capillary pericytes, 60 and scattered, p63-positive neoplastic basal epithelial cells. 66 Therefore, a combined approach using multiple myoepithelial markers is suggested in the WHO Breast 2019 as the best option to increase diagnostic accuracy. 67
Lobular Carcinoma In Situ
Two papers published in 1941 suggested, for the first time, considering LCIS as a distinct morphologic entity.24,40 The overview of the earliest stages of mammary carcinoma described by Muir 40 characterized LCIS as a lesion that originates in “acini,” whereas Foote and Stewart 24 introduced the term LCIS to describe “a disease of small lobular ducts and lobules.” Chen et al 15 in the WHO Breast 2019 define LCIS as a noninvasive neoplastic proliferation of discohesive cells originating from the terminal duct lobular units (TDLUs), with or without pagetoid involvement of terminal ducts and filling more than 50% of the acini in each TDLU (Fig. 1). This last morphological criterion is also the basis of the distinction between classic LCIS and atypical lobular hyperplasia (ALH), the latter involving less than 50% of the acini per TDLU (Fig. 1). 15
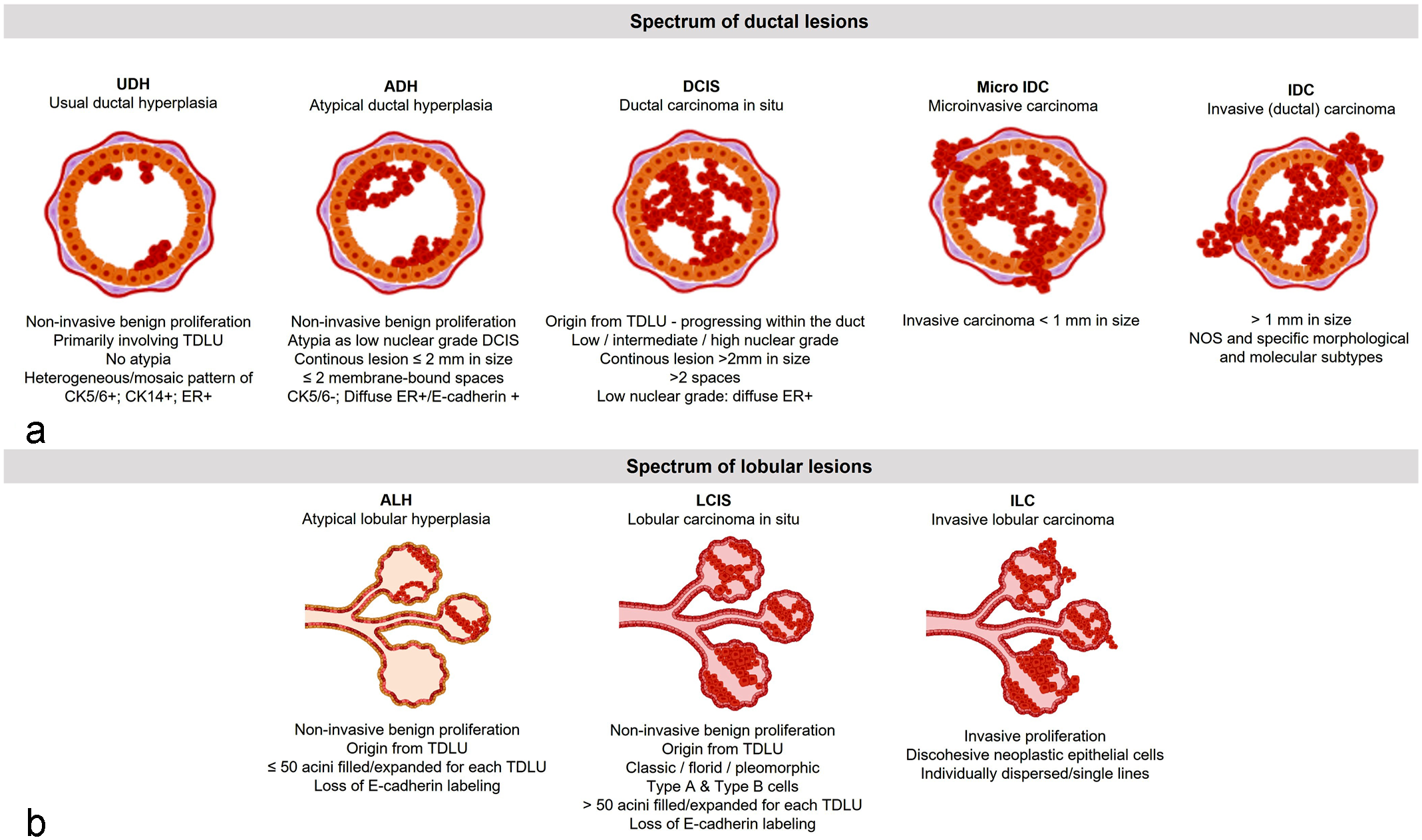
The main characteristics of (a) ductal and (b) lobular human breast lesions.
LCIS in women is a microscopic lesion that does not form a discrete palpable mass; consequently, the incidence of the disease is difficult to estimate.57,65 When it occurs alone in biopsied patients, LCIS constitutes 1% to 6% of mammary carcinomas and 30% to 50% of noninvasive carcinomas. 68
LCIS is considered a clonal neoplastic proliferation and a nonobligate precursor lesion associated with an increased risk of developing invasive breast cancer, approaching 1% to 2% per year, with a variable cumulative risk reported by different authors ranging from 14% at 10 years to 20% at 15 years.30,61,68 Breast cancer–specific mortality in patients with LCIS is considered low. 69 Mutation of the tumor suppressor gene CDH1, identified in 81% of LCIS cases, and its protein product (E-cadherin), which is involved in the maintenance of lobular architecture as well as in the β-catenin pathway, play an important role in the pathogenesis of both hereditary and spontaneous lobular lesions.18,62 Defective E-cadherin and the simultaneous downregulation of β-catenin result in loss of cell-cell adhesion, increased cell proliferation, and altered organization of the lobules, giving rise to the characteristic appearance of lobular lesions.18,62 Generally, LCIS can be histologically categorized as classical, pleomorphic, or florid. 15 According to the diagnostic criteria published by Chen et al 15 in the WHO Breast 2019, the essential parameters used to diagnose the different types of LCIS are both cellular morphology and architectural features, while the demonstration of loss of membranous E-cadherin expression, which is an immunohistochemical feature of all forms of LCIS, is considered only a desirable diagnostic criterion.
According to Wang et al, 63 myoepithelial cells generally maintain their continuous circumferential organization between the BM and LCIS; however, small gaps in myoepithelial continuity are observed when different antibodies are used for highlighting the myoepithelium. At present, histochemical/immunohistochemical staining for BM or myoepithelial cells is not considered mandatory for a diagnosis of LCIS. 15
Ductal Carcinoma In Situ
DCIS is a clinically, radiologically, and histologically heterogeneous, noninvasive and nonobligate precursor of invasive breast carcinoma, originating from the TDLU, and characterized by a proliferation of cohesive neoplastic epithelial cells confined to the mammary ductal-lobular system with possible extension to both the intralobular and extralobular ducts and with a range of architectural patterns, nuclear grades (see below), and distinct molecular subtypes. 46 Historically, DCIS presented as a palpable mass, but now, the vast majority are nonpalpable and detected by various radiologic techniques, mainly in geographical areas with population-based breast cancer–screening programs, where they comprise 15% to 30% of breast carcinomas. 46 The apparent incidence of DCIS has increased from 1.87 cases per 100,000 person-years in 1973–1975 to 32.5 cases per 100,000 person-years in 2005, with one case identified per 1000 screening mammograms.46,50 The breast cancer–specific survival of women with DCIS is extremely good. 23 Recent reports highlight that patients with DCIS aged >50 years have an equal risk of dying to that of the general female population. 23 Approximately 50% of patients with DCIS, treated with breast-conserving surgery, develop an invasive carcinoma. 51 Many factors, including younger age, large lesion size, higher grade disease, certain architectural patterns, comedonecrosis, and positive margins, have been variably associated with an increased risk of relapse. 51 Moreover, to date, no individual molecular prognostic marker has been demonstrated to be of clinical value in predicting the behavior of DCIS. 46
Several histologically nonuniform and nonharmonious classification systems for DCIS have been proposed over time. 5 DCIS was historically classified according to architectural pattern and categorized as solid, cribriform, micropapillary, or papillary with comedo and non-comedo types of necrosis. 46 The WHO Breast 2019 has recommended using the cytonuclear morphology instead of the architectural pattern. 46 As a consequence, DCIS is categorized as being of low, intermediate, or high nuclear grade. DCIS of low nuclear grade is composed of small, monomorphic cells, with nuclei 1.5 to 2 red blood cells (RBC) in size and uniform in shape, regular chromatin, inconspicuous nucleoli, rare mitotic figures, and inconspicuous areas of necrosis. DCIS of intermediate nuclear grade is composed of cells with moderate variability in size, shape, and polarization. The nuclei have variably coarse chromatin and sometimes have prominent nucleoli. In DCIS of intermediate nuclear grade, mitosis and necrosis may be present. DCIS of high nuclear grade is formed of large, atypical cells, most commonly with solid architecture. The nuclei are larger than 2.5 RBC and are typically pleomorphic, with irregular contours, coarse chromatin, and often prominent nucleoli. Mitoses are described as frequent, but no specific thresholds are proposed. 46 Interestingly, as pointed out by Badve and Gkömen-Polar in 2019, 5 the morphological changes in DCIS lie within the spectrum of usual ductal hyperplasia (UDH) and atypical ductal hyperplasia (ADH) at one end, and invasive and microinvasive carcinoma, the latter defined as a tumor with stromal invasion measuring less than 1 mm in diameter, 20 at the other end (Fig. 1). 67 The WHO Breast 2019 standardized the diagnostic criteria for several breast lesions including UDH, ADH, and DCIS; however, the distinction of low-grade DCIS from ADH still remains problematic, particularly in excisional biopsies (see below).46,67
IHC for high-molecular-weight keratins can assist the distinction of DCIS from UDH. It is reported that the loss of CK5/6 is a typical feature of DCIS and that UDH has a heterogeneous or mosaic pattern for high-molecular-weight CK, such as CK5/6, CK14, and 34βE12.46,67 Likewise, the strong and uniform expression of estrogen receptor (ER) can be used to support the diagnosis of DCIS because UDH typically displays patchy, weak-to-moderate labeling for ER.46,67 However, the distinction between ADH and DCIS is predominantly based on the combination of size/extent of the lesion, and architectural, cytological, and nuclear features.46,67 Page et al 43 proposed the involvement of 2 or more membrane-bound duct spaces as the cut-off point between ADH and DCIS for DCIS, whereas Tavassoli and Norris 59 and Allison et al 3 proposed a size of 2 and ≤2 mm, respectively, such that contiguous lesions less than or equal to 2 mm are classified as ADH. Based on these data, the WHO Breast 2019 reported that the size/extent criteria of >2 mm or more than 2 complete duct spaces are essential for distinguishing low-grade DCIS from ADH.46,67 When lesions of limited extent (ie, close to 2 mm) are identified or when the degree of involvement of ducts is borderline, particularly in core needle biopsies, a conservative approach is recommended in the WHO Breast 2019.3,46,67
IHC plays a limited role in the distinction of DCIS from ADH because both lesions lose the expression of CK and acquire that of ER in a similar manner.3,46,67
For distinguishing between DCIS and microinvasive/invasive ductal cancers (microIDC/IDC), IHC for myoepithelial markers can help but is not considered essential by the WHO Breast 2019. 46 The distinction is based on morphological evidence of a tumor less than 1 mm for microIDC or larger than 1 mm in greatest dimension for IDC, permeating the surrounding stroma, and disrupting the normal mammary units. 20
Canine and Feline In Situ Mammary Lesions
In Situ Canine Mammary Lesions
To the best of our knowledge, the first time that the term in situ was applied in dogs was in 1983 by Gilbertson et al. 25 They adapted the human classification devised by Black and Chabon 7 into the following 4 “grades”: (1) benign, (2) benign with precancerous changes, (3) in situ malignant tumors, and (4) invasive malignant tumors. Furthermore, Gilbertson et al proposed a grading system based on the degree of nuclear differentiation, degree of ductal epithelial proliferation, and lymphoid cellular reaction to evaluate the histology of the ductal epithelium (duct grade 1 = normotypic and normoplastic epithelium; duct grade 2 = normotypic and hyperplastic epithelium; duct grade 3 = moderately atypical epithelial proliferation; duct grade 4 = markedly atypical epithelial proliferation; and duct grade 5 = severely atypical epithelial proliferation, limited to the structural boundaries of the ductal system-in situ carcinoma). In particular, the in situ carcinoma was considered a severely atypical epithelial proliferation within the structural boundaries of the duct system and classified by the authors as histological stage 0. 25
The second edition of the WHO classification of mammary tumors of the dog and cat (1999) by Misdorp et al 36 officially introduced, for the first time, the histotype “noninfiltrating” (in situ) carcinoma as a proliferation of malignant cells within ducts/lobules with loss of architecture/cell polarity and lack of invasion of the BM, but provided no indication of how to evaluate invasion of the BM or how to distinguish ductal versus lobular CIS. Since then, most veterinary pathologists have used this definition. 36
In 2007, Antuofermo et al 4 defined canine DCIS as a precursor lesion of malignant transformation in the canine mammary gland, which was later confirmed by Mouser et al 39 and is similar to what is documented in HBC. In particular, Antuofermo et al 4 and Mouser et al 39 retrospectively examined canine mammary pre-neoplastic and pre-invasive lesions and described DCIS by adopting the morphological criteria used in human medicine (ie, a ductal lesion in which the epithelial proliferation fully involves at least 2 ductal cross sections showing both cellular and architectural atypia, such as the formation of discrete cribriform spaces lined by polarized epithelial cells). Antuofermo et al classified DCIS as low grade (monomorphic cells, no increase in nuclear size, diffuse, fine chromatin, and few prominent nucleoli or mitotic figures), intermediate grade (features intermediate between low and high grade), and high grade (pleomorphic cells, nuclear diameter >2.5 times that of normal ductal epithelial cells, vesicular chromatin, prominent or multiple nucleoli, and mitotic figures).4,39
A new standpoint was introduced by Soremno et al 58 in 2009, which included canine DCIS in the benign/dysplastic pre-malignant category, after providing clinical and histopathological evidence that spontaneous CMTs progress from benign to malignant.
Later in 2011 and 2017, Goldschmidt et al26,27 defined CIS, with no further specification of ductal versus lobular CIS, as a malignant, well-demarcated epithelial neoplasm that has not extended through the BM into the surrounding stromal tissue, and that is densely cellular, consisting of closely packed cells arranged in irregular tubules.
They emphasized the difficulty in differentiating CIS from lobular hyperplasia with atypia and from atypical epitheliosis.26,27 Besides morphology, no additional recommendations were proposed to precisely evaluate the extension of tumoral cells beyond the BM.26,27
Sanchez-Cespedes et al 53 evaluated the expression of calponin in 74 mammary gland lesions, classified according to the second edition of the WHO classification of mammary tumors of the dog and cat (1999), 36 demonstrating that the study of the myoepithelial cell layer integrity is not definitive for the diagnosis of malignancy and invasion in CMTs. 53
Further complicating the picture, some guidelines for the diagnosis of CMTs were established during 3 Brazilian mammary pathology meetings (2011, 2013, and 2017) and subsequently published introducing, for the first time in the veterinary literature, the term in situ as a type of tumor growth rather than a specific histotype.11–13 These guidelines also suggested criteria to distinguish between ductal and lobular in situ growths, using an approach similar to that applied in human pathology. The authors suggested that only CIS, and not hyperplastic and dysplastic conditions, could be considered as a precursor lesion of malignant transformation.12,13 These authors defined CIS as a “malignant epithelial proliferation in the ductal or lobular units of the mammary gland, occupying the entire lumen without discontinuity or absence of the basement membrane.” 11 They also introduced the term “in situ carcinoma areas with microinvasion areas” characterized by a small group of neoplastic cells (<1 mm) that invade the stroma beyond the BM. 11 As a conclusion, the authors postulated that periodic acid Schiff (PAS) reaction was helpful in evaluating the integrity of the BM, 11 but to the best of our knowledge, this approach is scarcely validated in veterinary medicine, 22 and it is not routinely performed in human pathology.
In a consensus review on the use of immunohistochemical markers on CMTs, Peña et al 45 pointed out that myoepithelial markers may not be able to distinguish in situ as a type of growth from invasive carcinomas, especially in dogs with complex tumors with proliferation of both interstitial and peripheral myoepithelial cells.
Studies investigating the performance of myoepithelial markers (p63, calponin, CD10, CK5, or alpha-SMA) in identifying a continuous peripheral layer of cells as a hallmark of canine CIS have provided some contradictory results.52–54
Recently, Chocteau et al 16 focused their research on 433 mammary carcinomas, histologically classified according to Goldschmidt (2011–2017),26,27 and histologically graded according to a modified Elston and Ellis system. 44 The in situ status, referring to a type of growth of any carcinoma, of 89 out 433 tumors was identified by the presence of a continuous layer of p63+ myoepithelial cells and assigned to histological stage 0 (tumor in situ, N0, M0). 16 Stage 0 tumors compared with non-in situ neoplasms (stage I–III) were associated with younger age of affected dogs (median 10.2 years), smaller tumor size (median 12.0 mm), very low probability of cancer progression within 1 year postdiagnosis, and a median overall survival time of about 3 years. 16
Therefore, based on the above-described literature and considering the most recent updated histologic classification system (2019) published by the Davis-Thompson Foundation, 72 there is a heterogeneous application of the term in situ in CMTs. Generally, there is an agreement on applying this terminology to a lesion that has not disrupted the preexisting anatomical borders; however, there are no standardized diagnostic criteria and no agreement as to whether this term should be used as a distinct histotype or as a qualifier of tumor growth to be applied to any histotype of CMTs. In addition, no detailed guidelines were provided on how to distinguish ductal versus lobular CIS, as routinely performed in women in situ breast neoplasm.15,46
Considering the above-described facts and controversies, we suggest that in future publications dealing with CIS, the authors precisely declare the histological classification system used specifying the use of the term in situ as a distinct histotype or as a qualifier of tumor growth of any histotype, as well as if and how the integrity of the peripheral myoepithelial layer is assessed.
In Situ Feline Mammary Tumors
The first time the term in situ was used to define a biological mammary entity in cats was in 1980 by Weijer to indicate “a sign at risk,” namely a lesion associated with the subsequent development of carcinoma in the same or other mammary glands of the same patient. 64 The term was later redefined by Castagnaro et al 14 in 1998 to characterize CIS and distinguish it from an infiltrative tumor employing an anti-actin antibody to demonstrate the myoepithelial layer.
In 1999, the 2nd edition of WHO classification of mammary tumors of the dog and cat reinforced the use of the term noninfiltrating CIS as a specific tumor histotype also in the cat. 36 Several studies published in recent years estimated the relative frequency of feline mammary CIS to be between 1.6% and 15.8% of mammary carcinomas, based on morphology alone.9,33,47,48,56
In 2010, Burrai et al 9 investigated the prevalence of UDH, ADH, and DCIS in feline mastectomy specimens. Based on morphology alone and human criteria, 9 the authors described the spontaneous development of these lesions in 28% of mastectomy specimens from female cats with clinical mammary disease. Similar to that reported by Antuofermo in dogs in 2007, 4 about half of feline UDH were associated with benign disease, whereas ADH and DCIS were generally associated with malignant mammary tumors.
In 2019, Chocteau et al, 17 as previously done in dogs, 16 used p63 immunolabelling to differentiate noninvasive FMTs (surrounded by a continuous layer of p63+ myoepithelial cells—stage 0) from invasive FMTs (lacking a continuous layer of p63+ myoepithelial cells) using the in situ terminology as a type of growth for any tumor histotype. They also proposed a histological staging system for FMTs and reported that at initial presentation, noninvasive FMTs (stage 0) significantly differed from invasive (stage I–III) tumors. Characteristically, compared with invasive FMTs, stage 0 FMTs were diagnosed at a younger age, were smaller and of lower histological grade, and had less frequent tumor-associated inflammation and central necrosis. 17
Although there are fewer studies, the application of the term in situ in FMTs is also not homogeneous and standardized. Again, in our opinion, applied criteria should be precisely declared within each study, possibly referring to veterinary classifications. As with CMTs, but with less complication due to the lack of complex/mixed FMTs, more than one marker could be used to detect a peripheral myoepithelial layer.
Canine and Feline CIS and IHC
Besides the studies investigating the myoepithelium, which have been discussed above, very few papers have explored the immunoreactivity of canine and feline hyperplastic lesions and CIS for estrogen receptor (ER), progesterone receptor (PR), HER2, and Ki-67, as utilized in HBC to predict behavior and prognosis.4,9,16,17,33–35,39,48,49 The available data in dogs and cats are scarce, somewhat conflicting, and not useful for the identification of CIS.
In 2002, Millanta et al 35 evaluated the expression of Ki-67 in feline mammary carcinomas, demonstrating that the 9 cases of CIS, classified according to Hampe and Misdorp, 28 had low proliferative rates, which were comparable to those registered in normal mammary tissue and adenosis.
The same authors in 2005 33 investigated the expression of ER and PR in 6 feline and 4 canine CIS classified according to Misdorp et al. 36 Canine CIS had a higher expression of ER and a lower expression of PR compared with normal mammary tissue, hyperplastic/dysplastic lesions, and benign tumors. 33 On the contrary, in cats CIS had a greater expression of PR compared with normal mammary tissue, hyperplastic/dysplastic lesions, and benign tumors, while ER expression was rarely observed in feline CIS. 33
In 2007, Antuofermo et al 4 investigated the expression of ER, PR, Ki-67, and HER2 in canine DCIS diagnosed according to morphologic human criteria. The authors demonstrated associations between the expression of some of these markers and DCIS grade, with ER found mainly expressed in intermediate-grade forms and Ki-67 expressed with highest percentages in high-grade forms. 4
In 2010, Mouser et al 39 added some information demonstrating that canine low-grade DCIS had a lower expression of ER, PR, and HER2 compared with the adjacent normal mammary gland and also a lower expression of PR compared with some hyperplastic mammary lesions.
In 2012, Ribeiro et al 49 classified 29 canine carcinomas arising in benign mixed tumors, diagnosed according to Misdorp et al, 36 into 4 subtypes based on their immunophenotypical profiles. The authors divided their 29 samples into those with an invasive malignant epithelial component and those with an in situ (noninvasive) epithelial component, the latter defined according to not well-specified parameters based on 2 texts from the human literature.19,31 The immunohistochemical panel of ER, PR, HER2, CK5, and human epidermal growth factor receptor 1 (EGFR) applied to these tumors showed that the majority had an in situ malignant component with an immunohistochemical profile compatible with a luminal A human subtype. 49
Reviewing CMTs and FMTs as a model for HBC, Abdelmegeed and Mohammed 2 in 2018 suggested that DCIS was the most similar category in dogs and humans in terms of morphological, pathological, and immunohistochemical characteristics.
More recently, Chocteau et al 17 considered the in situ noninvasive growth as stage 0 CMTs that showed higher ER and PR expression and lower Ki-67 proliferation index when compared with invasive CMTs.
In Situ Carcinoma and Molecular Investigations
Beetch et al in 2020, following their former works on DCIS histopathology, clinical outcome, and imaging,4,37,39 assessed DNA methylation alterations through the progression of canine triple-negative DCIS to triple-negative invasive mammary carcinoma. 6 Functional analysis of differentially methylated genes in DCIS and invasive tumors showed alteration in various processes commonly dysregulated in cancer, such as cell cycle progression, transcriptional regulation, apoptosis, and cellular signaling. 7
Mohammed et al 38 further demonstrated that ADH, DCIS, and invasive carcinomas represented a linear cancer progression in dogs, supporting the idea that molecular techniques could be helpful to stratify DCIS and to identify targets for prevention or therapy. Adding to this, canine DCIS showed overexpression of myoepithelial cell markers and core genes of the Wnt non-canonical pathway, which is known to predict metastasis and poor prognosis in human patients. 38
Conclusion
The different meanings given to the term carcinoma in situ during the last 30 years, mostly regarding its place and role in the complex mechanism of cancer progression, make this concept still problematic. This is more evident in CMTs and FMTs than in HBC for several reasons. In dogs and cats, the term in situ has been used in some studies to indicate a specific histotype—as for HBC—and in others to refer to a growth pattern of any tumor histotype. This review pointed out this lack of uniformity, which makes it difficult to compare results between studies and interferes with broad use of the term in diagnostic and clinical settings. In addition, even among those authors who share the same approach and use CIS as a separate tumor entity (ie, histotype), there is no agreement as to whether the human distinction of DCIS versus LCIS should be applied to dogs and cats and what the prognostic/clinical implications of such a distinction are.
Moreover, regardless of the way the term is applied (in situ as distinct histotype versus in situ as a qualifier of tumor growth for any histotype), there are no clearly stated and uniformly agreed-on diagnostic criteria (eg, morphology alone or analysis of the integrity of either the BM or the myoepithelial peripheral layer). This lack of standardization may be in part due to some historical inconsistency in the human pathology community when referring to CIS, which has been addressed in the most recent WHO classification of HBC (2019). 67
In conclusion, this review points out the facts and controversies regarding the nomenclature and classification issues of the term in situ in canine and feline mammary carcinoma and suggests a process of standardization in future publications. This endpoint would minimize subjective morphologic biases in the diagnosis of in situ lesions, which are strongly affected by the experience of pathologists, and lead to inconsistencies when classifying these intriguing, non-clear-cut canine and feline mammary lesions.
Supplemental Material
sj-docx-1-vet-10.1177_03009858221105060 – Supplemental material for Canine and feline in situ mammary carcinoma: A comparative review
Supplemental material, sj-docx-1-vet-10.1177_03009858221105060 for Canine and feline in situ mammary carcinoma: A comparative review by Giovanni P. Burrai, Valeria Baldassarre, Barbara Brunetti, Selina Iussich, Lorella Maniscalco, Francesca Mariotti, Alessandra Sfacteria, Cristiano Cocumelli, Valeria Grieco, Francesca Millanta, Orlando Paciello, Serenella Papparella, Roberta Rasotto, Mariarita Romanucci and Valentina Zappulli in Veterinary Pathology
Footnotes
Acknowledgements
Authors would like to thanks the Italian Association of Veterinary Pathology (AIPvet) that supported the constitution of a group of discussions on canine and feline mammary tumors composed by the authors of this review. The authors gratefully acknowledge Rachel Pittaway for critical reading the manuscript and for English language editing.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
